Mosquito Surveillance and Insecticide Resistance Monitoring Conducted by the Florida Keys Mosquito Control District, Monroe County, Florida, USA
Abstract
Simple Summary
Abstract
1. Introduction
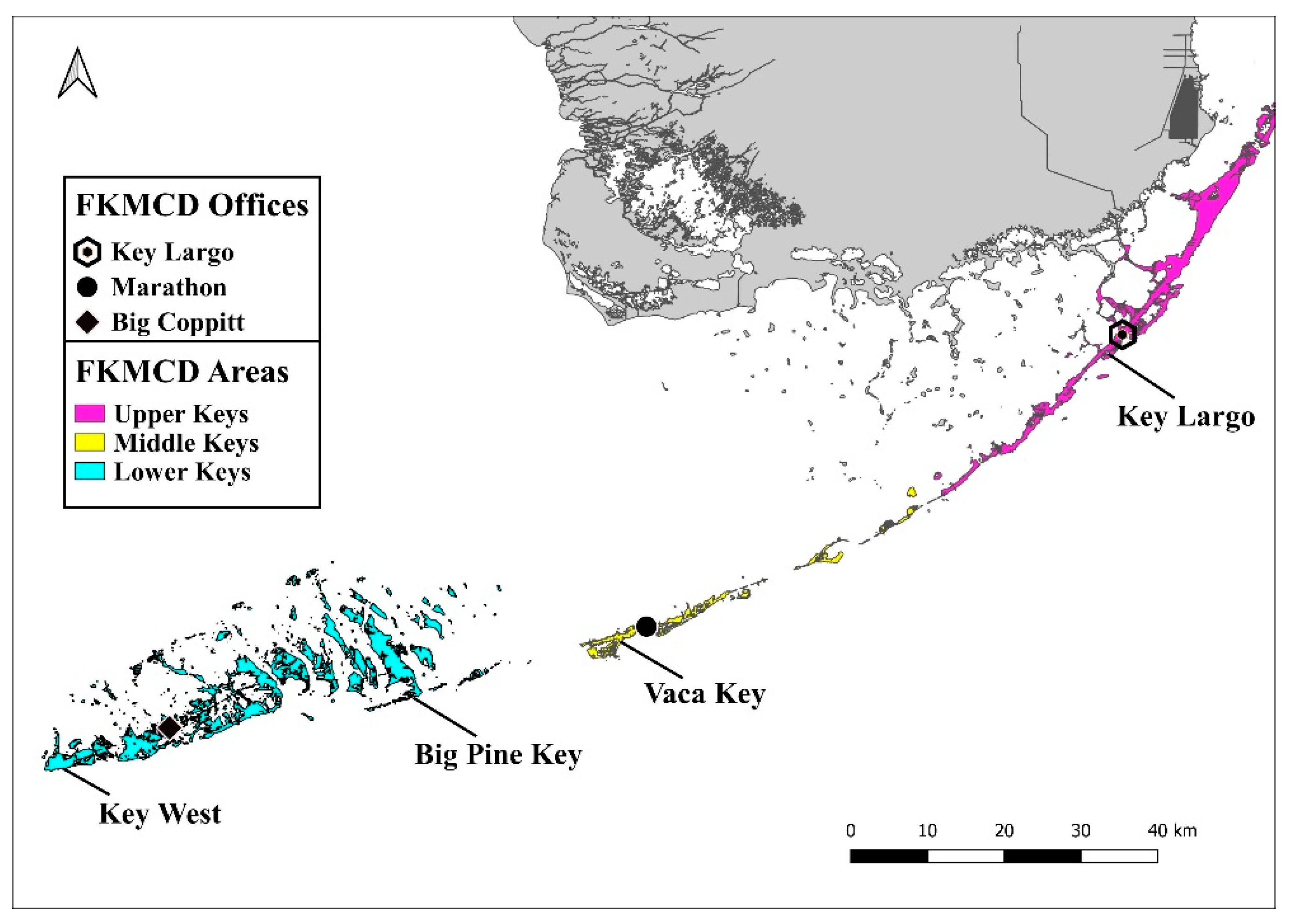
2. Adult Surveillance
2.1. Carbon Dioxide-Baited Light Traps
2.2. BG Sentinel Traps
2.3. Truck Traps
2.4. Gravid Traps and Oviposition Traps
2.5. Landing Rate Counts
3. Larval and Pupal Surveillance
4. Insecticide Resistance Monitoring
4.1. Adulticides
4.2. Larvicides
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Vector-Borne Diseases. Available online: www.who.int/news-room/fact-sheets/detail/vector-borne-diseases (accessed on 31 August 2022).
- Trout, A.; Baracco, G.; Rodriguez, M.; Barber, J.; Leal, A.; Radke, E.; Weis, K.; Stanek, D.; Stark, L.; Blackmore, C.; et al. Locally-acquired Dengue, 2009–2010. MMWR 2010, 59, 577–581. [Google Scholar]
- Likos, A.; Griffin, I.; Bingham, A.M. Local mosquito-borne transmission of Zika Virus—Miami-Dade and Broward Counties, Florida, June–August 2016. MMWR 2016, 65, 1032–1038. [Google Scholar] [CrossRef] [PubMed]
- Florida Health. Mosquito-Borne Disease Surveillance. Available online: https://www.floridahealth.gov/diseases-and-conditions/mosquito-borne-diseases/surveillance.html (accessed on 31 August 2022).
