WO3 and Ionic Liquids: A Synergic Pair for Pollutant Gas Sensing and Desulfurization
Abstract
1. Introduction
Ionic Liquids
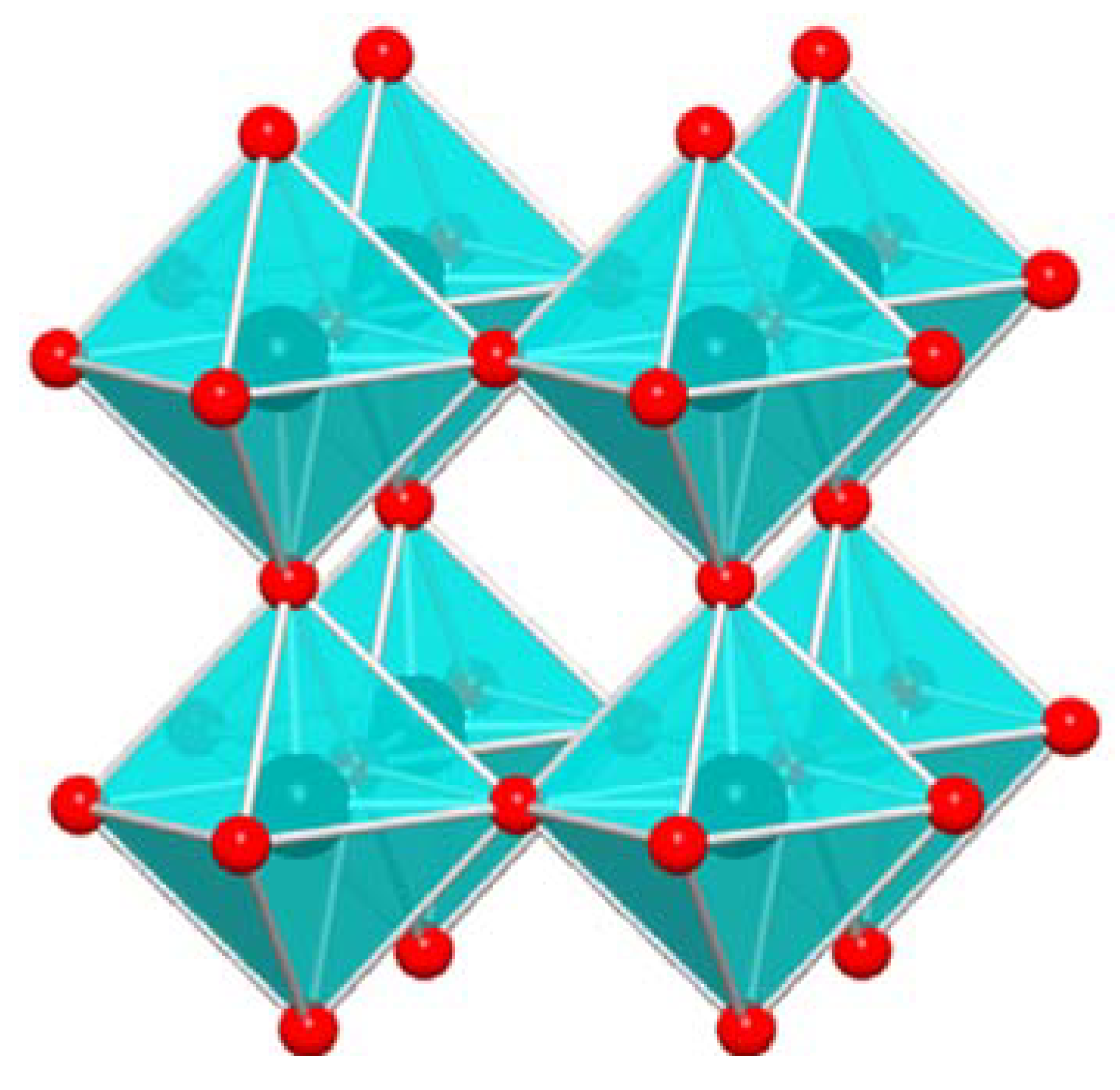
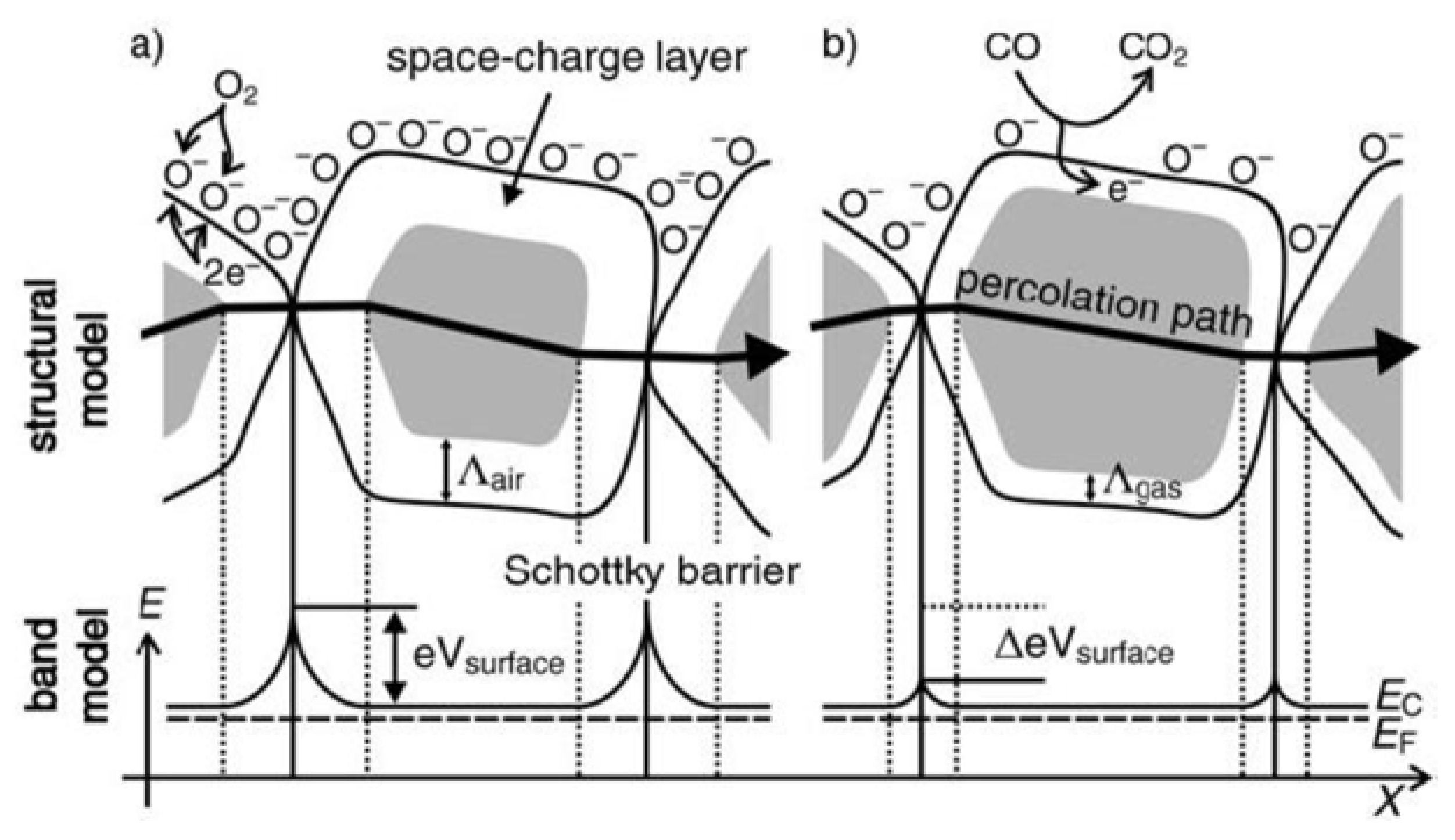
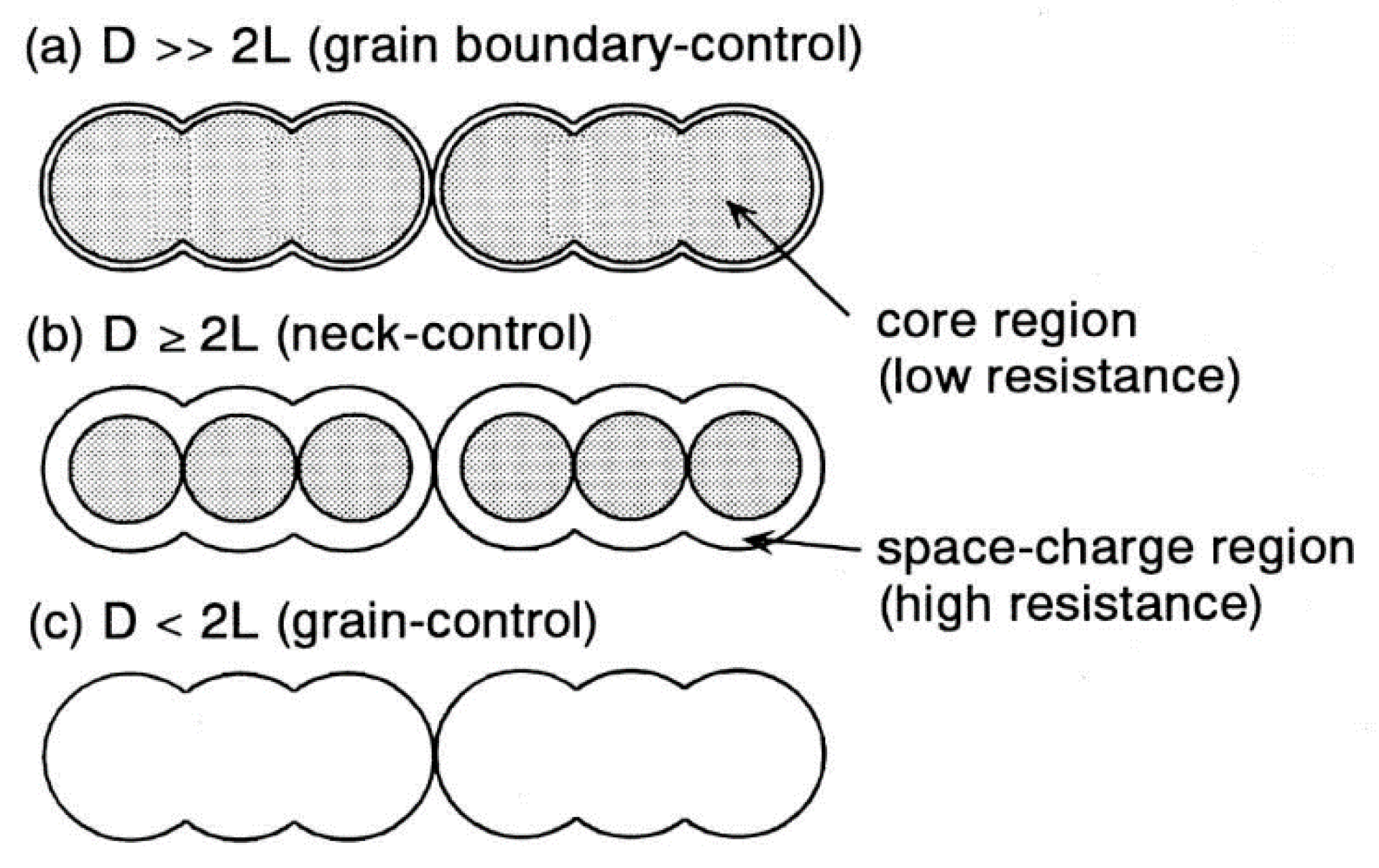
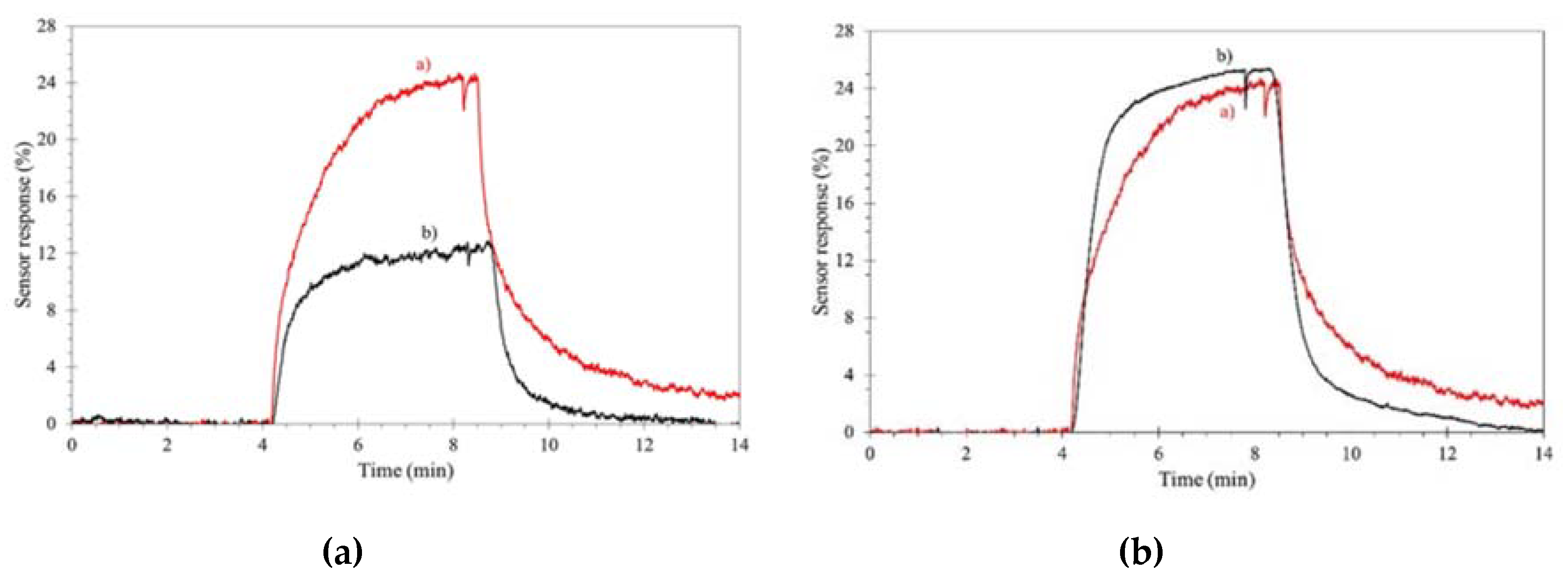
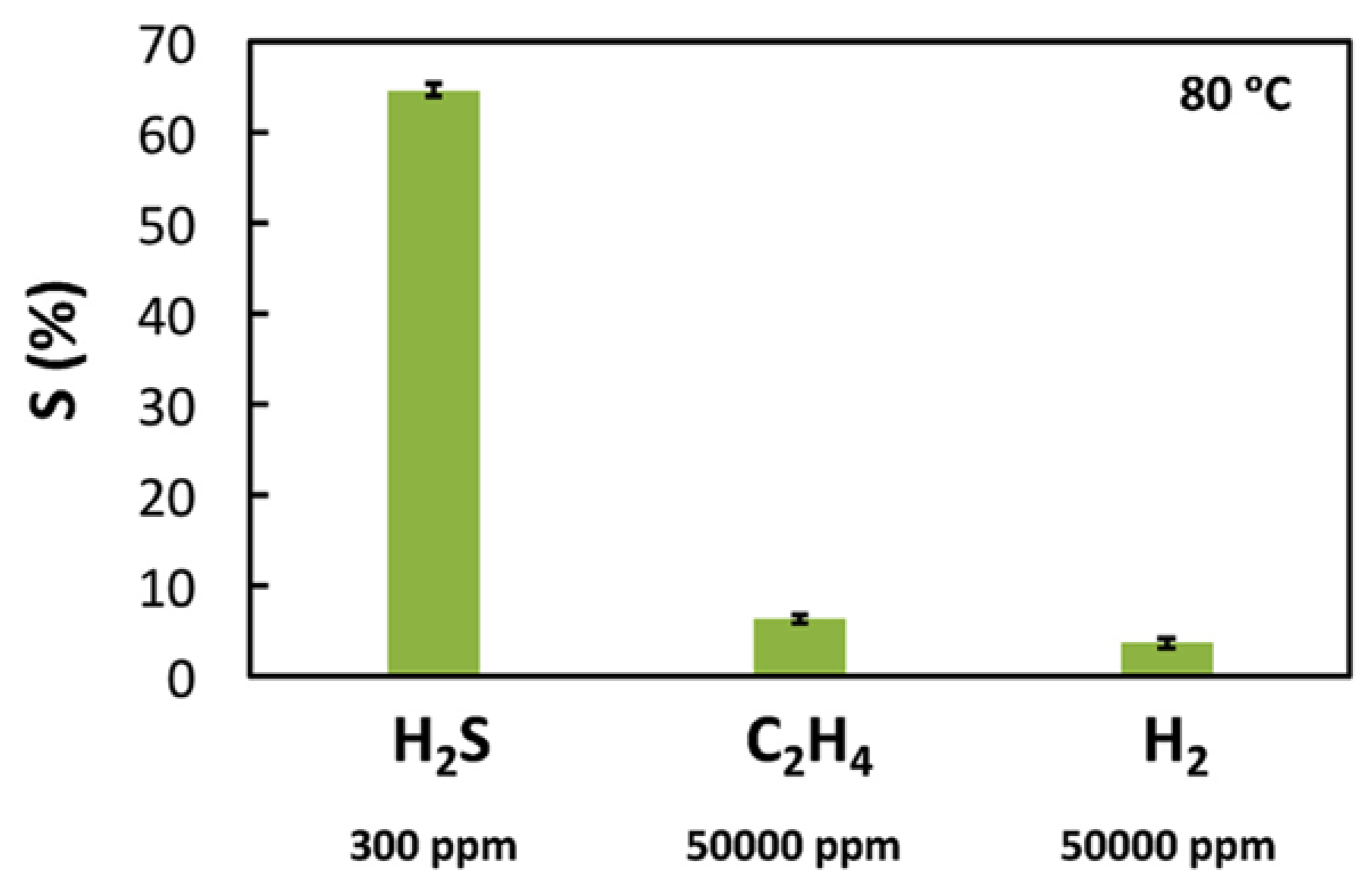
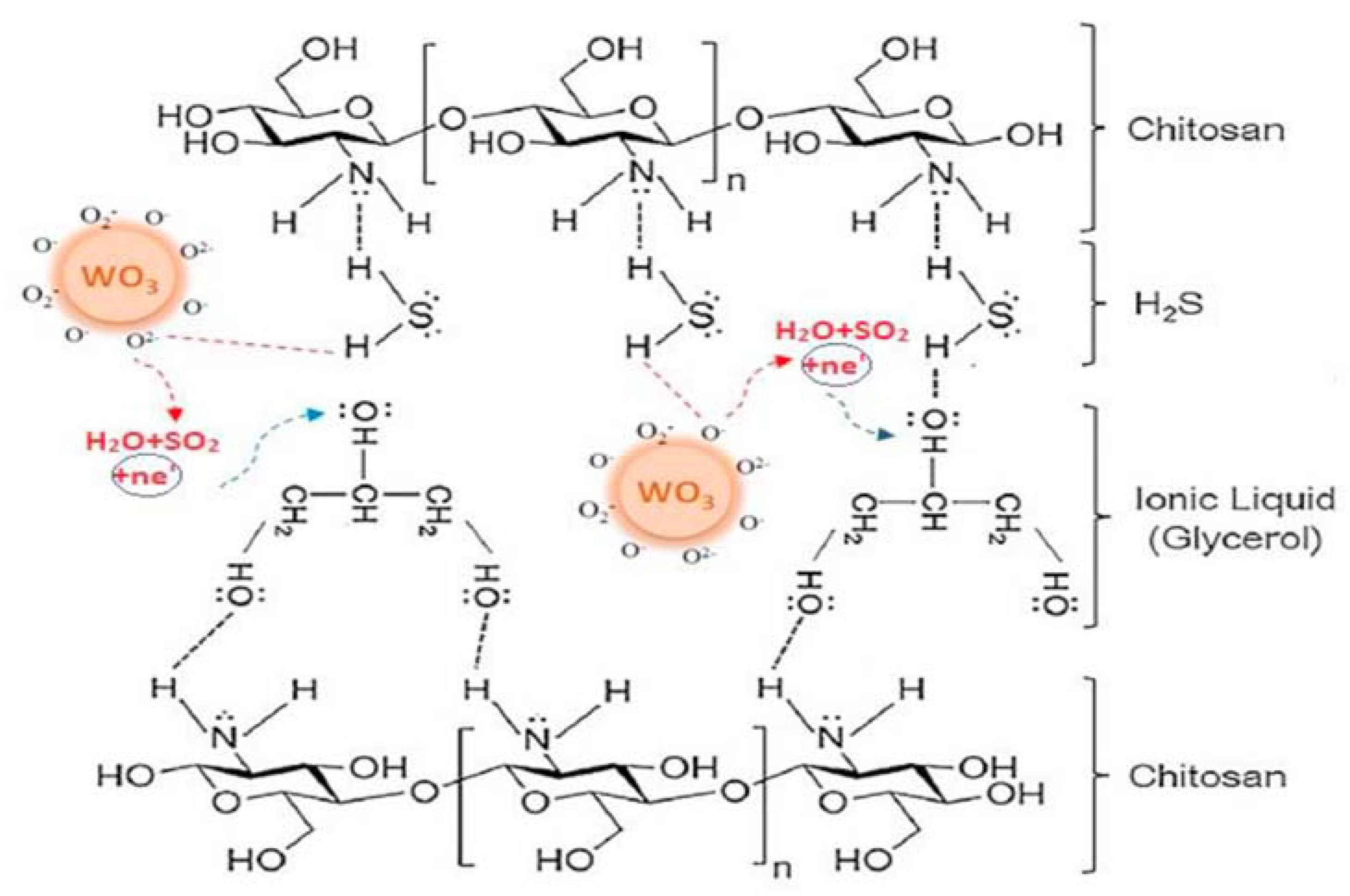
2. WO3 and Ionic Liquids in Pollutant Gas Sensing
- Fuel combustion from motor vehicles (e.g., cars and heavy-duty vehicles),
- Heat and power generation (e.g., oil and coal power plants and boilers),
- Industrial facilities (e.g., manufacturing factories, mines and oil refineries),
- Municipal and agricultural waste sites and waste incineration/burning,
- Residential cooking, heating and lighting with polluting fuels.
