Epigenetic Modifications in Generalized Autoimmune Epithelitis: Sjögren’s Syndrome and Primary Biliary Cholangitis
Abstract
:1. Introduction
2. Epigenetic Modifications in SjS and PBC
2.1. DNA Methylation/Hydroxymethylation
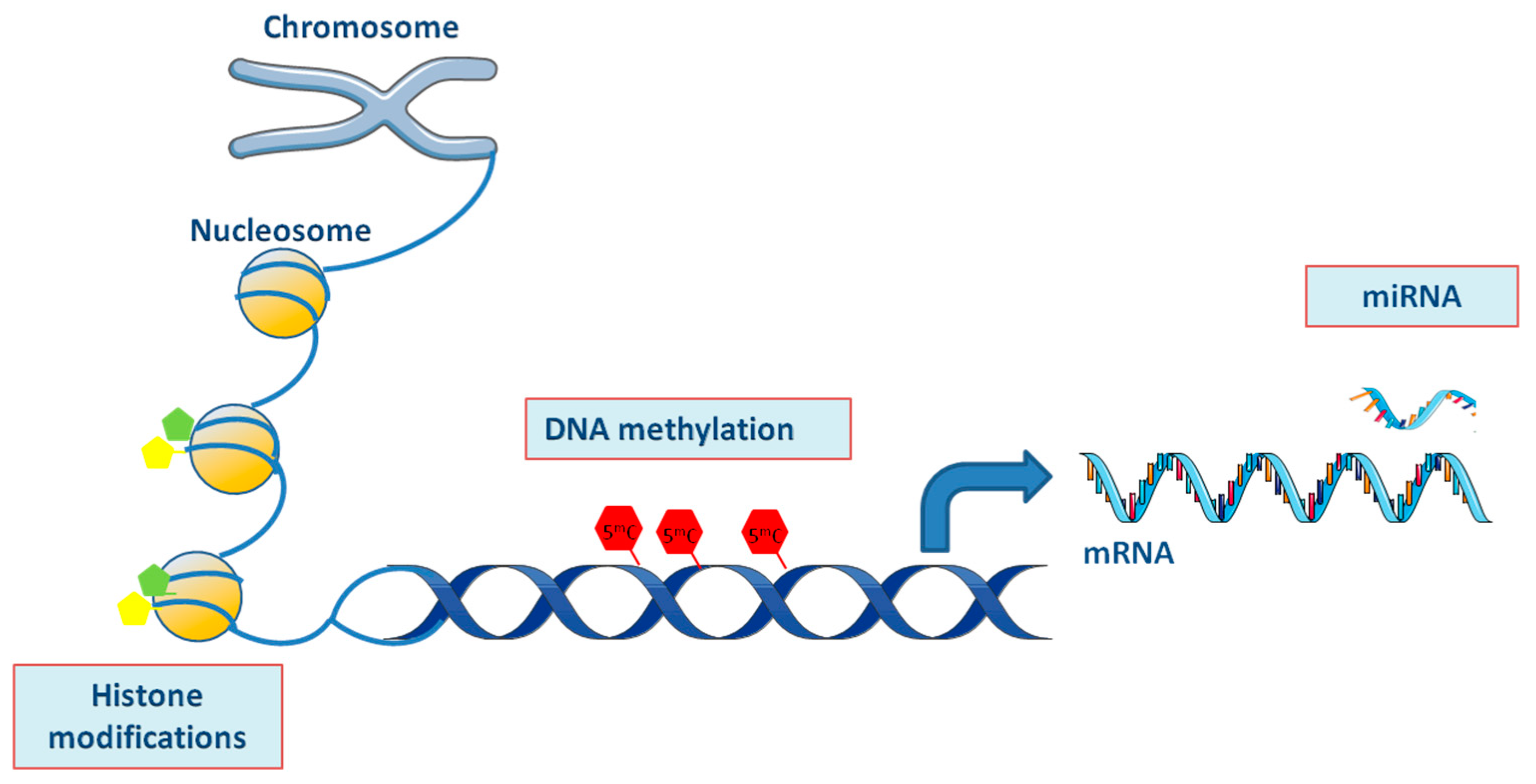
Description
2.2. DNA Methylation/Hydroxymethylation in SjS and PBC
2.3. Genome-Wide Analysis of DNA Methylation in SjS and PBC
2.4. Histone Modifications
Description
2.5. Histone Modifications in SjS and PBC
2.6. Micro-RNAs (miRNAs)
Description
2.7. miRNAs in SjS and PBC
3. Overview and Future Perspectives
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| SjS | Sjögren’s Syndrome |
| PBC | primary biliary cholangitis |
| ANA | antinuclear antibodies |
| AMA | antimitochondrial antibodies |
| SGECs | salivary glands epithelial cells |
| 5mC | 5-methylcytosine |
| DNMT | DNA methyltransferase |
| TET | ten eleven translocation methylcytosine dioxygenase |
| MBDs | methyl binding domains |
| MeCP | methyl-CpG-binding proteins |
| LINE-1 | long interspersed nuclear element |
| 5hmC | 5-hydroxymethylcytosine |
| IFN | interferon |
| GWAS | genome-wide association studies |
| CXCR3 | C-X-C motif chemokine receptor 3 |
| EWAS | epigenome-wide association studies |
| DMCs | differentially methylated CpGs |
| HLA-B | human leucocyte antigen B |
| IFI44L | interferon-induced protein 44 like |
| IFIT1 | interferon-induced protein with tetratricopeptide repeats 1 |
| SMARCA1 | SWI/SNF-related, matrix-associated, actin-dependent regulator of chromatin, subfamily A, member 1 |
| IL-6 | interleukin 6 |
| LIGHT | tumor necrosis factor superfamily member 14 |
| IL-17 | interleukin 17 |
| PBMCs | peripheral blood mononuclear cells |
| TRAIL | TNF-related apoptosis-inducing ligand |
| miRNAs | Micro-RNAs |
| UTR | untranslated region |
References
- Skopouli, F.N.; Moutsopoulos, H.M. Autoimmune epitheliitis: Sjogren’s syndrome. Clin. Exp. Rheumatol. 1994, 12, 9–11. [Google Scholar]
- Gao, J.; Liang Qiao, L.; Wang, B. Primary Biliary Cirrhosis is a Generalized Autoimmune Epithelitis. Int. J. Mol. Sci. 2015, 16, 6432–6446. [Google Scholar] [CrossRef]
- Selmi, C.; Gershwin, M.E. Chronic Autoimmune Epithelitis in Sjögren’s Syndrome and Primary Biliary Cholangitis: A Comprehensive Review. Rheumatol. Ther. 2017, 4, 263–279. [Google Scholar] [CrossRef]
- Carey, E.J.; Ali, A.H.; Lindor, K.D. Primary biliary cirrhosis. Lancet 2015, 386, 1565–1575. [Google Scholar] [CrossRef]
- Dalekos, G.N.; Lygoura, V.; Gatselis, N.K. Variants of primary biliary cholangitis: An updated mini review. J. Autoimmune Dis. Rheumatol. 2018, 6, 21–28. [Google Scholar] [CrossRef]
- Dalekos, G.N.; Gatselis, N.K. Variant and specific forms of autoimmune cholestatic liver diseases: A short review. Arch. Immunol. Ther. Exp. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Gatselis, N.K.; Zachou, K.; Lygoura, V.; Azariadis, K.; Arvaniti, P.; Spyrou, E.; Papadamou, G.; Koukoulis, G.K.; Dalekos, G.N.; Rigopoulou, E.I. Geoepidemiology, clinical manifestations and outcome of primary biliary cholangitis in Greece. Eur. J. Intern. Med. 2017, 42, 81–88. [Google Scholar] [CrossRef]
- Griffiths, L.; Dyson, J.K.; Jones, D.E. The new epidemiology of primary biliary cirrhosis. Semin. Liver Dis. 2014, 34, 318–328. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines: The diagnosis and management of primary biliary cholangitis. J. Hepatol. 2017, 67, 145–172. [Google Scholar] [CrossRef]
- Hirschfield, G.M.; Dyson, J.K.; Alexander, G.J.; Chapman, M.H.; Collier, J.; Hübscher, S.; Patanwala, I.; Pereira, S.P.; Thain, C.; Thorburn, D.; et al. The British Society of Gastroenterology/UK-PBC primary biliary cholangitis treatment andmanagement guidelines. Gut 2018, 67, 1568–1594. [Google Scholar] [CrossRef]
- Stefanidis, I.; Giannopoulou, M.; Liakopoulos, V.; Dovas, S.; Karasavvidou, F.; Zachou, K.; Koukoulis, G.K.; Dalekos, G.N. A case of membranous nephropathy associated with Sjögren syndrome, polymyositis and autoimmune hepatitis. Clin. Nephrol. 2008, 70, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Mavragani, C.P.; Moutsopoulos, H.M. Sjogren’s syndrome. Annu. Rev. Pathol. 2014, 9, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Casals, M.; Brito-Zeron, P.; Siso-Almirall, A.; Bosch, X. Primary Sjӧgren syndrome. BMJ 2012, 344, 3821. [Google Scholar] [CrossRef] [PubMed]
- Agmon-Levin, N.; Shapira, Y.; Selmi, C.; Barzilai, O.; Ram, M.; Szyper-Kravitz, M.; Sella, S.; Katz, BS.; Youinou, P.; Renaudineau, Y.; et al. A comprehensive evaluation of serum autoantibodies in primary biliary cirrhosis. J. Autoimmun. 2010, 34, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Tzioufas, A.G.; Tatouli, I.P.; Moutsopoulos, H.M. Autoantibodies in Sjögren’s syndrome: Clinical presentation and regulatory mechanisms. Presse Med. 2012, 41, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Gatselis, N.K.; Dalekos, G.N. Molecular diagnostic testing for primary biliary cholangitis. Expert Rev. Mol. Diagn. 2016, 16, 1001–1010. [Google Scholar] [CrossRef] [PubMed]
