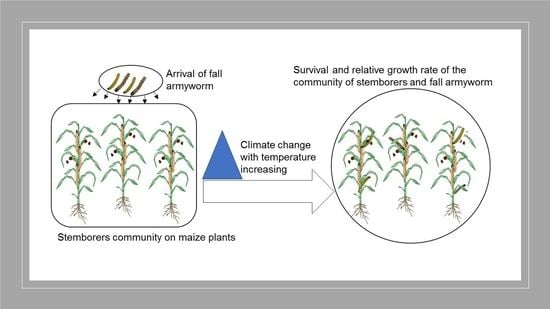
Influence of Temperature on the Interaction for Resource Utilization between Fall Armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), and a Community of Lepidopteran Maize Stemborers Larvae
Abstract
1. Introduction
2. Materials and Methods
2.1. Plants and Insects
2.2. Surrogate Stems
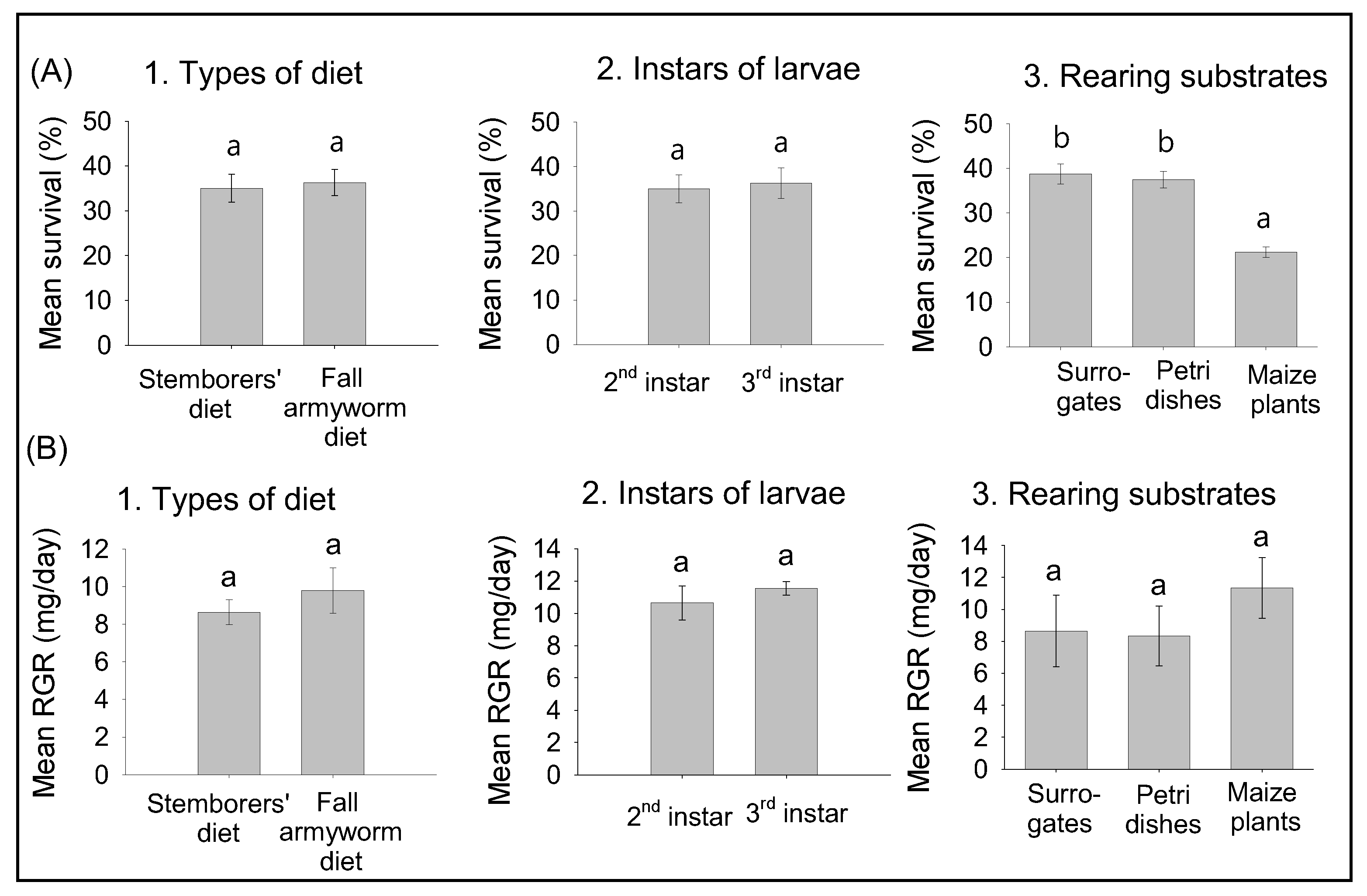
2.3. Preliminary Experiments: Effects of Diets, Instars, and Rearing Substrates on Survival and Relative Growth Rates of FAW Larvae
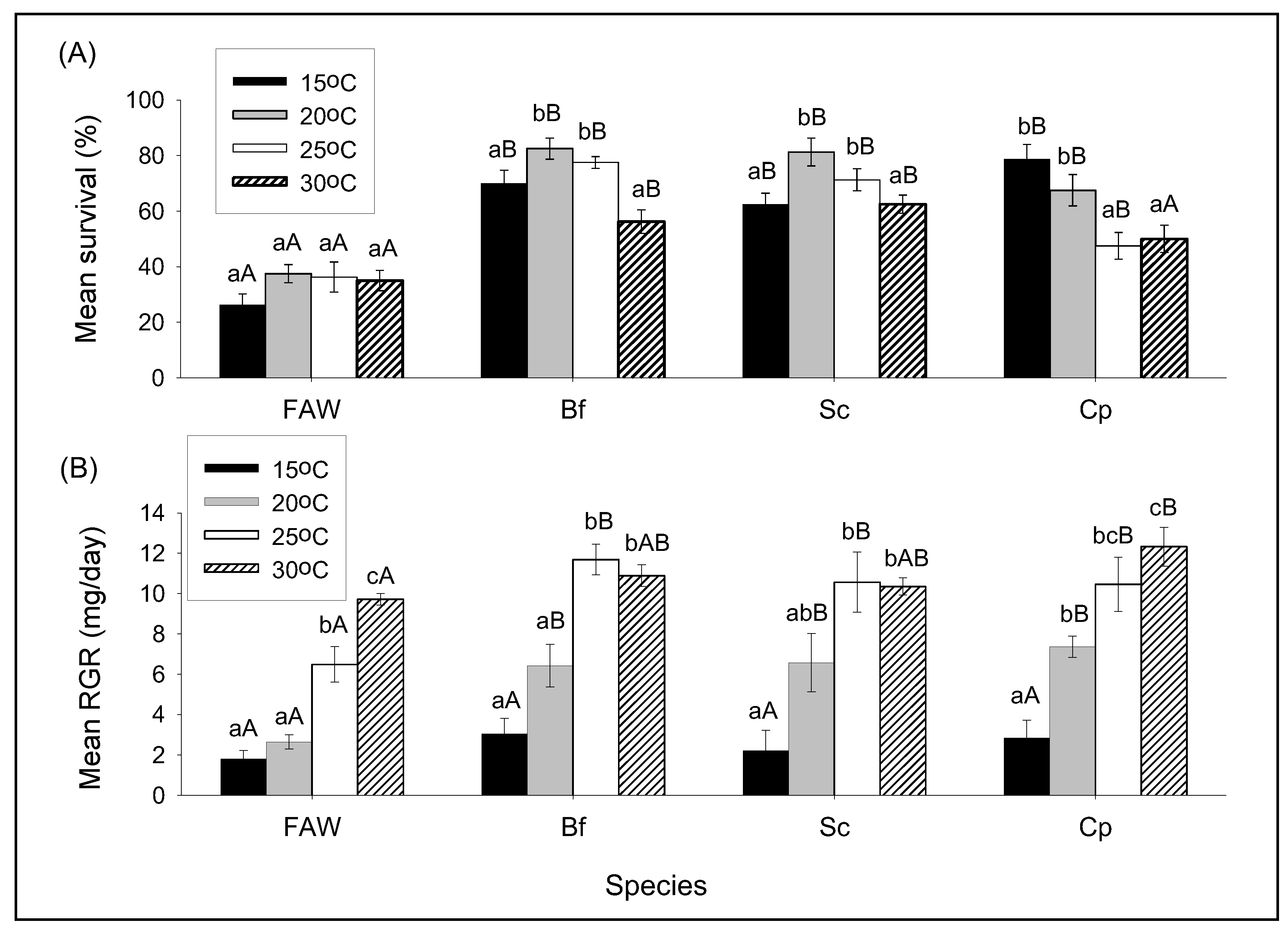
2.4. Influence of Different Constant Temperatures on Intra- and Interspecific Interactions within the FAW and a Community of Maize Stemborer Species
2.5. Data Analysis
3. Results
3.1. Effects of Diets, Larval Instars, and Rearing Substrates on Survival and RGR of FAW Larvae
3.2. Influence of Different Constant Temperatures on Intra- and Interspecific Interactions
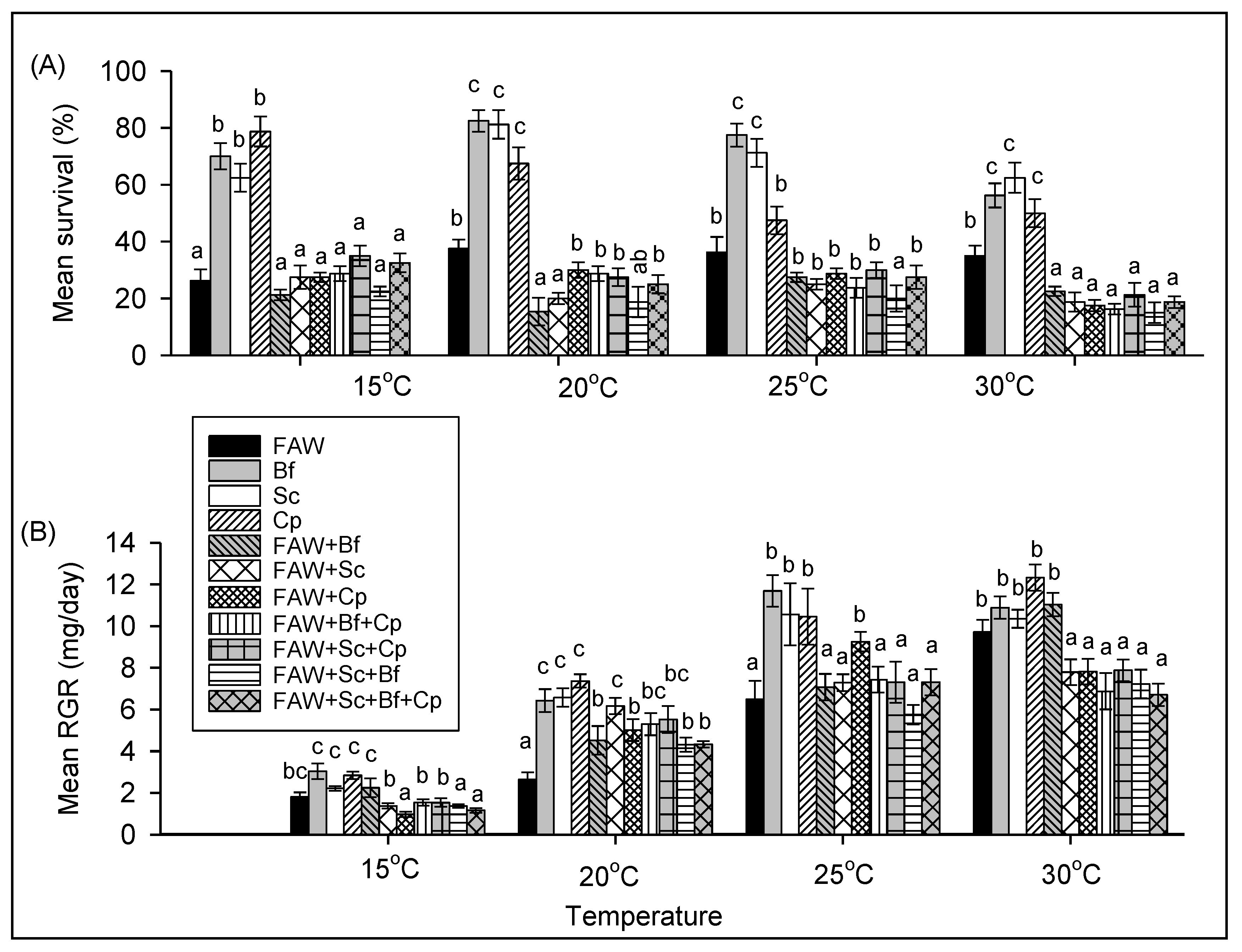
3.2.1. The Effect of Temperature on Survival and RGR of FAW and Stemborer Larvae
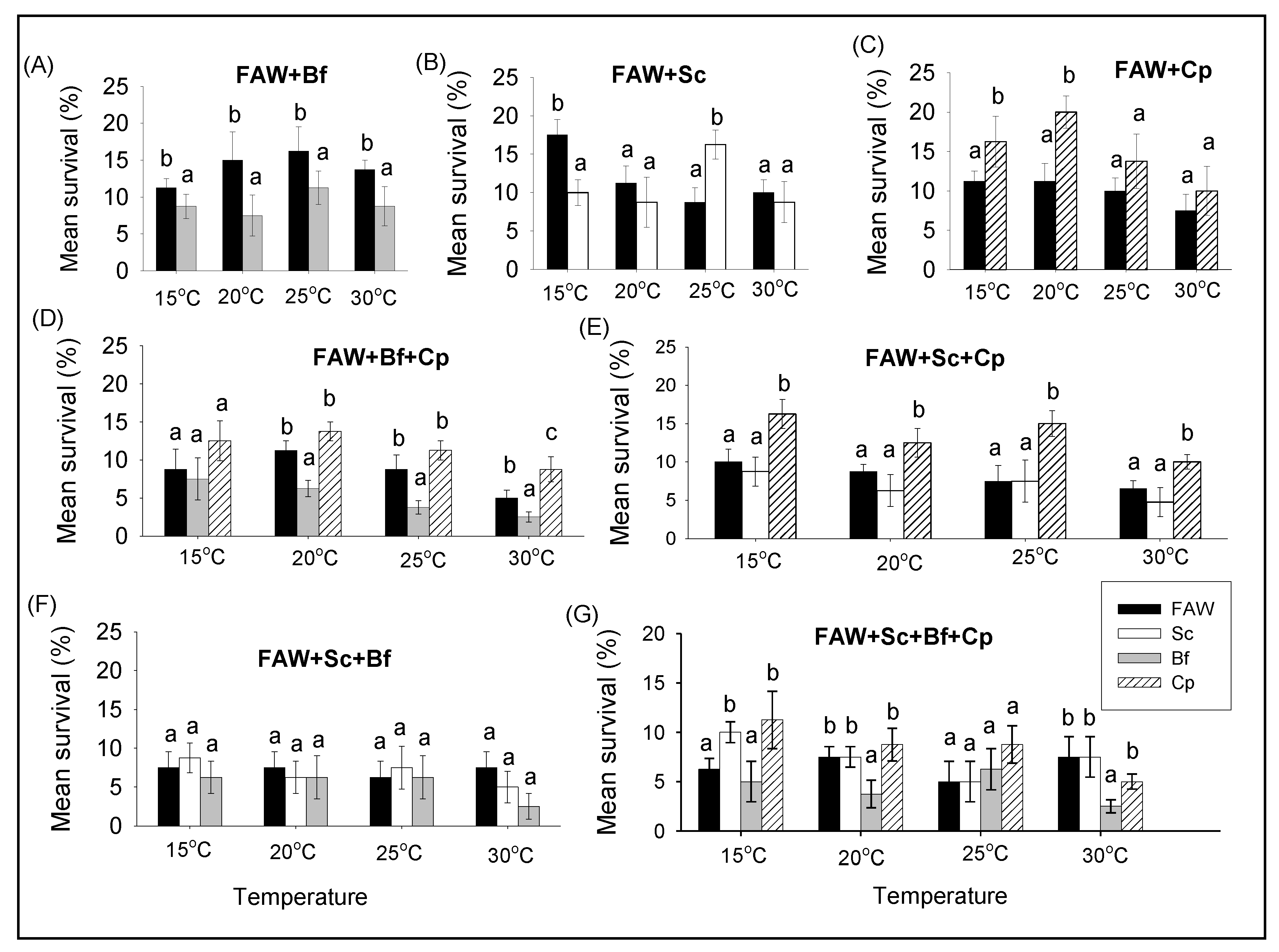
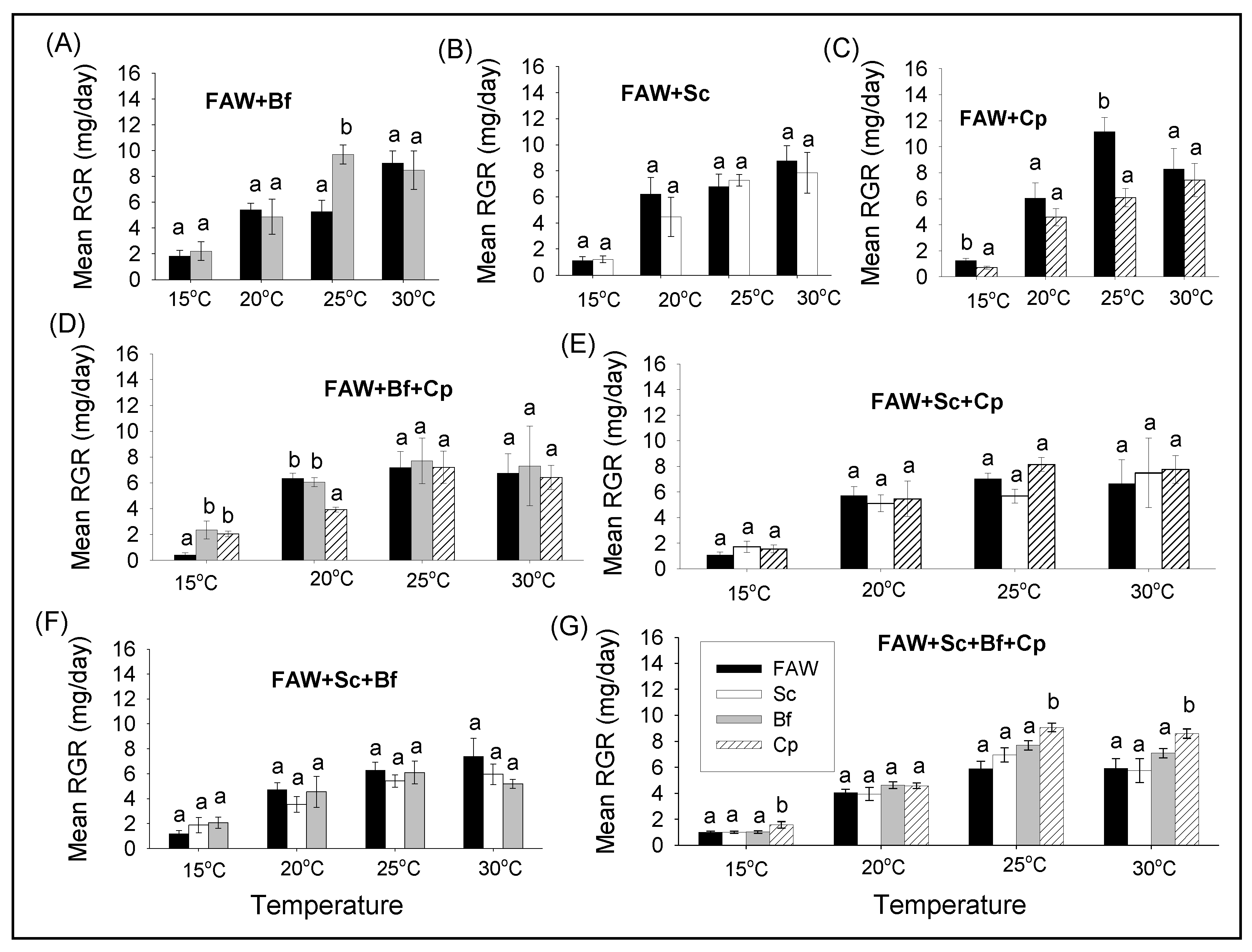
3.2.2. Comparison of Survival and RGR of FAW and Lepidopteran Stemborers in Multi-Species Combinations under Different Constant Temperatures
3.2.3. Comparison of Survival and RGR between Single and Multi-Species Combinations of Fall Armyworm and Lepidopteran Stemborers at Different Constant Temperatures
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kfir, R.; Overholt, W.A.; Khan, Z.R.; Polaszek, A. Biology and management of economicaly important lepidopteran cereal stem borers in Africa. Annu. Rev. Entomol. 2002, 47, 701–731. [Google Scholar] [CrossRef]
