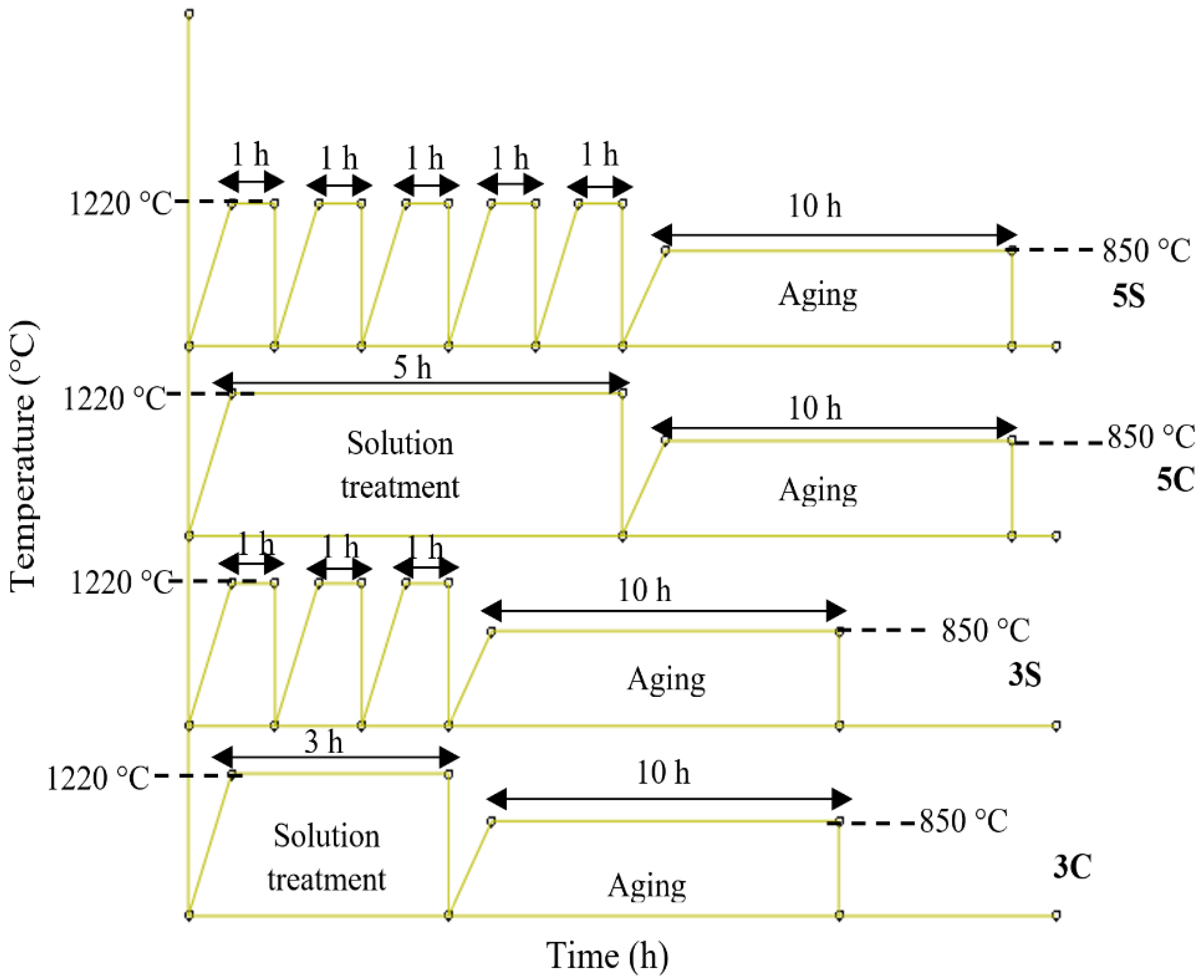
3.2. Solution Heat Treatment
The microstructures of the 3C and 5C samples are shown in
Figure 3a,c. As one can see, the solution heat treatment resulted in the dissolution and spheroidization of carbides leading to the homogeneity of the microstructure. The increase of solution time from 3 h to 5 h coarsened the grain size slightly but volume fraction of carbides almost remained unchanged. The increase of solution treatment time from 3 h to 5 h increased the grain size from ≈ 210 μm to ≈ 520 μm. The presence of parallel lines in the matrix is indicative of athermal ε martensite (hcp
1). According to the optical images, it seems that 5C sample has more athermal ε martensite than 3C sample. According to Huang’s work [
19], the volume fraction of athermal ε martensite was found to be smaller in fine grained alloy, but it concurrently increased with increasing grain size as confirmed in the present work (compare
Figure 3a,c). This can be attributed to the increase in the number of probable hcp embryos and lattice defects (twins and stacking faults) with increasing grain size and decrease of M
s temperature with decreasing grain size [
19].
Cyclic heat treatment significantly increased the volume fraction of athermal ε martensite, as shown in
Figure 3b,d, and also undissolved carbides can still be seen within the matrix and at the grain boundaries. It seems that the cyclic heat treatment was more effective in promoting γ
fcc → ε
hcp athermal martensite and provided more nucleation sites for ε martensite embryos. The thickness of the intergranular ε martensite varies in different regions and is closely related to the grain size; it was observed that the amount of athermal ε martensite in 5S sample was greater than that of the 3S sample. The increase of solution time and the number of quenching cycles increased the grain size and provided more nucleation sites for ε
hcp embryos (such as stacking fault defects) inducing more athermal ε martensite.
