Diversity and Global Distribution of Viruses of the Western Honey Bee, Apis mellifera
Abstract
1. Introduction
2. Diversity of Viruses of the Western Honey Bee
2.1. What Are the Viruses of the Western Honey Bee?
2.2. Diversity of Viruses of the Honey Bee
2.3. Symptoms of Viral Infections of the Honey Bee
3. Distribution of Viruses of the Western Honey Bee
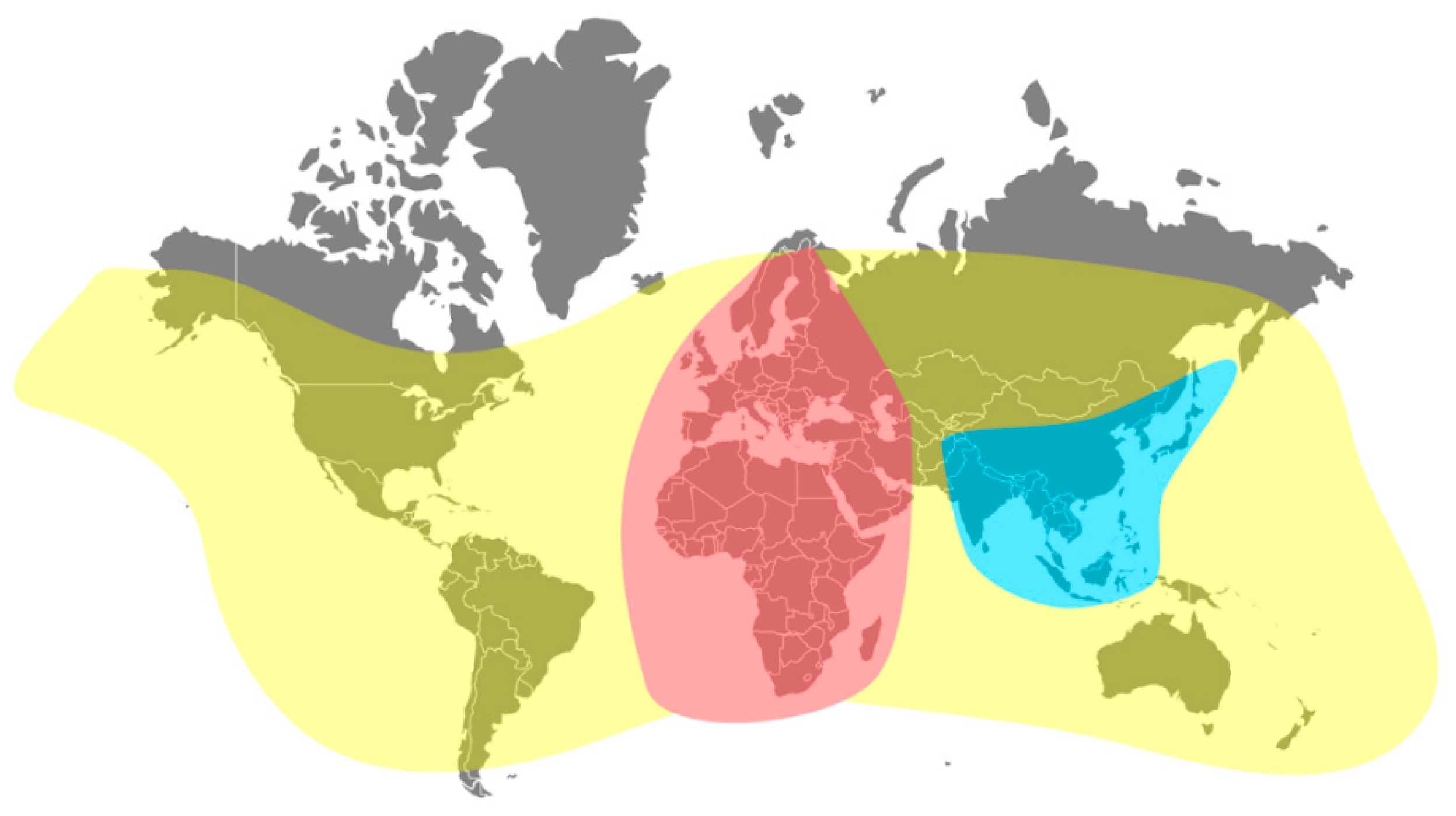
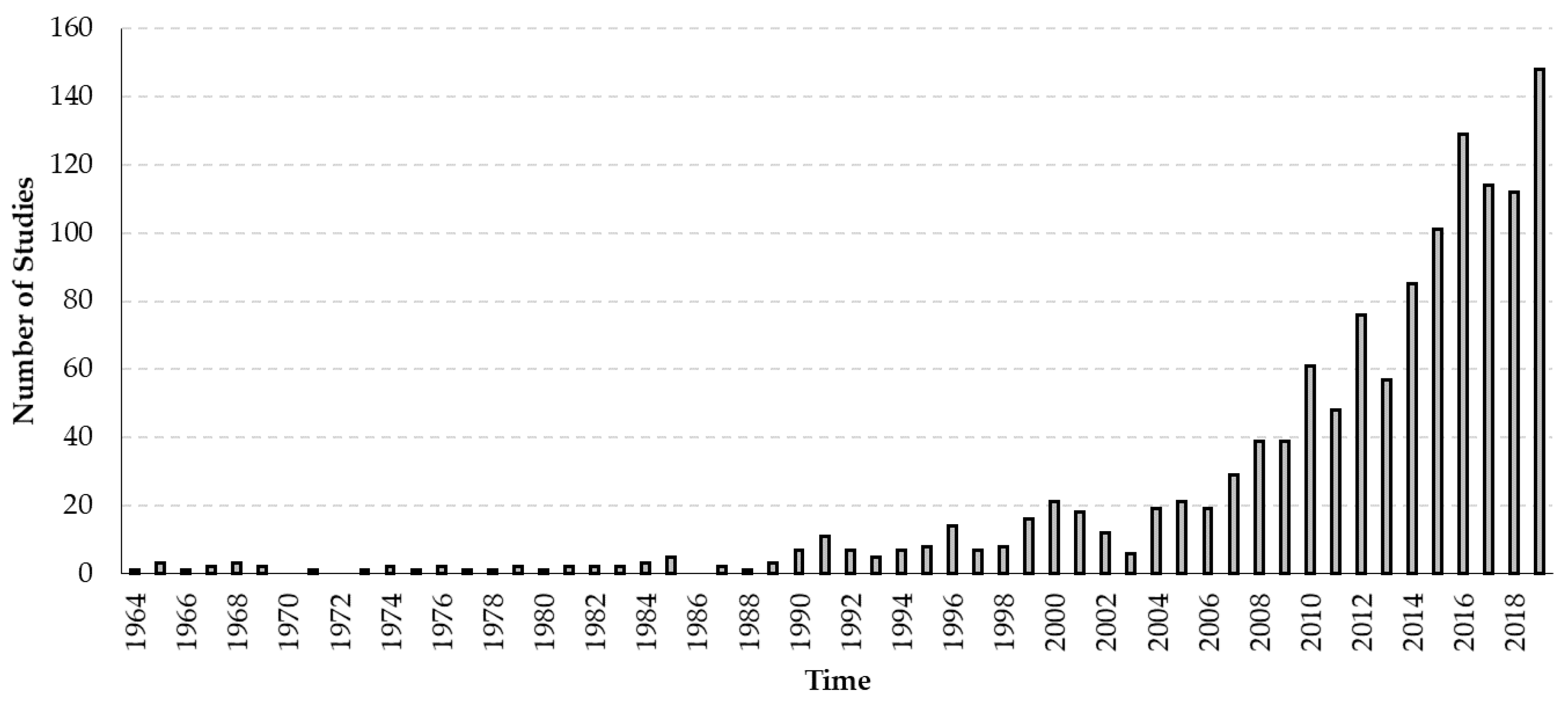
3.1. Global Dissemination of Viruses of the Honey Bee
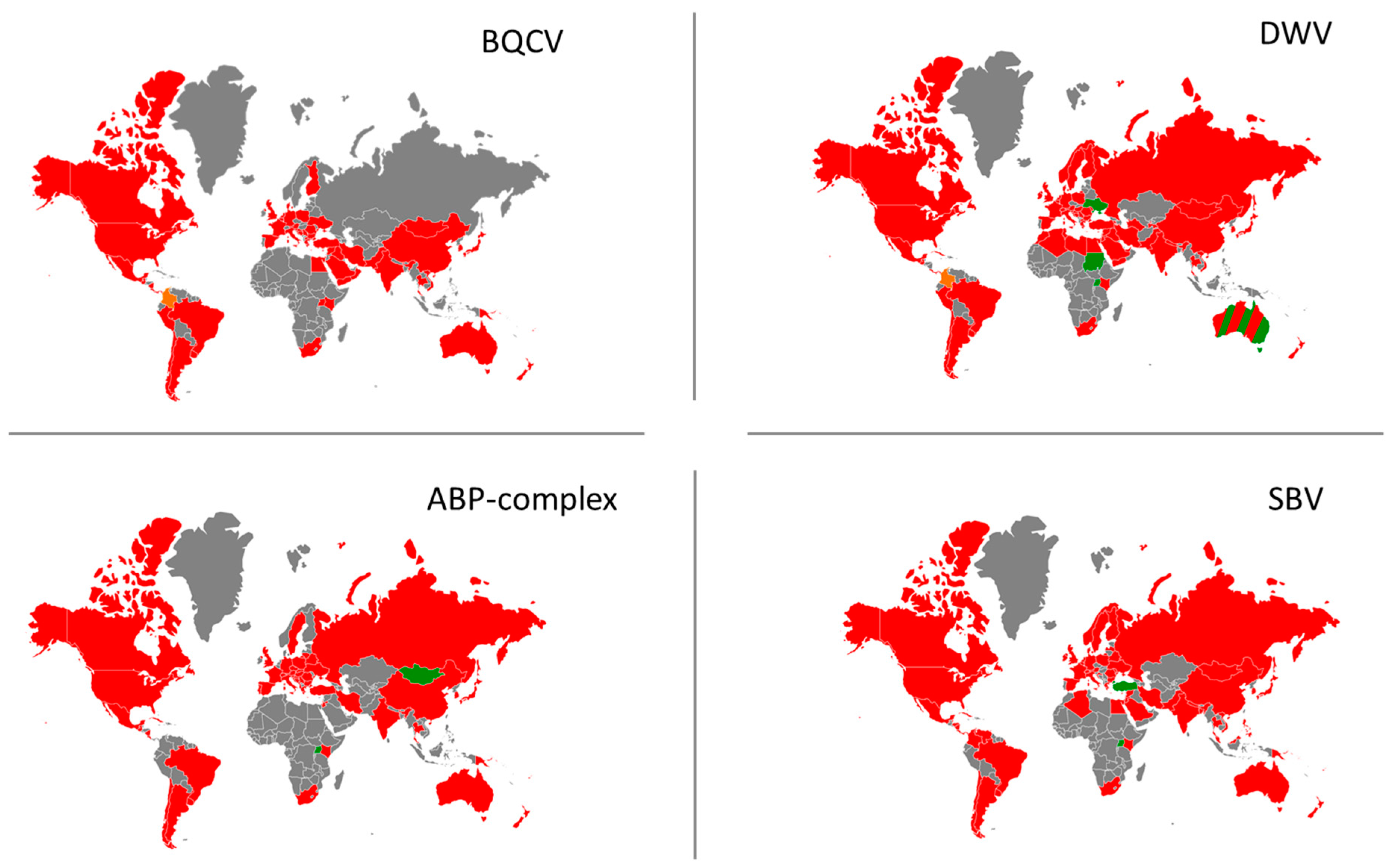
3.2. Current Geographical Distribution
3.3. Temporal Dynamics of Viruses Infecting Honey Bees
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kolar, C.S.; Lodge, D.M. Progress in invasion biology: Predicting invaders. Trends Ecol. Evol. 2001, 16, 199–204. [Google Scholar] [CrossRef]
- Tatem, A.J.; Rogers, D.J.; Hay, S.I. Global transport networks and infectious disease spread. Adv. Parasitol. 2006, 62, 293–343. [Google Scholar] [PubMed]
- Essl, F.; Dullinger, S.; Rabitsch, W.; Hulme, P.E.; Hülber, K.; Jarošík, V.; Kleinbauer, I.; Krausmann, F.; Kühn, I.; Nentwig, W.; et al. Socioeconomic legacy yields an invasion debt. Proc. Natl. Acad. Sci. USA 2011, 108, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Daszak, P.; Cunningham, A.A.; Hyatt, A.D. Emerging infectious diseases of wildlife—Threats to biodiversity and human health. Science 2000, 287, 443–449. [Google Scholar] [CrossRef]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef]
- Morens, D.M.; Folkers, G.K.; Fauci, A.S. Emerging infections: A perpetual challenge. Lancet Infect. Dis. 2008, 8, 710–719. [Google Scholar] [CrossRef]
- Bush, K.; Courvalin, P.; Dantas, G.; Davies, J.; Eisenstein, B.; Huovinen, P.; Jacoby, G.A.; Kishony, R.; Kreiswirth, B.N.; Kutter, E.; et al. Tackling antibiotic resistance. Nat. Rev. Microbiol. 2011, 9, 894–896. [Google Scholar] [CrossRef]
- Dawson, W.; Moser, D.; Van Kleunen, M.; Kreft, H.; Pergl, J.; Pyšek, P.; Weigelt, P.; Winter, M.; Lenzner, B.; Blackburn, T.M.; et al. Global hotspots and correlates of alien species richness across taxonomic groups. Nat. Ecol. Evol. 2017, 1, 1–7. [Google Scholar] [CrossRef]
- Colwell, R.R. Global climate and infectious disease: The cholera paradigm. Science 1996, 274, 2025–2031. [Google Scholar] [CrossRef]
- Lindgren, E.; Andersson, Y.; Suk, J.E.; Sudre, B.; Semenza, J.C. Monitoring EU emerging infectious disease risk due to climate change. Science 2012, 336, 418–419. [Google Scholar] [CrossRef]
- Mihara, T.; Koyano, H.; Hingamp, P.; Grimsley, N.; Goto, S.; Ogata, H. Taxon Richness of “Megaviridae” exceeds those of bacteria and archaea in the ocean. Microb. Environ. 2018, 33, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.C. The Evolution and Emergence of RNA Viruses; Oxford University Press: Oxford, UK, 2009. [Google Scholar]
- Bowen, D.G.; Walker, C.M. Adaptive immune responses in acute and chronic hepatitis C virus infection. Nature 2005, 436, 946–952. [Google Scholar] [CrossRef]
- Choisy, M.; Woelk, C.H.; Guégan, J.-F.; Robertson, D.L. Comparative study of adaptive molecular evolution in different human immunodeficiency virus groups and subtypes. J. Virol. 2004, 78, 1962–1970. [Google Scholar] [CrossRef] [PubMed]
- Pybus, O.G.; Rambaut, A. Evolutionary analysis of the dynamics of viral infectious disease. Nat. Rev. Genet. 2009, 10, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Duffy, S.; Shackelton, L.A.; Holmes, E.C. Rates of evolutionary change in viruses: Patterns and determinants. Nat. Rev. Genet. 2008, 9, 267–276. [Google Scholar] [CrossRef]
- Edwards, R.A.; Rohwer, F. Viral metagenomics. Nat. Rev. Microbiol. 2005, 3, 504–510. [Google Scholar] [CrossRef]
- Simmonds, P.; Adams, M.J.; Benkő, M.; Breitbart, M.; Brister, J.R.; Carstens, E.B.; Davison, A.J.; Delwart, E.; Gorbalenya, A.E.; Harrach, B.; et al. Virus taxonomy in the age of metagenomics. Nat. Rev. Microbiol. 2017, 15, 161–168. [Google Scholar] [CrossRef]
- Delwart, E. A Roadmap to the human virome. PLoS Pathog. 2013, 9, e1003146. [Google Scholar] [CrossRef]
- Brum, J.R.; Ignacio-Espinoza, J.C.; Roux, S.; Doulcier, G.; Acinas, S.G.; Alberti, A.; Chaffron, S.; Cruaud, C.; de Vargas, C.; Gasol, J.M.; et al. Patterns and ecological drivers of ocean viral communities. Science 2015, 348, 1261498. [Google Scholar] [CrossRef]
