Identification of the Flow Properties of a 0.54% Carbon Steel during Continuous Cooling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mathematical Approach
- The material behaves isotropic regarding all mechanical properties.
- Strain hardening effects are negligible compared to the phase transformation effects.
- Compared to the plastic deformations, the elastic, thermal and transformation strains are negligible small.
2.2. Materials
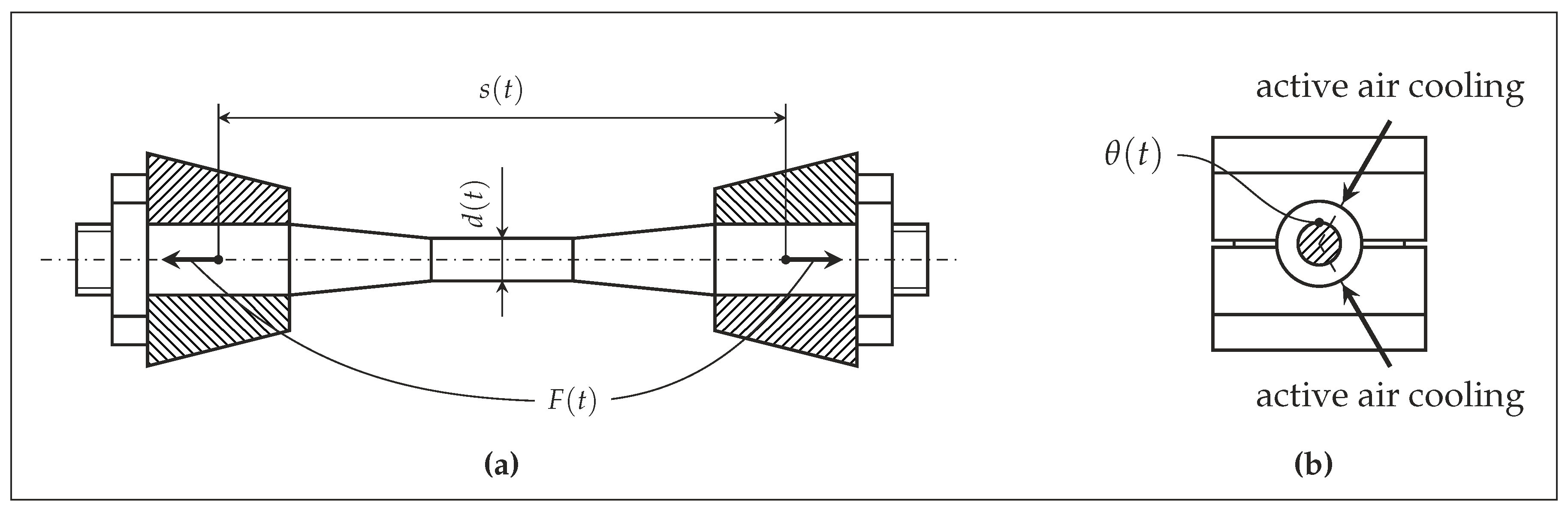
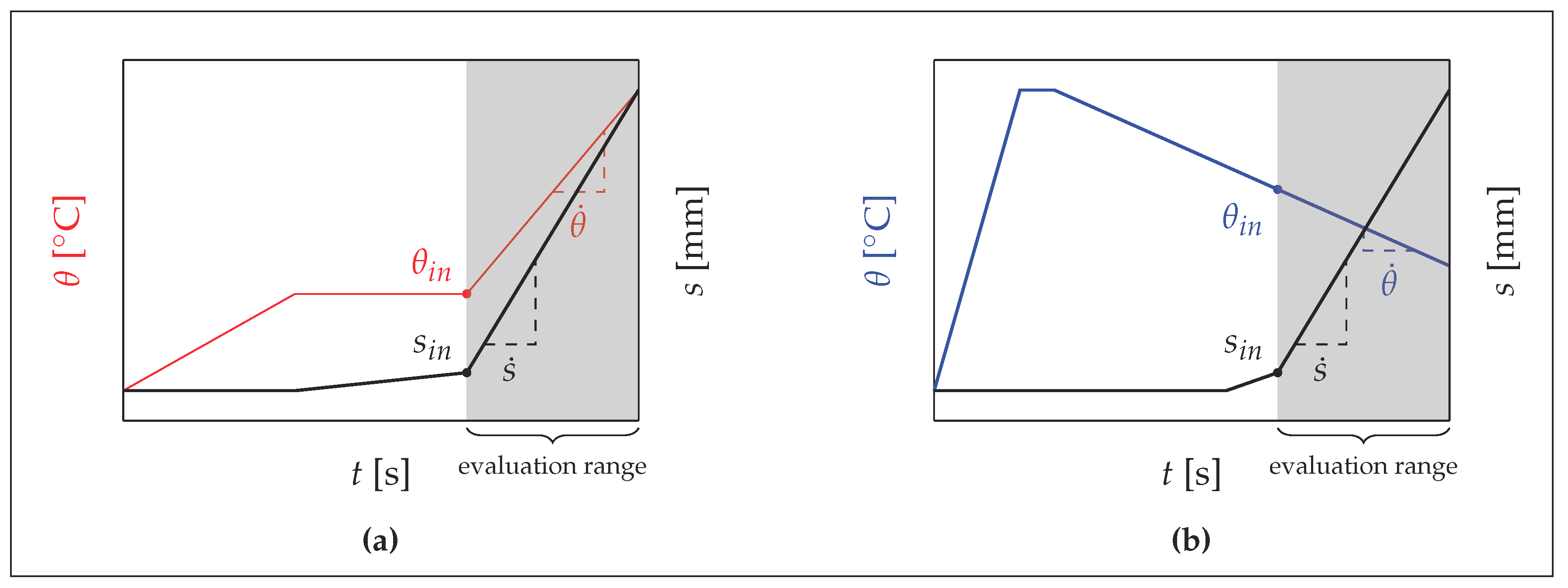
2.3. Experimental Setup
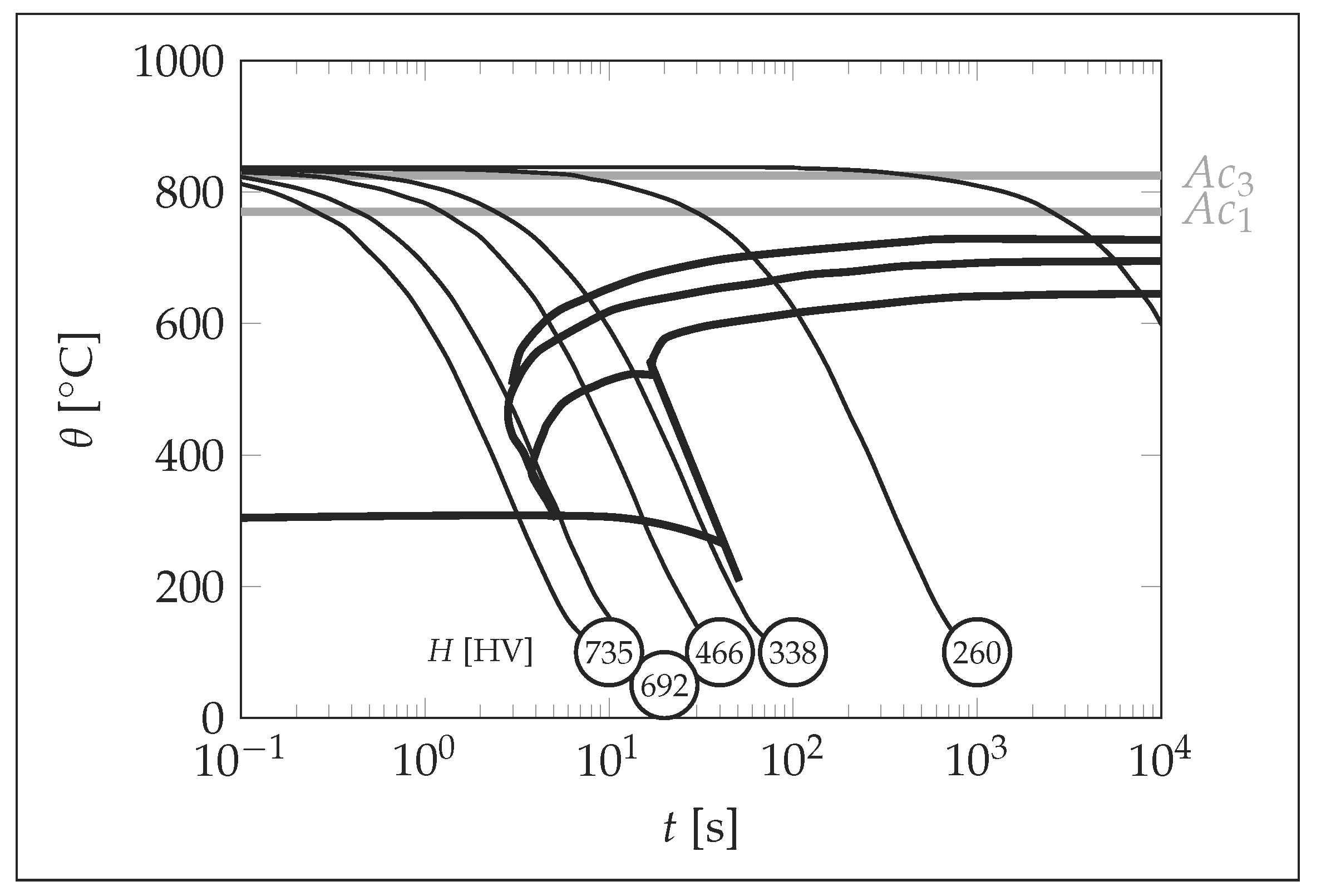
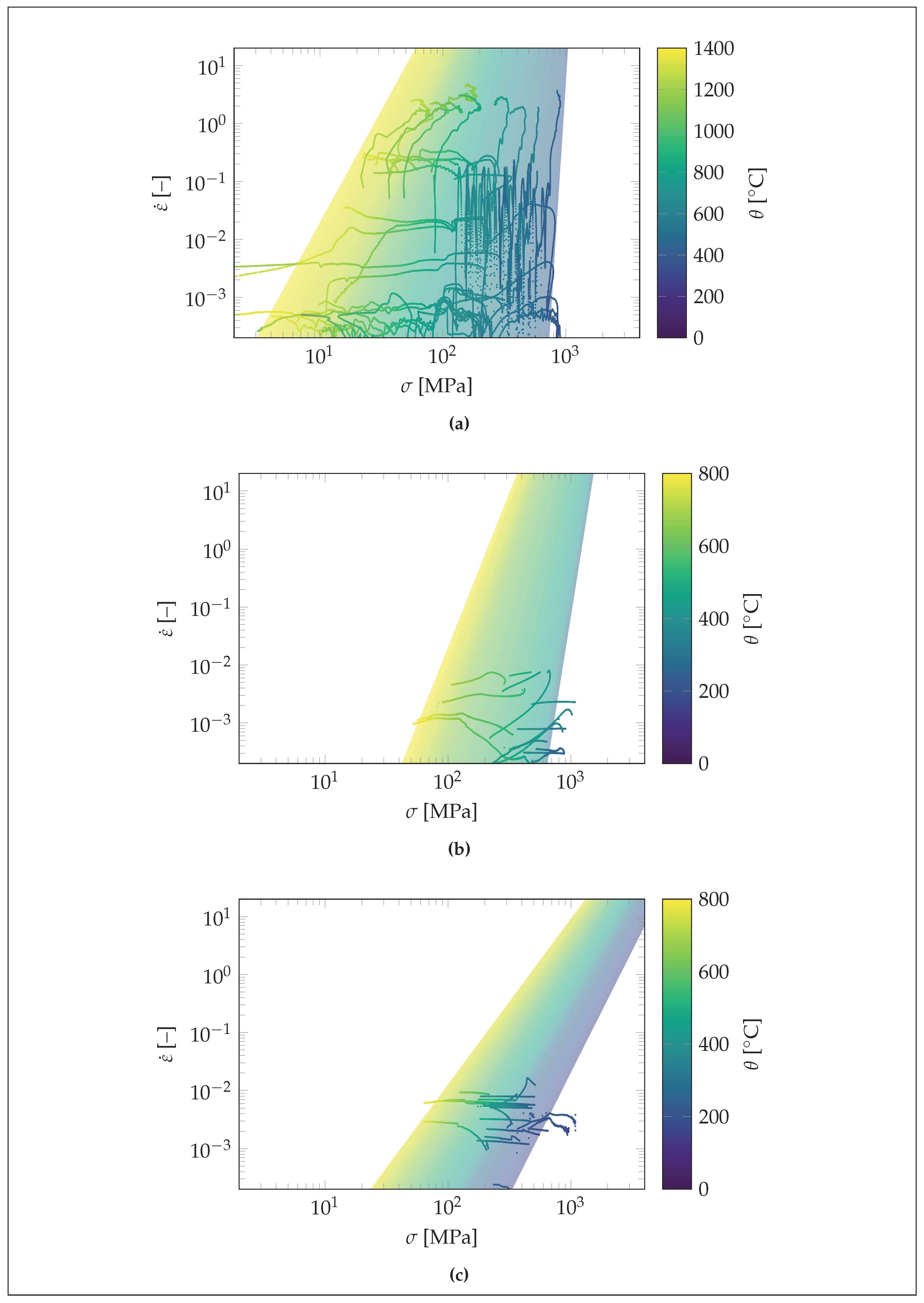
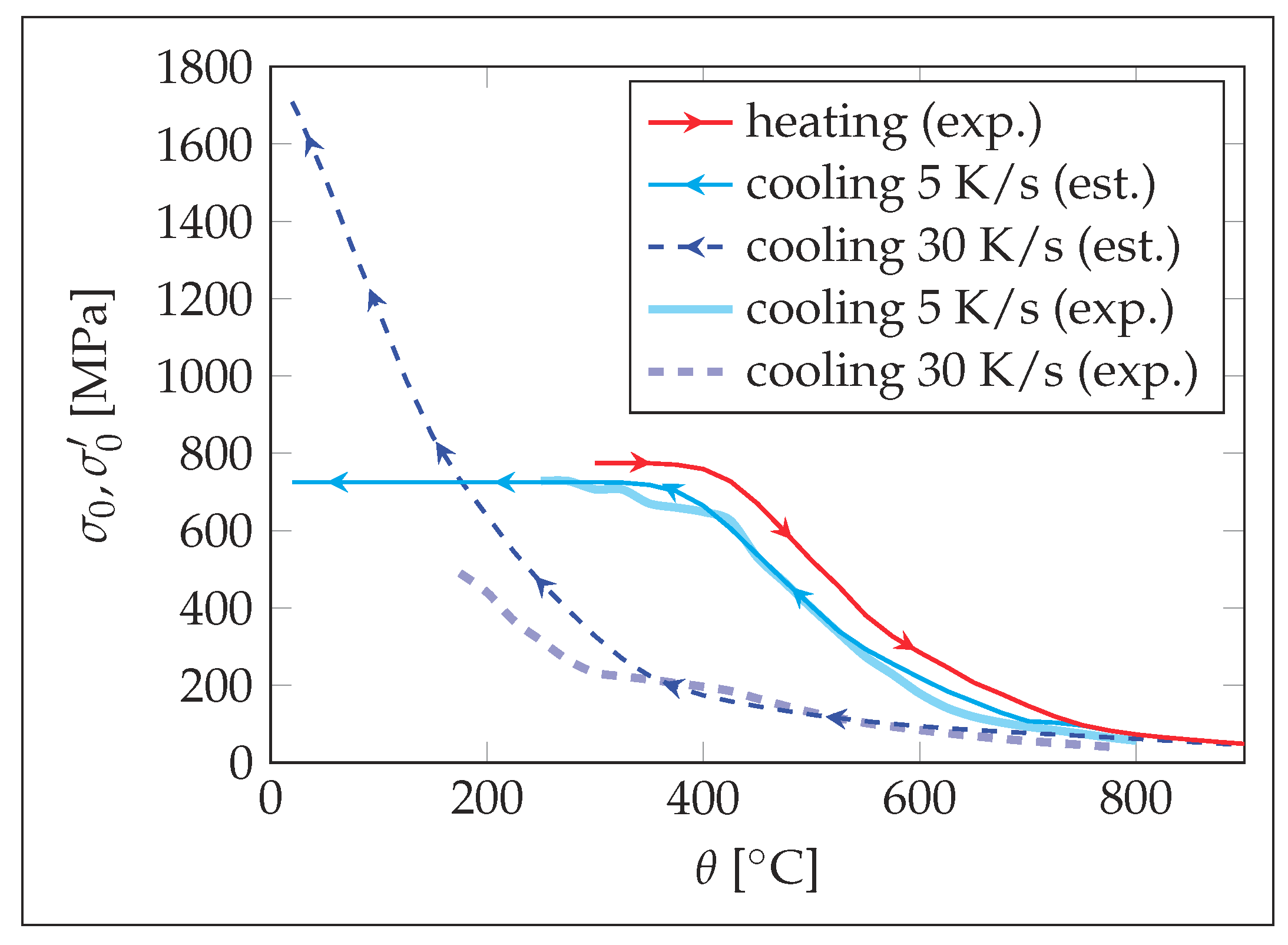
3. Results
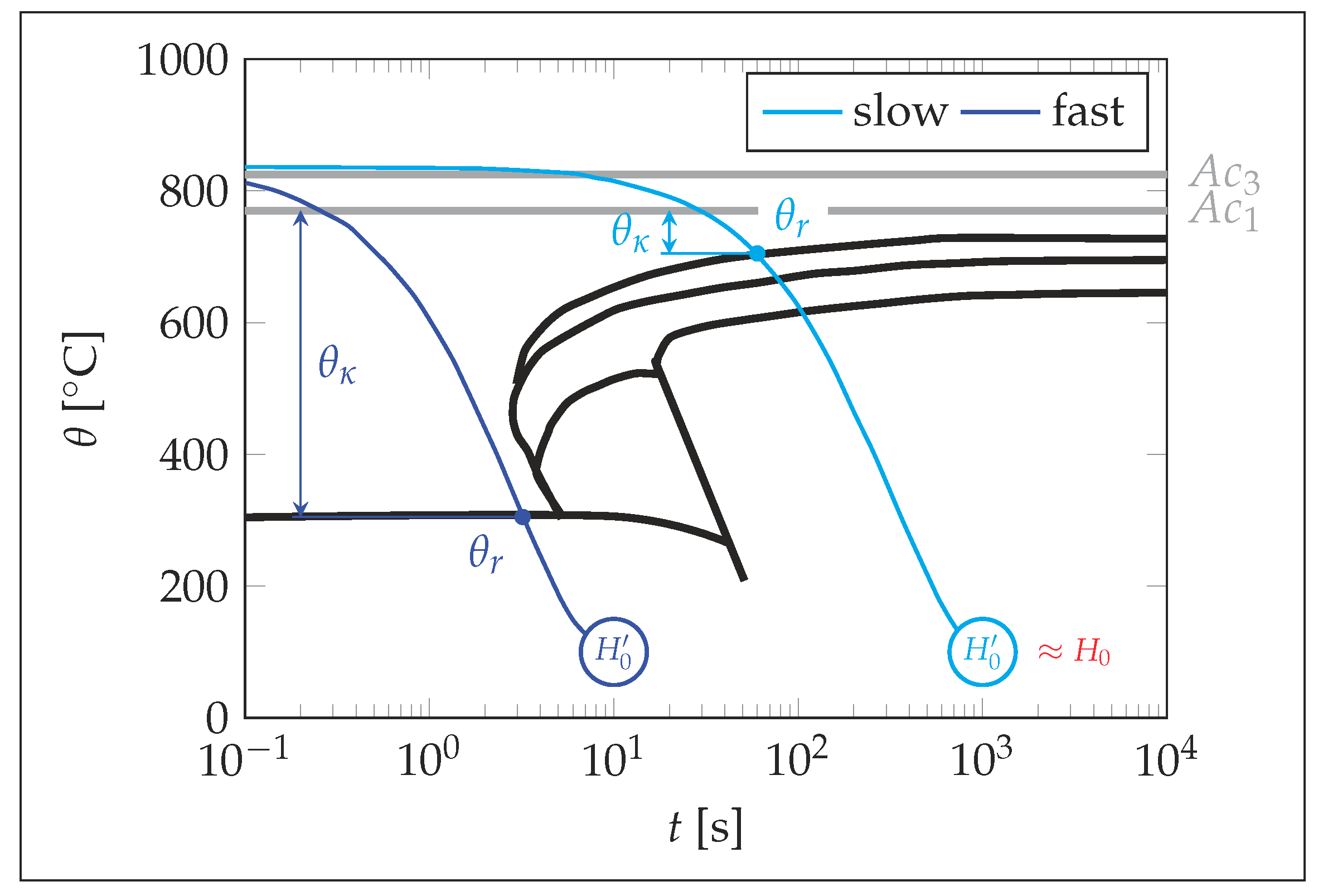
4. Discussion
5. Conclusions
- The model by Schmicker et al. is successfully applied to continuous cooling, without violating its consistency.
- The higher the cooling rate, the greater the differences in the flow properties at the same temperature during heating and cooling.