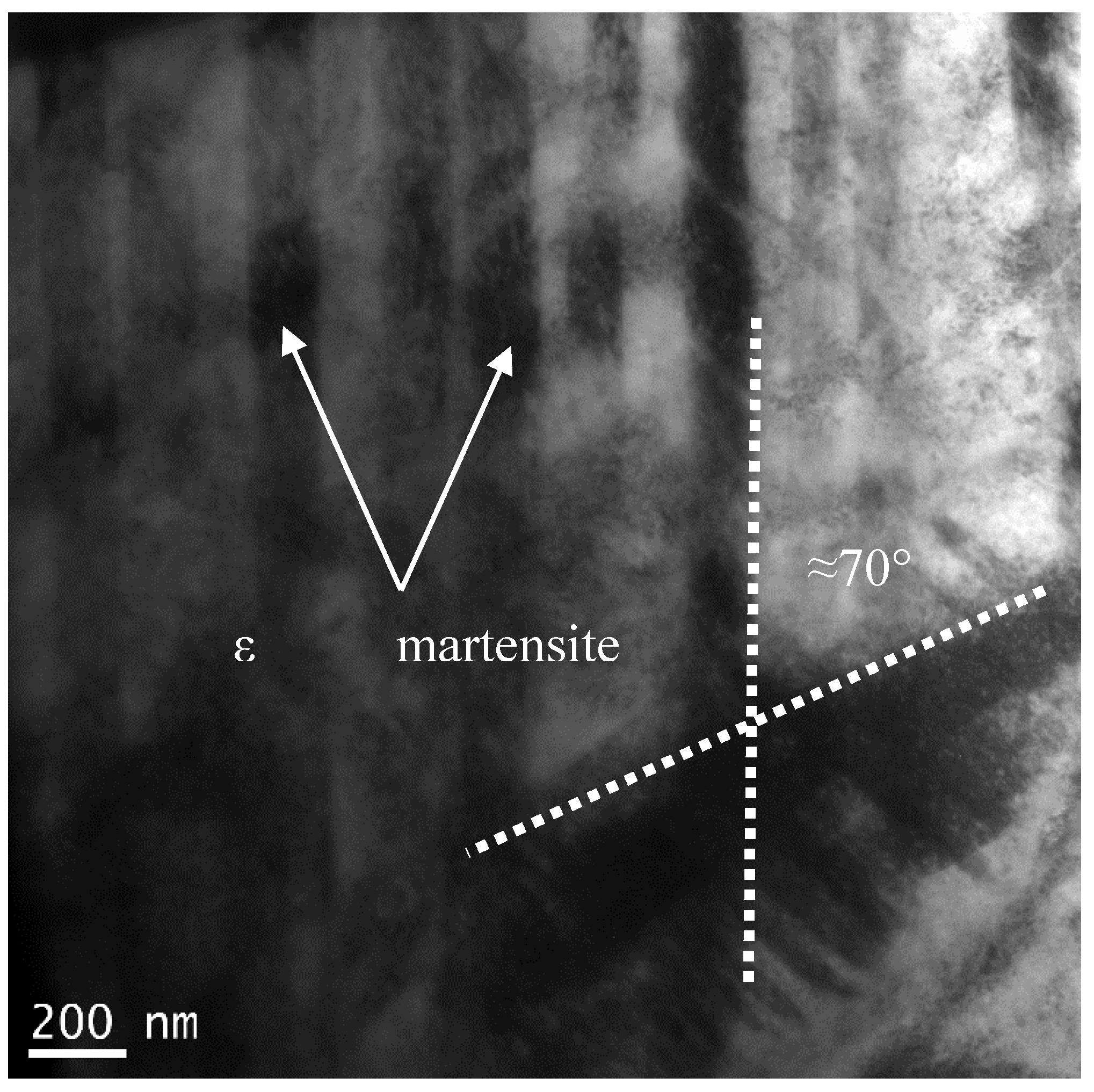
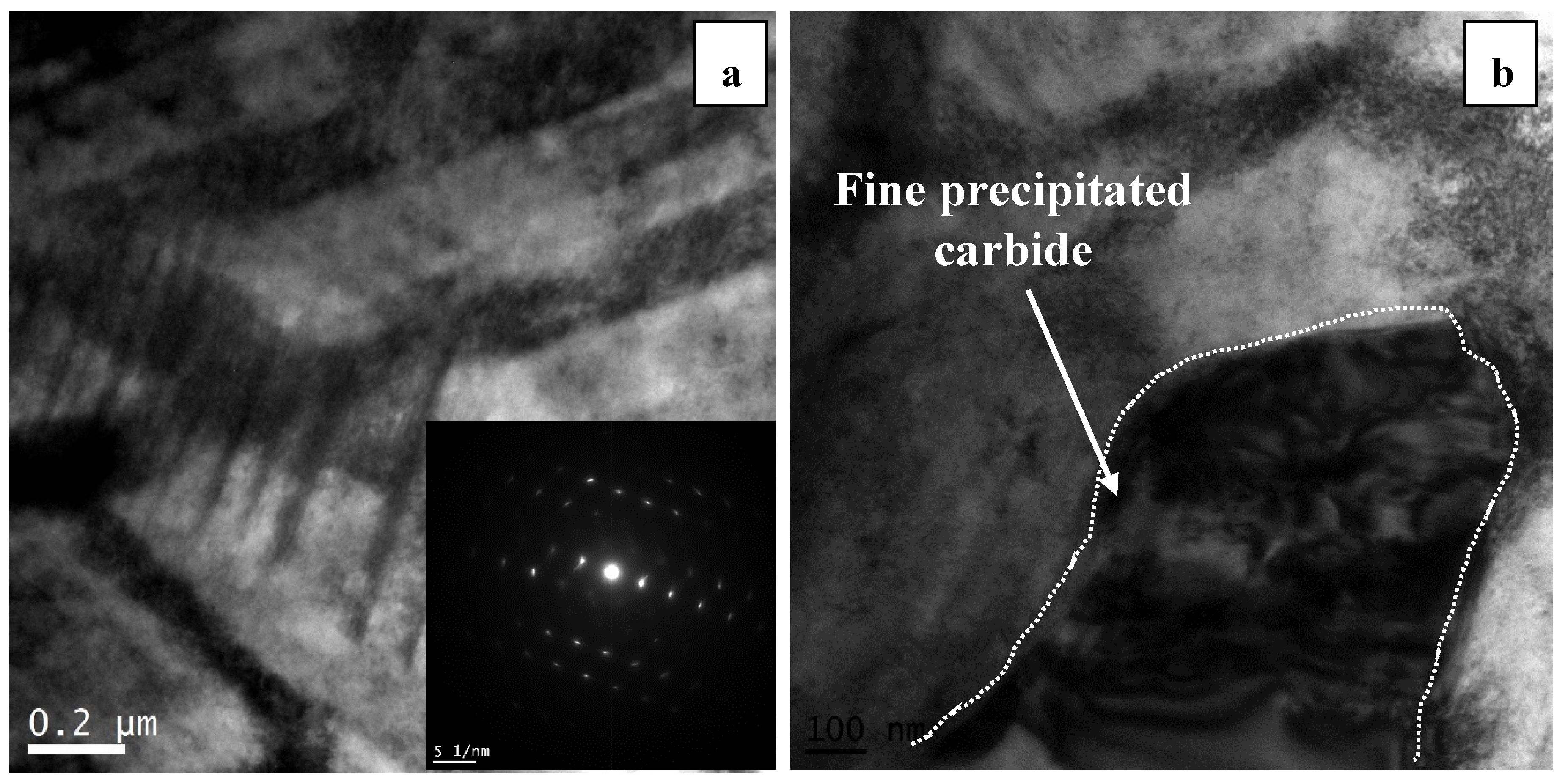
The bright field TEM image of the developed microstructure in sample 5S is shown in
Figure 4 in which cyclic quenching led to the generation of a series of parallel bands (intergranular striations) within the initial fcc grains. The angle between two athermal ε martensite bands is around 70° which is the same as the ideal angle of 70.52° between (111)
fcc planes on which ε martensite forms in accordance with Shoji–Nishiyama orientation relationship [
20].