- Gatselis, N.K.; Zachou, K.; Norman, G.L.; Gabeta, S.; Papamichalis, P.; Koukoulis, G.K.; Dalekos, G.N. Clinical significance of the fluctuation of primary biliary cirrhosis-related autoantibodies during the course of the disease. Autoimmunity 2013, 46, 471–479. [Google Scholar] [CrossRef]
- Rigopoulou, E.I.; Davies, E.T.; Pares, A.; Zachou, K.; Liaskos, C.; Bogdanos, DP.; Rodes, J.; Dalekos, GN.; Vergani, D. Prevalence and clinical significance of isotype specific antinuclear antibodies in primary biliary cirrhosis. Gut 2005, 54, 528–532. [Google Scholar] [CrossRef] [Green Version]
- Rigopoulou, E.I.; Dalekos, G.N. Anti-sp100 antibodies in primary biliary cirrhosis. Scand. J. Gastroenterol. 2004, 39, 406–407. [Google Scholar] [CrossRef]
- Nakamura, M.; Kondo, H.; Mori, T.; Komori, A.; Matsuyama, M.; Ito, M.; Takii, Y.; Koyabu, M.; Yokoyama, T.; Migita, K. Anti-gp210 and anti-centromere antibodies are different risk factors for the progression of primary biliary cirrhosis. Hepatology 2007, 45, 118–127. [Google Scholar] [CrossRef]
- Bogdanos, D.P.; Liaskos, C.; Pares, A.; Norman, G.; Rigopoulou, E.I.; Caballeria, L.; Dalekos, G.N.; Rodes, J.; Vergani, D. Anti-gp210 antibody mirrors disease severity in primary biliary cirrhosis. Hepatology 2007, 45, 1583–1584. [Google Scholar] [CrossRef] [PubMed]
- Granito, A.; Muratori, P.; Quarneti, C.; Pappas, G.; Cicola, R.; Muratori, L. Antinuclear antibodies as ancillary markers in primary biliary cirrhosis. Expert Rev. Mol. Diagn. 2012, 12, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Christodoulou, M.I.; Kapsogeorgou, E.K.; Moutsopoulos, H.M. Characteristics of the minor salivary gland infiltrates in Sjogren’s syndrome. J. Autoimmun. 2010, 34, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Kapsogeorgou, E.K.; Christodoulou, M.I.; Panagiotakos, D.B.; Paikos, S.; Tassidou, A.; Tzioufas, A.G.; Moutsopoulos, H.M. Minor salivary gland inflammatory lesions in Sjogren syndrome: Do they evolve? J. Rheumatol. 2013, 40, 1566–1571. [Google Scholar] [CrossRef] [PubMed]
- Scheuer, P.J. Ludwig Symposium on biliary disorders–part II. Pathologic features and evolution of primary biliary cirrhosis and primary sclerosing cholangitis. Mayo Clin. Proc. 1998, 73, 179–183. [Google Scholar] [CrossRef]
- Lindor, K.D.; Gershwin, E.M.; Poupon, R. Primary biliary cirrhosis. Hepatology 2009, 50, 291–308. [Google Scholar] [CrossRef] [PubMed]
- Gershwin, M.E.; Selmi, C.; Worman, H.J.; Gold, EB.; Watnik, M.; Utts, J.; Lindor, KD.; Kaplan, MM.; Vierling, JM.; USA PBC Epidemiology Group. Risk factors and comorbidities in primary biliary cirrhosis: A controlled interview-based study of 1032 patients. Hepatology 2005, 42, 1194–1202. [Google Scholar] [CrossRef] [Green Version]
- Hatzis, G.S.; Fragoulis, G.E.; Karatzaferis, A.; Delladetsima, I.; Barbatis, C.; Moutsopoulos, H.M. Prevalence and longterm course of primary biliary cirrhosis in primary Sjögren’s syndrome. J. Rheumatol. 2008, 35, 2012–2016. [Google Scholar]
- Corpechot, C.; Chretien, Y.; Chazouilleres, O.; Poupon, R. Demographic, lifestyle, medical and familial factors associated with primary biliary cirrhosis. J. Hepatol. 2010, 53, 162–169. [Google Scholar] [CrossRef]
- Floreani, A.; Franceschet, I.; Cazzagon, N.; Spinazzè, A.; Buja, A.; Furlan, P.; Baldo, V.; Gershwin, M.E. Extrahepatic autoimmune conditions associated with primary biliary cirrhosis. Clin. Rev. Allergy Immunol. 2015, 48, 192–197. [Google Scholar] [CrossRef]
- Tsianos, E.V.; Hoofnagle, J.H.; Fox, P.C.; Alspaugh, M.; Jones, E.A.; Schafer, D.F.; Moutsopoulos, H.M. Sjogren’s syndrome in patients with primary biliary cirrhosis. Hepatology 1990, 11, 730–734. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.I.; Fei, H.M.; Saito, I.; Sawada, S.; Chen, S.L.; Yi, D.; Chan, E.; Peebles, C.; Bugawan, T.L.; Erlich, H.A.; et al. Comparison of HLA class II genes in Caucasoid, Chinese, and Japanese patients with primary Sjogren’s syndrome. J. Immunol. 1993, 150, 3615–3623. [Google Scholar] [PubMed]
- Miyagawa, S.; Shinohara, K.; Nakajima, M.; Kidoguchi, K.; Fujita, T.; Fukumoto, T.; Yoshioka, A.; Dohi, K.; Shirai, T. Polymorphisms of HLA class II genes and autoimmune responses to Ro/SS-A-La/SS-B among Japanese subjects. Arthritis Rheum. 1998, 41, 927–934. [Google Scholar] [CrossRef]
- Lessard, C.J.; Li, H.; Adrianto, I.; Ice, J.A.; Rasmussen, A.; Grundahl, K.M.; Kelly, J.A.; Dozmorov, M.G.; Miceli-Richard, C.; Bowman, S. Variants at multiple loci implicated in both innate and adaptive immune responses are associated with Sjӧgren’s syndrome. Nat. Genet. 2013, 45, 1284–1292. [Google Scholar] [CrossRef] [PubMed]
- Gulamhusein, A.F.; Juran, B.D.; Lazaridis, K.N. Genome-wide association studies in primary biliary cirrhosis. Semin. Liver Dis. 2015, 35, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Leung, P.S.C.; Gershwin, M.E. Evolution of our understanding of PBC. Best Pract. Res. Clin. Gastroenterol. 2018, 34–35, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Leung, P.S.C.; Gershwin, M.E. Pathogen infections and primary biliary cholangitis. Clin. Exp. Immunol. 2019, 195, 25–34. [Google Scholar] [CrossRef]
