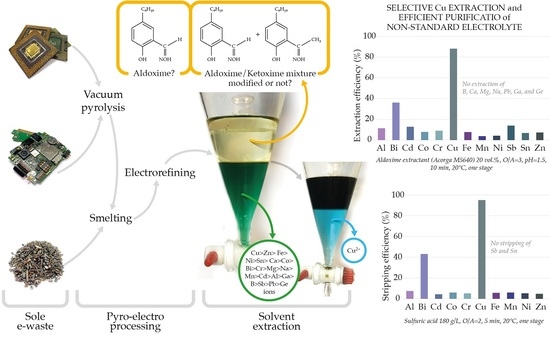
Influence of Electrolyte Impurities from E-Waste Electrorefining on Copper Extraction Recovery
Abstract
:Highlights:
- Electrolyte of highly complex chemical composition resulting from specially designed pyro-electrometallurgical e-waste process, aiming to reflect future obstacles considering sole e-waste recycling
- A one-factor solvent extraction methodology for the comparison of three commercial extractants in terms of efficiency, distribution coefficients, selectivity, and influence of impurities on Cu extraction from experimentally obtained electrolyte
- Feed pH, extractant, and stripping agent concentration affect solvent extraction most significantly through reaction equilibrium shifts and active centers availability
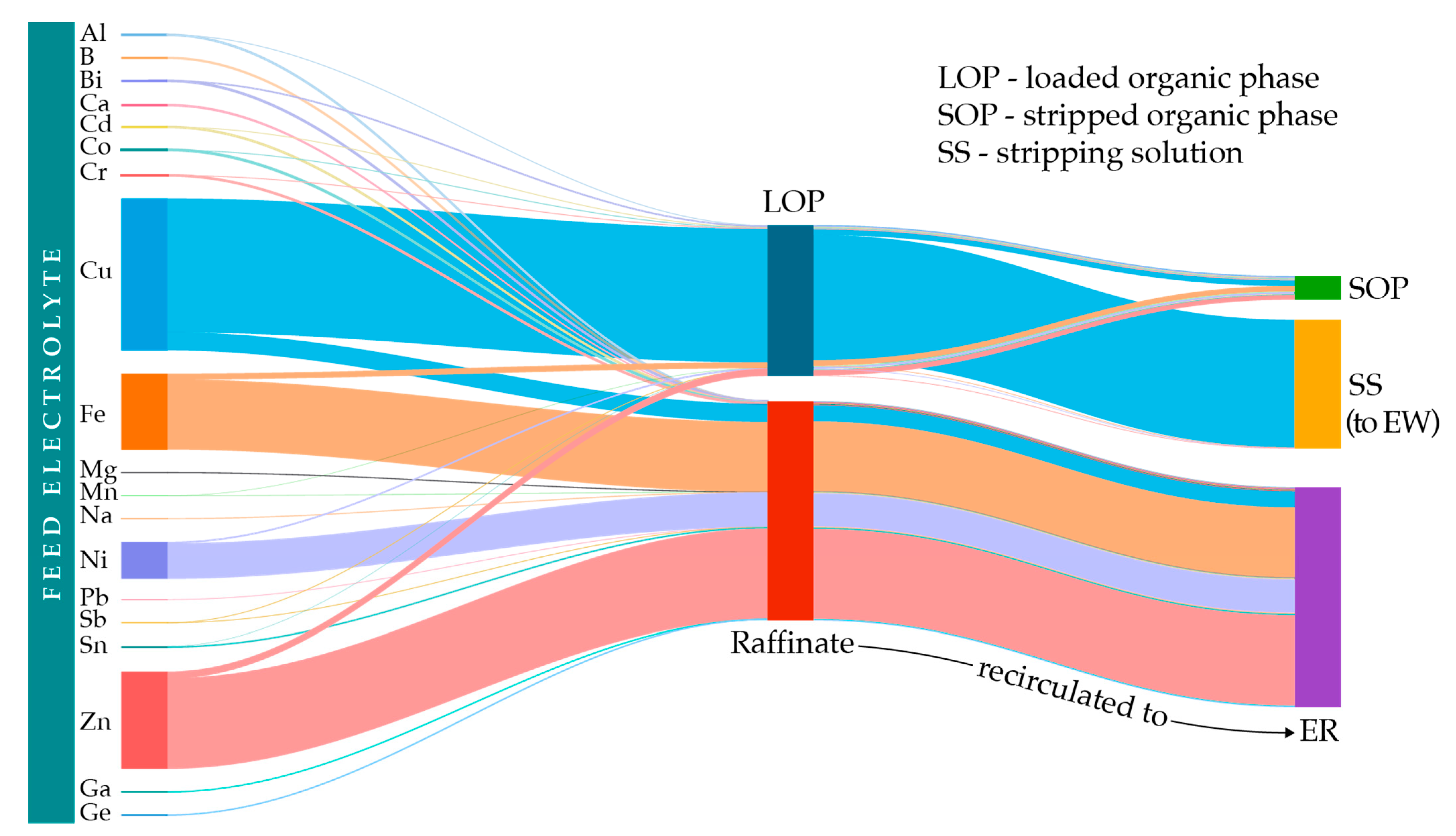
- Optimized process conditions enable selective Cu extraction among highly abundant Fe, Zn, Pb, Ni, and Sn and trace elements (i.e., Al, Co, Cr, Mg, Na, Sb, Ga, Ge)
- Transfer monitoring, distribution, and methodology for additional valorization of metals
1. Introduction
2. Experimental
2.1. Material
2.2. Sole E-Waste Preparation Process
2.3. Reagents and Procedure
2.4. Analytical Methods
3. Results and Discussion
3.1. Feed Composition
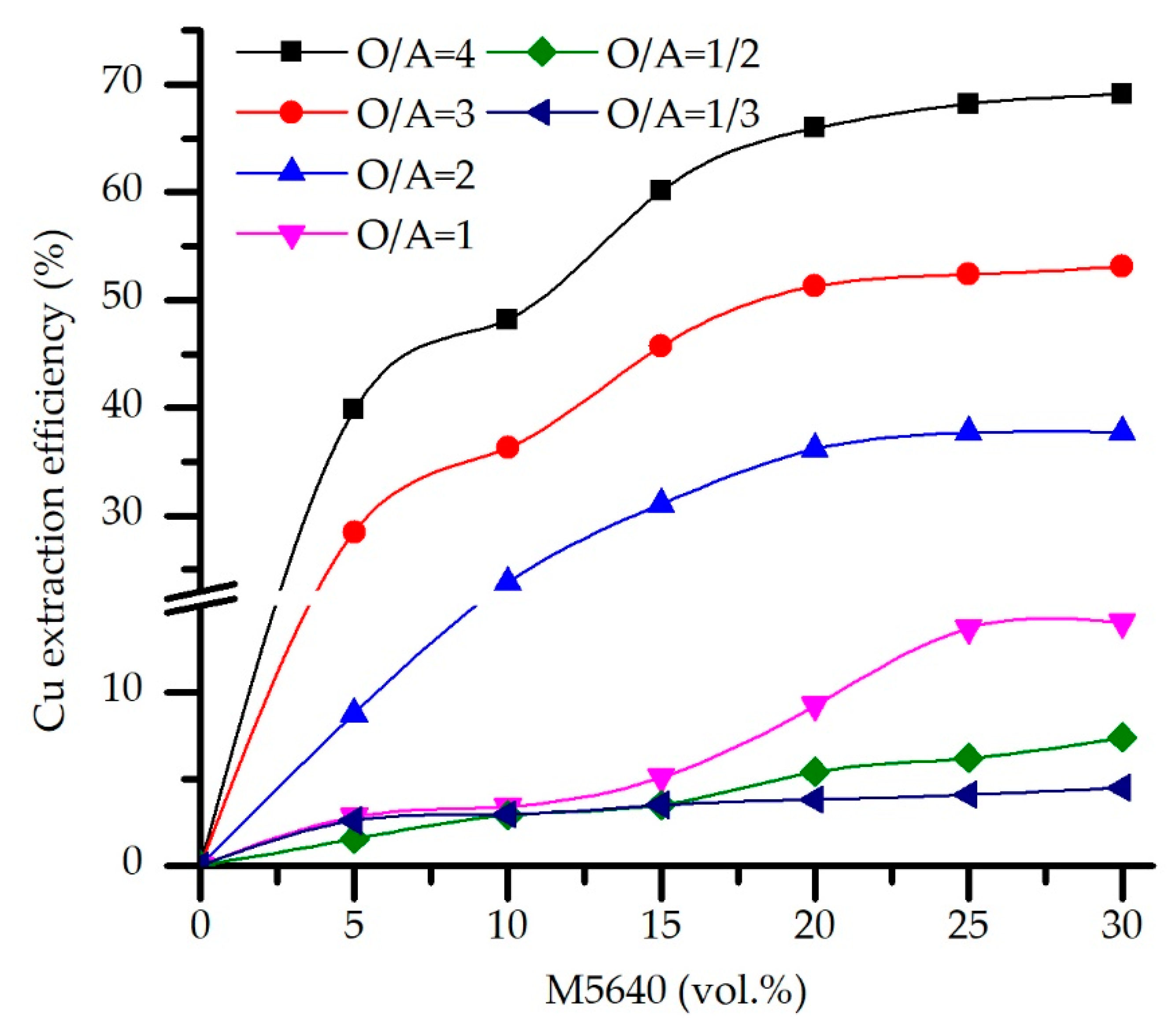
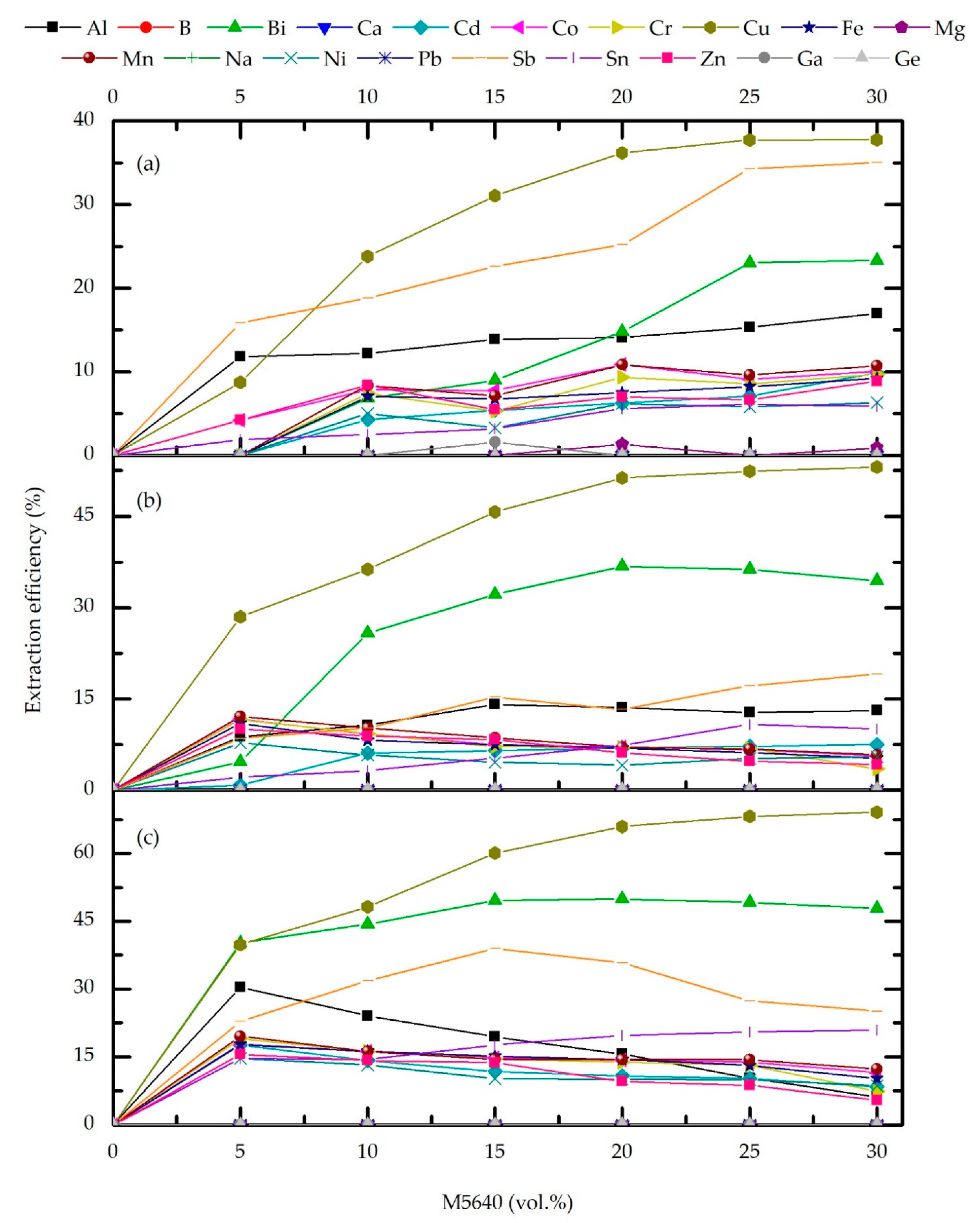
3.2. Solvent Extraction (SX) Optimization
3.2.1. Effect of Extractant Concentrations and O/A Ratio
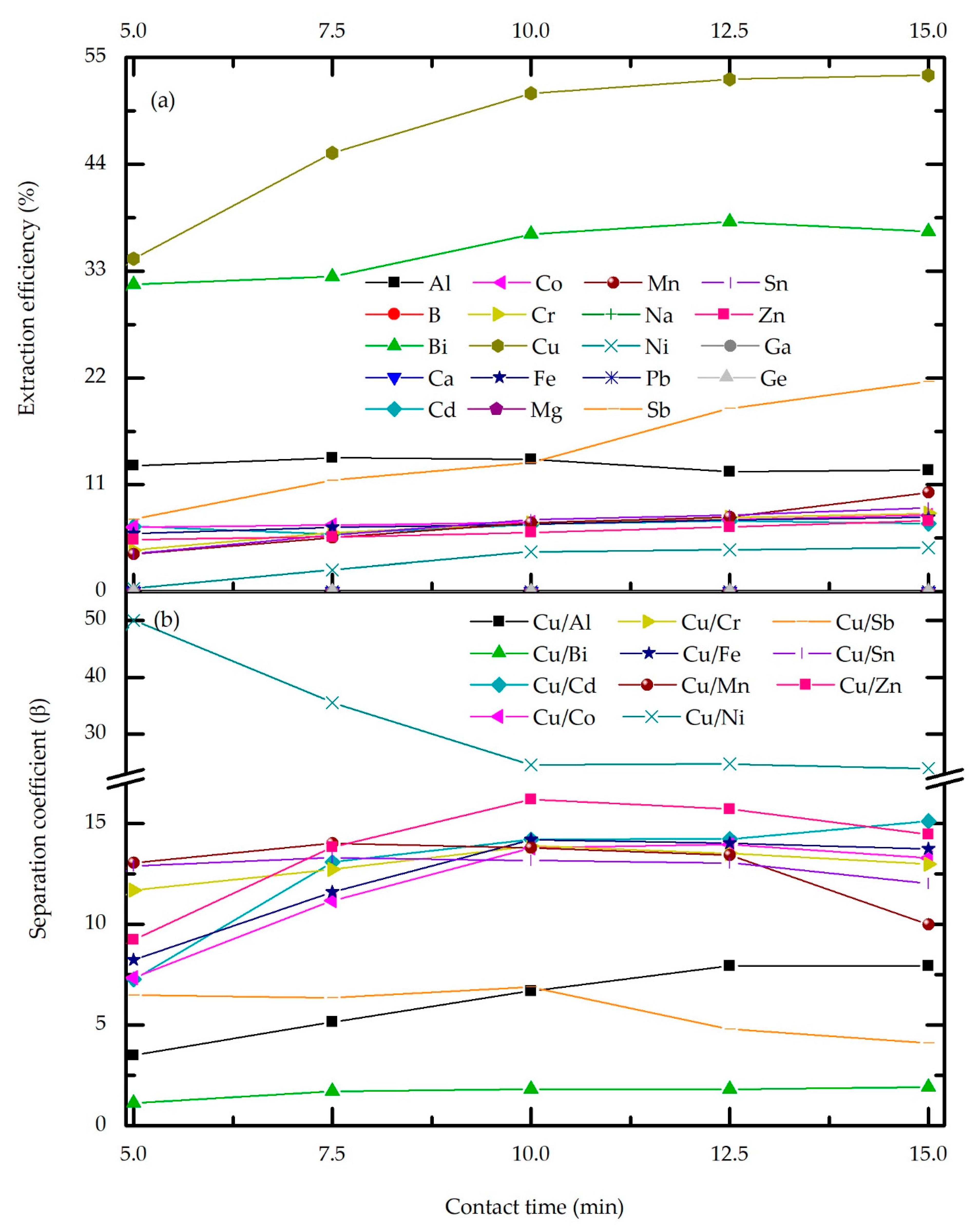
3.2.2. Effect of Phase Contact Time
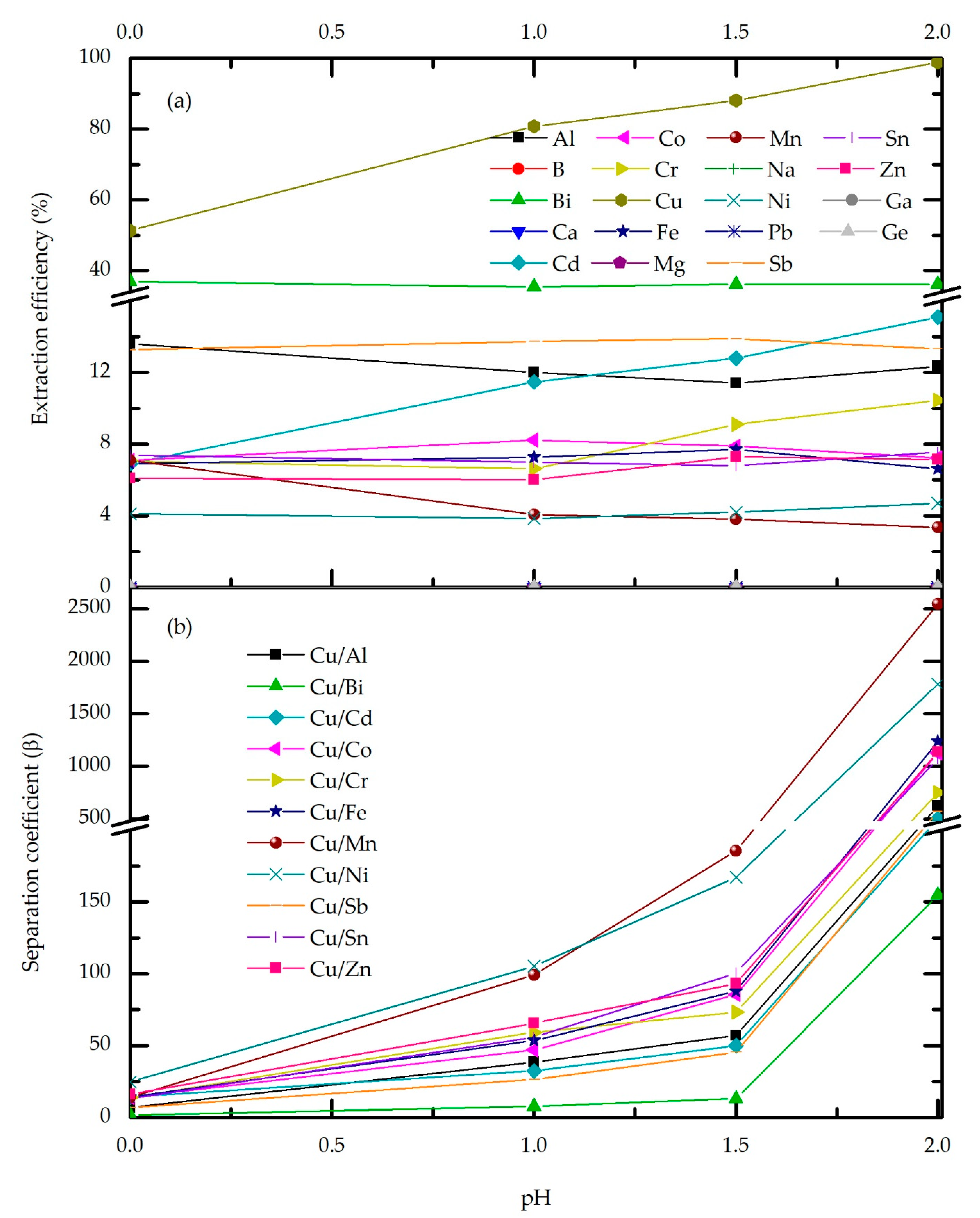
3.2.3. Effect of Feed pH
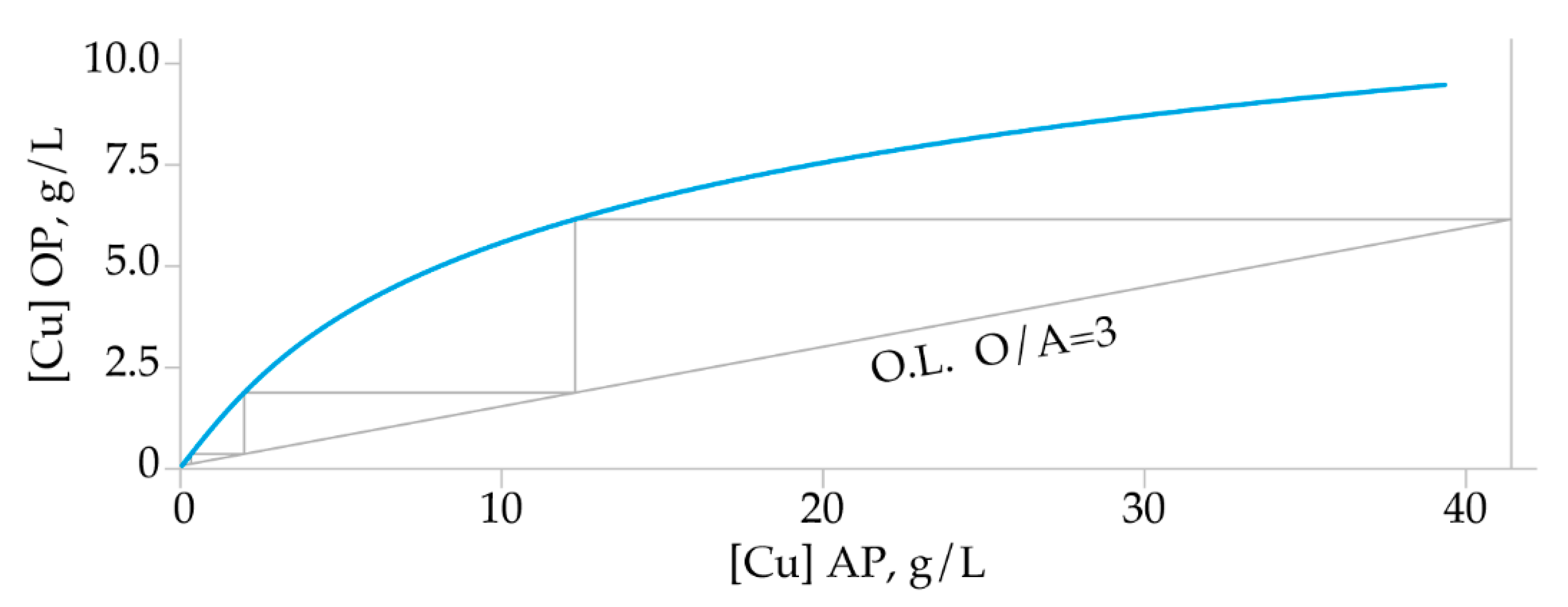
3.2.4. McCabe-Thiele Extraction Diagram
3.3. Stripping Optimization
