Development of Nanobainitic Microstructures in Carbo-Austempered Cast Steels: Heat Treatment, Microstructure and Properties
Abstract
1. Introduction
2. Materials and Methods
3. Results
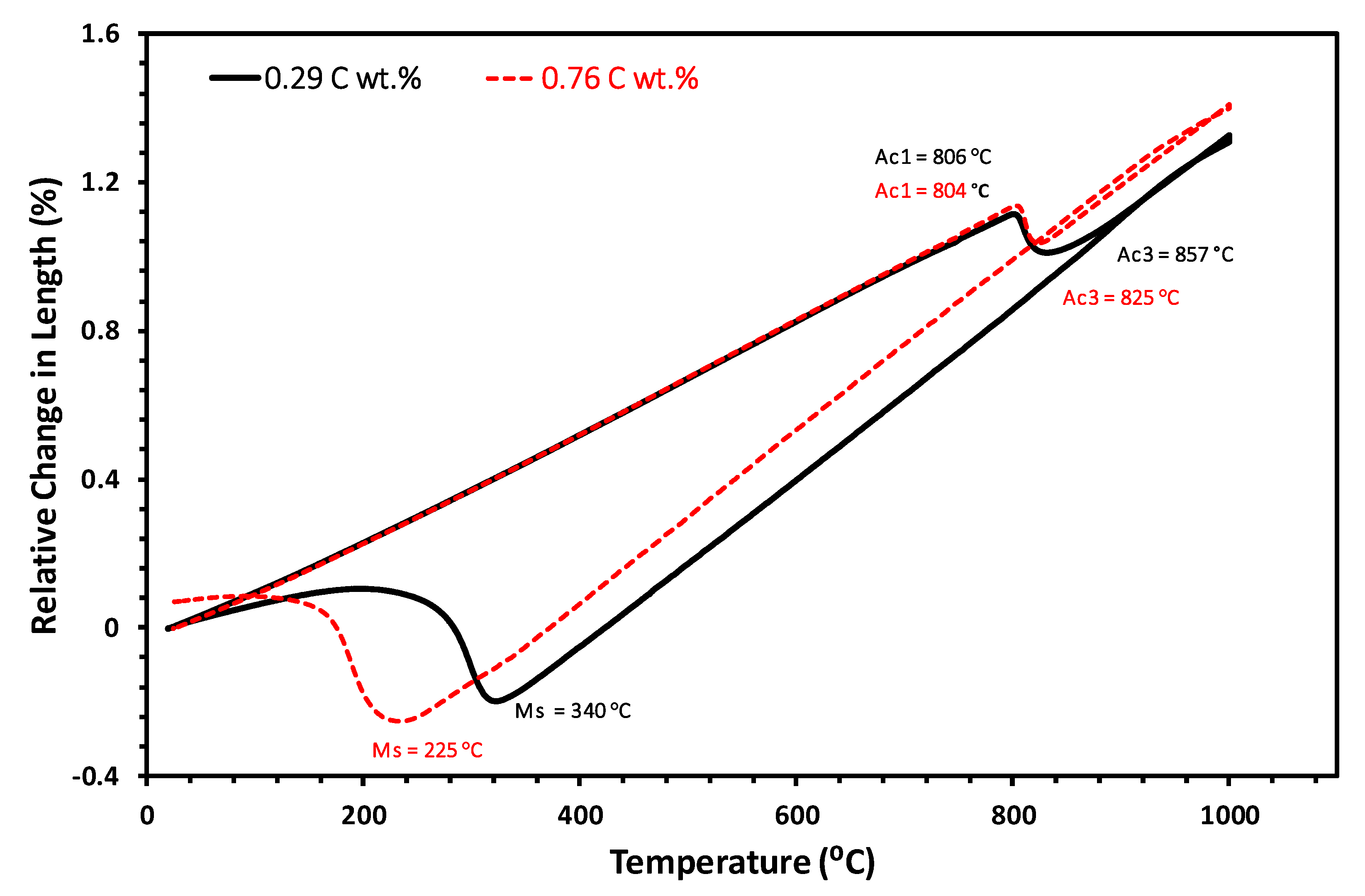
3.1. Determination of the Austenitization Conditions
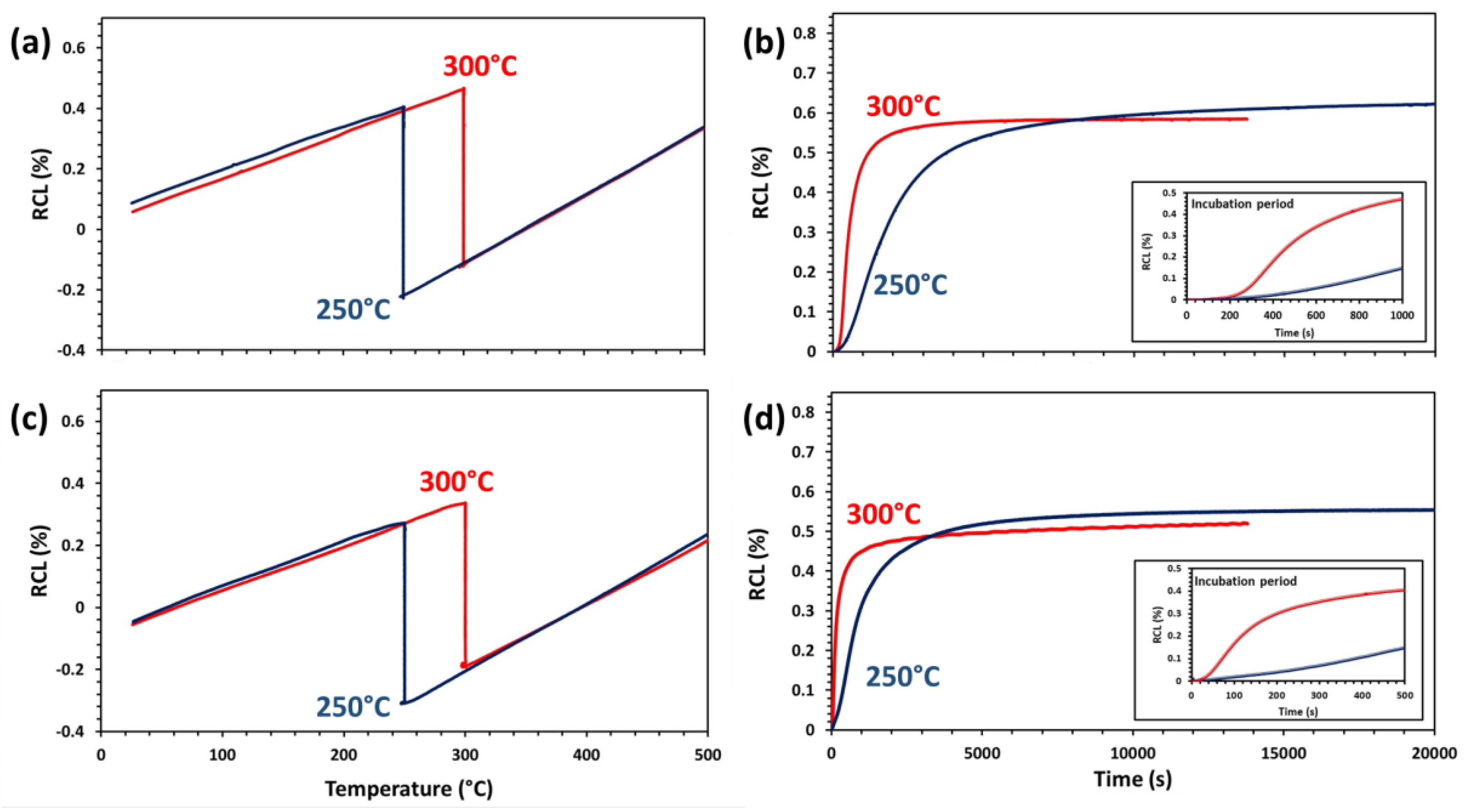
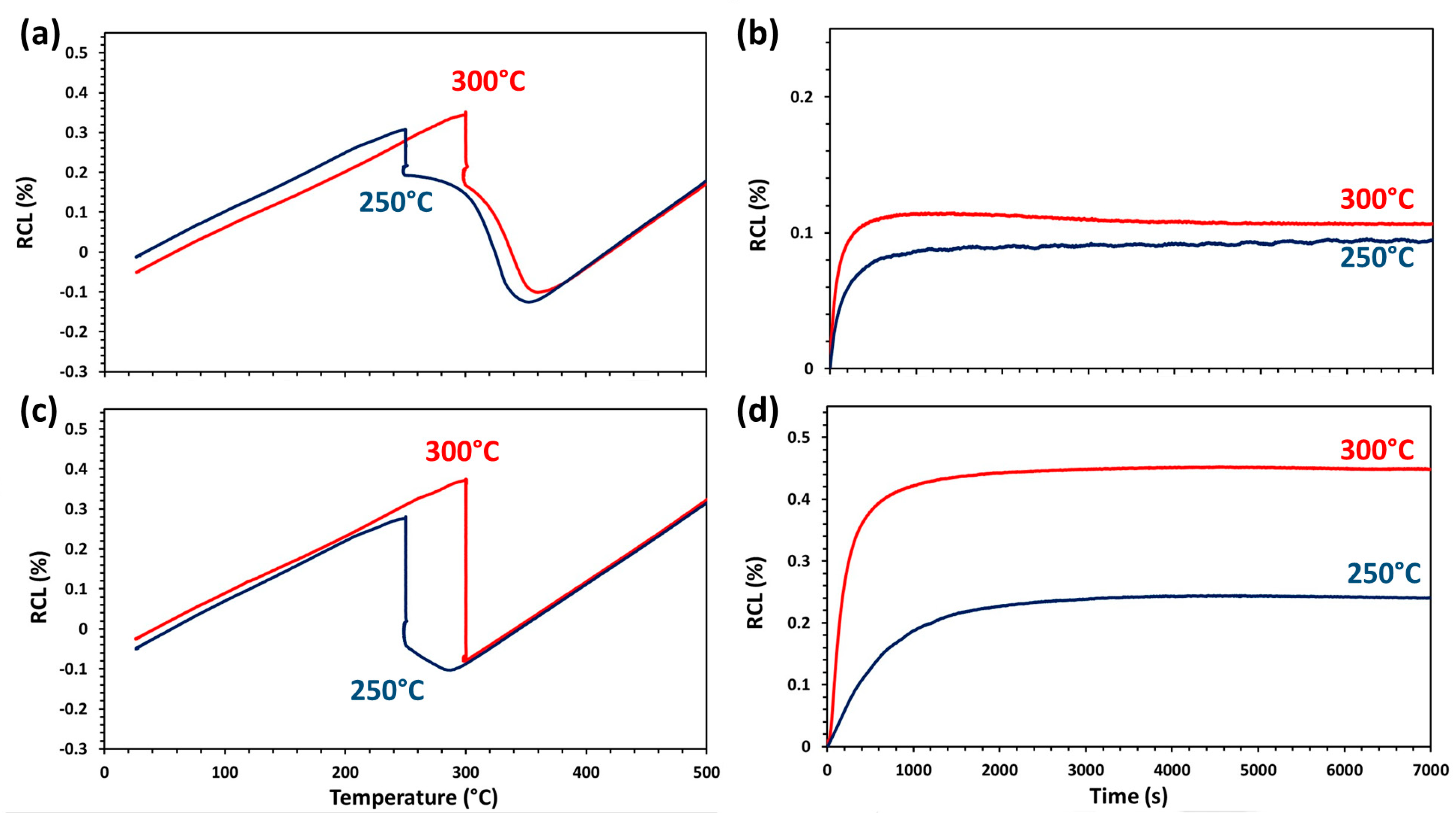
3.2. Characterization of Bainitic Transformation
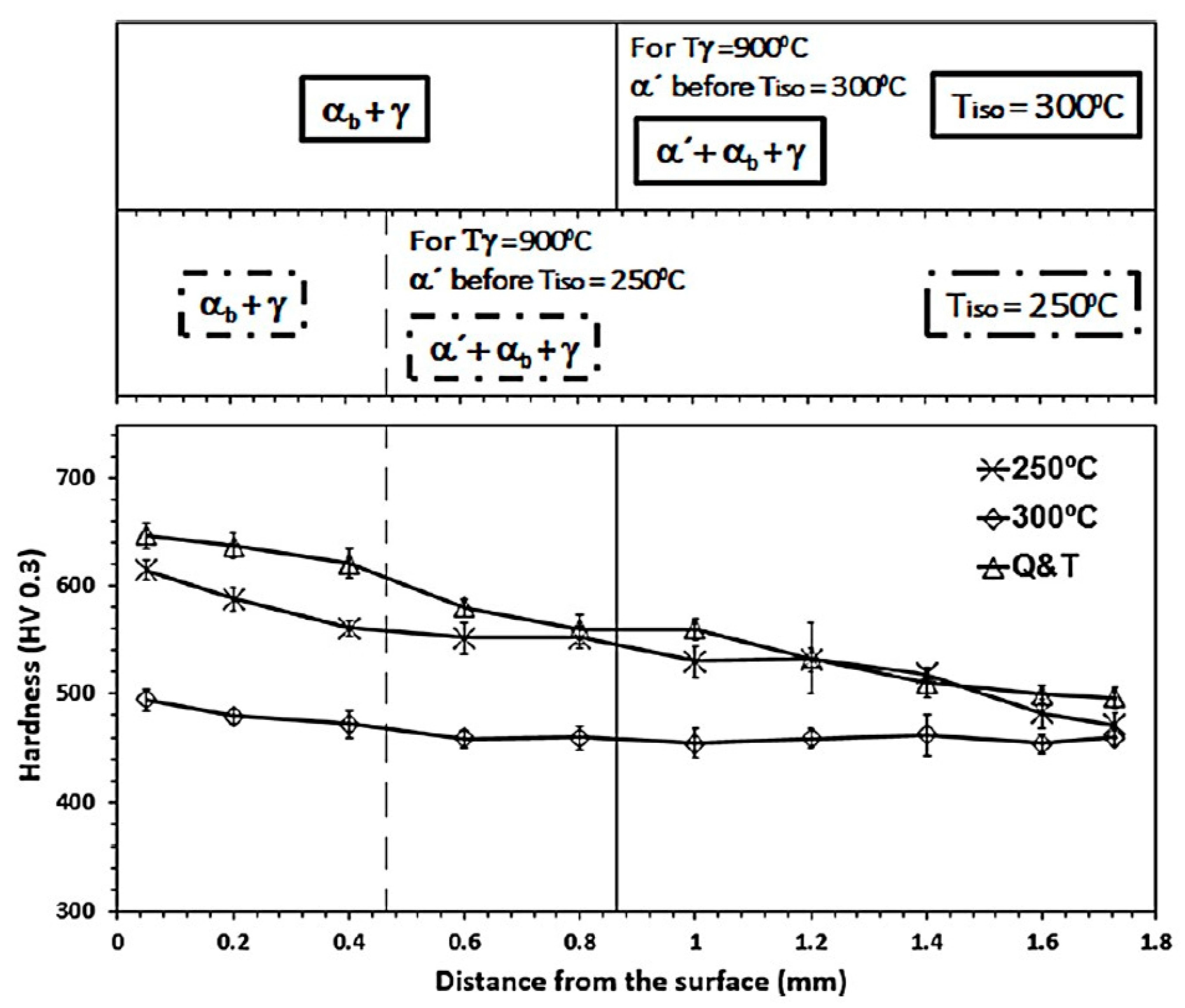
3.3. Mechanical Properties
4. Discussion
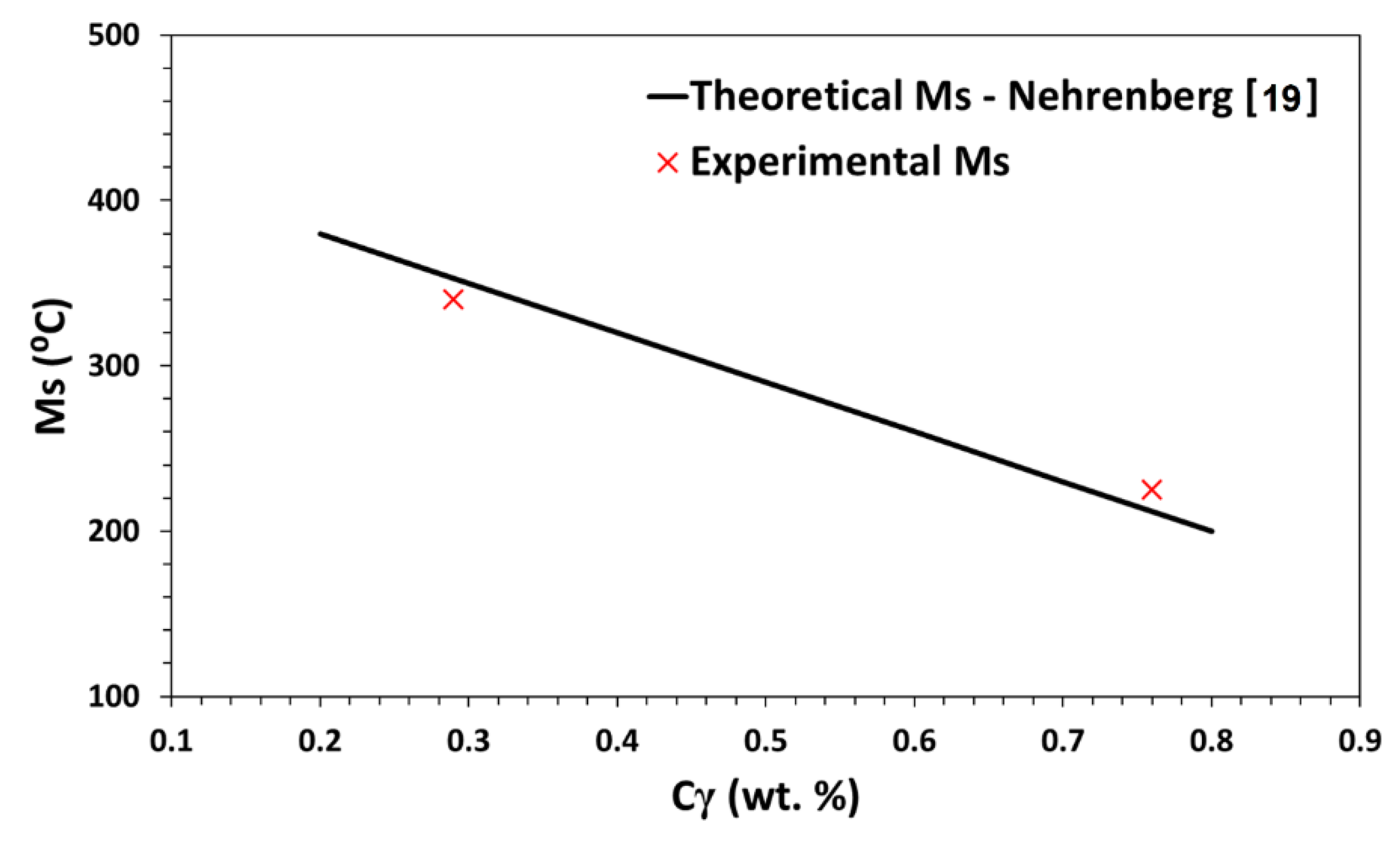
4.1. Bainitic Transformation
4.2. Mechanical Properties
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bhadeshia, H.K.D.H.; Edmonds, D.V. The mechanism of bainite formation in steels. Acta Metall. 1980, 28, 1265–1273. [Google Scholar] [CrossRef]
- Bhadeshia, H.K.D.H. Bainite in Steels. Transformations, Microstructure and Properties, 3rd ed.; Bhadeshia, H.K.D.H., Ed.; Institute of Materials, Minerals and Mining: London, UK, 2015. [Google Scholar]
- Garcia-Mateo, C.; Caballero, F.G. Understanding the mechanical properties of nanostructured bainite. In Handbook of Mechanical Nanostructuring; Aliofkhazraei, M., Ed.; Wiley: Weinheim, Germany, 2015; Volume 1, pp. 35–65. [Google Scholar]
