Development of New 14 Cr ODS Steels by Using New Oxides Formers and B as an Inhibitor of the Grain Growth
Abstract
1. Introduction
2. Materials and Methods
3. Results
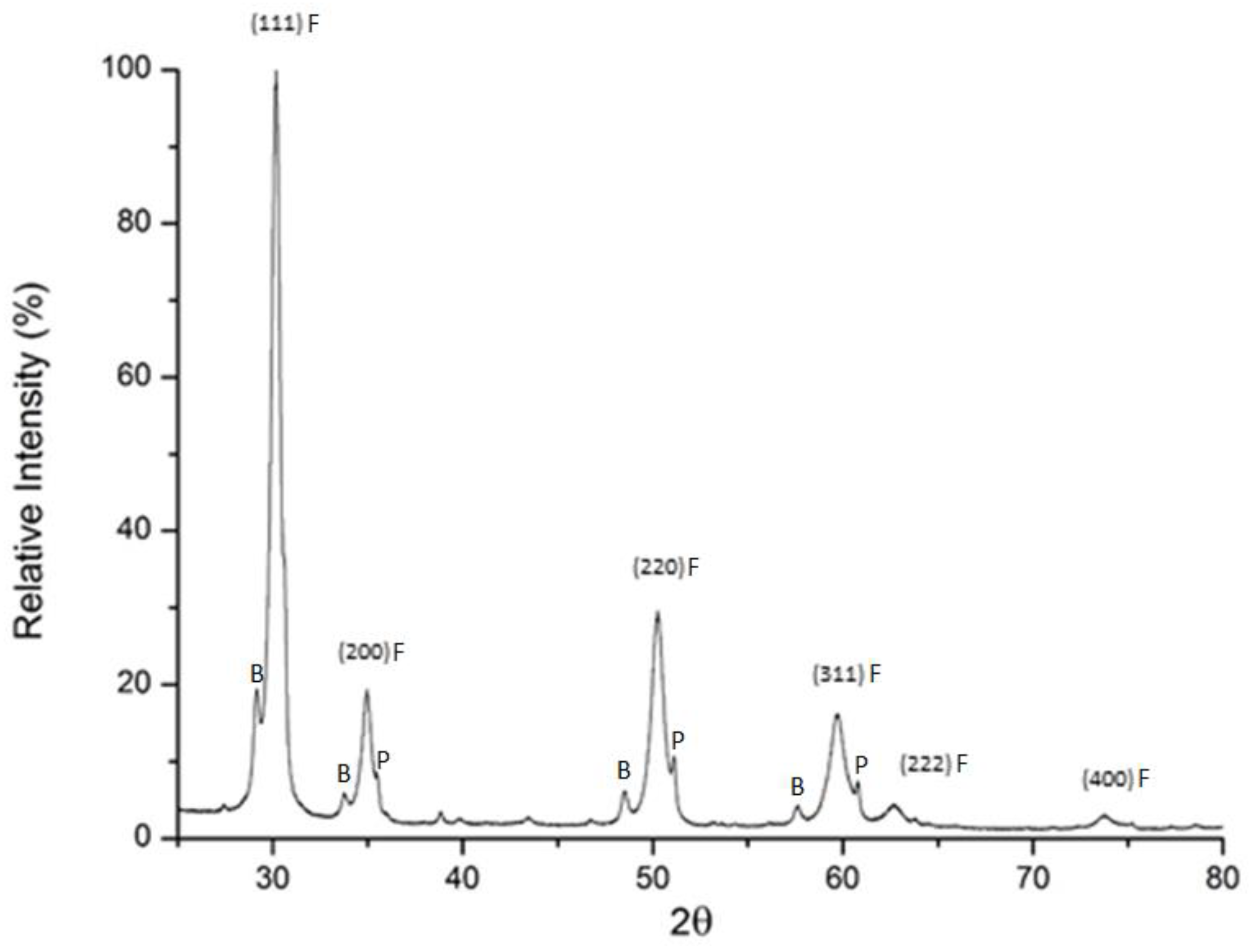
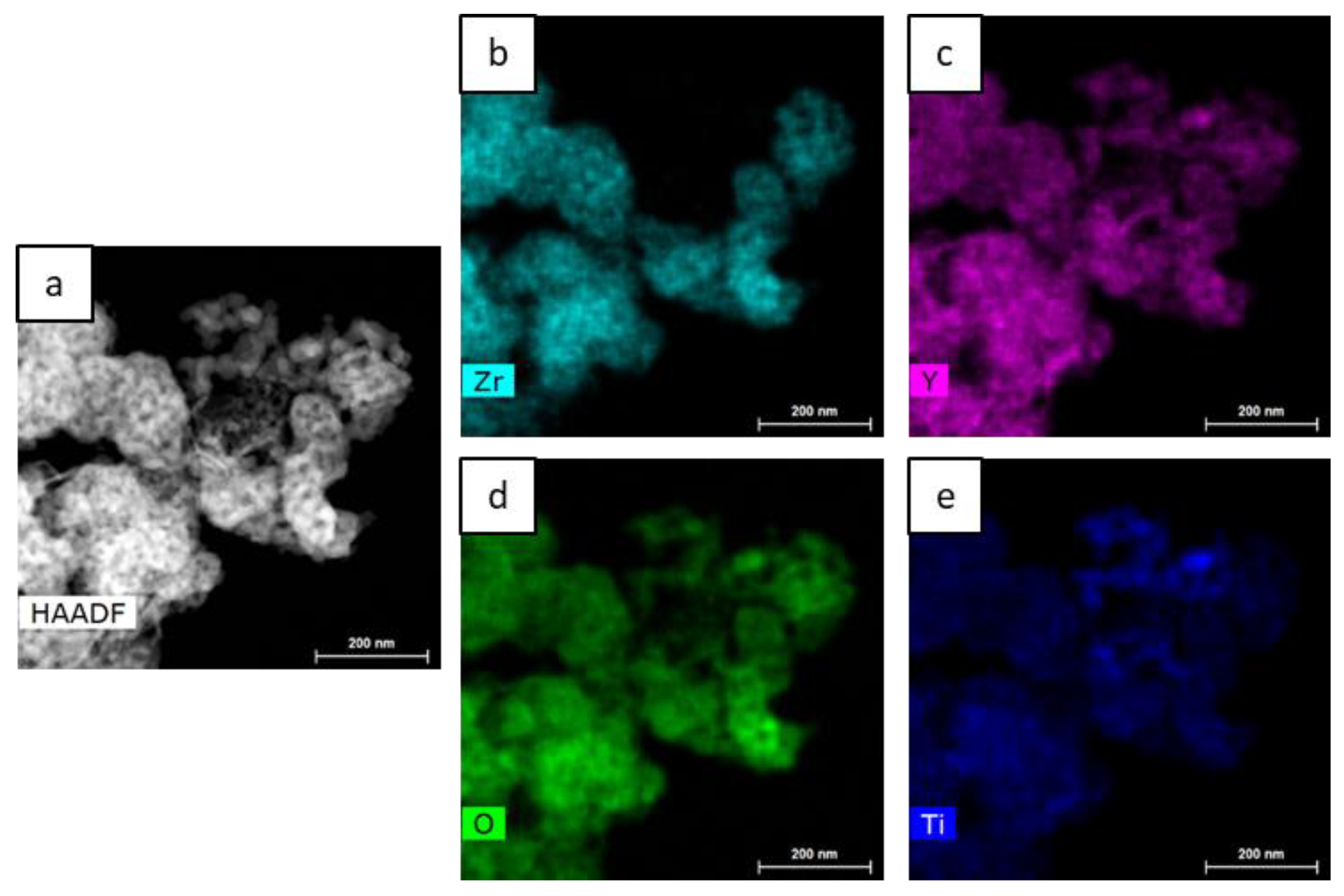
3.1. Characterization of Synthesized Complex Oxide
3.2. Characterization of Milled Powders
3.3. Microstructural Characterization
3.3.1. EBSD Analysis
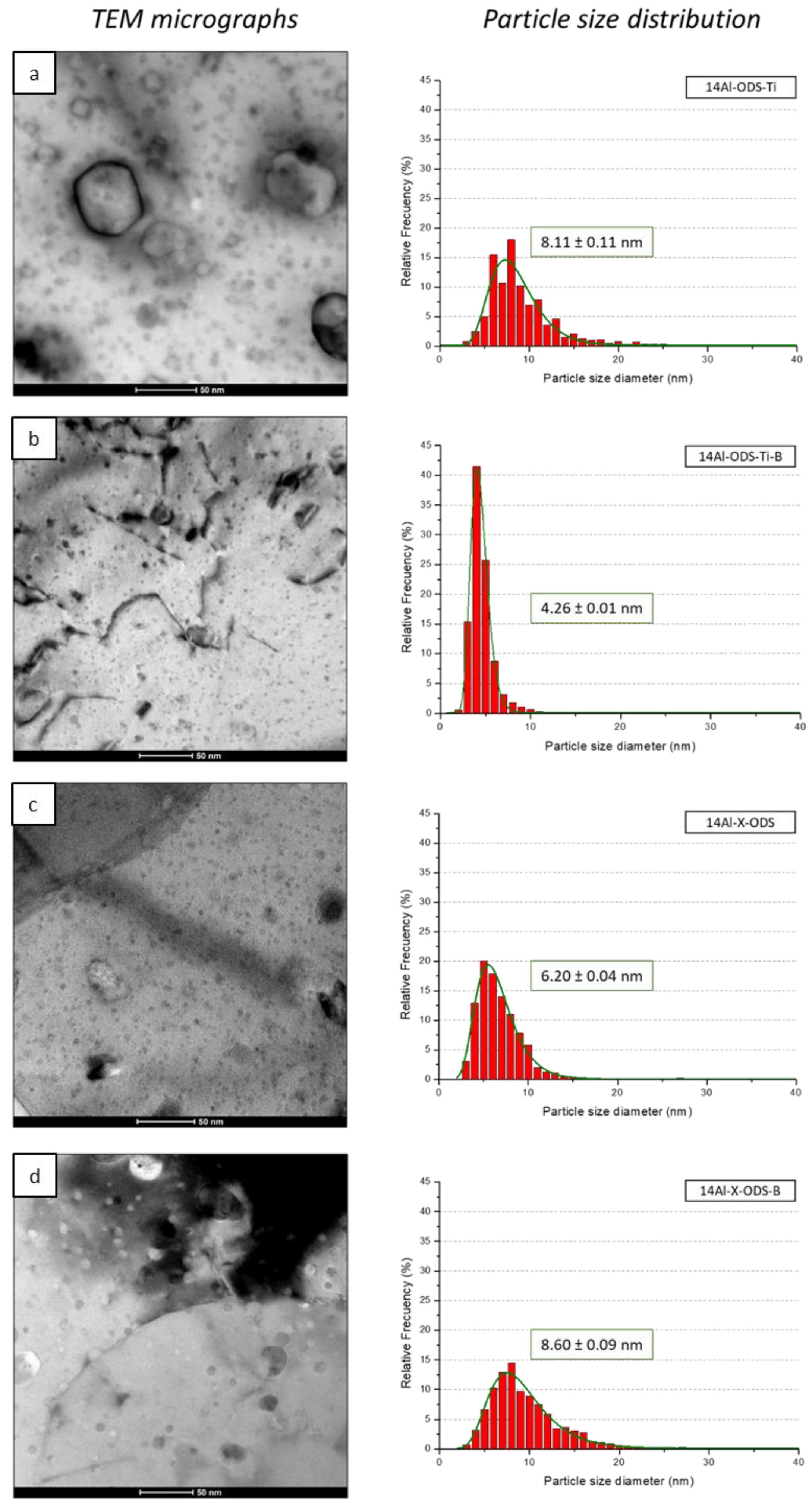
