The Effect of Species Soybean Vein Necrosis Orthotospovirus (SVNV) on Life Table Parameters of Its Vector, Soybean Thrips (Neohydatothrips variabilis Thysanoptera: Thripidae)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of Insects and Infected Thrips Population Development
2.2. Development of Uninfected Thrips Population
2.3. Confirmation of Virus Infection through Real Time PCR in Plants
2.4. Confirmation of Virus Infection through Real Time PCR in Thrips
2.5. Life Table Studies
2.6. Statistical Analysis
3. Results
3.1. SVNV Infection Status in the Infected Strain and Confirmation of Uninfected Population
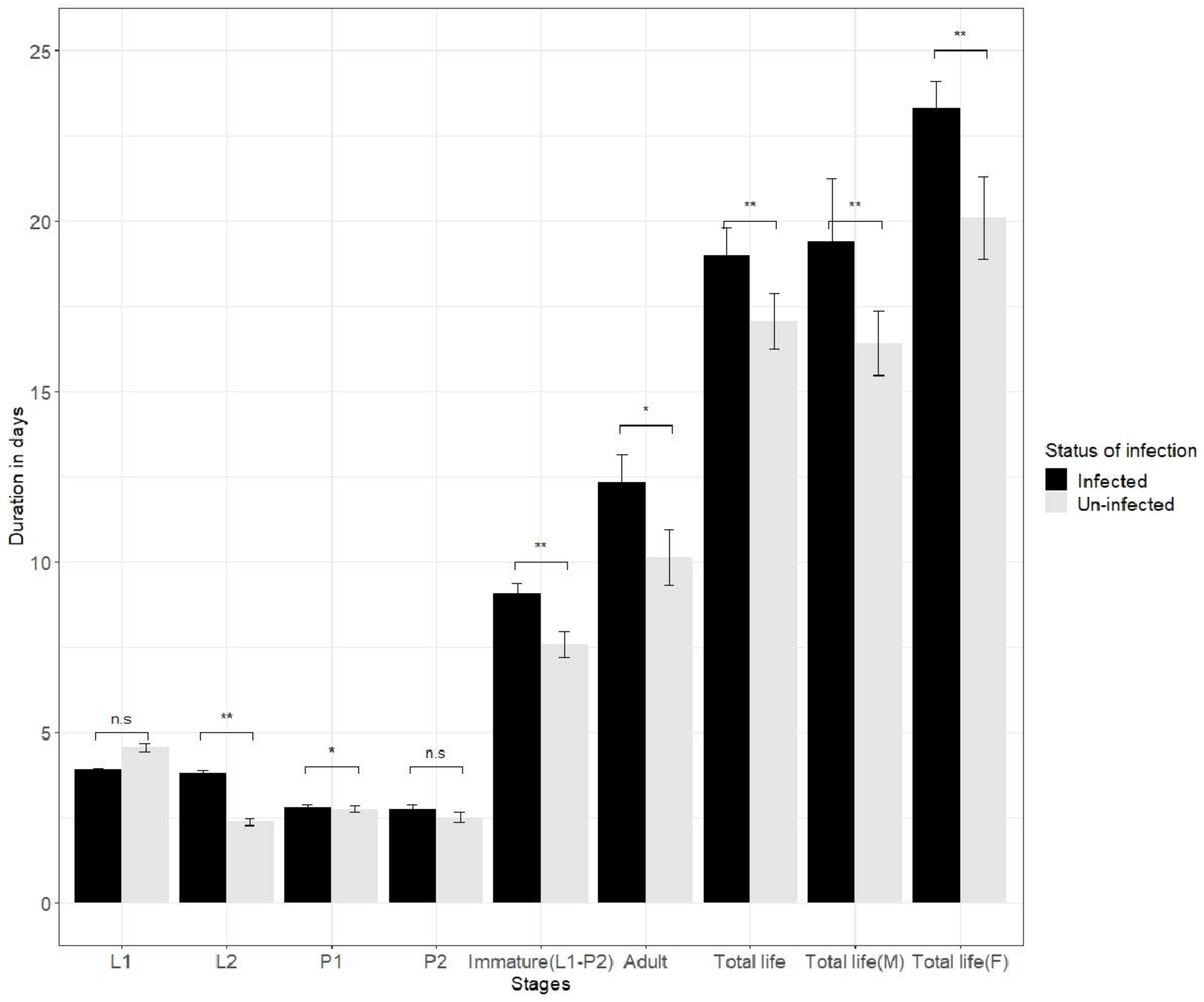
3.2. Impact of Virus on Thrips Biological Growth Parameters Such as Development Time, Adult Longevity and Fertility
3.3. Impact of SVNV on the Life Table Parameters
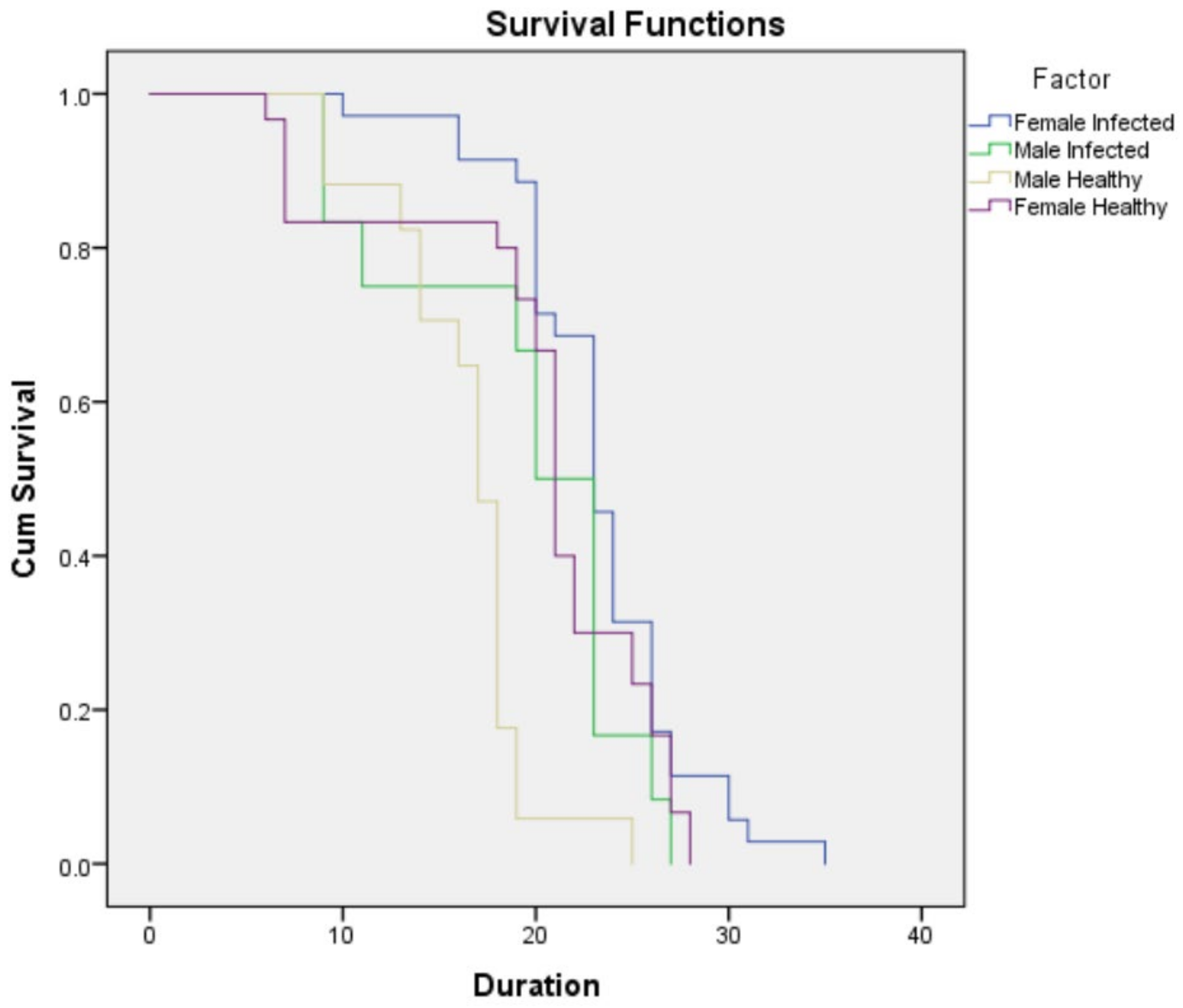
3.3.1. Survival and Mortality Parameters
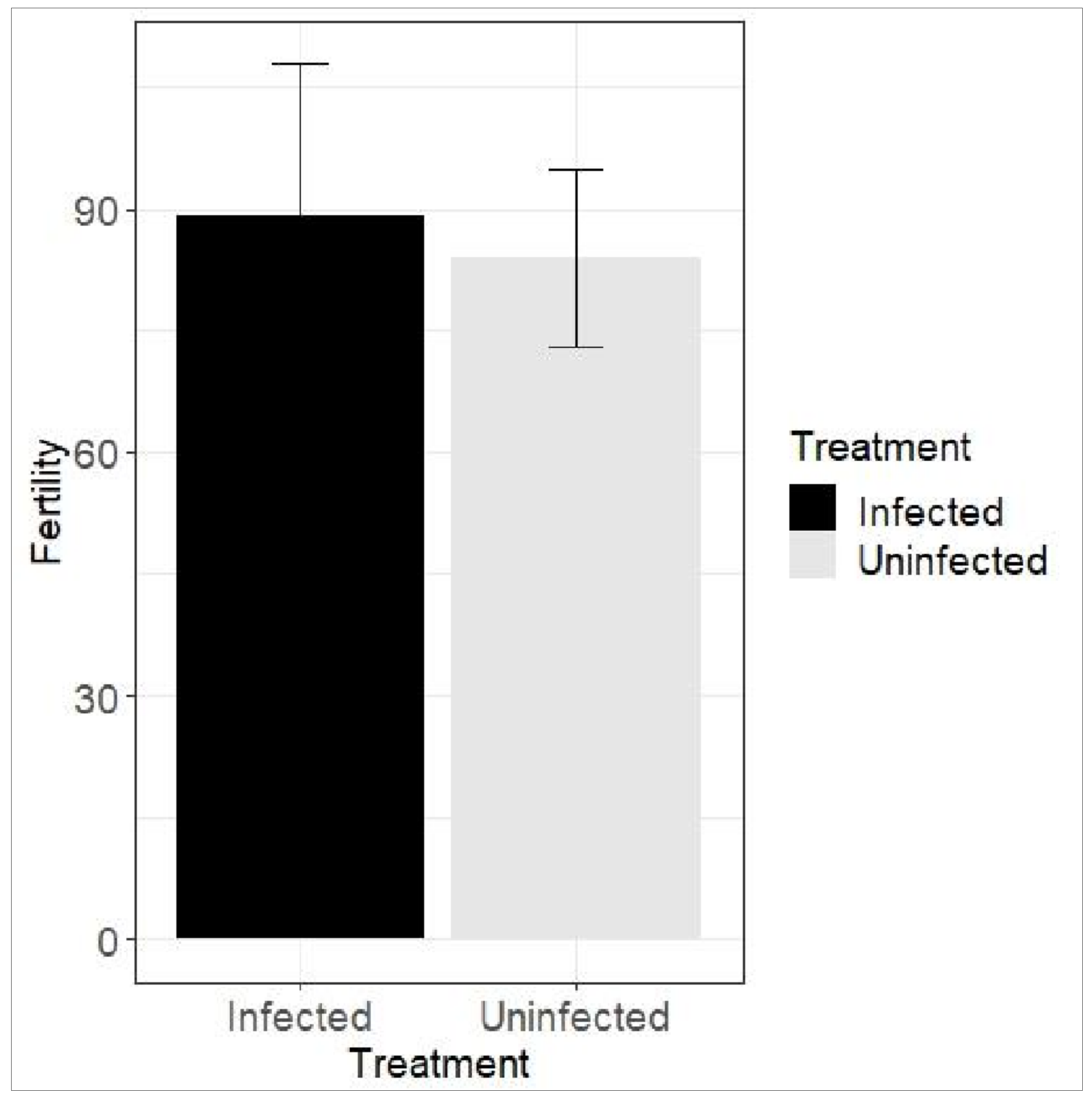
3.3.2. Effect of SVNV on Thrips Reproduction
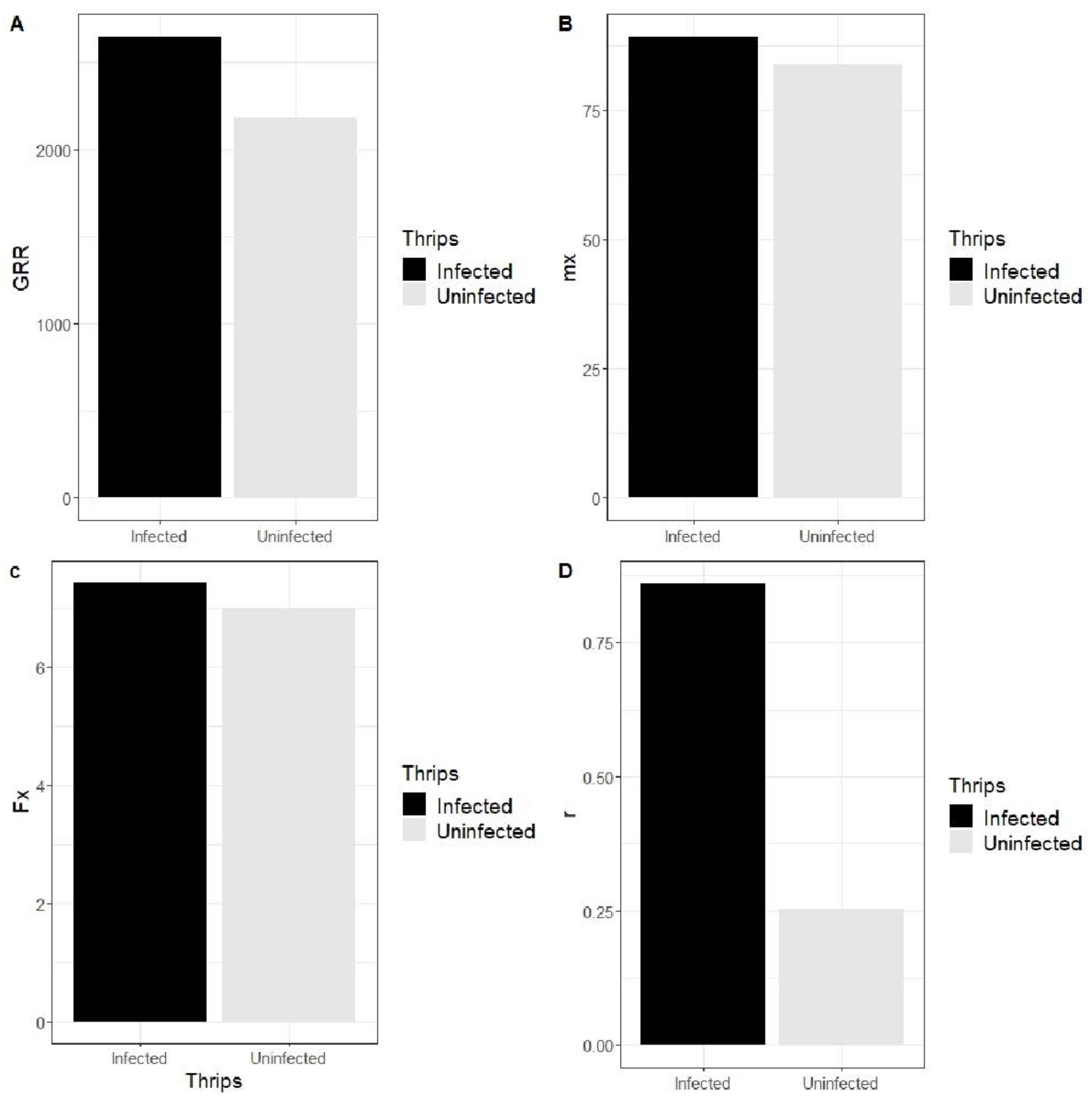
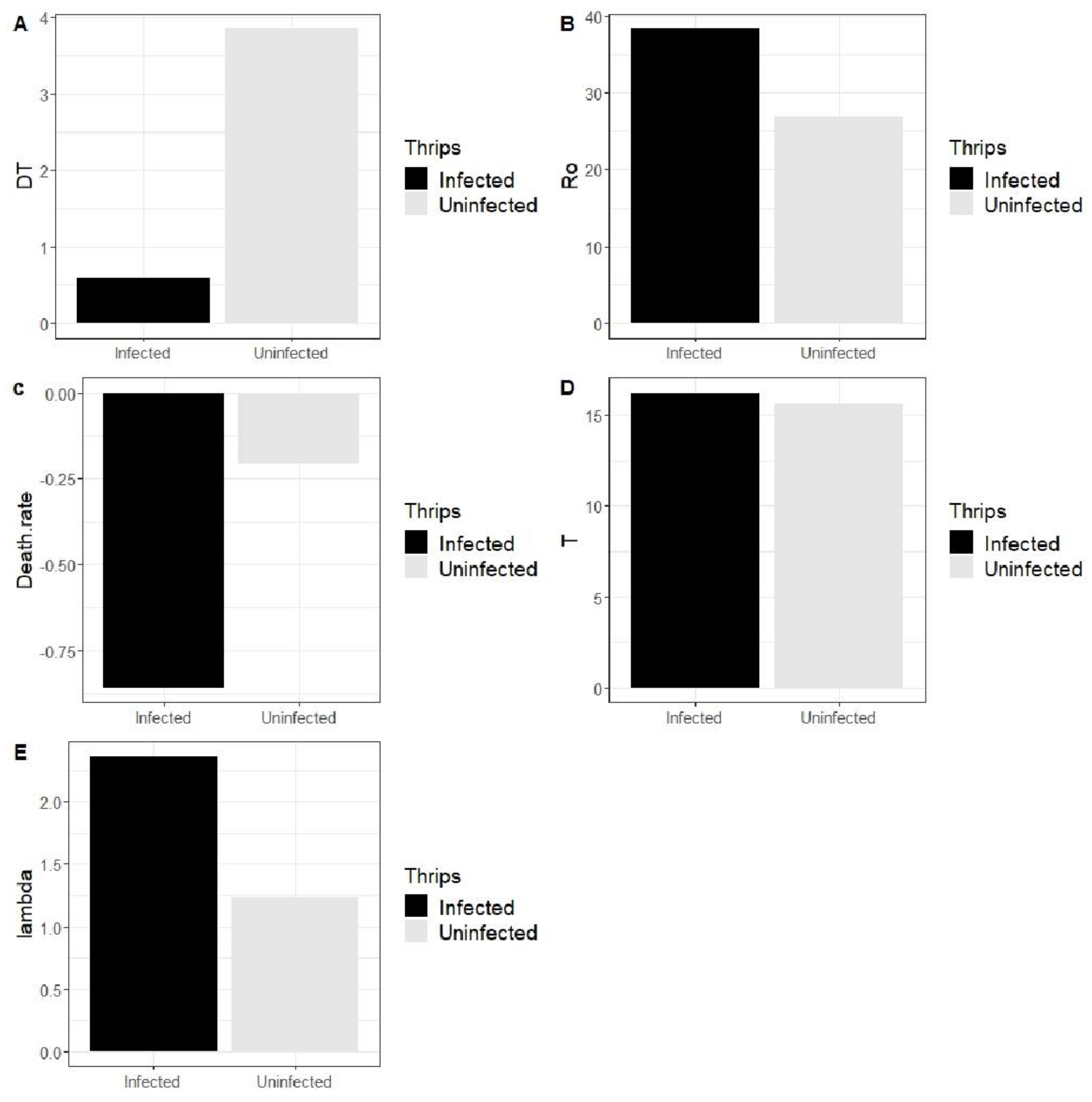
3.3.3. Effect of SVNV on Population Growth Parameters
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Groves, C.; German, T.; Dasgupta, R.; Mueller, D.; Smith, D.L. Seedtransmission of Soybeanveinnecrosisvirus: The first Tospovirus implicated in seed transmission. PLoS ONE 2016, 11, e0147342. [Google Scholar]
- Zhou, J.; Tzanetakis, I.E. Epidemiology of Soybeanveinnecrosis-associated virus. Phytopathology 2013, 103, 966–971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keough, S.A. Surveying Indiana Soybean for Soybean Vein Necrosis Virus (SVNV) and Analysis of SVNV on Life History Traits and Host Preference of Thrips Vectors. Ph.D. Thesis, Purdue University, West Lafayette, IN, USA, 2015. [Google Scholar]