- Kipkoech, A.K.; Schulthess, F.; Yabann, W.K.; Maritim, H.K.; Mithofer, D. Biological control of cereal stem borers in Kenya: A cost benefit approach. Ann. Soc. Entomol. Fr. 2006, 42, 519–528. [Google Scholar] [CrossRef]
- Le Ru, B.P.; Ong’amo, G.O.; Moyal, P.; Ngala, L.; Musyoka, B.; Abdullah, Z.; Cugala, D.; Defabachew, B.; Haile, T.A.; Matama, T.K.; et al. Diversity of lepidopteran stem borers on monocotyledonous plants in eastern Africa and the islands of Madagascar and Zanzibar revisited. Bull. Entomol. Res. 2006, 96, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Polaszek, A. African Cereal Stem Borers: Economic Importance, Natural Enemies and Control; CAB International: Wallingford, UK, 1998; p. 592. [Google Scholar]
- De Groote, H. Maize losses from stemborers in Kenya. Insect Sci. Its Appl. 2002, 22, 89–96. [Google Scholar] [CrossRef]
- Overholt, W.A.; Maes, K.V.N.; Goebel, F.R. Field Guide to the Stemborer Larvae of Maize, Sorghum and Sugarcane in Eastern and Southern Africa; ICIPE Science Press: Nairobi, Kenya, 2001; p. 31. [Google Scholar]
- Cock, M.J.W.; Beseh, P.K.; Buddie, A.G.; Cafá, G.; Crozier, J. Molecular methods to detect Spodoptera frugiperda in Ghana, and implications for monitoring the spread of invasive species in developing countries. Sci. Rep. 2017, 7, 4103. [Google Scholar] [CrossRef] [PubMed]
- Goergen, G.; Kumar, P.L.; Sankung, S.B.; Togola, A.; Tamò, M. First report of outbreaks of the fall armyworm Spodoptera frugiperda (J E Smith) (Lepidoptera, Noctuidae), a new alien invasive pest in West and Central Africa. PLoS ONE 2016, 11, e0165632. [Google Scholar] [CrossRef]