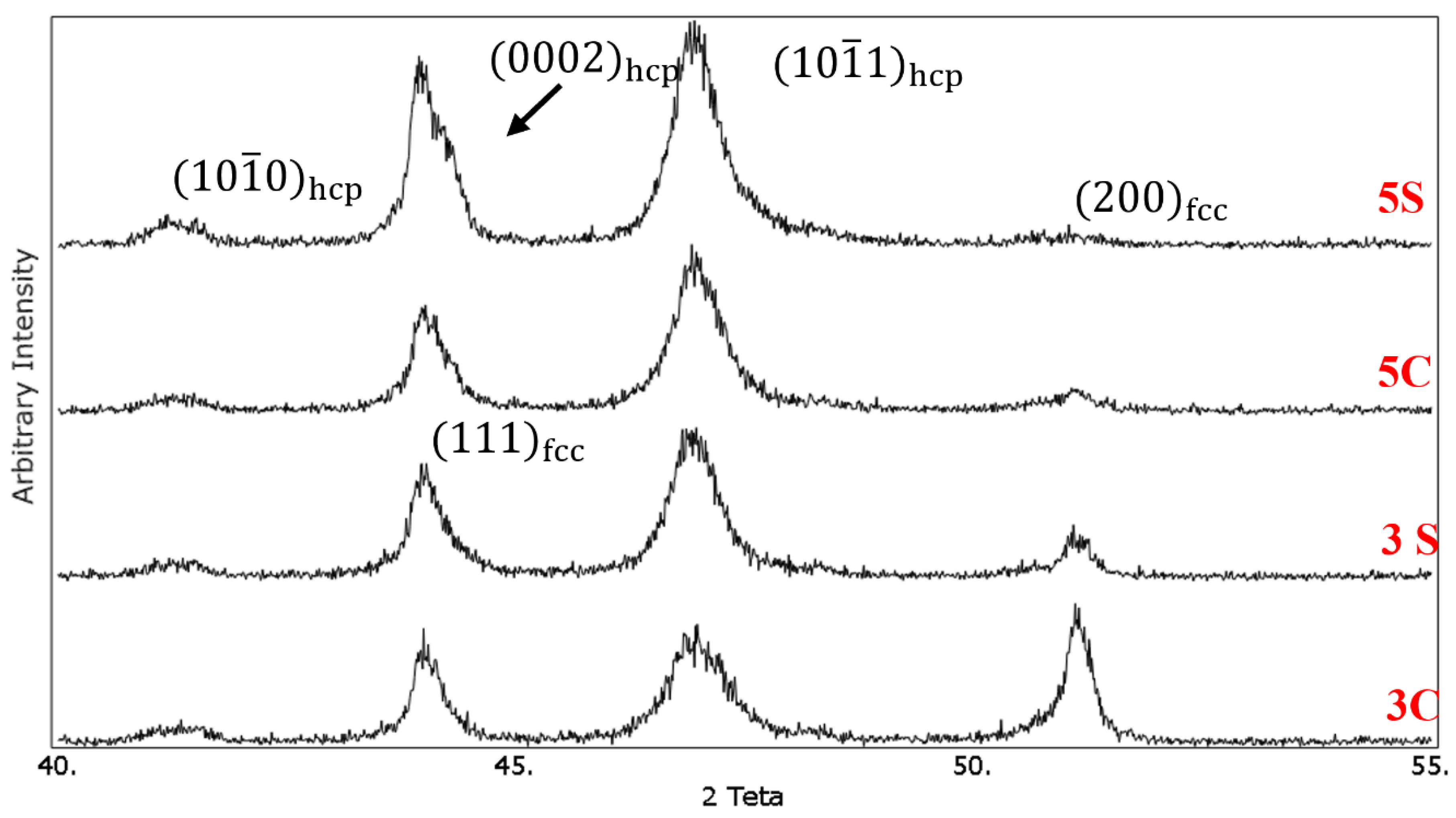
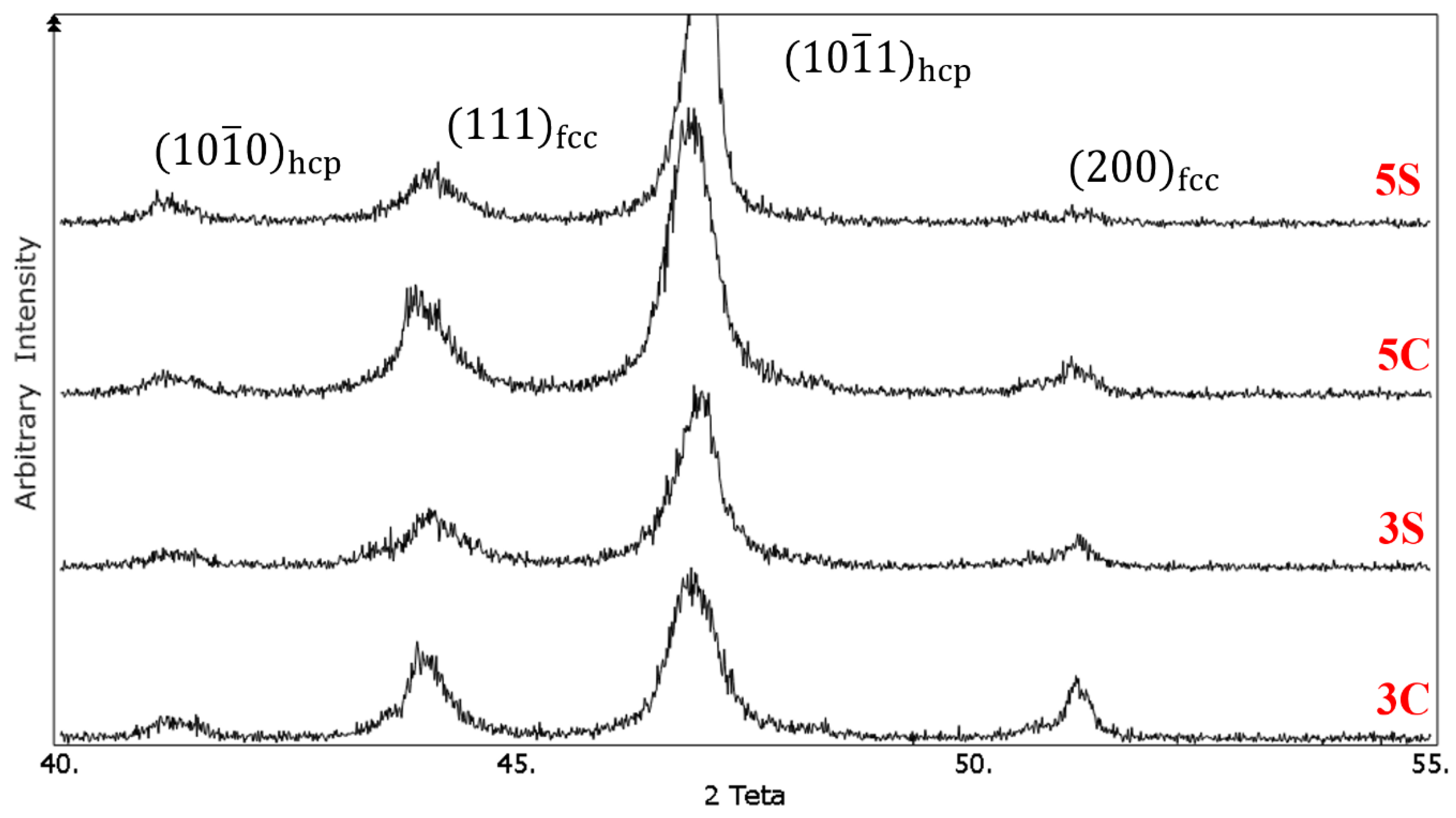
Figure 5 Shows the XRD patterns of the solution-treated samples under different conditions. The
and
diffraction peaks related to the fcc phase appeared at 2θ values of 43.75° and 50.75°, respectively. For the hcp phase,
,
and
peaks are located at 2θ values of 41°, 43.75°, and 46.75°, respectively. It was observed that cyclic solution heat treatment in sample 3S resulted in increasing the intensity of
peak and decreasing the intensity of
peak. The volume fraction of the hcp phase (
f hcp) was calculated quantitatively using Equation (1), and it was found that the volume fraction of the athermal ε martensite increased from 36% in sample 3C to 64% in sample 3S. It is interesting to note that the intensity of
peak for this sample nearly remained unchanged. The increase of quenching cycles from 3 to 5 increased the volume fraction of the athermal ε martensite. As one can see,
peak is almost vanished in 5C and 5S samples. In addition, new (0002)
hcp peak appeared at 2θ value of 43.75°. Using the same equation, the volume fraction of the athermal ε martensite in samples 5C and 5S was 81% and 90%, respectively. These results are in good agreement with the microstructural observation which was shown earlier.
Our previous studies [
8,
9] showed that quenching the Co-Cr-Mo alloy after solution treatment above 1100 °C promoted the formation of athermal ε martensite. It seems that the formation of high temperature defects which could act as potential sites for the nucleation of ε-martensite embryos is strongly favored at high cooling rates [
21]. In addition, the high cooling rate generates internal residual stress resulting in the partial transformation of metastable fcc → hcp ε martensite.
3.3. Isothermal Aging
In Co-Cr-Mo alloys, martensitic phase transformation can be induced in three different ways: (I) athermal martensite (hcp
1), (II) isothermal martensite (hcp
2, forming lamellar structure), and (III) stress-induced martensite. The formation of athermal martensite mainly depends upon the solution temperature rather than the holding time at this temperature and in most cases the volume fraction of this phase does not exceed 20% [
22]. This was attributed to the lack of enough defects necessary for spontaneous nucleation of ε martensite embryos during quenching [
21]. For this reason, isothermal phase transformation is usually conducted at temperatures within the range of 650–950 °C to complete the γ
fcc to ε