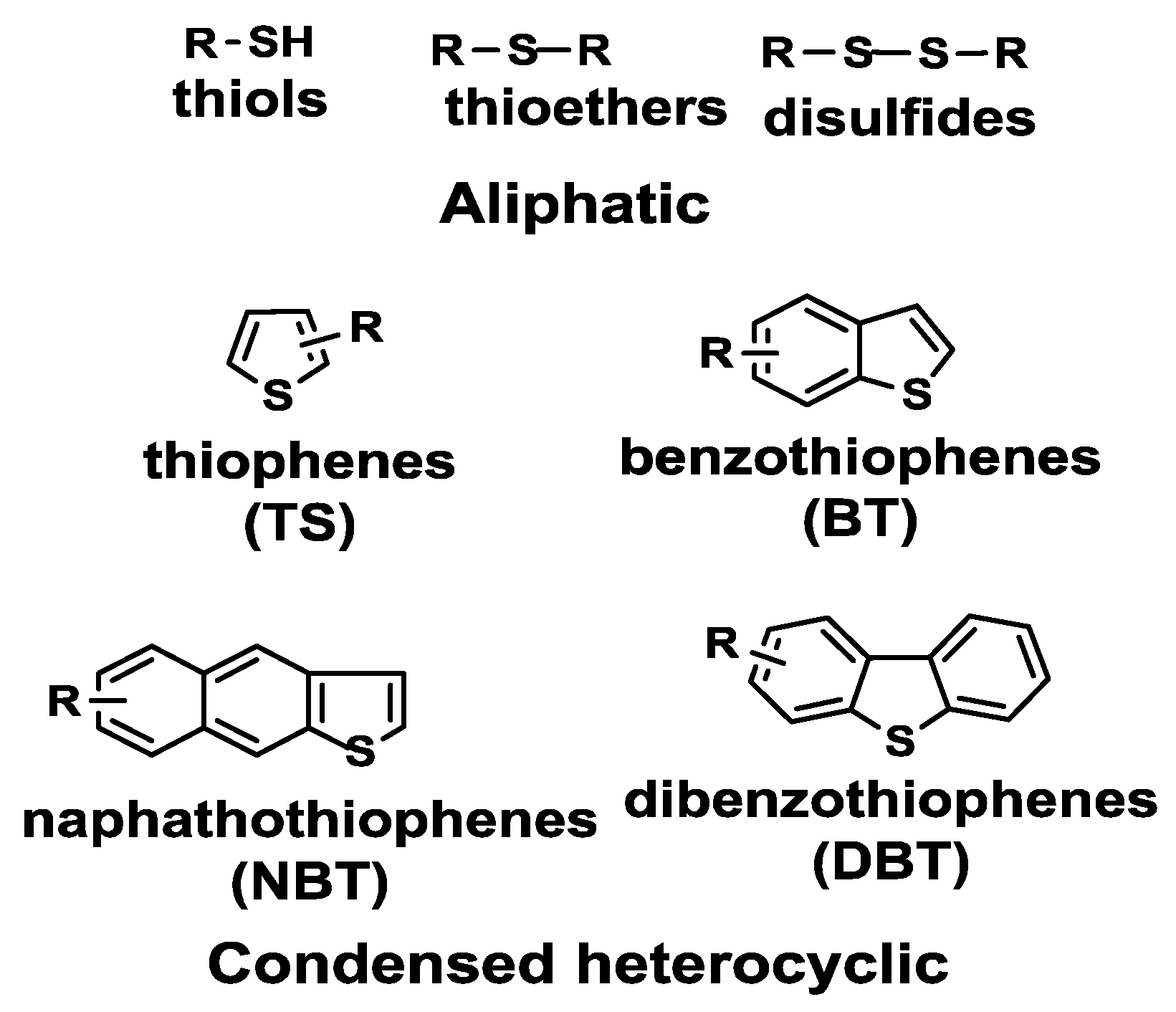
3. WO3 and Ionic Liquids in Fuel Desulfurization
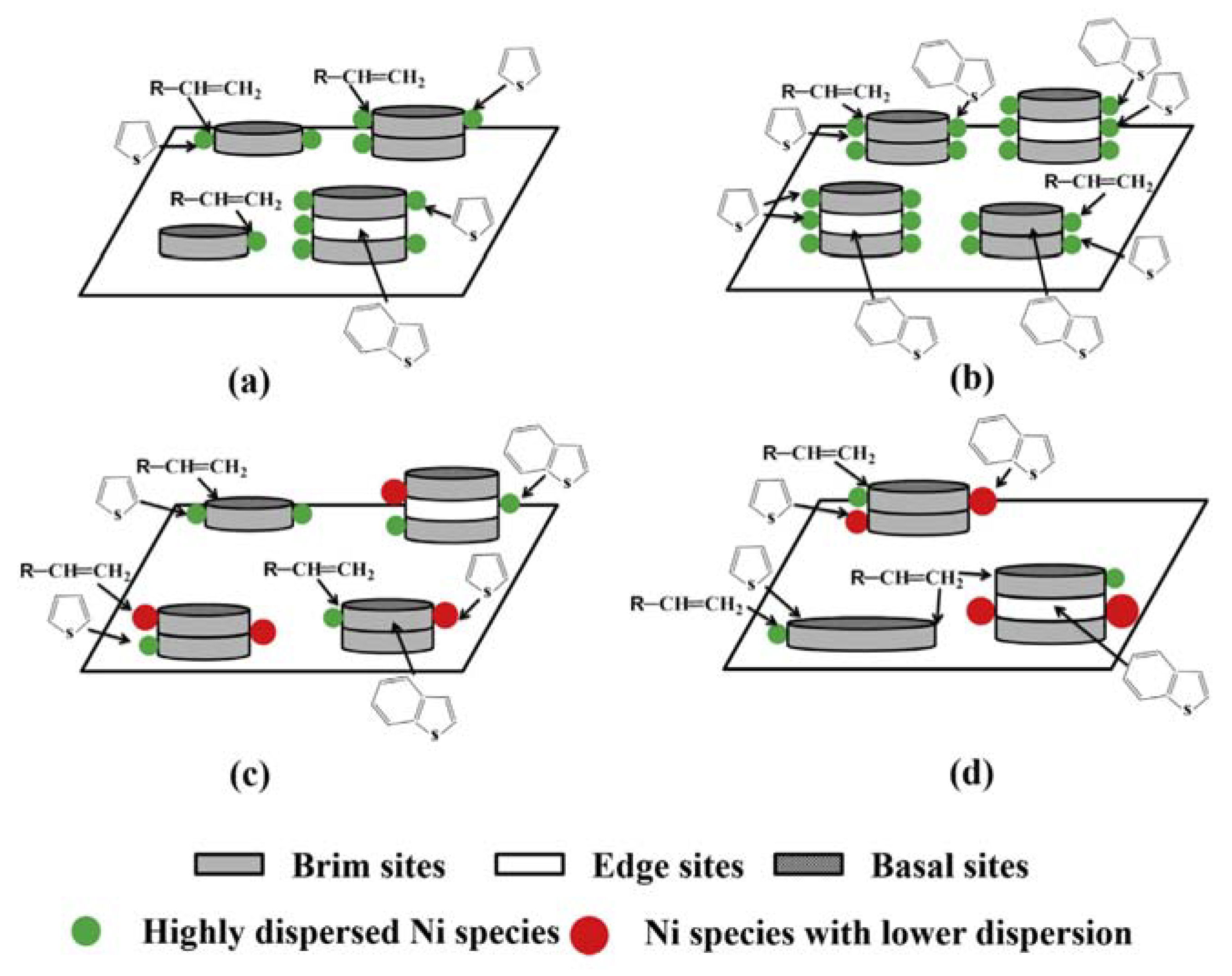
3.1. WO3 and Ionic Liquids in Hydrodesulfurization (HDS)
3.2. WO3 and Ionic Liquids in Oxidative Desulfurization (ODS)
4. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kwong, W.L.; Savvides, N.; Sorrell, C.C. Electrodeposited nanostructured WO3 thin films for photoelectrochemical applications. Electrochim. Acta 2012, 75, 371–380. [Google Scholar] [CrossRef]
- Yu, J.; Qi, L. Template-free fabrication of hierarchically flower-like tungsten trioxide assemblies with enhanced visible-light-driven photocatalytic activity. J. Hazard. Mater. 2009, 169, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Amano, F.; Ishinaga, E.; Yamakata, A. Effect of Particle Size on the Photocatalytic Activity of WO3 Particles for Water Oxidation. J. Phys. Chem. C 2013, 117, 22584–22590. [Google Scholar] [CrossRef]
- Long, H.; Zeng, W.; Zhang, H. Synthesis of WO3 and its gas sensing: A review. J. Mater. Sci.: Mater. Electron. 2015, 26, 4698–4707. [Google Scholar] [CrossRef]
- Grilli, M.L.; Chevallier, L.; Vona, M.L.D.; Licoccia, S.; Bartolomeo, E.D. Planar electrochemical sensors based on YSZ with WO3 electrode prepared by different chemical routes. Sensors Actuator. B Chem. 2005, 111, 91–95. [Google Scholar] [CrossRef]
- Lu, R.; Zhong, X.; Shang, S.; Wang, S.; Tang, M. Effects of sintering temperature on sensing properties of WO3 and Ag-WO3 electrode for NO2 sensor. R. Soc. Open Sci. 2018, 5, 171691. [Google Scholar] [CrossRef] [PubMed]
- Dutta, A.; Kaabbuathong, N.; Grilli, M.L.; Di Bartolomeo, E.; Traversa, E. Study of YSZ-Based Electrochemical Sensors with WO3 Electrodes in NO2 and CO Environments. J. Electrochem. Soc. 2003, 150, H33–H37. [Google Scholar] [CrossRef]
- Yamazoe, N. New approaches for improving semiconductor gas sensors. Sens. Actuators B Chem. 1991, 5, 7–19. [Google Scholar] [CrossRef]
- Granqvist, C.G. Electrochromic tungsten oxide films: Review of progress 1993–1998. Sol. Energy Mater. Sol. Cells 2000, 60, 201–262. [Google Scholar] [CrossRef]
- Masetti, E.; Grilli, M.L.; Dautzenberg, G.; Macrelli, G.; Adamik, M. Analysis of the influence of the gas pressure during the deposition of electrochromic WO3 films by reactive r.f. sputtering of W and WO3 target. Solar Energy Mater. Solar Cells 1999, 56, 259–269. [Google Scholar] [CrossRef]
- Rao, M.C. Structure and properties of WO3 thin films for electrochromic device application. J. Non-Oxide Glasses 2013, 5, 1–8. [Google Scholar]
- Arvizu, M.A.; Qu, H.-Y.; Cindemir, U.; Qiu, Z.; Rojas-González, E.A.; Primetzhofer, D.; Granqvist, C.G.; Österlund, L.; Niklasson, G.A. Electrochromic WO3 thin films attain unprecedented durability by potentiostatic pretreatment. J. Mater. Chem. A 2019, 7, 2908–2918. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.-J.; He, J.-L.; Jaing, C.-C.; Liang, C.-J.; Chou, C.-H.; Han, C.-Y.; Tien, C.-L. An All-Solid-State Electrochromic Device Based on WO3–Nb2O5 Composite Films Prepared by Fast-Alternating Bipolar-Pulsed Reactive Magnetron Sputtering. Coatings 2019, 9, 9. [Google Scholar] [CrossRef]
- Valerini, D.; Hernández, S.; Di Benedetto, F.; Russo, N.; Saracco, G.; Rizzo, A. Sputtered WO3 films for water splitting applications. Mater. Sci. Semicon. Proc. 2016, 42, 150–154. [Google Scholar] [CrossRef]
- Gerand, B.; Nowogrocki, G.; Guenot, J.; Figlarz, M. Structural study of a new hexagonal form of tungsten trioxide. J. Solid State Chem. 1979, 29, 429–434. [Google Scholar] [CrossRef]
- Sun, W.; Yeung, M.T.; Lech, A.T.; Lin, C.-W.; Lee, C.; Li, T.; Duan, X.; Zhou, J.; Kaner, R.B. High surface area tunnels in hexagonal WO3. Nano Lett. 2015, 15, 4834–4838. [Google Scholar] [CrossRef]
- Lee, T.; Lee, Y.; Jang, W.; Soon, A. Understanding the advantage of hexagonal WO3 as an efficient photoanode for solar water splitting: A first-principles perspective. J. Mater. Chem. A 2016, 4, 11498–11506. [Google Scholar] [CrossRef]
- Kollender, J.P.; Gallistl, B.; Mardare, A.I.; Hassel, A.W. Photoelectrochemical water splitting in a tungsten oxide—nickel oxide thin film material library. Electrochim. Acta 2014, 140, 275–281. [Google Scholar] [CrossRef]
- Wiseman, P.J.; Dickens, P.G. The crystal structure of cubic hydrogen tungsten bronze. J. Solid State Chem. 1973, 6, 374–377. [Google Scholar] [CrossRef]
- Mardare, C.C.; Hassel, A.W. Review on the Versatility of Tungsten Oxide Coatings. Phys. Status Solidi A 2019, 216, 1900047. [Google Scholar] [CrossRef]
- Biswas, S.K.; Baeg, J.O. A facile one-step synthesis of single crystalline hierarchical WO3 with enhanced activity for photoelectrochemical solar water oxidation. Int. J. Hydrog. Energy 2013, 8, 3177–3188. [Google Scholar] [CrossRef]
- Gullapalli, S.K.; Vemuri, R.S.; Ramana, C.V. Structural transformation induced changes in the optical properties of nanocrystalline tungsten oxide thin films. Appl. Phys. Lett. 2010, 96, 171903. [Google Scholar] [CrossRef]
- Inoue, T.; Ohtsuka, K.; Yoshida, Y.; Matsuura, Y.; Kajiyama, Y. Metal oxide semiconductor NO2 sensor. Sens. Actuators B 1995, 25, 388–391. [Google Scholar] [CrossRef]
- Kim, T.; Kim, Y.; Yoo, K.S.; Sung, G.; Jung, H. Sensing characteristics of dc reactive sputtered WO3 thin films as an NOx gas sensor. Sens. Actuators B Chem. 2000, 62, 102–108. [Google Scholar] [CrossRef]
- Qin, Y.X.; Ye, Z.H. DFT study on interaction of NO2 with the vacancy-defected WO3 nanowires for gas-sensing. Sens. Actuators B Chem. 2016, 222, 499–507. [Google Scholar] [CrossRef]
- Di Bartolomeo, E.; Grilli, M.L.; Yoon, J.W.; Traversa, E. Zirconia-Based Electrochemical NOx Sensors with Semiconducting Oxide Electrodes. J. Am. Ceram. Soc. 2004, 87, 1883–1889. [Google Scholar] [CrossRef]
- Di Bartolomeo, E.; Grilli, M.L.; Traversa, E. Sensing Mechanism of Potentiometric Gas Sensors Based on Stabilized Zirconia with Oxide Electrodes, Is It Always Mixed Potential? J. Electrochem. Soc. 2004, 151, H133–H139. [Google Scholar] [CrossRef]
