Pollutants and Their Interaction with Diseases of Social Hymenoptera
Abstract
1. Introduction
2. Major Classes of Pollutants Threatening Social Insects
3. Pathogens of Social Insects
4. Pollutants and Disease Susceptibility of Social Insects—Mechanisms of Interaction
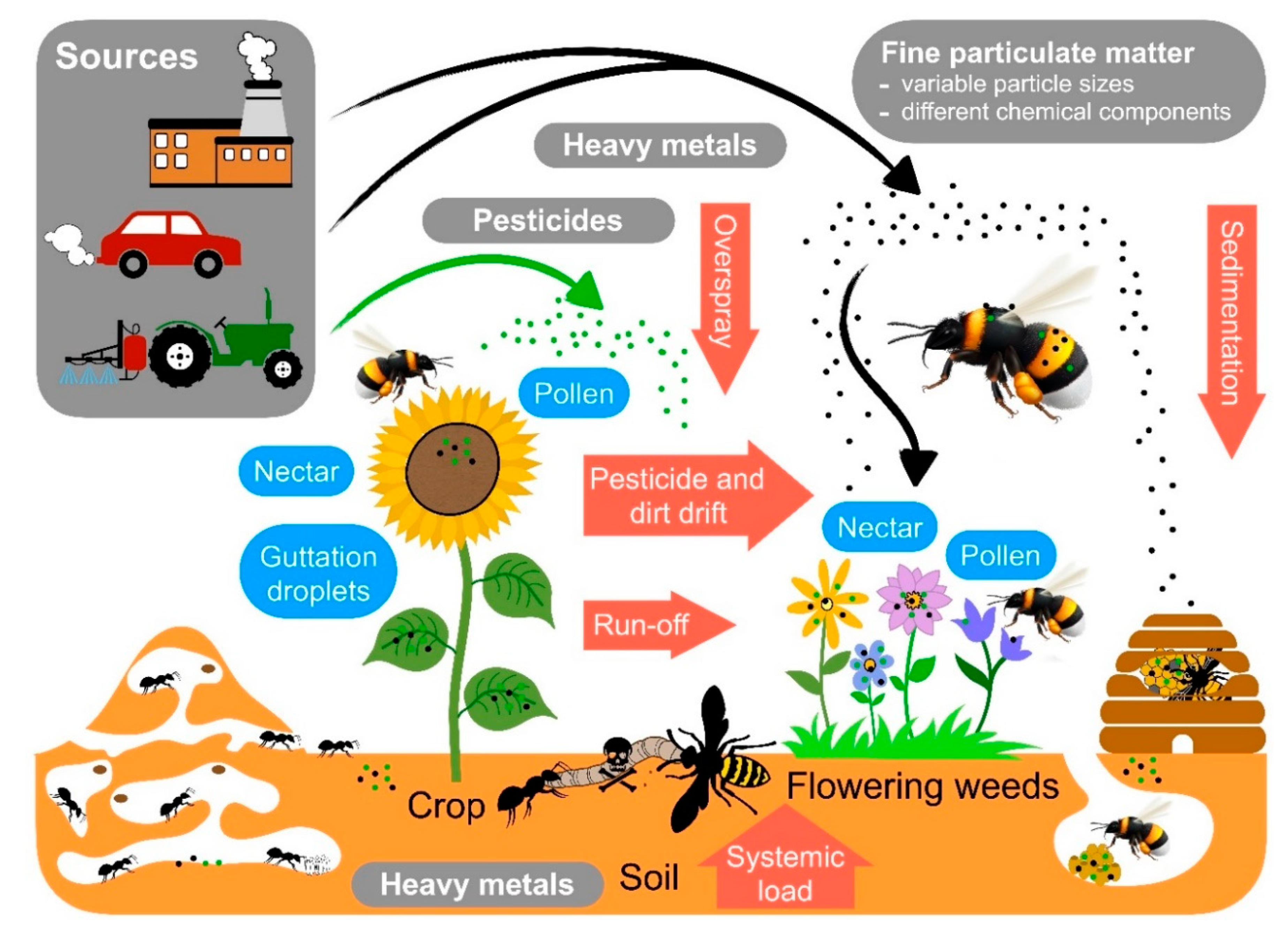
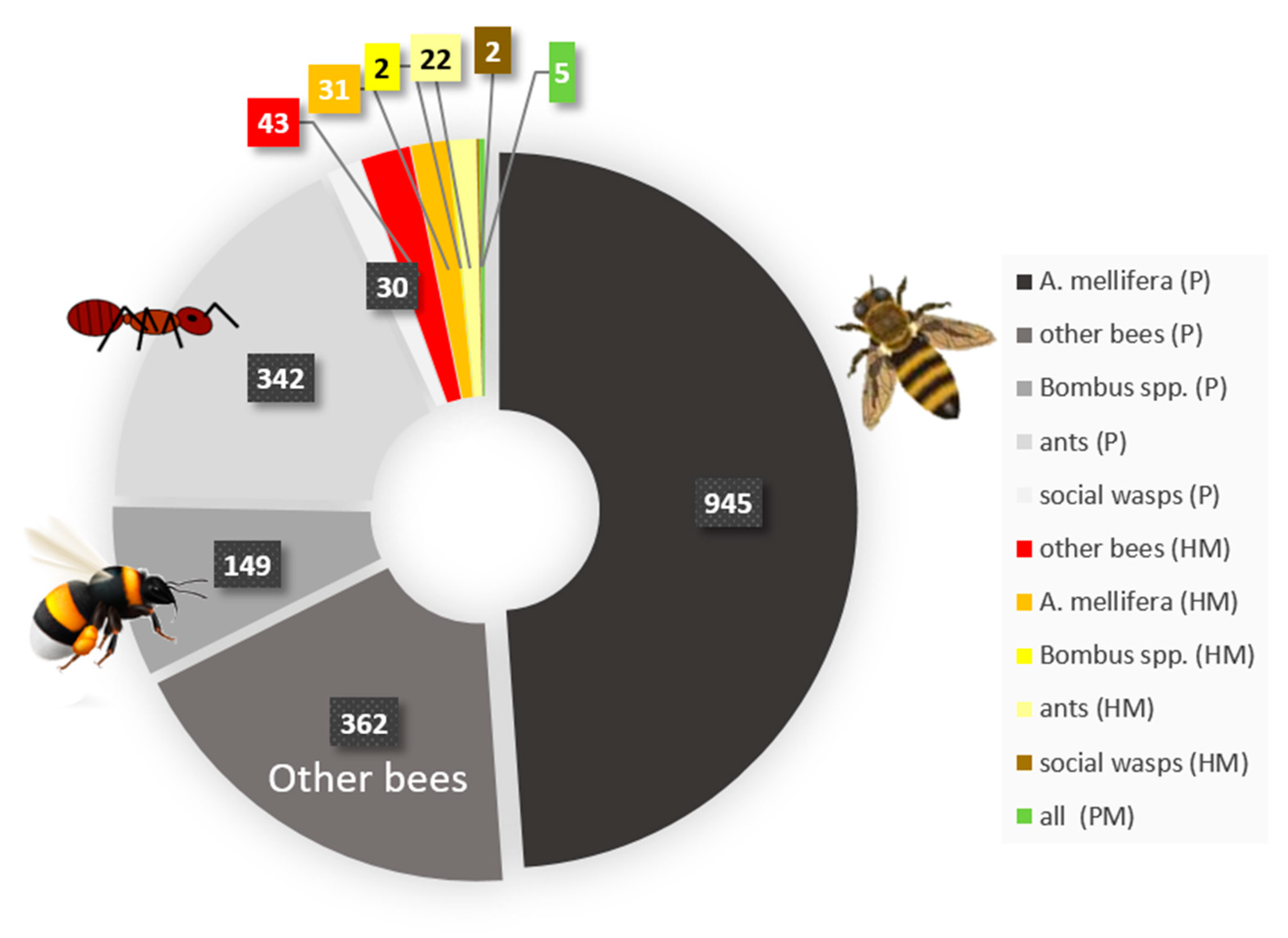
5. Pollutants Commonly Encountered by Social Insects and Their Interaction with Disease Susceptibility
6. Pollutants: Pesticides
7. Pollutants: Heavy Metals
8. Airborne Fine Particulate Matter
9. Outlook and Knowledge Gaps
Funding
Conflicts of Interest
References
- Sanchez-Bayo, F.; Wyckhuys, K.A.G. Worldwide decline of the entomofauna: A review of its drivers. Biol. Conserv. 2019, 232, 8–27. [Google Scholar] [CrossRef]
- Hallmann, C.A.; Sorg, M.; Jongejans, E.; Siepel, H.; Hofland, N.; Schwan, H.; Stenmans, W.; Muller, A.; Sumser, H.; Horren, T.; et al. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 2017, 12, e0185809. [Google Scholar] [CrossRef] [PubMed]
- Wagner, D.L. Insect declines in the anthropocene. Annu. Rev. Entomol. 2020, 65, 457–480. [Google Scholar] [CrossRef] [PubMed]
- Collison, E.; Hird, H.; Cresswell, J.; Tyler, C. Interactive effects of pesticide exposure and pathogen infection on bee health—A critical analysis. Biol. Rev. 2016, 91, 1006–1019. [Google Scholar] [CrossRef] [PubMed]
- Goulson, D. An overview of the environmental risks posed by neonicotinoid insecticides. J. Appl. Ecol. 2013, 50, 977–987. [Google Scholar] [CrossRef]
- Goulson, D.; Nicholls, E.; Botias, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 2015, 347, 1255957. [Google Scholar] [CrossRef]
- Sanchez-Bayo, F.; Goulson, D.; Pennacchio, F.; Nazzi, F.; Goka, K.; Desneux, N. Are bee diseases linked to pesticides?—A brief review. Environ. Int. 2016, 89–90, 7–11. [Google Scholar] [CrossRef]
- Aufauvre, J.; Biron, D.G.; Vidau, C.; Fontbonne, R.; Roudel, M.; Diogon, M.; Vigues, B.; Belzunces, L.P.; Delbac, F.; Blot, N. Parasite-insecticide interactions: A case study of Nosema ceranae and fipronil synergy on honeybee. Sci. Rep. 2012, 2, 326. [Google Scholar] [CrossRef]
- Czerwinski, M.A.; Sadd, B. Detrimental interactions of neonicotinoid pesticide exposure and bumblebee immunity. J. Exp. Zool. Part A-Ecol. Integr. Physiol. 2017, 327, 273–283. [Google Scholar] [CrossRef]
- Di Prisco, G.; Cavaliere, V.; Annoscia, D.; Varricchio, P.; Caprio, E.; Nazzi, F.; Gargiulo, G.; Pennacchio, F. Neonicotinoid clothianidin adversely affects insect immunity and promotes replication of a viral pathogen in honey bees. Proc. Natl. Acad. Sci. USA 2013, 110, 18466–18471. [Google Scholar] [CrossRef]
- Fauser-Misslin, A.; Sadd, B.; Neumann, P.; Sandrock, C. Influence of combined pesticide and parasite exposure on bumblebee colony traits in the laboratory. J. Appl. Ecol. 2014, 51, 450–459. [Google Scholar] [CrossRef]
- Hernandez Lopez, J.; Krainer, S.; Engert, A.; Schuehly, W.; Riessberger-Gallé, U.; Crailsheim, K. Sublethal pesticide doses negatively affect survival and the cellular responses in American foulbrood-infected honeybee larvae. Sci. Rep. 2017, 7, 40853. [Google Scholar] [CrossRef] [PubMed]
- O’Neal, S.T.; Anderson, T.D.; Wu-Smart, J.Y. Interactions between pesticides and pathogen susceptibility in honey bees. Curr. Opin. Insect Sci. 2018, 26, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Branchiccela, B.; Castelli, L.; Corona, M.; Diaz-Cetti, S.; Invernizzi, C.; de la Escalera, G.M.; Mendoza, Y.; Santos, E.; Silva, C.; Zunino, P.; et al. Impact of nutritional stress on the honeybee colony health. Sci. Rep. 2019, 9, 10156. [Google Scholar] [CrossRef]
- Di Pasquale, G.; Alaux, C.; Le Conte, Y.; Odoux, J.F.; Pioz, M.; Vaissiere, B.E.; Belzunces, L.P.; Decourtye, A. Variations in the availability of pollen resources affect honey bee health. PLoS ONE 2016, 11, e0162818. [Google Scholar] [CrossRef]
- Di Pasquale, G.; Salignon, M.; Le Conte, Y.; Belzunces, L.P.; Decourtye, A.; Kretzschmar, A.; Suchail, S.; Brunet, J.L.; Alaux, C. Influence of pollen nutrition on honey bee health: Do pollen quality and diversity matter? PLoS ONE 2013, 8, e72016. [Google Scholar] [CrossRef]
- Kaluza, B.F.; Wallace, H.M.; Heard, T.A.; Minden, V.; Klein, A.; Leonhardt, S.D. Social bees are fitter in more biodiverse environments. Sci. Rep. 2018, 8, 12353. [Google Scholar] [CrossRef]
- Gill, R.J.; Raine, N.E. Chronic impairment of bumblebee natural foraging behaviour induced by sublethal pesticide exposure. Funct. Ecol. 2014, 28, 1459–1471. [Google Scholar] [CrossRef]
- Arce, A.N.; David, T.I.; Randall, E.L.; Rodrigues, A.R.; Colgan, T.J.; Wurm, Y.; Gill, R.J. Impact of controlled neonicotinoid exposure on bumblebees in a realistic field setting. J. Appl. Ecol. 2017, 54, 1199–1208. [Google Scholar] [CrossRef]
- Colin, T.; Meikle, W.G.; Wu, X.B.; Barron, A.B. Traces of a neonicotinoid induce precocious foraging and reduce foraging performance in honey bees. Environ. Sci. Technol. 2019, 53, 8252–8261. [Google Scholar] [CrossRef]
- Gill, R.J.; Ramos-Rodriguez, O.; Raine, N.E. Combined pesticide exposure severely affects individual- and colony-level traits in bees. Nature 2012, 491, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Lunardi, S.J.; Zaluski, R.; Orsi, R.O. Evaluation of motor changes and toxicity of insecticides Fipronil and Imidacloprid in Africanized honey bees (Hymenoptera: Apidae). Sociobiology 2017, 64, 50–56. [Google Scholar] [CrossRef]
- Sivakoff, F.S.; Gardiner, M.M. Soil lead contamination decreases bee visit duration at sunflowers. Urban Ecosyst. 2017, 20, 1221–1228. [Google Scholar] [CrossRef]
- Siviter, H.; Koricheva, J.; Brown, M.J.F.; Leadbeater, E. Quantifying the impact of pesticides on learning and memory in bees. J. Appl. Ecol. 2018, 55, 2812–2821. [Google Scholar] [CrossRef]
- Stanley, D.A.; Russel, A.L.; Morrison, S.J.; Rogers, C.; Raine, N.E. Investigating the impacts of field-realistic exposure to a neonicotinoid pesticide on bumblebee foraging, homing ability and colony growth. J. Appl. Ecol. 2016, 53, 1440–1449. [Google Scholar] [CrossRef]
- Goulson, D.; Nicholls, E. The canary in the coalmine; bee declines as an indicator of environmental health. Sci. Prog. 2016, 99, 312–326. [Google Scholar] [CrossRef]