- Wilkerson, R.C.; Linton, Y.-M.; Strickman, D. Mosquitoes of the World; Johns Hopkins University Press: Baltimore, MD, USA, 2021; Volume 1 and Volume 2, pp. 1–1332. [Google Scholar]
- Ramírez, A.L.; Hurk, A.F.V.D.; Meyer, D.B.; Ritchie, S.A. Searching for the proverbial needle in a haystack: Advances in mosquito-borne arbovirus surveillance. Parasites Vectors 2018, 11, 320. [Google Scholar] [CrossRef]
- Environmental Protection Agency. Success in Mosquito Control: An Integrated Approach. Available online: https://www.epa.gov/mosquitocontrol/success-mosquito-control-integrated-approach (accessed on 31 August 2022).
- Floore, T.G. Mosquito larval control practices: Past and present. J. Am. Mosq. Control Assoc. 2006, 22, 527–533. [Google Scholar] [CrossRef]
- Latham, M.; Lloyd, A. Adulticides and Adulticiding. In Florida Mosquito Control 2018: The State of the Mission as Defined by Mosquito Controllers, Regulators, and Environmental Managers; Lloyd, A.M., Connelly, C.R., Carlson, D.B., Eds.; Florida Coordinating Council on Mosquito Control; University of Florida, Institute of Food and Agricultural Sciences; Florida Medical Entomology Laboratory: Vero Beach, FL, USA, 2018; pp. 87–115. [Google Scholar]
- Connelly, R.; Patterson, G. History of Mosquito Control in Florida. In Florida Mosquito Control 2018: The State of the Mission as Defined by Mosquito Controllers, Regulators, and Environmental Managers; Lloyd, A.M., Connelly, C.R., Carlson, D.B., Eds.; Florida Coordinating Council on Mosquito Control; University of Florida, Institute of Food and Agricultural Sciences; Florida Medical Entomology Laboratory: Vero Beach, FL, USA, 2018; pp. 5–12. [Google Scholar]
- Florida Legislature. Florida Statutes Online. Available online: http://www.leg.state.fl.us/STATUTES/ (accessed on 31 August 2022).
- Florida Department of State. Florida Administrative Code. Available online: https://www.flrules.org/default.asp (accessed on 31 August 2022).
- Snyder, J.R.; Herndon, A.; Robertson, W.B., Jr. South Florida Rockland. In Ecosystems of Florida; Myers, R.L., Ewel, J.J., Eds.; University of Central Florida Press: Orlando, FL, USA, 1990; pp. 230–277. [Google Scholar]
- Minno, M.C.; Emmel, T.C. Butterflies of the Florida Keys; Scientific Publishers: Gainesville, FL, USA, 1993; pp. 5–6. [Google Scholar]
- Stern, W.L.; Brizicky, G.K. The woods and flora of the Florida Keys. Introduction. Trop. Woods 1957, 107, 36–65. [Google Scholar]
- Corbett, D.R.; Chanton, J.; Burnett, W.; Dillon, K.; Rutkowski, C.; Fourqurean, J.W. Patterns of groundwater discharge into Florida Bay. Limnol. Oceanogr. 1999, 44, 1045–1055. [Google Scholar] [CrossRef]
- Browne, J.; Peck, S.B. The long-horned beetles of south Florida (Cerambycidae: Coleoptera): Biogeography and relationships with the Bahama Islands and Cuba. Can. J. Zool. 1996, 74, 2154–2169. [Google Scholar] [CrossRef]
- Hoel, D.; Kline, D.; Moore, D.; Burroughs, S. Mosquito Surveillance. In Florida Mosquito Control 2018: The State of the Mission as Defined by Mosquito Controllers, Regulators, and Environmental Managers; Lloyd, A.M., Connelly, C.R., Carlson, D.B., Eds.; Florida Coordinating Council on Mosquito Control; University of Florida, Institute of Food and Agricultural Sciences; Florida Medical Entomology Laboratory: Vero Beach, FL, USA, 2018; pp. 13–27. [Google Scholar]
- Dacko, N.M.; Nava, M.R.; Vitek, C.; Debboun, M. Chapter 7—Mosquito Surveillance. In Mosquitoes, Communities, and Public Health in Texas; Debboun, M., Reyna Nava, M., Rueda, L.M., Eds.; Academic Press: London, UK, 2020; pp. 221–247. [Google Scholar] [CrossRef]
- Osório, H.C.; Zé-Zé, L.; Amaro, F.; Alves, M.J. Mosquito surveillance for prevention and control of emerging mosquito-borne diseases in Portugal—2008–2014. Internat. J. Environ. Res. Pub. Health 2014, 11, 11583–11596. [Google Scholar] [CrossRef]
- Schwab, S.R.; Stone, C.M.; Fonseca, D.M.; Fefferman, N.H. The importance of being urgent: The impact of surveillance target and scale on mosquito-borne disease control. Epidemics 2018, 23, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Sudia, W.D.; Chamberlain, R.W. Battery-operated light trap, an improved model. Mosq. News 1962, 22, 126–129. [Google Scholar]
- Newhouse, V.F.; Chamberlain, R.W.; Johnston, J.G.; Sudia, W.D. Use of dry ice to increase mosquito catches of the CDC miniature light trap. Mosq. News 1966, 26, 30–35. [Google Scholar]
- Rudolfs, W. Chemotropism of mosquitoes. Bull. New Jersey Agric. Exper. Sta. 1922, 367, 1–23. [Google Scholar]
- Gillies, M.T. The role of carbon dioxide in host-finding by mosquitoes (Diptera: Culicidae): A review. Bull. Entomol. Res. 1980, 70, 525–532. [Google Scholar] [CrossRef]
- Cooperband, M.E.; Cardé, R.T. Orientation of Culex mosquitoes to carbon dioxide-baited traps: Flight manoeuvers and trapping efficiency. Med. Vet. Entomol. 2006, 20, 11–26. [Google Scholar] [CrossRef]