- Maekawa, T.; Tamaki, J.; Miura, N.; Yamazoe, N. Gold-loaded tungsten-oxide sensor for detection of ammonia in air. Chem. Lett. 1992, 4, 639–642. [Google Scholar] [CrossRef]
- Meixner, H.; Gerblinger, J.; Lampe, U.; Fleischer, M. Thin-film gas sensors based on semiconducting metal oxides. Sens. Actuators B 1995, 23, 119–125. [Google Scholar] [CrossRef]
- Ruokamo, I.; Karkkainen, T.; Huusko, J.; Ruokanen, T.; Blomberg, M.; Torvela, H.; Lantto, V. H2S response of WO3 thin film sensors manufactured by Silican processing technology. Sens. Actuators B 1994, 19, 486–488. [Google Scholar] [CrossRef]
- Zhu, L.F.; She, J.C.; Luo, J.Y.; Deng, S.Z.; Chen, J.; Xu, N.S. Study of Physical and Chemical Processes of H2 Sensing of Pt-Coated WO3 Nanowire Films. Phys. Chem. C 2010, 114, 15504–15509. [Google Scholar] [CrossRef]
- Shimizu, Y.; Matsunaga, N.; Hyodo, T.; Egashira, M. Improvement of SO2 Sensing Properties of WO3 by Noble Metal Loading. Sens. Actuators B Chem. 2011, 77, 35–40. [Google Scholar] [CrossRef]
- Grilli, M.L.; Di Bartolomeo, E.; Lunardi, A.; Chevallier, L.; Cordiner, S.; Traversa, E. Planar non-nernstian electrochemical sensors: Field test in the exhaust of a spark ignition engine. Sens. Actuators B Chem. 2005, 108, 319–332. [Google Scholar] [CrossRef]
- Grilli, M.L.; Kaabbuathong, N.; Dutta, A.; Di Bartolomeo, E.; Traversa, E. Electrochemical NO2 sensors with WO3 electrodes for high temperature applications. J. Ceramic Soc. Jpn. 2002, 110, 159–162. [Google Scholar] [CrossRef][Green Version]
- Yamazoe, N.; Shimanoe, K. Theory of Power Laws for Semiconductor Gas Sensors. Sens. Actuators B Chem. 2008, 128, 566–573. [Google Scholar] [CrossRef]
- Wang, C.; Yin, L.; Zhang, L.; Xiang, D.; Gao, R. Metal Oxide Gas Sensors: Sensitivity and Influencing Factors. Sensors 2010, 10, 2088–2106. [Google Scholar] [CrossRef]
- Xu, C.; Tamaki, J.; Miura, N.; Yamazoe, N. Grain size effects on gas sensitivity of porous SnO2-based elements. Sens. Actuators B 1991, 3, 147–155. [Google Scholar] [CrossRef]
- Lin, T.; Lv, X.; Li, S.; Wang, Q. The Morphologies of the Semiconductor Oxides and Their Gas-Sensing Properties. Sensors 2017, 17, 2779. [Google Scholar] [CrossRef]
- Li, X.L.; Lou, T.J.; Sun, X.M.; Li, Y.D. Highly sensitive WO3 hollow-sphere gas sensors. Inorg. Chem. 2004, 43, 5442–5449. [Google Scholar] [CrossRef]
- Yao, Y.; Ji, F.; Yin, M.; Ren, X.; Ma, Q.; Yan, J.; Liu, S.F. Ag Nanoparticle-sensitized WO3 hollow nanosphere for localized surface plasmon enhanced gas sensors. ACS Appl. Mat. Interface. 2016, 8, 18165–18172. [Google Scholar] [CrossRef]
- Kong, W.; Zhang, R.; Zhang, X.; Ji, L.; Yu, G.; Wang, T.; Luo, Y.; Shi, X.; Xu, Y.; Sun, X. WO3 nanosheets rich in oxygen vacancies for enhanced electrocatalytic N2 reduction to NH3. Nanoscale 2019, 11, 19274–19277. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Miyauchi, M.; Yamazaki, T.; Shen, Y. Facile synthesis and NO2 gas sensing of tungsten oxide nanorods assembled microspheres. Sens. Actuators B 2009, 140, 514–519. [Google Scholar] [CrossRef]
- Cao, B.; Chen, J.; Tang, X.; Zhou, W. Growth of monoclinic WO3 nanowire array for highly sensitive NO2 detection. J. Mater. Chem. 2009, 19, 2323–2327. [Google Scholar] [CrossRef]
- Cai, Z.-X.; Li, H.-Y.; Yang, X.-N.; Guo, X. NO sensing by single crystalline WO3 nanowires. Sens. Actuators B 2015, 219, 346–353. [Google Scholar] [CrossRef]
- Ji, H.; Zeng, W.; Li, Y. Gas sensing mechanisms of metal oxide semiconductors: A focus review. Nanoscale 2019, 11, 22664–22684. [Google Scholar] [CrossRef]
- Yan, H.; Zhang, X.; Zhou, S.; Xie, X.; Luo, Y.; Yu, Y. Synthesis of WO3 nanoparticles for photocatalytic O2 evolution by thermal decomposition of ammonium tungstate loading on g-C3N4. J. Alloys Compounds 2011, 509, L232–L235. [Google Scholar] [CrossRef]
- Gomez, C.; Sánchez Martínez, D.; Juarez, I.; Martinez, A.; Torres-Martínez, L. Facile synthesis of m-WO3 powders via precipitation in ethanol solution and evaluation of their photocatalytic activities. J. Photochem. Photobiol. A: Chem. 2013, 262, 28–33. [Google Scholar] [CrossRef]
- Kondalkar, V.V.; Kharade, R.R.; Mali, S.S.; Mane, R.M.; Patil, P.B.; Patil, P.S.; Choudhury, S.; Bhosale, P.N. Nanobrick-like WO3 thin films: Hydrothermalsynthesis and electrochromic application. Superlattices Microstruct. 2014, 73, 290–295. [Google Scholar] [CrossRef]
- Kida, T.; Nishiyama, A.; Hua, Z.; Suematsu, K.; Yuasa, M.; Shimanoe, K. WO3 nanolamella gas sensor: Porosity control using SnO2 nanoparticles for enhanced NO2 sensing. Langmuir 2014, 30, 2571–2579. [Google Scholar] [CrossRef]
- Meng, Z.; Fujii, A.; Hashishin, T.; Wada, N.; Sanada, T.; Tamaki, J.; Kojima, K.; Haneoka, h.; Suzuki, T. Morphological and crystal structural control of tungsten trioxide for highly sensitive NO2 gas sensors. J. Mater. Chem. C 2015, 3, 1134–1141. [Google Scholar] [CrossRef]
- Meulenkamp, E.A. Mechanism of WO3 Electrodeposition from Peroxy-Tungstate Solution. J. Electrochem. Soc. 1997, 144, 1664–1671. [Google Scholar] [CrossRef]
- Breedon, M.; Spizzirri, P.; Taylor, M.; du Plessis, J.; McCulloch, D.; Zhu, J.; Yu, L.; Hu, Z.; Rix, C.; Wlodarski, W.; et al. Synthesis of Nanostructured Tungsten Oxide Thin Films: A Simple, Controllable, Inexpensive, Aqueous Sol−Gel Method. Cryst. Growth Des. 2010, 10, 430–439. [Google Scholar] [CrossRef]
- Yamada, Y.; Tabata, K.; Yashima, T. The character of WO3 film prepared with RF sputtering. Solar Ener. Mater. Solar Cells 2007, 91, 29–37. [Google Scholar] [CrossRef]
- Boyadjiev, S.I.; Georgieva, V.; Stefan, N.; Stan, G.E.; Mihailescu, N.; Visan, A.; Mihailescu, I.N.; Besleaga, C.; Szilágyi, I.M. Characterization of PLD grown WO3 thin films for gas sensing. Appl. Surf. Sci. 2017, 417, 218–223. [Google Scholar] [CrossRef]
- Li, S.; Yao, Z.; Zhou, J.; Zhang, R.; Shen, H. Fabrication and characterization of WO3 thin films on silicon surface by thermal evaporation. Mater. Lett. 2017, 195, 213–216. [Google Scholar] [CrossRef]