- Kennedy, C.M.; Lonsdorf, E.; Neel, M.C.; Williams, N.M.; Ricketts, T.H.; Winfree, R.; Bommarco, R.; Brittain, C.; Burley, A.L.; Cariveau, D.; et al. A global quantitative synthesis of local and landscape effects on wild bee pollinators in agroecosystems. Ecol. Lett. 2013, 16, 584–599. [Google Scholar] [CrossRef]
- Wilson, E.O. The Insect Societies; The Belknap Press of Harvard University Press: Cambridge, MA, USA, 1971; p. 548. [Google Scholar]
- Bryden, J.; Gill, R.J.; Mitton, R.A.A.; Raine, N.E.; Jansen, V.A.A. Chronic sublethal stress causes bee colony failure. Ecol. Lett. 2013, 16, 1463–1469. [Google Scholar] [CrossRef]
- Siviter, H.; Brown, M.J.F.; Leadbeater, E. Sulfoxaflor exposure reduces bumblebee reproductive success. Nature 2018, 561, 109. [Google Scholar] [CrossRef]
- Bogdanov, S. Contaminants of bee products. Apidologie 2006, 37, 1–18. [Google Scholar] [CrossRef]
- Nascimento, A.S.; Chambo, E.D.; Oliveria, D.J.; Andrade, B.R.; Bonsucesso, J.S.; Carvalho, C.A.L. Honey from stingless bee as indicator of contamination with metals. Sociobiology 2018, 65, 727–736. [Google Scholar] [CrossRef]
- Nascimento, N.O.; Nalini, H.A.; Ataide, F.; Abreu, A.T.; Antonini, Y. Pollen storage by stingless bees as an environmental marker for metal contamination: Spatial and temporal distribution of metal elements. Sociobiology 2018, 65, 252–263. [Google Scholar] [CrossRef]
- Sanchez-Bayo, F.; Goka, K. Pesticide residues and bees—A risk assessment. PLoS ONE 2014, 9, e94482. [Google Scholar] [CrossRef] [PubMed]
- Skaldina, O.; Peraniemi, S.; Sorvari, J. Ants and their nests as indicators for industrial heavy metal contamination. Environ. Pollut. 2018, 240, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Urbini, A.; Sparvoli, E.; Turillazzi, S. Social paper wasps as bioindicators: A preliminary research with Polistes dominulus (Hymenoptera Vespidae) as a trace metal accumulator. Chemosphere 2006, 64, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Murcia Morales, M.; Goméz Ramos, M.J.; Parrilla Vasquez, P.; Díaz Galiano, F.J.; García Valverde, M.; Manuel Flores, J.; Fernández-Alba, A.R. Distribution of chemical residues in the beehive compartments and their transfer to the honeybee brood. Sci. Total Environ. 2020, 710, 136288. [Google Scholar] [CrossRef] [PubMed]
- Traynor, K.S.; Pettis, J.S.; Tarpy, D.R.; Mullin, C.A.; Frazier, J.L.; Frazier, M.; vanEngeldorp, D. In-hive pesticide exposome: Assessing risks to migratory honey bees from in-hive pesticide contamination in the Eastern United States. Sci. Rep. 2016, 6, 33207. [Google Scholar] [CrossRef]
- Ostiguy, N.; Eitzer, B. Overwintered brood comb honey: Colony exposure to pesticide residues. J. Apic. Res. 2014, 53, 413–421. [Google Scholar] [CrossRef]
- Chauzat, M.P.; Faucon, J.P. Pesticide residues in beeswax samples collected from honey bee colonies (Apis mellifera l.) in France. Pest Manag. Sci. 2007, 63, 1100–1106. [Google Scholar] [CrossRef]
- Hladun, K.R.; Di, N.; Liu, T.X.; Trumble, J.T. Metal contaminant accumulation in the hive: Consequences for whole-colony health and brood production in the honey bee (Apis mellifera L.). Environ. Toxicol. Chem. 2016, 35, 322–329. [Google Scholar] [CrossRef]
- Exley, C.; Rotheray, E.; Goulson, D. Bumblebee pupae contain high levels of aluminium. PLoS ONE 2015, 10, e0127665. [Google Scholar] [CrossRef] [PubMed]
- Crall, J.D.; Switzer, C.M.; Oppenheimer, R.L.; Versypt, A.N.F.; Dey, B.; Brown, A.; Eyster, M.; Guerin, C.; Pierce, N.E.; Combes, S.A.; et al. Neonicotinoid exposure disrupts bumblebee nest behavior, social networks, and thermoregulation. Science 2018, 362, 683. [Google Scholar] [CrossRef] [PubMed]
- Gathmann, A.; Tscharntke, T. Foraging ranges of solitary bees. J. Anim. Ecol. 2002, 71, 757–764. [Google Scholar] [CrossRef]
- Couvillon, M.J.; Schürch, R.; Ratnieks, F.L.W. Dancing bees communicate a foraging preference for rural lands in high-level agri-environment schemes. Curr. Biol. 2014, 24, 1212–1215. [Google Scholar] [CrossRef] [PubMed]
- Moron, D.; Grzes, I.M.; Skorka, P.; Szentgyorgyi, H.; Laskowski, R.; Potts, S.G.; Woyciechowski, M. Abundance and diversity of wild bees along gradients of heavy metal pollution. J. Appl. Ecol. 2012, 49, 118–125. [Google Scholar] [CrossRef]
- Moron, D.; Szentgyorgyi, H.; Skorka, P.; Potts, S.G.; Woyciechowski, M. Survival, reproduction and population growth of the bee pollinator, Osmia rufa ( Hymenoptera: Megachilidae), along gradients of heavy metal pollution. Insect Conserv. Divers. 2014, 7, 113–121. [Google Scholar] [CrossRef]
- Gradish, A.E.; van der Steen, J.; Scott-Dupree, C.D.; Cabrera, A.R.; Cutler, C.G.; Goulson, D.; Klein, O.; Lehmann, D.M.; Luckmann, J.; O’Neill, B.; et al. Comparison of pesticide exposure in honey bees (Hymenoptera: Apidae) and bumble bees (Hymenoptera: Apidae): Implications for risk assessments. Environ. Entomol. 2019, 48, 12–21. [Google Scholar] [CrossRef]
- Sgolastra, F.; Hinarejos, S.; Pitts-Singer, T.L.; Boyle, N.K.; Joseph, T.; Luckmann, J.; Raine, N.E.; Singh, R.; Williams, N.M.; Bosch, J. Pesticide exposure assessment paradigm for solitary bees. Environ. Entomol. 2019, 48, 22–35. [Google Scholar] [CrossRef]
- Cameron, A.S.; Sadd, B.M. Global trends in bumble bee health. Annu. Rev. Entomol. 2020, 65, 209–232. [Google Scholar] [CrossRef]
- Grozinger, M.C.; Flenniken, M.L. Bee viruses: Ecology, pathogenicity, and impacts. Annu. Rev. Entomol. 2019, 64, 205–226. [Google Scholar] [CrossRef]
- Schmid-Hempel, P. Parasites in Social Insects. Monographs in Behavior and Ecology; Princeton University Press: Princeton, NJ, USA, 1998; p. 409. [Google Scholar]
- Schmid-Hempel, P.; Stauffer, H.P. Parasites and flower choice of bumblebees. Anim. Behav. 1998, 55, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Harrison, F.J.; Roberts, S.P. Flight respiration and energetics. Annu. Rev. Physiol. 2000, 62, 179–205. [Google Scholar] [CrossRef]
- Gonzalez-Varo, J.P.; Biesmeijer, J.C.; Bonmarco, R.; Potts, S.G.; Schweiger, O.; Smith, H.G.; Steffan-Dewenter, I.; Szentgyorgyi, H.; Woyciechowski, M.; Vila, M. Combined effects of global change pressures on animal-mediated pollination. Trends Ecol. Evol. 2013, 28, 524–530. [Google Scholar] [CrossRef] [PubMed]
- Goulson, D.; Lye, G.C.; Darvill, B. Decline and conservation of bumble bees. Annu. Rev. Entomol. 2008, 53, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Vanbergen, A.J.; Baude, M.; Biesmeijer, J.C.; Britton, N.F.; Brown, M.J.F.; Brown, M.; Byden, J.; Budge, G.E.; Bull, J.C.; Carvell, C.; et al. Threats to an ecosystem service: Pressures on pollinators. Front. Ecol. Environ. 2013, 11, 251–259. [Google Scholar] [CrossRef]