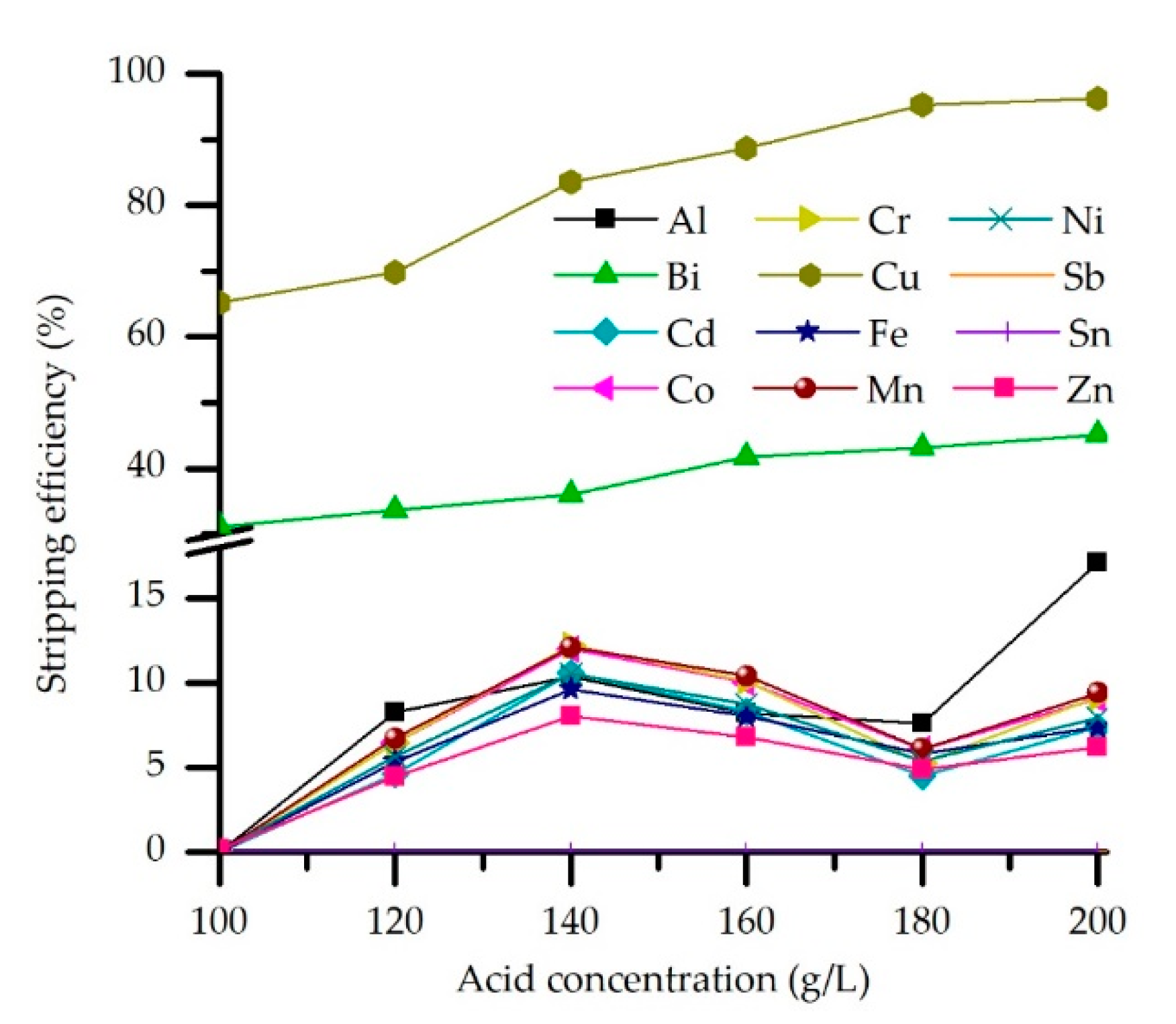
3.3.1. Effect of the Acid Concentration
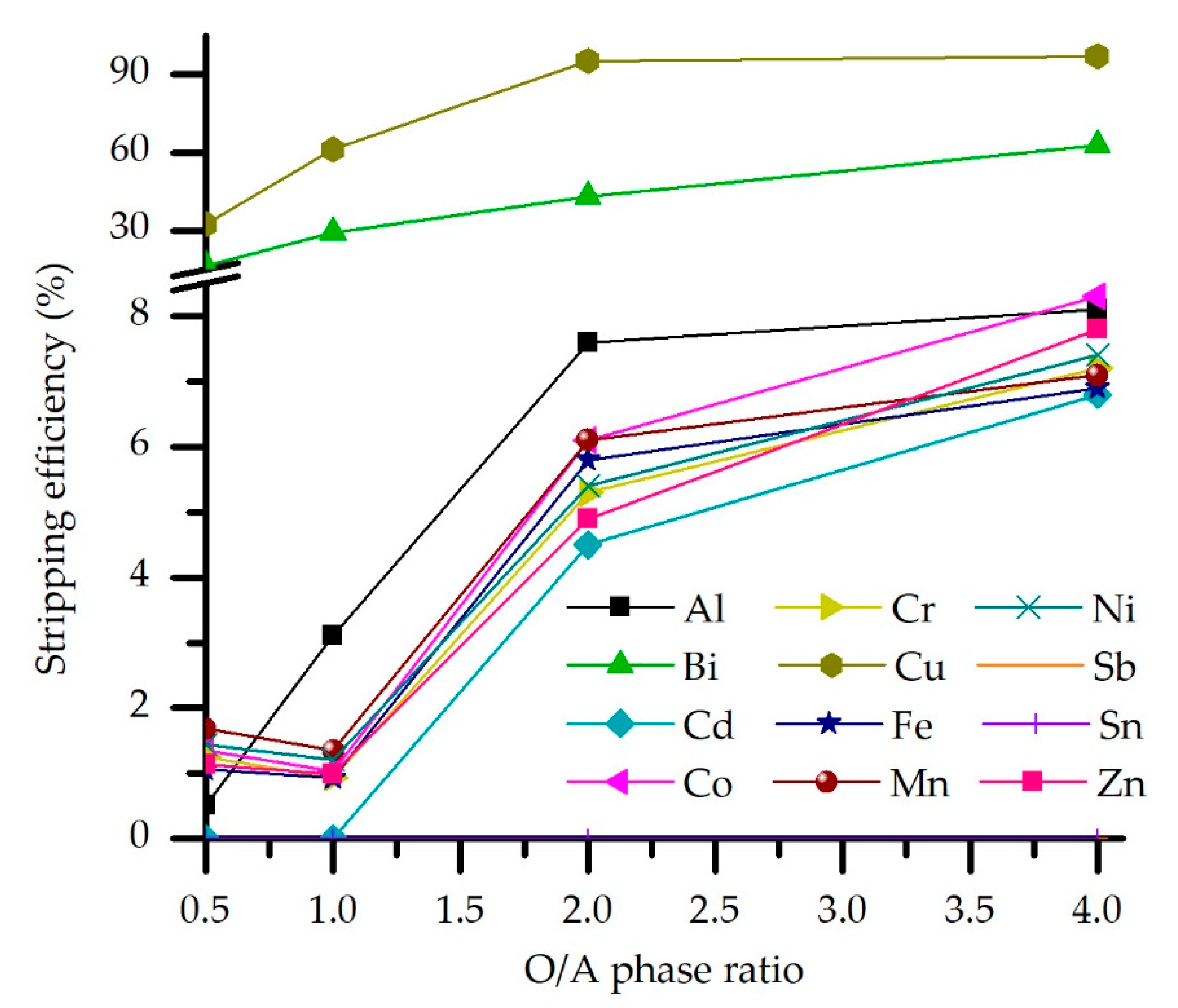
3.3.2. Effect of O/A Stripping Ratio
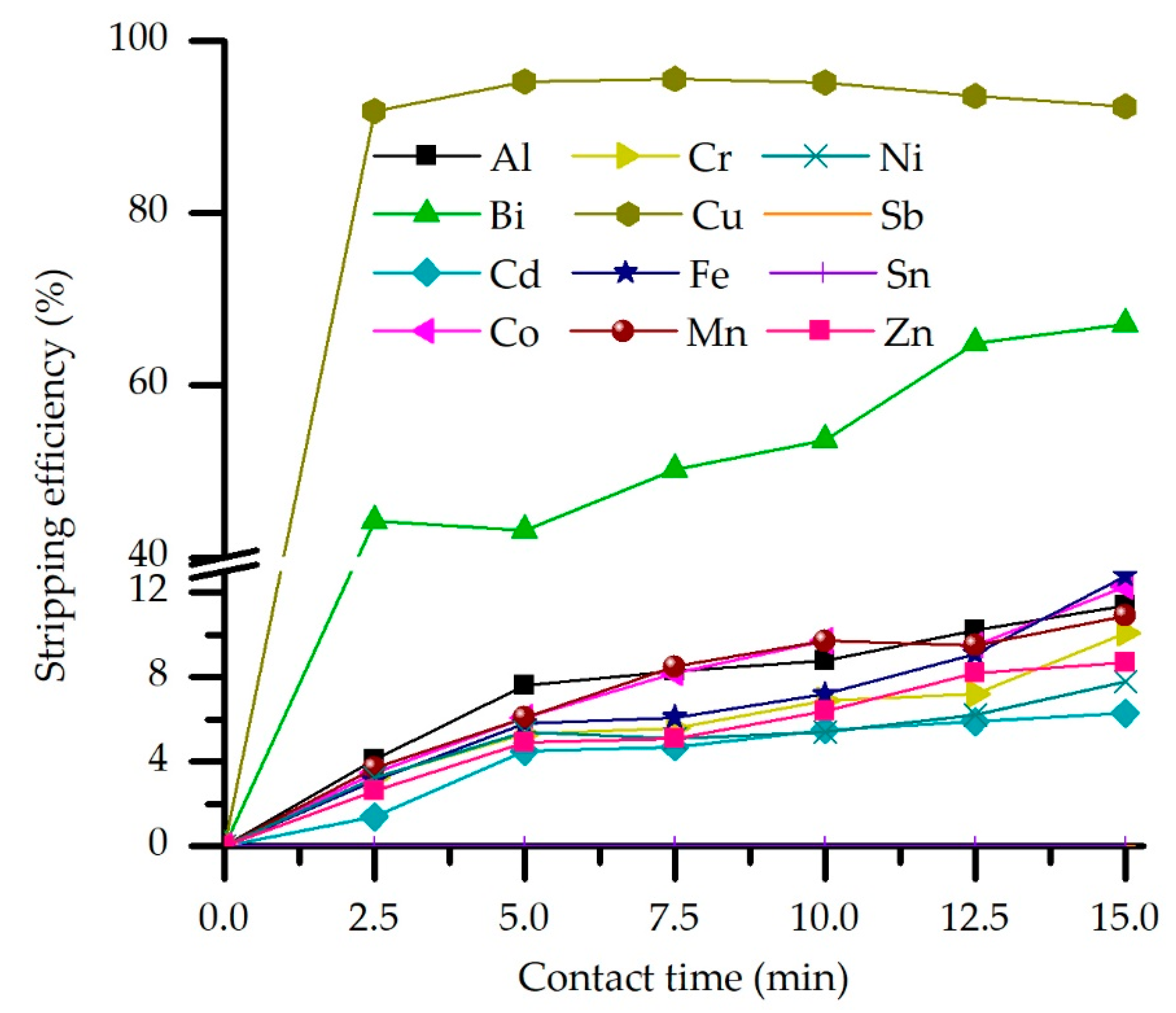
3.3.3. Effect of Stripping Contact Time
3.3.4. Effect of Extractant Concentration in the Loaded Organic Phase
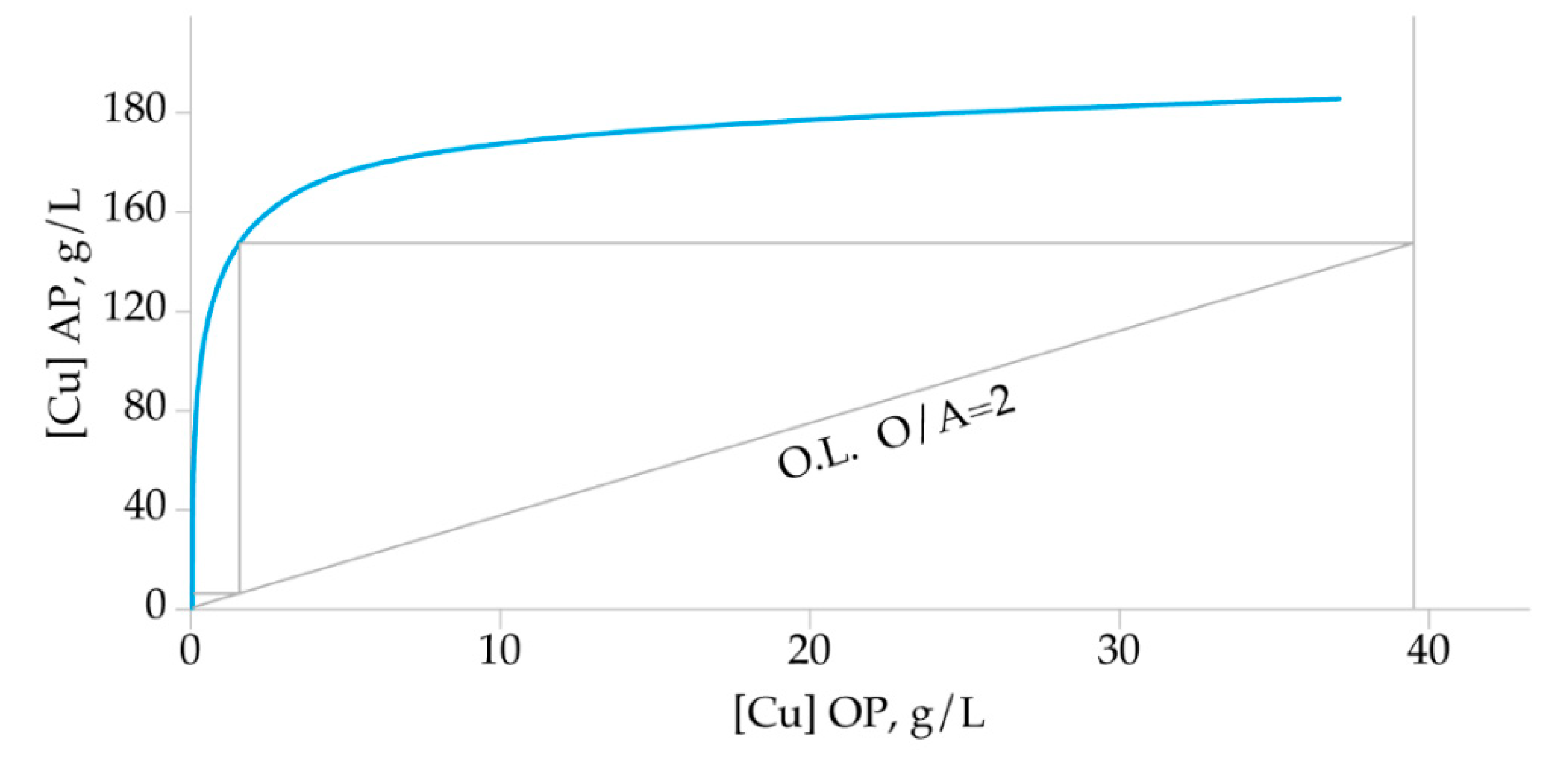
3.3.5. McCabe-Thiele Stripping Diagram
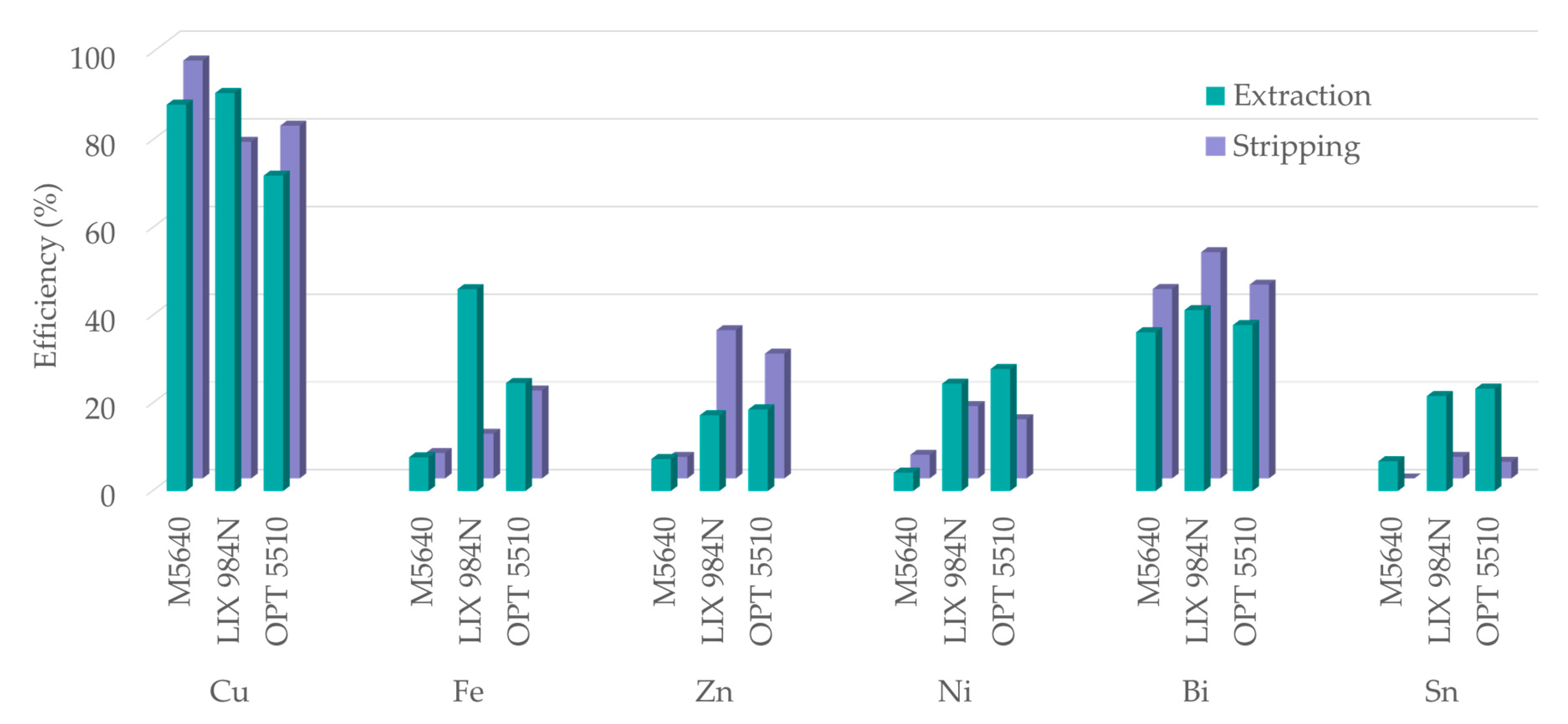
3.4. Solvent Extraction with LIX® 984N and OPT 5510
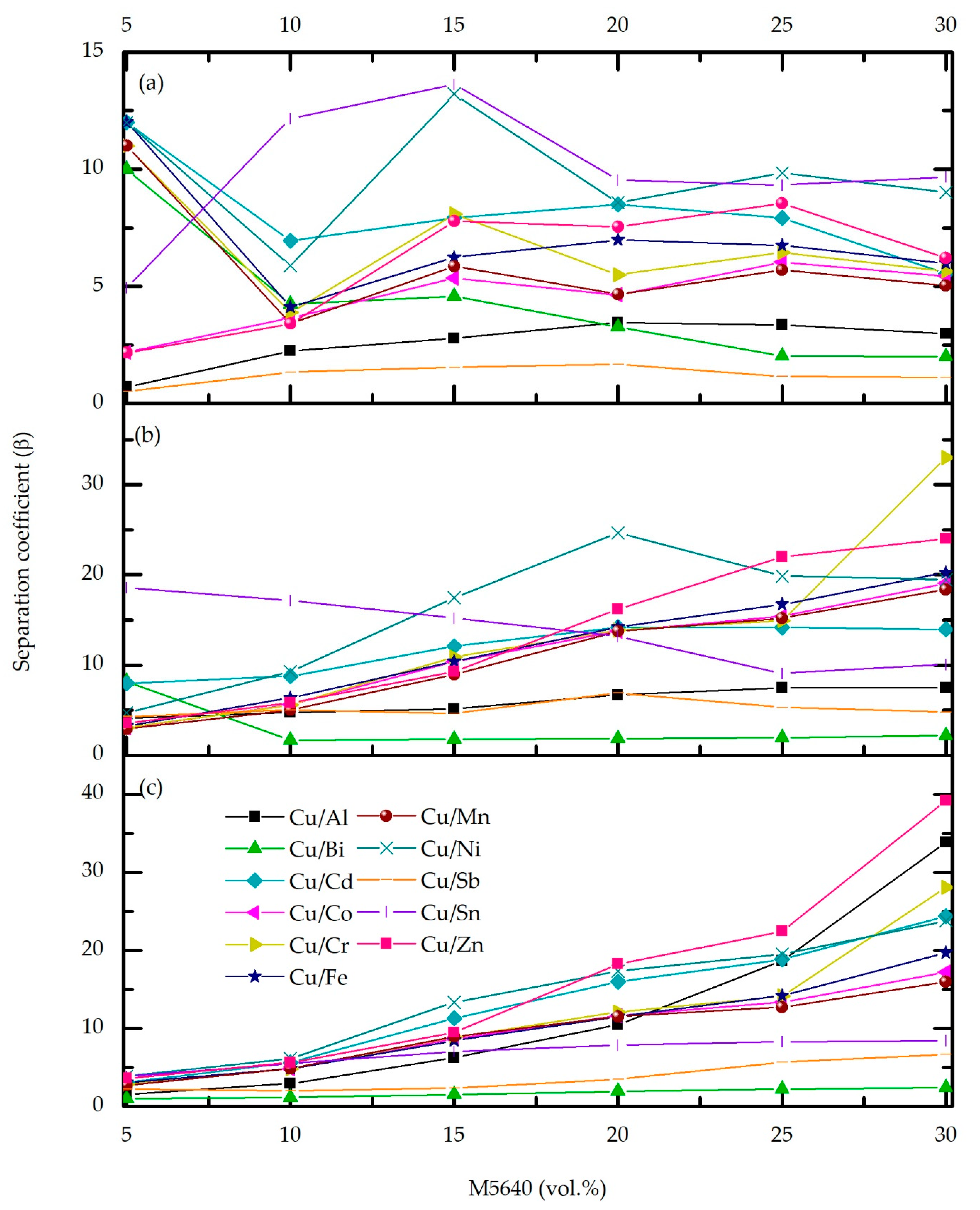
3.5. Copper and Metal Impurities Distribution
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Worell, E.; Reuter, M.A. Handbook of Recycling; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 978-0-12-396459-5. [Google Scholar]
- Dutta, D.; Panda, R.; Kumari, A.; Goel, S.; Jha, M.K. Sustainable Recycling Process for Metals Recovery from Used Printed Circuit Boards (PCBs). Sustain. Mater. Technol. 2018, 17, e00066. [Google Scholar] [CrossRef]
- Wang, L.; Li, Q.; Sun, X.; Wang, L. Separation and Recovery of Copper from Waste Printed Circuit Boards Leach Solution Using Solvent Extraction with Acorga M5640 as Extractant. Sep. Sci. Technol. 2019, 54, 1302–1311. [Google Scholar] [CrossRef]
- Ghosh, B.; Ghosh, M.K.; Parhi, P.; Mukherjee, P.S.; Mishra, B.K. Waste Printed Circuit Boards Recycling: An Extensive Assessment of Current Status. J. Clean. Prod. 2015, 94, 5–19. [Google Scholar] [CrossRef]
- Forti, V.; Baldé, C.P.; Kuehr, R.; Bel, G. The Global E-Waste Monitor 2020; United Nations University (UNU)/United Nations Institute for Training and Research (UNITAR)—co-hosted SCYCLE Programme, International Telecommunication Union (ITU) & International Solid Waste Association (ISWA): Bonn, Germany; Geneva, Switzerland; Rotterdam, The Netherlands, 2020; ISBN 978-92-808-9114-0. [Google Scholar]
