Depression of Pyrite in Seawater Flotation by Guar Gum
Abstract
1. Introduction
2. Methodology
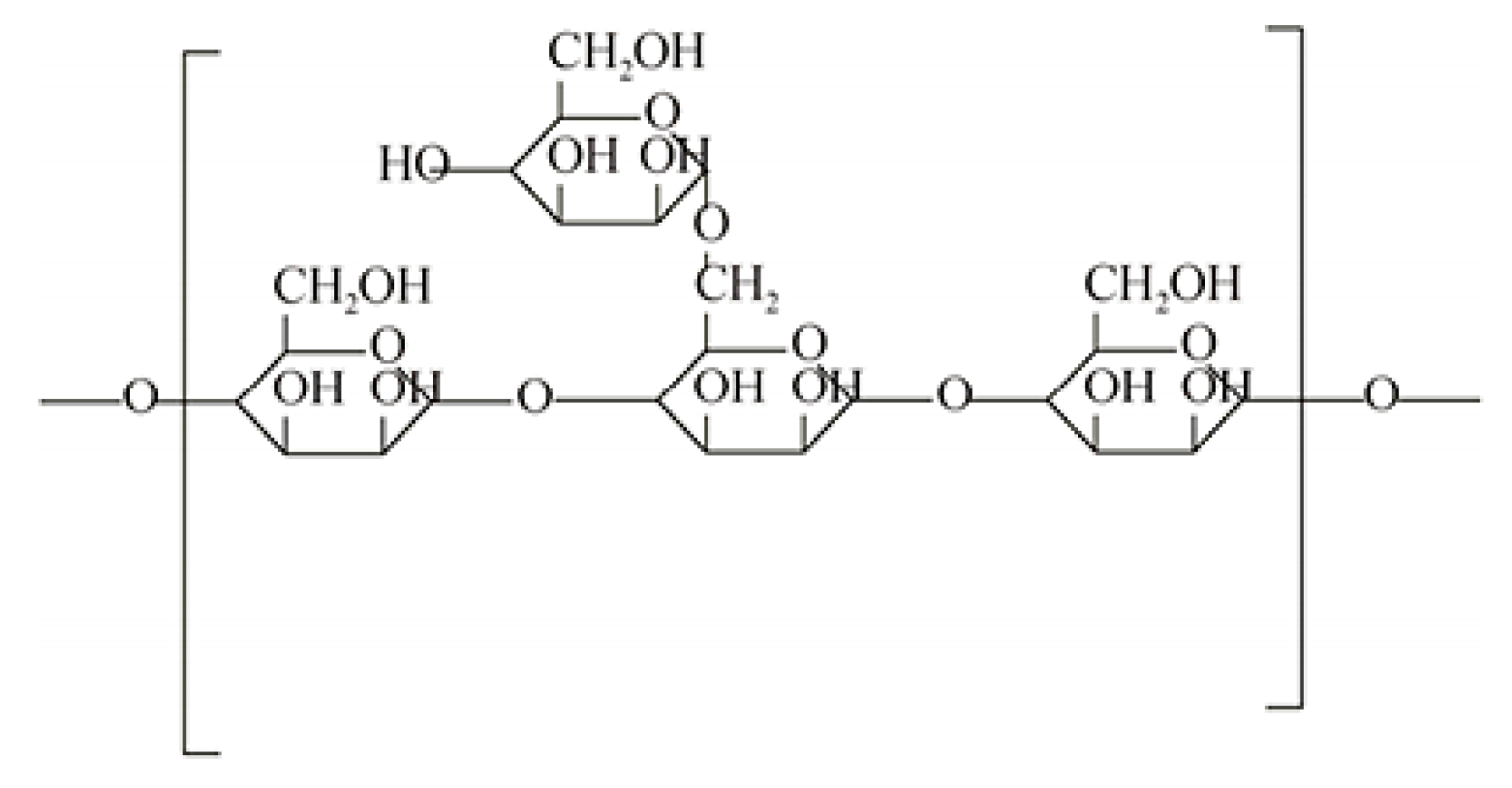
2.1. Materials
2.2. Preparation of Flotation Reagents
2.3. Microflotation Tests
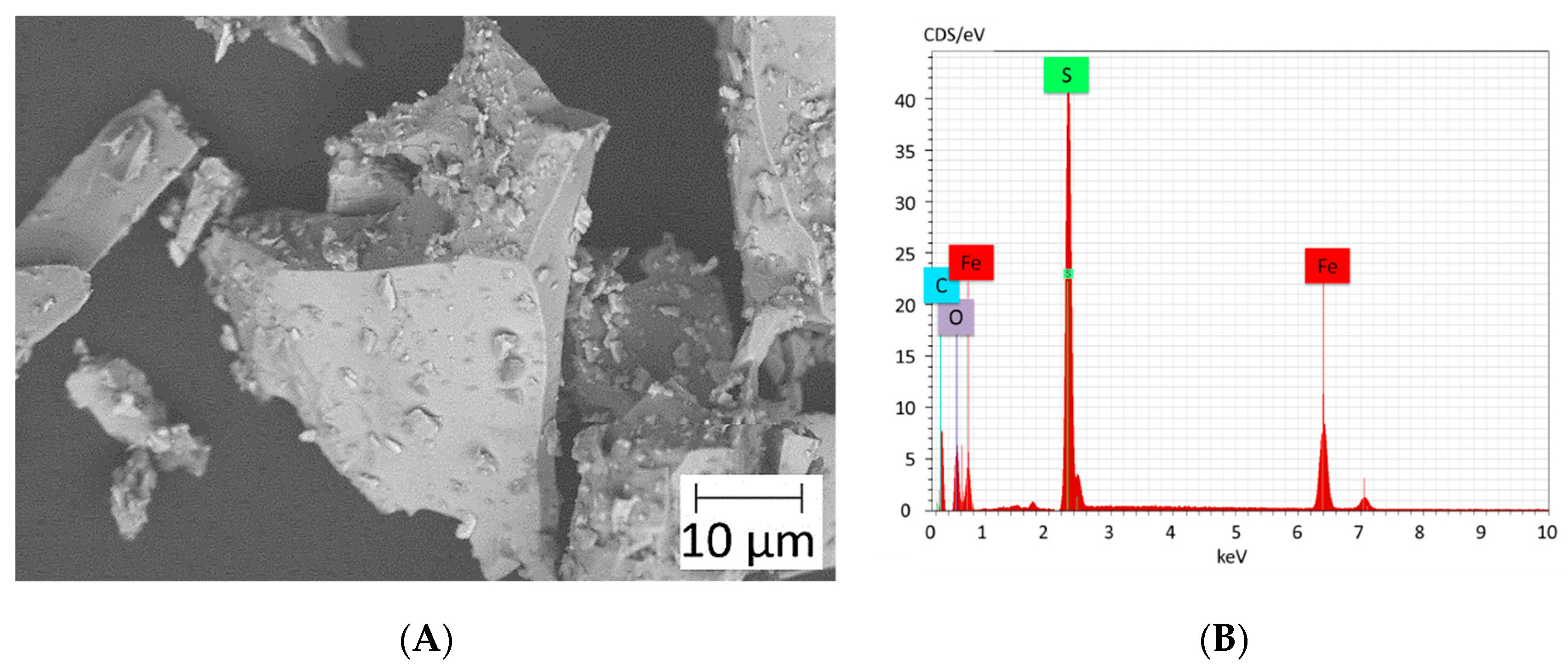
2.4. Aggregate Characterization
2.5. Fourier Transform Infrared (FTIR) Spectroscopy
2.6. Surface Tension
3. Results and Discussions
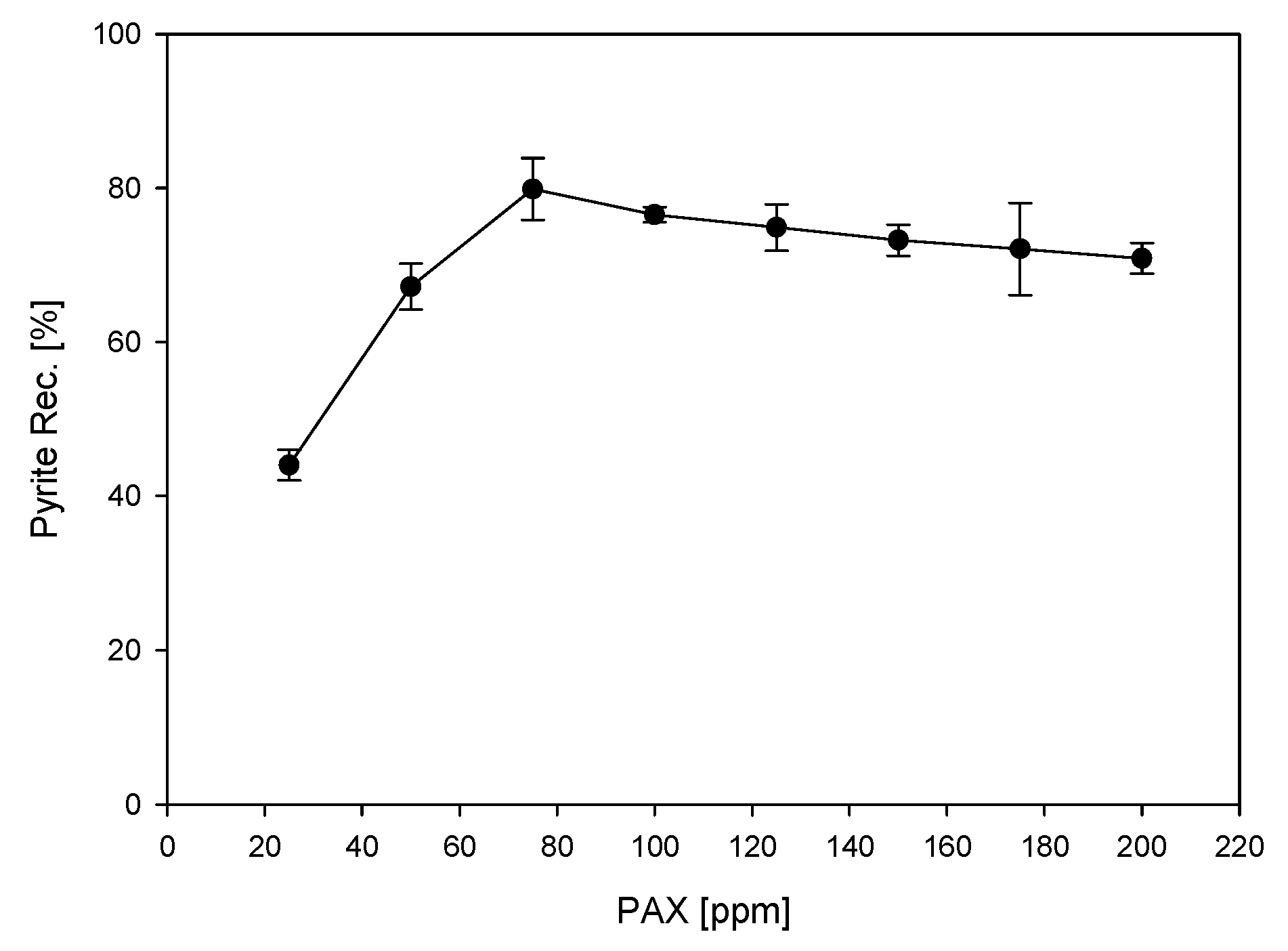
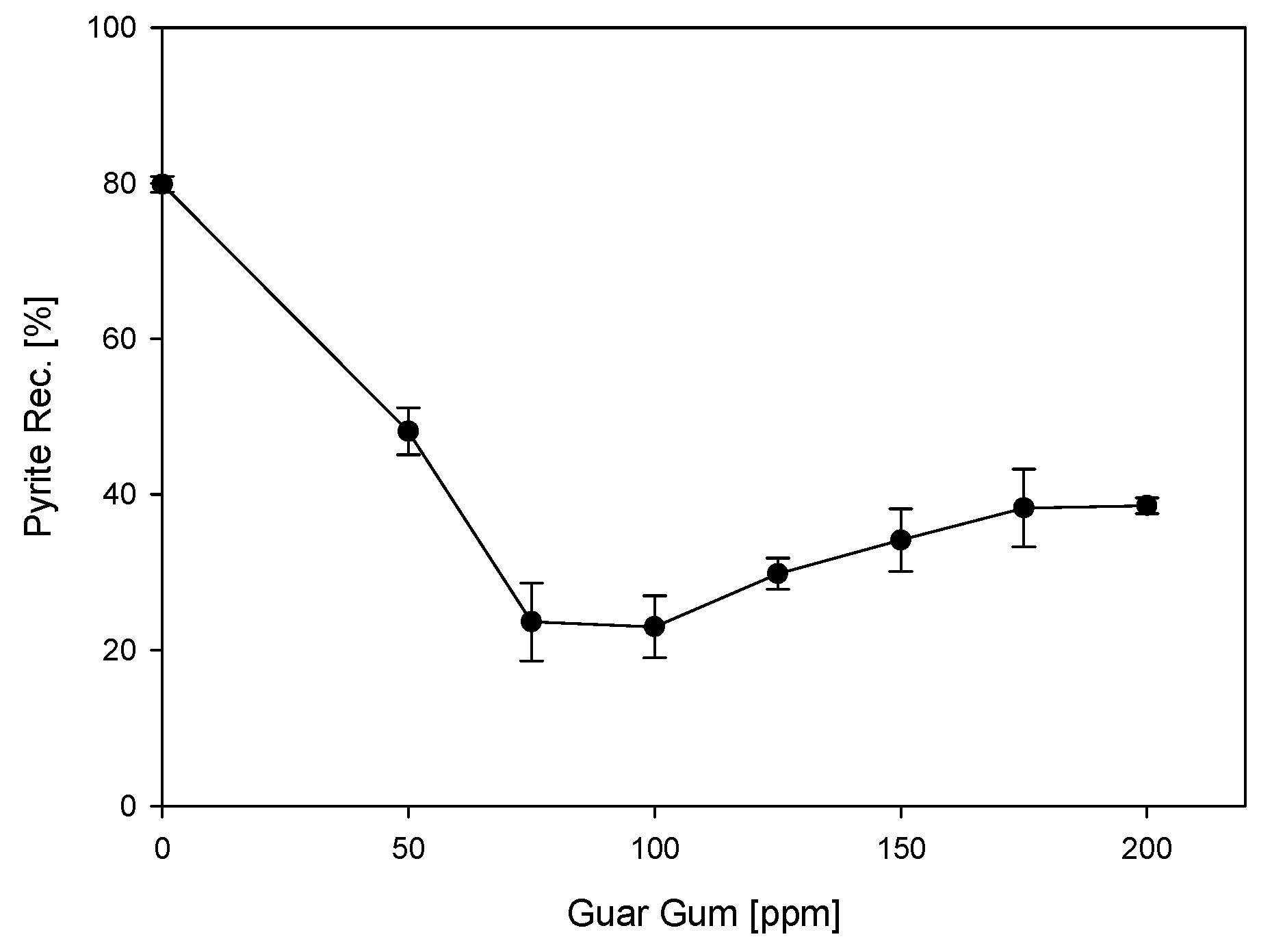
3.1. Microflotations
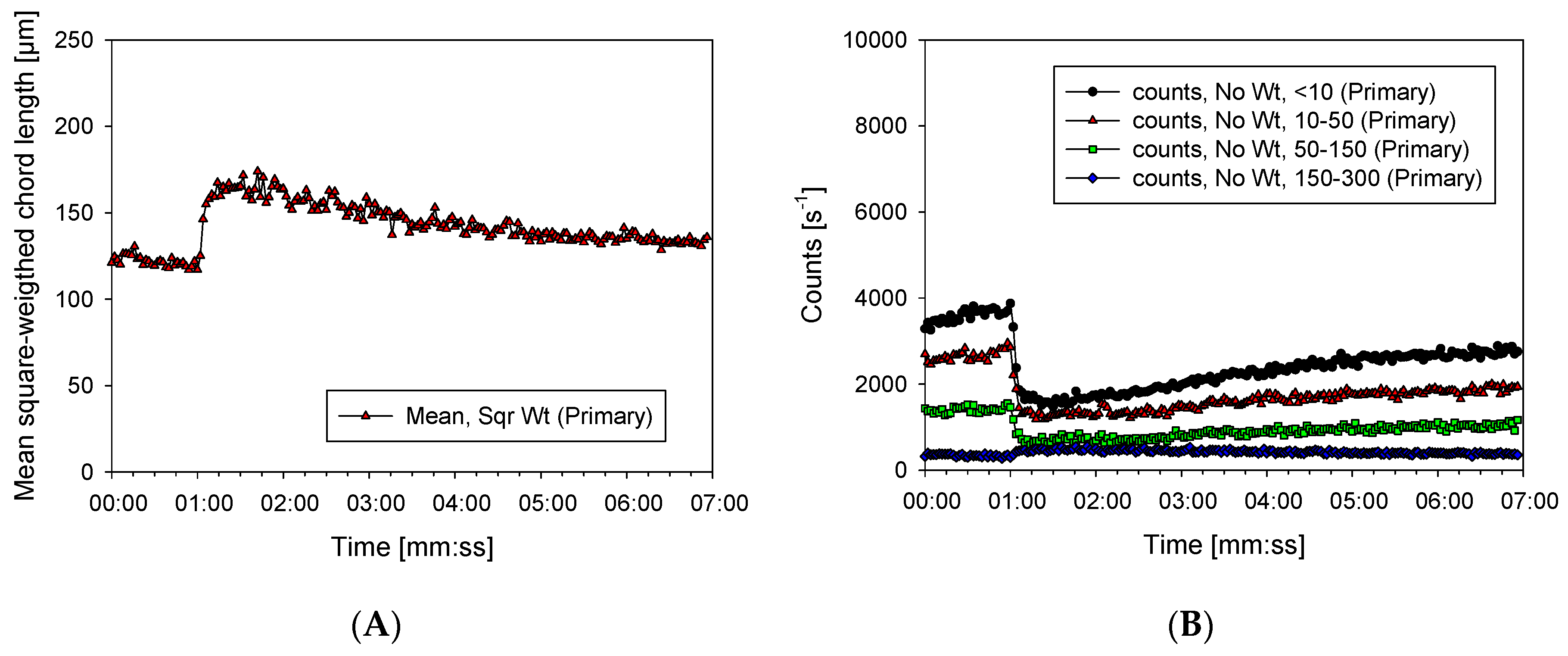
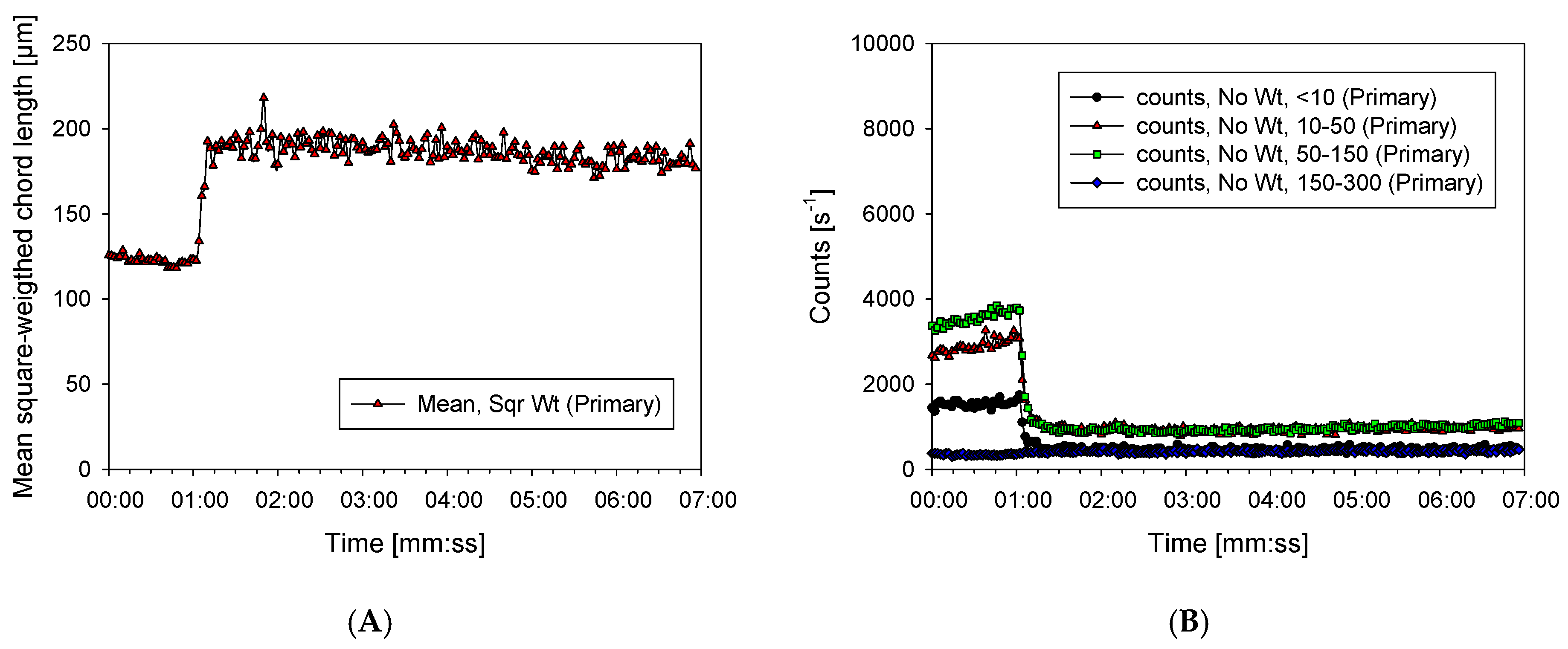
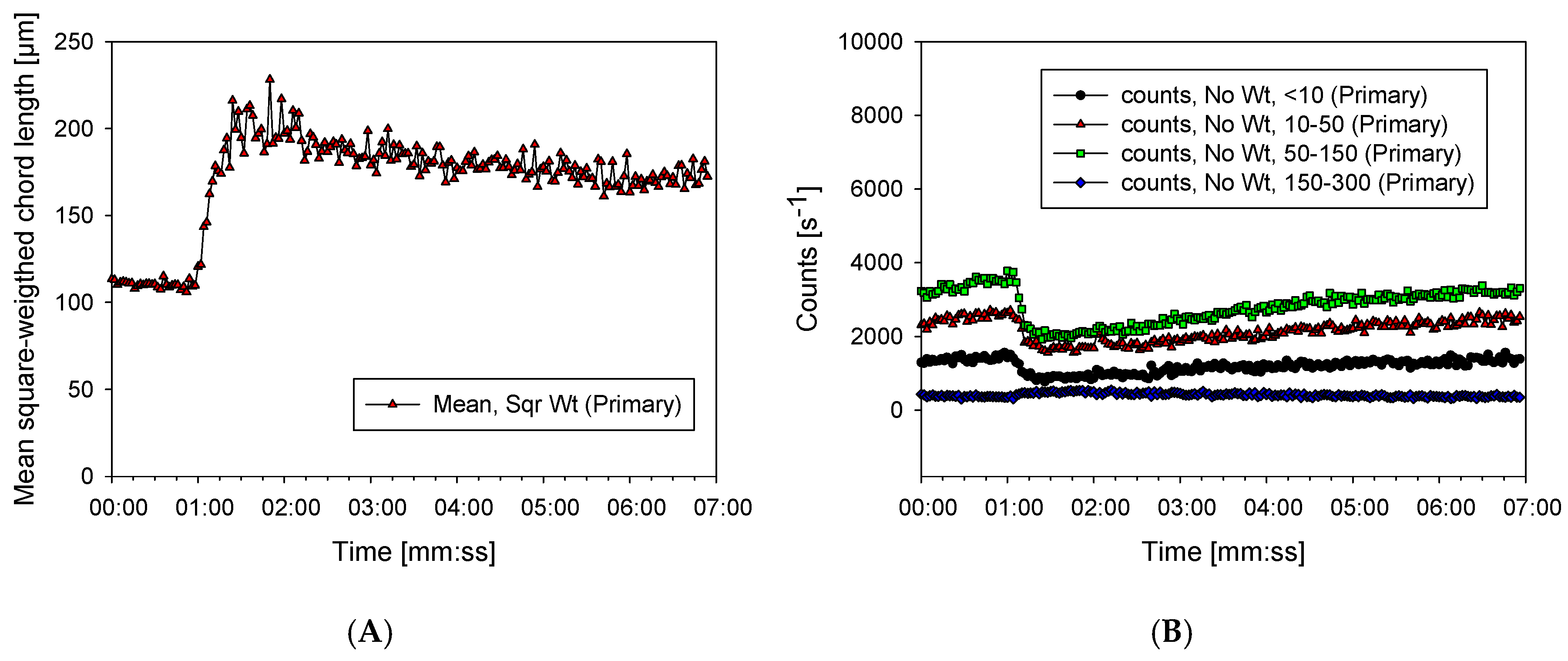
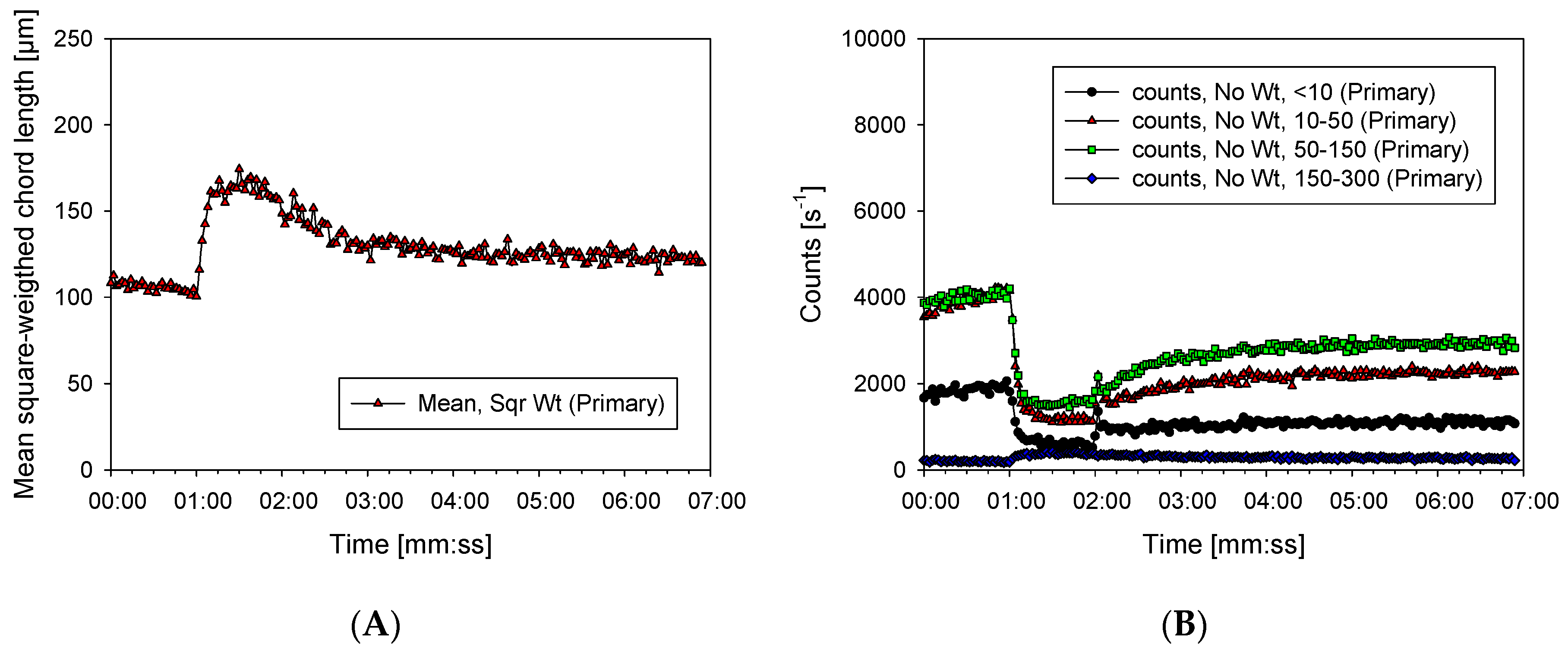
3.2. Characterization of Aggregates
3.3. FTIR Analysis
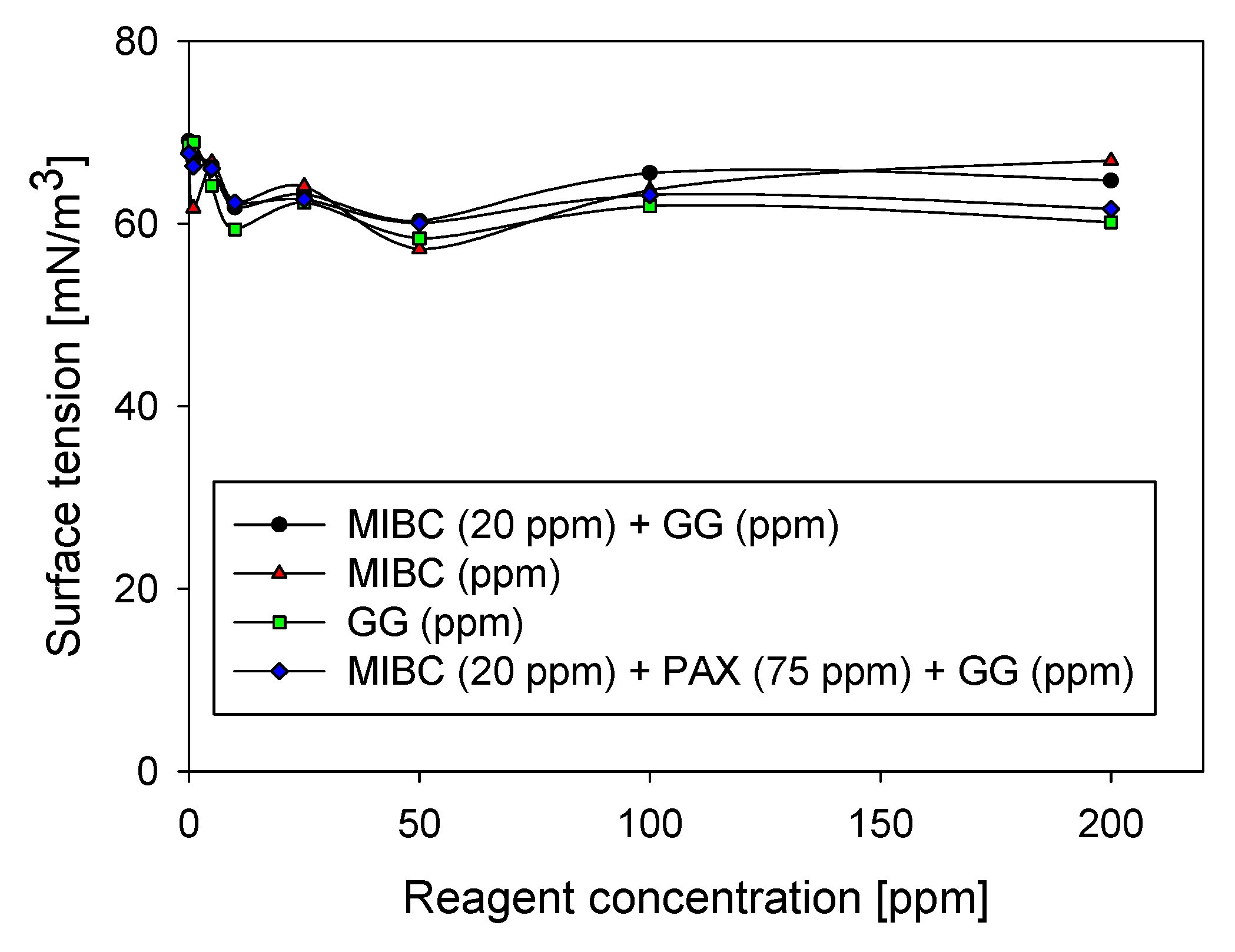
3.4. Surface Tension
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, X.-H.; Eric Forssberg, K.S. Mechanisms of pyrite flotation with xanthates. Int. J. Miner. Process. 1991, 33, 275–290. [Google Scholar] [CrossRef]
- Mu, Y.; Peng, Y.; Lauten, R.A. The depression of pyrite in selective flotation by different reagent systems—A Literature review. Miner. Eng. 2016, 96–97, 143–156. [Google Scholar] [CrossRef]
- Boulton, A.; Fornasiero, D.; Ralston, J. Depression of iron sulphide flotation in zinc roughers. Miner. Eng. 2001, 14, 1067–1079. [Google Scholar] [CrossRef]
- Jiang, C.L.; Wang, X.H.; Parekh, B.K.; Leonard, J.W. The surface and solution chemistry of pyrite flotation with xanthate in the presence of iron ions. Colloids Surf. A 1998, 136, 51–62. [Google Scholar] [CrossRef]
