Short-Time β-Alanine Supplementation on the Acute Strength Performance after High-Intensity Intermittent Exercise in Recreationally Trained Men
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
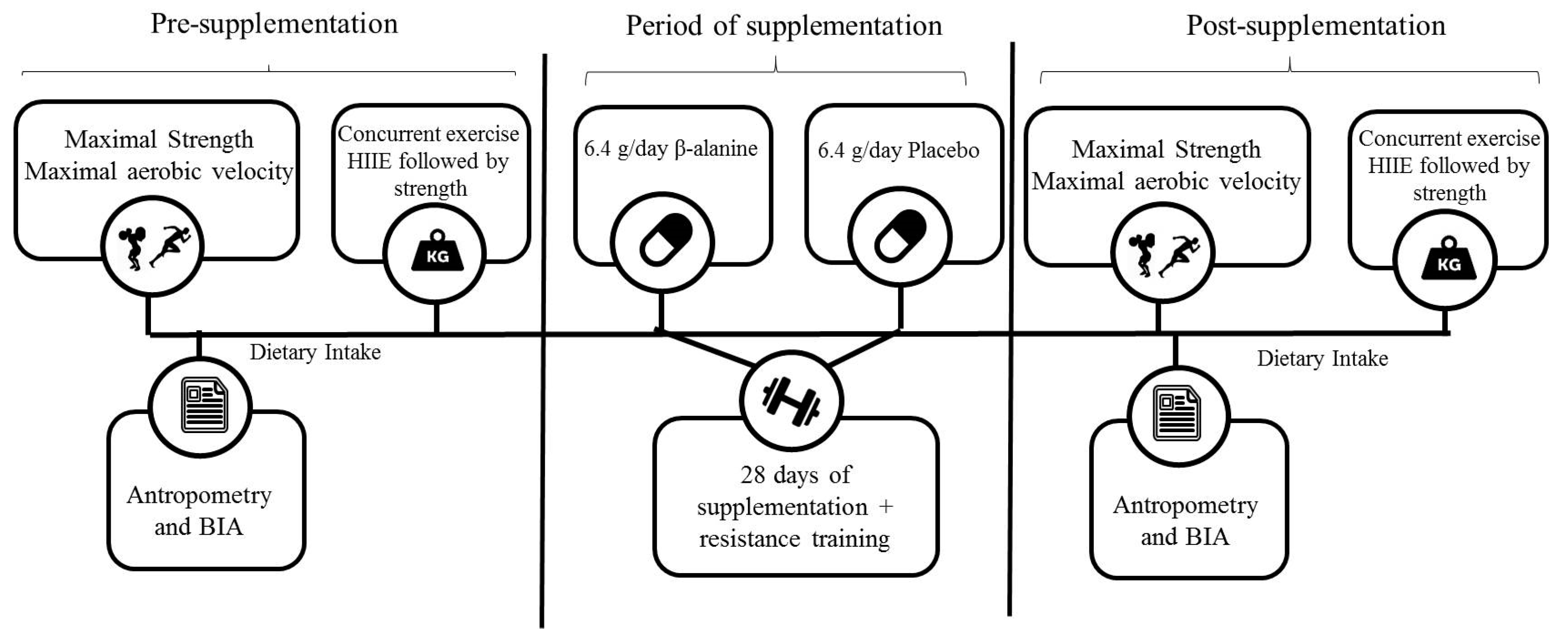
2.2. Experimental Design
2.3. Procedures
2.3.1. Maximal Aerobic Velocity and One-Maximum Repetition Test
2.3.2. Acute Concurrent Exercise Session
2.3.3. Resistance Training Protocol
2.3.4. Anthropometry, Body Composition, and Dietary Intake Assessment
2.3.5. Supplementation Procedure
2.3.6. Blood Lactate Concentration and Analysis
2.3.7. Statistical Analyses
3. Results
3.1. Maximal Strength and Maximal Aerobic Velocity Results
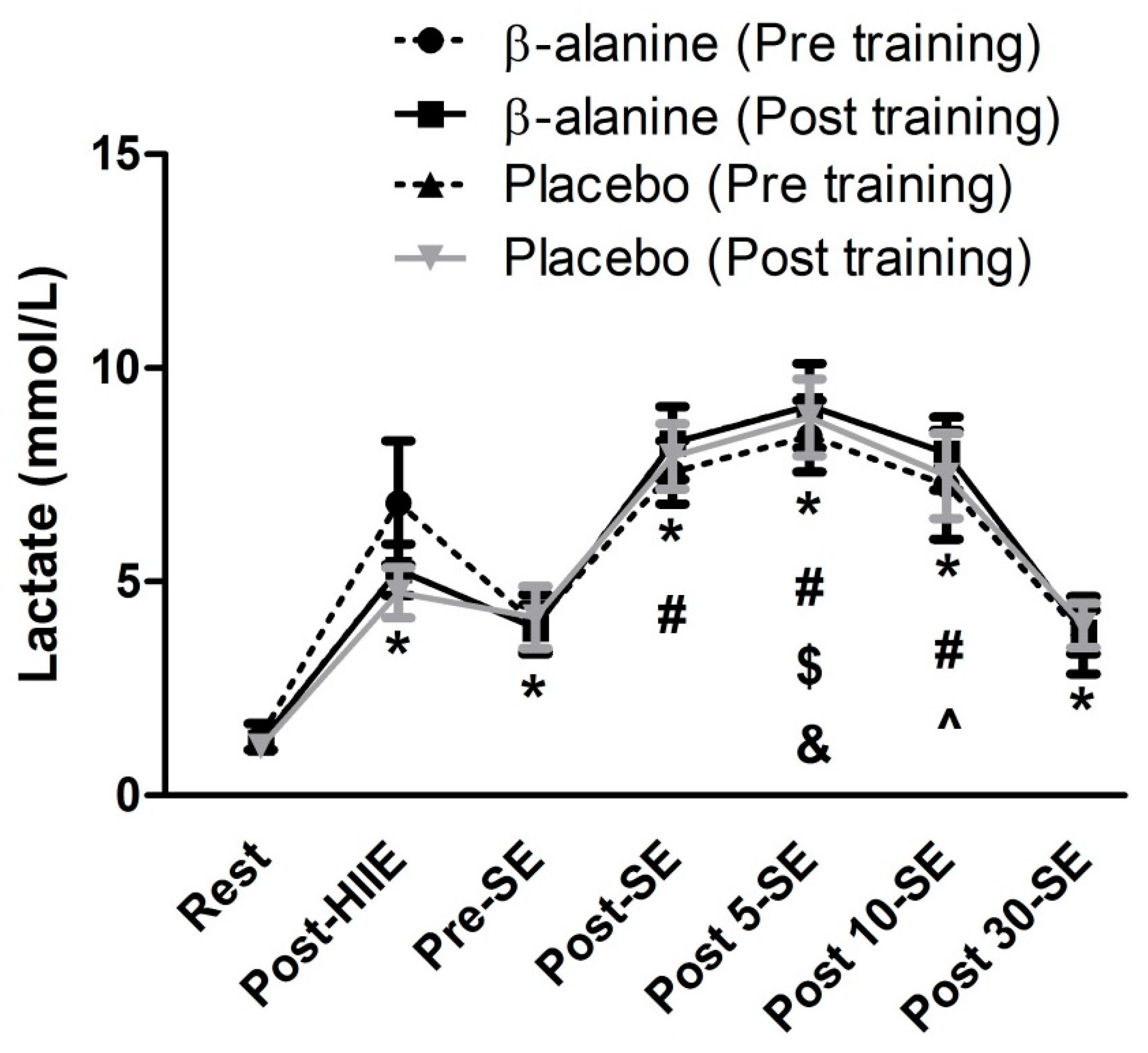
3.2. Acute Concurrent Exercise Results
3.3. Body Composition Results
3.4. Resistance Training Volume
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- de Souza, E.O.; Tricoli, V.; Franchini, E.; Paulo, A.C.; Regazzini, M.; Ugrinowitsch, C. Acute effect of two aerobic exercise modes on maximum strength and strength endurance. J. Strength. Cond. Res. 2007, 21, 1286–1290. [Google Scholar]
- Inoue, D.S.; Panissa, V.L.; Monteiro, P.A.; Gerosa-Neto, J.; Rossi, F.E.; Antunes, B.M.; Franchini, E.; Cholewa, J.M.; Gobbo, L.A.; Lira, F.S. Immunometabolic Responses to Concurrent Training: The Effects of Exercise Order in Recreational Weightlifters. J. Strength. Cond. Res. 2016, 30, 1960–1967. [Google Scholar]
- Leveritt, M.; Abernethy, P.J.; Barry, B.K.; Logan, P.A. Concurrent strength and endurance training. A review. Sports Med. 1999, 28, 413–427. [Google Scholar] [CrossRef] [PubMed]
- Panissa, V.L.; Tricoli, V.A.; Julio, U.F.; Ribeiro, N.; de Azevedo Neto, R.M.; Carmo, E.C.; Franchini, E. Acute effect of high-intensity aerobic exercise performed on treadmill and cycle ergometer on strength performance. J. Strength. Cond. Res. 2015, 29, 1077–1082. [Google Scholar] [CrossRef]
- Craig, B.W.; Lucas, J.; Pohlman, R.; Stelling, H. The Effects of Running, Weightlifting and a Combination of Both on Growth Hormone Release. J. Strength. Cond. Res. 1991, 5, 198–203. [Google Scholar]