- Caballero, F.G.; Morales-Rivas, L.; Garcia-Mateo, C. Retained austenite: Stability in a nanostructured bainitic steel. In Encyclopedia of Iron, Steel, and Their Alloys; Taylor & Francis: Abingdon, UK, 2016; pp. 3077–3087. [Google Scholar]
- Hayrynen, K.; Brandenberg, K.; Keough, J.R. Carbo-Austempering™—A New Wrinkle? SAE Technical Paper; SAE: Warrendale, PA, USA, 2002. [Google Scholar] [CrossRef]
- Damon, J.; Mühl, F.; Dietrich, S.; Schulze, V. A Comparative Study of kinetic models regarding bainitic transformation behavior in carburized case hardening steel 20MnCr5. Metall. Mater. Trans. A 2018, 50, 104–117. [Google Scholar] [CrossRef]
- Zhang, F.C.; Wang, T.S.; Zhang, P.; Zheng, C.L.; Lv, B.; Zhang, M.; Zheng, Y.Z. A novel method for the development of a low-temperature bainitic microstructure in the surface layer of low-carbon steel. Scr. Mater. 2008, 59, 294–296. [Google Scholar] [CrossRef]
- Zhang, F.; Yang, Z. Development of and perspective on high-performance nanostructured bainitic bearing steel. Engineering 2019, 5, 319–328. [Google Scholar] [CrossRef]
- Morales-Rivas, L.; Garcia-Mateo, C.; Sourmail, T.; Kuntz, M.; Rementeria, R.; Caballero, F. Ductility of nanostructured bainite. Metals 2016, 6, 302. [Google Scholar] [CrossRef]