- Yoshikawa, G.; Blanc-Mathieu, R.; Song, C.; Kayama, Y.; Mochizuki, T.; Murata, K.; Ogata, H.; Takemura, M. Medusavirus, a novel large DNA virus discovered from hot spring water. J. Virol. 2019, 93, e02130-18. [Google Scholar] [CrossRef]
- Roossinck, M.J. Move over, bacteria! Viruses make their mark as mutualistic microbial symbionts. J. Virol. 2015, 89, 6532–6535. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Lin, X.-D.; Tian, J.-H.; Chen, L.-J.; Chen, X.; Li, C.-X.; Qin, X.-C.; Li, J.; Cao, J.-P.; Eden, J.-S.; et al. Redefining the invertebrate RNA virosphere. Nature 2016, 540, 539–543. [Google Scholar] [CrossRef] [PubMed]
- De Miranda, J.R.; Bailey, L.; Ball, B.V.; Blanchard, P.; Budge, G.E.; Chejanovsky, N.; Chen, Y.-P.; Gauthier, L.; Genersch, E.; de Graaf, D.C.; et al. Standard methods for virus research in Apis mellifera. J. Apic. Res. 2013, 52, 1–56. [Google Scholar] [CrossRef]
- Di Prisco, G.; Iannaccone, M.; Ianniello, F.; Ferrara, R.; Caprio, E.; Pennacchio, F.; Capparelli, R. The neonicotinoid insecticide Clothianidin adversely affects immune signaling in a human cell line. Sci. Rep. 2017, 7, 13446. [Google Scholar] [CrossRef]
- Doublet, V.; Labarussias, M.; de Miranda, J.R.; Moritz, R.F.A.; Paxton, R.J. Bees under stress: Sublethal doses of a neonicotinoid pesticide and pathogens interact to elevate honey bee mortality across the life cycle. Environ. Microbiol. 2015, 17, 969–983. [Google Scholar] [CrossRef]
- Degrandi-Hoffman, G.; Chen, Y.; Watkins Dejong, E.; Chambers, M.L.; Hidalgo, G. Effects of oral exposure to fungicides on honey bee nutrition and virus levels. J. Econ. Entomol. 2015, 108, 2518–2528. [Google Scholar] [CrossRef]
- Tehel, A.; Brown, M.J.F.; Paxton, R.J. Impact of managed honey bee viruses on wild bees. Curr. Opin. Virol. 2016, 19, 16–22. [Google Scholar] [CrossRef]
- Grozinger, C.M.; Flenniken, M.L. Bee viruses: Ecology, pathogenicity, and impacts. Annu. Rev. Entomol. 2019, 64, 205–226. [Google Scholar] [CrossRef]
- McMenamin, A.J.; Flenniken, M.L. Recently identified bee viruses and their impact on bee pollinators. Curr. Opin. Insect Sci. 2018, 26, 120–129. [Google Scholar] [CrossRef]
- De Miranda, J.R.; Genersch, E. Deformed wing virus. J. Invertebr. Pathol. 2010, 103, S48–S61. [Google Scholar] [CrossRef]
- Levin, S.; Sela, N.; Chejanovsky, N. Two novel viruses associated with the Apis mellifera pathogenic mite Varroa destructor. Sci. Rep. 2016, 6, 37710. [Google Scholar] [CrossRef] [PubMed]
- Remnant, E.J.; Shi, M.; Buchmann, G.; Blacquière, T.; Holmes, E.C.; Beekman, M.; Ashe, A. A diverse range of novel rna viruses in geographically distinct honey bee populations. J. Virol. 2017, 91, e00158-17. [Google Scholar] [CrossRef] [PubMed]
- Levin, S.; Sela, N.; Erez, T.; Nestel, D.; Pettis, J.; Neumann, P.; Chejanovsky, N. New viruses from the ectoparasite mite Varroa destructor infesting Apis mellifera and Apis cerana. Viruses 2019, 11, 94. [Google Scholar] [CrossRef] [PubMed]
- Bailey, L. Honey bee viruses. Sci. Progress 1971, 59, 309–323. [Google Scholar]
- Bailey, L.; Ball, B.V. Honey Bee Pathology; Elsevier: Amsterdam, The Netherlands, 1991. [Google Scholar]
- Bailey, L.; Ball, B.V.; Perry, J.N. The prevalence of viruses of honey bees in Britain. Ann. Appl. Biol. 1981, 97, 109–118. [Google Scholar] [CrossRef]
- Chen, Y.; Evans, J.; Feldlaufer, M. Horizontal and vertical transmission of viruses in the honey bee, Apis mellifera. J. Invertebr. Pathol. 2006, 92, 152–159. [Google Scholar] [CrossRef]
- Yañez, O.; Zheng, H.-Q.; Su, X.-L.; Hu, F.-L.; Neumann, P.; Dietemann, V. Potential for virus transfer between the honey bees Apis mellifera and A. cerana. J. Apic. Res. 2015, 54, 179–191. [Google Scholar] [CrossRef]
- Koch, R. Uber bakteriologische Forschung. In Proceedings of the Verhandlung des X Internationalen Medichinischen Congresses, Berlin, Germany, 4–9 August 1890. [Google Scholar]
- Byrd, A.L.; Segre, J.A. Adapting Koch’s postulates. Science 2016, 351, 224–226. [Google Scholar] [CrossRef]
- Fredericks, D.N.; Relman, D.A. Sequence-based identification of microbial pathogens: A reconsideration of Koch’s postulates. Clin. Microbiol. Rev. 1996, 9, 18–33. [Google Scholar] [CrossRef]
- Cox-Foster, D.L.; Conlan, S.; Holmes, E.C.; Palacios, G.; Evans, J.D.; Moran, N.A.; Quan, P.-L.; Briese, T.; Hornig, M.; Geiser, D.M.; et al. A metagenomic survey of microbes in honey bee colony collapse disorder. Science 2007, 318, 283–287. [Google Scholar] [CrossRef]