hcp phase transformation. All the solution-treated samples were isothermally aged at 850 °C for 10 h to form isothermal martensite and complete the martensitic phase transformation. The optical images of the isothermally aged samples are shown in
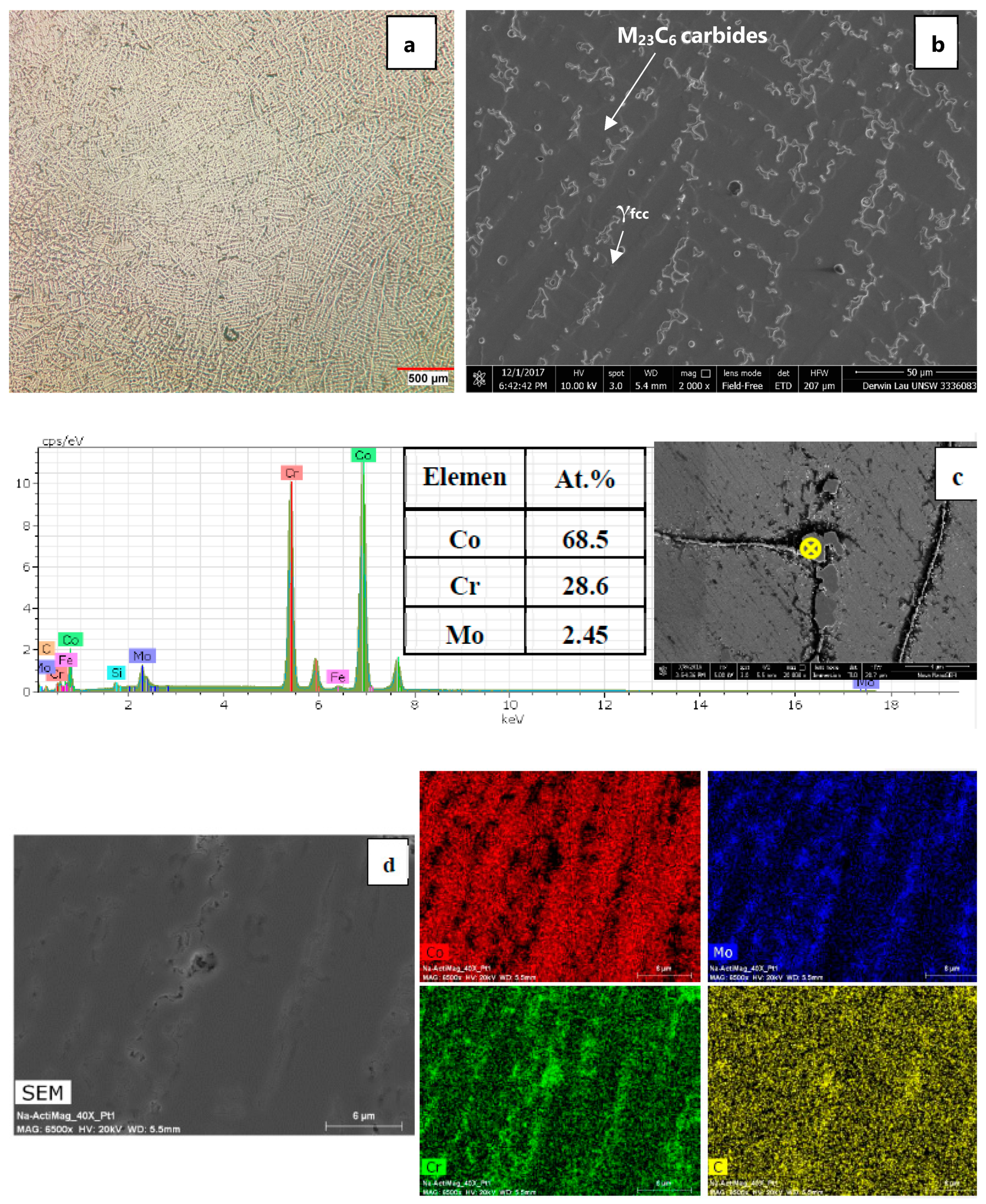
Figure 6a–d. No sign of perlite-type lamellae (hcp
2 phase) was observed in the matrix after isothermal aging and severe precipitation of M
23C
6 carbides was found to occur on the stacking faults. The kinetic of the isothermal fcc to hcp phase transformation is affected by the amount of lattice defects formed upon quenching. In other words, the increase of athermal martensite as a result of the cyclic solution treatment in the present work has slowed down the formation of perlite-type lamellae in the matrix. This could explain the absence of lamellae phase even after isothermal aging for 10 h at 850 °C. As shown earlier in XRD results, the cyclic solution treatment showed larger volume fraction of athermal martensite as compared to continuously solutionised samples. Massive and dense intergranular striations within the fcc grains are observed in the isothermally aged samples subjected to the cyclic solution treatment.
It is worth noting that the 5S sample shows higher amount of fcc (metastable) → hcp phase than 3S sample. It appears that the increase in the number of cyclic quenching contributed to the formation of heavily faulted regions and intersection of striations acted as potential nucleation sites for the precipitation of fine M
23C
6 carbides (see
Figure 6d). The M
23C
6 type carbides (which are fcc type with a lattice parameter similar to the fcc cobalt) are the typical carbides precipitating because of the minimal coherency strains. The intersection of these striations is shown in the bright field TEM image in
Figure 7a. It should be noted that these intersections are considered as dislocation dipoles whose high strain fields hinder the motion of slip dislocation [
23,
24]. The interaction between dislocations and these striations can influence the work hardening behavior of the Co-Cr-Mo alloy leading to the increase of hardness and strength.
Figure 7b shows the discontinuous precipitation of fine carbides on the intersection of striations (known as solute drag). This precipitation occurs as a result of preferential diffusion of elements to the faulted regions. Solute atoms such as carbon can migrate to the dislocations and stacking faults leading to the decrease of the stacking fault energy and widening the width of the stacking fault. This microsegregation which is known as Suzuki segregation can effectively cause solid-solution hardening [