- Ribeiro, N.; Ugrinowitsch, C. Acute effects of aerobic exercise performed with different volumes on strength performance and neuromuscular parameters. Eur. J. Sport Sci. 2018, 19, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Leveritt, M.; Abernethy, P.J. Acute effects of high-intensity endurance exercise on subsequent resistance activity. J. Strength. Cond. Res. 1999, 13, 47–51. [Google Scholar]
- Abernethy, P.J. Influence of acute endurance activity on isokinetic strength. J. Strength. Cond. Res. 1993, 7, 141–146. [Google Scholar]
- de Salles Painelli, V.; Alves, V.T.; Ugrinowitsch, C.; Benatti, F.B.; Artioli, G.G.; Lancha, A.H., Jr.; Gualano, B.; Roschel, H. Creatine supplementation prevents acute strength loss induced by concurrent exercise. Eur. J. Appl. Physiol. 2014, 114, 1749–1755. [Google Scholar] [CrossRef]
- Perez-Schindler, J.; Hamilton, D.L.; Moore, D.R.; Baar, K.; Philp, A. Nutritional strategies to support concurrent training. Eur. J. Sport Sci. 2015, 15, 41–52. [Google Scholar]
- Rossi, F.E.; Panissa, V.L.G.; Monteiro, P.A.; Gerosa-Neto, J.; Caperuto, E.C.; Cholewa, J.M.; Zagatto, A.M.; Lira, F.S. Caffeine supplementation affects the immunometabolic response to concurrent training. J. Exerc. Rehabil. 2017, 13, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Sale, C.; Saunders, B.; Harris, R.C. Effect of beta-alanine supplementation on muscle carnosine concentrations and exercise performance. Amino. Acids 2010, 39, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Saunders, B.; Elliott-Sale, K.; Artioli, G.G.; Swinton, P.A.; Dolan, E.; Roschel, H.; Sale, C.; Gualano, B. Beta-alanine supplementation to improve exercise capacity and performance: a systematic review and meta-analysis. Br. J. Sports Med. 2017, 51, 658–669. [Google Scholar] [CrossRef] [PubMed]
- Baguet, A.; Koppo, K.; Pottier, A.; Derave, W. Beta-alanine supplementation reduces acidosis but not oxygen uptake response during high-intensity cycling exercise. Eur. J. Appl. Physiol. 2010, 108, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Trexler, E.T.; Smith-Ryan, A.E.; Stout, J.R.; Hoffman, J.R.; Wilborn, C.D.; Sale, C.; Kreider, R.B.; Jäger, R.; Earnest, C.P.; Bannock, L.; et al. International society of sports nutrition position stand: Beta-Alanine. J. Int. Soc. Sports Nutr. 2015, 12, 30. [Google Scholar] [CrossRef] [PubMed]
