Leaching of Oxide Copper Ores by Addition of Weak Acid from Copper Smelters
Abstract
1. Introduction
1.1. Weak Acid
1.2. Weak Acid Treatment
1.3. Water Scarcity
2. Materials and Methods
2.1. Weak Acid Characterization
2.2. Copper Ore Characterization
2.3. Curing and Leaching Tests
2.4. Toxicity Test
3. Results and Discussion
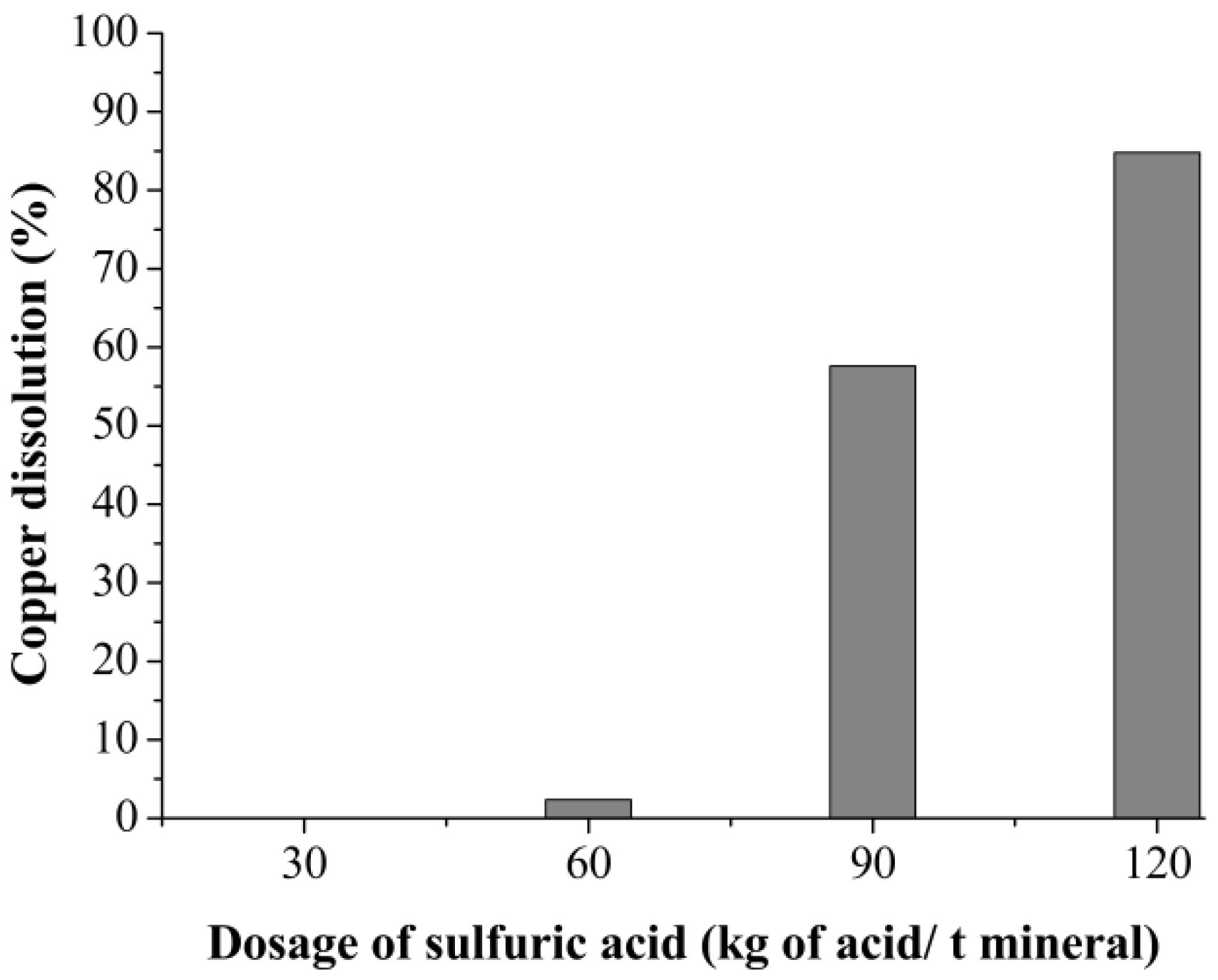
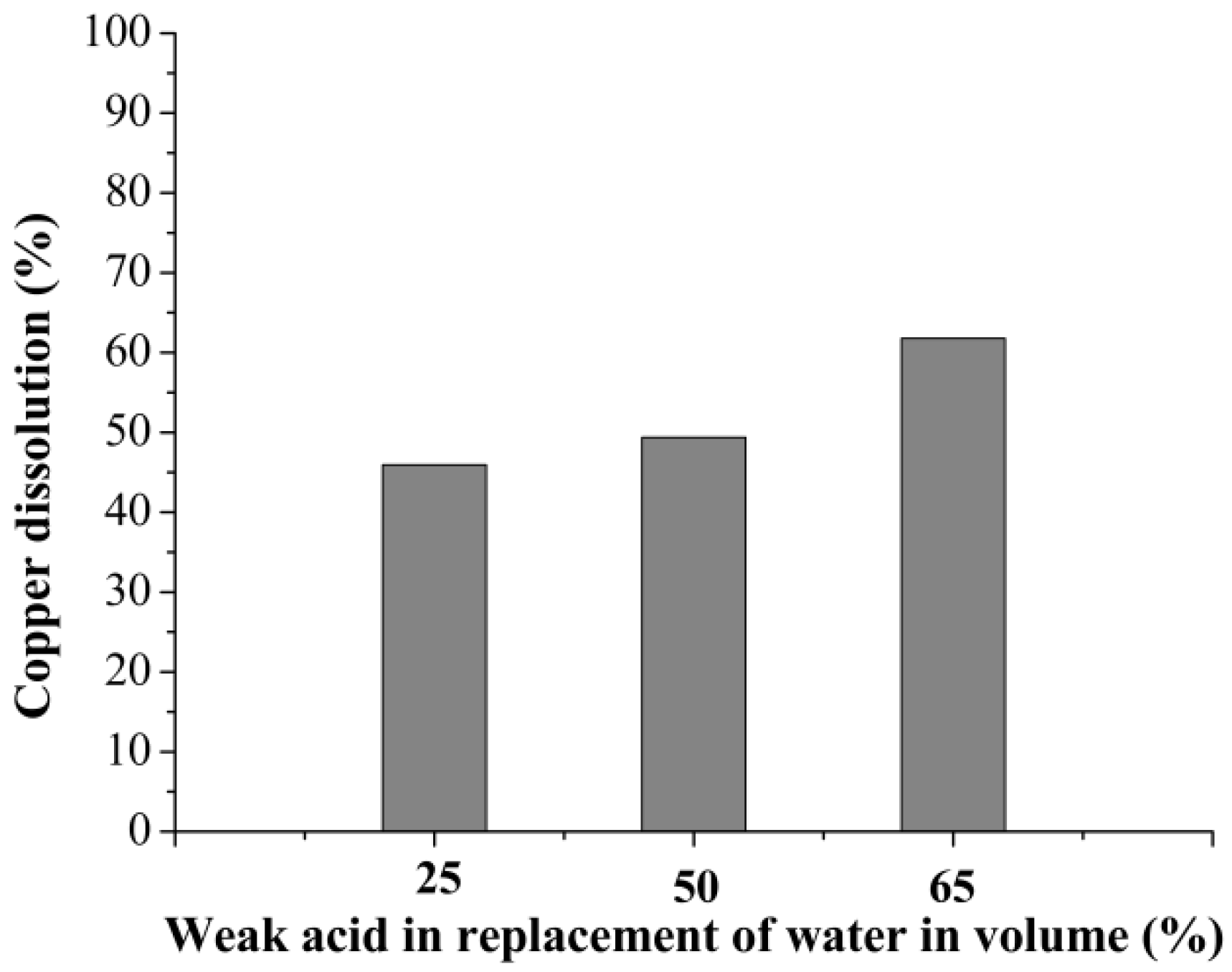
3.1. Curing
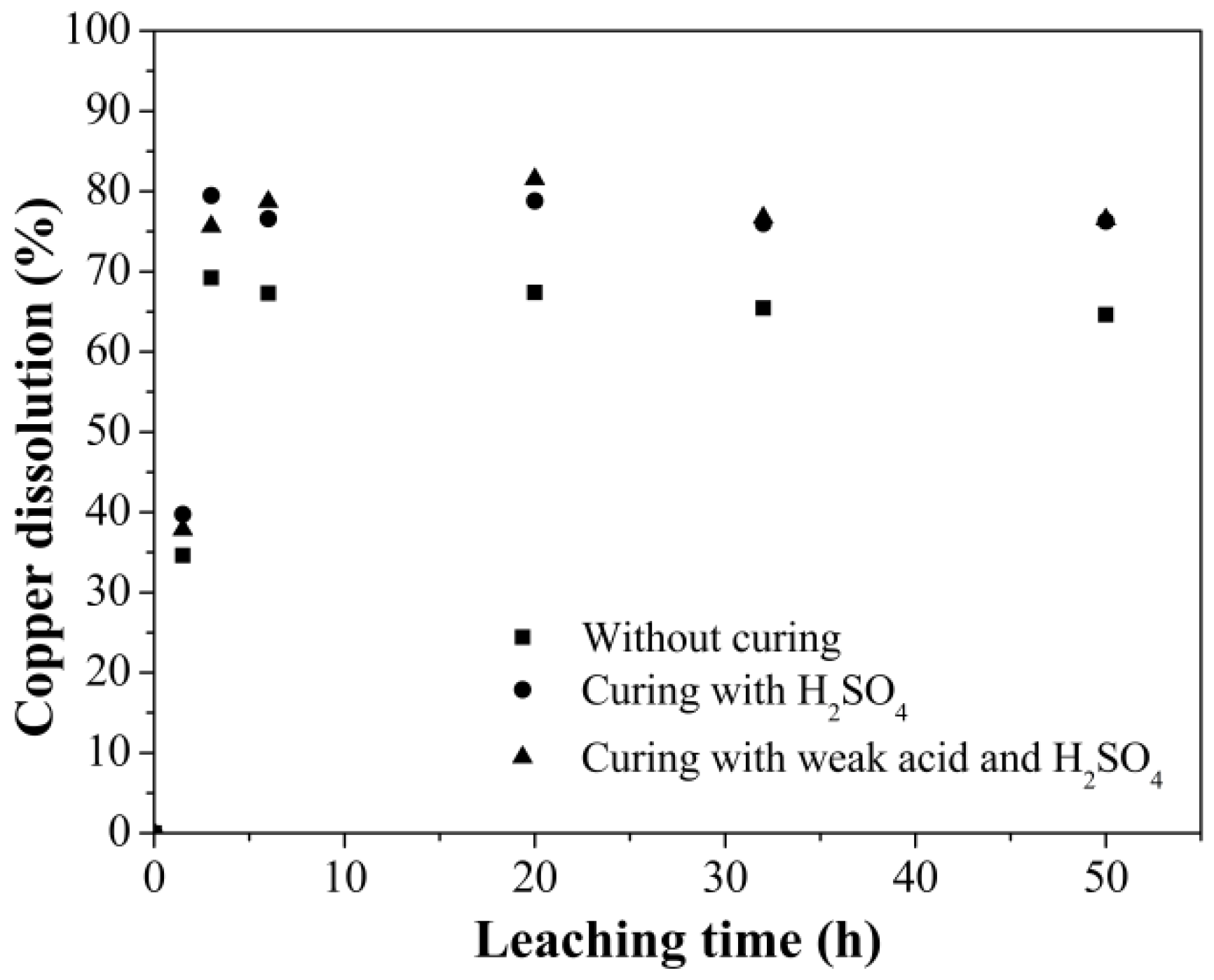
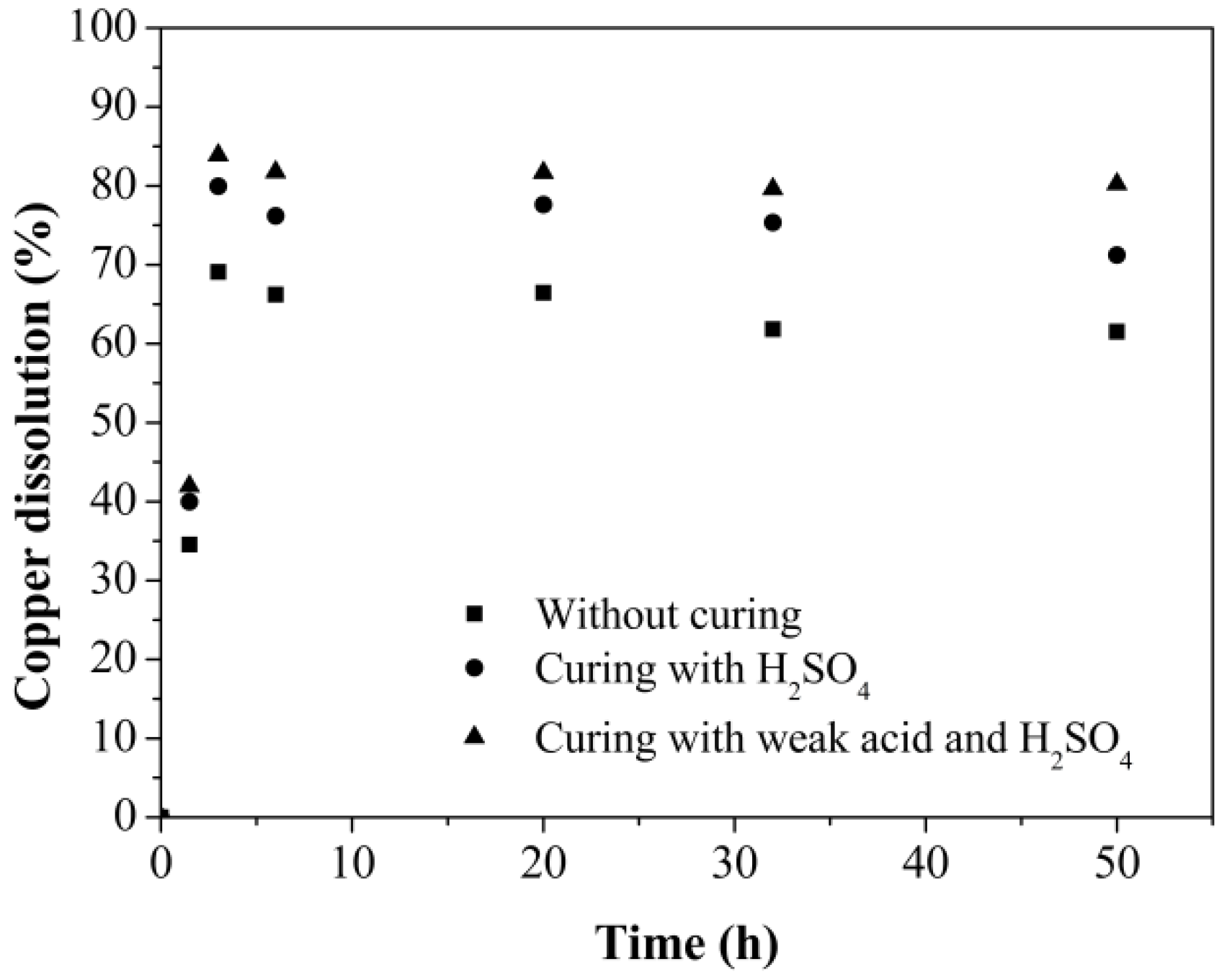
3.2. Leaching
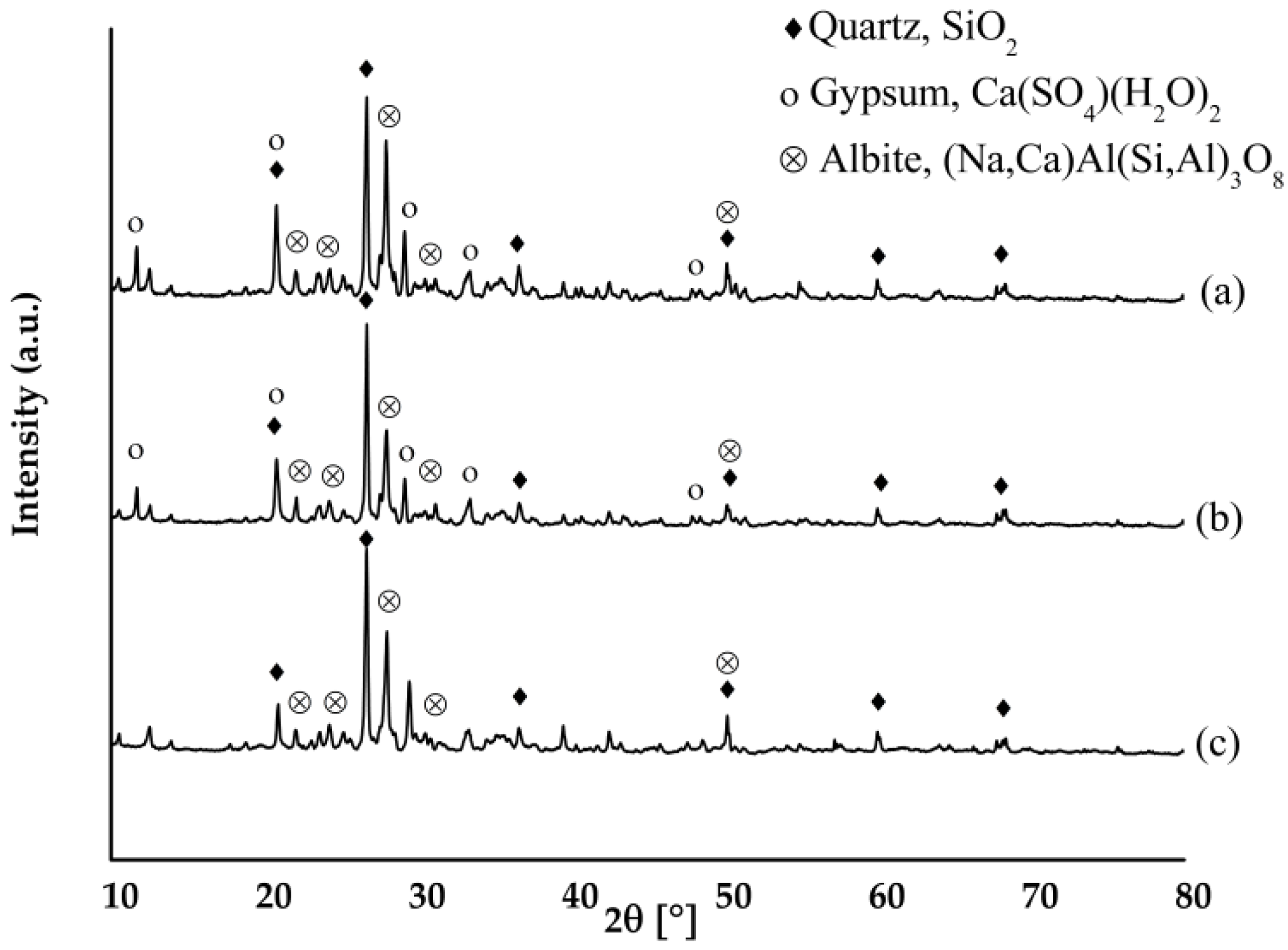
3.3. Residue of Leaching Tests
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sokić, M.; Marković, B.; Stanković, S.; Kamberović, Ž.; Štrbac, N.; Manojlović, V.; Petronijević, N. Kinetics of chalcopyrite leaching by hydrogen peroxide in sulfuric acid. Metals 2020, 9, 1173. [Google Scholar] [CrossRef]
- Guo, Z.; Zhu, D.; Pan, J.; Wu, T.; Zhang, F. Improving beneficiation of copper and iron from copper slag by modifying the molten copper slag. Metals 2016, 6, 86. [Google Scholar] [CrossRef]
- Drahota, P.; Filippi, M. Secondary arsenic minerals in the environment: A review. Environ. Int. 2009, 35, 1243–1255. [Google Scholar] [CrossRef] [PubMed]
- Filippou, D.; St-Germain, P.; Grammatikopoulos, T. Recovery of metal values from copper-Arsenic minerals and other related resources. Miner. Process. Extr. Metall. Rev. 2007, 28, 247–298. [Google Scholar] [CrossRef]
- Safarzadeh, M.S.; Moats, M.S.; Miller, J.D. An update to “recent trends in the processing of enargite concentrates”. Miner. Process. Extr. Metall. Rev. 2014, 35, 390–422. [Google Scholar] [CrossRef]
- Lee, J.; Kim, S.; Kim, B.; Lee, J. Effect of Mechanical Activation on the Kinetics of Copper Leaching from Copper Sulfide (CuS). Metals 2018, 8, 150. [Google Scholar] [CrossRef]
- Moskalyk, R.R.; Alfantazi, A.M. Review of copper pyrometallurgical practice: Today and tomorrow. Miner. Eng. 2003, 16, 893–919. [Google Scholar] [CrossRef]