25]. Having said that, the diffusion of carbon and chromium into the intersection of striations caused the precipitation of carbides and these carbides interact with the stacking faults locking the dissociated Shockley partial dislocations via solute segregation, as shown in
Figure 7b.
The XRD patterns of the isothermally aged samples at 850 °C for 10 h are shown in
Figure 8. In both 3C and 5C samples, similar to our previous work [
8], isothermal aging led to development of isothermal martensite. This can be seen by the increased intensity of
peak. In addition, after isothermal aging, the
peak in samples 3S and 5S shifted to the right which can be explained by the precipitation of carbides and decrease of lattice d-spacing. This peak shift was not noticeable after solution treatment in
Figure 5. The diffusion of Cr from the martensite and precipitation of Cr-rich carbides during isothermal aging decreased the lattice parameter of hcp phase shifting the diffraction peak towards higher angles. The slight peak broadening observed in the cyclically heat-treated samples is attributed to the accumulated residual stress introduced during cyclic quenching. As one can see, the martensitic phase transformation in continuous solutionised samples is not completed, since
and (111)
fcc peaks did not disappear thoroughly. Applying cyclic heat treatment in samples 3S and 5S resulted in the formation of almost fully martensitic structure as the intensity of
and (111)
fcc peaks decreased significantly in samples 3S and 5S after 10 h isothermal aging, as shown in
Figure 8. According to our previous work [
8], isothermal aging at 850 °C for 24 h in the solution-treated samples at 1230 °C for 3Continuous hours, did not result in complete martensite transformation (similar to the results obtained here). It seems that one quenching cycle only led to the formation of slight amount of hcp martensite. However, in this work, applying several cyclic quenching not only increased the volume fraction of athermal martensite but also contributed to the formation of heavily faulted regions providing nucleation sites for isothermal martensite and precipitation of fine carbides on the stacking faults. The effect of this process is alike the formation of stress-induced martensite (plastic straining of 10% and 20% via rolling) in our previous work [
9], in which complete martensitic phase transformation was observed just after isothermal aging at 850 °C for 8 h.