- Cayumil, R.; Khanna, R.; Rajarao, R.; Mukherjee, P.S.; Sahajwalla, V. Concentration of Precious Metals during Their Recovery from Electronic Waste. Waste Manag. 2016, 57, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.; Fellman, J.; Jokilaakso, A.; Klemettinen, L.; Marjakoski, M. Behavior of Waste Printed Circuit Board (WPCB) Materials in the Copper Matte Smelting Process. Metals 2018, 8, 887. [Google Scholar] [CrossRef] [Green Version]
- Rai, V.; Liu, D.; Xia, D.; Jayaraman, Y.; Gabriel, J.-C.P. Electrochemical Approaches for the Recovery of Metals from Electronic Waste: A Critical Review. Recycling 2021, 6, 53. [Google Scholar] [CrossRef]
- Hagelüken, C. Recycling of Electronic Scrap at Umicore’s Integrated Metals Smelter and Refinery. World Metall ERZMETALL 2006, 11, 152–161. [Google Scholar]
- Lennartsson, A.; Engström, F.; Samuelsson, C.; Björkman, B.; Pettersson, J. Large-Scale WEEE Recycling Integrated in an Ore-Based Cu-Extraction System. J. Sustain. Metall. 2018, 4, 222–232. [Google Scholar] [CrossRef] [Green Version]
- Ariizumi, M.; Takagi, M.; Inoue, O.; Oguma, N. Integrated processing of e-scrap at Naoshima smelter and refinery. In Proceedings of the Copper 2016, Kobe, Japan, 13–16 November 2016; Volume 6, p. RW 1–2. [Google Scholar]
- Kamberović, Ž.; Ranitović, M.; Korać, M.; Jovanović, N.; Tomović, B.; Gajić, N. Pyro-Refining of Mechanically Treated Waste Printed Circuit Boards in a DC Arc-Furnace. J. Sustain. Metall. 2018, 4, 251–259. [Google Scholar] [CrossRef]