- Cornman, S.R.; Schatz, M.C.; Johnston, S.J.; Chen, Y.-P.; Pettis, J.; Hunt, G.; Bourgeois, L.; Elsik, C.; Anderson, D.; Grozinger, C.M.; et al. Genomic survey of the ectoparasitic mite Varroa destructor, a major pest of the honey bee Apis mellifera. BMC Genom. 2010, 11, 602. [Google Scholar] [CrossRef] [PubMed]
- Runckel, C.; Flenniken, M.L.; Engel, J.C.; Ruby, J.G.; Ganem, D.; Andino, R.; DeRisi, J.L. Temporal analysis of the honey bee microbiome reveals four novel viruses and seasonal prevalence of known viruses, Nosema, and Crithidia. PLoS ONE 2011, 6, e20656. [Google Scholar] [CrossRef] [PubMed]
- Granberg, F.; Vicente-Rubiano, M.; Rubio-Guerri, C.; Karlsson, O.E.; Kukielka, D.; Belák, S.; Sánchez-Vizcaíno, J.M. Metagenomic Detection of viral pathogens in Spanish honeybees: Co-Infection by Aphid lethal paralysis, Israel acute paralysis and Lake Sinai viruses. PLoS ONE 2013, 8, e57459. [Google Scholar] [CrossRef] [PubMed]
- Schoonvaere, K.; Smagghe, G.; Francis, F.; de Graaf, D.C. Study of the metatranscriptome of eight social and solitary wild bee species reveals novel viruses and bee parasites. Front. Microbiol. 2018, 9, 177. [Google Scholar] [CrossRef]
- Schoonvaere, K.; De Smet, L.; Smagghe, G.; Vierstraete, A.; Braeckman, B.P.; de Graaf, D.C. Unbiased RNA shotgun metagenomics in social and solitary wild bees detects associations with Eukaryote parasites and new viruses. PLoS ONE 2016, 11, e0168456. [Google Scholar] [CrossRef]
- Edie, S.M.; Smits, P.D.; Jablonski, D. Probabilistic models of species discovery and biodiversity comparisons. Proc. Natl. Acad. Sci. USA 2017, 114, 3666–3671. [Google Scholar] [CrossRef]
- Roberts, J.M.K.; Anderson, D.L.; Durr, P.A. Metagenomic analysis of Varroa-free Australian honey bees (Apis mellifera) shows a diverse Picornavirales virome. J. Gen. Virol. 2018, 99, 818–826. [Google Scholar] [CrossRef]
- Locke, B.; Semberg, E.; Forsgren, E.; de Miranda, J.R. Persistence of subclinical deformed wing virus infections in honeybees following Varroa mite removal and a bee population turnover. PLoS ONE 2017, 12, e0180910. [Google Scholar] [CrossRef]
- De Miranda, J.R. Viruses in Bees. Bee World 2012, 89, 2–5. [Google Scholar] [CrossRef]
- Amiri, E.; Meixner, M.; Nielsen, S.L.; Kryger, P. Four categories of viral infection describe the health status of honey bee colonies. PLoS ONE 2015, 10, e0140272. [Google Scholar] [CrossRef]
- Schurr, F.; Tison, A.; Militano, L.; Cheviron, N.; Sircoulomb, F.; Rivière, M.-P.; Ribière-Chabert, M.; Thiéry, R.; Dubois, E. Validation of quantitative real-time RT-PCR assays for the detection of six honeybee viruses. J. Virol. Methods 2019, 270, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Yañez, O.; Jaffé, R.; Jarosch, A.; Fries, I.; Moritz, R.F.A.; Paxton, R.J.; de Miranda, J.R. Deformed wing virus and drone mating flights in the honey bee (Apis mellifera): Implications for sexual transmission of a major honey bee virus. Apidologie 2012, 43, 17–30. [Google Scholar] [CrossRef]
- Gauthier, L.; Tentcheva, D.; Tournaire, M.; Dainat, B.; Cousserans, F.; Colin, M.E.; Bergoin, M. Viral load estimation in asymptomatic honey bee colonies using the quantitative RT-PCR technique. Apidologie 2007, 38, 426–435. [Google Scholar] [CrossRef]
- Fievet, J.; Tentcheva, D.; Gauthier, L.; de Miranda, J.; Cousserans, F.; Colin, M.E.; Bergoin, M. Localization of deformed wing virus infection in queen and drone Apis mellifera L. Virol. J. 2006, 3, 16. [Google Scholar] [CrossRef]
- Traniello, I.M.; Bukhari, S.A.; Kevill, J.; Ahmed, A.C.; Hamilton, A.R.; Naeger, N.L.; Schroeder, D.C.; Robinson, G.E. Meta-analysis of honey bee neurogenomic response links Deformed wing virus type A to precocious behavioral maturation. Sci. Rep. 2020, 10, 3101. [Google Scholar] [CrossRef]
- Benaets, K.; Van Geystelen, A.; Cardoen, D.; De Smet, L.; de Graaf, D.C.; Schoofs, L.; Larmuseau, M.H.D.; Brettell, L.E.; Martin, S.J.; Wenseleers, T. Covert deformed wing virus infections have long-term deleterious effects on honeybee foraging and survival. Proc. R. Soc. B Biol. Sci. 2017, 284, 20162149. [Google Scholar] [CrossRef]
- Coulon, M.; Dalmon, A.; Di Prisco, G.; Prado, A.; Arban, F.; Dubois, E.; Ribière-Chabert, M.; Alaux, C.; Thiéry, R.; Le Conte, Y. Interactions between thiamethoxam and Deformed wing virus can drastically impair flight behaviour of honey bees. Front. Microbiol. 2020, (in press).