- Sisay, B.; Simiyu, J.; Malusi, P.; Likhayo, P.; Mendesil, E.; Elibariki, N.; Wakgari, M.; Ayalew, G.; Tefera, T. First report of the fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), natural enemies from Africa. J. Appl. Entomol. 2018, 142, 800–804. [Google Scholar] [CrossRef]
- Sokame, B.M. Fall Armyworm Larval Interactions with the Stemborers Community Statuts Observations in Smallholder Maize Fields in Central Region of Kenya; ICIPE: Nairobi, Kenya, 2017. [Google Scholar]
- Sokame, B.M.; Musyoka, B. Observation of Older Fall Armyworm Larvae in the Bored Holes and Tunnels Left by Stemborers and the Feeding of the Larvae on Corn with Stemborer Larvae in Smallholder Maize Fields in Kenya; ICIPE: Nairobi, Kenya, 2017. [Google Scholar]
- Holloway, J.D. Noctuidae. In African Cereal Stem Borers: Economic Importance, Taxonomy, Natural Enemies and Control; Polaszek, A., Ed.; CAB International: Wallingford, UK, 1998; pp. 79–86. [Google Scholar]
- Ntiri, E.S.; Calatayud, P.A.; Berg Van Den, J.; Schulthess, F.; Le Ru, B.P. Influence of temperature on intra- and interspecific resource utilization within a community of lepidopteran maize stemborers. PLoS ONE 2016, 11, e148735. [Google Scholar] [CrossRef]
- Bentivenha, J.P.; Montezano, D.G.; Hunt, T.E.; Baldin, E.L.; Peterson, J.A.; Victor, V.S.; Pannuti, L.E.; Vélez, A.M.; Paula-Moraes, S.V. Intraguild interactions and behavior of Spodoptera frugiperda and Helicoverpa spp. on maize. Pest Manag. Sci. 2017, 73, 2244–2251. [Google Scholar] [CrossRef]