- Forsén, O.; Aromaa, J.; Lundström, M. Primary Copper Smelter and Refinery as a Recycling Plant—A System Integrated Approach to Estimate Secondary Raw Material Tolerance. Recycling 2017, 2, 19. [Google Scholar] [CrossRef] [Green Version]
- Cui, J.; Zhang, L. Metallurgical Recovery of Metals from Electronic Waste: A Review. J. Hazard. Mater. 2008, 158, 228–256. [Google Scholar] [CrossRef] [PubMed]
- Djokić, J.; Jovančićević, B.; Brčeski, I.; Ranitović, M.; Gajić, N.; Kamberović, Ž. Leaching of Metastannic Acid from E-Waste by-Products. J. Mater. Cycles Waste Manag. 2020, 22, 1899–1912. [Google Scholar] [CrossRef]
- Vasilyev, F.; Virolainen, S.; Sainio, T. Modeling the Liquid–Liquid Extraction Equilibrium of Iron (III) with Hydroxyoxime Extractant and Equilibrium-Based Simulation of Counter-Current Copper Extraction Circuits. Chem. Eng. Sci. 2018, 175, 267–277. [Google Scholar] [CrossRef]
- Das, S.C.; Gopala Krishna, P. Effect of Fe(III) during Copper Electrowinning at Higher Current Density. Int. J. Miner. Process. 1996, 46, 91–105. [Google Scholar] [CrossRef]
- Kamberović, Ž.; Ranitović, M.; Korać, M.; Andjić, Z.; Gajić, N.; Djokić, J.; Jevtić, S. Hydrometallurgical Process for Selective Metals Recovery from Waste-Printed Circuit Boards. Metals 2018, 8, 441. [Google Scholar] [CrossRef] [Green Version]
- Kamberović, Ž.; Korać, M.; Ranitović, M. Hydrometallurgical process for extraction of metals from electronic waste-part II: Development of the processes for the recovery of copper from printed circuit boards (PCB). Metalurgija 2011, 17, 139–149. [Google Scholar]