- Trahar, W.J.; Senior, G.D.; Shannon, L.K. Interactions between sulphide minerals—The collectorless flotation of pyrite. Int. J. Miner. Process. 1994, 40, 287–321. [Google Scholar] [CrossRef]
- Chandra, A.P.; Gerson, A.R. The mechanisms of pyrite oxidation and leaching: A fundamental perspective. Surf. Sci. Rep. 2010, 65, 293–315. [Google Scholar] [CrossRef]
- Karthe, S.; Szargan, R.; Suoninen, E. Oxidation of pyrite surfaces: a photoelectron spectroscopic study. Appl. Surf. Sci. 1993, 72, 157–170. [Google Scholar] [CrossRef]
- Moslemi, H.; Gharabaghi, M. A review on electrochemical behavior of pyrite in the froth flotation process. J. Ind. Eng. Chem. 2017, 47, 1–18. [Google Scholar] [CrossRef]
- Hicyilmaz, C.; Emre Altun, N.; Ekmekci, Z.; Gokagac, G. Quantifying hydrophobicity of pyrite after copper activation and DTPI addition under electrochemically controlled conditions. Miner. Eng. 2004, 17, 879–890. [Google Scholar] [CrossRef]
- Kocabag, D.; Shergold, H.L.; Kelsall, G.H. Natural oleophilicity/hydrophobicity of sulphide minerals, II. Pyrite. Int. J. Miner. Process. 1990, 29, 211–219. [Google Scholar] [CrossRef]
- Zanin, M.; Lambert, H.; du Plessis, C.A. Lime use and functionality in sulphide mineral flotation: A review. Miner. Eng. 2019, 143, 105922. [Google Scholar] [CrossRef]