- Bloomingdale, C.; Irizarry, M.D.; Groves, R.L.; Mueller, D.S.; Smith, D.L. Seasonal population dynamics of Thrips (Thysanoptera) in Wisconsin and Iowas oybean fields. J. Econ. Entomol. 2016, 110, 133–141. [Google Scholar]
- Irizarry, M. Soybean Vein Necrosis Virus: Impacts of Infection on Yield Loss and Seed Quality and Expansion of Plant Host Range. Ph.D. Thesis, Iowa State University, Ames, IA, USA, 2016. [Google Scholar]
- Zhou, J.; Kantartzi, S.; Wen, R.-H.; Newman, M.; Hajimorad, M.; Rupe, J.; Tzanetakis, I. Molecular characterization of a new tospovirus infecting soybean. Virus Genes 2011, 43, 289. [Google Scholar]
- Abd El-Wahab, A.S.; El-Shazly, M.A. Identification and characterization of soybean vein necrosis virus (SVNV): A newly isolated thrips-borne tospovirus in Egypt. J. Virol. Sci. 2017, 1, 76–90. [Google Scholar]
- Abd El-Wahab, A. Molecular characterization and incidence of new tospovirus: Soybean Vein Necrosis Virus (SVNV) in Egypt. Braz. J. Biol. 2021, 84, 1–17. [Google Scholar] [CrossRef]
- Hameed, A. Tripartite Interactions among Soybean Vein Necrosis Orthotospovirus, Vector Thrips and Soybean Plants. Ph.D. Thesis, Pennsylvania State University, State College, PA, USA, 2021. [Google Scholar]
- Anderson, N.R.; Irizarry, M.D.; Bloomingdale, C.A.; Smith, D.L.; Bradley, C.A.; Delaney, D.P.; Kleczewski, N.M.; Sikora, E.J.; Mueller, D.S.; Wise, K.A. Effect of soybean vein necrosis on yield and seed quality of soybean. Can. J. Plant Pathol. 2017, 39, 334–341. [Google Scholar] [CrossRef]
- Mumford, R.A.; Barker, I.; Wood, K.R. The biology of the tospoviruses. Ann. Appl. Biol. 1996, 128, 159–183. [Google Scholar] [CrossRef]
- Pappu, H.; Jones, R.; Jain, R. Global status of tospovirus epidemics in diverse cropping systems: Successes achieved and challenges ahead. Virus Res. 2009, 141, 219–236. [Google Scholar] [CrossRef]