- vanEngelsdorp, D.; Evans, J.D.; Seagerman, C.; Mullin, C.; Haubruge, E.; Nguyen, B.K.; Frazier, M.; Frazier, J.; Cox-Foster, D.; Chen, Y.P.; et al. Colony Collapse Disorder: A descriptive study. PLoS ONE 2009, 4, e6481. [Google Scholar] [CrossRef] [PubMed]
- David, A.; Botias, C.; Abdul-Sada, A.; Nicholls, E.; Rotheray, E.L.; Hill, E.M.; Goulson, D. Widespread contamination of wildflower and bee-collected pollen with complex mixtures of neonicotinoids and fungicides commonly applied to crops. Environ. Int. 2016, 88, 169–178. [Google Scholar] [CrossRef]
- Tosi, S.; Costa, C.; Vesco, U.; Quaglia, G.; Guido, G. A 3-year survey of Italian honey bee-collected pollen reveals widespread contamination by agricultural pesticides. Sci. Total Environ. 2018, 615, 208–218. [Google Scholar] [CrossRef]
- Botias, C.; David, A.; Hill, E.M.; Goulson, D. Quantifying exposure of wild bumblebees to mixtures of agrochemicals in agricultural and urban landscapes. Environ. Pollut. 2017, 222, 73–82. [Google Scholar] [CrossRef]
- Botias, C.; David, A.; Hill, E.M.; Goulson, D. Contamination of wild plants near neonicotinoid seed-treated crops, and implications for non-target insects. Sci. Total Environ. 2016, 566, 269–278. [Google Scholar] [CrossRef]
- Barganska, Z.; Slebioda, M.; Namiesnik, J. Honey bees and their products: Bioindicators of environmental contamination. Crit. Rev. Environ. Sci. Technol. 2016, 46, 235–248. [Google Scholar] [CrossRef]
- Bromenshenk, J.J.; Gudatis, J.L.; Carlson, S.R.; Thomas, J.M.; Simmons, M.A. Population-dynamics of honey-bee nucleus colonies exposed to industrial pollutants. Apidologie 1991, 22, 359–369. [Google Scholar] [CrossRef]
- Conti, E.M.; Botre, F. Honeybees and their products as potential bioindicators of heavy metals contamination. Environ. Monit. Assess. 2001, 69, 267–282. [Google Scholar] [CrossRef]
- Costa, A.; Veca, M.; Barberis, M.; Tosti, A.; Notaro, G.; Nava, S.; Lazzari, M.; Agazzi, A.; Tangorra, F.M. Heavy metals on honeybees indicate their concentration in the atmosphere. A proof of concept. Ital. J. Anim. Sci. 2019, 18, 309–315. [Google Scholar] [CrossRef]
- Lazor, P.; Tomas, J.; Toth, T.; Toth, J.; Ceryova, S. Monitoring of air pollution and atmospheric deposition of heavy metals by analysis of honey. J. Microbiol. Biotechnol. Food Sci. 2012, 1, 522–533. [Google Scholar]
- Perugini, M.; Manera, M.; Grotta, L.; Abete, M.C.; Tarasco, R.; Amorena, M. Heavy metal (Hg, Cr, Cd, and Pb) contamination in urban areas and wildlife reserves: Honeybees as bioindicators. Biol. Trace Elem. Res. 2011, 140, 170–176. [Google Scholar] [CrossRef]
- Skorbilowicz, E.; Skorbilowicz, M.; Ciesluk, I. Bees as bioindicators of environmental pollution with metals in an urban area. J. Ecol. Eng. 2018, 19, 229–234. [Google Scholar] [CrossRef]
- Smith, K.E.; Weis, D.; Amini, M.; Shiel, A.E.; Lai, V.W.M.; Gordon, K. Honey as a biomonitor for a changing world. Nat. Sustain. 2019, 2, 223–232. [Google Scholar] [CrossRef]
- Szentgyorgyi, H.; Blinow, A.; Eremeeva, N.; Luzyanin, S.; Grzes, I.M.; Woyciechowski, M. Bumblebees (Bombidae) along pollution gradient—heavy metal accumulation, species diversity, and Nosema bombi infection level. Pol. J. Ecol. 2011, 59, 599–610. [Google Scholar]
- Dzugan, M.; Wesolowska, M.; Zagula, G.; Kaczmarski, M.; Czernicka, M.; Puchalski, C. Honeybees (Apis mellifera) as a biological barrier for contamination of honey by environmental toxic metals. Environ. Monit. Assess. 2018, 190, 101. [Google Scholar] [CrossRef]
- Gall, J.E.; Boyd, R.S.; Rajakaruna, N. Transfer of heavy metals through terrestrial food webs: A review. Environ. Monit. Assess. 2015, 187, 201. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, M.; Molero, R.; Gaju, M.; van der Steen, J.; Porrini, C.; Ruiz, J.A. Assessment of heavy metal pollution in Cordoba (Spain) by biomonitoring foraging honeybee. Environ. Monit. Assess. 2015, 187, 651. [Google Scholar] [CrossRef] [PubMed]
- Krakowska, A.; Muszynska, B.; Reczynski, W.; Opoka, W.; Turski, W. Trace metal analyses in honey samples from selected countries. A Potential Use Bio-Monit. Int. J. Environ. Anal. Chem. 2015, 95, 855–866. [Google Scholar]
- Lawal, O.A.; Ademolu, K.O.; Aina, S.A.; Abiade, A.N. Influence of nesting habitats on the gut enzymes activity and heavy metal composition of Apis mellifera andersonii L. (Hymenoptera: Apidae). Afr. Entomol. 2014, 22, 163–166. [Google Scholar] [CrossRef]
- Matin, G.; Kargar, N.; Buyukisik, H.B. Bio-monitoring of cadmium, lead, arsenic and mercury in industrial districts of Izmir, Turkey by using honey bees, propolis and pine tree leaves. Ecol. Eng. 2016, 90, 331–335. [Google Scholar] [CrossRef]
- Silici, S.; Uluozlu, O.D.; Tuzen, M.; Soylak, M. Honeybees and honey as monitors for heavy metal contamination near thermal power plants in Mugla, Turkey. Toxicol. Ind. Health 2016, 32, 507–516. [Google Scholar] [CrossRef]
- Yazgan, S.; Horn, H.; Isengard, H.D. Honey as bio indicator by screening the heavy metal content of the environment. Dtsch. Lebensm.-Rundsch. 2006, 102, 192–194. [Google Scholar]
- Polidori, C.; Pastor, A.; Jorge, A.; Pertusa, J. Ultrastructural alterations of midgut epithelium, but not greater wing fluctuating asymmetry, in paper wasps (Polistes dominula) from urban environments. Microsc. Microanal. 2018, 24, 183–192. [Google Scholar] [CrossRef]
- McEachran, A.D.; Blackwell, B.R.; Hanson, J.D.; Wooten, K.J.; Mayer, G.D.; Cox, S.B.; Smith, P.N. Antibiotics, bacteria, and antibiotic resistance genes: Aerial transport from cattle feed yards via particulate matter. Environ. Health Perspect. 2015, 123, 337–343. [Google Scholar] [CrossRef]
- Tapparo, A.; Marton, D.; Giorio, C.; Zanella, A.; Solda, L.; Marzaro, M.; Vivan, L.; Girolami, V. Assessment of the environmental exposure of honeybees to particulate matter containing neonicotinoid insecticides coming from corn coated seeds. Environ. Sci. Technol. 2012, 46, 2592–2599. [Google Scholar] [CrossRef]
- Marzaro, M.; Vivan, L.; Targa, A.; Mazzon, L.; Mori, N.; Greatti, M.; Toffolo, E.P.; Di Bernardo, A.; Giorio, C.; Marton, D.; et al. Lethal aerial powdering of honey bees with neonicotinoids from fragments of maize seed coat. Bull. Insectol. 2011, 64, 119–126. [Google Scholar]