- Cholewa, J.; Trexler, E.; Lima-Soares, F.; de Araujo Pessoa, K.; Sousa-Silva, R.; Santos, A.M.; Zhi, X.; Nicastro, H.; Cabido, C.E.T.; de Freitas, M.C.; et al. Effects of dietary sports supplements on metabolite accumulation, vasodilation and cellular swelling in relation to muscle hypertrophy: A focus on “secondary” physiological determinants. Nutrition 2018, 60, 241–251. [Google Scholar] [CrossRef]
- Kern, B.D.; Robinson, T.L. Effects of β-alanine supplementation on performance and body composition in collegiate wrestlers and football players. J. Strength. Cond. Res. 2011, 25, 1804–1815. [Google Scholar] [CrossRef]
- Outlaw, J.J.; Smith-Ryan, A.E.; Buckley, A.L.; Urbina, S.L.; Hayward, S.; Wingfield, H.L.; Campbell, B.; Foster, C.; Taylor, L.W.; Wilborn, C.D. Effects of beta-Alanine on Body Composition and Performance Measures in Collegiate Women. J. Strength. Cond. Res. 2016, 30, 2627–2637. [Google Scholar] [CrossRef]
- Kendrick, I.P.; Kim, H.J.; Harris, R.C.; Kim, C.K.; Dang, V.H.; Lam, T.Q.; Bui, T.T.; Wise, J.A. The effect of 4 weeks beta-alanine supplementation and isokinetic training on carnosine concentrations in type I and II human skeletal muscle fibres. Eur. J. Appl. Physiol. 2009, 106, 131–138. [Google Scholar] [CrossRef]
- Kresta, J.Y.; Oliver, J.M.; Jagim, A.R.; Fluckey, J.; Riechman, S.; Kelly, K.; Meininger, C.; Mertens-Talcott, S.U.; Rasmussen, C.; Kreider, R.B. Effects of 28 days of beta-alanine and creatine supplementation on muscle carnosine, body composition and exercise performance in recreationally active females. J. Int. Soc. Sports Nutr. 2014, 11, 55. [Google Scholar] [CrossRef]
- Harris, R.C.; Tallon, M.J.; Dunnett, M.; Boobis, L.; Coakley, J.; Kim, H.J.; Fallowfield, J.L.; Hill, C.A.; Sale, C.; Wise, J.A. The absorption of orally supplied beta-alanine and its effect on muscle carnosine synthesis in human vastus lateralis. Amino. Acids 2006, 30, 279–289. [Google Scholar] [CrossRef]
- Saunders, B.; Riani, L.; DE Salles Painelli, V.; DE Oliveira, L.F.; DA Eira Silva, V.; DA Silva, R.P.; Riani, L.; Franchi, M.; Gonçalves, L.S.; Harris, R.C.; et al. Twenty-four weeks of beta-alanine supplementation on carnosine content, related genes, and exercise. Med. Sci. Sports Exerc. 2017, 49, 896–906. [Google Scholar] [CrossRef]