- Pérez-Cortes, S.; Aguilera-Carvajal, Y.; Hurtado-Cruz, J.; Vargas-Norambuena, J.P. Modelación y simulación fluido-dinámica computacional de sistema de enfriamiento de gases para fundición de cobre mediante Convertidor Teniente con enfriador evaporativo. Rev. Int. Metod. Numer. Calc. Disen. Ing. 2017, 33, 179–187. [Google Scholar] [CrossRef]
- Torres, D.; Ayala, L.; Jeldres, R.I.; Cerecedo-sáenz, E.; Salinas-rodríguez, E.; Robles, P.; Toro, N. Leaching chalcopyrite with high MnO2 and chloride concentrations. Metals 2020, 10, 107. [Google Scholar] [CrossRef]
- Wang, Q.; Guo, X.; Tian, Q.; Chen, M.; Zhao, B. Reaction mechanism and distribution behavior of arsenic in the bottom blown copper smelting process. Metals 2017, 7, 302. [Google Scholar] [CrossRef]
- Habashi, F. Extractive Metallurgy Today, Progress and Problems. Miner. Process. Extr. Metall. Rev. 1992, 8, 17–33. [Google Scholar] [CrossRef]
- Mamyachenkov, S.V.; Anisimova, O.S.; Kostina, D.A. Improving the precipitation of arsenic trisulfide from washing waters of sulfuric-acid production of copper smelteries. Russ. J. Non-Ferrous Met. 2017, 58, 212–217. [Google Scholar] [CrossRef]
- Chen, Y.; Liao, T.; Li, G.; Chen, B.; Shi, X. Recovery of bismuth and arsenic from copper smelter flue dusts after copper and zinc extraction. Miner. Eng. 2012, 39, 23–28. [Google Scholar] [CrossRef]
- Barassi, G.M.; Klimsa, M.; Borrmann, T.; Cairns, M.J.; Kinkel, J.; Valenzuela, F. Lead sulfate nano- and microparticles in the acid plant blow-down generated at the sulfuric acid plant of the El Teniente mine, Chile. Environ. Sci. Process. Impacts 2014, 16, 2731–2741. [Google Scholar] [CrossRef] [PubMed]
- Dalewski, F. Removing arsenic from copper smelter gases. JOM 1999, 51, 24–26. [Google Scholar] [CrossRef]
- Berry, J.B.; Ferrada, J.B.; Dole, L.R.; Van Dyke, J.W. Sustainable production in the mining industry: By-product recovery. In Proceedings of the WM’00 Conference, Tuczon, AZ, USA, 10–15 April 2000. [Google Scholar]
- Chai, L.; Yue, M.; Yang, J.; Wang, Q.; Li, Q.; Liu, H. Formation of tooeleite and the role of direct removal of As(III) from high-arsenic acid wastewater. J. Hazard. Mater. 2016, 320, 620–627. [Google Scholar] [CrossRef]