- International, A.S.T.M. Standard Test Methods for Determining Average Grain Size; ASTM E112-13; ASTM International: West Conshohocken, PA, USA, 2013. [Google Scholar]
- International, A.S.T.M. Standard Test Method for Determining Volume Fraction by Systematic Manual Point Count; ASTM E562-19; ASTM International: West Conshohocken, PA, USA, 2019. [Google Scholar]
- Garcia-Mateo, C.; Jimenez, J.A.; Lopez-Ezquerra, B.; Rementeria, R.; Morales-Rivas, L.; Kuntz, M.; Caballero, F.G. Analyzing the scale of the bainitic ferrite plates by XRD, SEM and TEM. Mater. Charact. 2016, 122, 83–89. [Google Scholar] [CrossRef]
- Dyson, D.J.; Holmes, B. Effect of alloying additions on lattice parameter of austenite. J. Iron Steel Inst. 1970, 208, 469–474. [Google Scholar]
- International, A.S.T.M. Standard Test Methods for Tension Testing of Metallic Materials; ASTM E8/8M-16a; ASTM International: West Conshohocken, PA, USA, 2016. [Google Scholar]
- International, A.S.T.M. Standard Test Methods for Notched Bar Impact Testing of Metallic Materials; ASTM E23-16b; ASTM International: West Conshohocken, PA, USA, 2016. [Google Scholar]
- Parrish, G. Carburizing: Microstructures and Properties; ASM International: Novelty, OH, USA, 1999; p. 222. [Google Scholar]