- Using the data available in CCT diagrams, the properties during cooling can be approximated based on the properties determined at heating, which allows to increase the accuracy compared to the model without the presented adaptions.
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CCT | Continuous cooling transformation |
| RFW | Rotary friction welding |
| TTA | Time-temperature-austenitization |
References
- Mousavi, A.; Asghar, S.A.; Rahbar, A. Experimental and Numerical Analysis of the Friction Welding Process for the 4340 Steel and Mild Steel Combinations. Weld. J. 2008, 87, 178–186. [Google Scholar]
- Seli, H.; Ismail, A.I.M.; Rachman, E.; Ahmad, Z.A. Mechanical evaluation and thermal modelling of friction welding of mild steel and aluminium. J. Mater. Process. Technol. 2010, 210, 1209–1216. [Google Scholar] [CrossRef]
- Grant, B.; Preuss, M.; Withers, P.J.; Baxter, G.; Rowlson, M. Finite element process modelling of inertia friction welding advanced nickel-based superalloy. Mater. Sci. Eng. 2009, 513–514, 366–375. [Google Scholar] [CrossRef]
- Schmicker, D. A Holistic Approach on the Simulation of Rotary Friction Welding; ePubly GmbH: Berlin, Germany, 2015. [Google Scholar]
- Schmicker, D.; Naumenko, K.; Strackeljan, J. A robust simulation of Direct Drive Friction Welding with a modified Carreau fluid constitutive model. Comput. Methods Appl. Mech. Eng. 2013, 265, 186–194. [Google Scholar] [CrossRef]
- Schmicker, D.; Persson, P.O.; Strackeljan, J. Implicit Geometry Meshing for the simulation of Rotary Friction Welding. J. Comput. Phys. 2014, 270, 478–489. [Google Scholar] [CrossRef]
- Schmicker, D.; Paczulla, S.; Nitzschke, S.; Groschopp, S.; Naumenko, K.; Jüttner, S.; Strackeljan, J. Experimental identification of flow properties of a S355 structural steel for hot deformation processes. J. Strain Anal. Eng. Des. 2015, 50, 75–83. [Google Scholar] [CrossRef]
- Spittel, M.; Spittel, T.; Warlimont, H.; Landolt, H.; Börnstein, R.; Martienssen, W. (Eds.) Numerical Data and Functional Relationships in Science and Technology: New Series, Group VIII, Volume 2, Subvolume C, Part 1: Ferrous Alloys; Springer: Berlin, Germany, 2009. [Google Scholar]