- Luo, T.; Cui, J.; Hu, S.; Huang, Y.; Jing, C. Arsenic removal and recovery from copper smelting wastewater using TiO 2. Environ. Sci. Technol. 2010, 44, 9094–9098. [Google Scholar] [CrossRef]
- Guo, L.; Du, Y.; Yi, Q.; Li, D.; Cao, L.; Du, D. Efficient removal of arsenic from “dirty acid” wastewater by using a novel immersed multi-start distributor for sulphide feeding. Sep. Purif. Technol. 2015, 142, 209–214. [Google Scholar] [CrossRef]
- Cui, J.; Du, Y.; Xiao, H.; Yi, Q.; Du, D. A new process of continuous three-stage co-precipitation of arsenic with ferrous iron and lime. Hydrometallurgy 2014, 146, 169–174. [Google Scholar] [CrossRef]
- Lewis, A.E. Review of metal sulphide precipitation. Hydrometallurgy 2010, 104, 222–234. [Google Scholar] [CrossRef]
- Du, Y.; Lu, Q.; Chen, H.; Du, Y.; Du, D. A novel strategy for arsenic removal from dirty acid wastewater via CaCO3-Ca(OH)2-Fe(III) processing. J. Water Process Eng. 2016, 12, 41–46. [Google Scholar] [CrossRef]
- Basha, C.A.; Selvi, S.J.; Ramasamy, E.; Chellammal, S. Removal of arsenic and sulphate from the copper smelting industrial effluent. Chem. Eng. J. 2008, 141, 89–98. [Google Scholar] [CrossRef]
- Wang, A.; Zhou, K.; Zhang, X.; Zhou, D.; Peng, C.; Chen, W. Reductive removal of arsenic from waste acid containing high-acidity and arsenic levels through iodide and copper powder synergy. Chem. Eng. J. 2019, 373, 23–30. [Google Scholar] [CrossRef]
- Raghav, M.; Shan, J.; Sáez, A.E.; Ela, W.P. Scoping candidate minerals for stabilization of arsenic-bearing solid residuals. J. Hazard. Mater. 2013, 263, 525–532. [Google Scholar] [CrossRef]
- Ma, X.; Gomez, M.A.; Yuan, Z.; Zhang, G.; Wang, S.; Li, S.; Yao, S.; Wang, X.; Jia, Y. A novel method for preparing an As(V) solution for scorodite synthesis from an arsenic sulphide residue in a Pb refinery. Hydrometallurgy 2019, 183, 1–8. [Google Scholar] [CrossRef]
- Nazari, A.M.; Radzinski, R.; Ghahreman, A. Review of arsenic metallurgy: Treatment of arsenical minerals and the immobilization of arsenic. Hydrometallurgy 2017, 174, 258–281. [Google Scholar] [CrossRef]
