Fatty Acid and Related Potassium Kv2 Channel Blockers: Toxicity and Physiological Actions on Mosquitoes
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Electrophysiological Assays
2.2.1. Patch-Clamp Recordings
2.2.2. Drosophila Melanogaster Central Nervous System Recordings
2.3. Insects
2.4. Whole Insect Toxicity Assays
2.4.1. Larval Paralysis Assays
2.4.2. Topical Application
2.5. Data Analysis
3. Results
3.1. Electrophysiological Assays
3.1.1. Patch Clamp Recordings
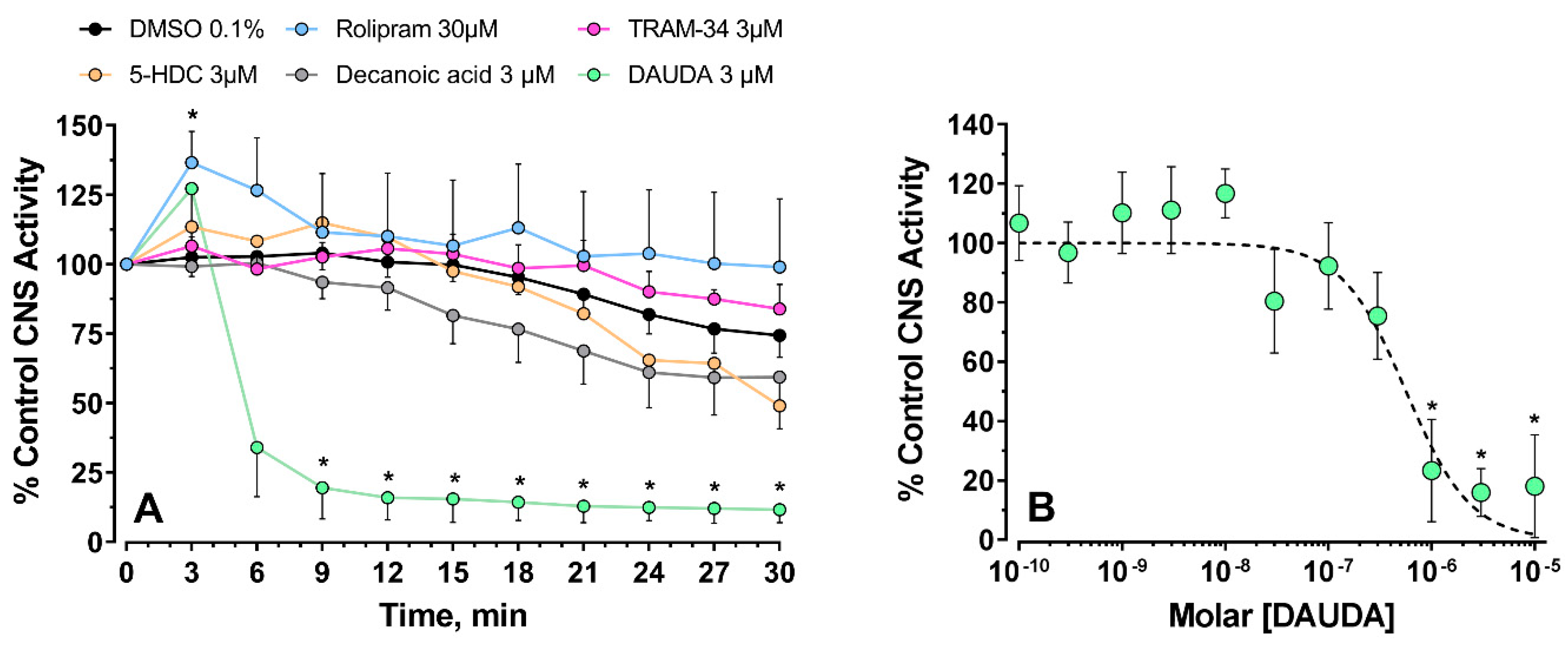
3.1.2. CNS Recordings
3.2. Larval Paralysis
3.3. Lethality by Topical Application
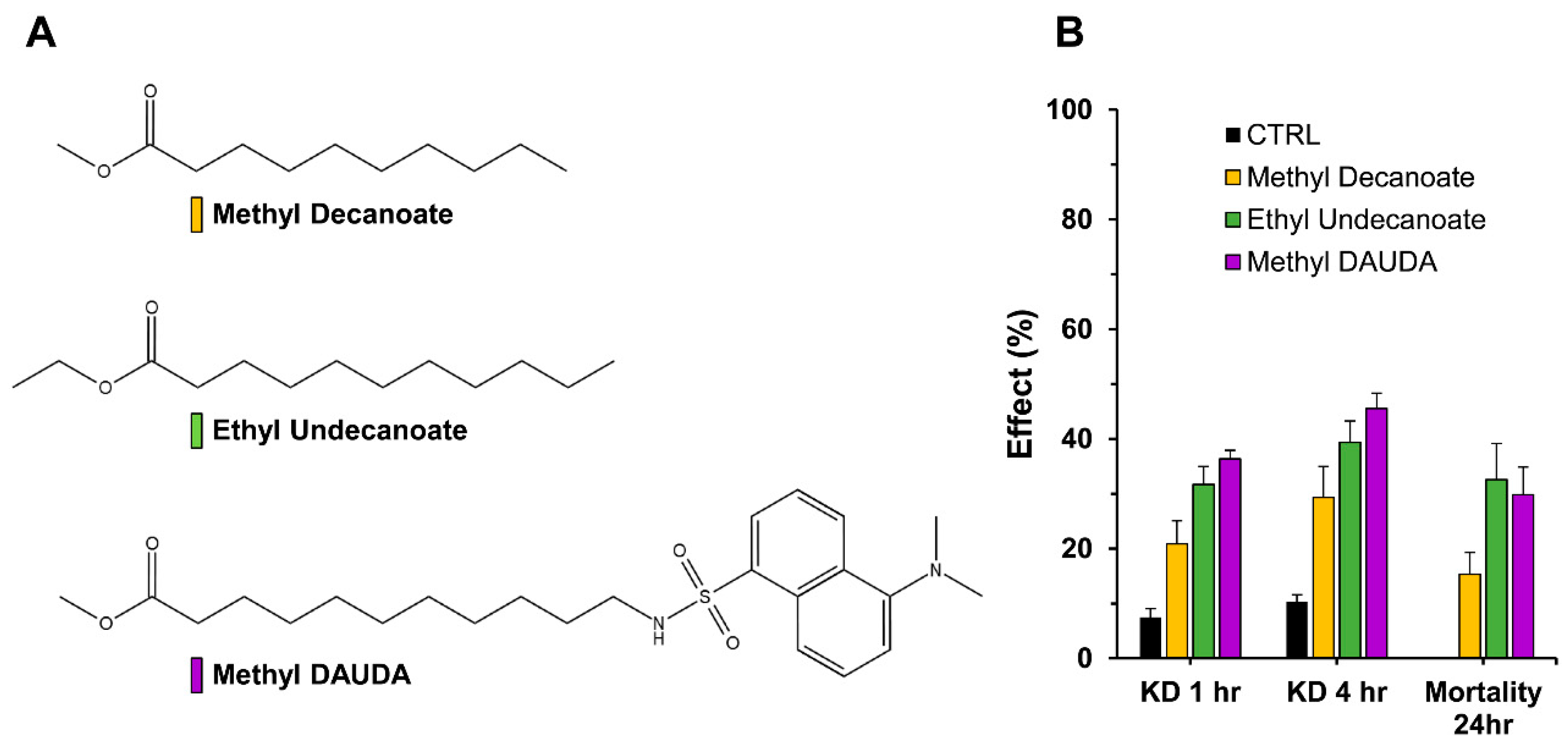
3.4. Evaluation of Permethrin Synergism
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hille, B. Ion Channels of Excitable Membranes; Sinauer: Sunderland, MA, USA, 2001; Volume 507. [Google Scholar]
- Catterall, W.A.; Cestèle, S.; Yarov-Yarovoy, V.; Yu, F.H.; Konoki, K.; Scheuer, T. Voltage-gated ion channels and gating modifier toxins. Toxicon 2007, 49, 124–141. [Google Scholar] [CrossRef] [PubMed]
- Jan, L.Y.; Jan, Y.N. Voltage-sensitive ion channels. Cell 1989, 56, 13–25. [Google Scholar] [CrossRef]
- Jan, L.Y.; Jan, Y.N. Voltage-gated potassium channels and the diversity of electrical signalling: Voltage-gated potassium channels. J. Physiol. 2012, 590, 2591–2599. [Google Scholar] [CrossRef] [PubMed]
- Salkoff, L.; Baker, K.; Butler, A.; Covarrubias, M.; Pak, M.D.; Wei, A. An essential ‘set’ of K+ channels conserved in flies, mice and humans. Trends Neurosci. 1992, 15, 161–166. [Google Scholar] [CrossRef]
- Dolly, J.O.; Parcej, D.N. Molecular properties of voltage-gated K+ channels. J. Bioenerg. Biomembr. 1996, 28, 231–253. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Singh, S. Unmasking of a novel potassium current in Drosophila by a mutation and drugs. J. Neurosci. 1999, 19, 6838–6843. [Google Scholar] [CrossRef] [PubMed]