3.3.2. TEM Observations
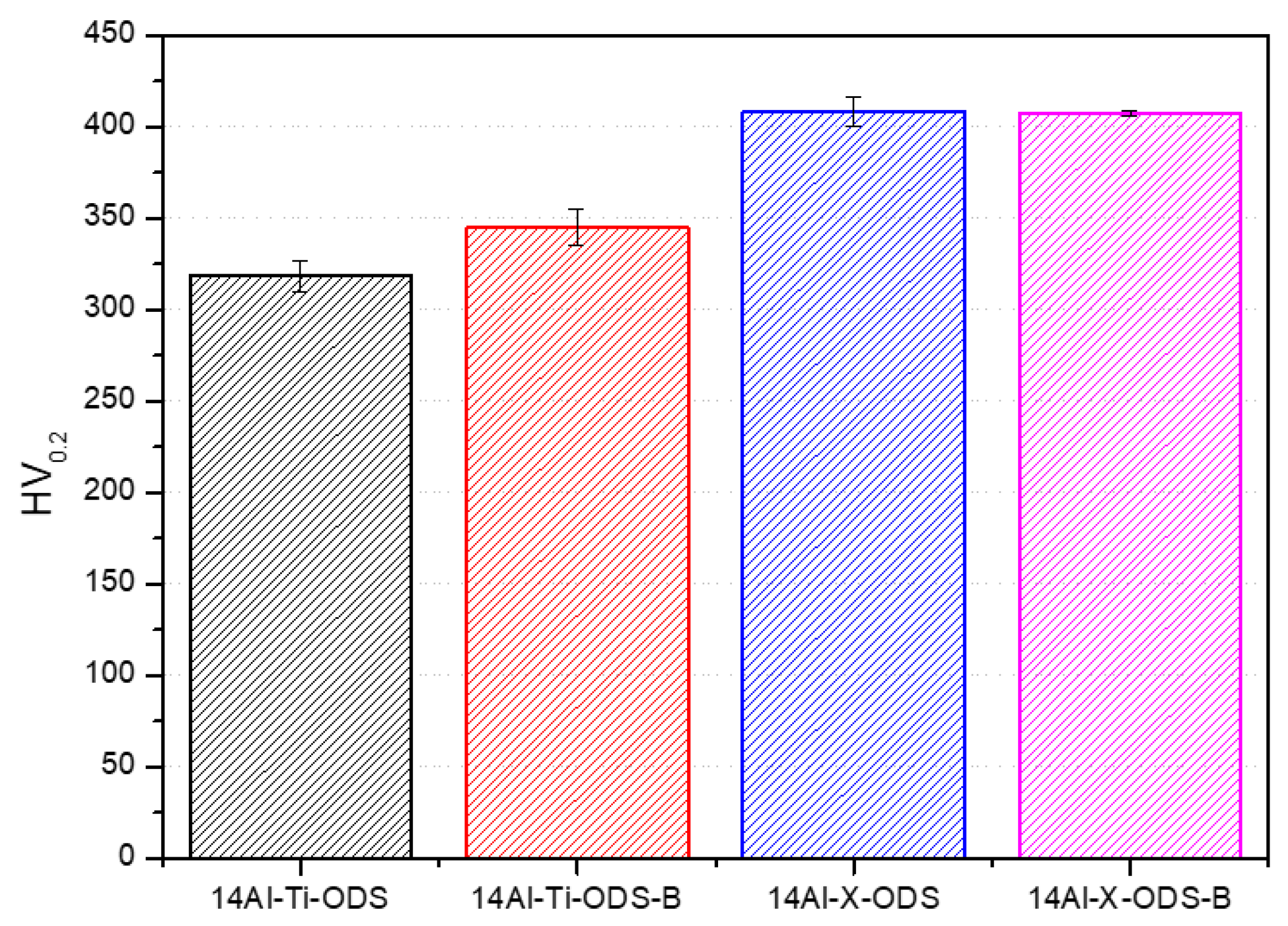
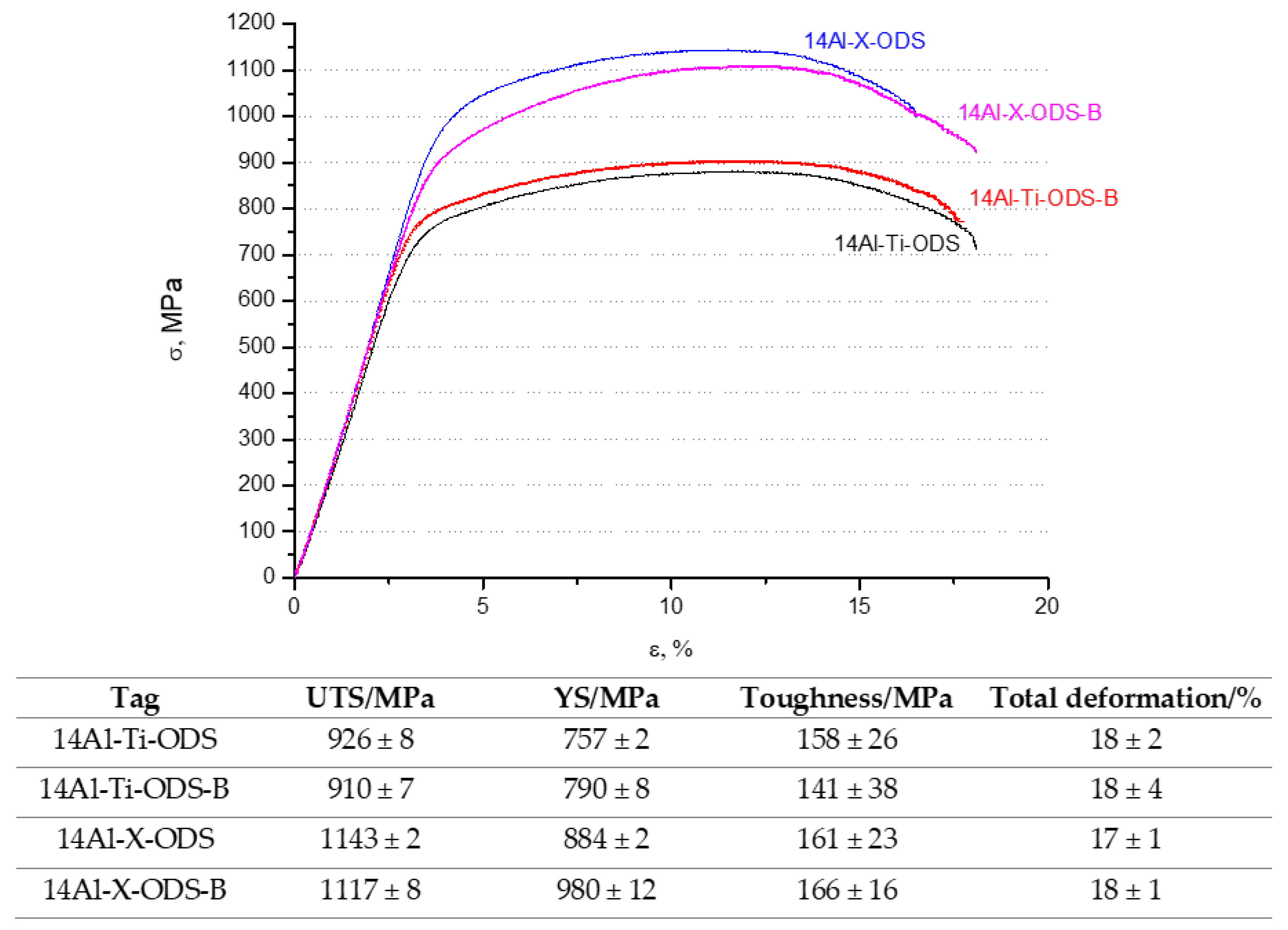
3.4. Mechanical Characterization
4. Conclusions
- The use of a unique precursor of oxide formers (Y-Ti-Zr-O) resulted in a better control of nano precipitates composition when preparing a F-ODS steel, thereby improving the precipitation of specific oxides containing Zr that led to a strengthened microstructure.
- A good precipitation morphologies (in the order of 1022 ox/m3) of the nano oxides was achieved, which may guarantee a fine pinning effect of the oxides over the dislocations and grain boundaries.
- The selected parameters in the SPS technique fully densified the milled powders while avoiding an excessive grain growth during the consolidation stage and maintaining a high density of dislocation. All of this occurred without the necessity of post heat or thermo-mechanical treatments.
- The tailored microstructure affected the final mechanical behavior of the material. The best values of microhardness and UTS were achieved for the 14Al-X-ODS and 14Al-X-ODS-B compositions (with increments of 50 HV0.2 and 200 MPa, respectively, in comparison with the reference composition), which were the ones that included the Y-Ti-Zr-O compound in their composition.