- Probert, P.M.; Leitch, A.C.; Dunn, M.P.; Meyer, S.K.; Palmer, J.M.; Abdelghany, T.M.; Lakey, A.F.; Cooke, M.P.; Talbot, H.; Wills, C.; et al. Identification of a xenobiotic as a potential environmental trigger in primary biliary cholangitis. J. Hepatol. 2018, 69, 1123–1135. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, A.; Leung, P.S.C.; Gershwin, M.E. Environmental basis of primary biliary cholangitis. Exp. Biol. Med. 2018, 243, 184–189. [Google Scholar] [CrossRef]
- Bogdanos, D.P.; Dalekos, G.N. Enzymes as target antigens of liver-specific autoimmunity: The case of cytochromes P450. Curr. Med. Chem. 2008, 15, 2285–2292. [Google Scholar] [CrossRef]
- Triantafyllopoulou, A.; Tapinos, N.; Moutsopoulos, H.M. Evidence for coxsackievirus infection in primary Sjögren’s syndrome. Arthritis Rheum. 2004, 50, 2897–2902. [Google Scholar] [CrossRef] [PubMed]
- Renaudineau, Y.; Youinou, P. Epigenetics and autoimmunity, with special emphasis on methylation. Keio J. Med. 2011, 60, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Renaudineau, Y.; Cha, S.; Ilei, G.; Brooks, W.H.; Selmi, C.; Tzioufas, A.; Pers, J.O.; Bombardieri, S.; Gershwin, M.E.; et al. Epigenetics in autoimmune disorders: Highlights of the 10th Sjögren’s Syndrome Symposium. Autoimmun. Rev. 2010, 9, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, K.D.; Helin, K. Role of TET enzymes in DNA methylation, development, and cancer. Genes Dev. 2016, 30, 733–750. [Google Scholar] [CrossRef] [PubMed]
- Konsta, O.; Thabet, Y.; Le Dantec, C.; Brooks, W.H.; Tzioufas, A.G.; Pers, J.O.; Renaudineau, Y. The contribution of epigenetics in Sjögren’s syndrome. Front. Genet. 2014, 5, 71. [Google Scholar] [CrossRef] [PubMed]
- Renaudineau, Y.; Ballestar, E. Epigenetics: DNA methylation signatures in Sjögren syndrome. Nat. Rev. Rheumatol. 2016, 12, 565–566. [Google Scholar] [CrossRef] [PubMed]
- Thabet, Y.; Le Dantec, C.; Ghedira, I.; Devauchelle, V.; Cornec, D.; Pers, J.O.; Renaudineau, Y. Epigenetic dysregulation in salivary glands from patients with primary Sjogren’s syndrome may be ascribed to infiltrating B cells. J. Autoimmun. 2013, 41, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Bordron ACharras, A.; Le Dantec, C.; Renaudineau, Y. Influence of epigenetic in Sjögren’s syndrome. Rev. Med. Interne. 2018, 39, 346–351. [Google Scholar]
- Charras, A.; Konsta, O.D.; Le Dantec, C.; Bagacean, C.; Kapsogeorgou, E.K.; Tzioufas, A.G.; Pers, J.O.; Bordron, A.; Renaudineau, Y. Cell-specific epigenome-wide DNA methylation profile in long-term cultured minor salivary gland epithelial cells from patients with Sjögren’s syndrome. Ann. Rheum. Dis. 2017, 76, 625–628. [Google Scholar] [CrossRef]
- Tanaka, A.; Leung, P.S.C.; Gershwin, M.E. The Genetics and epigenetics of primary biliary cholangitis. Clin. Liver Dis. 2018, 22, 443–455. [Google Scholar] [CrossRef]
- Lleo, A.; Zhang, W.; Zhao, M.; Tan, Y.; Bernuzzi, F.; Zhu, B.; Liu, Q.; Tan, Q.; Malinverno, F.; Valenti, L.; et al. DNA methylation profiling of the X chromosome reveals an aberrant demethylation on CXCR3 promoter in primary biliary cirrhosis. Clin. Epigenetics 2015, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Marzorati, S.; Lleo, A.; Carbone, M.; Tan, Y.; Bernuzzi, F.; Zhu, B.; Liu, Q.; Tan, Q.; Malinverno, F.; Valenti, L.; et al. The epigenetics of PBC: The link between genetic susceptibility and environemt. Clin. Res. Hepatol. Gastroenterol. 2016, 40, 650–659. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.Q.; Ma, H.D.; Lian, Z.X. Epigenetics and primary biliary cirrhosis: A comprehensive review and implications for autoimmunity. Clin. Rev. Allergy Immunol. 2016, 50, 390–403. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Z.; Yang, Y.; Chang, C.; Lu, Q. The epigenetic mechanism for discordance of autoimmunity in monozygotic twins. J. Autoimmun. 2017, 83, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Rosa, R.; Cristoferi, L.; Tanaka, A.; Invernizzi, P. Geoepidemiology and (epi-) genetics in primary biliary cholangitis. Best Pract. Res. Clin. Gastroenterol. 2018, 34–35, 11–15. [Google Scholar]
- Joshita, S.; Umemura, T.; Tanaka, E.; Ota, M. Genetics and epigenetics in the pathogenesis of primary biliary cholangitis. Clin. J. Gastroenterol. 2018, 11, 11–18. [Google Scholar] [CrossRef]
- Le Dantec, C.; Gazeau, P.; Mukherjee, S. How the environment influences epigenetics, DNA methylation, and autoimmune diseases. In Epigenetics and Dermatology; Lu, Q., Chang, C.C., Richardson, B.C., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 467–485. [Google Scholar]
- Konsta, O.D.; Charras, A.; Le Dantec, C.; Kapsogeorgeou, E.; Bordron, A.; Brooks, W.H.; Tzioufas, A.G.; Pers, J.O.; Renaudineau, Y. Epigenetic modifications in salivary glands from patients with Sjögren’s syndrome affect cytokeratin 19 expression. Bull. Group. Int. Rech. Sci. Stomatol. Odontol. 2016, 53, 1–10. [Google Scholar]