- Kuipers, H.; Verstappen, F.T.; Keizer, H.A.; Geurten, P.; van Kranenburg, G. Variability of aerobic performance in the laboratory and its physiologic correlates. Int. J. Sports Med. 1985, 6, 197–201. [Google Scholar] [CrossRef]
- Rossi, F.E.; Gerosa-Neto, J.; Zanchi, N.E.; Cholewa, J.M.; Lira, F.S. Impact of short and moderate rest intervals on the acute immunometabolic response to exhaustive strength exercise: Part I. J. Strength. Cond. Res. 2016, 30, 1563–1569. [Google Scholar] [CrossRef]
- Wang, Z.M.; Pierson, R.N., Jr.; Heymsfield, S.B. The five-level model: a new approach to organizing body-composition research. Am. J. Clin. Nutr. 1992, 56, 19–28. [Google Scholar] [CrossRef]
- Miot, H.A. Tamanho da amostra em estudos clínicos e experimentais. J. Vascul. Brasileiro 2011, 10, 275–278. [Google Scholar] [CrossRef]
- Fabricatore, A.N.; Wadden, T.A.; Moore, R.H.; Butryn, M.L.; Gravallese, E.A.; Erondu, N.E.; Heymsfield, S.B.; Nguyen, A.M. Attrition from randomized controlled trials of pharmacological weight loss agents: A systematic review and analysis. Obes. Rev. 2009, 10, 333–341. [Google Scholar] [CrossRef]
- Bassinello, D.; de Salles Painelli, V.; Dolan, E.; Lixandrao, M.; Cajueiro, M.; de Capitani, M.; Saunders, B.; Sale, C.; Artioli, G.G.; Gualano, B.; et al. Beta-alanine supplementation improves isometric, but not isotonic or isokinetic strength endurance in recreationally strength-trained young men. Amino. Acids 2018, 51, 27–37. [Google Scholar] [CrossRef]
- Mannion, A.F.; Jakeman, P.M.; Dunnett, M.; Harris, R.C.; Willan, P.L. Carnosine and anserine concentrations in the quadriceps femoris muscle of healthy humans. Eur. J. Appl. Physiol. Occup. Physiol. 1992, 64, 47–50. [Google Scholar] [CrossRef]
- Hoffman, J.; Ratamess, N.; Kang, J.; Mangine, G.; Faigenbaum, A.; Stout, J. Effect of creatine and beta-alanine supplementation on performance and endocrine responses in strength/power athletes. Int. J. Sport Nutr. Exerc. Metab. 2006, 16, 430–446. [Google Scholar] [CrossRef]
- Hoffman, J.; Ratamess, N.A.; Ross, R.; Kang, J.; Magrelli, J.; Neese, K.; Faigenbaum, A.D.; Wise, J.A. Beta-alanine and the hormonal response to exercise. Int. J. Sports Med. 2008, 29, 952–958. [Google Scholar] [CrossRef]