- Hribar, L.J. Mosquito (Diptera: Culicidae) collections in the Florida Keys, Monroe County, Florida, USA. Stud. Dipterol. 2002, 9, 679–691. [Google Scholar]
- Darsie, R.F., Jr. First report of Ochlerotatus condolescens (Dyar and Knab) (Diptera: Culicidae) in the United States. Proc. Entomol. Soc. Wash. 2003, 105, 1067–1068. [Google Scholar]
- Darsie, R.F., Jr.; Shroyer, D.A. Culex (Culex) declarator, a mosquito species new to Florida. J. Am. Mosq. Control Assoc. 2004, 20, 224–227. [Google Scholar]
- Darsie, R.F., Jr.; Vlach, J.J.; Fussell, E.M. New addition to the mosquito fauna of United States, Anopheles grabhamii (Diptera: Culicidae). J. Med. Entomol. 2002, 39, 430–431. [Google Scholar] [CrossRef]
- Hribar, L.J. Continued presence of Anopheles albimanus (Diptera: Culicidae) in Monroe County, Florida. Entomol. News 1999, 110, 317–319. [Google Scholar]
- Hribar, L.J. Uncommonly collected mosquitoes from the Florida Keys. Entomol. News 2001, 112, 121. [Google Scholar]
- Hribar, L.J. New and old mosquito records for extreme southern Florida (Diptera: Culicidae). Insecta Mundi 2007, 5, 1–3. [Google Scholar]
- Hribar, L.J.; Cerminara, C. Rediscovery of Aedes scapularis (Diptera: Culicidae) in the Florida Keys. Fla. Entomol. 2021, 104, 145. [Google Scholar] [CrossRef]
- Short, S.; Dos Santos, V.; Hribar, L.J. Range expansion of Aedes bahamensis (Diptera: Culicidae) into the Florida Keys. J. Vector Ecol. 2022, 47, 128–129. [Google Scholar] [CrossRef]
- Murray, H.L.; Pruszynski, C.A.; Leal, A.L.; Hribar, L.J. Establishment of Aedes albopictus (Diptera: Culicidae) in the Florida Keys, 2001–2017. J. Med. Entomol. 2018, 55, 1607–1612. [Google Scholar] [CrossRef] [PubMed]
- Pruszynski, C.A.; Hribar, L.J. New records of lower Diptera for the Florida Keys, USA. Stud. Dipterol. 2012, 19, 58–60. [Google Scholar]
- Boehmler, M.B. Culex coronator: A new species record for Monroe County, Florida. J. Am. Mosq. Control Assoc. 2022, 38, 96–98. [Google Scholar] [CrossRef] [PubMed]
- Boehmler, M.B. Aedeomyia (Aedeomyia) squamipennis (Diptera: Culicidae) new genus and species county record for Monroe County, Florida, USA. J. Am. Mosq. Control Assoc. 2022; in press. [Google Scholar]
- Boehmler, M.B.; Hribar, L.J. Aedes pertinax (Diptera: Culicidae) a lost and found new species record for Monroe County, FL. J. Am. Mosq. Control Assoc. 2022; in press. [Google Scholar]
- Hribar, L.J. Relative abundance of mosquito (Insecta: Diptera) species on Big Pine Key, Florida, USA. J. Vector Ecol. 2005, 30, 322–327. [Google Scholar] [PubMed]
- Hribar, L.J.; Vlach, J.J. Mosquito (Diptera: Culicidae) and biting midge (Diptera: Ceratopogonidae) collections in Florida Keys State Parks. Fla. Scient. 2001, 64, 219–223. [Google Scholar]
- Hribar, L.J.; DeMay, D.J.; Hemmen, M.J.; Leal, A.L. Observations on the distribution and seasonality of Ochlerotatus condolescens (Dyar & Knab) in the Florida Keys, USA (Diptera: Culicidae). Stud. Dipterol. 2005, 12, 361–362. [Google Scholar]
- DeMay, D.J.; Hribar, L.J. Species composition, seasonal distribution, and relative abundance of mosquitoes (Diptera: Culicidae) on Lignumvitae Key in the Florida Keys, USA. Trends Entomol. 2005, 4, 71–74. [Google Scholar]
- DeMay, D.J.; Hribar, L.J. Mosquito fauna of Key Largo, Florida. J. Am. Mosq. Control Assoc. 2008, 24, 471–477. [Google Scholar] [CrossRef]
- Leal, A.L.; Hribar, L.J. Mosquito fauna of wilderness islands within the National Key Deer Refuge and Great White Heron National Wildlife Refuge. J. Am. Mosq. Control Assoc. 2010, 26, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Hribar, L.J. Seasonality of Psorophora johnstonii (Diptera: Culicidae) on No Name Key, Florida. Entomol. News 2019, 128, 188–190. [Google Scholar] [CrossRef]
- Hribar, L.J.; Leal, A.L.; DeMay, D.J.; Tambasco, A.N. Mosquitoes (Insecta: Diptera: Culicidae) of the Florida Keys, Florida, United States of America. Check List J. Species List. Distrib. 2011, 7, 458–464. [Google Scholar] [CrossRef]
- Hribar, L.J.; Fussell, E.M.; Leal, A.L. Larviciding wilderness islands reduces aerial adulticide treatment of populated areas adjacent to national wildlife refuges. J. Am. Mosq. Control Assoc. 2011, 27, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Hribar, L.J.; DeMay, D.J.; Murray, H.L. Life and death of a trap site. Wing Beats 2018, 29, 33–34; 36–38. [Google Scholar]
- Vlach, J.J.; Hall, K.J.; Day, J.F.; Curtis, G.A.; Hribar, L.J.; Fussell, E.M. Interisland dispersal of the black salt marsh mosquito, Ochlerotatus taeniorhynchus (Diptera: Culicidae) in the Florida Keys. J. Am. Mosq. Control Assoc. 2006, 22, 615–621. [Google Scholar] [CrossRef]
- Hribar, L.J.; Vlach, J.J.; DeMay, D.J.; Stark, L.M.; Stoner, R.L.; Godsey, M.S.; Burkhalter, K.L.; Spoto, M.C.; James, S.S.; Smith, J.M.; et al. Mosquitoes infected with West Nile virus in the Florida Keys, Monroe County, Florida, USA. J. Med. Entomol. 2003, 40, 361–363. [Google Scholar] [CrossRef]