- Li, Y.; Chen, J.; Kang, D.; Guo, J. Depression of pyrite in alkaline medium and its subsequent activation by copper. Miner. Eng. 2012, 26, 64–69. [Google Scholar] [CrossRef]
- Cisternas, L.A.; Gálvez, E.D. The use of seawater in mining. Miner. Process. Extr. Metall. Rev. 2018, 39, 18–33. [Google Scholar] [CrossRef]
- Jeldres, R.I.; Calisaya, D.; Cisternas, L.A. An improved flotation test method and pyrite depression by an organic reagent during flotation in seawater. J. South. Afr. Inst. Min. Metall. 2017, 117, 499–504. [Google Scholar] [CrossRef]
- Castro, S. Challenges in flotation of Cu-Mo sulfide ores in sea water I. In Water in Mineral Processing; SME: Englewood, CO, USA, 2012; pp. 29–40. [Google Scholar]
- Jeldres, R.I.; Arancibia-Bravo, M.P.; Reyes, A.; Aguirre, C.E.; Cortes, L.; Cisternas, L.A. The impact of seawater with calcium and magnesium removal for the flotation of copper-molybdenum sulphide ores. Miner. Eng. 2017, 109, 10–13. [Google Scholar] [CrossRef]
- Li, W.; Li, Y.; Xiao, Q.; Wei, Z.; Song, S. The influencing mechanisms of sodium hexametaphosphate on chalcopyrite flotation in the presence of MgCl2 and CaCl2. Minerals 2018, 8, 150. [Google Scholar] [CrossRef]
- Li, Y.; Li, W.; Xiao, Q.; He, N.; Ren, Z.; Lartey, C.; Gerson, A. The influence of common monovalent and divalent chlorides on chalcopyrite flotation. Minerals 2017, 7, 111. [Google Scholar] [CrossRef]