- Persley, D.; Thomas, J.; Sharman, M. Tospoviruses—An Australian perspective. Australas. Plant Pathol. 2006, 35, 161–180. [Google Scholar] [CrossRef]
- Ullman, D.; Sherwood, J.; German, T. Thrips as vectors of plant pathogens. In Book Thrips as Crop Pests; Lewis, T., Ed.; CAB International: Wallingford, UK, 1997; Volume 1, pp. 539–565. [Google Scholar]
- Belliure, B.; Janssen, A.; Maris, P.C.; Peters, D.; Sabelis, M.W. Herbivore arthropods benefit from vectoring plant viruses. Ecol. Lett. 2005, 8, 70–79. [Google Scholar] [CrossRef]
- Keough, S.; Han, J.; Shuman, T.; Wise, K.; Nachappa, P. Effects of Soybean vein necrosis virus on lifehistory and host preference of its vector, Neohydatothrips variabilis, and evaluation of vector status of Frankliniella tritici and Frankliniella fusca. J. Econ. Entomol. 2016, 109, 1979–1987. [Google Scholar] [CrossRef]
- Han, J.; Nalam, V.J.; Yu, I.; Nachappa, P. Vector Competence of Thrips Species to Transmit Soybean Vein Necrosis Virus. Front. Microbiol. 2019, 10, 431. [Google Scholar] [CrossRef]
- Irwin, M.E.; Yeargan, K.V.; Marston, N.L. Spatial and seasonal patterns of phytophagous thrips in soybean fields with comments on sampling techniques. Environ. Entomol. 1979, 8, 131–140. [Google Scholar] [CrossRef]
- Shrestha, A.; Srinivasan, R.; Riley, D.G.; Culbreath, A.K. Direct and indirect effects of a thrips-transmitted Tospovirus on the preference and fitness of itsvector, Frankliniella fusca. Entomol. Exp. Appl. 2012, 145, 260–271. [Google Scholar] [CrossRef]
- Stumpf, C.F.; Kennedy, G.G. Effects of tomato spotted wilt virus (TSWV) isolates, host plants, and temperature on survival, size, and development time of Frankliniella fusca. Entomol. Exp. Appl. 2005, 114, 215–225. [Google Scholar] [CrossRef]
- Shalileh, S.; Ogada, P.A.; Moualeu, D.P.; Poehling, H.-M. Manipulation of Frankliniella occidentalis (Thysanoptera:Thripidae) by Tomato Spotted Wilt Virus (Tospovirus) Via the Host Plant Nutrients to Enhance Its Transmission and Spread. Environ. Entomol. 2016, 45, 1235–1242. [Google Scholar] [CrossRef] [Green Version]