- Spittel, M.; Spittel, T.; Warlimont, H.; Landolt, H.; Börnstein, R.; Martienssen, W. (Eds.) Numerical Data and Functional Relationships in Science and Technology: New Series, Group VIII, Volume 2, Subvolume C, Part 2: Non-ferrous Alloys - Light Metals; Springer: Berlin, Germany, 2011. [Google Scholar]
- Rech, J.; Hamdi, H.; Valette, S. Workpiece Surface Integrity. In Machining; Springer: London, UK, 2008; pp. 59–96. [Google Scholar] [CrossRef]
- Radaj, D. Heat Effects of Welding: Temperature Field, Residual Stress, Distortion; Springer: Berlin/Heidelberg, Germany, 1992. [Google Scholar] [CrossRef]
- Werkstoff-Datenblatt Saarstahl—C55R (Cm55). Available online: http://www.saarstahl.com/sag/downloads/download/11282 (accessed on 20 February 2019).
- Nürnberger, F.; Grydin, O.; Schaper, M.; Bach, F.W.; Koczurkiewicz, B.; Milenin, A. Microstructure Transformations in Tempering Steels during Continuous Cooling from Hot Forging Temperatures. Steel Res. Int. 2010, 81, 224–233. [Google Scholar] [CrossRef]
- Norton, F.H. The Creep of Steel at High Temperatures, 1st ed.; McGraw-Hill Book Company, Inc.: New York, NY, USA, 1929. [Google Scholar]
- Rößler, C.; Schmicker, D.; Naumenko, K.; Woschke, E. Adaption of a Carreau fluid law formulation for residual stress determination in rotary friction welds. J. Mater. Process. Technol. 2017. [Google Scholar] [CrossRef]
- Prandtl, L. Über die Härte plastischer Körper. Nachrich. Ges. Der Wiss. GÖTtingen-Math.-Phys. Kl. 1920, 1920, 74–85. [Google Scholar]
- Saeed, I. Untersuchungen über die Streuung und Anwendung von Fließkurven. In Fortschrittberichte der VDI-Zeitschriften; Grund- und Werkstoffe, VDI-Verlag: Düsseldorf, Germany, 1984; pp. 204–215. [Google Scholar]
- Ion, J.C.; Easterling, K.E.; Ashby, M.F. A second report on diagrams of microstructure and hardness for heat-affected zones in welds. Acta Metall. 1984, 32, 1949–1962. [Google Scholar] [CrossRef]
- Leblond, J.B.; Devaux, J. A new kinetic model for anisothermal metallurgical transformations in steels including effect of austenite grain size. Acta Metall. 1984, 32, 137–146. [Google Scholar] [CrossRef]