- Galbraith, D.A.; Fuller, Z.L.; Ray, A.M.; Brockmann, A.; Frazier, M.; Gikungu, M.W.; Martinez, J.F.I.; Kapheim, K.M.; Kerby, J.T.; Kocher, S.D.; et al. Investigating the viral ecology of global bee communities with high-throughput metagenomics. Sci. Rep. 2018, 8, 8879. [Google Scholar] [CrossRef]
- Dolan, P.T.; Whitfield, Z.J.; Andino, R. Mechanisms and concepts in RNA virus population dynamics and evolution. Annu. Rev. Virol. 2018, 5, 69–92. [Google Scholar] [CrossRef]
- Wilfert, L.; Long, G.; Leggett, H.C.; Schmid-Hempel, P.; Butlin, R.; Martin, S.J.M.; Boots, M. Honeybee disease: Deformed wing virus is a recent global epidemic in honeybees driven by Varroa mites. Science 2016, 351, 594–597. [Google Scholar] [CrossRef]
- McMahon, D.P.; Natsopoulou, M.E.; Doublet, V.; Fürst, M.A.; Weging, S.; Brown, M.J.F.; Gogol-Döring, A.; Paxton, R.J. Elevated virulence of an emerging viral genotype as a driver of honeybee loss. Proc. R. Soc. B Biol. Sci. 2016, 283, 20160811. [Google Scholar] [CrossRef] [PubMed]
- Gisder, S.; Möckel, N.; Eisenhardt, D.; Genersch, E. In vivo evolution of viral virulence: Switching of deformed wing virus between hosts results in virulence changes and sequence shifts. Environ. Microbiol. 2018, 20, 4612–4628. [Google Scholar] [CrossRef] [PubMed]
- Tehel, A.; Vu, Q.; Bigot, D.; Gogol-Döring, A.; Koch, P.; Jenkins, C.; Doublet, V.; Theodorou, P.; Paxton, R. The two prevalent genotypes of an emerging infectious disease, deformed wing virus, cause equally low pupal mortality and equally high wing deformities in host honey bees. Viruses 2019, 11, 114. [Google Scholar] [CrossRef] [PubMed]
- Ryabov, E.V.; Childers, A.K.; Chen, Y.; Madella, S.; Nessa, A.; vanEngelsdorp, D.; Evans, J.D. Recent spread of Varroa destructor virus-1, a honey bee pathogen, in the United States. Sci. Rep. 2017, 7, 17447. [Google Scholar] [CrossRef]
- Kevill, J.L.; de Souza, F.S.; Sharples, C.; Oliver, R.; Schroeder, D.C.; Martin, S.J. DWV-A lethal to honey bees (Apis mellifera): A colony level survey of DWV variants (A, B, and C) in England, Wales, and 32 states across the US. Viruses 2019, 11, 426. [Google Scholar] [CrossRef]
- Ongus, J.R.; Peters, D.; Bonmatin, J.-M.; Bengsch, E.; Vlak, J.M.; van Oers, M.M. Complete sequence of a picorna-like virus of the genus Iflavirus replicating in the mite Varroa destructor. J. Gen. Virol. 2004, 85, 3747–3755. [Google Scholar] [CrossRef]
- Ryabov, E.V.; Childers, A.K.; Lopez, D.; Grubbs, K.; Posada-Florez, F.; Weaver, D.; Girten, W.; vanEngelsdorp, D.; Chen, Y.; Evans, J.D. Dynamic evolution in the key honey bee pathogen Deformed wing virus: Novel insights into virulence and competition using reverse genetics. PLoS Biol. 2019, 17, e3000502. [Google Scholar] [CrossRef]
- Posada-Florez, F.; Childers, A.K.; Heerman, M.C.; Egekwu, N.I.; Cook, S.C.; Chen, Y.; Evans, J.D.; Ryabov, E.V. Deformed wing virus type A, a major honey bee pathogen, is vectored by the mite Varroa destructor in a non-propagative manner. Sci. Rep. 2019, 9, 12445. [Google Scholar] [CrossRef]
- Campbell, E.M.; Budge, G.E.; Watkins, M.; Bowman, A.S. Transcriptome analysis of the synganglion from the honey bee mite, Varroa destructor and RNAi knockdown of neural peptide targets. Insect Biochem. Mol. Biol. 2016, 70, 116–126. [Google Scholar] [CrossRef][Green Version]
- Mondet, F.; Kim, S.H.; de Miranda, J.R.; Beslay, D.; Le Conte, Y.; Mercer, A.R. Specific cues associated with honey bee social defence against Varroa destructor infested brood. Sci. Rep. 2016, 6, 25444. [Google Scholar] [CrossRef]
- Mondet, F.; Alaux, C.; Severac, D.; Rohmer, M.; Mercer, A.R.; Le Conte, Y. Antennae hold a key to Varroa-sensitive hygiene behaviour in honey bees. Sci. Rep. 2015, 5, 10454. [Google Scholar] [CrossRef] [PubMed]
- Natsopoulou, M.E.; McMahon, D.P.; Doublet, V.; Frey, E.; Rosenkranz, P.; Paxton, R.J. The virulent, emerging genotype B of Deformed wing virus is closely linked to overwinter honeybee worker loss. Sci. Rep. 2017, 7, 5242. [Google Scholar] [CrossRef] [PubMed]
- Mordecai, G.J.; Brettell, L.E.; Martin, S.J.; Dixon, D.; Jones, I.M.; Schroeder, D.C. Superinfection exclusion and the long-term survival of honey bees in Varroa-infested colonies. ISME J. 2016, 10, 1182–1191. [Google Scholar] [CrossRef] [PubMed]
- Dalmon, A.; Desbiez, C.; Coulon, M.; Thomasson, M.; Le Conte, Y.; Alaux, C.; Vallon, J.; Moury, B. Evidence for positive selection and recombination hotspots in Deformed wing virus (DWV). Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Ryabov, E.V.; Wood, G.R.; Fannon, J.M.; Moore, J.D.; Bull, J.C.; Chandler, D.; Mead, A.; Burroughs, N.; Evans, D.J. A virulent strain of Deformed wing virus (DWV) of honeybees (Apis mellifera) prevails after Varroa destructor-mediated, or in vitro, transmission. PLoS Pathog. 2014, 10, e1004230. [Google Scholar] [CrossRef]
- Moore, J.; Jironkin, A.; Chandler, D.; Burroughs, N.; Evans, D.J.; Ryabov, E.V. Recombinants between Deformed wing virus and Varroa destructor virus-1 may prevail in Varroa destructor-infested honeybee colonies. J. Gen. Virol. 2011, 92, 156–161. [Google Scholar] [CrossRef]
- Zioni, N.; Soroker, V.; Chejanovsky, N. Replication of Varroa destructor virus 1 (VDV-1) and a Varroa destructor virus 1-Deformed wing virus recombinant (VDV-1-DWV) in the head of the honey bee. Virology 2011, 417, 106–112. [Google Scholar] [CrossRef]
- Mordecai, G.J.; Wilfert, L.; Martin, S.J.; Jones, I.M.; Schroeder, D.C. Diversity in a honey bee pathogen: First report of a third master variant of the Deformed wing virus quasispecies. ISME J. 2015, 10, 1–10. [Google Scholar] [CrossRef]
- Kevill, J.L.; Highfield, A.; Mordecai, G.J.; Martin, S.J.; Schroeder, D.C. ABC assay: Method development and application to quantify the role of three DWV master variants in overwinter colony losses of European honey bees. Viruses 2017, 9, 1–14. [Google Scholar] [CrossRef]
- Genersch, E.; Aubert, M. Emerging and re-emerging viruses of the honey bee (Apis mellifera L.). Vet. Res. 2010, 41, 54. [Google Scholar] [CrossRef]
- Chen, Y.P.; Siede, R. Honey Bee Viruses. Adv. Virus Res. 2007, 70, 33–80. [Google Scholar] [PubMed]