- Stellingwerff, T.; Decombaz, J.; Harris, R.C.; Boesch, C. Optimizing human in vivo dosing and delivery of beta-alanine supplements for muscle carnosine synthesis. Amino. Acids 2012, 43, 57–65. [Google Scholar] [CrossRef]
- Hobson, R.M.; Saunders, B.; Ball, G.; Harris, R.C.; Sale, C. Effects of beta-alanine supplementation on exercise performance: A meta-analysis. Amino. Acids 2012, 43, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Morales-Alamo, D.; Losa-Reyna, J.; Torres-Peralta, R.; Martin-Rincon, M.; Perez-Valera, M.; Curtelin, D.; Ponce-González, J.G.; Santana, A.; Calbet, J.A. What limits performance during whole-body incremental exercise to exhaustion in humans? J. Physiol. 2015, 593, 4631–4648. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.E.; Walter, A.A.; Graef, J.L.; Kendall, K.L.; Moon, J.R.; Lockwood, C.M.; Fukuda, D.H.; Beck, T.W.; Cramer, J.T.; Stout, J.R. Effects of beta-alanine supplementation and high-intensity interval training on endurance performance and body composition in men; a double-blind trial. J. Int. Soc. Sports Nutr. 2009, 6, 5. [Google Scholar] [CrossRef]
- Kendrick, I.P.; Harris, R.C.; Kim, H.J.; Kim, C.K.; Dang, V.H.; Lam, T.Q.; Bui, T.T.; Smith, M.; Wise, J.A. The effects of 10 weeks of resistance training combined with beta-alanine supplementation on whole body strength, force production, muscular endurance and body composition. Amino. Acids 2008, 34, 547–554. [Google Scholar] [CrossRef]
- Schoenfeld, B.J.; Ogborn, D.; Krieger, J.W. Dose-response relationship between weekly resistance training volume and increases in muscle mass: A systematic review and meta-analysis. J. Sports. Sci. 2017, 35, 1073–1082. [Google Scholar] [CrossRef]
- Ohno, Y.; Oyama, A.; Kaneko, H.; Egawa, T.; Yokoyama, S.; Sugiura, T.; Ohira, Y.; Yoshioka, T.; Goto, K. Lactate increases myotube diameter via activation of MEK/ERK pathway in C2C12 cells. Acta Physiol. 2018, 223, e13042. [Google Scholar] [CrossRef] [PubMed]
- Ohno, Y.; Ando, K.; Ito, T.; Suda, Y.; Matsui, Y.; Oyama, A.; Kaneko, H.; Yokoyama, S.; Egawa, T.; Goto, K. Lactate Stimulates a Potential for Hypertrophy and Regeneration of Mouse Skeletal Muscle. Nutrients 2019, 11, 869. [Google Scholar] [CrossRef]