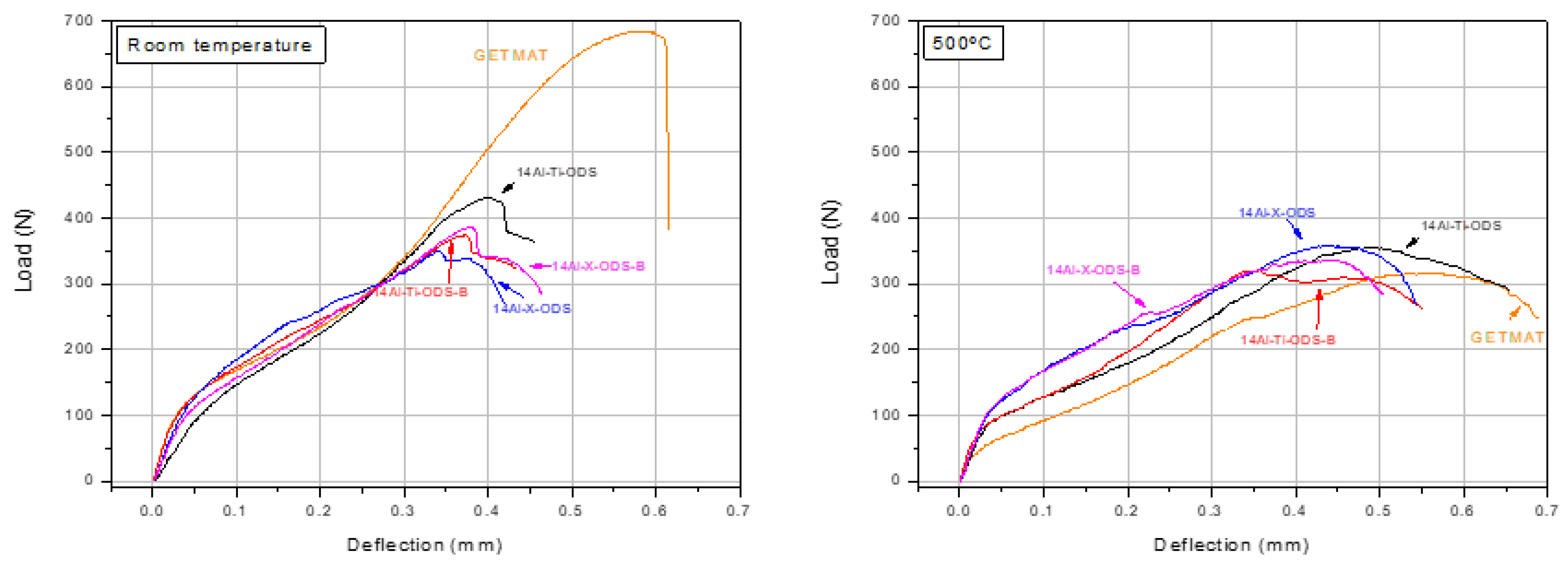
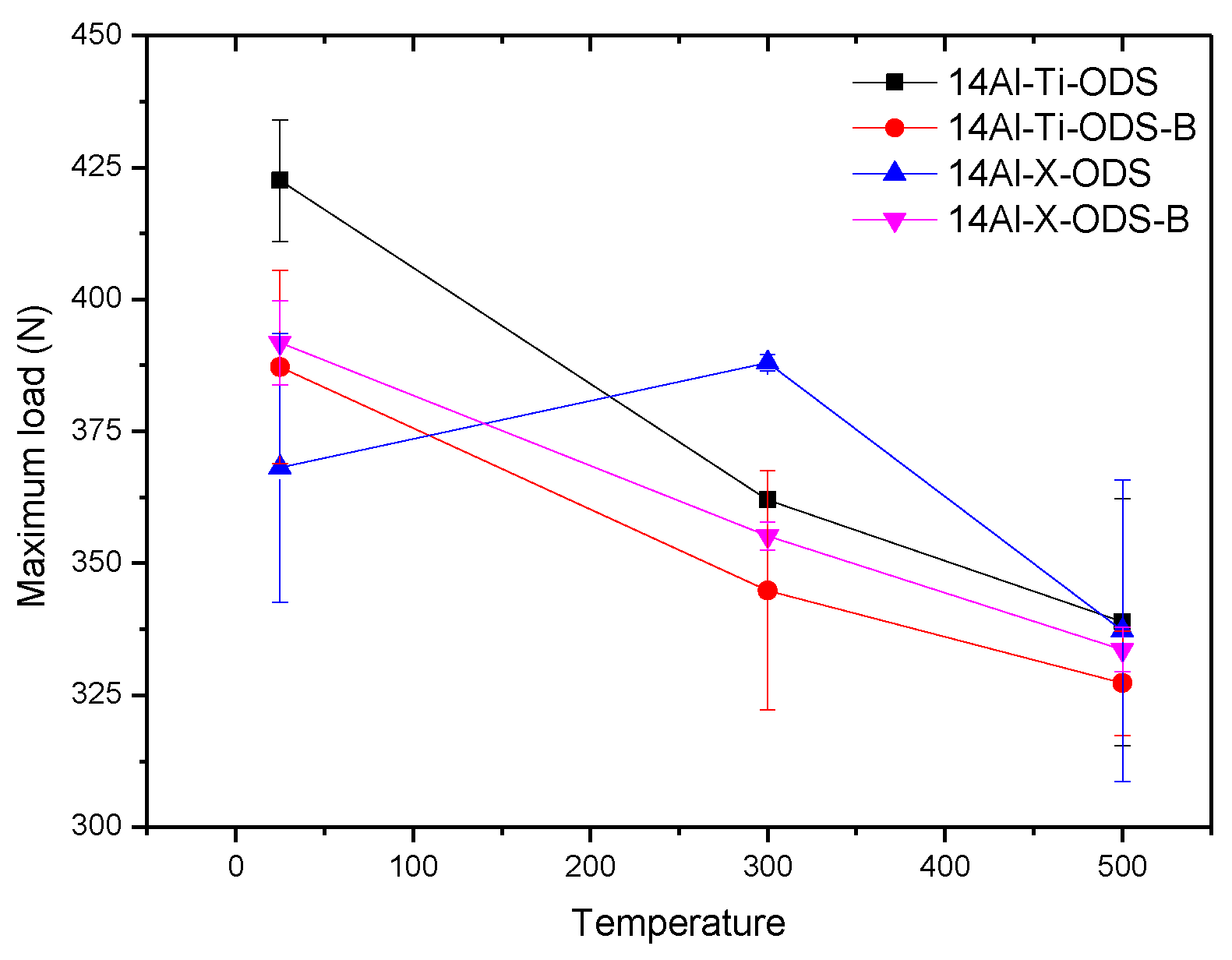
- Small punch tests demonstrated the outstanding performance of 14Al-X-ODS and 14Al-X-ODS-B, achieving similar or better results than the GETMAT material when the tests were performed at high temperatures.