- Maori, E.; Paldi, N.; Shafir, S.; Kalev, H.; Tsur, E.; Glick, E.; Sela, I. IAPV, a bee-affecting virus associated with Colony Collapse Disorder can be silenced by dsRNA ingestion. Insect Mol. Biol. 2009, 18, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Bailey, L.; Gibbs, A.J.; Woods, R.D. Two viruses from adult honey bees (Apis mellifera Linnaeus). Virology 1963, 21, 390–395. [Google Scholar] [CrossRef]
- Bailey, L.; Ball, B.V.; Woods, R.D. An Iridovirus from Bees. J. Gen. Virol. 1976, 31, 459–461. [Google Scholar] [CrossRef] [PubMed]
- Bailey, L.; Carpenter, J.M.; Woods, R.D. Properties of a filamentous virus of the honey bee (Apis mellifera). Virology 1981, 114, 1–7. [Google Scholar] [CrossRef]
- Bailey, L.; Woods, R.D. Three previously undescribed viruses from the honey bee. J. Gen. Virol. 1974, 25, 175–186. [Google Scholar] [CrossRef]
- Morse, R.A.; Flottum, K. Honeybee Pests, Predators and Diseases; A.I. Root: Medina, OH, USA, 1997. [Google Scholar]
- Bailey, L.; Gibbs, A.J.; Woods, R.D. The purification and properties of Chronic bee-paralysis virus. J. Gen. Virol. 1968, 2, 251–260. [Google Scholar] [CrossRef]
- Bailey, L. Viruses of honeybees. Bee World 1982, 63, 165–173. [Google Scholar] [CrossRef]
- Carreck, N.L.; Ball, B.V.; Martin, S.J. The epidemiology of Cloudy wing virus infections in honey bee colonies in the UK. J. Apic. Res. 2010, 49, 66–71. [Google Scholar] [CrossRef]
- Yue, C.; Schröder, M.; Gisder, S.; Genersch, E. Vertical-transmission routes for Deformed wing virus of honeybees (Apis mellifera). J. Gen. Virol. 2007, 88, 2329–2336. [Google Scholar] [CrossRef]
- Di Prisco, G.; Annoscia, D.; Margiotta, M.; Ferrara, R.; Varricchio, P.; Zanni, V.; Caprio, E.; Nazzi, F.; Pennacchio, F. A mutualistic symbiosis between a parasitic mite and a pathogenic virus undermines honey bee immunity and health. Proc. Natl. Acad. Sci. USA 2016, 113, 201523515. [Google Scholar] [CrossRef] [PubMed]
- Lanzi, G.; de Miranda, J.R.; Boniotti, M.B.; Cameron, C.E.; Lavazza, A.; Capucci, L.; Camazine, S.M.; Rossi, C. Molecular and biological characterization of Deformed wing virus of honeybees (Apis mellifera L.). Virology 2006, 80, 4998–5009. [Google Scholar] [CrossRef] [PubMed]
- Bailey, L.; Gibbs, A.J.; Woods, R.D. Sacbrood virus of the larval honey bee (Apis mellifera linnaeus). Virology 1964, 23, 425–429. [Google Scholar] [CrossRef]
- Anderson, D.L.; Giacon, H. Reduced Pollen Collection by Honey Bee (Hymenoptera: Apidae) colonies infected with Nosema apis and Sacbrood virus. J. Econ. Entomol. 1992, 85, 47–51. [Google Scholar] [CrossRef]
- Bailey, L.; Fernando, E.F.W. Effects of sacbrood virus on adult honey-bees. Ann. Appl. Biol. 1972, 72, 27–35. [Google Scholar] [CrossRef]
- Crane, E. Recent research on the world history of beekeeping. Bee World 1999, 80, 174–186. [Google Scholar] [CrossRef]
- Moritz, R.F.A.; Härtel, S.; Neumann, P. Global invasions of the western honeybee (Apis mellifera) and the consequences for biodiversity. Ecoscience 2005, 12, 289–301. [Google Scholar] [CrossRef]
- Mondet, F.; de Miranda, J.R.; Kretzschmar, A.; Le Conte, Y.; Mercer, A.R. On the front line: Quantitative virus dynamics in honeybee (Apis mellifera L.) colonies along a new expansion front of the parasite Varroa destructor. PLoS Pathog. 2014, 10, e1004323. [Google Scholar] [CrossRef]
- Ball, B.V.; Allen, M.F. The prevalence of pathogens in honey bee (Apis mellifera) colonies infested with the parasitic mite Varroa jacobsoni. Ann. Appl. Biol. 1988, 113, 237–244. [Google Scholar] [CrossRef]
- Martin, S.J.; Highfield, A.C.; Brettell, L.; Villalobos, E.M.; Budge, G.E.; Powell, M.; Nikaido, S.; Schroeder, D.C. Global Honey Bee Viral Landscape Altered by a Parasitic Mite. Science 2012, 336, 1304–1306. [Google Scholar] [CrossRef]
- Chen, Y.; Pettis, J.S.; Feldlaufer, M.F. Detection of multiple viruses in queens of the honey bee. J. Invertebr. Pathol. 2005, 90, 118–121. [Google Scholar] [CrossRef] [PubMed]
- Yue, C.; Schröder, M.; Bienefeld, K.; Genersch, E. Detection of viral sequences in semen of honeybees (Apis mellifera): Evidence for vertical transmission of viruses through drones. J. Invertebr. Pathol. 2006, 92, 105–108. [Google Scholar] [CrossRef] [PubMed]
- De Miranda, J.R.; Fries, I. Venereal and vertical transmission of deformed wing virus in honeybees (Apis mellifera L.). J. Invertebr. Pathol. 2008, 98, 184–189. [Google Scholar] [CrossRef]
- Amiri, E.; Kryger, P.; Meixner, M.D.; Strand, M.K.; Tarpy, D.R.; Rueppell, O. Quantitative patterns of vertical transmission of deformed wing virus in honey bees. PLoS ONE 2018, 13, e0195283. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Levitt, A.L.; Rajotte, E.G.; Holmes, E.C.; Ostiguy, N.; Vanengelsdorp, D.; Lipkin, W.I.; Depamphilis, C.W.; Toth, A.L.; Cox-Foster, D.L. RNA viruses in hymenopteran pollinators: Evidence of inter-taxa virus transmission via pollen and potential impact on non-Apis hymenopteran species. PLoS ONE 2010, 5, e14357. [Google Scholar] [CrossRef]
- Welch, A.; Drummond, F.; Tewari, S.; Averill, A.; Burand, J.P. Presence and prevalence of viruses in local and migratory honeybees (Apis mellifera) in Massachusetts. Appl. Environ. Microbiol. 2009, 75, 7862–7865. [Google Scholar] [CrossRef][Green Version]
- Alger, S.A.; Burnham, P.A.; Lamas, Z.S.; Brody, A.K.; Richardson, L.L. Home sick: Impacts of migratory beekeeping on honey bee (Apis mellifera) pests, pathogens, and colony size. PeerJ 2018, 6, e5812. [Google Scholar] [CrossRef]
- Oldroyd, B.P.; Wongsiri, S. Asian Honey Bees (Biology, Conservation, and Human Interactions); Harvard University Press: Cambridge, MA, USA, 2006. [Google Scholar]
- Hepburn, H.R.; Radloff, S.E. Honeybees of Asia. In Honeybees of Asia; Springer: Berlin, Germany, 2011; pp. 1–669. [Google Scholar]
- Yanez, O.; Piot, N.; Dalmon, A.; de Miranda, J.R.; Chantawannakul, P.; Panziera, D.; Amiri, E.; Smagghe, G.; Schroeder, D.C.; Chejanovsky, N. Bee viruses: Routes of infection in Hymenoptera. Front. Microbiol. 2020, (in press).
- Ray, M.A.; Lopez, D.L.; Martinez, J.F.I.; Galbraith, D.A.; Rose, R.; Van Engelsdorp, D.; Rosa, C.; Evans, J.D.; Grozinger, C.M. Distribution of recently identified bee-infecting viruses in managed honey bee (Apis mellifera) populations in the USA. Apidologie 2020, (in press).