- Deldicque, L.; Theisen, D.; Bertrand, L.; Hespel, P.; Hue, L.; Francaux, M. Creatine enhances differentiation of myogenic C2C12 cells by activating both p38 and Akt/PKB pathways. Am. J. Physiol. Cell Physiol. 2007, 293, 1263–1271. [Google Scholar] [CrossRef]
- Mate-Munoz, J.L.; Lougedo, J.H.; Garnacho-Castano, M.V.; Veiga-Herreros, P.; Lozano-Estevan, M.D.C.; Garcia-Fernandez, P.; de Jesús, F.; Guodemar-Pérez, J.; San Juan, A.F.; Domínguez, R. Effects of beta-alanine supplementation during a 5-week strength training program: a randomized, controlled study. J. Int. Soc. Sports Nutr. 2018, 15, 19. [Google Scholar] [CrossRef] [PubMed]
| Variables | Group | Pre | Post | Effect Size |
|---|---|---|---|---|
| Maximal strength (kg) | β-alanine | 340 ± 61 | 410 ± 71 * | 1.05 |
| Placebo | 317 ± 67 | 368 ± 77 * | 0.70 | |
| Maximal strength relative to body mass (kg/kg) | β-alanine | 4.2 ± 0.5 | 5.1 ± 0.6 * | 1.63 |
| Placebo | 4.1 ± 0.6 | 4.8 ± 0.7 * | 1.08 | |
| Maximal aerobic velocity (km/h) | β-alanine | 13.9 ± 1.0 | 13.9 ± 1.1 | 0.00 |
| Placebo | 13.2 ± 1.1 | 13.3 ± 1.0 | 0.10 | |
| Volume performed in leg press (kg) | β-alanine | 8754 ± 4179 | 11922 ± 3494 * | 0.82 |
| Placebo | 7376 ± 3394 | 10953 ± 4162 * | 0.95 | |
| Volume performed in leg press relative to body mass (kg/kg) | β-alanine | 107.2 ± 46.1 | 145.6 ± 33.6 * | 0.95 |
| Placebo | 96.2 ± 37.7 | 142.9 ± 46.3 * | 1.11 | |
| Body mass (kg) | β-alanine | 80.1 ± 8.0 | 80.8 ± 8.1 * | 0.08 |
| Placebo | 77.3 ± 15.8 | 77.5 ± 15.4 * | 0.01 | |
| Fat-free mass (kg) | β-alanine | 64.0 ± 5.7 | 65.8 ± 6.3 * | 0.30 |
| Placebo | 62.4 ± 9.2 | 63.1 ± 10.0 * | 0.07 | |
| Fat mass (kg) | β-alanine | 19.8 ± 6.5 | 19.5 ± 7.5 | 0.04 |
| Placebo | 19.6 ± 11.9 | 19.3 ± 11.7 | 0.03 | |
| Body fat (%) | β-alanine | 18.9 ± 5.7 | 17.8 ± 5.1 | −0.20 |
| Placebo | 17.3 ± 6.8 | 19.3 ± 11.7 | 0.22 | |
| Extracellular water (L) | β-alanine | 18.0 ± 1.8 | 18.6 ± 2.0 * | 0.32 |
| Placebo | 17.9 ± 2.7 | 18.1 ± 3.1 * | 0.07 | |
| Intracellular water (L) | β-alanine | 28.3 ± 2.9 | 29.1 ± 3.2 * | 0.26 |
| Placebo | 28.2 ± 4.7 | 28.4 ± 5.3 * | 0.04 | |
| Total body water (L) | β-alanine | 46.4 ± 4.8 | 47.7 ± 5.2 * | 0.26 |