- The mechanical behavior of the materials evaluated at room and high temperatures (500 °C) showed very promising and optimum values, comparable to the ones obtained in ODS steels developed with a more complex route.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Klueh, R.; Shingledecker, J.; Swindeman, R.; Hoelzer, D.T. Oxide dispersion-strengthened steels: A comparison of some commercial and experimental alloys. J. Nucl. Mater. 2005, 341, 103–114. [Google Scholar] [CrossRef]
- Kimura, A.; Ukai, S.; Fujiwara, M. Oxide Dispersion Strengthening Steels R&D for Advanced Water-Cooling Nuclear Systems. In Proceedings of the ICAPP, Pittsburgh, PA, USA, 13–17 June 2004; ISBN 0-89448-680-2. [Google Scholar]
- Lu, C.; Lu, Z.; Xie, R.; Liu, C.; Wang, L. Microstructure of HIPed and SPSed 9Cr-ODS steel and its effect on helium bubble formation. J. Nucl. Mater. 2016, 474, 65–75. [Google Scholar] [CrossRef]
- Kimura, A.; Sugano, R.; Matsushita, Y.; Ukai, S. Thermal Helium Desorption Behavior in Ad-vanced Ferritic Steels. J. Phys. Chem. Solids 2005, 66, 504–508. [Google Scholar] [CrossRef]
- Dou, P.; Kimura, A.; Okuda, T.; Inoue, M.; Ukai, S.; Ohnuki, S.; Fujisawa, T.; Abe, F. Effects of extrusion temperature on the nano-mesoscopic structure and mechanical properties of an Al-alloyed high-Cr ODS ferritic steel. J. Nucl. Mater. 2011, 417, 166–170. [Google Scholar] [CrossRef]
- Zhang, G.; Zhou, Z.; Mo, K.; Miao, Y.; Li, S.; Liu, X.; Wang, M.; Park, J.-S.; Almer, J.; Stubbins, J.F. The comparison of microstructures and mechanical properties between 14Cr-Al and 14Cr-Ti ferritic ODS alloys. Mater. Des. 2016, 98, 61–67. [Google Scholar] [CrossRef]
- Kimura, A.; Kasada, R.; Iwata, N.Y.; Kishimoto, H.; Zhang, C.; Isselin, J.; Dou, P.; Lee, J.; Muthukumar, N.; Okuda, T.; et al. Development of Al added high-Cr ODS steels for fuel cladding of next generation nuclear systems. J. Nucl. Mater. 2011, 417, 176–179. [Google Scholar] [CrossRef]
- Garcia-Rodriguez, N.; Campos, M.; Torralba, J.M.; Berger, M.H.; Bienvenu, Y. Capability of mechanical alloying and SPS technique to develop nanostructured high Cr, Al alloyed ODS steels. Mater. Sci. Technol. 2014, 30, 1676–1684. [Google Scholar] [CrossRef]
- Narita, T.; Ukai, S.; Ohtsuka, S.; Inoue, M. Effect of tungsten addition on microstructure and high temperature strength of 9CrODS ferritic steel. J. Nucl. Mater. 2011, 417, 158–161. [Google Scholar] [CrossRef]
- Boulnat, X.; Perez, M.; Fabrègue, D.; Cazottes, S.; De Carlan, Y. Characterization and modeling of oxides precipitation in ferritic steels during fast non-isothermal consolidation. Acta Mater. 2016, 107, 390–403. [Google Scholar] [CrossRef]
- García-Junceda, A.; Campos, M.; García-Rodríguez, N.; Torralba, J.M. On the Role of Alloy Composition and Sintering Parameters in the Bimodal Grain Size Distribution and Mechanical Properties of ODS Ferritic Steels. Met. Mater. Trans. A 2016, 47, 5325–5333. [Google Scholar] [CrossRef]
- Chen, C.-L.; Richter, A.; Kögler, R.; Talut, G. Dual beam irradiation of nanostructured FeCrAl oxide dispersion strengthened steel. J. Nucl. Mater. 2011, 412, 350–358. [Google Scholar] [CrossRef]
- Rajan, K.; Sarma, V.; Kutty, T.; Murty, B. Hot hardness behaviour of ultrafine grained ferritic oxide dispersion strengthened alloys prepared by mechanical alloying and spark plasma sintering. Mater. Sci. Eng. A 2012, 558, 492–496. [Google Scholar] [CrossRef]
- Dou, P.; Kimura, A.; Kasada, R.; Okuda, T.; Inoue, M.; Ukai, S.; Ohnuki, S.; Fujisawa, T.; Abe, F. TEM and HRTEM study of oxide particles in an Al-alloyed high-Cr oxide dispersion strengthened steel with Zr addition. J. Nucl. Mater. 2014, 444, 441–453. [Google Scholar] [CrossRef]
- Gao, R.; Zhang, T.; Wang, X.; Fang, Q.; Liu, C. Effect of zirconium addition on the microstructure and mechanical properties of ODS ferritic steels containing aluminum. J. Nucl. Mater. 2014, 444, 462–468. [Google Scholar] [CrossRef]
- García-Junceda, A.; García-Rodríguez, N.; Campos, M.; Cartón-Cordero, M.; Torralba, J.M. Effect of Zirconium on the Microstructure and Mechanical Properties of an Al-Alloyed ODS Steel Consolidated by FAHP. J. Am. Ceram. Soc. 2015, 98, 3582–3587. [Google Scholar] [CrossRef]
- Li, W.; Xu, H.; Sha, X.; Meng, J.; Wang, Z. Microstructure and Mechanical Properties of 14Cr-ODS Steels with Zr Addition. High Temp. Mater. Process. 2019, 38, 404–410. [Google Scholar] [CrossRef]
- Moeslang, A.; Klimenkov, M.; Lindau, R.; Materna-Morris, E.; Rolli, R.; Schneider, H.C. Neutron Irradiat-ed Ferritic-Martensitic 9CR-Steel: Effect of Nanoscaled ODS Particles and Helium Bubbles on Properties. 2016. Available online: https://inis.iaea.org/search/search.aspx?orig_q=RN:47104157 (accessed on 28 February 2018).