- Wang, C.-M.; Wen, C.-Y.; Chen, Y.-C.; Kao, K.-S.; Cheng, D.-L.; Peng, C.-H. Effect of Deposition Temperature on the Electrochromic Properties of Electron Beam-Evaporated WO3 Thin Films. Integr. Ferroelectr. 2014, 158, 62–68. [Google Scholar] [CrossRef]
- Blackman, I.C.S.; Parkin, P. Atmospheric Pressure Chemical Vapor Deposition of Crystalline Monoclinic WO3 and WO3-x Thin Films from Reaction of WCl6 with O-Containing Solvents and Their Photochromic and Electrochromic Properties. Chem. Mater. 2005, 17, 1583–1590. [Google Scholar] [CrossRef]
- Rogers, R.; Seddon, K.; Volkov, S. Green Industrial Applications of Ionic Liquids; Springer: Berlin/Heidelberg, Germany, 2002; Volume 818. [Google Scholar]
- Holbrey, J.D.; Rogers, R.D. Ionic Liquids in Synthesis; Wassercheid, P., Welton, T., Eds.; Wiley-VCH: Weinheim, Germany, 2008; Volume 1, pp. 57–174. [Google Scholar]
- Wang, B.; Qin, L.; Mu, T.; Xue, Z.; Gao, G. Are ionic liquids chemically stable? Chem. Rev. 2017, 117, 7113–7131. [Google Scholar] [CrossRef]
- Mahmood, H.; Moniruzzaman, M. Recent Advances of Using Ionic Liquids for Biopolymer Extraction and Processing. Biotechnol. J. 2019, 14, 1900072. [Google Scholar] [CrossRef]
- Verma, C.; Mishra, A.; Chauhan, S.; Verma, P.; Srivastava, V.; Quraishi, M.A.; Ebenso, E.E. Dissolution of cellulose in ionic liquids and their mixed cosolvents: A review. Sus. Chem. Pharm. 2019, 13, 100162. [Google Scholar] [CrossRef]
- Fang, Y.; Ma, P.; Cheng, H.; Tan, G.; Wu, J.; Zheng, J.; Zhou, X.; Fang, S.; Dai, Y.; Lin, Y. Synthesis of Low-Viscosity Ionic Liquids for Application in Dye-Sensitized Solar Cells. Chem. Asian J. 2019, 14, 4201–4206. [Google Scholar] [CrossRef] [PubMed]
- Lobregas, M.O.S.; Camacho, D.H. Gel polymer electrolyte system based on starch grafted with ionic liquid: Synthesis, characterization and its application in dye-sensitized solar cell. Electrochim. Acta 2019, 298, 219–228. [Google Scholar] [CrossRef]
- Dai, C.; Zhang, J.; Huang, C.; Lei, Z. Ionic Liquids in Selective Oxidation: Catalysts and Solvents. Chem. Rev. 2017, 117, 6929–6983. [Google Scholar] [CrossRef] [PubMed]
- Karimi, B.; Tavakolian, M.; Akbari, M.; Mansouri, F. Ionic Liquids in Asymmetric Synthesis: An Overall View from Reaction Media to Supported Ionic Liquid Catalysis. ChemCatChem 2018, 10, 3173–3205. [Google Scholar] [CrossRef]
- Kaur, G.; Sharma, A.; Banerjee, B. Ultrasound and Ionic Liquid: An Ideal Combination for Organic Transformations. ChemistrySelect 2018, 3, 5283–5295. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.; Motahharifar, N.; Sajjadi, M.; Aghbolagh, A.M.; Shokouhimehr, M.; Varma, R.S. Recent advances in N-formylation of amines and nitroarenes using efficient (nano)catalysts in eco-friendly media. Green Chem. 2019, 21, 5144–5167. [Google Scholar] [CrossRef]
- Sotgiu, G.; Chiarotto, I.; Feroci, M.; Orsini, M.; Rossi, L.; Inesi, A. An electrochemical alternative strategy to the synthesis of β-lactams. Part 3. Room-temperature ionic liquids vs molecular organic solvents. Electrochim. Acta 2008, 53, 7852–7858. [Google Scholar] [CrossRef]
- Feroci, M.; Chiarotto, I.; Inesi, A. Electrolysis of ionic liquids. A possible keystone for the achievement of green solvent-catalyst systems. Curr. Org. Chem. 2013, 17, 204–219. [Google Scholar] [CrossRef]
- D’Anna, F.; Marullo, S.; Vitale, P.; Noto, R. The Effect of the Cation π-Surface Area on the 3D Organization and Catalytic Ability of Imidazolium-Based Ionic Liquids. Eur. J. Org. Chem. 2011, 2011, 5681–5689. [Google Scholar] [CrossRef]
- D’Anna, F.; Marullo, S.; Noto, R. Aryl Azides Formation Under Mild Conditions: A Kinetic Study in Some Ionic Liquid Solutions. J. Org. Chem. 2010, 75, 767–771. [Google Scholar] [CrossRef]
- D’Anna, F.; Marullo, S.; Vitale, P.; Noto, R. Synthesis of aryl azides: A probe reaction to study the synergetic action of ultrasounds and ionic liquids. Ultrason. Sonochem. 2012, 19, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Marullo, S.; D’Anna, F.; Rizzo, C.; Noto, R. The ultrasounds–ionic liquids synergy on the copper catalyzed azide–alkyne cycloaddition between phenylacetylene and 4-azidoquinoline. Ultrason. Sonochem. 2015, 23, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, C.; D’Anna, F.; Marullo, S.; Noto, R. Task Specific Dicationic Ionic Liquids: Recyclable Reaction Media for the Mononuclear Rearrangement of Heterocycles. J. Org. Chem. 2014, 79, 8678–8683. [Google Scholar] [CrossRef] [PubMed]
- Pandolfi, F.; Feroci, M.; Chiarotto, I. Role of anion and cation in the 1-methyl-3-butylimidazolium ionic liquids BMImX: The Knoevenagel condensation. ChemistrySelect 2018, 3, 4745–4749. [Google Scholar] [CrossRef]
- Pandolfi, F.; Chiarotto, I.; Mattiello, L.; Petrucci, R.; Feroci, M. Two different selective ways in the deprotonation of β-bromopropionanilides: β-lactams or acrylanilides formation. ChemistrySelect 2019, 4, 12871–12874. [Google Scholar] [CrossRef]
- Chiarotto, I.; Feeney, M.M.M.; Feroci, M.; Inesi, A. Electrogenerated N-heterocyclic carbene: N-acylation of chiral oxazolidin-2-ones in ionic liquids. Electrochim. Acta 2009, 54, 1638–1644. [Google Scholar] [CrossRef]
- Chiarotto, I.; Feroci, M.; Orsini, M.; Sotgiu, G.; Inesi, A. Electrogenerated N-heterocyclic carbene: N-functionalization of benzoxazolones. Tetrahedron 2009, 65, 3704–3710. [Google Scholar] [CrossRef]
- Feroci, M.; Elinson, M.N.; Rossi, L.; Inesi, A. The double role of ionic liquids in organic electrosynthesis: Precursors of N-heterocyclic carbenes and green solvents. Henry reaction. Electrochem. Commun. 2009, 11, 1523–1526. [Google Scholar] [CrossRef]
- Feroci, M.; Chiarotto, I.; Vecchio Ciprioti, S.; Inesi, A. On the reactivity and stability of electrogenerated N-heterocyclic carbene in parent 1-butyl-3-methylimidazolium tetrafluoroborate: Formation and use of N-heterocyclic carbene-CO2 adduct as latent catalyst. Electrochim. Acta 2013, 109, 95–101. [Google Scholar] [CrossRef]
- Feroci, M.; Chiarotto, I.; D’Anna, F.; Gala, F.; Noto, R.; Ornano, L.; Zollo, G.; Inesi, A. N-Heterocyclic carbenes and parent cations: Acidity, nucleophilicity, stability, and hydrogen bonding-electrochemical study and ab initio calculations. ChemElectroChem 2016, 3, 1133–1141. [Google Scholar] [CrossRef]
