High Temperature Oxidation Behavior of Selective Laser Melting Manufactured IN 625
Abstract
1. Introduction
2. Materials and Methods
3. Results
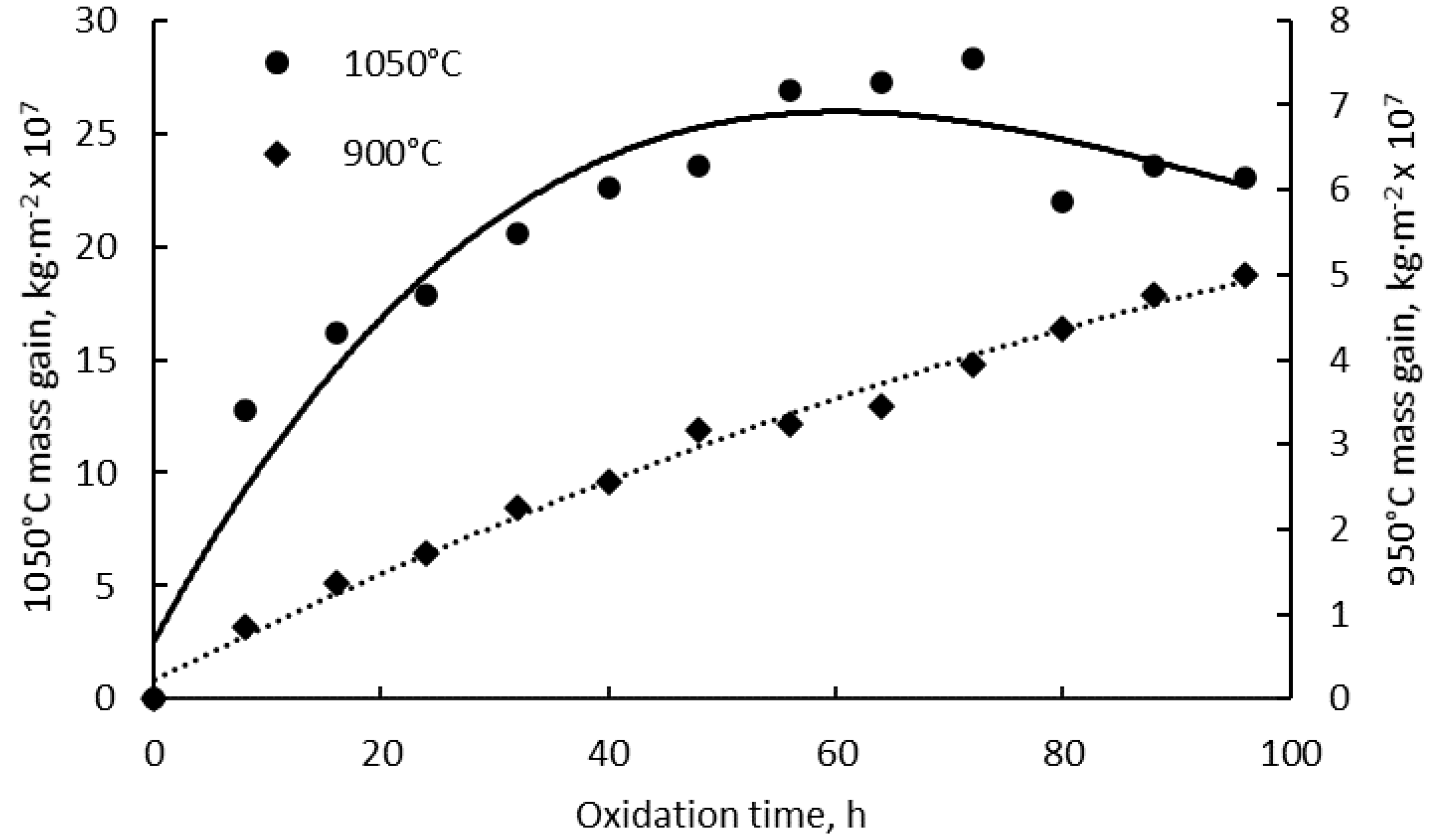
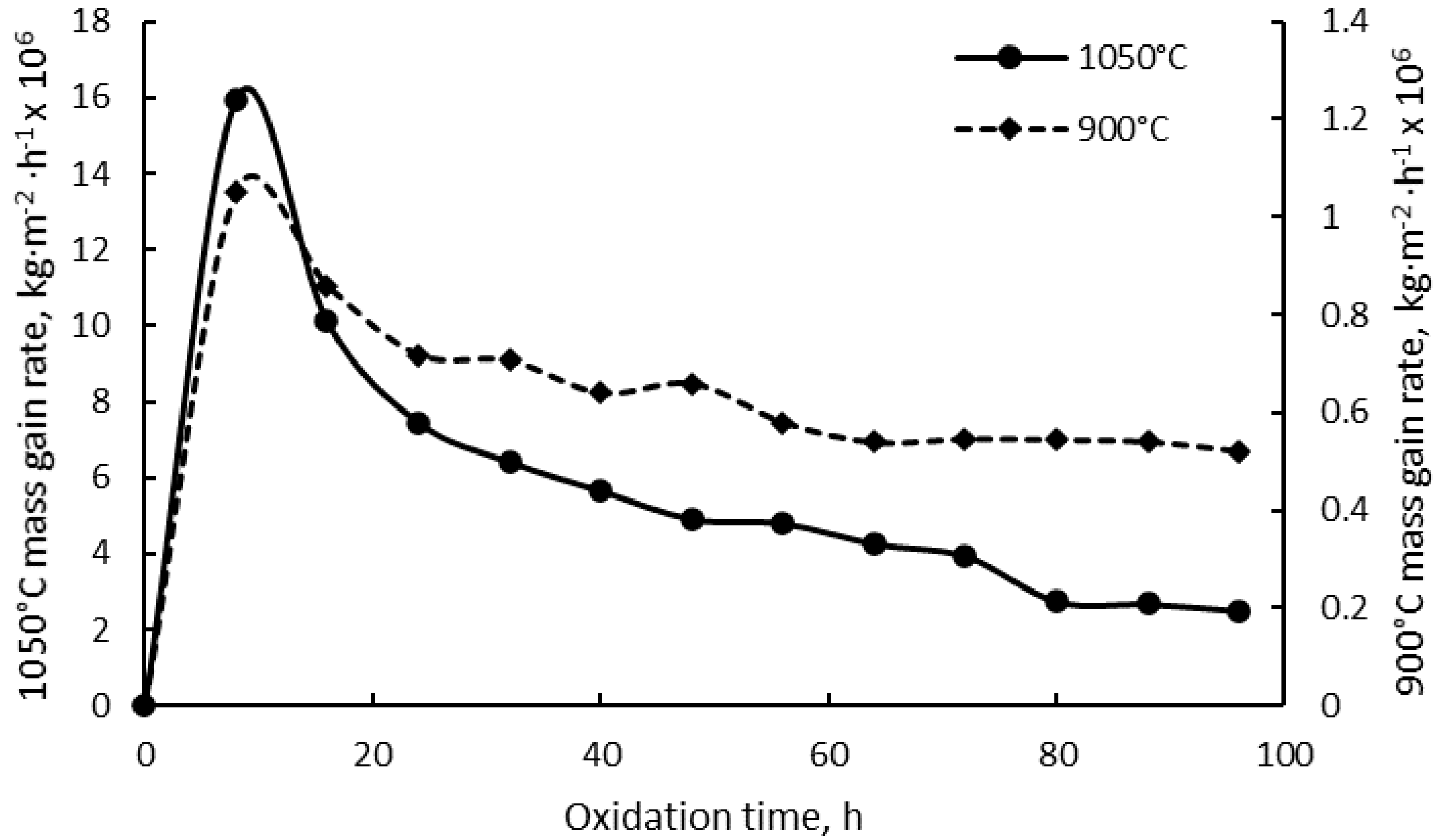
3.1. Mass Gain Analysis
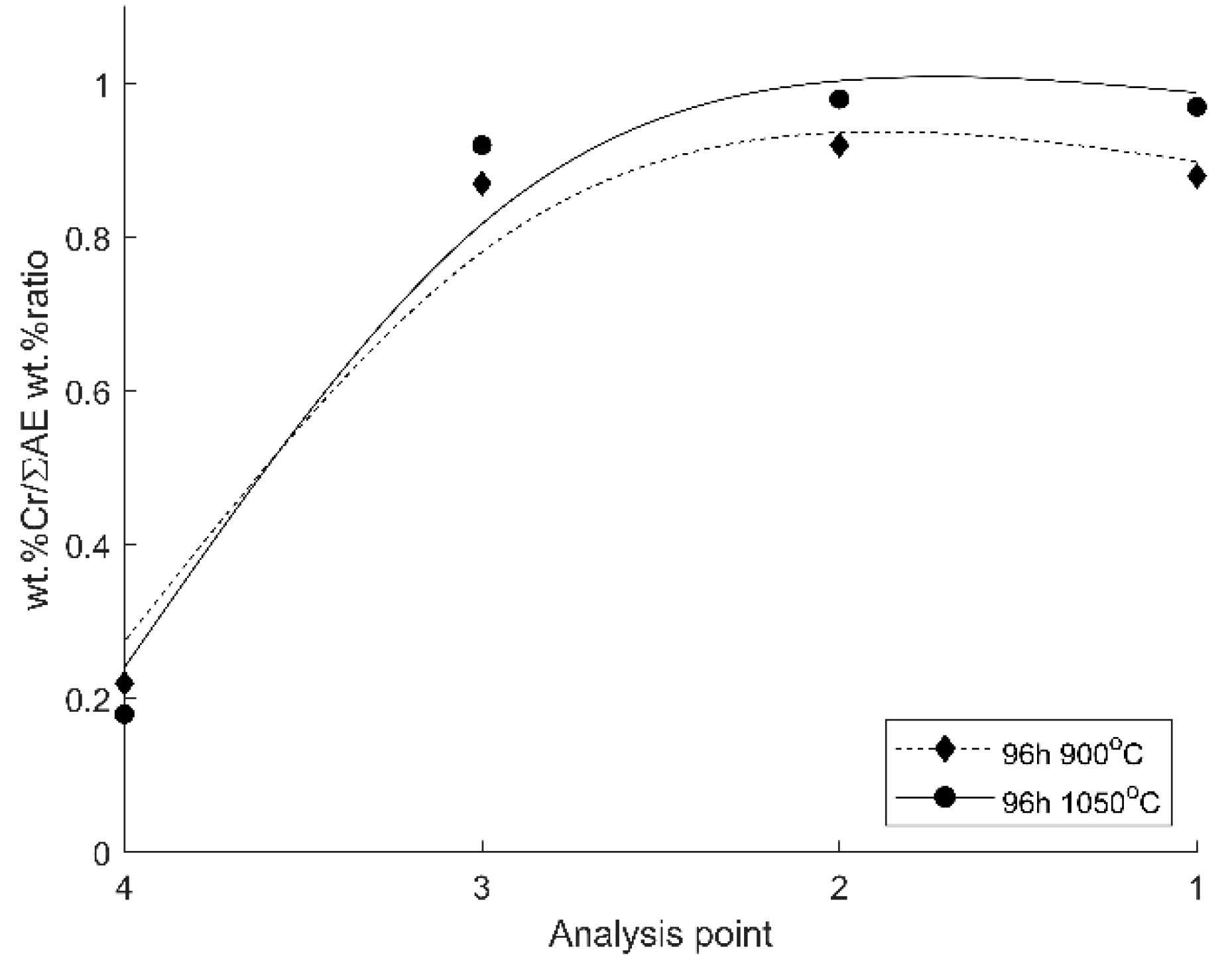
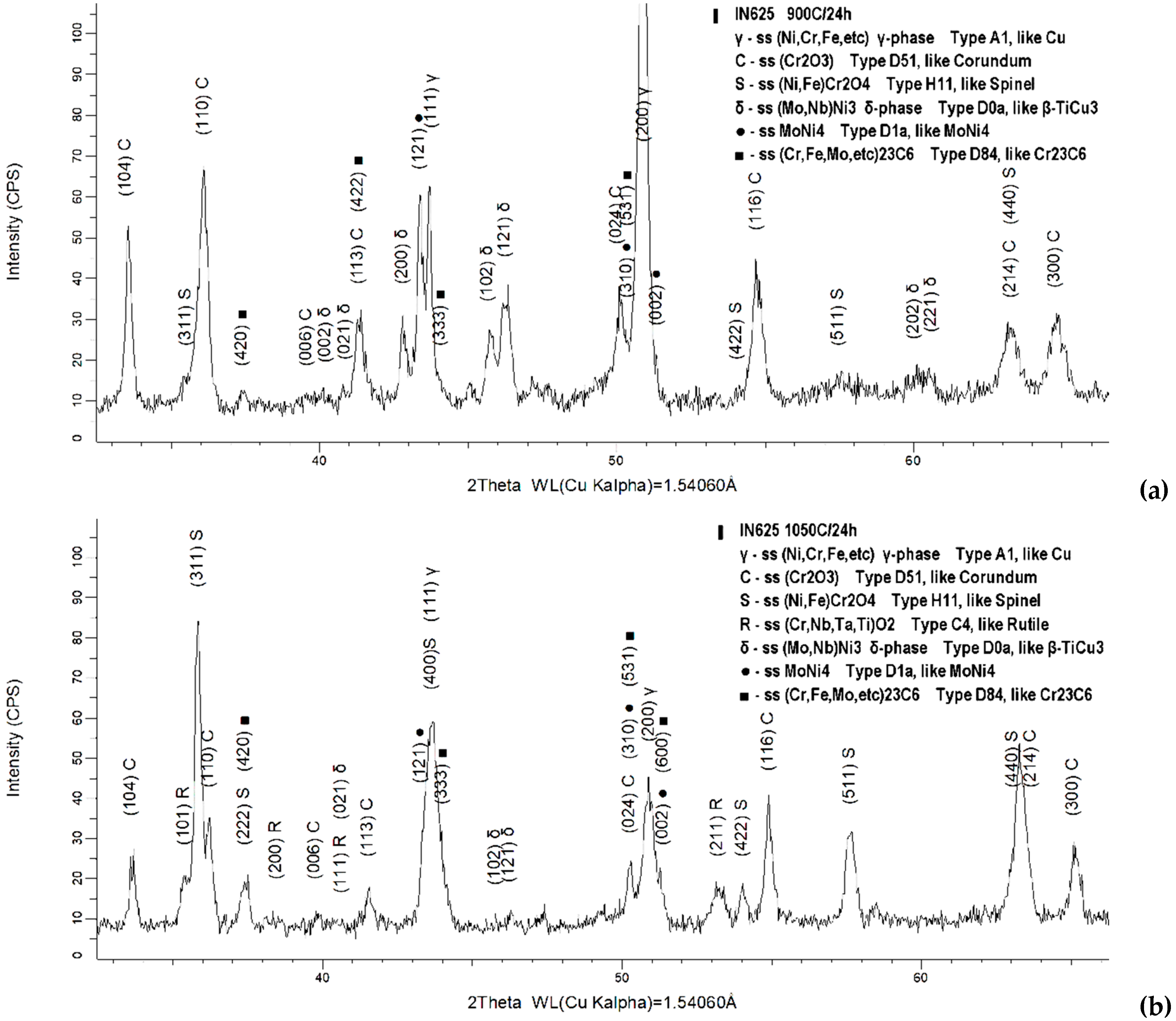
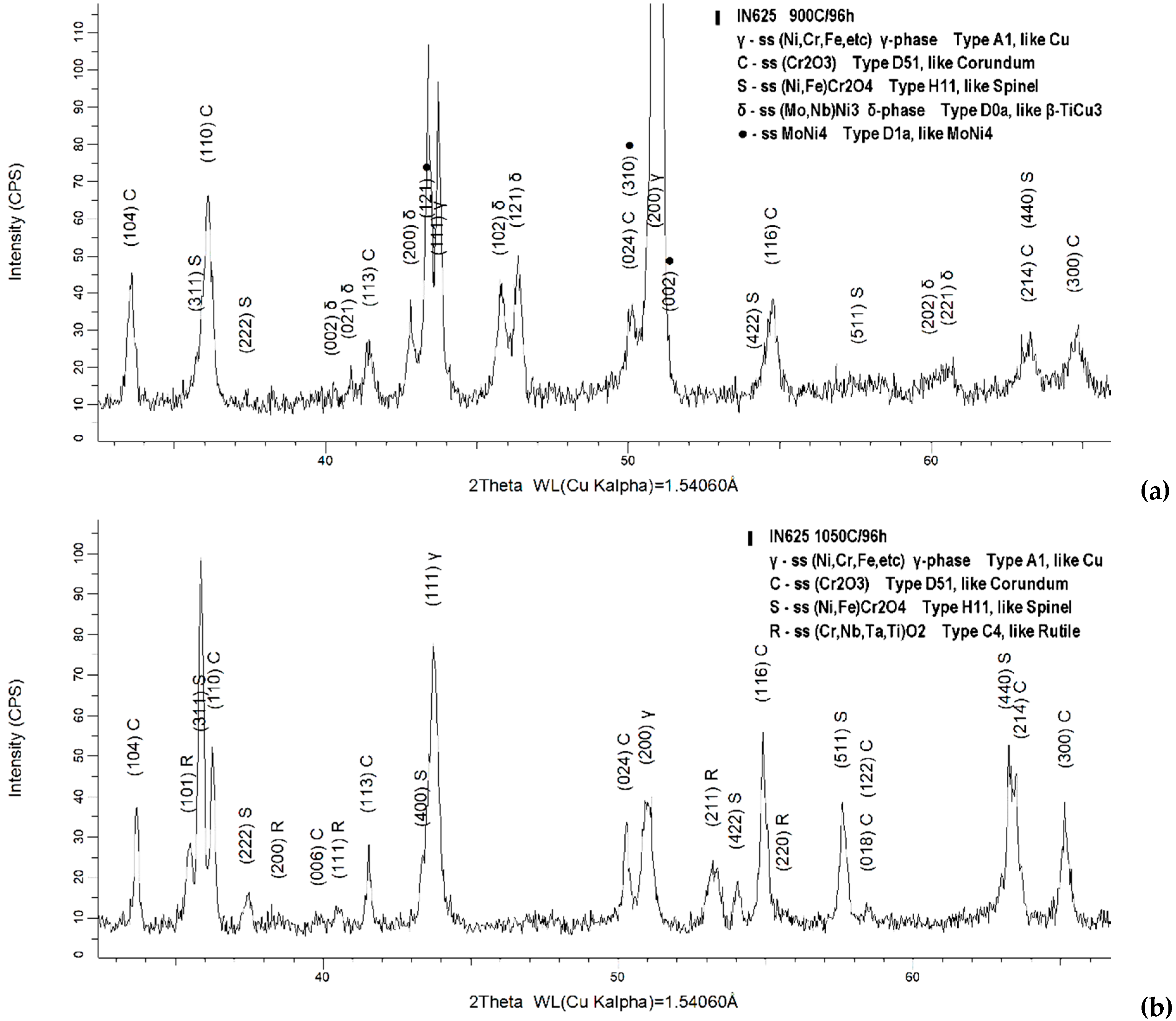
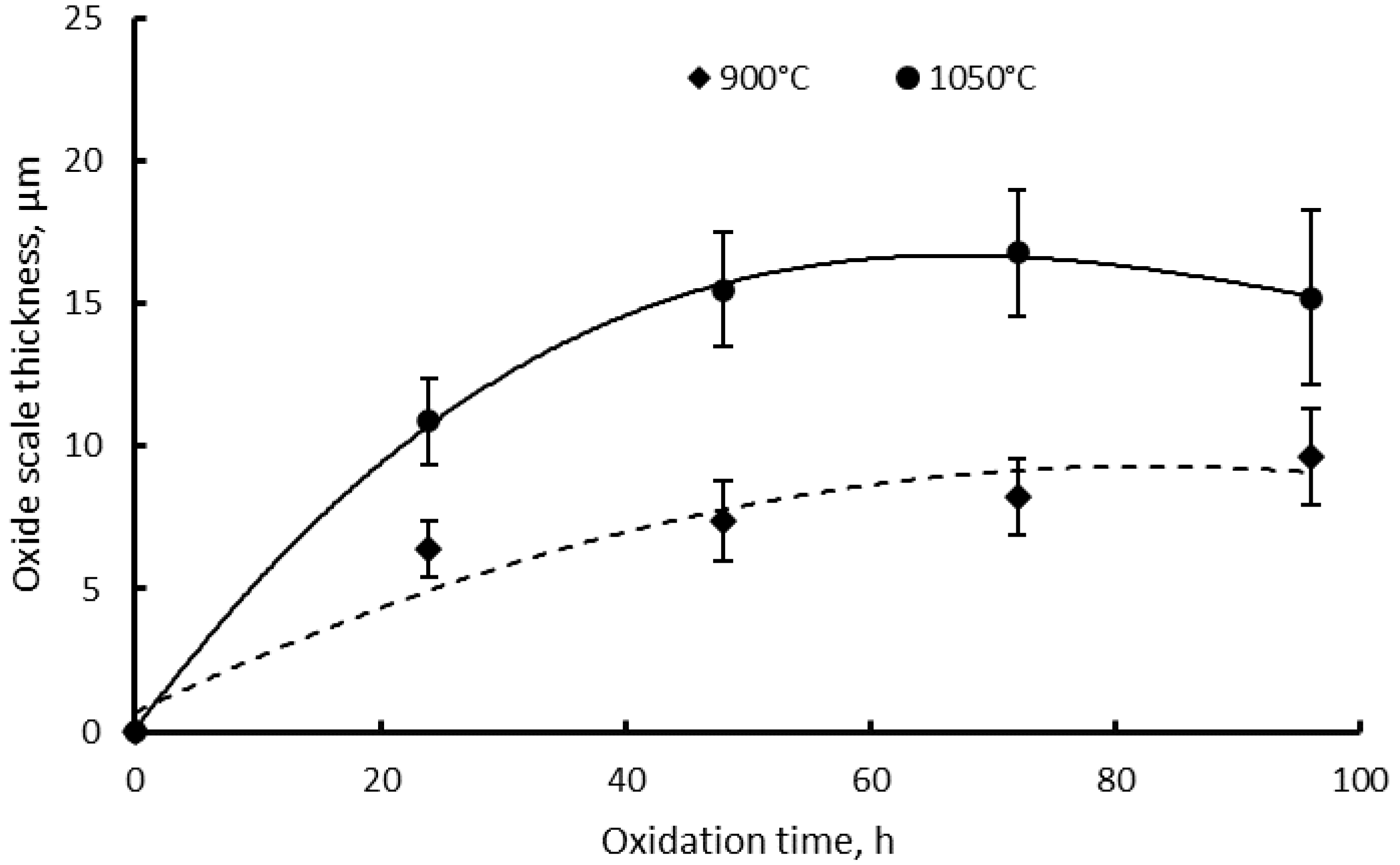
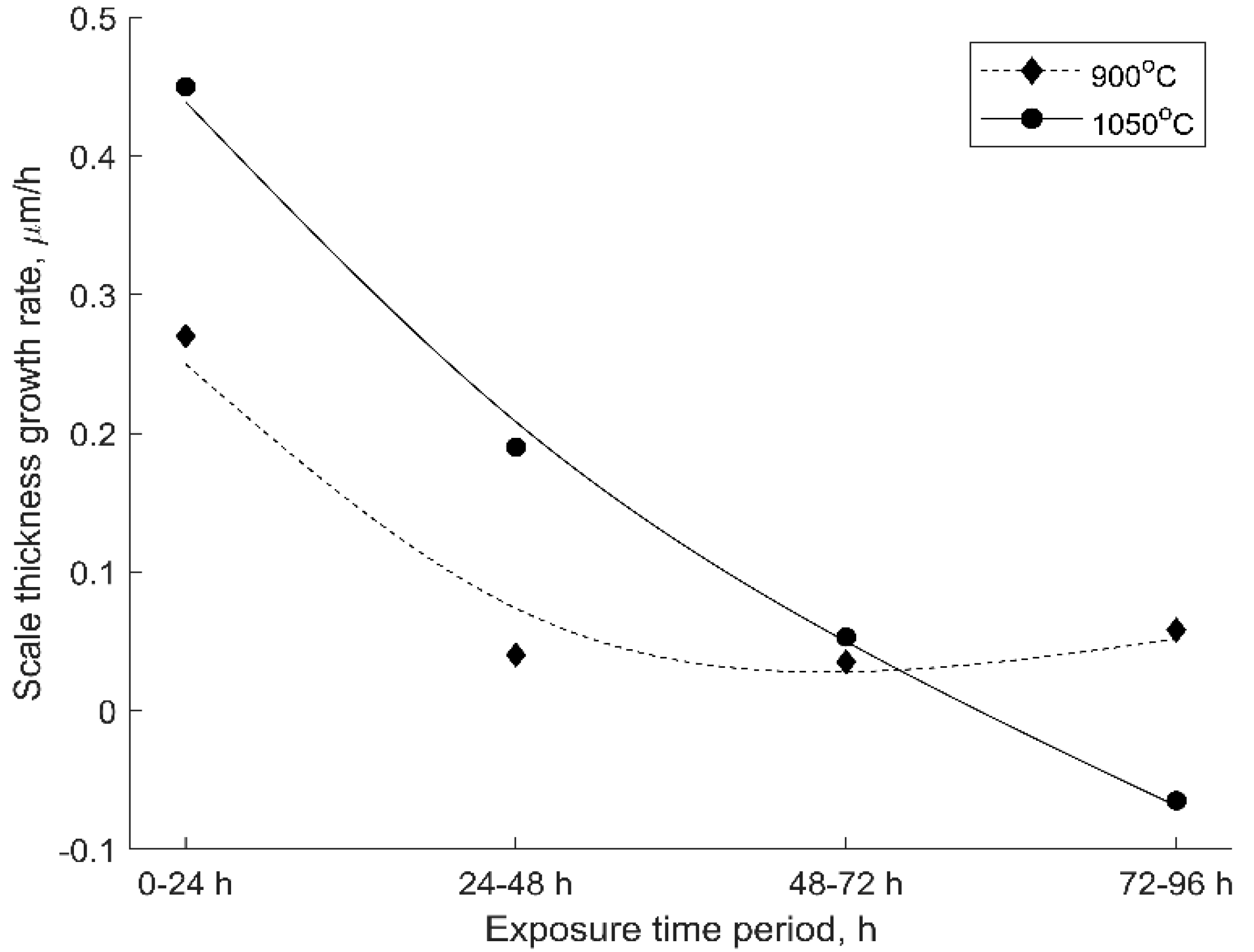
3.2. Oxide Scale Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Empirical Formula from PDF # | Assumed Approximate Formula | Symbol Phase Name | Structure Type |
|---|---|---|---|
| Cr0.08Fe0.65Ni0.27–PDF 04-020-3001 | (Ni, Cr, Fe, etc.) | γ γ-phase | Type A1, like Cu |
| Cr1.776Mo0.112O3–PDF 01-070-4368 | (Cr, Fe)2−2xMoxO3 | C ss Cr2O3 | Type D51, like Corundum |
| NiCr2O4–PDF 01-075-0198 | (Ni+2, Fe+2)(Cr+3, Fe+3)2O4 | S | Type H11, like Spinel |
| CrNbO4–PDF 01-081-0909 | ((Cr+3(Nb, Ta)+5)(1−x)/2(Ti+4)x)O−22 ((Cr+3(Nb, Ta)+5)(1−x)(Ti+4)2x)O−24 | R ss (Cr, Nb, Ta, Ti)O2 | Type C4, like Rutile |
| (Mo0.5Nb0.5)Ni3–PDF 04-003-9867 | (Mo, Nb)Ni3 | δ δ-phase | Type D0a, like β-TiCu3 |
| MoNi4–PDF 04-019-2633 | MoNi4 | ● | Type D1a, like MoNi4 |
| Cr23C6–PDF 04-007-8810 | (Cr, Fe, Mo, etc.)23C6 | ▰ complex carbid | Type D84, like Cr23C6 |
References
- Reed, R.C. The Superalloys: Fundamentals and Applications; Cambridge University Press: New York, NY, USA, 2006. [Google Scholar]
- Lund, C.H.; Wagner, H.J. Oxidation of Nickel-and Cobalt-Base Superalloys; DMIC Report 214; Battelle Memorial Institute: Columbus, OH, USA, 1965. [Google Scholar]
- Eiselstein, H.L.; Tillack, D.J. The Invention and Definition of Alloy 625. In Superalloys 718, 625 and Various Derivatives; TMS: Pittsburgh, PA, USA, 1991; pp. 1–14. [Google Scholar]
- DuPont, J.N.; Lippold, J.C.; Kiser, S.D. Welding Metallurgy and Weldability of Nickel-based Alloys; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2019. [Google Scholar]