- Konsta, O.D.; Le Dantec, C.; Charras, A.; Cornec, D.; Kapsogeorgou, E.K.; Tzioufas, A.G.; Pers, J.O.; Renaudineau, Y. Defective DNA methylation in salivary gland epithelial acini from patients with Sjogren’s syndrome is associated with SSB gene expression, anti-SSB/La detection, and lymphocyte infiltration. J. Autoimmun. 2016, 68, 30–38. [Google Scholar] [CrossRef]
- Cole, M.B.; Quach, H.; Quach, D.; Baker, A.; Taylor, K.E.; Barcellos, L.F.; Criswell, L.A. Epigenetic signatures of salivary gland inflammation in Sjögren’s syndrome. Arthritis Rheum. 2016, 68, 2936–2944. [Google Scholar] [CrossRef]
- Devauchelle-Pensec, V.; Pennec, Y.; Morvan, J.; Pers, J.O.; Daridon, C.; Jousse-Joulin, S.; Roudaut, A.; Jamin, C.; Renaudineau, Y.; Roué, I.Q.; et al. Improvement of Sjogren’s syndrome after two infusions of rituximab (anti-CD20). Arthritis Rheum. 2007, 57, 310–317. [Google Scholar] [CrossRef]
- Lagos, C.; Carvajal, P.; Castro, I.; Jara, D.; González, S.; Aguilera, S.; Barrera, M.J.; Quest, A.F.G.; Bahamondes, V.; Molina, C.; et al. Association of high 5-hydroxymethylcytosine levels with Ten Eleven Translocation 2 overexpression and inflammation in Sjögren’s syndrome patients. Clin. Immunol. 2018, 196, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Charras, A.; Arvaniti, P.; le Dantec, C.; Arleevskaya, M.I.; Zachou, K.; Dalekos, G.N.; Bordon, A.; Renaudineau, Y. JAK inhibitors suppress innate epigenetic reprogramming: A promise for patients with Sjögren’s syndrome. Clin. Rev. Allergy Immunol. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Mavragani, C.P.; Sagalovskiy, I.; Guo, Q.; Nezos, A.; Kapsogeorgou, E.K.; Lu, P.; Zhou, J.L.; Kirou, K.A.; Seshan, S.V.; Moutsopoulos, H.M.; et al. Long interspersed nuclear element-1 retroelements are expressed in patients with systemic autoimmune disease and induce type I interferon. Arthritis Rheumatol. 2016, 68, 2686–2696. [Google Scholar] [CrossRef] [PubMed]
- Mavragani, C.P.; Nezos, A.; Sagalovskiy, I.; Seshan, S.; Kirou, K.A.; Crow, M.K. Defective regulation of L1 endogenous retroelements in primary Sjogren’s syndrome and systemic lupus erythematosus: Role of methylating enzymes. J. Autoimmun. 2018, 88, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Lleo, A.; Liao, J.; Invernizzi, P.; Zhao, M.; Bernuzzi, F.; Ma, L.; Lanzi, G.; Ansari, A.A.; Coppel, R.L.; Zhang, P.; et al. Immunoglobulin M levels inversely correlate with CD40 ligand promoter methylation in patients with primary biliary cirrhosis. Hepatology 2012, 55, 153–160. [Google Scholar] [CrossRef]
- Cheung, A.C.; LaRusso, N.F.; Gores, G.J.; Lazaridis, K.N. Epigenetics in the primary biliary cholangitis and primary sclerosing cholangitis. Semin. Liver Dis. 2017, 37, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Huang, Y.; Liu, Y.; Sun, Y.; Zhou, Y.; Gu, M.; Chen, Y.; Xia, R.; Chen, S.; Deng, A.; et al. β-Arrestin 1 modulates functions of autoimmune T cells from primary biliary cirrhosis patients. J. ClinImmunol. 2011, 31, 346–355. [Google Scholar] [CrossRef]
- Selmi, C.; Cavaciocchi, F.; Lleo, A.; Cheroni, C.; De Francesco, R.; Lombardi, S.A.; De Santis, M.; Meda, F.; Raimondo, M.G.; Crotti, C.; et al. Genome-wide analysis of DNA methylation, copy number variation, and gene expression in monozygotic twins discordant for primary biliary cirrhosis. Front. Immunol. 2014, 5, 128. [Google Scholar] [CrossRef]
- Hall, J.C.; Baer, A.N.; Shah, A.A.; Criswell, L.A.; Shiboski, C.H.; Rosen, A.; Casciola-Rosen, L.l. Molecular subsetting of interferon pathways in Sjögren’s syndrome. Arthritis Rheumatol. 2015, 67, 2437–2446. [Google Scholar] [CrossRef]
- Imgenberg-Kreuz, J.; Sandling, J.K.; Carlsson Almlöf, J.; Nordlund, J.; Signér, L.; Norheim, K.B.; Omdal, R.; Rönnblom, L.; Eloranta, M.L.; Syvänen, A.C.; et al. Genome-wide DNA methylation analysis in multiple tissues in primary Sjögren’s syndrome reveals regulatory effects at interferon-induced genes. Ann. Rheum. Dis. 2016, 75, 2029–2036. [Google Scholar] [CrossRef]
- Richard, C.; Wang-Renault, S.; Boudaoud, S.; Busato, F.; Lallemand, C.; Bethune, K.; Belkhir, R.; Nocturne, G.; Mariette, X.; Tost, J. Overlap between differentially methylated DNA regions in blood B lymphocytes and genetic at-risk loci in primary Sjögren’s syndrome. Ann. Rheum. Dis. 2016, 75, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Konsta, O.D.; le Dantec, C.; Charras, A.; Brooks, W.H.; Arleevskaya, M.I.; Bordron, A.; Renaudineau, Y. An in silico approach reveals associations between genetic and epigenetic factors within regulatory elements in B cells from primary Sjögren’s syndrome patients. Front. Immunol. 2015, 6, 437. [Google Scholar] [CrossRef] [PubMed]