- Campos, M.S.; Zaldivar, P.C.J.; Ileana, M. Interspecific competition between Heliothis virescens (F.) and Spodoptera frugiperda Smith (Lepidoptera: Noctuidae) in the chickpea crop (Cicer arietinum L.). Revista Protección Vegetal 2013, 28, 171–177. [Google Scholar]
- Ntiri, E.S.; Calatayud, P.; Van Den Berg, J.; Le Ru, B.P. Spatio-temporal interactions between maize lepidopteran stemborer communities and possible implications from the recent invasion of Spodoptera frugiperda (Lepidoptera: Noctuidae) in Sub-Saharan Africa. Environ. Entomol. 2019, 48, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Onyango, F.O.; Ochieng’-Odero, J.E.R. Continuous rearing of the maize stem borer Busseola fusca on an artificial diet. Entomol. Exp. Appl. 1994, 73, 139–144. [Google Scholar] [CrossRef]
- Prasanna, B.M.; Huesing, J.E.; Eddy, R.; Peschke, V.M. Fall Armyworm in Africa: A Guide for Integrated Pest Management, 1st ed.; CIMMYT: Batan, Mexico, 2018. [Google Scholar]
- Da Silva, C.S.B.; Parra, J.R.P. New method for rearing Spodoptera frugiperda in laboratory shows that larval cannibalism is not obligatory. Rev. Bras. Entomol. 2013, 57, 347–349. [Google Scholar] [CrossRef]
- Ojeda-Avila, T.; Woods, H.A.; Raguso, R.A. Effects of dietary variation on growth, composition, and maturation of Manduca sexta (Sphingidae: Lepidoptera). J. Insect Physiol. 2003, 49, 293–306. [Google Scholar] [CrossRef]
- Lenth, R.V. Least-Squares Means: The R Package lsmeans. J. Stat. Softw. 2016, 69, 1–33. [Google Scholar] [CrossRef]
- Verhoeven, K.J.F.; Simonsen, K.L.; McIntyre, L.M. Implementing false discovery rate control: Increasing your power. Oikos 2005, 108, 643–647. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 27 January 2018).