- Donachie, M.J.; Donachie, S.J. Superalloys: A Technical Guide, 2nd ed.; ASM International: Cleveland, OH, USA, 2002. [Google Scholar]
- Park, S.J.; Seo, S.M.; Yoo, Y.S.; Jeong, H.W.; Jang, H.J. Statistical Study of the Effects of the Composition on the Oxidation Resistance of Ni-Based Superalloys. J. Nanomater. 2015, 1–11. [Google Scholar] [CrossRef]
- Stott, F.H. Principles of Growth and Adhesion of Oxide Scales. In The role of Active Elements in the Oxidation Behaviour High Temperature Metals and Alloys; Lang, E., Ed.; Elsevier Science Publishers Ltd.: London, UK, 1989; pp. 3–22. [Google Scholar]
- Birks, N.; Meier, G.H.; Pettit, F.S. Oxidation of alloys. In Introduction to the High Temperature Oxidation of Metals, 2nd ed.; Cambridge University Press: New York, NY, USA, 2006; pp. 101–162. [Google Scholar]
- Sato, A.; Moverare, J.J.; Hasselqvist, M.; Reed, R.C. On the Oxidation Resistance of Nickel-Based Superalloys. Adv. Mater. Res. 2011, 278, 174–179. [Google Scholar]
- Darolia, R. Development of strong, oxidation and corrosion resistant nickel-based superalloys: Critical review of challenges, progress and prospects. Int. Mat. Rev. 2019, 64, 355–380. [Google Scholar] [CrossRef]
- Brenneman, J.; Wei, J.; Sun, Z.; Liu, L.; Zou, G.; Zhou, Y. Oxidation behavior of GTD111 Ni-based superalloy at 900 °C in air. Corros. Sci. 2015, 100, 267–274. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, Z.; Wu, Z.; Hong, X.; Li, Z.; Xu, Y.; Li, J.; Ni, G.; Xiao, X. Oxide-Scale Evolution on a New Ni-Fe-Based Superalloy at High Temperature. Oxid. Met. 2019, 92, 49–65. [Google Scholar] [CrossRef]
- Chattopadhyay, B.; Wood, G.C. The transient oxidation of alloys. Oxid. Met. 1970, 2, 372–399. [Google Scholar] [CrossRef]
- Kitaguchi, H. Microstructure-Property Relationship in Advanced Ni-Based Superalloys. In Metallurgy–Advances in Materials and Processes; Pardhi, Y., Ed.; IntechOpen: Rijeka, Croatia, 2012; pp. 19–42. [Google Scholar]