- Altorok, N.; Coit, P.; Hughes, T.; Koelsch, K.A.; Stone, D.U.; Rasmussen, A.; Radfar, L.; Scofield, R.H.; Sivils, K.L.; Farris, A.D.; et al. Genome-wide DNA methylation patterns in naive CD4+ T cells from patients with primary Sjögren’s syndrome. Arthritis Rheumatol. 2014, 66, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Zhao, M.; Wu, X.; Gao, F.; Luo, Y.; Ma, L.; Liu, S.; Zhang, G.; Chen, J.; Li, F.; et al. Hypomethylation and overexpression of CD70 (TNFSF7) in CD4+ T cells of patients with primary Sjogren’s syndrome. J. Dermatol. Sci. 2010, 59, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Liang, G.; Yin, H.; Ngalamika, O.; Li, F.; Zhao, M.; Lu, Q. DNA hypermethylation leads to lower FOXP3 expression in CD4+ T cells of patients with primary Sjogren’s syndrome. Clin. Immunol. 2013, 148, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Mougeot, J.L.; Noll, B.D.; Bahrani Mougeot, F.K. Sjӧgren’s syndrome X-chromosome dose effect: An epigenetic perspective. Oral Dis. 2019, 25, 372–384. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Harris, V.M.; Cavett, J.; Kurien, B.T.; Liu, K.; Koelsch, K.A.; Fayaaz, A.; Chaudhari, K.S.; Radfar, L.; Lewis, D.; et al. Rare X chromosome abnormalities in Systemic Lupus erythematosus and Sjӧgren’s syndrome. Arthritis Rheumatol. 2017, 69, 2187–2192. [Google Scholar] [CrossRef]
- Harris, V.M.; Sharma, R.; Cavett, J.; Kurien, B.T.; Liu, K.; Koelsch, K.A.; Rasmussen, A.; Radfar, L.; Lewis, D.; Stone, D.U.; et al. Klinefelter’s syndrome (47, XXY) is in excess among men with Sjӧgren’s syndrome. Clin. Immunol. 2016, 168, 25–29. [Google Scholar] [CrossRef]
- Dieker, J.; Muller, S. Epigenetic histone code and autoimmunity. Clin. Rev. Allergy Immunol. 2010, 39, 78–84. [Google Scholar] [CrossRef]
- Kouzarides, T. Chromatin modifications and their function. Cell 2007, 128, 693–705. [Google Scholar] [CrossRef]
- Brooks, W.H.; Le Dantec, C.; Pers, J.O.; Youinou, P.; Renaudineau, Y. Epigenetics and autoimmunity. J. Autoimmun. 2010, 34, J207–J219. [Google Scholar] [CrossRef] [PubMed]
- Farh, K.K.; Marson, A.; Zhu, J.; Kleinewietfled, M.; Housley, W.J.; Beik, S.; Shoresh, N.; Whitton, H.; Ryan, R.J.H.; Shishkin, A.A.; et al. Genetic and epigenetic fine-mapping of causal autoimmune disease variants. Nature 2015, 518, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Lo, J.C.; Foster, A.; Yu, P.; Chen, H.M.; Wang, Y.; Tamada, K.; Chen, L.; Fu, Y.X. The regulation of T cell homeostasis and autoimmunity by T cell-derived LIGHT. J. Clin. Investig. 2001, 108, 1771–1780. [Google Scholar] [CrossRef] [PubMed]
- Zare-Shahabadi, A.; Renaudineau, Y.; Rezaei, N. MicroRNAs and multiple sclerosis: From physiopathology to ward therapy. Expert Opin. Ther. Targets 2013, 17, 1497–1507. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, P.M.; Perugorria, M.J.; Santos-Laso, A.; Bujanda, L.; Beuers, U.; Banales, J.M. Primary biliary cholangitis: A tale of epigenetically-induced secretory failure? J. Hepatol. 2018, 69, 1371–1383. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le Dantec, C.; Varin, M.M.; Brooks, W.H.; Pers, J.O.; Youinou, P.; Renaudineau, Y. Epigenetics and Sjögren’s syndrome. Curr. Pharm. Biotechnol. 2012, 13, 2046–2053. [Google Scholar] [CrossRef] [PubMed]
- Alevizos, I.; Alexander, S.; Turner, R.J.; Illei, G.G. MicroRNA expression profiles as biomarkers of minor salivary gland inflammation and dysfunction in Sjögren’s syndrome. Arthritis Rheum. 2011, 63, 535–544. [Google Scholar] [CrossRef]
- Kapsogeorgou, E.K.; Gourzi, V.C.; Manoussakis, M.N.; Moutsopoulos, H.M.; Tzioufas, A.G. Cellular microRNAs (miRNAs) and Sjögren’s syndrome: Candidate regulators of autoimmune response and autoantigen expression. J. Autoimmun. 2011, 37, 129–135. [Google Scholar] [CrossRef]
- Tandon, M.; Gallo, A.; Jang, S.I.; Illei, G.G.; Alevizos, I. Deep sequencing of short RNAs reveals novel microRNAs in minor salivary glands of patients with Sjögren’s syndrome. Oral Dis. 2012, 18, 127–131. [Google Scholar] [CrossRef]
- Zilahi, E.; Tarr, T.; Papp, G.; Griger, Z.; Sipka, S.; Zeher, M. Increased microRNA-146a/b, TRAF6 gene and decreased IRAK1 gene expressions in the peripheral mononuclear cells of patients with Sjögren’s syndrome. Immunol. Lett. 2012, 141, 165–168. [Google Scholar] [CrossRef]
- Chen, J.Q.; Papp, G.; Póliska, S.; Szabó, K.; Tarr, T.; Bálint, B.L.; Szodoray, P.; Zeher, M. MicroRNA expression profiles identify disease-specific alterations in systemic lupus erythematosus and primary Sjögren’s syndrome. PLoS ONE 2017, 12, e0174585. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, G.; Zhang, L.; Miansong, Z.; HongDong, H. Decreased microRNA-181a and -16 expression levels in the labial salivary glands of Sjögren syndrome patients. Exp. Ther. Med. 2017, 10, 426–432. [Google Scholar]