- de Andres, C.G.; Caballero, F.G.; Capdevila, C.; Alvarez, L.F. Application of dilatometric analysis to the study of solid-solid phase transformations in steels. Mater. Charact. 2002, 48, 101–111. [Google Scholar] [CrossRef]
- Sourmail, T.; Smanio, V. Determination of Ms temperature: Methods, meaning and influence of ‘slow start’ phenomenon. Mater. Sci. Technol. 2013, 29, 883–888. [Google Scholar] [CrossRef]
- Nehrenberg, A.E. The temperature range of martensite formation. Trans. ASM 1946, 167, 494–498. [Google Scholar]
- Garcia-Mateo, C.; Caballero, F.; Bhadeshia, H.K.D.H. Development of hard bainite. ISIJ Int. 2003, 43, 1238–1243. [Google Scholar] [CrossRef]
- Caballero, F.G.; Garcia-Mateo, C.; Sourmail, T. Bainitic Steel: Nanostructured. In Encyclopedia of Iron, Steel, and Their Alloys; Taylor & Francis: Abingdon, UK, 2016; pp. 271–290. [Google Scholar]
- Garcia-Mateo, C.; Caballero, F.G.; Bhadeshia, H.K.D.H. Acceleration of Low-temperature Bainite. ISIJ Int. 2003, 43, 1821–1825. [Google Scholar] [CrossRef]
- Li, Z.; Li, P.; Luo, Y.; Zhou, X.; Qi, L.; Li, S.; Wang, Z. Effect of austenitizing temperature and prior martensite on ultra-fine bainite transformation kinetics. Metals 2019, 9, 1309. [Google Scholar] [CrossRef]