- Min, X.B.; Liao, Y.P.; Chai, L.Y.; Yang, Z.H.; Xiong, S.; Liu, L.; Li, Q.Z. Removal and stabilization of arsenic from anode slime by forming crystal scorodite. Trans. Nonferrous Met. Soc. China (Engl. Ed.) 2015, 25, 1298–1306. [Google Scholar] [CrossRef]
- Nishimura, T.; Robins, R.G. Confirmation that tooeleite is a ferric arsenite sulfate hydrate, and is relevant to arsenic stabilisation. Miner. Eng. 2008, 21, 246–251. [Google Scholar] [CrossRef]
- Twidwell, L.G.; Plessas, K.O.; Comba, P.G.; Dahnke, D.R. Removal of Arsenic from Wastewaters and Stabilization of Arsenic Bearing Waste Solids: Summary of Experimental Studies. Miner. Process. Extr. Metall. Rev. 1995, 15, 237–246. [Google Scholar] [CrossRef]
- Agrawal, A.; Kumari, S.; Parveen, M.; Sahu, K.K. Exploitation of copper bleed stream for the extraction and recovery of copper and nickel by bis(2,4,4-trimethylpentyl)phosphinic acid. Miner. Process. Extr. Metall. Rev. 2012, 33, 339–351. [Google Scholar] [CrossRef]
- Agrawal, A.; Bagchi, D.; Kumari, S.; Pandey, B.D. An overview of process options and behavioral aspects of the copper values recovered from the copper bleed stream of a copper smelter developed at the national metallurgical laboratory. Miner. Process. Extr. Metall. Rev. 2009, 30, 136–162. [Google Scholar] [CrossRef]
- Northey, S.A.; Mudd, G.M.; Werner, T.T.; Haque, N.; Yellishetty, M. Sustainable water management and improved corporate reporting in mining. Water Resour. Ind. 2019, 21, 100104. [Google Scholar] [CrossRef]
- Northey, S.A.; Madrid López, C.; Haque, N.; Mudd, G.M.; Yellishetty, M. Production weighted water use impact characterisation factors for the global mining industry. J. Clean. Prod. 2018, 184, 788–797. [Google Scholar] [CrossRef]
- Lagos, G.; Peters, D.; Videla, A.; Jara, J.J. The effect of mine aging on the evolution of environmental footprint indicators in the Chilean copper mining industry 2001–2015. J. Clean. Prod. 2018, 174, 389–400. [Google Scholar] [CrossRef]
- Palomo-Briones, R.; Ovando-Franco, M.; Razo-Flores, E.; Celis, L.B.; Rangel-Méndez, J.R.; Vences-Álvarez, E.; Cruz, R.; Lázaro, I.; Briones-Gallardo, R. An Overview of Reclaimed Wastewater Reuse in Gold Heap Leaching. Miner. Process. Extr. Metall. Rev. 2016, 37, 274–285. [Google Scholar] [CrossRef]