- Lai, H. High Temperature Oxidation and Corrosion of Ni-Based Superalloy for industrial Gas Turbines. Ph.D. Thesis, Chalmers University of Technology, Gothenburg, Sweden, 2014. [Google Scholar]
- Pint, B.A.; Dryepondt, S.; Unocic, K.A. Oxidation of Superalloys in External Environments. In Proceedings of the 7th International Symposium on Superalloy 718 and Derivatives, Pittsburgh, PA, USA, 10–13 October 2010; Ott, E.A., Groh, J.R., Banik, A., Dempster, I., Gabb, T.P., Helmink, R., Liu, X., Michell, A., Sjoberg, G.P., Wusatowska-Sarnek, A., Eds.; TMS: Pittsburgh, PA, USA, 2010; pp. 861–875. [Google Scholar]
- Berthod, P. Kinetics of High Temperature Oxidation and Chromia Volatilization for a Binary Ni-Cr Alloy. Oxid. Met. 2005, 64, 235–252. [Google Scholar] [CrossRef]
- Huang, L.; Sun, X.F.; Guan, H.R.; Hu, Z.Q. Oxidation Behavior of a Single-Crystal Ni-base Superalloy in Air at 900, 1000 and 1100 °C. Oxid. Met. 2006, 65, 207–222. [Google Scholar] [CrossRef]
- Li, M.H.; Sun, X.F.; Jin, T.; Guan, H.R.; Hu, Z.Q. Oxidation Behavior of a Single-Crystal Ni-base Superalloy in Air–II: At 1000, 1100, and 1150 °C. Oxid. Met. 2003, 60, 195–210. [Google Scholar] [CrossRef]
- Li, M.H.; Sun, X.F.; Li, J.G.; Zhang, Z.Y.; Jin, T.; Guan, H.R.; Hu, Z.Q. Oxidation Behavior of a Single-Crystal Ni-Base Superalloy in Air I: At 800 and 900 °C. Oxid. Met. 2003, 59, 591–605. [Google Scholar] [CrossRef]
- Young, D.J. The Nature of High Temperature Oxidation. In High Temperature Oxidation and Corrosion of Metals; Burstein, T., Ed.; Elsevier: Cambridge, UK, 2008; Volume 1, pp. 1–27. [Google Scholar]
- Staszewska, K.; Scendo, M. Mechanism and Kinetics Oxidation of Inconel 617 and Inconel 625 Alloys. Tech. Issues 2016, 1, 82–89. [Google Scholar]
- Vesel, A.; Drenik, A.; Elersic, K.; Mozetic, M.; Kovac, J.; Gyergyek, T.; Stockel, J.; Varju, J.; Panek, R.; Balat-Pichelin, M. Oxidation of Inconel 625 superalloy upon treatment with oxygen or hydrogen plasma at high temperature. Appl. Surf. Sci. 2014, 305, 674–682. [Google Scholar] [CrossRef]