- Swartz, K.J.; MacKinnon, R. An inhibitor of the Kv2. 1 potassium channel isolated from the venom of a Chilean tarantula. Neuron 1995, 15, 941–949. [Google Scholar] [CrossRef]
- Mouhat, S.; Andreotti, N.; Jouirou, B.; Sabatier, J.-M. Animal toxins acting on voltage-gated potassium channels. Curr. Pharm. Des. 2008, 14, 2503–2518. [Google Scholar] [CrossRef] [PubMed]
- Honoré, E.; Barhanin, J.; Attali, B.; Lesage, F.; Lazdunski, M. External blockade of the major cardiac delayed-rectifier K+ channel (Kv1. 5) by polyunsaturated fatty acids. Proc. Natl. Acad. Sci. USA 1994, 91, 1937–1941. [Google Scholar] [CrossRef] [PubMed]
- Boland, L.M.; Drzewiecki, M.M. Polyunsaturated fatty acid modulation of voltage-gated ion channels. Cell Biochem. Biophys. 2008, 52, 59–84. [Google Scholar] [CrossRef] [PubMed]
- Bloomquist, J.R.; Mutunga, J.M.; Islam, R.M.; Verma, A.; Ma, M.; Totrov, M.M.; Carlier, P.R. Voltage-sensitive potassium Kv2 channels as new targets for insecticides. In Biopesticides: State of the Art and Future Opportunities; ACS Publications; American Chemical Society: Washington, DC, USA, 2014; pp. 71–81. [Google Scholar]
- Larson, N.R.; Carlier, P.R.; Gross, A.D.; Islam, R.M.; Ma, M.; Sun, B.; Totrov, M.M.; Yadav, R.; Bloomquist, J.R. Toxicology of potassium channel-directed compounds in mosquitoes. Neurotoxicology 2017, 60, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Ranson, H.; N’Guessan, R.; Lines, J.; Moiroux, N.; Nkuni, Z.; Corbel, V. Pyrethroid resistance in African anopheline mosquitoes: What are the implications for malaria control? Trends Parasitol. 2011, 27, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Ranson, H.; Lissenden, N. Insecticide Resistance in African Anopheles Mosquitoes: A Worsening Situation that Needs Urgent Action to Maintain Malaria Control. Trends Parasitol. 2016, 32, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Notsu, T.; Tanaka, I.; Takano, M.; Noma, A. Blockade of the ATP-sensitive K+ channel by 5-hydroxydecanoate in guinea pig ventricular myocytes. J. Pharmacol. Exp. Ther. 1992, 260, 702–708. [Google Scholar] [PubMed]