- Belliure, B.; Janssen, A.; Sabelis, M.W. Herbivore benefits from vectoring plant virus through reduction of period of vulnerability to predation. Oecologia 2008, 156, 797–806. [Google Scholar] [CrossRef] [Green Version]
- Nachappa, P.; Challacombe, J.; Margolies, D.C.; Nechols, J.R.; Whitfield, A.E.; Rotenberg, D. Tomato spotted wilt virus benefits its thrips vector by modulating metabolic and plant defense pathways in tomato. Front. Plant Sci. 2020, 11, 575564. [Google Scholar] [CrossRef]
- Ogada, P.A.; Moualeu, D.P.; Poehling, H.-M. Predictive models for Tomato spotted wilt virus spread dynamics, considering Frankliniella occidentalis specific life processes as influenced by the virus. PLoS ONE 2016, 11, e0154533. [Google Scholar] [CrossRef] [Green Version]
- Huang, L.-H.; Lin, M.-C. Life Table Parameters of Thrips palmi Karny (Thysanoptera: Thripidae) as Related to Its Management Strategy. Formos. Entomol. 2012, 32, 237–248. [Google Scholar]
- Badillo-Vargas, I.; Rotenberg, D.; Schneweis, D.; Hiromasa, Y.; Tomich, J.; Whitfield, A. Proteomic analysis of Frankliniella occidentalis and differentially-expressed proteins in response to Tomato spotted wilt virus infection. J. Virol. 2012, 86, 8793–8809. [Google Scholar] [CrossRef] [Green Version]
- Deangelis, J.D.; Sether, D.M.; Rossignol, P.A. Survival, Development, and Reproduction in Western Flower Thrips (Thysanoptera: Thripidae) Exposed to Impatiens Necrotic Spot Virus. Environ. Entomol. 1993, 22, 1308–1312. [Google Scholar] [CrossRef]
- Yadav, R.; Chang, N.T. Effects of temperature on the development and population growth of the melon thrips, Thrips palmi, on egg plant, Solanum melongena. J. Insect Sci. 2014, 14, 78. [Google Scholar] [CrossRef] [Green Version]