- Molins, R.; Andrieu, E. Analytical TEM study of the oxidation of nickel based superalloys. J. Phys. IV France 1993, 3, 469–475. [Google Scholar] [CrossRef][Green Version]
- Sand, T.; Geers, C.; Cao, Y.; Svensson, J.E.; Johansson, L.G. Effective Reduction of Chromium-oxy-hydroxide Evaporation from Ni-Base Alloy 690. Oxid. Met. 2019, 92, 259–279. [Google Scholar] [CrossRef]
- Santecchia, E.; Spigarelli, S.; Cabibbo, M. Materials Reuse in Laser Power Bed Fusion: Side Effects on the Laser-Metal Powder Interaction. Metals 2020, 10, 341. [Google Scholar] [CrossRef]
- Qin, H.; Xu, R.; Lan, P.; Wang, J.; Lu, W. Wear Performance of Metal Materials Fabricated by Powder Bed Fusion: A Literature Review. Metal 2020, 10, 304. [Google Scholar] [CrossRef]
- Nagahari, T.; Nagoya, T.; Kakehi, K.; Sato, N.; Nakano, S. Microstructure and Creep Properties of Ni-Base Superalloy IN718 Built up by Selective Laser Melting in a Vacuum Environment. Metals 2020, 10, 362. [Google Scholar] [CrossRef]
- Kang, Y.J.; Yang, S.; Kim, Y.K.; AlMangour, B.; Lee, K.A. Effect of post-treatment on the microstructure and high-temperature oxidation behaviour of additively manufactured Inconel 718 alloy. Corros. Sci. 2019, 158, 108082. [Google Scholar] [CrossRef]
- Buscail, H.; Rolland, R.; Issartel, C.; Rabaste, F.; Riffard, F.; Aranda, L.; Vilasi, M. Effects of water vapour on the oxidation of a nickel-base 625 alloy between 900 and 1100 °C. J. Mater. Sci. 2011, 46, 5903–5915. [Google Scholar] [CrossRef]
- Chyrkin, A.; Huczkowsiki, P.; Shemet, V.; Singheiser, L.; Quadakkers, W.J. Sub-Scale Depletion and Enrichment Process During High Temperature Oxidation of the Nickel Base Alloy 625 in the Temperature Range 900–1000 °C. Oxid. Met. 2011, 75, 143–166. [Google Scholar] [CrossRef]
- N’dah, E.; Hierro, M.P.; Borrero, K.; Perez, F.J. Study of the Cyclic Oxidation Resistance of Superalloy IN 625: Lifetime Predicted by COSP-Modelling Program. Oxid. Met. 2007, 68, 9–21. [Google Scholar] [CrossRef]