- Zhou, X.T.; Taylor, M.P.; Davies, P.J. Tracing natural and industrial contamination and lead isotopic compositions in an Australian native bee species. Environ. Pollut. 2018, 242, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.T.; Taylor, M.P.; Davies, P.J.; Prasad, S. Identifying sources of environmental contamination in European honey bees (Apis mellifera) using trace elements and lead isotopic compositions. Environ. Sci. Technol. 2018, 52, 991–1001. [Google Scholar] [CrossRef] [PubMed]
- Bell, J.N.B.; Power, S.A.; Jarraud, N.; Agrawal, M.; Davies, C. The effects of air pollution on urban ecosystems and agriculture. Int. J. Sustain. Dev. World Ecol. 2011, 18, 226–235. [Google Scholar] [CrossRef]
- Lukowski, A.; Popek, R.; Jagiello, R.; Maderek, E.; Karolewski, P. Particulate matter on two Prunus spp. decreases survival and performance of the folivorous beetle Gonioctena quinquepunctata. Environ. Sci. Pollut. Res. 2018, 25, 16629–16639. [Google Scholar]
- Negri, I.; Mavris, C.; Di Prisco, G.; Caprio, E.; Pellecchia, M. Honey bees (Apis mellifera, L.) as active samplers of airborne particulate matter. PLoS ONE 2015, 10, e132491. [Google Scholar] [CrossRef]
- Pellecchia, M.; Negri, I. Particulate matter collection by honey bees (Apis mellifera, L.) near to a cement factory in Italy. Peerj 2018, 6, E5322. [Google Scholar] [CrossRef]
- Tan, Y.Q.; Dion, E.; Monteiro, A. Haze smoke impacts survival and development of butterflies. Sci. Rep. 2018, 8, 15667. [Google Scholar] [CrossRef]
- Vanderstock, A.M.; Latty, T.; Leonard, R.J.; Hochuli, D.F. Mines over matter: Effects of foliar particulate matter on the herbivorous insect, Helicoverpa armigera. J. Appl. Entomol. 2019, 143, 77–87. [Google Scholar] [CrossRef]
- Peterson, E.M.; Wooten, K.J.; Subbiah, S.; Anderson, T.A.; Longing, S.; Smith, P.N. Agrochemical mixtures detected on wildflowers near cattle feed yards. Environ. Sci. Technol. Lett. 2017, 4, 216–220. [Google Scholar] [CrossRef]
- Kreider, M.L.; Panko, J.M.; McAtee, B.L.; Sweet, L.I.; Finley, B.L. Physical and chemical characterization of tire-related particles: Comparison of particles generated using different methodologies. Sci. Total Environ. 2010, 408, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Pant, P.; Baker, S.J.; Shukla, A.; Maikawa, C.; Pollitt, K.J.G.; Harrison, R.M. The PM10 fraction of road dust in the UK and India: Characterization, source profiles and oxidative potential. Sci. Total Environ. 2015, 530, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Pant, P.; Harrison, R.M. Estimation of the contribution of road traffic emissions to particulate matter concentrations from field measurements: A review. Atmos. Environ. 2013, 77, 78–97. [Google Scholar] [CrossRef]
- Dominici, F.; Wang, Y.; Correia, A.W.; Ezzati, M.; Pope, C.A.; Dockery, D.W. Chemical composition of fine particulate matter and life expectancy in 95 US counties between 2002 and 2007. Epidemiology 2015, 26, 556–564. [Google Scholar] [CrossRef]
- Kelly, F.J.; Fuller, G.W.; Walton, H.A.; Fussell, J.C. Monitoring air pollution: Use of early warning systems for public health. Respirology 2012, 17, 7–19. [Google Scholar] [CrossRef]
- Kim, K.H.; Jahan, S.A.; Kabir, E. A review on human health perspective of air pollution with respect to allergies and asthma. Environ. Int. 2013, 59, 41–52. [Google Scholar] [CrossRef]
- Calatayud-Vernich, P.; Calatayud, F.; Simo, E.; Pico, Y. Pesticide residues in honey bees, pollen and beeswax: Assessing beehive exposure. Environ. Pollut. 2018, 241, 106–114. [Google Scholar] [CrossRef]
- Chauzat, M.P.; Martel, A.C.; Cougoule, N.; Porta, P.; Lachaize, J.; Zeggane, S.; Aubert, M.; Carpentier, P.; Faucon, J.P. An assessment of honeybee colony matrices, Apis mellifera (Hymenoptera Apidae) to monitor pesticide presence in continental France. Environ. Toxicol. Chem. 2011, 30, 103–111. [Google Scholar] [CrossRef]
- Daniele, G.; Giroud, B.; Jabot, C.; Vulliet, E. Exposure assessment of honeybees through study of hive matrices: Analysis of selected pesticide residues in honeybees, beebread, and beeswax from French beehives by LC-MS/MS. Environ. Sci. Pollut. Res. 2018, 25, 6145–6153. [Google Scholar] [CrossRef]
- Gonzalez-Martin, M.I.; Revilla, I.; Betances-Salcedo, E.V.; Vivar-Quintana, A.M. Pesticide residues and heavy metals in commercially processed propolis. Microchem. J. 2018, 143, 423–429. [Google Scholar] [CrossRef]
- Manning, R. Chemical residues in beebread, honey, pollen and wax samples collected from bee hives placed on canola crops in Western Australia. J. Apic. Res. 2018, 57, 696–708. [Google Scholar] [CrossRef]
- Porrini, C.; Caprio, E.; Tesoriero, D.; Di Prisco, G. Using honey bee as bioindicator of chemicals in Campanian agroecosystems (South Italy). Bull. Insectol. 2014, 67, 137–146. [Google Scholar]
- Ravoet, J.; Reybroeck, W.; de Graaf, D.C. Pesticides for apicultural and/or agricultural application found in Belgian honey bee wax combs. Bull. Environ. Contam. Toxicol. 2015, 94, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Valdovinos-Flores, C.; Alcantar-Rosales, V.M.; Gaspar-Ramirez, O.; Saldana-Loza, L.M.; Dorantes-Ugalde, J.A. Agricultural pesticide residues in honey and wax combs from Southeastern, Central and Northeastern Mexico. J. Apic. Res. 2017, 56, 667–679. [Google Scholar] [CrossRef]
- Kammoun, S.; Mulhauser, B.; Aebi, A.; Mitchell, E.A.D.; Glauser, G. Ultra-trace level determination of neonicotinoids in honey as a tool for assessing environmental contamination. Environ. Pollut. 2019, 247, 964–972. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, B.D. Eradication of populations of an invasive ant in northern Australia: Successes, failures and lessons for management. Biodivers. Conserv. 2011, 20, 3267–3278. [Google Scholar] [CrossRef]
- Buczkowski, G.; Wossler, T.C. Controlling invasive Argentine ants, Linepithema humile, in conservation areas using horizontal insecticide transfer. Sci. Rep. 2019, 9, 19495. [Google Scholar] [CrossRef]
- Calibeo, D.; Oi, F.; Oi, D.; Mannion, C. Insecticides for suppression of Nylanderia fulva. Insects 2017, 8, 93. [Google Scholar] [CrossRef]
- Sakamoto, Y.; Hayashi, T.I.; Inoue, M.N.; Ohnishi, H.; Kishimoto, T.; Goka, K. Effects of fipronil on non-target ants and other invertebrates in a program for eradication of the Argentine ant, Linepithema humile. Sociobiology 2019, 66, 227–238. [Google Scholar] [CrossRef]