- Hribar, L.J.; Stark, L.M.; Stoner, R.L.; DeMay, D.J.; Nordholt, A.L.; Hemmen, M.J.; Vlach, J.J.; Fussell, E.M. Isolation of West Nile virus from mosquitoes (Diptera: Culicidae) in the Florida Keys, Monroe County, Florida. Carib. J. Sci. 2004, 40, 362–367. [Google Scholar]
- Turell, M.J.; Dohm, D.J.; Mores, C.N.; Terracina, L.; Wallette, D.L., Jr.; Hribar, L.J.; Pecor, J.E.; Blow, J.A. Potential for North American mosquitoes (Diptera: Culicidae) to transmit Rift Valley Fever Virus. J. Am. Mosq. Control Assoc. 2008, 24, 502–507. [Google Scholar] [CrossRef]
- Hribar, L.J. Relation of Culex bahamensis Dyar & Knab, 1906 (Diptera: Culicidae) to rainfall and tides on Grassy Key, Florida, USA. Ann. Med. Entomol. 2002, 11, 11–16. [Google Scholar]
- Hribar, L.J. Influence of meteorological variables on mosquito catch by dry ice-baited light traps in the Florida Keys, USA. Fla. Scient. 2017, 80, 165–171. [Google Scholar]
- Hribar, L.J.; DeMay, D.J.; Lund, U.J. The association between meteorological variables and abundance of Aedes taeniorhynchus in the Florida Keys. J. Vector Ecol. 2010, 35, 339–346. [Google Scholar] [CrossRef]
- Hribar, L.J. Dataset for mosquito collections on Big Pine Key, Florida, USA. Data Brief 2019, 26, 1–3. [Google Scholar] [CrossRef]
- Hribar, L.J. Deinocerites cancer Theobald (Diptera: Culicidae) on Grassy Key and Long Key in the Florida Keys, USA. Res. Ideas Outcomes 2019, 5, e36159. [Google Scholar] [CrossRef]
- Hribar, L.J. Dataset for mosquitoes (Diptera: Culicidae) from Vaca Key, Monroe County, Florida USA. Biodiver. Data J. 2020, 8, e55059. [Google Scholar] [CrossRef] [PubMed]
- Hribar, L.J. Dataset for mosquitoes (Diptera: Culicidae) from Cross Key, Monroe County, Florida, USA. Res. Ideas Outcomes 2021, 7, e73768. [Google Scholar] [CrossRef]
- Hribar, L.J. Expanding basic entomological knowledge by using mosquito surveillance bycatch. Ann. Entomol. Soc. Am. 2020, 113, 439–446. [Google Scholar] [CrossRef]
- Kröckel, U.; Rose, A.; Eiras, A.E.; Geier, M. New tools for surveillance of adult Yellow Fever Mosquitoes: Comparison of trap catches with human landing rates in an urban environment. J. Am. Mosq. Control Assoc. 2006, 22, 229–238. [Google Scholar] [CrossRef]
- Pruszynski, C. Notes from the field: Lizards ate my data! Wing Beats 2019, 30, 25–27. [Google Scholar]
- Hribar, L. Diptera other than Culicidae captured in the BG Sentinel mosquito trap. Fly Times 2011, 46, 18. [Google Scholar]
- Maciel-de-Freitas, R.; Eiras, Á.E.; Lourenço-de-Oliveira, R. Field evaluation of effectiveness of the BG-Sentinel, a new trap for capturing adult Aedes aegypti (Diptera: Culicidae). Mem. Inst. Oswaldo Cruz 2006, 101, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Bhalala, H.; Arias, J.R. The Zumba mosquito trap and BG-Sentinel trap: Novel surveillance tools for host-seeking mosquitoes. J. Am. Mosq. Control Assoc. 2009, 25, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.R.; Long, S.A.; Webb, C.E.; Bitzhenner, M.; Geier, M.; Russell, R.C.; Ritchie, S.A. Aedes aegypti population sampling using BG-Sentinel traps in North Queensland Australia: Statistical considerations for trap deployment and sampling strategy. J. Med. Entomol. 2007, 44, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Farajollahi, A.; Kesavaraju, B.; Price, D.C.; Gregory, M.; Healy, S.P.; Gaugler, R.; Nelder, M.P. Field efficacy of BG-Sentinel and industry-standard traps for Aedes albopictus (Diptera: Culicidae) and West Nile Virus surveillance. J. Med. Entomol. 2009, 46, 919–925. [Google Scholar] [CrossRef]
- Williams, C.R.; Long, S.A.; Russell, R.C.; Scott, A. Field efficacy of the BG-Sentinel compared With CDC backpack aspirators and CO2 -baited EVS traps for collection of adult Aedes aegypti in Cairns, Queensland, Australia. J. Am. Mosq. Control Assoc. 2006, 22, 296–300. [Google Scholar] [CrossRef]
- Meeraus, W.H.; Armistead, J.S.; Arias, J.R. Field comparison of novel and gold standard traps for collecting Aedes albopictus in Northern Virginia. J. Am. Mosq. Control Assoc. 2008, 24, 244–248. [Google Scholar] [CrossRef]
- Graham, A.S.; Pruszynski, C.A.; Hribar, L.J.; DeMay, D.J.; Tambasco, A.N.; Hartley, A.E.; Fussell, E.M.; Michael, S.F.; Isern, S. Mosquito-associated Dengue virus, Key West, Florida, USA. 2010. Emerg. Infect. Dis. 2011, 17, 2074–2075. [Google Scholar] [CrossRef]
- Pruszynski, C.A.; Hribar, L.J.; Mickle, R.; Leal, A.L. A large scale biorational approach using Bacillus thuringiensis israeliensis (Strain AM65-52) for managing Aedes aegypti populations to prevent Dengue, Chikungunya and Zika transmission. PLoS ONE 2017, 12, e0170079. [Google Scholar] [CrossRef]
- Baragona, S. Army Laboratories Work Together to Defeat Dengue. Available online: https://www.army.mil/article/51064/army_laboratories_work_together_to_defeat_dengue (accessed on 31 August 2022).