- Julliet, C.; Oudriss, A.; Balmain, J.; Feaugas, X.; Pedreza, F. Characterization and Oxidation resistance of additive manufactured and forged IN718 Ni-based superalloys. Corros. Sci. 2018, 142, 266–276. [Google Scholar] [CrossRef]
- Colas, J.; Charpentier, L.; Balat-Pichelin, M. Oxidation in Air at 1400 K and Optical Properties of Inconel 625, FeCrAlloy and Kanthal Super ER. Oxid. Met. 2020, 93, 355–370. [Google Scholar] [CrossRef]
- Whitney, E.; Simkovich, G.; Fink, J. High Temperature Oxidation of a Modified Alloy 625. In Superalloys 718, 625, 706 and Various Derivatives; Loria, E.A., Ed.; TMS: Pittsburg, PA, USA, 1997; pp. 695–704. [Google Scholar]
- Kumar, L.; Venkataramani, R.; Sundararaman, M.; Mukhopadhyay, P.; Garg, S.P. Studies on the Oxidation Behavior of Inconel 625 Between 873 and 1523 K. Oxid. Met. 1996, 45, 221–244. [Google Scholar] [CrossRef]
- Encinas-Orpesa, A.; Drew, G.L.; Hardy, M.C.; Leggett, A.J.; Nichollas, J.R.; Simms, N.J. Effects of Oxidation and Hot Corrosion in a Nickel Disc Alloy. In Superalloys 2018; Reed, R.C., Green, K.A., Caron, P., Gabb, T.P., Fahrmann, M.G., Huron, E.S., Woodard, S.A., Eds.; TMS: Pittsburg, PA, USA, 2008; pp. 609–618. [Google Scholar]
- Cruchley, S.; Evans, H.E.; Taylor, M.P.; Hardy, M.C.; Stekovic, S. Chromia layer growth on a Ni-based superalloy: Sub-parabolic kinetics and the role of titanium. J. Corr. Sci. 2013, 75, 58–66. [Google Scholar] [CrossRef]
- Gorman, D.M.; Higginson, R.L.; Du, H.; McColvin, G.; Fry, A.T.; Thomson, R.C. Microstructural Analysis of IN617 and IN 625 Oxidised in the Presence of Steam for use in Ultra-Supercritical Power Plant. Oxid. Met. 2013, 79, 553–566. [Google Scholar] [CrossRef]
- Mariano de Sousa Malafaia, A.; Brino de Oliveira, R.; Latu-Romain, L.; Wouters, Y.; Baldan, R. Isothermal oxidation of Inconel 625 superalloy at 800 and 1000 °C: Microstructure and oxide layer characterization. J. Mater. Char. 2020, 161, 110160. [Google Scholar] [CrossRef]