- Birch, L.C. The intrinsic rate of natural increase of an insect population. J. Anim. Ecol. 1948, 17, 15. [Google Scholar] [CrossRef]
- Southwood, T.R.E.; Henderson, P.A. Ecological Methods; Wiley-Blackwell: Hoboken, NJ, USA, 2000; p. 575. [Google Scholar]
- Wagner, W.E., III. Using IBM® SPSS® Statistics for Research Methods and Social Science Statistics; Sage Publications: Thousand Oaks, CA, USA, 2016. [Google Scholar]
- Sether, D.M. Transmission Efficiency and Life Table Parameters of Western Flower Thrips, Frankliniella occidentalis (Pergande) Exposed to Tomato Spotted Wilt Virus-Impatiens Serotype. Master’s Thesis, Oregon State University, Corvallis, OR, USA, 1991. [Google Scholar]
- Maris, P.; Joosten, N.; Goldbach, R.; Peters, D. Tomato spotted wilt virus infection improves host suitability for its vector Frankliniella occidentalis. Phytopathology 2004, 94, 706–711. [Google Scholar] [CrossRef] [Green Version]
- Nachappa, P.; Margolies, D.C.; Nechols, J.R.; Whitfield, A.E.; Rotenberg, D. Tomato Spotted Wilt Virus Benefits a Non-Vector Arthropod, Tetranychus Urticae, by Modulating Different Plant Responses in Tomato. PLoS ONE 2013, 8, e75909. [Google Scholar] [CrossRef] [Green Version]
- Ogada, P.; Maiss, E.; Poehling, H.M. Influence of tomato spotted wilt virus on performance and behaviour of western flowerthrips (Frankliniella occidentalis). J. Appl. Entomol. 2013, 137, 488–498. [Google Scholar] [CrossRef]
- Fereres, A.; Kampmeier, G.; Irwin, M. Aphid attraction and preference for soybean and pepper plants infected with Potyviridae. Ann. Entomol. Soc. Am. 1999, 92, 542–548. [Google Scholar] [CrossRef]
- Castle, S.; Berger, P. Rates of growth and increase of Myzus persicae on virus-infected potatoes according to type of virusvector relationship. Entomol. Exp. Appl. 1993, 69, 51–60. [Google Scholar] [CrossRef]