- Kay, J. To Fight Zika, Key West Releases Bacteria-Carrying Mosquitoes. Orlando Sentinel 2017. Available online: https://orlandosentinel.com/politics/os-mosquitoess-bacteria-key-west-20170418-story.html (accessed on 31 August 2022).
- Wright, J.A.; Larson, R.T.; Richardson, A.G.; Cote, N.M.; Stoops, C.A.; Clark, M.; Obenauer, P.J. Comparison of BG-Sentinel(R) Trap and oviposition cups for Aedes aegypti and Aedes albopictus surveillance in Jacksonville, Florida, USA. J. Am. Mosq. Control Assoc. 2015, 31, 26–31. [Google Scholar] [CrossRef]
- Barrera, R.; Mackay, A.; Amador, M. An improved trap to capture adult container-inhabiting mosquitoes. J. Am. Mosq. Control Assoc. 2013, 29, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Ball, T.S.; Ritchie, S.R. Sampling biases of the BG-Sentinel Trap with respect to physiology, age, and body size of adult Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 2010, 47, 649–656. [Google Scholar] [CrossRef] [PubMed]
- Pruszynski, C.A.; Stenn, T.; Acevedo, C.; Leal, A.L.; Burkett-Cadena, N.D. Human blood feeding by Aedes aegypti (Diptera: Culicidae) in the Florida Keys and a review of the literature. J. Med. Entomol. 2020, 57, 1640–1647. [Google Scholar] [CrossRef] [PubMed]
- Pruszynski, C.A. Dataset for Aedes aegypti (Diptera: Culicidae) and Culex quinquefasciatus (Diptera: Culicidae) Collections from Key West, Florida, USA, 2010–2020. Data Brief 2022, 41, 107907. [Google Scholar] [CrossRef]
- Rogers, A. Pasco County Mosquito Control District; Personal Communication: Odessa, FL, USA, 2022. [Google Scholar]
- Center for Disease Control and Prevention Zika Virus. Case Counts in the United States. Available online: https://www.cdc.gov/zika/geo/united-states.html (accessed on 31 August 2022).
- Antoniou, E.; Orovou, E.; Sarella, A.; Iliadou, M.; Rigas, N.; Palaska, E.; Iatrakis, G.; Dagla, M. Zika Virus and the risk of developing microcephaly in infants: A systematic review. Internat. J. Environ. Res. Public Health 2020, 17, 3806. [Google Scholar] [CrossRef]
- Pruszynski, C.; Boehmler, M.; Murray, H. Releasing Oxitec mosquitoes in the Florida Keys. Wing Beats 2022, 33, 40–44. [Google Scholar]
- Chamberlain, J.C.; Lawson, F.R. A mechanical trap for the sampling of aerial insect populations. Mosq. News 1945, 5, 4–7. [Google Scholar]
- Bidlingmayer, W.L. Field activity studies of Culicoides furens. Ann. Entomol. Soc. Am. 1961, 54, 149–156. [Google Scholar] [CrossRef]
- Bidlingmayer, W.L. Use of the truck trap for evaluating adult mosquito populations. Mosq. News 1966, 26, 139–143. [Google Scholar]
- Carroll, M.K.; Bourg, J.A. The night-time flight activity and relative abundance of fifteen species of Louisiana mosquitoes. Mosq. News 1977, 47, 661–664. [Google Scholar]
- Pruszynski, C. The truck trap circles back: Revisiting a method of passive mosquito trapping. Wing Beats 2014, 25, 16, 19–20. [Google Scholar]
- Bidlingmayer, W.L. A comparison of trapping methods for adult mosquitoes: Species response and environmental influence. J. Med. Entomol. 1967, 4, 200–220. [Google Scholar] [CrossRef] [PubMed]
- Bidlingmayer, W.L. The measurement of adult mosquito population changes—Some considerations. J. Am. Mosq. Control Assoc. 1985, 1, 328–348. [Google Scholar] [PubMed]
- Day, J.F. Mosquito oviposition behavior and vector control. Insects 2016, 7, 65. [Google Scholar] [CrossRef] [PubMed]
- Reiter, P. A portable, battery-powered trap for collecting gravid Culex mosquitoes. Mosq. News 1983, 43, 496–498. [Google Scholar]
- Isoe, J.; Beehler, J.W.; Millar, J.G.; Mulla, M.S. Oviposition responses of Culex tarsalis and Culex quinquefasciatus to aged Bermuda grass infusions. J. Am. Mosq. Control Assoc. 1995, 11, 39–44. [Google Scholar]
- Burkett-Cadena, N.; Mullen, G.R. Field comparison of Bermuda-hay infusion to infusions of emergent aquatic vegetation for collecting adult female mosquitoes. J. Am. Mosq. Control Assoc. 2007, 23, 117–123. [Google Scholar] [CrossRef]
- Buxton, P.A.; Hopkins, G.H.E. Researches in Polynesia and Melanesia—Entomology. IV. Experiments performed on Aedes variegatus and Aedes argenteus. Mem. Ser. Lond. Sch. Trop. Med. Hyg. 1927, 1, 125–256. [Google Scholar]
- Fay, R.W.; Eliason, D.A. A preferred oviposition site as a surveillance method for Aedes aegypti. Mosq. News 1966, 26, 531–535. [Google Scholar]
- Brelsfoard, C.L.; Mains, J.W.; Mulligan, S.; Cornel, A.; Holeman, J.; Kluh, S.; Leal, A.; Hribar, L.J.; Morales, H.; Posey, T.; et al. Aedes aegypti males as vehicles for insecticide delivery. Insects 2019, 10, 230. [Google Scholar] [CrossRef]
- Scott, M.L.; Hribar, L.J.; Leal, A.L.; McAllister, J.C. Characterization of pyrethroid resistance mechanisms in Aedes aegypti from the Florida Keys. Amer. J. Trop. Med. Hyg. 2021, 104, 104–1111. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.F. Landing rates and bite counts for nuisance evaluation. In Proceedings of the Seventy-Sixth Annual Meeting of the New Jersey Mosquito Control Association, Asbury Park, NJ, USA, 15–17 March 1989; pp. 34–37. [Google Scholar]