- Abe, F. Effect of Boron on Microstructure and Creep Strength of Advanced Ferritic Power Plant Steels. Procedia Eng. 2011, 10, 94–99. [Google Scholar] [CrossRef]
- Shulga, A.V. A comparative study of the mechanical properties and the behavior of carbon and boron in stainless steel cladding tubes fabricated by PM HIP and traditional technologies. J. Nucl. Mater. 2013, 434, 133–140. [Google Scholar] [CrossRef]
- Abe, F.; Tabuchi, M.; Tsukamoto, S. Mechanisms for boron effect on microstructure and creep strength of ferritic power plant steels. Energy Mater. 2009, 4, 166–174. [Google Scholar] [CrossRef]
- Jayakumar, T.; Mathew, M.D.; Laha, K.; Sandhya, R. Materials development for fast reactor applications. Nucl. Eng. Des. 2013, 265, 1175–1180. [Google Scholar] [CrossRef]
- Abe, F.; Tabuchi, M.; Kondo, M.; Tsukamoto, S. Suppression of Type IV fracture and improvement of creep strength of 9Cr steel welded joints by boron addition. Int. J. Press. Vessel. Pip. 2007, 84, 44–52. [Google Scholar] [CrossRef]
- Abe, F. Progress in Creep-Resistant Steels for High Efficiency Coal-Fired Power Plants. J. Press. Vessel. Technol. 2016, 138, 040804. [Google Scholar] [CrossRef]
- Sulima, I. Role of boron addition on the consolidation and properties of steel composites prepared by SPS. Bull. Mater. Sci. 2015, 38, 1831–1841. [Google Scholar] [CrossRef]
- Torralba, J.M.; Fuentes-Pacheco, L.; García-Rodríguez, N.; Campos, M. Development of high performance powder metallurgy steels by high-energy milling. Adv. Powder Technol. 2013, 24, 813–817. [Google Scholar] [CrossRef]
- Hilger, I.; Boulnat, X.; Hoffmann, J.; Testani, C.; Bergner, F.; De Carlan, Y.; Ferraro, F.; Ulbricht, A. Fabrication and characterization of oxide dispersion strengthened (ODS) 14Cr steels consolidated by means of hot isostatic pressing, hot extrusion and spark plasma sintering. J. Nucl. Mater. 2016, 472, 206–214. [Google Scholar] [CrossRef]
- Schaedler, T.A.; Francillon, W.; Gandhi, A.S.; Grey, C.P.; Sampath, S.; Levi, C.G. Phase evolution in the YO1.5–TiO2–ZrO2 system around the pyrochlore region. Acta Mater. 2005, 53, 2957–2968. [Google Scholar] [CrossRef]
- Macía, E.; García-Junceda, A.; Serrano, M.; Hernández-Mayoral, M.; Diaz, L.A.; Campos, M. Effect of the heating rate on the microstructure of a ferritic ODS steel with four oxide formers (Y-Ti-Al-Zr) consolidated by spark plasma sintering (SPS). J. Nucl. Mater. 2019, 518, 190–201. [Google Scholar] [CrossRef]
- García-Junceda, A.; Macía, E.; Garbiec, D.; Serrano, M.; Torralba, J.M.; Campos, M. Effect of Small Variations in Zr Content on the Microstructure and Properties of Ferritic ODS Steels Consolidated by SPS. Metals 2020, 10, 348. [Google Scholar] [CrossRef]
- Hilger, I.; Tegel, M.; Gorley, M.; Grant, P.S.; Weißgärber, T.; Kieback, B. The structural changes of Y2O3 in ferritic ODS alloys during milling. J. Nucl. Mater. 2014, 447, 242–247. [Google Scholar] [CrossRef]
- Song, X.; Liu, X.; Zhang, J. Neck Formation and Self-Adjusting Mechanism of Neck Growth of Conducting Powders in Spark Plasma Sintering. J. Am. Ceram. Soc. 2006, 89, 494–500. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, Y.; Yu, L.; Ma, Z.; Guo, Q.; Huang, Y.; Li, H. Microstructure characteristic and mechanical property of transformable 9Cr-ODS steel fabricated by spark plasma sintering. Mater. Des. 2017, 132, 158–169. [Google Scholar] [CrossRef]
- Boulnat, X.; Perez, M.; Fabrègue, D.; Douillard, T.; Mathon, M.-H.; De Carlan, Y. Microstructure Evolution in Nano-reinforced Ferritic Steel Processed By Mechanical Alloying and Spark Plasma Sintering. Met. Mater. Trans. A 2013, 45, 1485–1497. [Google Scholar] [CrossRef]