- Chilean Copper Commission (Cochilco). Yearbook Copper Statistics and Other Minerals 1999–2018; Chilean Government; Chilean Copper Commission (Cochilco): Santiago, Chile, 2019. [Google Scholar]
- Chilean Copper Commission (Cochilco). Water Consumption in Copper Mining 2017; Chilean Copper Commission (Cochilco): Santiago, Chile, 2018. [Google Scholar]
- Aitken, D.; Rivera, D.; Godoy-Faúndez, A.; Holzapfel, E. Water scarcity and the impact of the mining and agricultural sectors in Chile. Sustainability 2016, 8, 128. [Google Scholar] [CrossRef]
- Mhlongo, S.; Mativenga, P.T.; Marnewick, A. Water quality in a mining and water-stressed region. J. Clean. Prod. 2018, 171, 446–456. [Google Scholar] [CrossRef]
- Cisternas, L.A.; Gálvez, E.D. The use of seawater in mining. Miner. Process. Extr. Metall. Rev. 2018, 39, 18–33. [Google Scholar] [CrossRef]
- Cruz, C.; Reyes, A.; Jeldres, R.I.; Cisternas, L.A.; Kraslawski, A. Using Partial Desalination Treatment To Improve the Recovery of Copper and Molybdenum Minerals in the Chilean Mining Industry. Ind. Eng. Chem. Res. 2019, 58, 8915–8922. [Google Scholar] [CrossRef]
- Ikumapayi, F.; Rao, K.H. Recycling process water in complex sulfide ore flotation: Effect of calcium and sulfate on sulfide minerals recovery. Miner. Process. Extr. Metall. Rev. 2015, 36, 45–64. [Google Scholar] [CrossRef]
- Castillo, J.; Sepúlveda, R.; Araya, G.; Guzmán, D.; Toro, N.; Pérez, K.; Rodríguez, M.; Navarra, A. Leaching of White Metal in a NaCl-H2SO4 System under Environmental Conditions. Minerals 2019, 9, 319. [Google Scholar] [CrossRef]
- Hernández, P.C.; Taboada, M.E.; Herreros, O.O.; Torres, C.M.; Ghorbani, Y. Chalcopyrite dissolution using seawater-based acidic media in the presence of oxidants. Hydrometallurgy 2015, 157, 325–332. [Google Scholar] [CrossRef]
- Velásquez-Yévenes, L.; Quezada-Reyes, V. Influence of seawater and discard brine on the dissolution of copper ore and copper concentrate. Hydrometallurgy 2018, 180, 88–95. [Google Scholar] [CrossRef]