- Suyantara, G.P.W.; Hirajima, T.; Miki, H.; Sasaki, K. Floatability of molybdenite and chalcopyrite in artificial seawater. Miner. Eng. 2018, 115, 117–130. [Google Scholar] [CrossRef]
- Cao, Z.; Chen, X.; Peng, Y. The role of sodium sulfide in the flotation of pyrite depressed in chalcopyrite flotation. Miner. Eng. 2018, 119, 93–98. [Google Scholar] [CrossRef]
- Göktepe, F. Effect of H2O2 and NaSH addition to change the electrochemical potential in flotation of chalcopyrite and pyrite minerals. Miner. Process. Extr. Metall. Rev. 2010, 32, 24–29. [Google Scholar] [CrossRef]
- Hassanzadeh, A.; Hasanzadeh, M. Chalcopyrite and pyrite floatabilities in the presence of sodium sulfide and sodium metabisulfite in a high pyritic copper complex ore. J. Dispers. Sci. Technol. 2017, 38, 782–788. [Google Scholar] [CrossRef]
- Khmeleva, T.N.; Skinner, W.; Beattie, D.A.; Georgiev, T.V. The effect of sulphite on the xanthate-induced flotation of copper-activated pyrite. Physicochem. Prob. Miner. Process. 2002, 36, 185–195. [Google Scholar]
- Kar, B.; Sahoo, H.; Rath, S.S.; Das, B. Investigations on different starches as depressants for iron ore flotation. Miner. Eng. 2013, 49, 1–6. [Google Scholar] [CrossRef]
- Lopez Valdivieso, A.; Sánchez López, A.A.; Song, S.; García Martínez, H.A.; Licón Almada, S. Dextrin as a regulator for the selective flotation of chalcopyrite, galena and pyrite. Can. Metall. Q. 2007, 46, 301–309. [Google Scholar] [CrossRef]