- Plentovich, S.; Swenson, C.; Reimer, N.; Richardson, M.; Garon, N. The effects of hydramethylnon on the tropical fire ant, Solenopsis geminata (Hymenoptera: Formicidae), and non-target arthropods on Spit Island, Midway Atoll, Hawaii. J. Insect Conserv. 2010, 14, 459–465. [Google Scholar] [CrossRef]
- Pisa, L.W.; Amaral-Rogers, V.; Belzunces, L.P.; Bonmatin, J.M.; Downs, C.A.; Goulson, D.; Kreutzweiser, D.P.; Krupke, C.; Liess, M.; McField, M.; et al. Effects of neonicotinoids and fipronil on non-target invertebrates. Environ. Sci. Pollut. Res. 2015, 22, 68–102. [Google Scholar] [CrossRef] [PubMed]
- Wood, T.J.; Goulson, D. The environmental risks of neonicotinoid pesticides: A review of the evidence post 2013. Environ. Sci. Pollut. Res. 2017, 24, 17285–17325. [Google Scholar] [CrossRef] [PubMed]
- Alburaki, M.; Chen, D.; Skinner, J.A.; Meikle, W.G.; Tarpy, D.R.; Adamczyk, J.; .Stewart, S.D. Honey bee survival and pathogen prevalence: From the perspective of landscape and exposure to pesticides. Insects 2018, 9, 65. [Google Scholar] [CrossRef] [PubMed]
- Calatayud-Vernich, P.; Calatayud, F.; Simo, E.; Aguilar, J.A.P.; Pico, Y. A two-year monitoring of pesticide hazard in-hive: High honey bee mortality rates during insecticide poisoning episodes in apiaries located near agricultural settings. Chemosphere 2019, 232, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Calatayud-Vernich, P.; Calatayud, F.; Simo, E.; Suarez-Varela, M.M.; Pico, Y. Influence of pesticide use in fruit orchards during blooming on honeybee mortality in 4 experimental apiaries. Sci. Total Environ. 2016, 541, 33–41. [Google Scholar] [CrossRef]
- Tsvetkov, N.; Samson-Robert, O.; Sood, K.; Patel, H.S.; Malena, D.A.; Gajiwala, P.H.; Maciukiewicz, P.; Fournier, V.; Zayed, A. Chronic exposure to neonicotinoids reduces honey bee health near corn crops. Science 2017, 356, 1395. [Google Scholar] [CrossRef]
- Woodcock, B.A.; Bullock, J.M.; Shore, R.F.; Heard, M.S.; Pereira, M.G.; Redhead, J.; Ridding, L.; Dean, H.; Sleep, D.; Henrys, P.; et al. Country-specific effects of neonicotinoid pesticides on honey bees and wild bees. Science 2017, 356, 1393. [Google Scholar] [CrossRef]
- Woodcock, B.A.; Isaac, N.J.B.; Bullock, J.M.; Roy, D.B.; Garthwaite, D.G.; Crowe, A.; Pywell, R.F. Impacts of neonicotinoid use on long-term population changes in wild bees in England. Nat. Commun. 2016, 7, 12459. [Google Scholar] [CrossRef]
- Aghamirlou, H.M.; Khadem, M.; Rahmani, A.; Sadeghian, M.; Mahvi, A.H.; Akbarzadeh, A.; Nazmara, S. Heavy metals determination in honey samples using inductively coupled plasma-optical emission spectrometry. J. Environ. Health Sci. Eng. 2015, 13, 39. [Google Scholar] [CrossRef]
- Altunatmaz, S.S.; Tarhan, D.; Aksu, F.; Barutcu, U.B.; Or, M.E. Mineral element and heavy metal (cadmium, lead and arsenic) levels of bee pollen in Turkey. Food Sci. Technol. 2017, 37, 136–141. [Google Scholar] [CrossRef]
- Bratu, I.; Georgescu, C. The comparative study of heavy metals contamination of honey and flowers coming from a chemically polluted area. Rev. Chim. 2005, 56, 118–120. [Google Scholar]
- Celechovska, O.; Vorlova, L. Groups of honey—Physicochemical properties and heavy metals. Acta Vet. Brno 2001, 70, 91–95. [Google Scholar] [CrossRef]
- Formicki, G.; Gren, A.; Stawarz, R.; Zysk, B.; Gal, A. Metal content in honey, propolis, wax, and bee pollen and implications for metal pollution monitoring. Pol. J. Environ. Stud. 2013, 22, 99–106. [Google Scholar]
- Kovacik, J.; Gruz, J.; Biba, O.; Hedbavny, J. Content of metals and metabolites in honey originated from the vicinity of industrial town Kosice (eastern Slovakia). Environ. Sci. Pollut. Res. 2016, 23, 4531–4540. [Google Scholar] [CrossRef] [PubMed]
- Leita, L.; Muhlbachova, G.; Cesco, S.; Barabttini, R.; Mondini, C. Investigation of the use of honey bees and honey bee products to assess heavy metals contamination. Environ. Monit. Assess. 1996, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ruschioni, S.; Riolo, P.; Minuz, R.L.; Stefano, M.; Cannella, M.; Porrini, C.; Isidoro, N. Biomonitoring with honeybees of heavy metals and pesticides in nature reserves of the Marche region (Italy). Biol. Trace Elem. Res. 2013, 154, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Sitarz-Palczak, E.; Kalembkiewicz, J.; Galas, D. Evaluation of the content of selected heavy metals in samples of Polish honeys. J. Ecol. Eng. 2015, 16, 130–138. [Google Scholar] [CrossRef]
- Spiric, D.; Ciric, J.; Dordevic, V.; Nikolic, D.; Jankovic, S.; Nikolic, A.; Petrovic, Z.; Katanic, N.; Teodorovic, V. Toxic and essential element concentrations in different honey types. Int. J. Environ. Anal. Chem. 2019, 99, 474–485. [Google Scholar] [CrossRef]
- Burden, C.M.; Elmore, C.; Hladun, K.R.; Trumble, J.T.; Smith, B.H. Acute exposure to selenium disrupts associative conditioning and long-term memory recall in honey bees (Apis mellifera). Ecotoxicol. Environ. Saf. 2016, 127, 71–79. [Google Scholar] [CrossRef]
- Burden, C.M.; Morgan, M.O.; Hladun, K.R.; Amdam, G.V.; Trumble, J.T.; Smith, B.H. Acute sublethal exposure to toxic heavy metals alters honey bee (Apis mellifera) feeding behavior. Sci. Rep. 2019, 9, 4253. [Google Scholar] [CrossRef]
- Eeva, T.; Sorvari, J.; Kolvunen, V. Effects of heavy metal pollution on red wood ant (Formica s. str.) populations. Environ. Pollut. 2004, 132, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Nisbet, C.; Guler, A.; Biyik, S. Effects of different environmental conditions on the cognitive function of honeybee (Apis mellifera L.) and mineral content of honey. Ank. Univ. Vet. Fak. Derg. 2019, 66, 95–101. [Google Scholar]
- Sgolastra, F.; Blasioli, S.; Renzi, T.; Tosi, S.; Medrzycki, P.; Molowny-Horas, R.; Porrini, C.; Braschi, I. Lethal effects of Cr(III) alone and in combination with propiconazole and clothianidin in honey bees. Chemosphere 2018, 191, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Skaldina, O.; Sorvari, J. Ecotoxicological Effects of Heavy Metal Pollution on Economically Important Terrestrial Insects, in Networking of Mutagens in Environmental Toxicology; Kesari, K.K., Ed.; Springer: Cham, Switzerland, 2009; pp. 137–144. [Google Scholar]
- Sorvari, J.; Eeva, T. Pollution diminishes intra-specific aggressiveness between wood ant colonies. Sci. Total Environ. 2010, 408, 3189–3192. [Google Scholar] [CrossRef] [PubMed]