- Chyrkin, A.; Huczkowsiki, P.; Shemet, V.; Singheiser, L.; Quadakkers, W.J. Predicting Subsurface Enrichment/Depletion Processes During High-Temperature Oxidation of Alloy 625 Thin Foils. Australas. Corros. Assoc. 2012, 2, 1556. [Google Scholar]
- Garcia-Fresnillo, L.; Chyrkin, A.; Böhme, C.; Barnikel, J.; Schmitz, F.; Quadakkers, W.J. Oxidation behaviour and microstructural stability of alloy 625 during long-term exposure in steam. J. Mater. Sci. 2014, 49, 6127–6142. [Google Scholar] [CrossRef]





| Chemical Elem. | Al | C | Co | Cr | Fe | Mn | Mo | Nb | Si | Ti | Ni |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Specification [wt%] | <0.4 | <0.1 | <1.0 | 20–23 | 3–5 | <0.5 | 8–10 | 3.15–4.15 | <0.5 | <0.4 | Bal. |
| UK81572 [wt%] | 0.06 | 0.02 | 0.1 | 20.7 | 4.1 | 0.01 | 8.9 | 3.77 | 0.01 | 0.07 | 62.26 |
| Exposure Time [h] | EDS Microarea | O [wt%] | Nb [wt%] | Mo [wt%] | Cr [wt%] | Fe [wt%] | Ni [wt%] |
|---|---|---|---|---|---|---|---|
| 24 | 1 | 24.85 | 4.24 | 5.99 | 38.75 | 2.30 | 23.87 |
| 2 | 28.74 | 4.64 | 6.87 | 33.13 | 2.22 | 24.40 | |
| 3 | 28.51 | 5.68 | 8.38 | 27.11 | 2.16 | 28.17 | |
| 4 | 5.26 | 5.70 | 12.35 | 16.84 | 4.51 | 55.34 | |
| 48 | 1 | 27.86 | 2.25 | 2.68 | 53.75 | 2.06 | 11.41 |
| 2 | 22.81 | 2.95 | 4.13 | 44.19 | 1.86 | 24.06 | |
| 3 | 11.76 | 2.27 | 4.02 | 37.94 | 3.20 | 40.81 | |
| 4 | 9.04 | 3.04 | 7.83 | 28.55 | 4.47 | 47.07 | |
| 72 | 1 | 16.87 | 1.73 | 1.97 | 69.53 | 1.34 | 8.57 |
| 2 | 17.57 | 1.87 | 1.95 | 68.33 | 1.30 | 8.98 | |
| 3 | 19.54 | 1.81 | 2.24 | 63.12 | 1.63 | 11.67 | |
| 4 | 6.30 | 10.23 | 12.84 | 19.85 | 3.13 | 47.65 | |
| 96 | 1 | 25.91 | 2.30 | 2.91 | 57.33 | 2.33 | 9.22 |
| 2 | 39.50 | 1.78 | 1.96 | 53.96 | 1.06 | 1.74 | |
| 3 | 29.33 | 3.91 | 3.95 | 57.94 | 1.12 | 3.75 | |
| 4 | 3.98 | 11.57 | 16.93 | 8.88 | 3.29 | 55.33 |
| Exposure Time [h] | EDS Microarea | O [wt%] | Nb [wt%] | Mo [wt%] | Cr [wt%] | Fe [wt%] | Ni [wt%] |
|---|---|---|---|---|---|---|---|
| 24 | 1 | 30.31 | 2.72 | - | 65.77 | - | 1.19 |
| 2 | 25.56 | 1.88 | - | 71.07 | - | 1.49 | |
| 3 | 21.19 | 4.85 | - | 67.54 | - | 6.42 | |