- Smithers, N.; Bolivar, J.H.; Lee, A.G.; East, J.M. Characterizing the fatty acid binding site in the cavity of potassium channel KcsA. Biochemistry 2012, 51, 7996–8002. [Google Scholar] [CrossRef] [PubMed]
- Reeves, M.L.; Leigh, B.K.; England, P. The identification of a new cyclic nucleotide phosphodiesterase activity in human and guinea-pig cardiac ventricle. Implications for the mechanism of action of selective phosphodiesterase inhibitors. Biochem. J. 1987, 241, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Terrenoire, C.; Houslay, M.D.; Baillie, G.S.; Kass, R.S. The cardiac I Ks potassium channel macromolecular complex includes the phosphodiesterase PDE4D3. J. Biol. Chem. 2009, 284, 9140–9146. [Google Scholar] [CrossRef] [PubMed]
- Wulff, H.; Miller, M.J.; Hänsel, W.; Grissmer, S.; Cahalan, M.D.; Chandy, K.G. Design of a potent and selective inhibitor of the intermediate-conductance Ca2+-activated K+ channel, IKCa1: A potential immunosuppressant. Proc. Natl. Acad. Sci. USA 2000, 97, 8151–8156. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, J.J.; Zhu, Y.; Zhang, Q.-Y.; Mongin, A.A.; Hough, L.B. TRAM-34, a putatively selective blocker of intermediate-conductance, calcium-activated potassium channels, inhibits cytochrome P450 activity. PLoS ONE 2013, 8, e63028. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, K.; Sasaki, Y.; Kassai, T. The insecticidal activity of mixtures of pyrethroids and organophosphates or carbamates against the insecticide-resistant green rice leafhopper, Nephotettix cincticeps Uhler. J. Pestic. Sci. 1984, 9, 67–72. [Google Scholar] [CrossRef]