- Wang, Z.; Xie, X.; Xiao, S.; Liu, J. Adsorption behavior of glucose on pyrite surface investigated by TG, FTIR and XRD analyses. Hydrometallurgy 2010, 102, 87–90. [Google Scholar] [CrossRef]
- Sarquís, P.E.; Menéndez-Aguado, J.M.; Mahamud, M.M.; Dzioba, R. Tannins: the organic depressants alternative in selective flotation of sulfides. J. Clean. Prod. 2014, 84, 723–726. [Google Scholar] [CrossRef]
- Rutledge, J.; Anderson, C. Tannins in mineral processing and extractive metallurgy. Metals 2015, 5, 1520–1542. [Google Scholar] [CrossRef]
- Mu, Y.; Peng, Y.; Lauten, R.A. Electrochemistry aspects of pyrite in the presence of potassium amyl xanthate and a lignosulfonate-based biopolymer depressant. Electrochim. Acta 2015, 174, 133–142. [Google Scholar] [CrossRef]
- Lopez-Valdivieso, A.; Sanchez-López, A.A.; Padilla-Ortega, E.; Robledo-Cabrera, A.; Galvez, E.; Cisternas, L.A. Pyrite depression by dextrin in flotation with xanthates. Adsorption and floatability studies. Physicochem. Prob. Miner. Process. 2018, 54, 1159–1171. [Google Scholar] [CrossRef]
- Mathur, N.K. Industrial Galactomannan Polysaccharides; CRC Press: Boca Raton, FL, USA, 2016; ISBN 9780429105142. [Google Scholar]
- Laskowski, J.S.; Liu, Q.; O’Connor, C.T. Current understanding of the mechanism of polysaccharide adsorption at the mineral/aqueous solution interface. Int. J. Miner. Process. 2007, 84, 59–68. [Google Scholar] [CrossRef]