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous Inference in General Parametric Models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef] [PubMed]
- Venables, W.N.; Ripley, B.D. Modern Applied Statistics with S, 4th ed.; Springer: New York, NY, USA, 2002; ISBN 0-387-95457-0. [Google Scholar]
- Hope, R.M. Rmisc: Ryan Miscellaneous. 2016. Available online: https://cran.r-project.org/web/packages/Rmisc/Rmisc.pdf (accessed on 13 November 2019).
- Fox, J.; Weisberg, S. An R Companion to Applied Regression, 3rd ed.; Sage: Thousand Oaks, CA, USA, 2019; Available online: https://socialsciences.mcmaster.ca/jfox/Books/Companion/ (accessed on 13 November 2019).
- Ogle, D.H.; Wheeler, P.; Dinno, A. FSA: Fisheries Stock Analysis; Harper & Row Publishers: New York, NY, USA; Leslie, MI, USA, 2019; R Package Version 0.8.26; Available online: https://github.com/droglenc/FSA (accessed on 24 October 2019).
- Revelle, W. psych: Procedures for Personality and Psychological Research; Version = 1.8.12.; Northwestern University: Evanston, IL, USA, 2018; Available online: https://CRAN.R-project.org/package=psych (accessed on 24 October 2019).
- Graves, S.; Piepho, H.-P.; Selzer, L. Visualizations of Paired Comparisons. 2015. Available online: https://cran.r-project.org/web/packages/multcompView/multcompView.pdf (accessed on 15 November 2019).
- Sarmento, R.D.A.; Aguiar, D.S.W.R.; de Aguiar, R.A.S.S.; Viera, S.M.J.; de Oliveira, H.G.; Holtz, A.M. Biology review, occurence and control of Spodoptera frugiperda Smith (Lepidoptera: Noctuidae) in corn in Brazil. Biosci. J. 2002, 18, 41–48. [Google Scholar]
- Farias, S.P.R.; Barbosa, J.C.; Busoli, A.C. Sequencial sampling based on taylor’s power law for the survey of Spodoptera frugiperda for the corn crop. Sci. Agric. 2001, 58, 395–399. [Google Scholar] [CrossRef]
- Da Silva, M. Factors limiting the efficiency of insecticides to control Spodoptera frugiperda Smith in maize. Ciência Rural 1999, 29, 383–387. [Google Scholar] [CrossRef][Green Version]
- Chapman, J.W.; Williams, T.; Martínez, A.M.; Cisneros, J. Does cannibalism in Spodoptera frugiperda (Lepidoptera: Noctuidae) reduce the risk of predation? Behav. Ecol. Sociobiol. 2000, 48, 321–327. [Google Scholar] [CrossRef]
- De Polanía, I.Z.; Maldonado, H.A.; Cruz, R.M.; Sánchez, J.D. Spodoptera frugiperda: Response of different populations to the Cry1Ab toxin. Rev. Colomb. Entomol. 2009, 35, 34–41. [Google Scholar]
- Goussain, M.M.; Moraes, J.C.; Carvalho, J.G.; Nogueira, N.L.; Rossi, E.M.L. Effect of silicon application on corn plants upon the biological development of the fall Armyworm Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae). Neotrop. Entomol. 2002, 31, 305–310. [Google Scholar] [CrossRef]
- Chapman, J.W.; Williams, T.; Escribano, A.; Caballero, P.; Cave, R.D.; Goulson, D. Age-related cannibalism and horizontal transmission of a nuclear polyhedrosis virus in larval Spodoptera frugiperda. Ecol. Entomol. 1999, 24, 268–275. [Google Scholar] [CrossRef]
- Chapman, J.W.; Williams, T.; Escribanoc, A.; Caballero, P.; Cave, R.D. Fitness consequences of cannibalism in the fall armyworm, Spodoptera frugiperda. Behav. Ecol. 1999, 10, 298–303. [Google Scholar] [CrossRef]
- Breden, F.; Chippendale, G.M. Effect of larval density and cannibalism on growth and development of the Southwestern corn borer, Diatraea grandiosella, and the European corn borer, Ostrinia nubilalis (Lepidoptera: Pyralidae). J. Kans. Entomol. Soc. 1989, 62, 307–315. [Google Scholar]
- Bonhof, M.J.; Overholt, W.A.Z. Impact of solar radiation, rainfall and cannibalism on disappearance of maize stemborers in Kenya. Insect Sci. Appl. 2001, 21, 403–407. [Google Scholar] [CrossRef]
- Violle, C.; Nemergut, D.R.; Pu, Z.; Jiang, L. Phylogenetic limiting similarity and competitive exclusion. Ecol. Lett. 2011, 14, 782–787. [Google Scholar] [CrossRef]
- Denno, R.F.; Mcclure, M.S.; Ott, J.R. Interspecific interactions in phytophagous insects: Competition Reexamined and Resurrected. Annu. Rev. Entomol. 1995, 40, 297–331. [Google Scholar] [CrossRef]