- Jüttner, S.; Körner, M. Entwicklung eines Reibgesetzes zur Erfassung des Drehzahleinflusses bei der Reibschweißprozesssimulation; Otto von Guericke University Library: Magdeburg, Germany, 2019. [Google Scholar] [CrossRef]
- Seyffarth, P.; Meyer, B.; Scharff, A. Großer Atlas Schweiss-ZTU-Schaubilder. In Fachbuchreihe Schweisstechnik; Dt. Verl. für Schweisstechnik DVS-Verl.: Düsseldorf, Germany, 1992; Volume 110. [Google Scholar]
| C | Si | Mn | P | S |
|---|---|---|---|---|
| 0.54 | 0.21 | 0.63 | 0.008 | 0.006 |
| Heating | Cooling | |
|---|---|---|
| ≥300 | ≤800 | |
| 1–2000 K/s | 5 K/s, 30 K/s | |
| 0.3 mm | ||
| 0.003–30 mm/s | 0.03–0.3 mm/s | |
| Heating | Cooling | ||
|---|---|---|---|
| 5 K/s | 30 K/s | ||
| 0.9084 | 0.9421 | 0.8830 | |
| Heating | Cooling | |||||
|---|---|---|---|---|---|---|
| 5 K/s | 30 K/s | |||||
| n | n | n | ||||
| [MPa] | [-] | [MPa] | [-] | [MPa] | [-] | |
| 200 | – | – | – | – | 440.4 | 4.13 |
| 250 | – | – | 728.3 | 13.26 | 315.7 | 3.88 |