- Jiménez-Martínez, E.; Bosque-Pérez, N.; Berger, P.; Zemetra, R. Life history of the bird cherry-oat aphid, Rhopalosiphum padi (Homoptera:Aphididae), on transgenic and untransformed wheat challenged with Barley yellow dwarf virus. J. Econ. Entomol. 2004, 97, 203–212. [Google Scholar] [CrossRef]
- Jiu, M.; Zhou, X.-P.; Tong, L.; Xu, J.; Yang, X.; Wan, F.-H.; Liu, S.-S. Vector-virus mutualism accelerates population increase of an invasive whitefly. PLoS ONE 2007, 2, e182. [Google Scholar] [CrossRef] [Green Version]
- Srinivasan, R.; Alvarez, J.M.; Bosque-Pérez, N.A.; Eigenbrode, S.D.; Novy, R.G. Effect of an alternate weed host, hairy nightshade, Solanum sarrachoides, on the biology of the two most important Potato leaf roll virus (Luteoviridae:Polerovirus) vectors, Myzus persicae and Macrosiphum euphorbiae (Aphididae:Homoptera). Environ. Entomol. 2008, 37, 592–600. [Google Scholar]
- Blua, M.; Perring, T. Alatae production and population increase of aphid vectors on virus-infected host plants. Oecologia 1992, 92, 65–70. [Google Scholar] [CrossRef]
- Garcia, L.; Kennedy, G.; Brandenburg, R. Survival and reproductive success of tobacco thrips on three Tomato spotted wilt virus infected and noninfected peanut cultivars. Peanut Sci. 2000, 27, 49–52. [Google Scholar] [CrossRef]
- Wijkamp, I.; Goldbach, R.; Peters, D. Propagation of tomato spotted wilt virus in Frankliniella occidentalis does neither result in pathological effects nor in trans ovarial passage of the virus. Entomol. Exp. Appl. 1996, 81, 285–292. [Google Scholar] [CrossRef]
- Roca, E.; Aramburu, J.; Moriones, E. Comparative host reactions and Frankliniella occidentalis transmission of different isolates of tomato spotted wilt tospovirus from Spain. Plant Pathol. 1997, 46, 407–415. [Google Scholar] [CrossRef]