- Shi, S.; He, Y.; Ji, X.; Jiang, M.; Cheng, J. Do rice water weevils and rice stem borers compete when sharing a host plant? J. Zhejiang Univ. Sci. B 2008, 9, 572–577. [Google Scholar] [CrossRef][Green Version]
- Pedruski, M.T.; Fussmann, G.F.; Gonzalez, A. Predicting the outcome of competition when fitness inequality is variable. R. Soc. Open Sci. 2015, 2, 150274. [Google Scholar] [CrossRef]
- Tilman, D.; Kilham, S.S.; Kilham, P. Phytoplankton community ecology: The role of limiting nutrients. Annu. Rev. Ecol. Syst. 1982, 13, 349–372. [Google Scholar] [CrossRef]
- Inbar, M. Interspecific competition among phloem-feeding insects mediated by induced host-plant sinksi. Ecology 1995, 76, 1506–1515. [Google Scholar] [CrossRef]
- Kaplan, I.; Denno, R.F. Interspecific interactions in phytophagous insects revisited: A quantitative assessment of competition theory. Ecol. Lett. 2007, 10, 977–994. [Google Scholar] [CrossRef]
- Nooten, S.S.; Andrew, N.R.; Hughes, L. Potential impacts of climate change on insect communities: A transplant experiment. PLoS ONE 2014, 9, e85987. [Google Scholar] [CrossRef]
- Lemoine, N.P.; Burkepile, D.E.; Parker, J.D. Variable effects of temperature on insect herbivory. PeerJ 2014, 2, e376. [Google Scholar] [CrossRef]
- Johnson, C.A.; Coutinho, R.M.; Berlin, E.; Dolphin, K.E.; Heyer, J.; Kim, B.; Leung, A.; Sabellon, J.L.; Amarasekare, P. Effects of temperature and resource variation on insect population dynamics: The bordered plant bug as a case study. Funct. Ecol. 2016, 30, 1122–1131. [Google Scholar] [CrossRef]
- Howe, R. Temperature effects on embryonic development in insects. Annu. Rev. Entomol. 1967, 12, 15–42. [Google Scholar] [CrossRef]
- Bale, J.S.; Masters, G.J.; Hodkinson, I.D.; Awmack, C.; Bezemer, T.M.; Brown, V.K.; Butterfield, J.; Buse, A.; Coulson, J.C.; Farrar, J.; et al. Herbivory in global climate change research: Direct effects of rising temperature on insect herbivores. Glob. Chang. Biol. 2002, 8, 1–16. [Google Scholar] [CrossRef]
- Sporleder, M.; Kroschel, J.; Quispe, M.R.G.; Lagnaoui, A. A temperature-based simulation model for the potato tuberworm, Phthorimaea operculella Zeller (Lepidoptera; Gelechiidae). Physiol. Ecol. 2004, 33, 477–486. [Google Scholar] [CrossRef]
- Hodkinson, I.D. Terrestrial insects along elevation gradients: Species and community responses to altitude. Biol. Rev. 2005, 80, 489–513. [Google Scholar] [CrossRef] [PubMed]
- Dangles, O.; Carpio, C.; Barragan, A.; Zeddam, J.-L.; Silvain, J. Temperature as a key driver of ecological sorting among invasive pest species in the tropical andes. Ecol. Appl. 2008, 18, 1795–1809. [Google Scholar] [CrossRef] [PubMed]
- Speight, M.R.; Hunter, M.D.; Watt, A.D. Ecology of Insects: Concepts and Applications, 2nd ed.; Environmental Entomology, Entomological Society of America: Lanham, MD, USA, 2008; Available online: https://doi.org/10.1603/022.038.0448 (accessed on 12 September 2018).
- Stokes, K.; Stiling, P. Indirect competitive effects of stemborers on a gall community. Entomol. Exp. Appl. 2015, 154, 23–27. [Google Scholar] [CrossRef]
- Guofa, Z.; Overholt, W.A.; Mochiah, M.B. Changes in the distribution of lepidopteran maize stemborers in Kenya from the 1950s to 1990s. Int. J. Trop. Insect Sci. 2001, 21, 395–402. [Google Scholar] [CrossRef]
- Ong’amo, G.; Le Ru, B.; Dupas, S.; Moyal, P.; Muchugu, E.; Calatayud, P.-A.; Silvain, J.-F. The role of wild host plants in the abundance of lepidopteran stem borers along altitudinal gradient in Kenya. Ann. Soc. Entomol. Fr. 2006, 42, 363–370. [Google Scholar] [CrossRef]
- Ong’amo, G.; Le Ru, B.; Dupas, S.; Moyal, P.; Calatayud, P.-A.; Silvain, J.-F. Distribution, pest status and agro-climatic preferences of lepidopteran stem borers of maize in Kenya. Ann. Soc. Entomol. Fr. 2006, 42, 171–177. [Google Scholar] [CrossRef]
| Between Temperatures | Statistic Values | Between Species | Statistic Values | ||
|---|---|---|---|---|---|
| O.R. (95% CI) | p-Value | O.R. (95% CI) | p-Value | ||
| FAW | 1.11 (0.90–1.37) | 0.29 | 15 °C | 1.95 (1.57–2.45) | <0.0001 |
| Bf | 0.79 (0.63–0.98) | 0.04 | 20 °C | 1.51 (1.22–1.89) | 0.0001 |
| Sc | 0.95 (0.77–1.18) | 0.66 | 25 °C | 1.11 (0.91–1.36) | 0.02 |
| Cp | 0.62 (0.50–0.77) | <0.0001 | 30 °C | 1.22 (1.01–1.50) | 0.04 |