- Navarro-López, A.; Sietsma, J.; Santofimia, M.J. Effect of prior athermal martensite on the isothermal transformation kinetics below Ms in a low-C high-Si steel. Metall. Mater. Trans. A 2016, 47, 1028–1039. [Google Scholar] [CrossRef]
- Saha Podder, A. Tempering of a Mixture of Bainite and Retained Austenite. Ph.D. Thesis, University of Cambridge, Cambridge, UK, 2011. [Google Scholar]
- Kramer, P.C. An Investigation of Rolling-Sliding Contact Fatigue Damage of Carburized Gear Steels. Ph.D. Thesis, Colorado School of Mines, Golden, CO, USA, 2014. [Google Scholar]
- Garcia-Mateo, C.; Caballero, F.G. Ultra-high-strength bainitic steels. ISIJ Int. 2005, 45, 1736–1740. [Google Scholar] [CrossRef]
- Garcia-Mateo, C.; Caballero, F.G. Nanocrystalline bainitic steels for industrial applications. In Nanotechnology for Energy Sustainability; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2017; pp. 707–724. [Google Scholar] [CrossRef]
- Santajuana, M.A.; Eres-Castellanos, A.; Ruiz-Jimenez, V.; Allain, S.; Geandier, G.; Caballero, F.G.; Garcia-Mateo, C. Quantitative assessment of the time to end bainitic transformation. Metals 2019, 9, 925. [Google Scholar] [CrossRef]