- Wang-Renault, S.W.; Boudaoud, S.; Nocturne, G.; Roche, E.; Sigrist, N.; Daviaud, C.; Bugge Tinggaard, A.; Renault, V.; Deleuze, J.F.; Mariette, X.; et al. Deregulation of microRNA expression in purified T and B lymphocytes from patients with primary Sjögren’s syndrome. Ann. Rheum. Dis. 2018, 77, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Michael, A.; Bajracharya, S.D.; Yuen, P.S.; Zhou, H.; Star, R.A.; Illei, G.G.; Alevizos, I. Exosomes from human saliva as a source of microRNA biomarkers. Oral Dis. 2010, 16, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Zheng, L.Y.; Zhang, P.; Yu, C.Q. miR-146a and miR-155 expression in PBMCs from patients with Sjogren’s syndrome. J. Oral Pathol. Med. 2014, 43, 792–797. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Ma, W.; Yi, F.; Yang, Y.J.; Lin, W.; Chen, H.; Zhang, X.; Zhang, L.H.; Zhang, F.; Du, Q. MicroRNA profiling in Chinese patients with primary Sjogren syndrome reveals elevated miRNA-181a in peripheral blood mononuclear cells. J. Rheumatol. 2014, 41, 2208–2213. [Google Scholar] [CrossRef] [PubMed]
- Gourzi, V.C.; Kapsogeorgou, E.K.; Kyriakidis, N.C.; Tzioufas, A.G. Study of microRNAs (miRNAs) that are predicted to target the autoantigens Ro/SSA and La/SSB in primary Sjogren’s syndrome. Clin. Exp. Immunol. 2015, 182, 14–22. [Google Scholar] [CrossRef]
- Gallo, A.; Jang, S.I.; Ong, H.L.; Perez, P.; Tandon, M.; Ambudkar, I.; Illei, G.; Alevizos, I. Targeting the Ca2+ sensor STIM1 by exosomal transfer of Ebv-miR-BART13-3p is associated with Sjogren’s syndrome. EbioMedicine 2016, 10, 216–226. [Google Scholar] [CrossRef]
- Yang, Y.; Peng, L.; Ma, W.; Yi, F.; Zhang, Z.; Chen, H.; Guo, Y.; Wang, L.; Zhao, L.D.; Zheng, W.; et al. Autoantigen-targeting microRNAs in Sjogren’s syndrome. Clin. Rheumatol. 2016, 35, 911–917. [Google Scholar] [CrossRef]
- Debant, M.; Burgos, M.; Hemon, P.; Buscaglia, P.; Fali, T.; Melayah, S.; Le Coux, N.; Vandier, C.; Potier-Cartereau, M.; Pers, J.O.; et al. STIM1 at the plasma membrane as a new target in progressive chronic lymphocytic leukemia. J. Immunother. Cancer 2019, 7, 111. [Google Scholar] [CrossRef]
- Mukherjee, S.; Karolak, A.; Debant, M.; Buscaglia, P.; Renaudineau, Y.; Mignen, O.; Guida, W.C.; Brooks, W.H.; et al. Molecular dynamics simulations of membrane-bound STIM1 to investigate conformational changes during STIM1 activation upon calcium release. J. Chem. Inf. Model. 2017, 57, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Pauley, K.M.; Cha, S.; Chan, E.K. microRNA in autoimmunity and autoimmune diseases. J. Autoimmun. 2009, 32, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Padgett, K.A.; Lan, R.Y.; Leung, P.C.; Lleo, A.; Dawson, K.; Pfeiff, J.; Mao, T.K.; Coppel, R.L.; Ansari, A.A.; Gershwin, M.E. Primary biliary cirrhosis is associated with altered hepatic microRNA expression. J. Autoimmun. 2009, 32, 246–253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qin, B.; Huang, F.; Liang, Y.; Yang, Z.; Zhong, R. Analysis of altered microRNA expression profiles in peripheral blood mononuclear cells from patients with primary biliary cirrhosis. J. Gastroenterol. Hepatol. 2013, 28, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Ninomiya, M.; Kondo, Y.; Funayama, R.; Nagashima, T.; Kogure, T.; Kakazu, E.; Kimura, O.; Ueno, Y.; Nakayama, K.; Shimosegawa, T. Distinct microRNAs expression profile in primary biliary cirrhosis and evaluation of miR 505-3p and miR197-3p as novel biomarkers. PLoS ONE 2013, 8, 66086. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, T.; Morishita, A.; Nomura, T.; Tani, J.; Miyoshi, H.; Yoneyama, H.; Iwama, H.; Himoto, T.; Masaki, T. Identification of microRNA profiles associated with refractory primary biliary cirrhosis. Mol. Med. Rep. 2016, 14, 3350–3356. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.Y.; Hou, Y.Q.; Luo, L.J.; Li, A.O. Altered expression of miR-92a correlates with Th17 cell frequency in patients with primary biliary cirrhosis. Int. J. Mol. Med. 2016, 38, 131–138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, Y.; Pan, T.; Ye, Y.; Ge, G.; Chen, L.; Wen, D.; Zou, S. Serum microRNAs as potential biomarkers of primary biliary cirrhosis. PLoS ONE 2014, 9, 111424. [Google Scholar] [CrossRef]
- Banales, J.M.; Saez, E.; Uriz, M.; Sarvide, S.; Urribarri, A.D.; Splinter, P.; Tietz Bogert, P.S.; Bujanda, L.; Prieto, J.; Medina, J.F.; et al. Up-regulation of microRNA 506 leads to decreased Cl-/HCO3- anion exchanger 2 expression in biliary epithelium of patients with primary biliary cirrhosis. Hepatology 2012, 56, 687–697. [Google Scholar] [CrossRef]
- Esparza-Baquer, A.; Labiano, I.; Bujanda, L.; Perugorria, M.J.; Banales, J.M. MicroRNAs incholangiopathies: Potential diagnostic and therapeutic tools. Clin. Res. Hepatol. Gastroenterol. 2016, 40, 15–27. [Google Scholar] [CrossRef]
- Ananthanarayanan, M.; Banales, J.M.; Guerra, M.T.; Spirli, C.; Munoz-Garrido, P.; Mitchell-Richards, K.; Tafur, D.; Saez, E.; Nathanson, M.H. Post-translational regulation of the type III inositol 1,4,5-trisphosphate receptor by miRNA-506. J. Biol. Chem. 2015, 290, 184–196. [Google Scholar] [CrossRef] [PubMed]
- Olaizola, P.; Lee-Law, P.Y.; Arbelaiz, A.; Lapitz, A.; Perugorria, M.J.; Bujanda, L.; Banales, J.M. MicroRNAs and extracellular vesicles in cholangiopathies. Biochim. Biophys. Acta 2018, 1864, 1293–1307. [Google Scholar] [CrossRef] [PubMed]