3.4. Grain Refinement by Reverse Transformation
Figure 9a–d show the near-surface microstructures of different solution-treated samples after aging at 850 °C for 10 h. In samples 3S and 5S, a new type of grain refinement was observed similar to what we reported before [
11,
12]. In our previous works [
11,
12], austenite grain refinement was achieved through a two-step heat treatment process in which pearlitic-type constitutes (M
23C
6 + hcp
2 martensite, lamellar structure) firstly developed during isothermal aging and then the interface of the lamellar phase was used as the initiation sites for the reverse austenite phase transformation at temperatures 1000–1240 °C. However, in the present work, it seems reasonable to suppose that the nucleation sites for reverse austinites and its concomitant grain refinement method was different than what we observed and reported in our previous studies.
In consecutive cyclic quenching, the interface of athermal ε martensite was harnessed to induce a new type of grain refinement and develop new refined austenite grains.
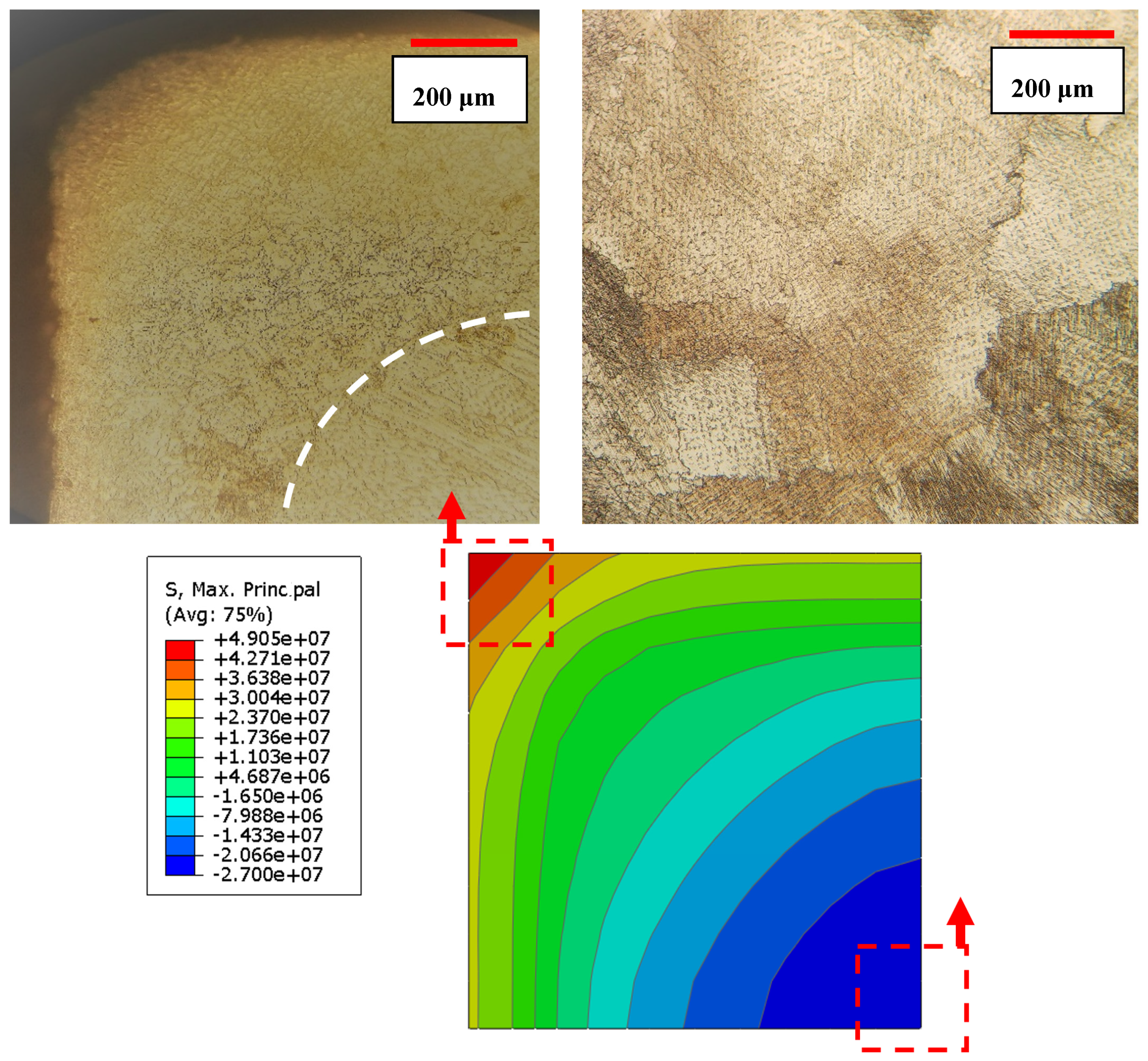
Figure 9b and d show 3S and 5S samples aged for 10 h. As one can see, the severity of grain refinement is significant beneath the surface and decreases by moving away from the free surface. The grain size measurement showed that the average grain size decreased from 283 ± 4 μm in as-cast sample to 16 ± 7 μm in 5S sample. The grain refinement is attributed to the generation of the residual thermal stress, mostly near the free surface during cyclic quenching. The increase of number of cyclic quenching resulted in more severe grain refinement and uniform distribution of carbides in the matrix. In addition, it was observed that the depth of the affected area increased by increasing the number of cyclic quenching.
The depth of the affected area in 5S sample was within the range of 400–500 μm, as shown in
Figure 10. Cyclic quenching and imposing thermal stresses particularly near the surface have been quite effective and refined the grain size near the surface which can potentially lead to enhanced surface properties such as hardness whereas the inner core material remains unaffected.