| C | Si | Mn | Cr | Observations |
|---|---|---|---|---|
| 0.29 | 1.87 | 0.55 | 0.92 | core steel |
| 0.76 | 1.82 | 0.53 | 0.93 | case steel |
| C (wt.%) | T (°C) | ||
|---|---|---|---|
| Ac1 | Ac3 | Ms | |
| 0.29 | 806 | 857 | 340 |
| 0.76 | 804 | 825 | 225 |
| Tγ (°C) | tγ (min) | Vα (vol %) | Vγ (= 1 − Vα) (vol %) | Cγ (wt.%) | HV10 |
|---|---|---|---|---|---|
| 900 | 30 | 0 | 100 | 0.29 | 513 ± 9 |
| 15 | 0 | 100 | 0.29 | 525 ± 11 | |
| 850 | 15 | 20 ± 3 | 80 ± 3 | 0.36 | 485 ± 10 |
| 30 | 14 ± 2 | 86 ± 2 | 0.33 | 490 ± 8 | |
| 840 | 15 | 25 ± 2 | 75 ± 1 | 0.38 | 450 ± 9 |
| 30 | 20 ± 3 | 80 ± 2 | 0.36 | 466 ± 6 | |
| 830 | 15 | 32 ± 2 | 68 ± 2 | 0.43 | 410 ± 12 |
| 30 | 28 ± 3 | 72 ± 3 | 0.40 | 420 ± 10 |
| C (wt.%) | Tγ (°C) | tγ (min) | CRiso (°C/s) | Tiso (°C) | tiso (min) |
|---|---|---|---|---|---|
| 0.29 | 900 | 15 | 35 | 250 | 480 |
| 300 | 240 | ||||
| 830 | 250 | 480 | |||
| 300 | 240 | ||||
| 0.76 | 900 | 250 | 480 | ||
| 300 | 240 | ||||
| 830 | 250 | 480 | |||
| 300 | 240 |
| Alloy C (wt.%) | 0.29 | 0.76 | ||||||
|---|---|---|---|---|---|---|---|---|
| Isothermally treated samples | ||||||||
| Tγ(°C)/tγ(min) | 900/15 | 830/15 | 900/15 | 830/15 | ||||