- Erice, O.; Munoz-Garrido, P.; Vaquero, J.; Perugorria, M.J.; Fernandez-Barrena, M.G.; Saez, E.; Santos-Laso, A.; Arbelaiz, A.; Jimenez-Agüero, R.; Fernandez-Irigoyen, J.; et al. MicroRNA-506 promotes primary biliary cholangitis-like features in cholangiocytes and immune activation. Hepatology 2018, 67, 1420–1440. [Google Scholar] [CrossRef] [PubMed]
- Van Niekerk, J.; Kersten, R.; Beuers, U. Role of bile Acids and the biliary HCO3- umbrella in the pathogenesis of primary biliary cholangitis. Clin. Liver Dis. 2018, 22, 457–479. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Anso, E.; Castillo, J.E.; Diez, J.; Medina, J.F.; Prieto, J. Immunohistochemical detection of chloride/bicarbonate anion exchangers in human liver. Hepatology 1994, 19, 1400–1406. [Google Scholar] [CrossRef] [PubMed]
- Arenas, F.; Hervias, I.; Uriz, M.; Joplin, R.; Prieto, J.; Medina, J.F. Combination of ursodeoxycholic acid and glucocorticoids upregulates the AE2 alternate promoter in human liver cells. J. Clin. Investig. 2008, 118, 695–709. [Google Scholar] [CrossRef] [Green Version]
- Medina, J.F. Role of the anion exchanger 2 in the pathogenesis and treatment of primary biliary cirrhosis. Dig. Dis. 2011, 29, 103–112. [Google Scholar] [CrossRef]
- Uriarte, I.; Banales, J.M.; Saez, E.; Arenas, F.; Oude Elferink, R.P.; Prieto, J.; Medina, J.F. Bicarbonate secretion of mouse cholangiocytes involves Na(+)-HCO(3)(-) cotransport in addition to Na(+)-independent Cl(-)/HCO(3)(-) exchange. Hepatology 2010, 51, 891–902. [Google Scholar] [CrossRef]
- Beuers, U.; Hohenester, S.; de Buy Wenniger, L.J.; Kremer, A.E.; Jansen, P.L.; Elferink, R.P. The biliary HCO(3)(-) umbrella: A unifying hypothesis on pathogenetic and therapeutic aspects of fibrosingcholangiopathies. Hepatology 2010, 52, 1489–1496. [Google Scholar] [CrossRef]
- Hohenester, S.; Wenniger, L.M.; Paulusma, C.C.; van Vliet, S.J.; Jefferson, D.M.; Elferink, R.P.; Beuers, U. A biliary HCO3- umbrella constitutes a protective mechanism against bile acid induced injury in human cholangiocytes. Hepatology 2012, 55, 173–183. [Google Scholar] [CrossRef]
- Medina, J.F.; Martinez, A.; Vazquez, J.J.; Prieto, J. Decreased anion exchanger 2 immunoreactivity in the liver of patients with primary biliary cirrhosis. Hepatology 1997, 25, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Melero, S.; Spirli, C.; Zsembery, A.; Medina, J.F.; Joplin, R.E.; Duner, E.; Zuin, M.; Neuberger, J.M.; Prieto, J.; Strazzabosco, M. Defective regulation of cholangiocyte Cl-/HCO3(-) and Na+/H+ exchanger activities in primary biliary cirrhosis. Hepatology 2002, 35, 1513–1521. [Google Scholar] [CrossRef] [PubMed]
- Salas, J.T.; Banales, J.M.; Sarvide, S.; Recalde, S.; Ferrer, A.; Uriarte, I.; Oude Elferink, R.P.; Prieto, J.; Medina, J.F. Ae2a,b deficient mice develop antimitochondrial antibodies and other features resembling primary biliary cirrhosis. Gastroenterology 2008, 134, 1482–1493. [Google Scholar] [CrossRef] [PubMed]
- Van der Ree, M.H.; de Vree, J.M.; Stelma, F.; Willemse, S.; van der Valk, M.; Rietdijk, S.; Molenkamp, R.; Schinkel, J.; van Nuenen, A.C.; Beuers, U. Safety, tolerability, and antiviral effect of RG-101 in patients with chronic hepatitis C: A phase 1B, double-blind, randomised controlled trial. Lancet 2017, 389, 709–717. [Google Scholar] [CrossRef]
- Jones, P.A.; Issa, J.P.; Baylin, S. Targeting the cancer epigenome for therapy. Nat. Rev. Genet. 2016, 17, 630–641. [Google Scholar] [CrossRef]
- Pera, B.; Tang, T.; Marullo, R.; Yang, S.N.; Ahn, H.; Patel, J.; Elstrom, R.; Ruan, J.; Furman, R.; Leonard, J.; et al. Combinatorial epigenetic therapy in diffuse large B cell lymphoma pre-clinical models and patients. Clin. Epigenet. 2016, 8, 79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fardi, M.; Solali, S.; Hagh, M.F. Epigenetic mechanisms as a new approach in cancer treatment: An updated review. Genes Dis. 2018, 5, 304–311. [Google Scholar] [CrossRef]
- Mahmood, N.; Rabbani, S.A. DNA Methylation Readers and Cancer: Mechanistic and Therapeutic Applications. Front. Oncol. 2019, 9, 489. [Google Scholar] [CrossRef]
- Hull, E.E.; Montgomery, M.R.; Leyva, K.J. HDAC Inhibitors as Epigenetic Regulators of the Immune System: Impacts on Cancer Therapy and Inflammatory Diseases. BioMed Res. Int. 2016, 2016. [Google Scholar] [CrossRef]
- Sippl, W.; Jung, M.; Mannhold, R.; Buschmann, H.; Holenz, J. Epigenetic Drug Discovery; Wiley: Hoboken, NJ, USA, 2019. [Google Scholar]
- Mazzone, R.; Zwergel, C.; Artico, M.; Taurone, S.; Ralli, M.; Greco, A.; Mai, A. The emerging role of epigenetics in human autoimmune disorders. Clin. Epigenetics 2019, 11, 34. [Google Scholar] [CrossRef]
- Imgenberg-Kreuz, J.; Sandling, J.K.; Nordmark, G. Epigenetic alterations in primary Sjögren’s syndrome—An overview. Clin. Immunol. 2018, 196, 12–20. [Google Scholar] [CrossRef] [PubMed]
| Study | Method | Sample | Findings |
|---|---|---|---|
| Huet al, 2011 [68] | Chromatin Immunoprecipitation (ChIP) | 60 PBC patients (55 women, 5 men) | Increased H4 histone acetylation of promoters for CD40L, LIGHT, IL-17, IFNγ and deacetylation of H4 histone of TRAIL, Apo2, HDAC7A promoter in T cells. |