- Sorvari, J.; Rantala, L.M.; Rantala, M.J.; Hakkarainen, H.; Eeva, T. Heavy metal pollution disturbs immune response in wild ant populations. Environ. Pollut. 2007, 145, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Szentgyorgyi, H.; Moron, D.; Nawrocka, A.; Tofilski, A.; Woyciechowski, M. Forewing structure of the solitary bee Osmia bicornis developing on heavy metal pollution gradient. Ecotoxicology 2017, 26, 1031–1040. [Google Scholar] [CrossRef]
- Kampa, M.; Castanas, E. Human health effects of air pollution. Environ. Pollut. 2008, 151, 362–367. [Google Scholar] [CrossRef]
- Järup, L. Hazards of heavy metal contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef]
- Tragust, S.; Tartally, A.; Espadaler, X.; Billen, J. Histopathology of Laboulbeniales (Ascomycota: Laboulbeniales): Ectoparasitic fungi on ants (Hymenoptera: Formicidae). Myrmecol. News 2016, 23, 81–89. [Google Scholar]
- Tragust, S.; Feldhaar, H.; Espadaler, X.; Pedersen, J.S. Rapid increase of the parasitic fungus Laboulbenia formicarum in supercolonies of the invasive garden ant Lasius neglectus. Biol. Invasions 2015, 17, 2795–2801. [Google Scholar] [CrossRef]
- Bos, N.; Kankaanpaa-Kukkonen, V.; Freitak, D.; Stucki, D.; Sundstrom, L. Comparison of twelve ant species and their susceptibility to fungal infection. Insects 2019, 10, 271. [Google Scholar] [CrossRef] [PubMed]
- Valles, S.M.; Rivers, A.R. Nine new RNA viruses associated with the fire ant Solenopsis invicta from its native range. Virus Genes 2019, 55, 368–380. [Google Scholar] [CrossRef] [PubMed]
- Li, J.L.; Qin, H.R.; Wu, J.; Sadd, B.M.; Wang, X.H.; Evans, J.D.; Peng, W.J.; Chen, Y.P. The prevalence of parasites and pathogens in Asian honeybees Apis cerana in China. PLoS ONE 2012, 7, e47955. [Google Scholar] [CrossRef] [PubMed]
- Manfredini, F.; Shoemaker, D.; Grozinger, C.M. Dynamic changes in host-virus interactions associated with colony founding and social environment in fire ant queens (Solenopsis invicta). Ecol. Evol. 2016, 6, 233–244. [Google Scholar] [CrossRef]
- Porter, S.D.; Valles, S.M.; Oi, D.H. Host specificity and colony impacts of the fire ant pathogen, Solenopsis invicta virus 3. J. Invertebr. Pathol. 2013, 114, 1–6. [Google Scholar] [CrossRef]
- Tufts, D.M.; Hunter, W.B.; Bextine, B. Discovery and effects of Texas Solenopsis invicta virus [SINV-1 (TX5)] on red imported fire ant populations. J. Invertebr. Pathol. 2010, 104, 180–185. [Google Scholar] [CrossRef]
- Valles, S.M.; Porter, S.D. Dose response of red imported fire ant colonies to Solenopsis invicta virus 3. Arch. Virol. 2015, 160, 2407–2413. [Google Scholar] [CrossRef]
- Valles, S.M. Positive-strand RNA viruses infecting the red imported fire ant, Solenopsis invicta. Psyche 2012, 2012, 1–14. [Google Scholar] [CrossRef]
- Cordes, N.; Huang, W.F.; Strange, J.P.; Cameron, S.A.; Griswold, T.L.; Lozier, J.D.; Solter, L.F. Interspecific geographic distribution and variation of the pathogens Nosema bombi and Crithidia species in United States bumble bee populations. J. Invertebr. Pathol. 2012, 109, 209–216. [Google Scholar] [CrossRef]
- Brown, M.J.F.; Loosli, R.; Schmid-Hempel, P. Condition-dependent expression of virulence in a trypanosome infecting bumblebees. Oikos 2000, 91, 421–427. [Google Scholar] [CrossRef]
- Brown, M.J.F.; Schmid-Hempel, R.; Schmid-Hempel, P. Strong context-dependent virulence in a host-parasite system: Reconciling genetic evidence with theory. J. Anim. Ecol. 2003, 72, 994–1002. [Google Scholar] [CrossRef]
- Fauser, A.; Sandrock, C.; Neumann, P.; Sadd, B. Neonicotinoids override a parasite exposure impact onhibernation success of a key bumblebee pollinator. Ecol. Entomol. 2017, 42, 306–314. [Google Scholar] [CrossRef]
- Genersch, E. Honey bee pathology: Current threats to honey bees and beekeeping. Appl. Microbiol. Biotechnol. 2010, 87, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Goblirsch, M. Nosema ceranae disease of the honey bee (Apis mellifera). Apidologie 2018, 49, 131–150. [Google Scholar] [CrossRef]
- Paris, L.; El Alaoui, H.; Delbac, F.; Diogon, M. Effects of the gut parasite Nosema ceranae on honey bee physiology and behavior. Curr. Opin. Insect Sci. 2018, 26, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Lemaitre, B.; Hoffmann, J. The host defense of Drosophila melanogaster. Annu. Rev. Immunol. 2007, 25, 697–743. [Google Scholar] [CrossRef]
- Otti, O.; Tragust, S.; Feldhaar, H. Unifying external and internal immune defences. Trends Ecol. Evol. 2014, 29, 625–634. [Google Scholar] [CrossRef]
- Cremer, S.; Armitage, S.A.O.; Schmid-Hempel, P. Social immunity. Curr. Biol. 2007, 17, R693–R702. [Google Scholar] [CrossRef]
- Broderick, N.A.; Welchman, D.P.; Lemaitre, B. Recognition and Response to Microbial Infection in Drosophila, in Insect Infection and Immunity; Rolff, J., Reynolds, S.E., Eds.; Oxford University Press: New York, NY, USA, 2009; pp. 13–33. [Google Scholar]
- Wilson-Rich, N.; Bonoan, R.E.; Taylor, E.; Lwanga, L.; Starks, P.T. An improved method for testing invertebrate encapsulation response as shown in the honey bee. Insectes Soc. 2019, 66, 91–105. [Google Scholar] [CrossRef]
- Allander, K.; Schmid-Hempel, P. Immune defence reaction in bumble-bee workers after a previous challenge and parasitic coinfection. Funct. Ecol. 2000, 14, 711–717. [Google Scholar] [CrossRef]
- Sorvari, J.; Hakkarainen, H.; Rantala, M.J. Immune defense of ants is associated with changes in habitat characteristics. Environ. Entomol. 2008, 37, 51–56. [Google Scholar] [CrossRef]
- Li, X.C.; Schuler, M.A.; Berenbaum, M.R. Molecular mechanisms of metabolic resistance to synthetic and natural xenobiotics. Annu. Rev. Entomol. 2007, 52, 231–253. [Google Scholar] [CrossRef] [PubMed]
- Berenbaum, M.R.; Johnson, R.M. Xenobiotic detoxification pathways in honey bees. Curr. Opin. Insect Sci. 2015, 10, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Collison, E.J.; Hird, H.; Tyler, C.R.; Cresswell, J.E. Effects of neonicotinoid exposure on molecular and physiological indicators of honey bee immunocompetence. Apidologie 2018, 49, 196–208. [Google Scholar] [CrossRef]
- Alaux, C.; Brunet, J.L.; Dussaubat, C.; Mondet, F.; Tchamitchan, S.; Cousin, M.; Brillard, J.; Baldy, A.; Belzunces, L.P.; Le Conte, Y. Interactions between Nosema microspores and a neonicotinoid weaken honeybees (Apis mellifera). Environ. Microbiol. 2010, 12, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Reeves, A.M.; O’Neal, S.T.; Fell, R.D.; Brewster, C.C.; Anderson, T.D. In-hive acaricides alter biochemical and morphological indicators of honey bee nutrition, immunity, and development. J. Insect Sci. 2018, 18, 8. [Google Scholar] [CrossRef]