- McElhany, P.; Real, L.A.; Power, A.G. Vector preference and disease dynamics: A study of barley yellow dwarf virus. Ecology 1995, 76, 444–457. [Google Scholar] [CrossRef]
- Jeger, M.; Holt, J.; VanDenBosch, F.; Madden, L. Epidemiology of insect-transmitted plant viruses: Modelling disease dynamics and control interventions. Physiol. Entomol. 2004, 29, 291–304. [Google Scholar] [CrossRef]
- Sisterson, M.S. Effects of insect-vector preference for healthy or infected plants on pathogen spread: Insights from a model. J. Econ. Entomol. 2008, 101, 1–8. [Google Scholar] [CrossRef]
- Chapman, T.W.; Crespi, B. High relatedness and inbreeding in two species of haplodiploid eusocial thrips (Insecta: Thysanoptera) revealed by microsatellite analysis. Behav. Ecol. Sociobiol. 1998, 43, 301–306. [Google Scholar] [CrossRef]
- Kageyama, D.; Narita, S.; Watanabe, M. Insect sex determination manipulated by their endosymbionts: Incidences, mechanisms and implications. Insects 2012, 3, 161–199. [Google Scholar] [CrossRef] [Green Version]
- Widana Gamage, S.M.; Rotenberg, D.; Schneweis, D.J.; Tsai, C.-W.; Dietzgen, R.G. Transcriptome-wide responses of adult melon thrips (Thrips palmi) associated with capsicum chlorosis virus infection. PLoS ONE 2018, 13, e0208538. [Google Scholar]
| Status of Infection | Stage | Proportion of Individual Surviving (Lx) | The Probability of Surviving the Period (Npx) | lx*px | Percent Probability of Death 100 qx | Cumulative Number of Days Lived beyond Age × Tx |
|---|---|---|---|---|---|---|
| L1 | 1 | 0.85 | 0.85 | 15 | 19.26 | |
| L2 | 0.85 | 0.788 | 0.67 | 21.176 | 15.4 | |
| P1 | 0.67 | 0.91 | 0.61 | 8.955 | 12.08 | |
| P2 | 0.61 | 0.836 | 0.51 | 16.393 | 8.93 | |
| Infected Thrips | Adult male age = (10 days) | 0.19 | 0.105 | 0.02 | 89.474 | 1.05 |
| Adult male age = (15 days) | 0 | 0 | 0 | 100 | 0 | |
| Adult female age = (10 days) | 0.627451 | 0.719 | 0.451 | 28.125 | 8.93 | |