- Velásquez-Yévenes, L.; Torres, D.; Toro, N. Leaching of chalcopyrite ore agglomerated with high chloride concentration and high curing periods. Hydrometallurgy 2018, 181, 215–220. [Google Scholar] [CrossRef]
- Wittholz, M.K.; O’Neill, B.K.; Colby, C.B.; Lewis, D. Estimating the cost of desalination plants using a cost database. Desalination 2008, 229, 10–20. [Google Scholar] [CrossRef]
- Ziolkowska, J.R. Is Desalination Affordable?—Regional Cost and Price Analysis. Water Resour. Manag. 2015, 29, 1385–1397. [Google Scholar] [CrossRef]
- Bouffard, S.C. Review of agglomeration practice and fundamentals in heap leaching. Miner. Process. Extr. Metall. Rev. 2005, 26, 233–294. [Google Scholar] [CrossRef]
- Tiwari, M.K.; Bajpai, S.; Dewangan, U.K.; Tamrakar, R.K. Suitability of leaching test methods for fly ash and slag: A review. J. Radiat. Res. Appl. Sci. 2015, 8, 523–537. [Google Scholar] [CrossRef]
- Reig, M.; Vecino, X.; Valderrama, C.; Gibert, O.; Cortina, J.L. Application of selectrodialysis for the removal of As from metallurgical process waters: Recovery of Cu and Zn. Sep. Purif. Technol. 2018, 195, 404–412. [Google Scholar] [CrossRef]
- Lu, J.; Dreisinger, D.; West-Sells, P. Acid curing and agglomeration for heap leaching. Hydrometallurgy 2017, 167, 30–35. [Google Scholar] [CrossRef]
- Paktunc, D.; Bruggeman, K. Solubility of nanocrystalline scorodite and amorphous ferric arsenate: Implications for stabilization of arsenic in mine wastes. Appl. Geochem. 2010, 25, 674–683. [Google Scholar] [CrossRef]


| Element | mg/L | Element | mg/L | Element | mg/L |
|---|---|---|---|---|---|
| Zn | 38,195.3 | Mg | 233.0 | Li | 2.0 |
| As | 3676.0 | Bi | 90.9 | Hg | 0.01 |
| Cu | 2424.0 | Pb | 87.8 | Co | 0.0 |
| Fe | 1570.0 | Sb | 80.0 | Se | 0.0 |
| Cd | 1172.0 | Mo | 54.0 | Sr | 0.0 |
| Ca | 664.0 | Cr | 28.0 | Ti | 0.0 |
| Ni | 188.0 | Tl | 7.0 | V | 0.0 |
| Element | Amount (wt%) |
|---|---|
| Cu | 1.59 |
| Fe | 10.65 |
| As | 0.004 |
| Cd | 0.0003 |
| Ca | 4.17 |
| Co | 0.0063 |
| Pb | 0.015 |
| Li | 0.0012 |
| Mg | 1.02 |
| Mn | 0.09 |
| Sr | 0.006 |
| Ti | 0.10 |
| Zn | 0.08 |
| Bi | 0.0625 |
| Hg | 0.0004 |
| Mineral | Amount (wt%) |
|---|---|
| Albite | 22.1 |
| Quartz | 25.6 |
| Anorthite | 16.2 |