- Lister, T.E.; Wang, P.; Anderko, A. Recovery of Critical and Value Metals from Mobile Electronics Enabled by Electrochemical Processing. Hydrometallurgy 2014, 149, 228–237. [Google Scholar] [CrossRef] [Green Version]
- Robinson, B.H. E-Waste: An Assessment of Global Production and Environmental Impacts. Sci. Total Environ. 2009, 408, 183–191. [Google Scholar] [CrossRef]
- Kaya, M. Recovery of Metals and Nonmetals from Electronic Waste by Physical and Chemical Recycling Processes. Waste Manag. 2016, 57, 64–90. [Google Scholar] [CrossRef]
- Jiang, F.; Yin, S.; Zhang, L.; Peng, J.; Ju, S.; Miller, J.D.; Wang, X. Solvent Extraction of Cu(II) from Sulfate Solutions Containing Zn(II) and Fe(III) Using an Interdigital Micromixer. Hydrometallurgy 2018, 177, 116–122. [Google Scholar] [CrossRef]
- Tuncuk, A.; Stazi, V.; Akcil, A.; Yazici, E.Y.; Deveci, H. Aqueous Metal Recovery Techniques from E-Scrap: Hydrometallurgy in Recycling. Miner. Eng. 2012, 25, 28–37. [Google Scholar] [CrossRef]
- Agarwal, S.; Ferreira, A.E.; Santos, S.M.C.; Reis, M.T.A.; Ismael, M.R.C.; Correia, M.J.N.; Carvalho, J.M.R. Separation and Recovery of Copper from Zinc Leach Liquor by Solvent Extraction Using Acorga M5640. Int. J. Miner. Process. 2010, 97, 85–91. [Google Scholar] [CrossRef]
- Ochromowicz, K.; Chmielewski, T. Solvent Extraction of Copper(II) from Concentrated Leach Liquors. Physicochem. Probl. Miner. Process. 2013, 49, 357–367. [Google Scholar] [CrossRef]
- Habashi, F. A Textbook of Hydrometallurgy; Métallurgie Extractive Québec: Sainte-Foy, QC, Canada, 1999; ISBN 978-2-9803247-7-2. [Google Scholar]
- Jha, M.K.; Gupta, D.; Choubey, P.K.; Kumar, V.; Jeong, J.; Lee, J. Solvent Extraction of Copper, Zinc, Cadmium and Nickel from Sulfate Solution in Mixer Settler Unit (MSU). Sep. Purif. Technol. 2014, 122, 119–127. [Google Scholar] [CrossRef]