- Le Conte, Y.; Navajas, M. Climate change: Impact on honey bee populations and diseases. Rev. Sci. Tech. Off. Int. Epiz. 2008, 27, 499–510. [Google Scholar]
- Koetz, A. Ecology, Behaviour and Control of Apis cerana with a focus on relevance to the australian incursion. Insects 2013, 4, 558–592. [Google Scholar] [CrossRef] [PubMed]
- Shybanov, S.; Kharina, A.; Stakhurska, O.; Snihur, G.; Kompanets, T. Detection of honey bee viruses on the territory of ukraine. Agrofor Int. J. 2018, 2, 140–146. [Google Scholar] [CrossRef]
- Haddad, N.J.; Noureddine, A.; Al-Shagour, B.; Loucif-Ayad, W.; El-Niweiri, M.A.A.; Anaswah, E.; Hammour, W.A.; El-Obeid, D.; Imad, A.; Shebl, M.A.; et al. Distribution and variability of deformed wing virus of honeybees (Apis mellifera) in the Middle East and North Africa. Insect Sci. 2017, 24, 103–113. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.J.; Brettell, L.E. Deformed wing virus in honeybees and other insects. Annu. Rev. Virol. 2019, 6, 49–69. [Google Scholar] [CrossRef]
- Roberts, J.M.K.; Anderson, D.L.; Durr, P.A. Absence of Deformed wing virus and Varroa destructor in Australia provides unique perspectives on honeybee viral landscapes and colony losses. Sci. Rep. 2017, 7, 6925. [Google Scholar] [CrossRef]
- Tozkar, C.Ö.; Kence, M.; Kence, A.; Huang, Q.; Evans, J.D. Metatranscriptomic analyses of honey bee colonies. Front. Genet. 2015, 6, 100. [Google Scholar] [CrossRef]
- Kajobe, R.; Marris, G.; Budge, G.; Laurenson, L.; Cordoni, G.; Jones, B.; Wilkins, S.; Cuthbertson, A.G.S.; Brown, M.A. First molecular detection of a viral pathogen in Ugandan honey bees. J. Invertebr. Pathol. 2010, 104, 153–156. [Google Scholar] [CrossRef]
- Abou Kubaa, R.; Molinatto, G.; Khaled, B.; Daher-Hjaij, N.; Heinoun, K.; Saponari, M. First detection of black queen cell virus, Varroa destructor macula-like virus, Apis mellifera filamentous virus and Nosema ceranae in Syrian honey bees Apis mellifera syriaca. Bull. Insectol. 2018, 71, 217–224. [Google Scholar]
- De Miranda, J.R.; Cordoni, G.; Budge, G. The Acute bee paralysis virus–Kashmir bee virus–Israeli acute paralysis virus complex. J. Invertebr. Pathol. 2010, 103, S30–S47. [Google Scholar] [CrossRef]
- Tsevegmid, K.; Neumann, P.; Yañez, O. The Honey bee pathosphere of Mongolia: European viruses in central Asia. PLoS ONE 2016, 11, e0151164. [Google Scholar] [CrossRef]
- Tentcheva, D.; Gauthier, L.; Zappulla, N.; Dainat, B.; Cousserans, F.; Colin, M.E.; Bergoin, M. Prevalence and seasonal variations of six bee viruses in Apis mellifera L. and Varroa destructor mite populations in France. Appl. Environ. Microbiol. 2004, 70, 7185–7191. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.; Rivkin, H.; Slabezki, Y.; Chejanovsky, N. Dynamics of the presence of Israeli acute paralysis virus in honey bee colonies with Colony Collapse Disorder. Viruses 2014, 6, 2012–2027. [Google Scholar] [CrossRef] [PubMed]
- Blažytė-Čereškienė, L.; Skrodenytė-Arbačiauskienė, V.; Radžiutė, S.; Čepulytė-Rakauskienė, R.; Apšegaitė, V.; Būda, V. A three-year survey of honey bee viruses in Lithuania. J. Apic. Res. 2016, 55, 176–184. [Google Scholar] [CrossRef]
- Porrini, C.; Mutinelli, F.; Bortolotti, L.; Granato, A.; Laurenson, L.; Roberts, K.; Gallina, A.; Silvester, N.; Medrzycki, P.; Renzi, T.; et al. The status of honey bee health in Italy: Results from the nationwide bee monitoring network. PLoS ONE 2016, 11, e0155411. [Google Scholar] [CrossRef]
- Traynor, K.S.; Rennich, K.; Forsgren, E.; Rose, R.; Pettis, J.; Kunkel, G.; Madella, S.; Evans, J.; Lopez, D.; van Engelsdorp, D. Multiyear survey targeting disease incidence in US honey bees. Apidologie 2016, 47, 325–347. [Google Scholar] [CrossRef]
- Grassly, N.C.; Fraser, C. Seasonal infectious disease epidemiology. Proc. R. Soc. B Biol. Sci. 2006, 273, 2541–2550. [Google Scholar] [CrossRef]
- Alaux, C.; Ducloz, F.; Crauser, D.; Le Conte, Y. Diet effects on honeybee immunocompetence. Biol. Lett. 2010, 6, 562–565. [Google Scholar] [CrossRef]
- Antúnez, K.; Anido, M.; Branchiccela, B.; Harriet, J.; Campa, J.; Invernizzi, C.; Santos, E.; Higes, M.; Martín-Hernández, R.; Zunino, P. Seasonal variation of honeybee pathogens and its association with pollen diversity in Uruguay. Microb. Ecol. 2015, 70, 522–533. [Google Scholar] [CrossRef]
- Branchiccela, B.; Castelli, L.; Corona, M.; Díaz-Cetti, S.; Invernizzi, C.; Martínez de la Escalera, G.; Mendoza, Y.; Santos, E.; Silva, C.; Zunino, P.; et al. Impact of nutritional stress on the honeybee colony health. Sci. Rep. 2019, 9, 10156. [Google Scholar] [CrossRef]
- Smart, M.; Pettis, J.; Rice, N.; Browning, Z.; Spivak, M. Linking measures of colony and individual honey bee health to survival among apiaries exposed to varying agricultural land use. PLoS ONE 2016, 11, e0152685. [Google Scholar] [CrossRef]
- Manley, R.; Boots, M.; Wilfert, L. Emerging viral disease risk to pollinating insects: Ecological, evolutionary and anthropogenic factors. J. Appl. Ecol. 2015, 52, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Ball, B.V. Varroa jacobsoni as a virus vector. In Present Status of Varroatosis in Europe and Progress in the Varroa Mite Control; The Stationery Commission of the European Communities: Luxembourg, 1989; pp. 241–244. [Google Scholar]
- Carreck, N.L.; Ball, B.V.; Wilson, J.K. Virus succession in honeybee colonies infested with Varroa destructor. Apiacta 2002, 37, 33–38. [Google Scholar]
- Nordström, S. Distribution of Deformed wing virus within honey bee (Apis mellifera) brood cells infested with the ectoparasitic mite Varroa destructor. Exp. Appl. Acarol. 2003, 29, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Beaurepaire, A.L.; Krieger, K.J.; Moritz, R.F.A. Seasonal cycle of inbreeding and recombination of the parasitic mite Varroa destructor in honeybee colonies and its implications for the selection of acaricide resistance. Infect. Genet. Evol. 2017, 50, 49–54. [Google Scholar] [CrossRef]
- Diao, Q.; Yang, D.; Zhao, H.; Deng, S.; Wang, X.; Hou, C.; Wilfert, L. Prevalence and population genetics of the emerging honey bee pathogen DWV in Chinese apiculture. Sci. Rep. 2019, 9, 12042. [Google Scholar] [CrossRef]
- Martin, S.J. The role of Varroa and viral pathogens in the collapse of honeybee colonies: A modelling approach. J. Appl. Ecol. 2001, 38, 1082–1093. [Google Scholar] [CrossRef]
- Dainat, B.; Evans, J.D.; Chen, Y.P.; Gauthier, L.; Neumanna, P. Dead or alive: Deformed wing virus and Varroa destructor reduce the life span of winter honeybees. Appl. Environ. Microbiol. 2012, 78, 981–987. [Google Scholar] [CrossRef]
- Francis, R.M.; Nielsen, S.L.; Kryger, P.; Martin, B.S. Varroa-virus interaction in collapsing honey bee colonies. PLoS ONE 2013, 8, e57540. [Google Scholar] [CrossRef]
- Thaduri, S.; Locke, B.; Granberg, F.; de Miranda, J.R. Temporal changes in the viromes of Swedish Varroa-resistant and Varroa-susceptible honeybee populations. PLoS ONE 2018, 13, e0206938. [Google Scholar] [CrossRef]
- Locke, B. Natural Varroa mite-surviving Apis mellifera honeybee populations. Apidologie 2016, 47, 1–16. [Google Scholar] [CrossRef]
- Martin, S.J.; Medina, L.M. Africanized honeybees have unique tolerance to Varroa mites. Trends Parasitol. 2004, 20, 112–114. [Google Scholar] [CrossRef] [PubMed]
- Pirk, C.W.; Strauss, U.; Yusuf, A.A.; Demares, F. Honeybee health in Africa—A review. Apidologie 2015, 47, 276–300. [Google Scholar] [CrossRef]
- Rosenkranz, P. Honey bee (Apis mellifera L.) tolerance to Varroa jacobsoni Oud. in South America. Apidologie 1999, 30, 159–172. [Google Scholar] [CrossRef]
- Strauss, U.; Human, H.; Gauthier, L.; Crewe, R.M.; Dietemann, V.; Pirk, C.W.W. Seasonal prevalence of pathogens and parasites in the savannah honeybee (Apis mellifera scutellata). J. Invertebr. Pathol. 2013, 114, 45–52. [Google Scholar] [CrossRef]
- Coulon, M.; Schurr, F.; Martel, A.-C.; Cougoule, N.; Bégaud, A.; Mangoni, P.; Dalmon, A.; Alaux, C.; Le Conte, Y.; Thiéry, R.; et al. Metabolisation of thiamethoxam (a neonicotinoid pesticide) and interaction with the Chronic bee paralysis virus in honeybees. Pestic. Biochem. Physiol. 2018, 144, 10–18. [Google Scholar] [CrossRef]
- Sumpter, D.J.T.; Martin, S.J. The dynamics of virus epidemics in Varroa-infested honey bee colonies. J. Anim. Ecol. 2004, 73, 51–63. [Google Scholar] [CrossRef]