| Between Temperatures | Statistic Values | Between Species | Statistic Values | ||
|---|---|---|---|---|---|
| F | p-Value | F | p-Value | ||
| FAW | 19.14 | <0.0001 | 15 °C | 0.35 | 0.78 |
| Bf | 14.09 | <0.0001 | 20 °C | 3.81 | 0.01 |
| Sc | 8.36 | 0.0002 | 25 °C | 3.66 | 0.02 |
| Cp | 13.01 | <0.0001 | 30 °C | 2.80 | 0.04 |
| Temperature | FAW+Bf | FAW+Sc | FAW+Cp | FAW+Bf+Cp | ||||
|---|---|---|---|---|---|---|---|---|
| O.R. (95% CI) | p-Value | O.R. (95% CI) | p-Value | O.R. (95% CI) | p-Value | O.R. (95% CI) | p-Value | |
| 15 °C | 1.24 (0.82–1.89) | 0.03 | 0.52 (0.19–1.30) | 0.01 | 1.53 (0.61–2.93) | 0.03 | 1.24 (0.73–2.14) | 0.42 |
| 20 °C | 2.17 (1.04–3.54) | 0.04 | 0.75 (0.25–2.13) | 0.59 | 1.97 (0.82–2.95) | 0.01 | 1.14 (0.68–1.92) | 0.04 |
| 25 °C | 0.65 (0.25–1.61) | 0.03 | 2.02 (0.78–5.66) | 0.02 | 1.43 (0.54–3.90) | 0.46 | 1.18 (0.66–2.14) | 0.03 |
| 30 °C | 0.60 (0.21–1.61) | 0.04 | 0.86 (0.28–2.52) | 0.78 | 1.37 (0.45–4.34) | 0.57 | 1.45 (0.72–2.06 | 0.03 |
| Temperature | FAW+Sc+Cp | FAW+Sc+Bf | FAW+Sc+Bf+Cp | |||||
| O.R. (95% CI) | p-Value | O.R. (95% CI) | p-Value | O.R. (95% CI) | p-Value | |||
| 15 °C | 1.35 (0.83–2.25) | 0.02 | 0.91 (0.50–1.65) | 0.76 | 1.14 (0.79–1.65) | 0.02 | ||
| 20 °C | 1.25 (0.73–2.19) | 0.03 | 0.90 (0.47–1.69) | 0.75 | 1.00 (0.67–1.47) | 0.03 | ||
| 25 °C | 1.53 (0.90–2.67) | 0.01 | 1.00 (0.53–1.87) | 1.00 | 1.24 (0.82–1.89) | 0.30 | ||
| 30 °C | 1.21 (0.65–2.26) | 0.03 | 0.57 (0.25–1.19) | 0.15 | 0.78 (0.50–1.20) | 0.02 | ||
| Temperature | FAW+Bf | FAW+Sc | FAW+Cp | FAW+Bf+Cp | ||||
|---|---|---|---|---|---|---|---|---|
| F | p-Value | F | P-value | F | p-Value | F | p-Value | |
| 15 °C | 0.22 | 0.64 | 0.04 | 0.83 | 6.61 | 0.02 | 6.67 | 0.007 |
| 20 °C | 0.16 | 0.69 | 1.78 | 0.20 | 1.32 | 0.26 | 16.75 | <0.0001 |
| 25 °C | 14.87 | 0.001 | 0.27 | 0.61 | 14.38 | 0.002 | 0.13 | 0.87 |
| 30 °C | 0.11 | 0.74 | 0.23 | 0.63 | 0.16 | 0.69 | 0.06 | 0.93 |
| Temperature | FAW+Sc+Cp | FAW+Sc+Bf | FAW+Sc+Bf+Cp | |||||
| F | p-Value | F | p-Value | F | p-Value | |||
| 15 °C | 0.96 | 0.39 | 1.43 | 0.28 | 3.70 | 0.03 | ||
| 20 °C | 0.06 | 0.94 | 0.68 | 0.52 | 1.04 | 0.40 | ||
| 25 °C | 0.99 | 0.39 | 0.46 | 0.63 | 4.86 | 0.04 | ||
| 30 °C | 0.13 | 0.87 | 0.57 | 0.58 | 4.08 | 0.04 | ||
| Comparisons | 15 °C | 20 °C | 25 °C | 30 °C | ||||
|---|---|---|---|---|---|---|---|---|
| O.R. (95% CI) | p-Value | O.R. (95% CI) | p-Value | O.R. (95% CI) | p-Value | O.R. (95% CI) | p-Value | |
| FAW vs. FAW+Bf | 0.75 (0.36–1.57) | 0.45 | 0.48 (0.23–0.95) | 0.04 | 0.74 (0.38–1.45) | 0.39 | 0.53 (0.36–0.93) | 0.008 |
| FAW vs. FAW+Sc | 1.06 (0.52–2.15) | 0.85 | 0.41 (0.20–0.83) | 0.01 | 0.58 (0.29–1.15) | 0.12 | 0.42 (0.20–0.87) | 0.02 |
| FAW vs. FAW+Cp | 1.06 (0.52–2.15) | 0.85 | 0.71 (0.36–1.37) | 0.31 | 0.70 (0.36–1.37) | 0.31 | 0.39 (0.18–0.81) | 0.01 |
| FAW vs. FAW+Bf+Cp | 1.13 (0.56–2.28 | 0.72 | 0.67 (0.34–1.30) | 0.24 | 0.54 (0.27–1.08) | 0.08 | 0.36 (0.16–0.75) | 0.007 |
| FAW vs. FAW+Sc+Cp | 1.51 (0.77–3.00) | 0.23 | 0.63 (0.32–1.22) | 0.17 | 0.75 (0.38–1.45) | 0.40 | 0.50 (0.24–1.00) | 0.05 |
| FAW vs. FAW+Sc+Bf | 0.81 (0.39–1.68) | 0.58 | 0.38 (0.18–0.78) | 0.009 | 0.43 (0.21–0.88) | 0.02 | 0.32 (0.14–0.69) | 0.004 |
| FAW vs. FAW+Sc+Bf+Cp | 1.26 (0.64–2.51) | 0.49 | 0.55 (0.27–1.08) | 0.08 | 0.66 (0.33–1.29) | 0.23 | 0.42 (0.20–0.87) | 0.02 |
| Bf vs. FAW+Bf | 0.11 (0.05–0.23) | <0.0001 | 0.06 (0.02–0.13) | <0.0001 | 0.11 (0.05–0.23) | <0.0001 | 0.22 (0.11–0.44) | <0.0001 |
| Bf vs. FAW+Bf+Sc | 0.12 (0.05–0.24) | <0.0001 | 0.04 (0.02–0.10) | <0.0001 | 0.07 (0.03–0.15) | <0.0001 | 0.13 (0.06–0.28) | <0.0001 |
| Bf vs. FAW+Bf+Cp | 0.17 (0.08–0.33) | <0.0001 | 0.09 (0.04–0.18) | <0.0001 | 0.09 (0.04–0.18) | <0.0001 | 0.15 (0.06–0.30) | <0.0001 |
| Bf vs. FAW+Bf+Sc+Cp | 0.20 (0.10–0.39) | <0.0001 | 0.07 (0.03–0.14) | <0.0001 | 0.11 (0.05–0.22) | <0.0001 | 0.17 (0.08–0.35) | <0.0001 |
| Sc vs. FAW+Sc | 0.22 (0.11–0.43) | <0.0001 | 0.05 (0.02–0.12) | <0.0001 | 0.13 (0.06–0.26) | <0.0001 | 0.13 (0.06–0.27) | <0.0001 |
| Sc vs. FAW+Bf+Sc | 0.17 (0.08–0.34) | <0.0001 | 0.05 (0.02–0.11) | <0.0001 | 0.10 (0.04–0.20) | <0.0001 | 0.10 (0.04–0.22) | <0.0001 |