- O’Neal, S.T.; Reeves, A.M.; Fell, R.D.; Brewster, C.C.; Anderson, T.D. Chlorothalonil exposure alters virus susceptibility and markers of immunity, nutrition, and development in honey bees. J. Insect Sci. 2019, 19, 14. [Google Scholar]
- Paris, L.; Roussel, M.; Pereira, B.; Delbac, F.; Diogon, M. Disruption of oxidative balance in the gut of the western honeybee Apis mellifera exposed to the intracellular parasite Nosema ceranae and to the insecticide fipronil. Microb. Biotechnol. 2017, 10, 1702–1717. [Google Scholar] [CrossRef]
- Carneiro, L.S.; Martinez, L.C.; Goncalves, W.G.; Santana, L.M.; Serraeo, J.E. The fungicide iprodione affects midgut cells of non-target honey bee Apis mellifera workers. Ecotoxicol. Environ. Saf. 2020, 189, 109991. [Google Scholar] [CrossRef]
- Lämsä, J.; Kuusela, E.; Tuomi, J.; Juntunen, S.; Watts, P.C. Low dose of neonicotinoid insecticide reduces foraging motivation of bumblebees. Proc. R. Soc. B-Biol. Sci. 2018, 285, 2018506. [Google Scholar]
- Moret, Y.; Schmid-Hempel, P. Survival for immunity: The price of immune system activation for bumblebee workers. Science 2000, 290, 1166–1168. [Google Scholar] [CrossRef] [PubMed]
- Ardia, D.R.; Gantz, J.E.; Schneider, B.C.; Strebel, S. Costs of immunity in insects: An induced immune response increases metabolic rate and decreases antimicrobial activity. Funct. Ecol. 2012, 26, 732–739. [Google Scholar] [CrossRef]
- Klein, S.; Cabirol, A.; Devaud, J.M.; Barron, A.B.; Lihoreau, M. Why bees are so vulnerable to environmental stressors. Trends Ecol. Evol. 2017, 32, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Boucias, D.G.; Stokes, C.; Storey, G.; Pendland, J.C. The effects of imidacloprid on the termite Reticulitermes flavipes and its interaction with the mycopathogen Beauveria bassiana. Pflanzenschutz-Nachr. Bayer 1996, 49, 103–144. [Google Scholar]
- Raymann, K.; Moran, N.A. The role of the gut microbiome in health and disease of adult honey bee workers. Curr. Opin. Insect Sci. 2018, 26, 97–104. [Google Scholar] [CrossRef]
- Engel, P.; Kwong, W.K.; McFrederick, Q.; Anderson, K.E.; Barribeau, S.M.; Chandler, J.A.; Cornman, R.S.; Dainat, J.; de Miranda, J.R.; Doublet, V.; et al. The bee microbiome: Impact on bee health and model for evolution and ecology of host-microbe interactions. Mbio 2016, 7, e02164. [Google Scholar] [CrossRef]
- Hamdi, C.; Balloi, A.; Essanaa, J.; Crotti, E.; Gonella, E.; Raddadi, N.; Ricci, I.; Boudabous, A.; Borin, S.; Manino, A.; et al. Gut microbiome dysbiosis and honeybee health. J. Appl. Entomol. 2011, 135, 524–533. [Google Scholar] [CrossRef]
- Rothman, J.A.; Leger, L.; Kirkwood, J.S.; McFrederick, Q.S. Cadmium and selenate exposure affects the honey bee microbiome and metabolome, and bee-associated bacteria show potential for bioaccumulation. Appl. Environ. Microbiol. 2019, 85, e01411-19. [Google Scholar] [CrossRef]
- Yang, Y.; Ma, S.L.; Yan, Z.X.; Liu, F.; Diao, Q.Y.; Dai, P.L. Effects of three common pesticides on survival, food consumption and midgut bacterial communities of adult workers Apis cerana and Apis mellifera. Environ. Pollut. 2019, 249, 860–867. [Google Scholar] [CrossRef]
- Motta, E.V.S.; Raymann, K.; Moran, N.A. Glyphosate perturbs the gut microbiota of honey bees. Proc. Natl. Acad. Sci. USA 2018, 115, 10305–10310. [Google Scholar] [CrossRef]
- Kakumanu, M.L.; Reeves, A.M.; Anderson, T.D.; Rodrigues, R.R.; Williams, M.A. Honey bee gut microbiome is altered by in-hive pesticide exposures. Front. Microbiol. 2016, 7, 1255. [Google Scholar] [CrossRef] [PubMed]
- Blot, N.; Veillat, L.; Rouze, R.; Delatte, H. Glyphosate, but not its metabolite AMPA, alters the honeybee gut microbiota. PLoS ONE 2019, 14, e0215466. [Google Scholar] [CrossRef] [PubMed]
- Lucky, A. Urban ants of North America and Europe: Identification, biology, and management. Syst. Entomol. 2009, 34, 406–407. [Google Scholar] [CrossRef]
- Lutinski, J.A.; Lopes, B.C.; de Morais, A.B.B. Urban ant diversity (Hymenoptera: Formicidae) in ten cities of southern Brazil. Biota Neotrop. 2013, 13, 332–342. [Google Scholar] [CrossRef]
- Savage, A.M.; Hackett, B.; Guenard, B.; Youngsteadt, E.K.; Dunn, R.R. Fine-scale heterogeneity across Manhattan’s urban habitat mosaic is associated with variation in ant composition and richness. Insect Conserv. Divers. 2015, 8, 216–228. [Google Scholar] [CrossRef]
- Youngsteadt, E.; Appler, R.H.; Lopez-Uribe, M.M.; Tarpy, D.R.; Frank, S.D. Urbanization increases pathogen pressure on feral and managed honey bees. PLoS ONE 2015, 10, e0142031. [Google Scholar] [CrossRef]
- Johnson, R.M. Honey bee toxicology. Annu. Rev. Entomol. 2015, 60, 415–434. [Google Scholar] [CrossRef]
- Fairbrother, A.; Purdy, J.; Anderson, T.; Fell, R. Risks of neonicotinoid insecticides to honeybees. Environ. Toxicol. Chem. 2014, 33, 719–731. [Google Scholar] [CrossRef]
- Lopez, D.R.; Ahumada, D.A.; Diaz, A.C.; Guerrero, J.A. Evaluation of pesticide residues in honey from different geographic regions of Colombia. Food Control 2014, 37, 33–40. [Google Scholar] [CrossRef]
- Luken, D.J.; von der Ohe, W. A research about different residues in pollen and honey samples. Hazards Pestic. Bees 2018, 462, 198–202. [Google Scholar]
- Mullin, C.A.; Frazier, M.; Frazier, J.L.; Ashcraft, S.; Simonds, R.; vanEngelsdorp, D.; Pettis, J.S. High levels of miticides and agrochemicals in North American apiaries: Implications for honey bee health. PLoS ONE 2010, 5, e9754. [Google Scholar] [CrossRef]
- Stanley, D.A.; KSmith, E.; Raine, N.E. Bumblebee learning and memory is impaired by chronic exposure to a neonicotinoid pesticide. Sci. Rep. 2015, 5, 16508. [Google Scholar] [CrossRef]
- Baron, G.L.; Raine, N.E.; Brown, M.J.F. General and species-specific impacts of a neonicotinoid insecticide on the ovary development and feeding of wild bumblebee queens. Proc. R. Soc. B-Biol. Sci. 2017, 284, 20170123. [Google Scholar] [CrossRef] [PubMed]
- Brandt, A.; Gorenflo, A.; Siede, R.; Meixner, M.; Büchler, R. The neonicotinoids thiacloprid, imidacloprid, and clothianidin affect the immunocompetence of honey bees (Apis mellifera L.). J. Insect Physiol. 2016, 86, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Brandt, A.; Grikscheidt, K.; Siede, R.; Grosse, R.; Meixner, M.D.; Büchler, R. Immunosuppression in honeybee queens by the neonicotinoids thiacloprid and clothianidin. Sci. Rep. 2017, 7, 4673. [Google Scholar] [CrossRef]