- Headlee, T.J. The Mosquitoes of New Jersey and their Control. N.J. Agric. Exper. Sta. Bull. 1921, 348, 1–229. [Google Scholar]
- Martinez, J.; Showering, A.; Oke, C.; Jones, R.T.; Logan, J.G. Differential attraction in mosquito–human interactions and implications for disease control. Phil. Trans. R. Soc. 2021, 376, 20190811. [Google Scholar] [CrossRef] [PubMed]
- Achee, N.L.; Youngblood, L.; Bangs, M.J.; Lavery, J.V.; James, S. Considerations for the use of human participants in vector biology research: A tool for investigators and regulators. Vector-Borne Zoonotic Dis. 2015, 15, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Jamrozik, E.; de la Fuente-Núñez, V.; Reis, A.; Ringwald, P.; Selgelid, M.J. Ethical aspects of malaria control and research. Malaria J. 2015, 14, 518. [Google Scholar] [CrossRef]
- Gama, R.A.; da Silva, I.M.; Geier, M.; Eiras, A.E. Development of the BG-Malaria trap as an alternative to human-landing catches for the capture of Anopheles darlingi. Mem. Inst. Oswaldo Cruz 2013, 108, 763–771. [Google Scholar] [CrossRef]
- Tambwe, M.M.; Saddler, A.; Kibondo, U.A.; Mashauri, R.; Kreppel, K.S.; Govella, N.J.; Moore, S.J. Semi-field evaluation of the exposure-free mosquito electrocuting trap and BG-Sentinel trap as an alternative to the human landing catch for measuring the efficacy of transfluthrin emanators against Aedes aegypti. Parasites Vectors 2021, 14, 265. [Google Scholar] [CrossRef]
- Gimnig, J.E.; Walker, E.D.; Otieno, P.; Kosgei, J.; Olang, G.; Ombok, M.; Williamson, J.; Marwanga, D.; Abong’o, D.; Desai, M.; et al. Incidence of malaria among mosquito collectors conducting human landing catches in western Kenya. Am. J. Trop. Med. Hyg. 2013, 88, 301–308. [Google Scholar] [CrossRef]
- World Health Organization. Ethical Issues Associated with Vector-Borne Diseases. Report of a WHO Scoping Meeting, Geneva, 23–24 February 2017; World Health Organization: Geneva, Switzerland, 2017; pp. 25, 39. [Google Scholar]
- Geier, M.; Weber, M.; Rose, A.; Obermayr, U.; Abadam, C.; Kiser, J.; Pruszynski, C.; Doyle, M.A. Smart Internet of Things (loT) Device for Monitoring Mosquito Trap Counts in the Field While Drinking Coffee at Your Desk. Available online: https://www.researchgate.net/publication/294580125 (accessed on 27 July 2022).
- Pruszynski, C. The BG-Counter: A new surveillance trap that remotely measures mosquito density in real time. Wing Beats 2016, 27, 13–18. [Google Scholar]
- Day, C.A.; Richards, S.L.; Reiskind, M.H.; Doyle, M.S.; Byrd, B.D. Context-dependent accuracy of the BG-Counter remote mosquito surveillance device in North Carolina. J. Am. Mosq. Control Assoc. 2020, 36, 74–80. [Google Scholar] [CrossRef]
- Johnson, B.J.; Manby, R.; Devine, G.J. What happens on islands, doesn’t stay on islands: Patterns of synchronicity in mosquito nuisance and host-seeking activity between a mangrove island and adjacent coastal development. Urban Ecosyst. 2020, 23, 1321–1333. [Google Scholar] [CrossRef]
- Southey, R. Omniana; Or, Horae Otiosiores: Volume 1; Longman: Hurst, Rees, Orme, and Brown, Paternoster Row: London, UK, 1812; p. 364. [Google Scholar]
- Howard, L.O. Mosquitoes: How They Live; How They Carry Disease; How they Are Classified; How They May Be Destroyed; McClure, Phillips & Co.: New York, NY, USA, 1901; p. 241. [Google Scholar]
- Ritchie, S.A.; Johnson, E.S. Aedes taeniorhynchus (Diptera: Culicidae) oviposition patterns in a Florida mangrove forest. J. Med. Entomol. 1991, 28, 496–500. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, S.A.; Addison, D.S. Oviposition preferences of Aedes taeniorhynchus (Diptera: Culicidae) in Florida mangrove forests. Environ. Entomol. 1992, 21, 737–744. [Google Scholar] [CrossRef]
- O’Malley, C. Seven ways to a successful dipping career. Wing Beats 1995, 6, 23–24. [Google Scholar]
- Nielsen, E.T.; Evans, D.G. Duration of the pupal stage of Aedes taeniorhynchus with a discussion of the velocity of development as a function of temperature. Oikos 1960, 11, 200–222. [Google Scholar] [CrossRef]
- Hribar, L.J.; Vlach, J.J.; DeMay, D.J.; James, S.S.; Fahey, J.S.; Fussell, E.M. Mosquito Larvae (Culicidae) and other Diptera associated with containers, storm drains, and sewage treatment plants in the Florida Keys, Monroe County, Florida. Fla. Entomol. 2004, 87, 199–203. [Google Scholar] [CrossRef]
- Hribar, L.J. Larval Habitats of potential mosquito vectors of West Nile Virus in the Florida Keys. J. Water Health 2007, 5, 97–100. [Google Scholar] [CrossRef]
- Boehmler, M.B. Outbreak of Locally-Transmitted Dengue in Key Largo, Florida, Florida Keys Mosquito Control District, 100701 Overseas Highway: Key Largo, FL, USA, 2022; manuscript in preparation.