| Tiso(°C)/tiso(min) | 250/480 | 300/240 | 250/480 | 300/240 | 250/480 | 300/240 | 250/480 | 300/240 |
| tαb (nm) | 168 ± 13 | 250 ± 13 | 135 ± 15 | 170 ± 16 | 58 ± 6 | 96 ± 18 | 54 ± 6 | 102 ± 18 |
| tγt (nm) | 146 ± 14 | 195 ± 15 | 90 ± 14 | 140 ± 10 | 44 ± 6 | 70 ± 10 | 45 ± 9 | 65 ± 11 |
| Vαb (Vol %) | 25.5 | 34.5 | 25.3 | 58.5 | 85.5 | 83.3 | 86.7 | 85.1 |
| Vα′ (Vol %) | 70 | 56 | 36 | - | - | - | - | - |
| Vα (Vol %) | - | - | 32.3 ± 3 | 33 ± 3 | - | - | - | - |
| Vγ (Vol %) | 4.5 ± 3 | 9.5 ± 3 | 6.7 ± 3 | 8.5 ± 3 | 14.5 ± 3 | 16.7 ± 3 | 13.3 ± 3 | 14.9 ± 3 |
| Cγ (wt.%) | 1.54 ± 0.12 | 1.67 ± 0.12 | 1.63 ± 0.12 | 0.79 ± 0.12 | 1.03 ± 0.12 | 1.24 ± 0.12 | 1.20 ± 0.12 | 1.33 ± 0.12 |
| HV10 | 490 ± 9 | 440 ± 7 | 380 ± 6 | 340 ± 8 | 579 ± 4 | 513 ± 5 | 576 ± 6 | 510 ± 10 |
| Ms (°C) | 356 | 374 | 291 | - | - | - | - | - |
| Tγ/tγ (°C/min) | Tiso/tiso (°C/min) | YS (MPa) | UTS (MPa) | TE (%) | V-Notch (J) |
|---|---|---|---|---|---|
| 900/15 | Q&T | 846 ± 13 | 911 ± 15 | 6.0 ± 0.5 | 4 ± 0.5 |
| 900/15 | 250/480 | 1220 ± 7 | 1310 ± 16 | 8.0 ± 0.4 | 6 ± 0.4 |
| 300/240 | 1025 ± 26 | 1110 ± 17 | 8.5 ± 0.4 | 8 ± 0.8 | |
| 830/15 | 250/480 | 1289 ± 5 | 1390 ± 26 | 9.0 ± 0.2 | 9 ± 0.3 |