- Gauthier, M.; Aras, P.; Paquin, J.; Boily, M. Chronic exposure to imidacloprid or thiamethoxam neonicotinoid causes oxidative damages and alters carotenoid-retinoid levels in caged honey bees (Apis mellifera). Sci. Rep. 2018, 8, 16274. [Google Scholar] [CrossRef] [PubMed]
- Tesovnik, T.; Zorc, M.; Ristanic, M.; Glavinic, U.; Stevanovic, J.; Narat, M.; Stanimirovic, Z. Exposure of honey bee larvae to thiamethoxam and its interaction with Nosema ceranae infection in adult honey bees. Environ. Pollut. 2020, 256, 113443. [Google Scholar] [CrossRef]
- Tesovnik, T.; Cizelj, I.; Zorc, M.; Citar, M.; Bozic, J.; Glavan, G.; Narat, M. Immune related gene expression in worker honey bee (Apis mellifera carnica) pupae exposed to neonicotinoid thiamethoxam and Varroa mites (Varroa destructor). PLoS ONE 2017, 12, e0187079. [Google Scholar] [CrossRef]
- Aufauvre, J.; Misme-Aucouturier, B.; Vigues, B.; Texier, C.; Delbac, F.; Blot, N. Transcriptome analyses of the honeybee response to Nosema ceranae and insecticides. PLoS ONE 2014, 9, e91686. [Google Scholar] [CrossRef]
- McMenamin, A.J.; Brutscher, L.M.; Glenny, W.; Flenniken, M.L. Abiotic and biotic factors affecting the replication and pathogenicity of bee viruses. Curr. Opin. Insect Sci. 2016, 16, 14–21. [Google Scholar] [CrossRef]
- Vidau, C.; Diogon, M.; Aufauvre, J.; Fontbonne, R.; Vigues, B.; Brunet, J.L.; Texier, C.; Biron, D.G.; Blot, N.; El Alaoui, H.; et al. Exposure to sublethal doses of fipronil and thiacloprid highly increases mortality of honeybees previously infected by Nosema ceranae. PLoS ONE 2011, 6, e21550. [Google Scholar] [CrossRef] [PubMed]
- Kairo, G.; Biron, D.G.; Ben Abdelkader, F.; Bonnet, M.; Tchamitchian, S.; Cousin, M.; Dussaubat, C.; Benoit, B.; Kretzschmar, A.; Belzunces, L.P.; et al. Nosemaceranae, fipronil and their combination compromise honey bee reproduction via changes in male physiology. Sci. Rep. 2017, 7, 8556. [Google Scholar] [CrossRef] [PubMed]
- Dussaubat, C.; Maisonnasse, A.; Crauser, D.; Tchamitchian, S.; Bonnet, M.; Cousin, M.; Kretzschmar, A.; Brunet, J.L.; Le Conte, Y. Combined neonicotinoid pesticide and parasite stress alter honeybee queens’ physiology and survival. Sci. Rep. 2016, 6, 31430. [Google Scholar] [CrossRef] [PubMed]
- Coulon, M.; Schurr, F.; Martel, A.C.; Cougoule, N.; Begaud, A.; Mangoni, P.; Di Prisco, G.; Dalmon, A.; Alaux, C.; Ribiere-Chabert, M. Influence of chronic exposure to thiamethoxam and chronic bee paralysis virus on winter honey bees. PLoS ONE 2019, 14, e0220703. [Google Scholar] [CrossRef]
- Straub, L.; Williams, G.R.; Vidondo, B.; Khongphinitbunjong, K.; Retschnig, G.; Schneeberger, A.; Chantawannakul, P.; Dietemann, V.; Neumann, P. Neonicotinoids and ectoparasitic mites synergistically impact honeybees. Sci. Rep. 2019, 9, 8159. [Google Scholar] [CrossRef]
- Giglio, A.; Ammendola, A.; Battistella, S.; Naccarato, A.; Pallavicini, A.; Simeon, E.; Tagarelli, A.; Giulianini, P.G. Apis mellifera ligustica, Spinola 1806 as bioindicator for detecting environmental contamination: A preliminary study of heavy metal pollution in Trieste, Italy. Environ. Sci. Pollut. Res. 2017, 24, 659–665. [Google Scholar] [CrossRef]
- Raes, H.; Cornelis, R.; Rzeznik, U. Distribution, accumulation and depuration of administered lead in adult honeybees. Sci. Total Environ. 1992, 113, 269–279. [Google Scholar] [CrossRef]
- Polykretis, P.; Delfino, G.; Petrocelli, I.; Cervo, R.; Tanteri, G.; Montori, G.; Perito, B.; Branca, J.J.V.; Morucci, G.; Gulisano, M. Evidence of immunocompetence reduction induced by cadmium exposure in honey bees (Apis mellifera). Environ. Pollut. 2016, 218, 826–834. [Google Scholar] [CrossRef]
- Dabour, K.; Al Naggar, Y.; Masry, S.; Naiem, E.; Giesy, J.P. Cellular alterations in midgut cells of honey bee workers (Apis millefera L.) exposed to sublethal concentrations of CdO or PbO nanoparticles or their binary mixture. Sci. Total Environ. 2019, 651, 1356–1367. [Google Scholar] [CrossRef]
- Bové, H.; Bongaerts, E.; Slenders, E.; Bijnens, E.M.; Saenen, N.D.; Gyselaers, W.; Van Eyken, P.; Plusquin, M.; Roeffaers, M.B.J.; Ameloot, M.; et al. Ambient black carbon particles reach the fetal side of human placenta. Nat. Commun. 2019, 10, 3866. [Google Scholar] [CrossRef]
- Kelly, F.J.; Fussell, J.C. Linking ambient particulate matter pollution effects with oxidative biology and immune responses. Cell. Environ. Stressors Biol. Med. 2015, 1340, 84–94. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Kabir, E.; Kabir, S. A review on the human health impact of airborne particulate matter. Environ. Int. 2015, 74, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.S.; Duan, F.K.; He, K.B.; Ma, Y.L. Review on recent progress in observations, source identifications and countermeasures of PM2.5. Environ. Int. 2016, 86, 150–170. [Google Scholar] [CrossRef] [PubMed]
- Kelly, F.J.; Fussell, J.C. Size, source and chemical composition as determinants of toxicity attributable to ambient particulate matter. Atmos. Environ. 2012, 60, 504–526. [Google Scholar] [CrossRef]
- Zaric, N.M.; Deljanin, I.; Ilijevic, K.; Stanisavljevic, L.; Ristic, M.; Grzetic, I. Honeybees as sentinels of lead pollution: Spatio-temporal variations and source appointment using stable isotopes and Kohonen self-organizing maps. Sci. Total Environ. 2018, 642, 56–62. [Google Scholar] [CrossRef]
- Sheldon, M.; Pinion, C.; Klyza, J.; Zimeri, A.M. Pesticide contamination in central Kentucky urban honey: A pilot study. J. Environ. Health 2019, 82, 8–13. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feldhaar, H.; Otti, O. Pollutants and Their Interaction with Diseases of Social Hymenoptera. Insects 2020, 11, 153. https://doi.org/10.3390/insects11030153
Feldhaar H, Otti O. Pollutants and Their Interaction with Diseases of Social Hymenoptera. Insects. 2020; 11(3):153. https://doi.org/10.3390/insects11030153
Chicago/Turabian StyleFeldhaar, Heike, and Oliver Otti. 2020. "Pollutants and Their Interaction with Diseases of Social Hymenoptera" Insects 11, no. 3: 153. https://doi.org/10.3390/insects11030153
APA StyleFeldhaar, H., & Otti, O. (2020). Pollutants and Their Interaction with Diseases of Social Hymenoptera. Insects, 11(3), 153. https://doi.org/10.3390/insects11030153