| Orthoclase | 5.6 |
| Calcite | 5.7 |
| Chlorite | 5.7 |
| Hornblende | 4.5 |
| Titanite | 3.1 |
| Epidote | 2.7 |
| Hematite | 4.2 |
| Cuprite | 0.9 |
| Muscovite | 2.7 |
| Tenorite | 0.4 |
| Other minerals | 0.6 |
| Total | 100.0 |
| Set | Dosage of H2SO4 (kg H2SO4/t ore) | Weak acid in Water Replacement (%) |
|---|---|---|
| 1 | 30 | 0 |
| 60 | ||
| 90 | ||
| 120 | ||
| 2 | From set 1 | 25 |
| 50 | ||
| 65 |
| Solid Fraction (%) | Sulfuric Acid (g/L) | Weak acid Replacement to Sulfuric Acid (%) | Curing Conditions |
|---|---|---|---|
| 20 | 25 | 0 | Without curing |
| Curing with H2SO4 (90 g/t) and water | |||
| Curing with weak acid, H2SO4, and water (60% weak acid, and 90 g/t of H2SO4 contributed by weak acid and concentrated sulfuric acid) | |||
| 20 | 25 | 100 | Without curing |
| Curing with H2SO4 (90 g/t) and water | |||
| Curing with weak acid, H2SO4, and water (60% weak acid and 90 g/t of H2SO4 contributed by weak acid and concentrated sulfuric acid) |
| Element g/L | With Sulfuric Acid | With Weak Acid | ||||
|---|---|---|---|---|---|---|
| Without Curing | Curing with H2SO4 and Water | Curing with Weak Acid, H2SO4, and Water | Without Curing | Curing with H2SO4 and Water | Curing with Weak Acid, H2SO4, and Water | |
| Cd | 0.00 | 0.00 | 419- | 0.31 | 0.32 | 0.44 |
| Zn | 0.02 | 0.00 | 1.95 | 8.25 | 9.37 | 12.17 |
| Sb | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Pb | 0.02 | 0.02 | 0.01 | 0.01 | 0.00 | 0.00 |
| As | 0.00 | 0.00 | 0.18 | 0.80 | 1.07 | 1.46 |
| Cu | 3.03 | 3.48 | 3.49 | 3.66 | 4.06 | 4.48 |
| Fe | 0.35 | 2.34 | 2.25 | 0.74 | 2.38 | 2.38 |
| Mn | 0.12 | 0.14 | 0.15 | 0.15 | 0.14 | 0.14 |
| Hg | <0.001 (*) | <0.001 (*) | <0.001 (*) | <0.001 (*) | <0.001 (*) | <0.001 (*) |
| Bi | 0.0112 | 0.0110 | 0.0128 | 0.0159 | 0.0263 | 0.0301 |
| Amount (wt%) | With Sulfuric Acid | With Weak Acid | ||||
|---|---|---|---|---|---|---|
| Without Curing | Curing with H2SO4 and Water | Curing with Weak Acid, H2SO4, and Water | Without Curing | Curing with H2SO4 and Water | Curing with Weak Acid, H2SO4, and Water | |
| Cd | 0.0026 | 0.0250 | 0.0031 | 0.0244 | 0.040 | 0.0065 |