| 300/480 | 1106 ± 11 | 1179 ± 25 | 10 ± 0.2 | 12 ± 0.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ríos-Diez, O.; Aristizábal-Sierra, R.; Serna-Giraldo, C.; Jimenez, J.A.; Garcia-Mateo, C. Development of Nanobainitic Microstructures in Carbo-Austempered Cast Steels: Heat Treatment, Microstructure and Properties. Metals 2020, 10, 635. https://doi.org/10.3390/met10050635
Ríos-Diez O, Aristizábal-Sierra R, Serna-Giraldo C, Jimenez JA, Garcia-Mateo C. Development of Nanobainitic Microstructures in Carbo-Austempered Cast Steels: Heat Treatment, Microstructure and Properties. Metals. 2020; 10(5):635. https://doi.org/10.3390/met10050635
Chicago/Turabian StyleRíos-Diez, Oscar, Ricardo Aristizábal-Sierra, Claudia Serna-Giraldo, Jose A. Jimenez, and Carlos Garcia-Mateo. 2020. "Development of Nanobainitic Microstructures in Carbo-Austempered Cast Steels: Heat Treatment, Microstructure and Properties" Metals 10, no. 5: 635. https://doi.org/10.3390/met10050635
APA StyleRíos-Diez, O., Aristizábal-Sierra, R., Serna-Giraldo, C., Jimenez, J. A., & Garcia-Mateo, C. (2020). Development of Nanobainitic Microstructures in Carbo-Austempered Cast Steels: Heat Treatment, Microstructure and Properties. Metals, 10(5), 635. https://doi.org/10.3390/met10050635