| 4 | 3.59 | 6.27 | 14.33 | 13.91 | 5.05 | 56.85 | |
| 48 | 1 | 30.98 | 3.24 | - | 64.15 | - | 1.63 |
| 2 | 24.79 | 2.1 | - | 71.51 | - | 1.6 | |
| 3 | 22.38 | 4.49 | - | 62.85 | - | 10.28 | |
| 4 | 1.65 | 6.83 | 13.84 | 10.5 | 4.88 | 62.31 | |
| 72 | 1 | 29.07 | 1.92 | - | 67.02 | - | 1.99 |
| 2 | 24.11 | 1.77 | - | 72.48 | - | 1.64 | |
| 3 | 21.87 | 2.19 | - | 62.64 | - | 13.31 | |
| 4 | 2.61 | 6.74 | 14.07 | 9.44 | 5.19 | 61.95 | |
| 96 | 1 | 25.6 | 2.43 | - | 70.35 | - | 1.62 |
| 2 | 16.07 | 1.38 | - | 81.14 | - | 1.41 | |
| 3 | 18.78 | 5.74 | - | 68.87 | - | 6.61 | |
| 4 | 2.2 | 12.79 | 15.89 | 7.12 | 3.53 | 58.47 |
| kp Reported by Author | Corresponding kp in kg2·m−4·s−1 or Calculated from the Plot Reported by the Author | Material and Oxidation Conditions | Reference |
|---|---|---|---|
| 7.23 × 10−11 kg2·m−4·s−1 | - | AM IN 625, 900 °C/96 h | present work |
| 1.67 × 10−9 kg2·m−4·s−1 | - | AM IN 625, 1050 °C/96 h | |
| - | 2.8 × 10−9 kg2·m−4·s−1 | IN 625 (1 mm thick sheet) 1100 °C/36 h | J. Colas [34] |
| 2 × 10−3 mg2·cm−4·h−1 | 6 × 10−11 kg2·m−4·s−1 | IN625 (3 mm thick sheet) 900 °C/50 h | E N’dah [32] |
| 3 × 10−2 mg2·cm−4·h−1 | 7 × 10−10 kg2·m−4·s−1 | IN625 (3 mm thick sheet) 1000 °C/50 h | |
| - | 3 × 10−11 kg2·m−4·s−1 | IN625 (3 mm thick sheet) 900 °C/100 h | |
| - | 1 × 10−10 kg2·m−4·s−1 | IN625 (3 mm thick sheet) 1000 °C/100 h | |
| - | 3.23 × 10−9 kg2·m−4·s−1 | IN 625, cast, 1100 °C/60 h | Whitney et.al., [35] |
| 9.79 × 10−6 mg2·cm−4·s−1 | 9.79 × 10−10 kg2·m−4·s−1 | IN 625 (6 mm sheet) 1000 °C/96 h | Staszewska et. al., [22] |
| 9.90 × 10−6 mg2·cm−4·s−1 | 9.90 × 10−10 kg2·m−4·s−1 | IN 625 (6 mm sheet) 1100 °C/96 h | |
| - | 5.58–6.25 × 10−9 kg2·m−4·s−1 | IN 625 (1 mm sheet) 1054 °C/90 min. (depending on the PO2) | Kumar et.al., [36] |
| 3.2 × 10−13 g2·cm−4·s−1 | 3.2 × 10−11 kg2·m−4·s−1 | SY625 (1.6 mm thick sheet) 900 °C/50 h | Buscail et.al., [30] |
| 7.4 × 10−12 g2·cm−4·s−1 | 7.4 × 10−10 kg2·m−4·s−1 | SY625 (1.6 mm thick sheet) 1000 °C/50 h | |