One may attribute the observed grain refinement to the recrystallization process. The origin of a recrystallized grain is always a pre-existing area which is highly oriented or heavily faulted in relation to the surrounding material. In metals with a low value of γ
SFE, the difficulty of cross slip and/or climbing of dislocations reduces the ability of the material to accommodate plastic deformation by slip alone (basic mechanisms responsible for recovery), and therefore deformation twinning may occur. The development of subgrains are not usually seen in metals with lower stacking fault energy such as stainless steel and Cobalt-base alloys, because recrystallization usually occurs prior to significant recovery. The other possibility to explain the grain refinement mechanism is the nucleation of the reversed austenite (γ
fcc) on the athermal ε martensite phase which is heavily faulted and can provide the potential nucleation sites for the new fcc grains and lead to the grain refinement near the surface layer, as shown in SEM micrographs in
Figure 10c–d. Severe precipitation of M
23C
6 carbides on the heavily faulted region (ε martensite) and considerable grain refinement of surface layer can lead to the enhanced surface properties such as hardness and wear resistance.
The finite element analysis (FEA) using an Abaqus software was utilized to estimate the amount of thermal stress induced during three cycles of quenching. The sample was initially heated to 1220 °C to make it homogenies and stress-free. The surface was then quenched in cold water until the sample reached the temperature of 5 °C. The current analysis was carried out in Abaqus/Standard through sequential thermal-stress solution procedure. In the sequential thermal-stress simulation, first, transient heat transfer analysis was performed, and then nodal temperatures were used as a loading of thermal-stress analysis. The sample is modeled with three-dimensional continuum elements which consists of 1000 first-order brick elements.
Figure 11 shows the effective stress developed after quenching from 1220 °C. It should be noted that for decreasing the computational cost due to the symmetry of the cubic sample, only 1/8 of sample was considered for the simulation. As one can see from
Figure 11, stress distribution after quenching is not uniform. The severity of introduced stress is maximum in the middle of the sample edge. It seems that the residual thermal stresses are accumulative during cyclic quenching. In other words, the thermal stresses build up in the high stress regions and this would provide nucleation sites and enough driving force for the athermal martensitic phase transformation.
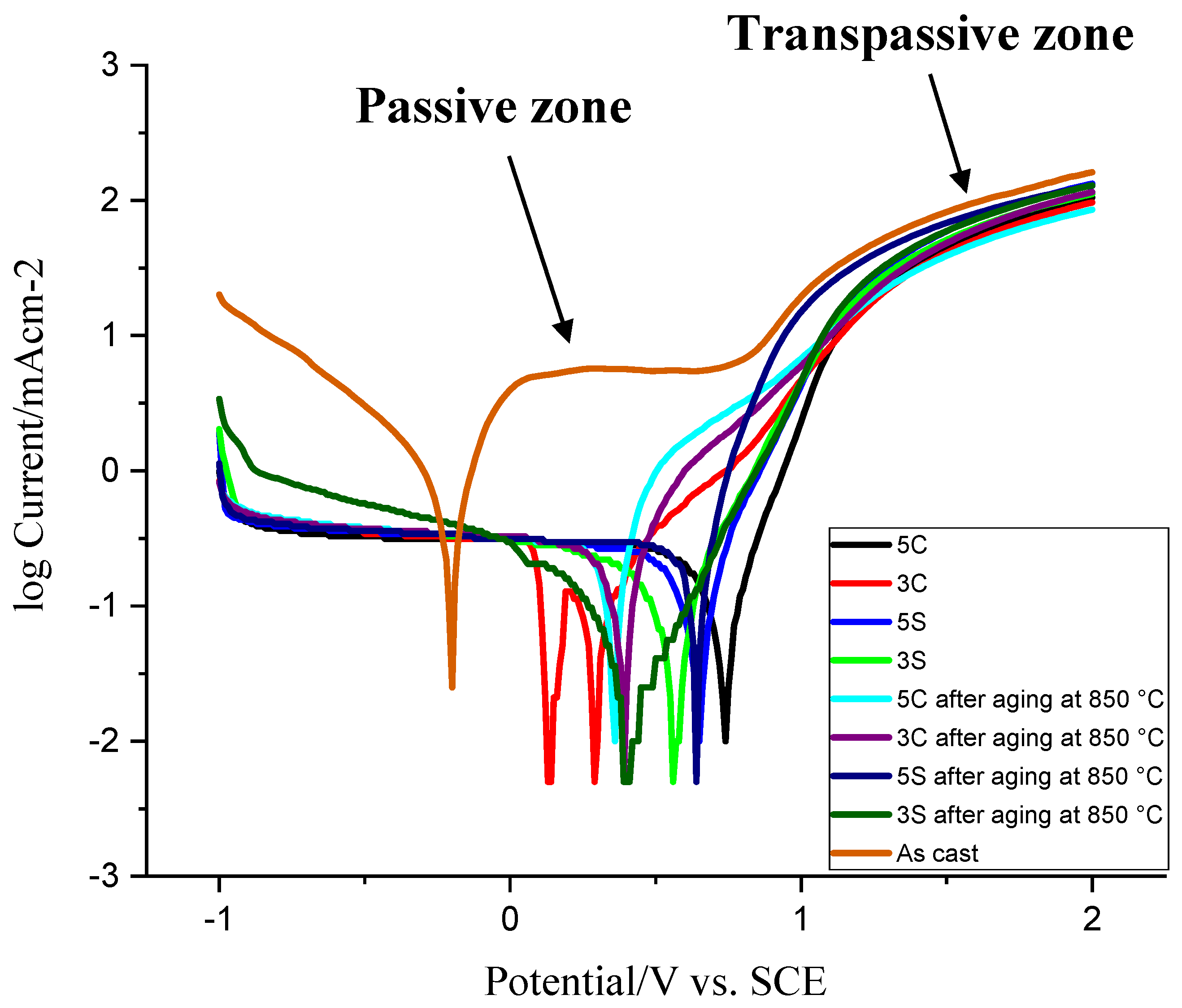
Figure 12 shows the Tafel plot curves of the samples subjected to different thermal treatment conditions in the 3.5 wt.%. NaCl aqueous solution.