- Murray, H.L.; Pruszynski, C.A.; Hribar, L.J. Ground applications of Vectobac® WDG with A1 Super-Duty Mist Sprayer® and Micronair® AU5000 Atomizer for suppression of Aedes aegypti populations in the Florida Keys. J. Am. Mosq. Control Assoc. 2021, 37, 271–279. [Google Scholar] [CrossRef]
- Hribar, L.J.; Whiteside, M.E. Seasonal habitat use by immature Aedes aegypti (Linneaus) (Diptera: Culicidae) in the Florida Keys, USA. Stud. Dipterol. 2010, 17, 237–251. [Google Scholar]
- Hribar, L.J.; Whiteside, M.E. Productivity of container habitats for pupal Culex quinquefasciatus Say and Culex nigripalpus Theobald (Diptera: Culicidae) in the Florida Keys. Trends Entomol. 2011, 7, 33–36. [Google Scholar]
- O’Meara, G.F. The Asian Tiger Mosquito in Florida; Publication Number EENY-319; Electronic Data Information Service—University of Florida, Institute of Food and Agricultural Sciences: Gainesville, FL, USA, 1997. [Google Scholar]
- Medlock, J.M.; Hansford, K.M.; Schaffner, F.; Versteirt, V.; Hendrickx, G.; Zeller, H.; Van Bortel, W. A review of the invasive mosquitoes in Europe: Ecology, public health risks, and control options. Vector-Borne Zoonotic Dis. 2012, 12, 435–447. [Google Scholar] [CrossRef] [PubMed]
- Wong, P.-S.J.; Li, M.I.; Chong, C.-S.; Ng, L.-C.; Tan, C.-H. Aedes (Stegomyia) albopictus (Skuse): A potential vector of Zika virus in Singapore. PLoS Negl. Trop. Dis. 2013, 7, e2348. [Google Scholar] [CrossRef] [PubMed]
- Amraoui, F.; Vazeille, M.; Failloux, A.B. French Aedes albopictus are able to transmit yellow fever virus. Euro. Surveill. 2016, 21, 30361. [Google Scholar] [CrossRef] [PubMed]
- O’Meara, G.F.; Evans, L.F.; Gettman, A.D.; Cuda, J.P. Spread of Aedes albopictus and decline of Aedes aegypti (Diptera: Culicidae) in Florida. J. Med. Entomol. 1995, 32, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Reiskind, M.H.; Lounibos, L.P. Spatial and temporal patterns of abundance of Aedes aegypti L. (Stegomyia aegypti) and Aedes albopictus (Skuse) [Stegomyia albopictus (Skuse)] in southern Florida. Med. Vet. Entomol. 2013, 27, 421–429. [Google Scholar] [CrossRef]
- Hopperstead, K.A.; Reiskind, M.H. Recent changes in the local distribution of Aedes aegypti (Diptera: Culicidae) in South Florida, USA. J. Med. Entomol. 2016, 53, 836–842. [Google Scholar] [CrossRef]
- Tambasco, A.N.; Hribar, L.J. First record of Toxorhynchites rutilus rutilus (Coquillett) (Diptera: Culicidae) from the Florida Keys, USA. Stud. Dipterol. 2013, 20, 68–70. [Google Scholar]
- Rawlins, S.C.; Martinez, R.; Wiltshire, S.; Legall, G. A comparison of Surveillance Systems for the Dengue Vector Aedes aegypti in Port of Spain, Trinidad. J. Am. Mosq. Control Assoc. 1998, 14, 131–136. [Google Scholar] [PubMed]
- Codeço, C.T.; Lima, A.W.S.; Araújo, S.C.; Lima, J.B.P.; Maciel-de-Freitas, R.; Honório, N.A.; Galardo, A.K.R.; Braga, I.A.; Coelho, G.E.; Valle, D. Surveillance of Aedes aegypti: Comparison of house index with four alternative traps. PLoS Negl. Trop. Dis. 2015, 9, e0003475. [Google Scholar] [CrossRef]
- Cuervo-Parra, J.A.; Cortes, T.R.; Ramirez-Lepe, M. Mosquito-borne diseases, pesticides used for mosquito control, and development of resistance to insecticides. In Insecticides Resistance; Trdan, S., Ed.; InTech Open: London, UK, 2016. [Google Scholar]
- Labbé, P.; David, J.-P.; Alout, H.; Milesi, P.; Djogbénou, L.; Pasteur, N.; Weill, M. Evolution of resistance to insecticide in disease vectors. In Genetics and Evolution of Infectious Diseases; Tibayrenc, M., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 313–339. [Google Scholar] [CrossRef]
- Carrasco, D.; Lefèvre, T.; Moiroux, N.; Pennetier, C.; Chandre, F.; Cohuet, A. Behavioral adaptations of mosquito vectors to insecticide control. Curr. Opin. Ins. Sci. 2019, 34, 48–54. [Google Scholar] [CrossRef]
- Estep, A.S.; Sanscrainte, N.D.; Waits, C.M.; Bernard, S.J.; Lloyd, A.M.; Lucas, K.J.; Buckner, E.A.; Vaidyanathan, R.; Morreale, R.; Conti, L.A.; et al. Quantification of permethrin resistance and kdr alleles in Florida strains of Aedes aegypti (L.) and Aedes albopictus (Skuse). PLOS Negl. Trop. Dis. 2018, 12, e0006544. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). CONUS Manual for Evaluating Insecticide Resistance in Mosquitoes Using the CDC Bottle Bioassay Kit. Available online: https://www.cdc.gov/mosquitoes/mosquito-control/professionals/cdc-bottle-bioassay.html (accessed on 13 May 2022).