| 5.25 × 10−11 g2·cm−4·s−1 | 5.25 × 10−9 kg2·m−4·s−1 | SY625 (1.6 mm thick sheet) 1100 °C/50 h | |
| 2.8 × 10−12 g2·cm−4·s−1 | 2.8 × 10−10 kg2·m−4·s−1 | Ni-30 wt% Cr alloy, cast (2 mm thick specimen) 1000 °C/50 h | Berthod et.al., [17] |
| 0.35 × 10−10 g2·cm−4·s−1 | 0.35 × 10−8 kg2·m−4·s−1 | Ni-Cr alloy (14.9 wt% Cr) 1096 °C/8 h | Lund et.al., [2] |
| 3.79 × 10−10 g2·cm−4·s−1 | 3.79 × 10−8 kg2·m−4·s−1 | Ni-Cr alloy (11.1 wt% Cr) 1096 °C/8 h | |
| 1.8–3.3 × 10−13 g2·cm−4·s−1 | 1.8–3.3 × 10−11 kg2·m−4·s−1 | AM IN 718, 800 °C/48 h | Juillet et.al., [33]. |
| 3.13 × 10−5 mg2·cm−4·s−1 | 3.13 × 10−9 kg2·m−4·s−1 | AM IN718 (2 mm thick specimens) Solution treatment + aging, 900 °C/24 h | Kang et.al., [29] |
| 1.81 × 10−5 mg2·cm−4·s−1 | 1.81 × 10−9 kg2·m−4·s−1 | AM IN718 (2 mm thick specimens) Solution treatment + aging 1000 °C/24 h | |
| 8.55 × 10−4 mg2·cm−4·s−1 | 8.55 × 10−8 kg2·m−4·s−1 | AM IN718 (2 mm thick specimens), HIPed + ageing, 900 °C/24 h | |
| 7.16 × 10−4 mg2·cm−4·s−1 | 7.16 × 10−8 kg2·m−4·s−1 | AM IN718 (2 mm thick specimens), HIPed + ageing 1000 °C/24 h |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Condruz, M.R.; Matache, G.; Paraschiv, A.; Badea, T.; Badilita, V. High Temperature Oxidation Behavior of Selective Laser Melting Manufactured IN 625. Metals 2020, 10, 668. https://doi.org/10.3390/met10050668
Condruz MR, Matache G, Paraschiv A, Badea T, Badilita V. High Temperature Oxidation Behavior of Selective Laser Melting Manufactured IN 625. Metals. 2020; 10(5):668. https://doi.org/10.3390/met10050668
Chicago/Turabian StyleCondruz, Mihaela Raluca, Gheorghe Matache, Alexandru Paraschiv, Teodor Badea, and Viorel Badilita. 2020. "High Temperature Oxidation Behavior of Selective Laser Melting Manufactured IN 625" Metals 10, no. 5: 668. https://doi.org/10.3390/met10050668
APA StyleCondruz, M. R., Matache, G., Paraschiv, A., Badea, T., & Badilita, V. (2020). High Temperature Oxidation Behavior of Selective Laser Melting Manufactured IN 625. Metals, 10(5), 668. https://doi.org/10.3390/met10050668