Table 2 lists the potentiodynamic polarization data attained from
Figure 12 including corrosion current density, polarization resistance, Tafel constants, and corrosion rate. The open circuit potential (OCP, E
corro) of the as-cast sample is more negative than other thermally treated samples. As one can see, the corrosion resistance of all thermally treated samples is better than the as-cast sample. In other words, the as-cast sample shows higher anodic reactivity compared to other samples. This could be attributed to the coarse dendritic structure with interdendritic carbides precipitated during solidification (see
Figure 2). During anodic cycle of the Tafel plot, the passivation behavior was observed in the as-cast sample (from 0.1 to 0.8 V) whereas the current density of all other thermally treated samples was increased gradually before converging and following the same trend. According to the anodic part of the Tafel plot, the passivation behavior began earlier in isothermally treated samples 3C and 5C as compared to other isothermally treated samples (the slope of anodic curves was not as steep as other samples). Stable current density (from 0 to 0.8 V) in the as-cast sample shows the formation of dense protective oxide layer on the surface. At 0.8 V, the current density starts to increase sharply which is related to the passive-transpassive oxidation. The reaction of Cr
2O
3 with hydrogen reactivates the anodic dissolution of passive layer leading to oxide layer breakdown (Cr
2O
3 + 6H
+ → 2Cr
3+ + 3H
2O) [
26,
27].
It was shown that the protective oxide layer in Co-Cr-Mo alloy in simulated physiological solution (SPS) at E < 0.3 V consists mainly of Cr
2O
3 and the fraction of CoO increased with potential and, in the region from 0.5 to 0.7 V, the fractions of Cr and Co were found to be approximately the same [
26]. Generally, all thermally treated samples showed lower corrosion current density and higher polarization resistance as compared to the as-cast sample. This indicates the rate of electrochemical reaction is slow because of the dissolution and spheroidization of carbides, reduction of carbide size, less dendritic microstructure, and homogenization of the microstructure. Among all solutionised samples, sample 3S showed better corrosion behavior (≈1/10 of the as-cast sample). No features or spikes (indicative of the protective oxide layer breakdown) were observed during scan which indicates good corrosion resistance of the thermally treated samples. Isothermal aging at 850 °C for 10 h decreased the corrosion resistance of 5C and 5S samples by ≈ 96% and 13%, respectively. However, it improved the corrosion resistance of 3C and 3S samples by ≈ 6% and 38%, respectively. It should be noted that the corrosion resistance of all isothermally aged samples is still much better than the as-cast sample which contains coarse dendritic microstructure, microsegregation, and coarse M
23C
6 carbides in the matrix and on the grain boundaries. As discussed earlier, the amount of athermal ε martensite is favored by the cyclic solution heat treatment. A maximum volume fraction of 64% was previously reported in the water atomized Co-Cr-Mo powders [
26]. The amount of athermal ε martensite exceeded 80% in chill casting of Co-Cr alloy [
26]. Generally, the amount of athermal ε martensite is influenced by the annealing temperature and higher annealing temperature leads to greater dislocation and fault density [
26]. The development of athermal martensite is restricted by the strain fields at the γ
fcc/ε
hcp interface or local variations in chemical composition that stabilizes the fcc phase (carbon-enriched regions or Cr and Mo-depleted regions). In the present study, we showed that not only the annealing temperature but also the annealing time (3 h and 5 h) and type of heat treatment (continuous versus cyclic) could affect the volume fraction of athermal ε martensite and this would influence the corrosion properties of Co-Cr-Mo alloy. In fact, repetitive cyclic heat treatment creates more vacancies and stacking faults as compared to the continuous heat treatment providing more nucleation sites for athermal ε martensite and leading to larger amount of athermal ε martensite in the matrix.
The decrease of the corrosion resistance in 5C and 5S samples after isothermal aging is attributed to the considerable volume fraction of carbides and ε martensite. It has been accepted that the corrosion rate of Co-Cr-Mo alloy increased with the development of HCP phase in the microstructure. It seems that solution treatment for continuous 5 h or 5 repetitive cycles has led to considerable dissolution of primary M
23C
6 carbides in the matrix and formation of athermal ε martensite. This athermal ε martensite later provides heterogeneous nucleation sites for the precipitation of secondary carbides in the matrix. Carbides themselves are very good corrosion resistant. However, the depletion of Cr adjacent to carbides leads to localized corrosion (microgalvanic corrosion). This is followed by the formation of pits and crevices in the matrix which accelerate the corrosion and release metal ions in the human body. The size, density and distribution of these carbides could have significant influence on the wear and corrosion properties of Co-Cr-Mo alloy. Generally, Co-Cr-Mo alloy consisting of the γ
fcc is desirable for ductility and the presence of ε
hcp martensite makes this alloy resistant against wear and corrosion [
28]. Montero et al. [
29] showed that the kinetics of fcc → hcp phase transformation in low carbon Co-Cr-Mo alloy was considerably affected by the introduction of defects (twins and stacking faults) upon quenching after solution treatment. In other words, if fcc phase transforms into hcp phase during quenching, the kinetics of isothermal martensite transformation (hcp
2 phase forming lamellar structure) is significantly slowed down. Unlike Monterlo’s study, no lamellar phase was observed in the current work even after isothermal aging at 850 °C for 10 h. This could be due to the fact that cyclic heat treatment has resulted in the formation of considerable volume fraction of athermal ε martensite and this has reduced the kinetics of isothermal martensite.
Upon aging, athermal ε martensite continues to grow and fine carbides begin to precipitate on the ε martensite. In Monterlo’s study [
29], the corrosion resistance of isothermally aged low carbon Co-Cr-Mo alloy was shown to decrease only in those samples subjected to short period of aging where there were significant volume fractions of lattice defects (hcp embryos). Upon the reduction of lattice defects as a result of phase transformation, corrosion resistance was no longer affected by the fcc to hcp phase transformation since the chemical composition did not change during phase transformation.
The improvement of corrosion resistance in the 3S sample in the present study could be explained based upon the volume fraction of lattice defects and stacking faults formed during cyclic quenching and consumption of these defects due to the precipitation of carbides during isothermal aging and absence of lamellar phase. However, in 5S sample, it seems that applying cyclic solution treatment for five times created more lattice defects and stacking faults compared to sample 3S, and isothermal aging at 850 °C for 10 h was not enough to decrease the density of these defects. This perhaps has led to the deterioration of the corrosion resistance in sample 5S. In addition, the grain refinement, and fine and uniform distribution of the secondary carbides due to the cyclic heat treatment have modified the microstructure of the surface and contributed to the superior corrosion resistance of the cyclic heat-treated samples as compared to the as-cast one. Therefore, cyclic solution treatment is proposed as an effective method to enhance the surface characteristics and biocompatibility of the Co-Cr-Mo implant alloy.