- Kurtak, D.; Meyer, R.; Ocran, M.; Ouédraogo, M.; Renaud, P.; Sawadogo, R.; Tele, B. Management of insecticide resistance in control of the Simulium damnosum complex by the Onchocerciasis Control Programme, West Africa: Potential use of negative correlation between organophosphate resistance and pyrethroid susceptibility. Med. Vet. Entomol. 1987, 1, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, J.; Corbel, V.; Darriet, F.; Chandre, F.; Hougard, J.-M. Topical applications of pyrethroid and organophosphate mixtures revealed positive interactions against pyrethroid-resistant Anopheles gambiae. J. Am. Mosq. Control Assoc. 2004, 20, 438–443. [Google Scholar] [PubMed]
- Corbel, V.; Chandre, F.; Darriet, F.; Lardeux, F.; Hougard, J. Synergism between permethrin and propoxur against Culex quinquefasciatus mosquito larvae. Med. Vet. Entomol. 2003, 17, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Taylor-Wells, J.; Gross, A.D.; Jiang, S.; Démares, F.; Clements, J.S.; Carlier, P.R.; Bloomquist, J.R. Toxicity, mode of action, and synergist potential of flonicamid against mosquitoes. Pestic. Biochem. Phys. 2018. [Google Scholar] [CrossRef]
- Villaverde, J.J.; Sandín-España, P.; Sevilla-Morán, B.; López-Goti, C.; Alonso-Prados, J.L. Biopesticides from natural products: Current development, legislative framework, and future trends. BioResources 2016, 11, 5618–5640. [Google Scholar] [CrossRef]
- Okano, K.; Mitsuhashi, N.; Tokuyama, H. Total synthesis of PDE-I and-II by copper-mediated double aryl amination. Tetrahedron 2013, 69, 10946–10954. [Google Scholar] [CrossRef]