- Ma, X.; Pawlik, M. Adsorption of guar gum onto quartz from dilute mixed electrolyte solutions. J. Colloid Interface Sci. 2006, 298, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Pawlik, M. Role of background ions in guar gum adsorption on oxide minerals and kaolinite. J. Colloid Interface Sci. 2007, 313, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Mailula, T.D.; Bradshaw, D.J.; Harris, P.J. The effect of copper sulphate addition on the recovery of chromite in the flotation of UG2 ore. J. S. Afr. Inst. Min. Metall. 2003, 103. [Google Scholar]
- Shortridge, P.; Harris, P.; Bradshaw, D.; Koopal, L. The effect of chemical composition and molecular weight of polysaccharide depressants on the flotation of talc. Int. J. Miner. Process. 2000, 59, 215–224. [Google Scholar] [CrossRef]
- Bicak, O.; Ekmekci, Z.; Bradshaw, D.J.; Harris, P.J. Adsorption of guar gum and CMC on pyrite. Miner. Eng. 2007, 20, 996–1002. [Google Scholar] [CrossRef]
- Jeldres, M.; Piceros, E.C.; Toro, N.; Torres, D.; Robles, P.; Leiva, W.H.; Jeldres, R.I. Copper tailing flocculation in seawater: Relating the yield stress with fractal aggregates at varied mixing conditions. Metals 2019, 9, 1295. [Google Scholar] [CrossRef]
- Hu, Y.; Sun, W.; Wang, D. Electrochemistry of Flotation of Sulphide Minerals; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 978-3-540-92178-3. [Google Scholar]