- Chiarotto, I.; Mattiello, L.; Pandolfi, F.; Rocco, D.; Feroci, M. NHC in imidazolium acetate ionic liquids: Actual or potential presence? Front. Chem. 2018, 6, 355. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.I.M.; Awwad, F.; Greish, Y.E.; Abu-Hani, A.F.S.; Mahmoud, S.T. Fabrication of low temperature and fast response H2S gas sensor based on organic-metal oxide hybrid nanocomposite membrane. Org. Electron. 2020, 76, 105486. [Google Scholar] [CrossRef]
- Bahadori, M.; Tangestaninejad, S.; Bertmer, M.; Moghadam, M.; Mirkhani, V.; Mohammadpoor−Baltork, I.; Kardanpour, R.; Zadehahmadi, F. Task-Specific Ionic Liquid Functionalized–MIL–101(Cr) as a Heterogeneous and Efficient Catalyst for the Cycloaddition of CO2 with Epoxides Under Solvent Free Conditions. ACS Sustain. Chem. Eng. 2019, 7, 3962–3973. [Google Scholar] [CrossRef]
- Eyckens, D.J.; Champion, M.E.; Fox, B.L.; Yoganantharajah, P.; Gibert, Y.; Welton, T.; Henderson, L.C. Solvate Ionic Liquids as Reaction Media for Electrocyclic Transformations. Eur. J. Org. Chem. 2016, 2016, 913–917. [Google Scholar] [CrossRef]
- Eyckens, D.J.; Demir, B.; Walsh, T.R.; Welton, T.; Henderson, L.C. Determination of Kamlet–Taft parameters for selected solvate ionic liquids. Phys. Chem. Chem. Phys. 2016, 18, 13153–13157. [Google Scholar] [CrossRef]
- Lui, M.Y.; Crowhurst, L.; Hallett, J.P.; Hunt, P.A.; Niedermeyer, H.; Welton, T. Salts dissolved in salts: Ionic liquid mixtures. Chem. Sci. 2011, 2, 1491–1496. [Google Scholar] [CrossRef]
- Dupont, J. From Molten Salts to Ionic Liquids: A “Nano” Journey. Acc. Chem. Res. 2011, 44, 1223–1231. [Google Scholar] [CrossRef]
- Dupont, J.; Suarez, P.A.Z. Physico-chemical processes in imidazolium ionic liquids. Phys. Chem. Chem. Phys. 2006, 8, 2441–2452. [Google Scholar] [CrossRef]
- Carvalho, T.O.; Carvalho, P.H.P.R.; Correa, J.R.; Guido, B.C.; Medeiros, G.A.; Eberlin, M.N.; Coelho, S.E.; Domingos, J.B.; Neto, B.A.D. Palladium Catalyst with Task-Specific Ionic Liquid Ligands: Intracellular Reactions and Mitochondrial Imaging with Benzothiadiazole Derivatives. J. Org. Chem. 2019, 84, 5118–5128. [Google Scholar] [CrossRef]
- Qian, W.; Tan, X.; Su, Q.; Cheng, W.; Xu, F.; Dong, L.; Zhang, S. Transesterification of Isosorbide with Dimethyl Carbonate Catalyzed by Task-Specific Ionic Liquids. ChemSusChem 2019, 12, 1169–1178. [Google Scholar] [CrossRef]
- Xie, W.; Shao, L. Phosphorylation of Corn Starch in an Ionic Liquid. Starch/Stärke 2009, 61, 702–708. [Google Scholar] [CrossRef]
- El Sayed, S.; Bordet, A.; Weidenthaler, C.; Hetaba, W.; Luska, K.L.; Leitner, W. Selective Hydrogenation of Benzofurans Using Ruthenium Nanoparticles in Lewis Acid-Modified Ruthenium-Supported Ionic Liquid Phases. ACS Catal. 2020, 10, 2124–2130. [Google Scholar] [CrossRef]
- Fatehi, A.; Ghorbani-Vaghei, R.; Alavinia, S.; Mahmoodi, J. Synthesis of Quinazoline Derivatives Catalyzed by a New Efficient Reusable Nanomagnetic Catalyst Supported with Functionalized Piperidinium Benzene-1,3-Disulfonate Ionic Liquid. ChemistrySelect 2020, 5, 944–951. [Google Scholar] [CrossRef]
- Zhao, Q.; Yang, C.; Fang, M.; Jiang, T. Performance of Brönsted-Lewis acidic ionic liquids supported Ti-SBA-15 for the esterification of acetic acid to benzyl alcohol. Appl. Catal. A General 2020, 594, 117470. [Google Scholar] [CrossRef]
- Billeci, F.; D’Anna, F.; Gunaratne, H.Q.N.; Plechkova, N.V.; Seddon, K.R. “Sweet” ionic liquid gels: Materials for sweetening of fuels. Green Chem. 2018, 20, 4260–4276. [Google Scholar] [CrossRef]
- Guo, P.; Su, A.; Wei, Y.; Liu, X.; Li, Y.; Guo, F.; Li, J.; Hu, Z.; Sun, J. Healable, Highly Conductive, Flexible, and Nonflammable Supramolecular Ionogel Electrolytes for Lithium-Ion Batteries. ACS Appl. Mater. Interfaces 2019, 11, 19413–19420. [Google Scholar] [CrossRef] [PubMed]
- Kuddushi, M.; Mata, J.; Malek, N. Self-Sustainable, self-healable, Load Bearable and Moldable stimuli responsive ionogel for the Selective Removal of Anionic Dyes from aqueous medium. J. Mol. Liq. 2020, 298, 112048. [Google Scholar] [CrossRef]
- Marullo, S.; Rizzo, C.; Dintcheva, N.T.; Giannici, F.; D’Anna, F. Ionic liquids gels: Soft materials for environmental remediation. J. Colloid Interface Sci. 2018, 517, 182–193. [Google Scholar] [CrossRef]
- Rizzo, C.; Marullo, S.; Campodonico, P.R.; Pibiri, I.; Dintcheva, N.T.; Noto, R.; Millan, D.; D’Anna, F. Self-Sustaining Supramolecular Ionic Liquid Gels for Dye Adsorption. ACS Sustain. Chem. Eng. 2018, 6, 12453–12462. [Google Scholar] [CrossRef]
- Billeci, F.; D’Anna, F.; Feroci, M.; Cancemi, P.; Feo, S.; Forlino, A.; Tonnelli, F.; Seddon, K.R.; Gunaratne, H.Q.N.; Plechkova, N.V. When Functionalization Becomes Useful: Ionic Liquids with a “Sweet” Appended Moiety Demonstrate Drastically Reduced Toxicological Effects. ACS Sustain. Chem. Eng. 2020, 8, 926–938. [Google Scholar] [CrossRef]
- Egorova, K.S.; Ananikov, V.P. Toxicity of Ionic Liquids: Eco(cyto)activity as Complicated, but Unavoidable Parameter for Task-Specific Optimization. ChemSusChem 2014, 7, 336–360. [Google Scholar] [CrossRef] [PubMed]
- Hulsbosch, J.; De Vos, D.E.; Binnemans, K.; Ameloot, R. Biobased Ionic Liquids: Solvents for a Green Processing Industry? ACS Sustain. Chem. Eng. 2016, 4, 2917–2931. [Google Scholar] [CrossRef]
- Socha, A.M.; Parthasarathi, R.; Shi, J.; Pattathil, S.; Whyte, D.; Bergeron, M.; George, A.; Tran, K.; Stavila, V.; Venkatachalam, S.; et al. Efficient biomass pretreatment using ionic liquids derived from lignin and hemicellulose. Proc. Natl. Acad. Sci. USA 2014, 111, E3587–E3595. [Google Scholar] [CrossRef] [PubMed]
- Worlds Health Organization (WHO). Health Topics. Available online: https://www.who.int/health-topics/air-pollution#tab=tab_1 (accessed on 14 February 2020).
- Worlds Health Organization (WHO). Available online: https://www.who.int/airpollution/ambient/pollutants/en/) (accessed on 14 February 2020).