- Kumari, A.; Jha, M.K.; Lee, J.; Singh, R.P. Clean Process for Recovery of Metals and Recycling of Acid from the Leach Liquor of PCBs. J. Clean. Prod. 2016, 112, 4826–4834. [Google Scholar] [CrossRef]
- Li, X.; Wei, C.; Deng, Z.; Li, C.; Fan, G.; Rong, H.; Zhang, F. Extraction and Separation of Indium and Copper from Zinc Residue Leach Liquor by Solvent Extraction. Sep. Purif. Technol. 2015, 156, 348–355. [Google Scholar] [CrossRef]
- Deep, A.; Kumar, P.; Carvalho, J.M.R. Recovery of Copper from Zinc Leaching Liquor Using ACORGA M5640. Sep. Purif. Technol. 2010, 76, 21–25. [Google Scholar] [CrossRef]
- Ferreira, A.E.; Agarwal, S.; Machado, R.M.; Gameiro, M.L.F.; Santos, S.M.C.; Reis, M.T.A.; Ismael, M.R.C.; Correia, M.J.N.; Carvalho, J.M.R. Extraction of Copper from Acidic Leach Solution with Acorga M5640 Using a Pulsed Sieve Plate Column. Hydrometallurgy 2010, 104, 66–75. [Google Scholar] [CrossRef]
- Vander Linden, J. Selective Recuperation of Copper by Supported Liquid Membrane (SLM) Extraction. J. Membr. Sci. 1998, 139, 125–135. [Google Scholar] [CrossRef]
- Kongolo, K.; Ngoie, N.; Francis, K.; Patric, T. Improving the efficiency of solvent extraction of copper by combination of hydroxyoximic extractants. In Proceedings of the International Conference on Metal Solvent Extraction 2015, Three Gorges, China, 10–14 November 2015; pp. 117–126. [Google Scholar]
- Asghari, H.; Safarzadeh, M.S.; Asghari, G.; Moradkham, D. The Effect of Impurities on the Extraction of Copper from Sulfate Medium Using LIX®984N in Kerosene. Russ. J. Non-Ferrous Metals 2009, 50, 89–96. [Google Scholar] [CrossRef]