- Bingham, G.; Strode, C.; Tran, L.; Thi Khoa, P.; Pates Jamet, H. Can piperonyl butoxide enhance the efficacy of pyrethroids against pyrethroid-resistant Aedes aegypti? Trop. Med. Intl. Health. 2011, 16, 492–500. [Google Scholar] [CrossRef] [PubMed]
- Hribar, L.J.; Murray, H.L. Toxicity of tau-fluvalinate, lambda-cyhalothrin, and alpha-cypermethrin to Aedes aegypti, 2019. Arthropod Mgmt. Tests 2019, 44, 1–2. [Google Scholar] [CrossRef]
- Hribar, L.J.; Murray, H.L.; McIntire, S.G.; Pruszynski, C.A. Effects of mosquito control adulticides on sterile Cochliomyia hominivorax (Diptera: Calliphoridae). J. Econ. Entomol. 2018, 111, 959–966. [Google Scholar] [CrossRef]
- Zhong, H.; Hribar, L.J.; Daniels, J.C.; Feken, M.A.; Brock, C.; Trager, M.D. Aerial ultra-low-volume application of naled: Impact on nontarget imperiled butterfly larvae (Cyclargus thomasi bethunebakeri) and efficacy against adult mosquitoes (Aedes taeniorhynchus). Environ. Entomol. 2010, 39, 1961–1972. [Google Scholar] [CrossRef] [PubMed]
- PACVEC (Pacific Southwest Regional Center of Excellence in Vector-Borne Diseases). Larval Mosquito Resistance Testing Videos. Available online: https://pacvec.us/larval-mosquito-resistance-testing-video/ (accessed on 18 July 2022).
- Murray, H.L. Problem-solving problem storm drains in the Florida Keys. Wing Beats 2020, 31, 15–20. [Google Scholar]
- Hribar, L.J. Increased water hardness in catch basins treated with spinosad (Natular XRT) extended release tablets. J. Fla. Mosq. Control Assoc. 2020, 67, 60–63. [Google Scholar] [CrossRef]
- Floore, T.G.; Smith, J.P.; Shaffer, K.R.; Schreiber, E.T. Laboratory bioassay studies to determine methoprene susceptibility in a natural population of Ochlerotatus taeniorhynchus from the Florida Keys. J. Am. Mosq. Control Assoc. 2002, 18, 111–113. [Google Scholar]
- Pruszynski, C.A. Laboratory bioassays to determine methoprene susceptibility in Aedes aegypti populations in the Florida Keys, 2021. Arthropod Mnmt. Tests 2022, 47, 1–2. [Google Scholar] [CrossRef]
- Hribar, L.J.; Murray, H.L. Toxicity of naled and eugenol to mosquito larvae, 2018. Arthropod Mnmt. Tests 2019, 44, 1–2. [Google Scholar] [CrossRef]
| Trap Type | ||||
|---|---|---|---|---|
| ABC/CDC | BG Sentinel | Oviposition | LRC (Per Day) | |
| Lower Keys | 33 | 18 | 6 | 121 |
| Middle Keys | 10 | 3 | 0 | 46 |
| Upper Keys | 19 | 11 | 0 | 56 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hribar, L.J.; Boehmler, M.B.; Murray, H.L.; Pruszynski, C.A.; Leal, A.L. Mosquito Surveillance and Insecticide Resistance Monitoring Conducted by the Florida Keys Mosquito Control District, Monroe County, Florida, USA. Insects 2022, 13, 927. https://doi.org/10.3390/insects13100927
Hribar LJ, Boehmler MB, Murray HL, Pruszynski CA, Leal AL. Mosquito Surveillance and Insecticide Resistance Monitoring Conducted by the Florida Keys Mosquito Control District, Monroe County, Florida, USA. Insects. 2022; 13(10):927. https://doi.org/10.3390/insects13100927
Chicago/Turabian StyleHribar, Lawrence J., Michael B. Boehmler, Heidi L. Murray, Catherine A. Pruszynski, and Andrea L. Leal. 2022. "Mosquito Surveillance and Insecticide Resistance Monitoring Conducted by the Florida Keys Mosquito Control District, Monroe County, Florida, USA" Insects 13, no. 10: 927. https://doi.org/10.3390/insects13100927
APA StyleHribar, L. J., Boehmler, M. B., Murray, H. L., Pruszynski, C. A., & Leal, A. L. (2022). Mosquito Surveillance and Insecticide Resistance Monitoring Conducted by the Florida Keys Mosquito Control District, Monroe County, Florida, USA. Insects, 13(10), 927. https://doi.org/10.3390/insects13100927