- Bloomquist, J.R. Mode of action of atracotoxin at central and peripheral synapses of insects. Invertebr. Neurosci. 2003, 5, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Islam, R.M.; Bloomquist, J.R. A method for assessing chemically-induced paralysis in headless mosquito larvae. MethodsX 2015, 2, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Wheeler, M.W.; Park, R.M.; Bailer, A.J. Comparing median lethal concentration values using confidence interval overlap or ratio tests. Environ. Toxicol. Chem. 2006, 25, 1441–1444. [Google Scholar] [CrossRef] [PubMed]
- Luzhkov, V.B.; Åqvist, J. Mechanisms of tetraethylammonium ion block in the KcsA potassium channel. FEBS let. 2001, 495, 191–196. [Google Scholar] [CrossRef]
- Siegler, E.H.; Popenoe, C.H. The Fatty Acids as Contact Insecticides. J. Econ. Entomol. 1925, 18, 292–299. [Google Scholar] [CrossRef]
- Anneken, D.J.; Both, S.; Christoph, R.; Fieg, G.; Steinberner, U.; Westfechtel, A. Fatty Acids. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006. [Google Scholar]
- Chandler, D.; Bailey, A.S.; Tatchell, G.M.; Davidson, G.; Greaves, J.; Grant, W.P. The development, regulation and use of biopesticides for integrated pest management. Philos. Trans. R. Soc. B 2011, 366, 1987–1998. [Google Scholar] [CrossRef] [PubMed]
- Gross, A.; Norris, E.; Kimber, M.; Bartholomay, L.; Coats, J. Essential oils enhance the toxicity of permethrin against Aedes aegypti and Anopheles gambiae. Med. Vet. Entomol. 2017, 31, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Salgado, V. The neurotoxic insecticidal mechanism of the nonsteroidal ecdysone agonist RH-5849: K+ channel block in nerve and muscle. Pestic. Biochem. Physiol. 1992, 43, 1–13. [Google Scholar] [CrossRef]
- Corbel, V.; Stankiewicz, M.; Bonnet, J.; Grolleau, F.; Hougard, J.; Lapied, B. Synergism between insecticides permethrin and propoxur occurs through activation of presynaptic muscarinic negative feedback of acetylcholine release in the insect central nervous system. NeuroToxicology 2006, 27, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.G. Piperonyl Butoxide: The Insecticide Synergist; Academic Press: San Diego, CA, USA, 1998. [Google Scholar]