- Banza, A.N.; Gock, E.; Kongolo, K. Base Metals Recovery from Copper Smelter Slag by Oxidising Leaching and Solvent Extraction. Hydrometallurgy 2002, 67, 63–69. [Google Scholar] [CrossRef]
- Naveed, S.; Nawaz, Z. Copper Extraction from Copper Rolling Mills Scraps using Solvent “aryl aldoxime, 2-hydroxy-5-nonylbenzaldoxime” (ACORGA-M5640). J. Chem. Soc. Pak. 2006, 28, 44–50. [Google Scholar]
- De la Torre, E.; Vargas, E.; Ron, C.; Gámez, S. Europium, Yttrium, and Indium Recovery from Electronic Wastes. Metals 2018, 8, 777. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Wang, Y.; Cui, L.; Gao, J.; Guo, Y.; Cheng, F. A Sustainable Approach for Advanced Removal of Iron from CFA Sulfuric Acid Leach Liquor by Solvent Extraction with P507. Sep. Purif. Technol. 2020, 251, 117371. [Google Scholar] [CrossRef]
- Ocaña, N.; Alguacil, F.J. Solvent Extraction of Iron(III) by MOC-55 TD: Experimental Equilibrium Study and Demonstration of Lack of Influence on Copper(II) Extraction from Sulphate Solutions. Hydrometallurgy 1998, 48, 239–249. [Google Scholar] [CrossRef]
- Sridhar, V.; Verma, J.K. Recovery of Copper, Nickel and Zinc from Sulfate Solutions by Solvent Extraction Using LIX 984N. E-J. Chem. 2011, 8, S434–S438. [Google Scholar] [CrossRef]
- Inoue, K.; Mirvaliev, R.; Yoshizuka, K.; Ohto, K.; Babasaki, S.-Y. Solvent extraction of TIN(IV) with PC-88A from sulfuric acid solutions containing chloride ions. Solvent. Extr. Res. Dev. Jpn. 2001, 8, 21–25. [Google Scholar]

| Element | Metallic Granulate | Anodes | Electrolyte | ||
|---|---|---|---|---|---|
| % | % | ppm | mg/L | g/L | |
| Cu | 69.12 | 83.40 | - | - | 41.37 |
| Fe | 5.90 | 3.90 | - | - | 20.67 |