| Sc vs. FAW+Sc+Cp | 0.32 (0.16–0.61) | 0.0005 | 0.08 (0.04–0.18) | <0.0001 | 0.17 (0.08–0.33) | <0.0001 | 0.16 (0.07–0.32) | <0.0001 |
| Sc vs. FAW+Bf+Sc+Cp | 0.28 (0.14–0.54) | 0.0001 | 0.07 (0.03–0.15) | <0.0001 | 0.15 (0.07–0.30) | <0.0001 | 0.13 (0.06–0.27) | <0.0001 |
| Cp vs. FAW+Cp | 0.10 (0.04–0.20) | <0.0001 | 0.20 (0.10–0.39) | <0.0001 | 0.44 (0.22–0.85) | 0.01 | 0.21 (0.10–0.42) | <0.0001 |
| Cp vs. FAW+Bf+Cp | 0.10 (0.05–0.21) | <0.0001 | 0.20 (0.10–0.39) | <0.0001 | 0.34 (0.17–0.67) | 0.002 | 0.19 (0.08–0.39) | <0.0001 |
| Cp vs. FAW+Sc+Cp | 0.14 (0.07–0.28) | <0.0001 | 0.18 (0.09–0.35) | <0.0001 | 0.47 (0.24–0.90) | 0.024 | 0.26 (0.13–0.53) | 0.0002 |
| Cp vs. FAW+Bf+Sc+Cp | 0.12 (0.06–0.25) | <0.0001 | 0.16 (0.07–0.31) | <0.0001 | 0.41 (0.21–0.80) | 0.009 | 0.23 (0.11–0.46) | <0.0001 |
| Comparisons | 15 °C | 20 °C | 25 °C | 30 °C | ||||
|---|---|---|---|---|---|---|---|---|
| F | p-Value | F | p-Value | F | p-Value | F | p-Value | |
| FAW vs. FAW+Bf | 0.79 | 0.38 | 6.34 | 0.02 | 0.59 | 0.45 | 5.87 | 0.02 |
| FAW vs. FAW+Sc | 0.34 | 0.56 | 9.69 | 0.006 | 0.09 | 0.76 | 12.03 | 0.002 |
| FAW vs. FAW+Cp | 13.25 | 0.002 | 14.27 | 0.001 | 24.34 | 0.0001 | 7.77 | 0.01 |
| FAW vs. FAW+Bf+Cp | 0.30 | 0.59 | 17.91 | 0.0005 | 0.1.79 | 0.19 | 10.68 | 0.004 |
| FAW vs. FAW+Sc+Cp | 1.33 | 0.30 | 15.99 | 0.0009 | 0.57 | 0.46 | 7.14 | 0.01 |
| FAW vs. FAW+Sc+Bf | 6.34 | 0.02 | 21.83 | 0.0002 | 1.73 | 0.20 | 12.57 | 0.002 |
| FAW vs. FAW+Sc+Bf+Cp | 12.13 | 0.002 | 19.61 | 0.0003 | 1.23 | 0.28 | 37.86 | <0.0001 |
| Bf vs. FAW+Bf | 0.75 | 0.39 | 9.98 | 0.005 | 22.15 | 0.0001 | 0.008 | 0.92 |
| Bf vs. FAW+Bf+Sc | 64.26 | <0.0001 | 14.91 | 0.001 | 39.72 | <0.0001 | 12.5 | 0.002 |
| Bf vs. FAW+Bf+Cp | 67.61 | <0.0001 | 2.89 | 0.10 | 18.45 | 0.0004 | 21.73 | 0.0002 |
| Bf vs. FAW+Bf+Sc+Cp | 80.87 | <0.0001 | 23.43 | 0.0001 | 19.74 | 0.0003 | 42.83 | <0.0001 |
| Sc vs. FAW+Sc | 22.05 | 0.0001 | 0.12 | 0.92 | 4.51 | 0.04 | 16.44 | 0.0008 |
| Sc vs. FAW+Bf+Sc | 34.72 | <0.0001 | 17.68 | 0.0006 | 37.16 | <0.0001 | 9.00 | 0.008 |
| Sc vs. FAW+Sc+Cp | 8.65 | 0.008 | 3.61 | 0.07 | 19.4 | 0.0003 | 5.32 | 0.03 |
| Sc vs. FAW+Bf+Sc+Cp | 43.41 | <0.0001 | 28.12 | <0.0001 | 13.39 | 0.001 | 31.78 | <0.0001 |
| Cp vs. FAW+Cp | 108.1 | <0.0001 | 13.95 | 0.001 | 3.40 | 0.08 | 11.43 | 0.003 |
| Cp vs. FAW+Bf+Cp | 8.18 | 0.01 | 10.72 | 0.004 | 16.01 | 0.0009 | 16.82 | 0.0008 |
| Cp vs. FAW+Sc+Cp | 29.99 | <0.0001 | 6.48 | 0.02 | 8.42 | 0.009 | 9.15 | 0.008 |
| Cp vs. FAW+Bf+Sc+Cp | 95.69 | <0.0001 | 61.09 | <0.0001 | 16.4 | 0.0007 | 25.93 | <0.0001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sokame, B.M.; Rebaudo, F.; Malusi, P.; Subramanian, S.; Kilalo, D.C.; Juma, G.; Calatayud, P.-A. Influence of Temperature on the Interaction for Resource Utilization between Fall Armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), and a Community of Lepidopteran Maize Stemborers Larvae. Insects 2020, 11, 73. https://doi.org/10.3390/insects11020073
Sokame BM, Rebaudo F, Malusi P, Subramanian S, Kilalo DC, Juma G, Calatayud P-A. Influence of Temperature on the Interaction for Resource Utilization between Fall Armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), and a Community of Lepidopteran Maize Stemborers Larvae. Insects. 2020; 11(2):73. https://doi.org/10.3390/insects11020073
Chicago/Turabian StyleSokame, Bonoukpoè Mawuko, François Rebaudo, Peter Malusi, Sevgan Subramanian, Dora Chao Kilalo, Gerald Juma, and Paul-André Calatayud. 2020. "Influence of Temperature on the Interaction for Resource Utilization between Fall Armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), and a Community of Lepidopteran Maize Stemborers Larvae" Insects 11, no. 2: 73. https://doi.org/10.3390/insects11020073
APA StyleSokame, B. M., Rebaudo, F., Malusi, P., Subramanian, S., Kilalo, D. C., Juma, G., & Calatayud, P.-A. (2020). Influence of Temperature on the Interaction for Resource Utilization between Fall Armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), and a Community of Lepidopteran Maize Stemborers Larvae. Insects, 11(2), 73. https://doi.org/10.3390/insects11020073