- García-Junceda, A.; Hernández-Mayoral, M.; Serrano, M. Influence of the microstructure on the tensile and impact properties of a 14Cr ODS steel bar. Mater. Sci. Eng. A 2012, 556, 696–703. [Google Scholar] [CrossRef]




| ODS Steel | Prealloyed | Oxide Formers | Complex Oxide | Other Alloying Elements | ||||
|---|---|---|---|---|---|---|---|---|
| Fe | Cr | Al | W | Y2O3 | Ti | Y-Ti-Zr-O | B | |
| 14Al-Ti-ODS | bal | 14 | 5 | 3 | 0.25 | 0.4 | - | - |
| 14Al-Ti-ODS-B | bal | 14 | 5 | 3 | 0.25 | 0.4 | - | 0.1 |
| 14Al-X-ODS | bal | 14 | 5 | 3 | - | - | 1.62 | - |
| 14Al-X-ODS-B | bal | 14 | 5 | 3 | - | - | 1.97 | 0.1 |
| Tag | d50 (µm) | L (nm) | µε (%) | Dislocations Density (m-2) | σdis (MPa) |
|---|---|---|---|---|---|
| Preal. powder | 30 | 43.10 | 0.217 | 1.07 × 1015 | 713 |
| 14Al-Ti-ODS | 38 | 11.94 | 0.769 | 1.36 × 1016 | 2522 |
| 14Al-Ti-ODS-B | 73 | 11.99 | 0.767 | 1.45 × 1016 | 2598 |
| 14Al-X-ODS | 38 | 11.60 | 0.790 | 1.51 × 1016 | 2655 |
| 14Al-X-ODS-B | 54 | 11.40 | 0.808 | 1.37 × 1016 | 2529 |
| ODS Steel | 14Al-Ti-ODS | 14Al-Ti-ODS-B | 14Al-X-ODS | 14Al-X-ODS-B |
|---|---|---|---|---|
| Mean size of coarse grains (> 1 µm) | 3.03 µm | 2.98 µm | 2.48 µm | 2.83 µm |
| Mean size of ultrafine grains (< 1 µm) | 0.59 µm | 0.64 µm | 0.26 µm | 0.28 µm |
| % UFG area | 5% | 13% | 13% | 16% |
| ODS Composition | Dislocations Density (m−2) | Dis (MPa) |
|---|---|---|
| 14Al-Ti-ODS | 3.27 × 1015 | 1235 |
| 14Al-Ti-ODS-B | 2.40 × 1015 | 1056 |
| 14Al-X-ODS | 2.24 × 1015 | 1022 |
| 14Al-X-ODS-B | 2.74 × 1015 | 1128 |
| ODS Composition | Precipitates Density (Precipitates/m3) | σp (MPa) |
|---|---|---|
| 14Al-Ti-ODS | 2.76 × 1022 | 323 |
| 14Al-Ti-ODS-B | 7.43 × 1022 | 384 |
| 14Al-X-ODS | 4.03 × 1022 | 341 |
| 14Al-X-ODS-B | 1.70 × 1022 | 261 |
| ODS Composition | Type of Precipitates | ||||
|---|---|---|---|---|---|
| Ti-W/Ti-Zr/Ti-Zr-W (60–80 nm) | Y-Al-O (8–20 nm) | Y-Al-Zr-O (6–20 nm) | Y-Al-Ti-Zr-O (6–20 nm) | Y-Zr-O (3–10 nm) | |
| 14Al-Ti-ODS | ✓ | ✓ | - | - | - |
| 14Al-Ti-ODS-B | ✓ | ✓ | - | - | - |
| 14Al-X-ODS | ✓ | ✓ | ✓ | ✓ | ✓ |
| 14Al-X-ODS-B | ✓ | ✓ | ✓ | ✓ | ✓ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meza, A.; Macía, E.; García-Junceda, A.; Díaz, L.A.; Chekhonin, P.; Altstadt, E.; Serrano, M.; Rabanal, M.E.; Campos, M. Development of New 14 Cr ODS Steels by Using New Oxides Formers and B as an Inhibitor of the Grain Growth. Metals 2020, 10, 1344. https://doi.org/10.3390/met10101344
Meza A, Macía E, García-Junceda A, Díaz LA, Chekhonin P, Altstadt E, Serrano M, Rabanal ME, Campos M. Development of New 14 Cr ODS Steels by Using New Oxides Formers and B as an Inhibitor of the Grain Growth. Metals. 2020; 10(10):1344. https://doi.org/10.3390/met10101344
Chicago/Turabian StyleMeza, Alberto, Eric Macía, Andrea García-Junceda, Luis Antonio Díaz, Paul Chekhonin, Eberhard Altstadt, Marta Serrano, María Eugenia Rabanal, and Mónica Campos. 2020. "Development of New 14 Cr ODS Steels by Using New Oxides Formers and B as an Inhibitor of the Grain Growth" Metals 10, no. 10: 1344. https://doi.org/10.3390/met10101344
APA StyleMeza, A., Macía, E., García-Junceda, A., Díaz, L. A., Chekhonin, P., Altstadt, E., Serrano, M., Rabanal, M. E., & Campos, M. (2020). Development of New 14 Cr ODS Steels by Using New Oxides Formers and B as an Inhibitor of the Grain Growth. Metals, 10(10), 1344. https://doi.org/10.3390/met10101344