| Zn | 0.0205 | 0.0288 | 0.0268 | 0.044 | 0.045 | 0.030 |
| Sb | 0.0114 | 0.0096 | 0.0100 | 0.0113 | 0.0114 | 0.0108 |
| Pb | 0.173 | 0.172 | 0.170 | 0.214 | 0.1808 | 0.163 |
| As | 0.182 | 0.602 | 0.191 | 1.766 | 1.144 | 0.856 |
| Cu | 0.199 | 0.220 | 0.220 | 0.510 | 0.204 | 0.191 |
| Fe | 7.57 | 8.653 | 8.418 | 8.508 | 8.380 | 8.053 |
| Mn | 0.042 | 0.338 | 0.034 | 0.038 | 0.029 | 0.030 |
| Hg | <0.001 (*) | <0.001 (*) | <0.001 (*) | <0.001 (*) | <0.001 (*) | <0.001 (*) |
| Bi | 0.043 | 0.0795 | 0.053 | 0.209 | 0.171 | 0.139 |
| Test | Condition | Element Concentration (mg/L) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Pb | Cd | Hg | Cr | Ba | Se | As | Ag | ||
| TCLP | Without weak acid | 0.384 | 0.019 | <0.001 * | 0.029 | 14.330 | <0.3 * | 0.704 | <0.007 * |
| With weak acid | 0.516 | 0.559 | <0.001 * | 0.036 | 14.720 | <0.3 * | 2.141 | <0.007 * | |
| SPLP | Without weak acid | 0.159 | 0.010 | <0.001 * | 0.026 | 12.300 | <0.3 * | 0.608 | <0.007 * |
| With weak acid | 0.347 | 0.532 | <0.001 * | 0.036 | 10.230 | <0.3 * | 2.071 | 0.008 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araya, G.; Toro, N.; Castillo, J.; Guzmán, D.; Guzmán, A.; Hernández, P.; Jeldres, R.I.; Sepúlveda, R. Leaching of Oxide Copper Ores by Addition of Weak Acid from Copper Smelters. Metals 2020, 10, 627. https://doi.org/10.3390/met10050627
Araya G, Toro N, Castillo J, Guzmán D, Guzmán A, Hernández P, Jeldres RI, Sepúlveda R. Leaching of Oxide Copper Ores by Addition of Weak Acid from Copper Smelters. Metals. 2020; 10(5):627. https://doi.org/10.3390/met10050627
Chicago/Turabian StyleAraya, Giselle, Norman Toro, Jonathan Castillo, Danny Guzmán, Alexis Guzmán, Pía Hernández, Ricardo I. Jeldres, and Rossana Sepúlveda. 2020. "Leaching of Oxide Copper Ores by Addition of Weak Acid from Copper Smelters" Metals 10, no. 5: 627. https://doi.org/10.3390/met10050627
APA StyleAraya, G., Toro, N., Castillo, J., Guzmán, D., Guzmán, A., Hernández, P., Jeldres, R. I., & Sepúlveda, R. (2020). Leaching of Oxide Copper Ores by Addition of Weak Acid from Copper Smelters. Metals, 10(5), 627. https://doi.org/10.3390/met10050627