- Duk-Dong, L.; Dae-Sik, L. Environmental gas sensors. IEEE Sensors J. 2001, 1, 214–224. [Google Scholar] [CrossRef]
- Rai, A.C.; Kumar, P.; Pilla, F.; Skouloudis, A.N.; Di Sabatino, S.; Ratti, C.; Yasar, A.; Rickerby, D. End-user perspective of low-cost sensors for outdoor air pollution monitoring. Sci. Total Environ. 2017, 607–608, 691–705. [Google Scholar] [CrossRef]
- Pang, X.; Shaw, M.D.; Gillot, S.; Lewis, A.C. The impacts of water vapour and co-pollutants on the performance of electrochemical gas sensors used for air quality monitoring. Sens. Actuators B Chem. 2018, 266, 674–684. [Google Scholar] [CrossRef]
- Moseley, P.T. Progress in the development of semiconducting metal oxide gas sensors: A review. Meas. Sci. Technol. 2017, 28, 082001. [Google Scholar] [CrossRef]
- Dong, C.; Zhao, R.; Yao, L.; Ran, Y.; Zhang, X.; Wang, Y. A review on WO3 based gas sensors: Morphology control and enhanced sensing properties. J. Alloys Compd. 2020, 820, 153194. [Google Scholar] [CrossRef]
- Hariharan, V.; Gnanavel, B.; Sathiyapriya, R.; Aroulmoji, V.A. A Review on Tungsten Oxide (WO3) and their Derivatives for Sensor Applications. Int. J. Adv. Sci. Eng. 2019, 5, 1163–1168. [Google Scholar] [CrossRef]
- Sari, W.P.; Leigh, S.; Covington, J. Tungsten Oxide Based Sensor for Oxygen Detection. Proceedings 2018, 2, 952. [Google Scholar] [CrossRef]
- Staerz, A.; Somacescu, S.; Epifani, M.; Russ, T.; Weimar, U.; Barsan, N. WO3 Based Gas Sensors. Proceedings 2018, 2, 826. [Google Scholar] [CrossRef]
- Li, Z.; Li, J.; Song, L.; Gong, H.; Niu, Q. Ionic liquid-assisted synthesis of WO3 particles with enhanced gas sensing properties. J. Mat. Chem. A 2013, 1, 15377–15382. [Google Scholar] [CrossRef]
- Zhang, Y.; Cheng, X.; Zhang, X.; Major, Z.; Xu, Y.; Gao, S.; Zhao, H.; Huo, L. Ionic liquid-assisted synthesis of tungsten oxide nanoparticles with enhanced NO2 sensing properties at near room temperature. Appl. Surf. Sci. 2020, 505, 144533. [Google Scholar] [CrossRef]
- Favard, A.Y.X.; Anguille, S.; Moulin, P.; Seguin, J.-L.; Aguir, K.; Bendahan, M. Ionic Liquids Filter for Humidity Effect Reduction on Metal Oxide Gas Sensor Response. Sensors Transduc. 2018, 222, 6–11. [Google Scholar]
- Abu-Hani, A.F.S.; Awwad, F.; Greish, Y.E.; Ayesh, A.I.; Mahmoud, S.T. Design, fabrication, and characterization of low-power gas sensors based on organic-inorganic nano-composite. Org. Electron. 2017, 42, 284–292. [Google Scholar] [CrossRef]
- Daves, W.; Ersoez, B. Electrochemical Gas Sensor. International Patent Application No. WO2018/234185 A1, 27 December 2018. [Google Scholar]
- Bhutto, A.W.; Abro, R.; Gao, S.; Abbas, T.; Chen, X.; Yu, G. Oxidative desulfurization of fuel oils using ionic liquids: A review. J. Taiwan Inst. Chem. Eng. 2016, 62, 84–97. [Google Scholar] [CrossRef]
- Daage, M.; Chianelli, R.R. Structure-Function Relations in Molybdenum Sulfide Catalysts: The “Rim-Edge” Model. J. Catal. 1994, 149, 414–427. [Google Scholar] [CrossRef]
- Topsøe, H. The role of Co–Mo–S type structures in hydrotreating catalysts. Appl. Catal. A General 2007, 322, 3–8. [Google Scholar] [CrossRef]