- López Valdivieso, A.; Sánchez López, A.A.; Song, S. On the cathodic reaction coupled with the oxidation of xanthates at the pyrite/aqueous solution interface. Int. J. Miner. Process. 2005, 77, 154–164. [Google Scholar] [CrossRef]
- López Valdivieso, A.; Celedón Cervantes, T.; Song, S.; Robledo Cabrera, A.; Laskowski, J. Dextrin as a non-toxic depressant for pyrite in flotation with xanthates as collector. Miner. Eng. 2004, 17, 1001–1006. [Google Scholar] [CrossRef]
- Rath, R.K.; Subramanian, S.; Pradeep, T. Surface chemical studies on pyrite in the presence of polysaccharide-based flotation depressants. J. Colloid Interface Sci. 2000, 229, 82–91. [Google Scholar] [CrossRef]
- Kongolo, M.; Benzaazoua, M.; de Donato, P.; Drouet, B.; Barrès, O. The comparison between amine thioacetate and amyl xanthate collector performances for pyrite flotation and its application to tailings desulphurization. Miner. Eng. 2004, 17, 505–515. [Google Scholar] [CrossRef]
- Leppinen, J.O. FTIR and flotation investigation of the adsorption of ethyl xanthate on activated and non-activated sulfide minerals. Int. J. Miner. Process. 1990, 30, 245–263. [Google Scholar] [CrossRef]
- Tan, S.N.; Pugh, R.J.; Fornasiero, D.; Sedev, R.; Ralston, J. Foaming of polypropylene glycols and glycol/MIBC mixtures. Miner. Eng. 2005, 18, 179–188. [Google Scholar] [CrossRef]
- Wang, Y.; Lauten, R.A.; Peng, Y. The effect of biopolymer dispersants on copper flotation in the presence of kaolinite. Miner. Eng. 2016, 96–97, 123–129. [Google Scholar] [CrossRef]