| 300 | 774.3 | 32.93 | 706.8 | 13.03 | 232.8 | 3.69 |
| 350 | 774.3 | 32.93 | 670.3 | 12.64 | 215.1 | 3.64 |
| 400 | 759.3 | 32.01 | 648.4 | 12.41 | 196.4 | 3.59 |
| 450 | 669.9 | 27.16 | 530.6 | 11.19 | 165.9 | 3.49 |
| 500 | 523.3 | 20.90 | 399.8 | 9.82 | 129.9 | 3.36 |
| 550 | 381.1 | 16.14 | 274.0 | 8.44 | 102.8 | 3.24 |
| 600 | 285.0 | 13.35 | 178.9 | 7.28 | 84.6 | 3.15 |
| 650 | 207.0 | 11.21 | 118.6 | 6.43 | 68.8 | 3.06 |
| 700 | 147.1 | 9.58 | 93.1 | 6.02 | 56.0 | 2.98 |
| 750 | 97.2 | 8.14 | 74.6 | 5.68 | 46.0 | 2.90 |
| 800 | 73.0 | 7.37 | 56.9 | 5.32 | ||
| 850 | 59.6 | 6.91 | ||||
| 900 | 48.6 | 6.50 | ||||
| 950 | 39.4 | 6.13 | ||||
| 1000 | 32.7 | 5.83 | ||||
| 1050 | 26.5 | 5.53 | ||||
| 1100 | 22.0 | 5.29 | ||||
| 1150 | 18.4 | 5.08 | ||||
| 1200 | 17.0 | 4.98 | ||||
| 1250 | 13.5 | 4.74 | ||||
| 1300 | 10.1 | 4.47 | ||||
| Heating | Cooling | |||
|---|---|---|---|---|
| 5 K/s | 30 K/s | |||
| [HV] | 322 | 311 | 733 | |
| [] | – | 690 | 305 | |
| [] | 720 | |||
| [] | 850 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rößler, C.; Schmicker, D.; Sherepenko, O.; Halle, T.; Körner, M.; Jüttner, S.; Woschke, E. Identification of the Flow Properties of a 0.54% Carbon Steel during Continuous Cooling. Metals 2020, 10, 104. https://doi.org/10.3390/met10010104
Rößler C, Schmicker D, Sherepenko O, Halle T, Körner M, Jüttner S, Woschke E. Identification of the Flow Properties of a 0.54% Carbon Steel during Continuous Cooling. Metals. 2020; 10(1):104. https://doi.org/10.3390/met10010104
Chicago/Turabian StyleRößler, Christoph, David Schmicker, Oleksii Sherepenko, Thorsten Halle, Markus Körner, Sven Jüttner, and Elmar Woschke. 2020. "Identification of the Flow Properties of a 0.54% Carbon Steel during Continuous Cooling" Metals 10, no. 1: 104. https://doi.org/10.3390/met10010104
APA StyleRößler, C., Schmicker, D., Sherepenko, O., Halle, T., Körner, M., Jüttner, S., & Woschke, E. (2020). Identification of the Flow Properties of a 0.54% Carbon Steel during Continuous Cooling. Metals, 10(1), 104. https://doi.org/10.3390/met10010104