| Lleo et al., 2012 [66] | Bisulfite sequencing | 20 PBC women, 20 healthy women, 9 women with psoriasis, 9 women with DM1 | Hypomethylationof CD40L gene promoter |
| Selmi et al., 2014 [69] | DNA immunoprecipitation (MeDIP) | 3 pairs of monozygotic female twins, 8 pairs of sisters discordant for PBC | Differentmethylation • twins—60 gene regions • sisters—14 gene regions Downregulation of: CXCR5, HLA-B, IFI44L, IFIT1, and SMARCA1, Upregulation of IL-6 |
| Lleo et al., 2015 [51] | Bisulfite sequencing | 30 women with PBC30 healthy women | 20, 15, and 9 distinct gene promoters with a significant difference in methylation profile in CD4(+), CD8(+), and CD14(+) T cells respectively |
| Authors | miRNAs | Tissue | Design | Findings |
|---|---|---|---|---|
| Michael et al., 2009 [95] | hsa-miR-203 hsa-miR-768-3p hsa-miR-574-3p | saliva | SjS vs. controls | exosomal microRNAs as disease biomarkers |
| Pauley et al., 2011 [103] | miR-146a | PBMC | SjS vs. controls | Overexpression of miR-146 and miR-155 |
| Alevizos et al., 2011 [88] | miR-768-3p miR-574-3p | MSG | SjS vs. controls | miR-768-3p and miR-574 inversely correlated with focus score |
| Kapsogeorgou et al., 2011 [89] | miR-16 miR-200b, Let-7b miR-223 miR-181a | PBMCs, SG, SGEC | SjS vs. sicca controls and seronegative SjS patients | downregulation of let-7b in SGECs of antibody-positive patients |
| Tandon et al., 2012 [90] | miR-4524 miR-5100 | MSG | SjS vs. controls | differential expression of miR-5100 |
| Zilahi et al., 2012 [91] | miR-146a miR-146b | PBMCs | SjS vs. controls | Overexpression of miR-146a/b |
| Shi et al., 2014 [96] | miR-146a miR-155 | PBMCs | SjS vs. controls | Overexpression of miR-146a, downregulation of miR-155 |
| Peng et al., 2014 [97] | miR-181a | PBMCs | SjS vs. controls | Overexpression of miR-181a |
| Gourzi et al., 2015 [98] | miR16, miR200b-3p, miR223, miR483-5p | MSG, SGEC, PBMC | SjS vs. sicca controls | dysregulation of miR-16, miR-200b-3p, miR-223and miR-483-5p |
| Gallo et al., 2016 [99] | miR-BART13-3p | MSG | SjS vs. controls | elevation of miR-BART13-3p affects calcium pathway |
| Yang et al., 2016 [100] | miR-1207-5p, miR-4695-3p | MSG | SjS vs. controls | downregulation of miR-1207-5p and miR-4695-3p expression |
| Chen et al., 2017 [92] | miR-150-5p | PBMC | SjS vs. controls | reduced miR-150-5p expression |
| Wang et al., 2017 [93] | miR-181a miR-16 | MSG | SjS vs. controls | decreased miR-181a/miR-16 expression associated with focus score |
| Wang-Renault et al., 2018 [94] | miR-30b-5p | CD19(+) B, CD4(+) T cells | SjS vs. controls | miR-30b-5p inversely correlates with BAFF expression in B cells |
| Authors | miRNAs | Tissue | Design | Findings |
|---|---|---|---|---|
| Padgett et al., 2009 [104] | miR-122a, miR-26a, miR-328, miR-299-5p | Liver | PBC vs. controls | Downregulation of miR-122a and miR-26a and the increased expression of miR-328 and miR-299-5p |
| Banales et al., 2012 [110] | miR-506 | Liver | PBC vs. controls | miR-506 overexpression impairs biliary bicarbonate secretion |
| Qin et al., 2013 [105] | miR-15a-5p, miR-20a-5p, miR-140-3p, miR-106b-5p, miR-3654, miR-181a-5p | PBMCs | PBC vs. controls | altered expression of 17 miRNAs involved in cell differentiation and signal transduction |
| Ninomiya et al.,2013 [106] | hsa-miR-505-3p, miR-197-3p | Sera | PBC vs. controls | decreased expression of hsa-miR-505-3p and miR-197-3p |
| Tan et al., 2014 [109] | hsa-miR-122-5p, hsa-miR-141-3p, hsa-miR-26b-5p | Sera | PBC vs. controls | hsa-miR-122-5p, hsa-miR-141-3p, and hsa-miR-26b-5p as disease biomarkers |
| Sakamoto et al., 2016 [107] | miRNA-122, miRNA-378, miRNA-4311, miRNA-4714-3p | Sera | Treatment effective vs. resistant patients | miRNA profile can be a useful approach for the characterization of PBC development |
| Liang et al., 2016 [108] | miR-92a | Sera, PBMCs | PBC vs. controls | altered miR-92a expression is associated with Th17 cell differentiation |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arvaniti, P.; Zachou, K.; Lyberopoulou, A.; Gatselis, N.K.; Brooks, W.H.; Dalekos, G.N.; Renaudineau, Y. Epigenetic Modifications in Generalized Autoimmune Epithelitis: Sjögren’s Syndrome and Primary Biliary Cholangitis. Epigenomes 2019, 3, 15. https://doi.org/10.3390/epigenomes3030015
Arvaniti P, Zachou K, Lyberopoulou A, Gatselis NK, Brooks WH, Dalekos GN, Renaudineau Y. Epigenetic Modifications in Generalized Autoimmune Epithelitis: Sjögren’s Syndrome and Primary Biliary Cholangitis. Epigenomes. 2019; 3(3):15. https://doi.org/10.3390/epigenomes3030015
Chicago/Turabian StyleArvaniti, Pinelopi, Kalliopi Zachou, Aggeliki Lyberopoulou, Nikolaos K. Gatselis, Wesley H. Brooks, George N. Dalekos, and Yves Renaudineau. 2019. "Epigenetic Modifications in Generalized Autoimmune Epithelitis: Sjögren’s Syndrome and Primary Biliary Cholangitis" Epigenomes 3, no. 3: 15. https://doi.org/10.3390/epigenomes3030015