- Kweka, E.J.; Mazigo, H.D.; Mapunda, G.; Yewhalaw, D. Piperonyl Butoxide: An Enhancing Arsenal for an Adomant Foe. J. Transm. Dis. Immun. 2017, 1, 15. [Google Scholar]
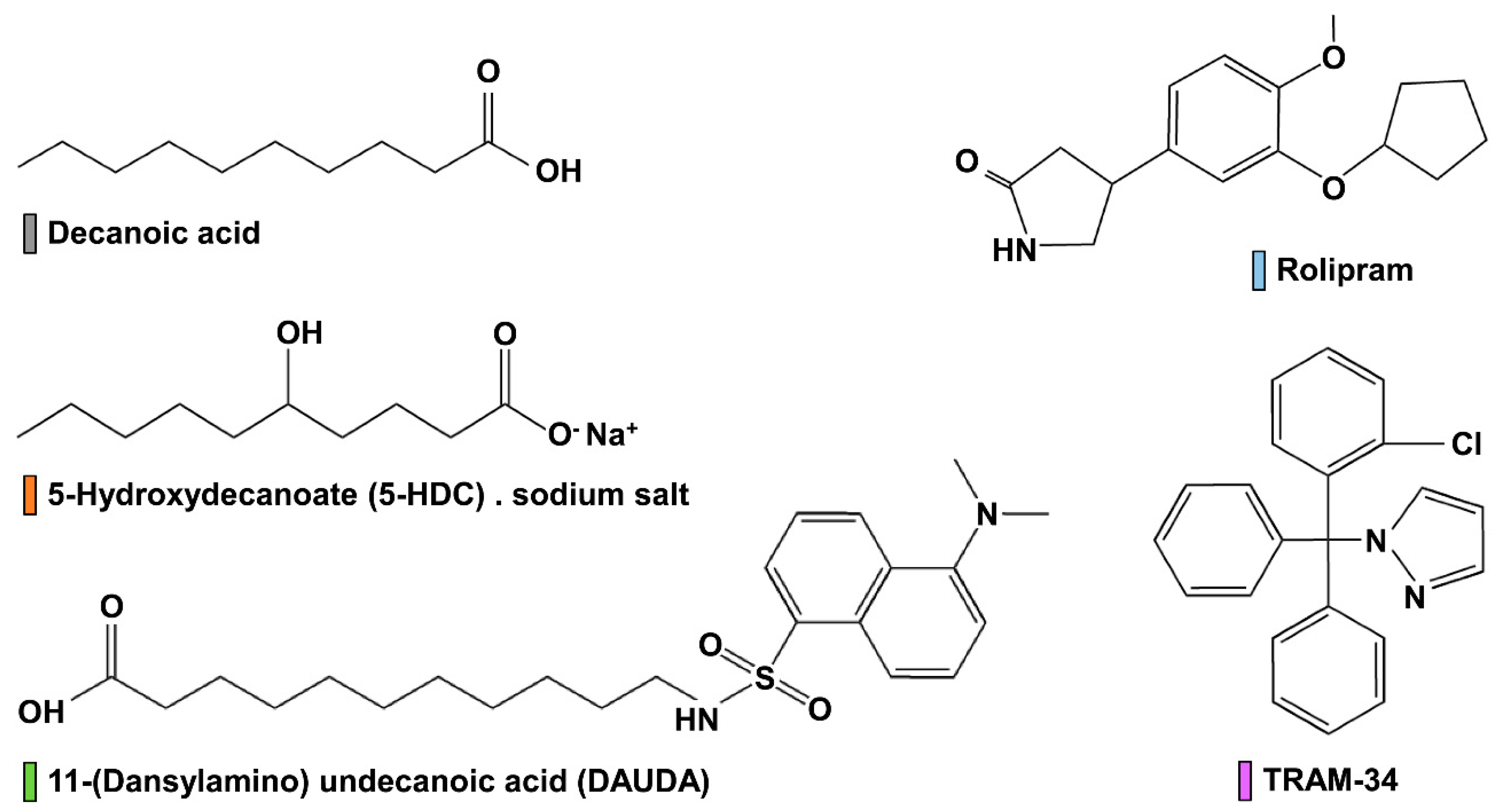

| Compounds | Patch Clamp | Headless Larvae | Intact Larvae | Topical Application |
|---|---|---|---|---|
| IC50 | PC50 | % Paralysis | LD50 | |
| (µM) | (ppm) | (250 ppm) | (µg/mg BW) | |
| Rolipram | N.A. | N.A. | 3.0 a | 20.9 a |
| [0–8.9] | [4.2–1.3 × 105] | |||
| TRAM-34 | N.A. | N.A. | 6.4 a | 15.6 a |
| [0.1–12.7] | [2.9–5.0 × 106] | |||
| 5-HDC | 30 a | 173 a | 13.8 ab | 2.6 ab |
| [19–57] | [90–480] | [5.6–22.0] | [1.6–6.3] | |
| Decanoic acid | 0.61 b | 32 b | 29.5 b | 0.8 b |
| [0.3–1.2] | [26–42] | [21.5–37.6] | [0.3–2.9] | |
| DAUDA | 1.18 b | 21 b | 23.2 b | 8.9 ab |
| [0.6–2.2] | [9–83] | [18.9–27.5] | [3.5–65] |
| Compounds | 1 h | 4 h | 24 h | |||
|---|---|---|---|---|---|---|
| KD50 | Slope | KD50 | Slope | LD50 | Slope | |
| Rolipram | 1.18 ab | 0.96 | 1.93 a | 0.7 | 20.9 a | 0.9 |
| [0.72–2.86] | (0.07) | [0.91–12.3] | (0.06) | [4.23–1.3 × 105] | (0.11) | |
| TRAM-34 | 0.74 a | 1.5 | 0.81 a | 0.94 | 15.6 a | 0.75 |
| [0.54–1.10] | (0.07) | [0.55–1.43] | (0.06) | [2.86–5.0 × 106] | (0.08) | |
| 5-HDC | 0.71 a | 1.44 | 0.91 a | 1.28 | 2.62 ab | 1.29 |
| [0.58–0.91] | (0.07) | [0.66–1.41] | (0.07) | [1.60–6.28] | (0.09) | |
| Decanoic acid | 0.44 a | 1.41 | 0.33 b | 1.56 | 0.80 b | 1.77 |
| [0.22–1.01] | (0.07) | [0.22–0.52] | (0.08) | [0.29–2.92] | (0.09) | |
| DAUDA | 1.91 b | 1.1 | 1.7 a | 0.71 | 8.90 a | 0.84 |
| [1.23–3.35] | (0.06) | [0.84–5.03] | (0.05) | [3.82–64.9] | (0.06) | |
| Compound | Synergist | 1 h | 4 h | 24 h | |||
|---|---|---|---|---|---|---|---|
| Dose (ng) | KD50 | Slope | KD50 | Slope | LD50 | Slope | |
| Permethrin | 0 | 0.08 a | 3 | 0.08 a | 2.22 | 0.10 ab | 2.36 |