| Adult female (age = 15 days) | 0.23 | 0.348 | 0.08 | 65.217 | 1.74 | |
| Adult female (age = 20 days) | 0.08 | 0 | 0 | 100 | 0 | |
| L1 | 1 | 0.94 | 0.94 | 6 | 24.19 | |
| L2 | 0.94 | 0.936 | 0.88 | 6.383 | 20.52 | |
| P1 | 0.88 | 0.932 | 0.82 | 6.818 | 16.97 | |
| Uninfected Thrips | P2 | 0.82 | 0.817 | 0.67 | 18.293 | 14.36 |
| Adult male age = (10 days) | 0.22 | 0.409 | 0.09 | 59.091 | 4.09 | |
| Adult male age = (15 days) | 0.09 | 0 | 0 | 100 | 0 | |
| Adult female age = (10 days) | 0.45 | 0.956 | 0.43 | 4.444 | 12.11 | |
| Adult female (age = 15 days) | 0.43 | 0.512 | 0.22 | 48.837 | 2.56 | |
| Adult female (age = 20 days) | 0.22 | 0 | 0 | 100 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hameed, A.; Rosa, C.; Rajotte, E.G. The Effect of Species Soybean Vein Necrosis Orthotospovirus (SVNV) on Life Table Parameters of Its Vector, Soybean Thrips (Neohydatothrips variabilis Thysanoptera: Thripidae). Insects 2022, 13, 632. https://doi.org/10.3390/insects13070632
Hameed A, Rosa C, Rajotte EG. The Effect of Species Soybean Vein Necrosis Orthotospovirus (SVNV) on Life Table Parameters of Its Vector, Soybean Thrips (Neohydatothrips variabilis Thysanoptera: Thripidae). Insects. 2022; 13(7):632. https://doi.org/10.3390/insects13070632
Chicago/Turabian StyleHameed, Asifa, Cristina Rosa, and Edwin G. Rajotte. 2022. "The Effect of Species Soybean Vein Necrosis Orthotospovirus (SVNV) on Life Table Parameters of Its Vector, Soybean Thrips (Neohydatothrips variabilis Thysanoptera: Thripidae)" Insects 13, no. 7: 632. https://doi.org/10.3390/insects13070632
APA StyleHameed, A., Rosa, C., & Rajotte, E. G. (2022). The Effect of Species Soybean Vein Necrosis Orthotospovirus (SVNV) on Life Table Parameters of Its Vector, Soybean Thrips (Neohydatothrips variabilis Thysanoptera: Thripidae). Insects, 13(7), 632. https://doi.org/10.3390/insects13070632