- Remnant, E.J.; Mather, N.; Gillard, T.L.; Yagound, B.; Beekman, M. Direct transmission by injection affects competition among RNA viruses in honeybees. Proc. R. Soc. B Biol. Sci. 2019, 286, 20182452. [Google Scholar] [CrossRef]
- Fries, I.; Camazine, S. Implications of horizontal and vertical pathogen transmission for honey bee epidemiology. Apidologie 2001, 32, 199–214. [Google Scholar] [CrossRef]
- Fürst, M.A.; McMahon, D.P.; Osborne, J.L.; Paxton, R.J.; Brown, M.J.F. Disease associations between honeybees and bumblebees as a threat to wild pollinators. Nature 2014, 506, 364–366. [Google Scholar] [CrossRef]
- Klein, A.-M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B Biol. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef]
- Aizen, M.A.; Harder, L.D. The global stock of domesticated honey bees is growing slower than agricultural demand for pollination. Curr. Biol. 2009, 19, 915–918. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.D.; Aronstein, K.A.; Chen, Y.P.; Hetru, C.; Imler, J.-L.; Jiang, H.; Kanost, M.; Thompson, G.J.; Zou, Z.; Hultmark, D. Immune pathways and defence mechanisms in honey bees Apis mellifera. Insect Mol. Biol. 2006, 15, 645–656. [Google Scholar] [CrossRef] [PubMed]
- Doublet, V.; Poeschl, Y.; Gogol-Döring, A.; Alaux, C.; Annoscia, D.; Aurori, C.; Barribeau, S.M.; Bedoya-Reina, O.C. Unity in defence: Honeybee workers exhibit conserved molecular responses to diverse pathogens. BMC Genom. 2017, 18, 207. [Google Scholar] [CrossRef] [PubMed]
- Engel, P.; Kwong, W.K.; McFrederick, Q.; Anderson, K.E.; Barribeau, S.M.; Chandler, J.A.; Cornman, R.S.; Dainat, J.; de Miranda, J.R.; Doublet, V.; et al. The bee microbiome: Impact on bee health and model for evolution and ecology of host-microbe interactions. MBio 2016, 7, e02164-15. [Google Scholar] [CrossRef]
- White, G.F. Sacbrood, a Disease of Bees; US Department of Agriculture, Bureau of Entomology: Washington, DC, USA, 1913.
- Loope, K.J.; Baty, J.W.; Lester, P.J.; Wilson Rankin, E.E. Pathogen shifts in a honeybee predator following the arrival of the Varroa mite. Proc. R. Soc. B Biol. Sci. 2019, 286, 20182499. [Google Scholar] [CrossRef]
- Mazzei, M.; Cilia, G.; Forzan, M.; Lavazza, A.; Mutinelli, F.; Felicioli, A. Detection of replicative Kashmir bee virus and Black queen cell virus in Asian hornet Vespa velutina (Lepelieter 1836) in Italy. Sci. Rep. 2019, 9, 10091. [Google Scholar] [CrossRef]
- Schläppi, D.; Lattrell, P.; Yañez, O.; Chejanovsky, N.; Neumann, P. Foodborne transmission of Deformed wing virus to ants (Myrmica rubra). Insects 2019, 10, 394. [Google Scholar] [CrossRef]
- Fletcher, D.J.C. The African Bee, Apis Mellifera Adansonii, in Africa. Annu. Rev. Entomol. 1978, 23, 151–171. [Google Scholar] [CrossRef]
- Genersch, E.; Yue, C.; Fries, I.; de Miranda, J.R. Detection of Deformed wing virus, a honey bee viral pathogen, in bumble bees (Bombus terrestris and Bombus pascuorum) with wing deformities. J. Invertebr. Pathol. 2006, 91, 61–63. [Google Scholar] [CrossRef]
- Dalmon, A.; Gayral, P.; Decante, D.; Klopp, C.; Bigot, D.; Thomasson, M.; Herniou, E.A.; Alaux, C.; Le Conte, Y. Viruses in the invasive hornet Vespa velutina. Viruses 2019, 11, 1041. [Google Scholar] [CrossRef]
- Meeus, I.; de Miranda, J.R.; de Graaf, D.C.; Wäckers, F.; Smagghe, G. Effect of oral infection with Kashmir bee virus and Israeli acute paralysis virus on bumblebee (Bombus terrestris) reproductive success. J. Invertebr. Pathol. 2014, 121, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Gisder, S.; Genersch, E. Viruses of commercialized insect pollinators. J. Invertebr. Pathol. 2017, 147, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Swevers, L.; Rombouts, C.; Meeus, I.; Meulebroek, L.; Vanhaecke, L.; Smagghe, G. A metabolomics approach to unravel Cricket paralysis virus infection in silkworm Bm5 cells. Viruses 2019, 11, 861. [Google Scholar] [CrossRef] [PubMed]
- De Guzman, L.I.; Williams, G.R.; Khongphinitbunjong, K.; Chantawannakul, P. Ecology, life history, and management of Tropilaelaps Mites. J. Econ. Entomol. 2017, 110, 319–332. [Google Scholar] [CrossRef] [PubMed]
- Phokasem, P.; de Guzman, L.I.; Khongphinitbunjong, K.; Frake, A.M.; Chantawannakul, P. Feeding by Tropilaelaps mercedesae on pre- and post-capped brood increases damage to Apis mellifera colonies. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
| Genome | Order | Family | Virus | Major Strains | Accession Number | TRANSMISSION | SEASON | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ORAL-FECAL | VARROA | CONTACT | VERTICAL | ENVIRONMENT | SPRING | SUMMER | AUTUMN | WINTER | |||||||
| ssRNA(+) | Picornavirales | Dicistroviridae | Acute bee paralysis virus | ABPV | AF150629 | + | + | - | + | + | + | +++ | ++ | + | |
| KBV | AY275710 | + | + | - | + | + | + | ++ | +++ | ? | |||||
| IAPV | EF219380 | + | + | - | + | + | + | ++ | ++ | ? | |||||
| Aphid lethal paralysis virus | ALPV | AF536531 | ? | ? | ? | ? | + | - | +++ | - | ? | ||||
| Apis dicistrovirus | ADV | KY354239 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Big Sioux river virus | BSRV | KY826434 | ? | ? | ? | ? | + | - | +++ | ++ | ? | ||||
| Black queen cell virus | BQCV | AF183905 | + | ~ | - | + | + | + | +++ | + | + | ||||
| Bundaberg bee virus 1 c | QLD-6 | MG995706 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Bundaberg bee virus 2 * c | QLD-4 | MG995700 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Empeyrat virus c | NT-5 | KU754505 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| QLD-7 | MG995702 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||||
| Hobart bee virus 1 * c | TAS-7 | MG995722 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Perth bee virus 1 * c | WA2-13 | MG995730 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Perth bee virus 2 * c | WA1-14 | MG995726 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Renmark bee virus 1 * c | SA-7 | MG995710 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Rhopalosiphum padi virus c | RhPV | AF022937 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Robinvale bee virus 1 * c | VN1-10 | MG995714 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Robinvale bee virus 2 * c | VN1-57 | MG995719 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Robinvale bee virus 3 * c | VN1-50 | MG995718 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Iflaviridae | Bundaberg bee virus 4 c | QLD-11 | MG995705 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| Bundaberg bee virus 5 c | QLD-13 | MG995706 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Bundaberg bee virus 6 c | QLD-14 | MG995707 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| NT-12 | MG995697 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||||
| Darwin bee virus 2 * c | NT-6 | MG995694 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| NT-17 | MG995699 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||||
| Deformed wing virus | DWV-A | AY292384 | + | + | - | + | + | + | ++ | +++ | +++ | ||||
| DWV-B | AY251269 | + | + | - | + | + | + | + | ++ | + | |||||
| DWV-C | ERS657948 | ? | ? | ? | ? | ? | + | + | +++ | +++ | |||||
| (Egypt bee virus*) | DWV-D | n.a. | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Moku virus | MV | KU645789 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Perth bee virus 3 * c | WA2-20 | MG995731 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| VN2-2 | MG995723 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||||
| VN2-6 | MG995724 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||||
| Renmark bee virus 2 * c | SA-5 | MG995709 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Robinvale bee virus 4 c | VN3-43 | MG995721 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Sacbrood virus | SBV | AF092924 | + | - | - | ? | + | +++ | ++ | + | ? | ||||
| TSBV | KM884995 | + | ? | ? | ? | ? | ? | ? | ? | ? | |||||
| Slow bee paralysis virus | SBPV | EU035616 | + | + | - | ? | + | - | - | - | ? | ||||