| [0.07–0.09] | (0.1) | [0.06–0.10] | (0.04) | [0.08–0.12] | (0.08) | ||
| + Rolipram | 300 | 0.08 a | 2.67 | 0.11 a | 1.5 | 0.15 a | 2.1 |
| [0.07–0.10] | (0.14) | [0.08–0.16] | (0.12) | [0.11–0.21] | (0.13) | ||
| + TRAM-34 | 300 | 0.11 a | 2.03 | 0.09 ab | 1.49 | 0.10 ab | 2.14 |
| [0.08–0.13] | (0.13) | [0.03–0.15] | (0.11) | [0.05–0.18] | (0.13) | ||
| + 5-HDC | 300 | 0.03 b | 1.99 | 0.06 ab | 1.67 | 0.06 b | 2.16 |
| [0.02–0.04] | (0.15) | [0.02–0.09] | (0.12) | [0.03–0.09] | (0.14) | ||
| + Decanoic acid | 250 | 0.03 b | 1.92 | 0.04 ab | 1.06 | 0.14 ab | 2.17 |
| [0.02–0.04] | (0.09) | [0.02–0.07] | (0.07) | [0.07–0.48] | (0.11) | ||
| + DAUDA | 500 | 0.06 ab | 1.11 | 0.03 b | 1.04 | 0.15 a | 1.46 |
| [0.03–0.09] | (0.06) | [0.02–0.04] | (0.06) | [0.10–0.29] | (0.06) | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Démares, F.; Coquerel, Q.; Richoux, G.; Linthicum, K.; Bloomquist, J. Fatty Acid and Related Potassium Kv2 Channel Blockers: Toxicity and Physiological Actions on Mosquitoes. Insects 2018, 9, 155. https://doi.org/10.3390/insects9040155
Démares F, Coquerel Q, Richoux G, Linthicum K, Bloomquist J. Fatty Acid and Related Potassium Kv2 Channel Blockers: Toxicity and Physiological Actions on Mosquitoes. Insects. 2018; 9(4):155. https://doi.org/10.3390/insects9040155
Chicago/Turabian StyleDémares, Fabien, Quentin Coquerel, Gary Richoux, Kenneth Linthicum, and Jeffrey Bloomquist. 2018. "Fatty Acid and Related Potassium Kv2 Channel Blockers: Toxicity and Physiological Actions on Mosquitoes" Insects 9, no. 4: 155. https://doi.org/10.3390/insects9040155
APA StyleDémares, F., Coquerel, Q., Richoux, G., Linthicum, K., & Bloomquist, J. (2018). Fatty Acid and Related Potassium Kv2 Channel Blockers: Toxicity and Physiological Actions on Mosquitoes. Insects, 9(4), 155. https://doi.org/10.3390/insects9040155