- Shan, S.; Yuan, P.; Han, W.; Shi, G.; Bao, X. Supported NiW catalysts with tunable size and morphology of active phases for highly selective hydrodesulfurization of fluid catalytic cracking naphtha. J. Catal. 2015, 330, 288–301. [Google Scholar] [CrossRef]
- Hossain, M.N.; Park, H.C.; Choi, H.S. A Comprehensive Review on Catalytic Oxidative Desulfurization of Liquid Fuel Oil. Catalyst 2019, 9, 12. [Google Scholar] [CrossRef]
- Sun, H.; Wu, P.; He, J.; Liu, M.; Zhu, L.; Zhu, F.; Chen, G.; He, M.; Zhu, W. Fabrication of oxygen-defective tungsten oxide nanorods for deep oxidative desulfurization of fuel. Pet. Sci. 2018, 15, 849–856. [Google Scholar] [CrossRef]
- Rongxiang, Z.; Xiuping, L.; Jianxun, S.; Weiwei, S.; Xiaohan, G. Preparation of WO3/C Composite and Its Application in Oxidative Desulfurization of Fuel. China Pet. Process. Petrochem. Technol. 2017, 19, 65–73. [Google Scholar]
- Li, X.; Zhao, R.; Mao, C. Polycrystalline Phase WO3/g-C3N4 as a High Efficient Catalyst for Removal of DBT in Model Oil. China Pet. Process. Petrochem. Technol. 2019, 21, 36–45. [Google Scholar]
- Zhang, M.; Zhu, W.; Li, H.; Xun, S.; Li, M.; Li, Y.; Wei, Y.; Li, H. Fabrication and characterization of tungsten-containing mesoporous silica for heterogeneous oxidative desulfurization. Chin. J. Catal. 2016, 37, 971–978. [Google Scholar] [CrossRef]
- Xun, S.; Hou, C.; Li, H.; He, M.; Ma, R.; Zhang, M.; Zhu, W.; Li, H. Synthesis of WO3/mesoporous ZrO2 catalyst as a high-efficiency catalyst for catalytic oxidation of dibenzothiophene in diesel. J. Mat. Sci. 2018, 53, 15927–15938. [Google Scholar] [CrossRef]
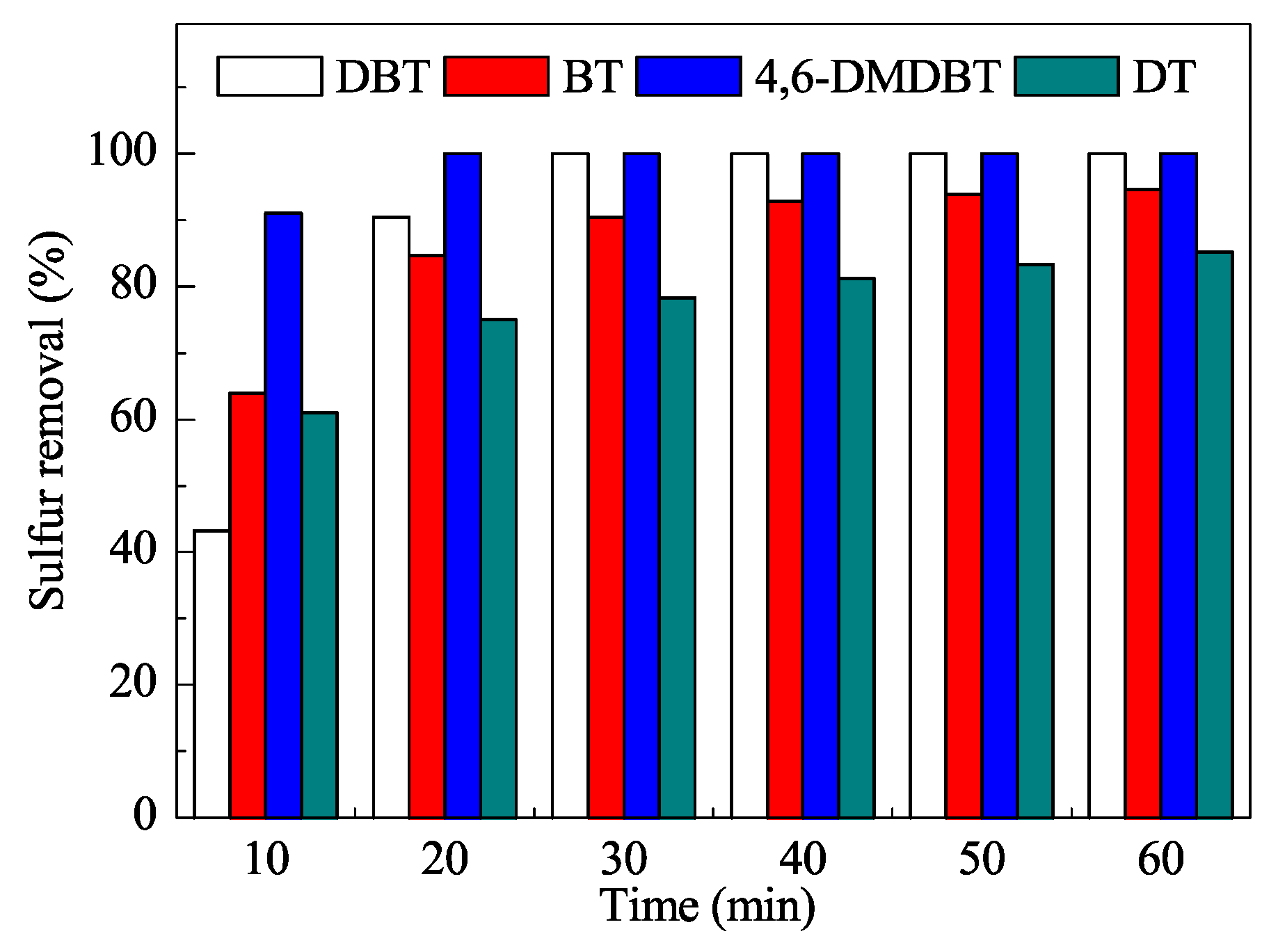
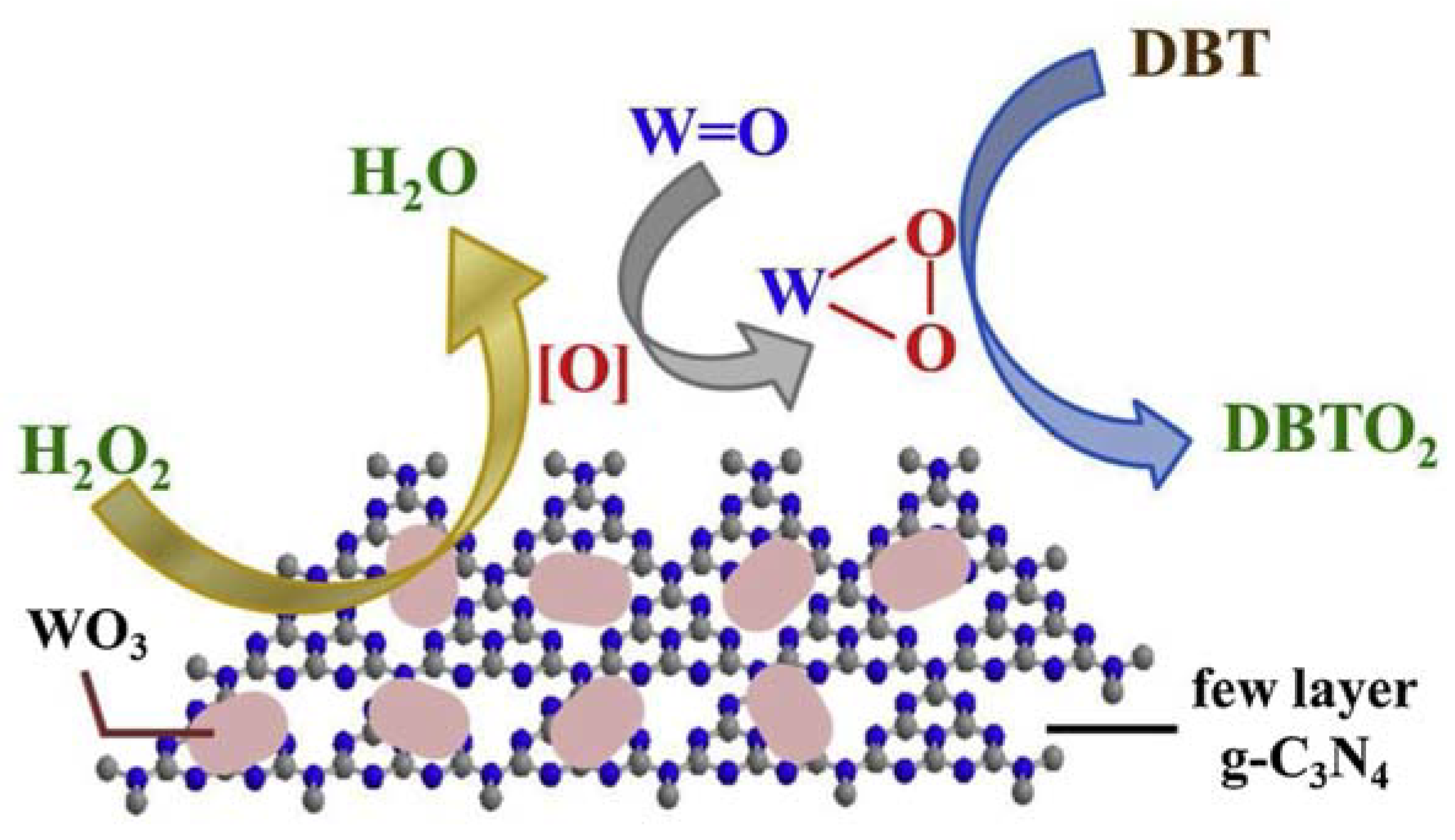
- Ma, R.; Guo, J.; Wang, D.; He, M.; Xun, S.; Gu, J.; Zhu, W.; Li, H. Preparation of highly dispersed WO3/few layer g-C3N4 and its enhancement of catalytic oxidative desulfurization activity. Colloid. Surface. A: Physicochem. Eng. Aspects 2019, 572, 250–258. [Google Scholar] [CrossRef]
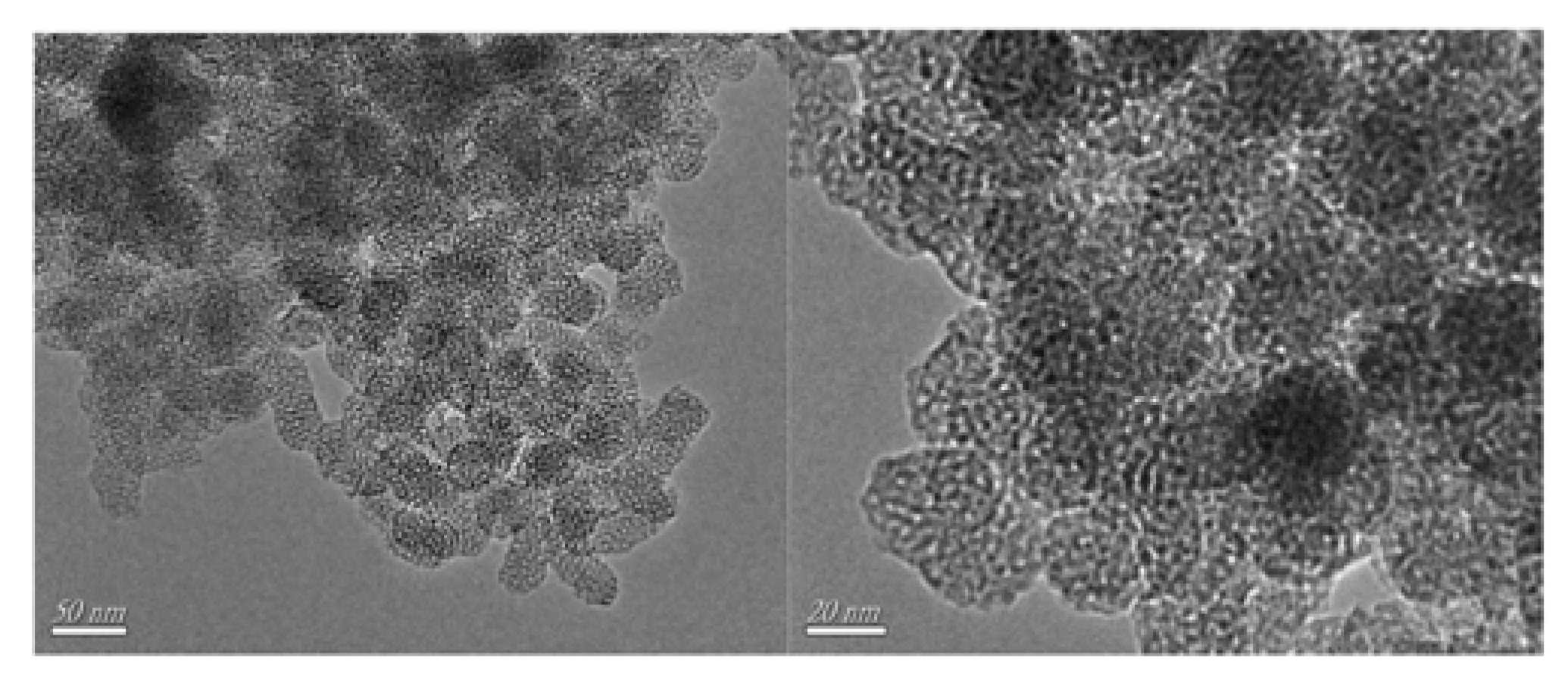
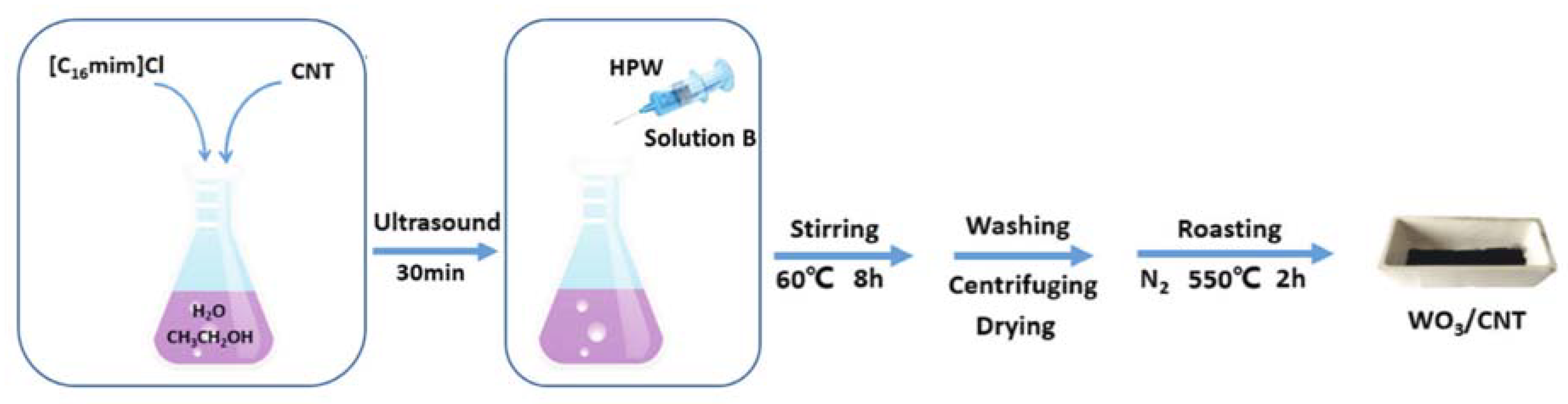
- Wang, C.; Li, A.; Xu, J.; Wen, J.; Zhang, H.; Zhang, L. Preparation of WO3/CNT catalysts in presence of ionic liquid [C16mim]Cl and catalytic efficiency in oxidative desulfurization. J. Chem. Technol. Biotechnol. 2019, 94, 3403–3412. [Google Scholar] [CrossRef]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Anna, F.; Grilli, M.L.; Petrucci, R.; Feroci, M. WO3 and Ionic Liquids: A Synergic Pair for Pollutant Gas Sensing and Desulfurization. Metals 2020, 10, 475. https://doi.org/10.3390/met10040475
D’Anna F, Grilli ML, Petrucci R, Feroci M. WO3 and Ionic Liquids: A Synergic Pair for Pollutant Gas Sensing and Desulfurization. Metals. 2020; 10(4):475. https://doi.org/10.3390/met10040475
Chicago/Turabian StyleD’Anna, Francesca, Maria Luisa Grilli, Rita Petrucci, and Marta Feroci. 2020. "WO3 and Ionic Liquids: A Synergic Pair for Pollutant Gas Sensing and Desulfurization" Metals 10, no. 4: 475. https://doi.org/10.3390/met10040475
APA StyleD’Anna, F., Grilli, M. L., Petrucci, R., & Feroci, M. (2020). WO3 and Ionic Liquids: A Synergic Pair for Pollutant Gas Sensing and Desulfurization. Metals, 10(4), 475. https://doi.org/10.3390/met10040475