| Zn | 12.71 | 5.40 | - | - | 26.55 |
| Ni | 1.68 | 2.32 | - | - | 9.89 |
| Sn | 5.89 | 7.27 | - | 453.1 | - |
| Pb | 3.30 | 3.52 | - | 3.1 | - |
| Bi | n.a. | 0.04 | - | 112.9 | - |
| Ca | n.a. | 0.10 | - | 346.3 | - |
| Co | 0.01 | 0.05 | - | 163.4 | - |
| Cr | 0.12 | 0.03 | - | 95.8 | - |
| Mg | n.a. | 0.03 | - | 61.1 | - |
| Ag | 0.64 | 0.64 | - | <DL | - |
| Au | 30 ppm | 0.11 | - | <DL | - |
| Al | 0.12 | - | 63 | 19.3 | - |
| Cd | 0.04 | - | 62 | 21.7 | - |
| Mn | 0.10 | - | 84 | 29.7 | - |
| B | n.a. | - | 32 | 8.7 | - |
| Na | n.a. | - | 120 | 41.8 | - |
| Sb | n.a. | - | 23 | 5.8 | - |
| Ga | n.a. | - | 47 | 12.5 | - |
| Ge | n.a. | - | 4 | 1.2 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Djokić, J.; Radovanović, D.; Nikolovski, Z.; Andjić, Z.; Kamberović, Ž. Influence of Electrolyte Impurities from E-Waste Electrorefining on Copper Extraction Recovery. Metals 2021, 11, 1383. https://doi.org/10.3390/met11091383
Djokić J, Radovanović D, Nikolovski Z, Andjić Z, Kamberović Ž. Influence of Electrolyte Impurities from E-Waste Electrorefining on Copper Extraction Recovery. Metals. 2021; 11(9):1383. https://doi.org/10.3390/met11091383
Chicago/Turabian StyleDjokić, Jovana, Dragana Radovanović, Zlatko Nikolovski, Zoran Andjić, and Željko Kamberović. 2021. "Influence of Electrolyte Impurities from E-Waste Electrorefining on Copper Extraction Recovery" Metals 11, no. 9: 1383. https://doi.org/10.3390/met11091383
APA StyleDjokić, J., Radovanović, D., Nikolovski, Z., Andjić, Z., & Kamberović, Ž. (2021). Influence of Electrolyte Impurities from E-Waste Electrorefining on Copper Extraction Recovery. Metals, 11(9), 1383. https://doi.org/10.3390/met11091383