- Ramos, O.; Castro, S.; Laskowski, J.S. Copper–molybdenum ores flotation in sea water: Floatability and frothability. Miner. Eng. 2013, 53, 108–112. [Google Scholar] [CrossRef]





| Ion | Concentration [mg/L] | Method of Analysis |
|---|---|---|
| 9950 | Atomic absorption spectrometry | |
| 1250 | Atomic absorption spectrometry | |
| 400 | Atomic absorption spectrometry | |
| 380 | Atomic absorption spectrometry | |
| 19,450 | Argentometric method | |
| 150 | Acid–base volumetry |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castellón, C.I.; Piceros, E.C.; Toro, N.; Robles, P.; López-Valdivieso, A.; Jeldres, R.I. Depression of Pyrite in Seawater Flotation by Guar Gum. Metals 2020, 10, 239. https://doi.org/10.3390/met10020239
Castellón CI, Piceros EC, Toro N, Robles P, López-Valdivieso A, Jeldres RI. Depression of Pyrite in Seawater Flotation by Guar Gum. Metals. 2020; 10(2):239. https://doi.org/10.3390/met10020239
Chicago/Turabian StyleCastellón, César I., Eder C. Piceros, Norman Toro, Pedro Robles, Alejandro López-Valdivieso, and Ricardo I. Jeldres. 2020. "Depression of Pyrite in Seawater Flotation by Guar Gum" Metals 10, no. 2: 239. https://doi.org/10.3390/met10020239
APA StyleCastellón, C. I., Piceros, E. C., Toro, N., Robles, P., López-Valdivieso, A., & Jeldres, R. I. (2020). Depression of Pyrite in Seawater Flotation by Guar Gum. Metals, 10(2), 239. https://doi.org/10.3390/met10020239