| Placebo | 44.8 ± 6.7 | 45.0 ± 7.6 * | 0.03 | |
| Resistance (ohms) | β-alanine | 457.1 ± 32.9 | 449.7 ± 33.3 | 0.24 |
| Placebo | 477.1 ± 56.4 | 470.9 ± 70.8 | 0.10 | |
| Reactance (ohms) | β-alanine | 52.3 ± 6.4 | 51.1 ± 8.0 | 0.16 |
| Placebo | 53.8 ± 6.3 | 52.2 ± 7.1 | 0.24 |
| Period | Group | Total Weekly Volume (kg) | Effect Size |
|---|---|---|---|
| Week 1 * | Beta | 27.048 ± 5582 | NA |
| Placebo | 24.499 ± 6053 | NA | |
| Week 2 # | Beta | 32.705 ± 5484 | 1.02 |
| Placebo | 27.958 ± 5759 | 0.59 | |
| Week 3 | Beta | 33.521 ± 5386 | 1.18 |
| Placebo | 29.370 ± 5871 | 0.82 | |
| Week 4 | Beta | 35.323 ± 5631 | 1.48 |
| Placebo | 29.574 ± 5283 | 0.90 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freitas, M.C.; Cholewa, J.; Panissa, V.; Quizzini, G.; de Oliveira, J.V.; Figueiredo, C.; Gobbo, L.A.; Caperuto, E.; Zanchi, N.E.; Lira, F.; et al. Short-Time β-Alanine Supplementation on the Acute Strength Performance after High-Intensity Intermittent Exercise in Recreationally Trained Men. Sports 2019, 7, 108. https://doi.org/10.3390/sports7050108
Freitas MC, Cholewa J, Panissa V, Quizzini G, de Oliveira JV, Figueiredo C, Gobbo LA, Caperuto E, Zanchi NE, Lira F, et al. Short-Time β-Alanine Supplementation on the Acute Strength Performance after High-Intensity Intermittent Exercise in Recreationally Trained Men. Sports. 2019; 7(5):108. https://doi.org/10.3390/sports7050108
Chicago/Turabian StyleFreitas, Marcelo Conrado, Jason Cholewa, Valéria Panissa, Giovanni Quizzini, João Vitor de Oliveira, Caique Figueiredo, Luis Alberto Gobbo, Erico Caperuto, Nelo Eidy Zanchi, Fabio Lira, and et al. 2019. "Short-Time β-Alanine Supplementation on the Acute Strength Performance after High-Intensity Intermittent Exercise in Recreationally Trained Men" Sports 7, no. 5: 108. https://doi.org/10.3390/sports7050108
APA StyleFreitas, M. C., Cholewa, J., Panissa, V., Quizzini, G., de Oliveira, J. V., Figueiredo, C., Gobbo, L. A., Caperuto, E., Zanchi, N. E., Lira, F., & Rossi, F. E. (2019). Short-Time β-Alanine Supplementation on the Acute Strength Performance after High-Intensity Intermittent Exercise in Recreationally Trained Men. Sports, 7(5), 108. https://doi.org/10.3390/sports7050108