| Varroa destructor virus-2 a | VDV-2 | KX578271 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Nodaviridae | Apis Nora virus | ANV | KY354240 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| Nodamura-like virus * | ? | PEHZ00000000 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Secoviridae | Seco-like virus * | ? | PEHZ00000000 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| Tobacco ringspot virus | TRSV | U50869 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| ? | Arkansas bee virus ** | ArkBV | n.a. | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| Berkeley bee virus ** | BerkBPV | n.a. | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| ? | Bundaberg bee virus 7 * c | QLD-8 | MG995703 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| Darwin bee virus 5 c | NT1 | MG995692 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Perth bee virus 6 * c | WA2-62 | MG995732 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Perth bee virus 7 * c | WA1-16 | MG995727 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Perth bee virus 8 * c | WA1-18 | MG995728 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Perth bee virus 9 * c | WA1-9 | MG995725 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Renmark bee virus 4 c | SA-10 | MG995708 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Renmark bee virus 5 * c | SA-8 | MG995711 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Robinvale bee virus 7 * c | VN1-35 | MG995717 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Robinvale bee virus 9 * c | VN3-31 | MG995720 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| ? | Darwin bee virus 6 c | NT-8 | MG995696 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| Perth bee virus 4 * c | WA2-63 | MG995733 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Perth bee virus 5 * c | WA1-24 | MG995729 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Renmark bee virus 3 c | SA4 | MG995708 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Robinvale bee virus 5 * c | VN1-15 | MG995715 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Robinvale bee virus 8 c | VN1-22 | MG995716 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| ? | Bundaberg bee virus 8 * c | QLD-9 | MG995704 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| Darwin bee virus 7 * c | NT-15 | MG995698 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| ? | Darwin bee virus 8 * c | NT-7 | MG995695 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| Robinvale bee virus 6 * c | VN1-8 | MG995713 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| Tymovirales | Tymoviridae | Bee Macula-like virus | BeeMLV | KT162925 | ? | + | ? | ? | + | + | ++ | +++ | ? | ||
| Varroa Tymo-like virus * a | VTLV | KT162926 | ? | + | ? | ? | ? | ? | ? | ? | ? | ||||
| ? | ? | Cloudy wing virus ** | CWV | n.a. | ? | - | ~ | ? | ? | + | + | + | ? | ||
| ? | ? | Chronic bee paralysis virus | CBPV | EU122230 | + | - | + | ? | + | ++ | ++ | + | ? | ||
| ? | Bee virus ** | BVX | n.a. | + | ? | ? | ? | ? | +++ | + | + | ? | |||
| BVY | n.a. | + | ? | ? | ? | ? | + | +++ | + | ? | |||||
| Lake Sinai virus | LSV-1 | HQ871931 | ? | ? | ? | ? | + | ++ | +++ | ++ | ? | ||||
| LSV-2 | HQ888865 | ? | ? | ? | ? | + | +++ | + | + | ? | |||||
| LSV-3 | MH267700 | ? | ? | ? | ? | + | + | + | + | ? | |||||
| LSV-4 | KM886903 | ? | ? | ? | ? | + | + | + | + | ? | |||||
| ? | ? | Varroa destructor virus-3 a | VDV-3 | KX578272 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||
| ? | Varroa destructor virus-4 a | VDV-4 | MK032464 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| ssRNA(-) | Articulavirales | Orthomyxoviridae | Varroa orthomyxovirus-1 a | VOV-1 | MK032465 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |
| Bunyavirales | Arenaviridae | Apis bunyavirus-1 * | ABV-1 | KY354236 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||
| Phasmaviridae | Apis bunyavirus-2 * | ABV-2 | KY354237 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |||
| Mononegavirales | Rhabdoviridae | Apis rhabdovirus-1 | ARV-1 | MH267691 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||
| Apis rhabdovirus-2 | ARV-2 | KY354234 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| dsRNA | ? | Partitiviridae | Partiti-like virus * | ? | PEHZ00000000 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |
| ssDNA | ? | Circoviridae | Circo-like virus-1 * b | ? | PEHZ00000000 | ? | ? | ? | ? | ? | ? | ? | ? | ? | |
| Circo-like virus-2 * b | ? | PEHZ00000000 | ? | ? | ? | ? | ? | ? | ? | ? | ? | ||||
| dsDNA | Megavirales | Baculoviridae | Apis mellifera filamentous virus b | AmFV | MH243376 | + | ? | ? | ? | ? | +++ | + | + | ? | |
| Iridoviridae | Apis iridescent virus * b | AIV | AF042340 | ? | ? | ? | ? | ? | + | ++ | + | ? | |||
| Virus | Tropism | Symptoms | Refs |
|---|---|---|---|
| Acute bee paralysis virus complex | Nervous system, cytoplasm of fat body cells, brain and hypopharyngeal glands | Trembling, inability to fly, gradual darkening and loss of hair from the thorax and abdomen, crawling on the ground and upward on grass, rapid death for highly infected bees | [84,85,86] |
| Apis iridovirus | NA | Iridescence of most internal organs | [87] |
| Apis mellifera filamentous virus | NA | Milky-white hemolymph | [88] |
| Bee virus X | NA | Shortened lifespan of adult bees | [89] |
| Bee virus Y | NA | Shortened lifespan of adult bees | [37] |
| Black queen cell virus | Gut tissue | Yellowish queen larvae with sac-appearance that resembles SBV and with time evolves to dark brown, infected pupae turn brown and die, dark brown to black colored walls in queen cells, significantly shortened life span in adult bees | [36,84,90] |
| Chronic bee paralysis virus | Nervous system, alimentary tract, mandibular and hypopharyngeal glands | Syndrome 1: trembling of the wings and bodies, bloated abdomen, inability to fly, crawling on the ground and upward on grass, gather in groups in the warmest areas of the nest, death within few days | [84,91] |
| Syndrome 2 (’black robbers’): hairless (thus appearing smaller), darker, greasy in appearance, shiny, suffer nibbling attacks by the healthy bees, death within few days | |||
| Cloudy wing virus | Tracheal tissue and thoracic muscles | Opaque wings, shortened lifespan of adult bees | [92,93] |
| Deformed wing virus | Whole body, including the queen ovaries, queen fat body, spermatheca, and drone seminal vesicles, tissues of wings, head, thorax, legs, hemolymph and gut | Crumpled or aborted wings, shortened abdomens, paralysis, severely shortened adult life span for emerging worker and drone bees, modified responsiveness to sucrose, impaired learning, impaired foraging behavior | [59,64,94,95,96] |
| Invertebrate iridescent virus Type 6 | NA | Flightless clustering bees | [87] |
| Sacbrood virus | Hypopharyngeal glands of worker bees, cytoplasm of fat, muscle and tracheal-end cells of larvae | Pupation failure, ’sac’ phenotype: swollen larvae filled with ecdysial fluid full of viral particles, precocious foraging, reduction of adult life span and metabolic activities, impaired foraging activity | [97,98,99] |
| Slow bee paralysis virus | Nervous system | Paralysis of the two anterior legs a day or two before death | [89] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beaurepaire, A.; Piot, N.; Doublet, V.; Antunez, K.; Campbell, E.; Chantawannakul, P.; Chejanovsky, N.; Gajda, A.; Heerman, M.; Panziera, D.; et al. Diversity and Global Distribution of Viruses of the Western Honey Bee, Apis mellifera. Insects 2020, 11, 239. https://doi.org/10.3390/insects11040239
Beaurepaire A, Piot N, Doublet V, Antunez K, Campbell E, Chantawannakul P, Chejanovsky N, Gajda A, Heerman M, Panziera D, et al. Diversity and Global Distribution of Viruses of the Western Honey Bee, Apis mellifera. Insects. 2020; 11(4):239. https://doi.org/10.3390/insects11040239
Chicago/Turabian StyleBeaurepaire, Alexis, Niels Piot, Vincent Doublet, Karina Antunez, Ewan Campbell, Panuwan Chantawannakul, Nor Chejanovsky, Anna Gajda, Matthew Heerman, Delphine Panziera, and et al. 2020. "Diversity and Global Distribution of Viruses of the Western Honey Bee, Apis mellifera" Insects 11, no. 4: 239. https://doi.org/10.3390/insects11040239
APA StyleBeaurepaire, A., Piot, N., Doublet, V., Antunez, K., Campbell, E., Chantawannakul, P., Chejanovsky, N., Gajda, A., Heerman, M., Panziera, D., Smagghe, G., Yañez, O., de Miranda, J. R., & Dalmon, A. (2020). Diversity and Global Distribution of Viruses of the Western Honey Bee, Apis mellifera. Insects, 11(4), 239. https://doi.org/10.3390/insects11040239











