Genetic Diversity of Mitochondrial DNA of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) Associated with Cassava and the Occurrence of Cassava Mosaic Disease in Zambia
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling Methodology
2.3. Extraction of Whitefly DNA, PCR and Sequencing
2.3.1. 2013 Samples
2.3.2. 2015 Samples
2.4. Data Analysis
2.5. Phylogenetic and Sequence Analysis
2.6. Population Genetic Analysis
3. Results
3.1. CMD Symptoms and Sooty Mold
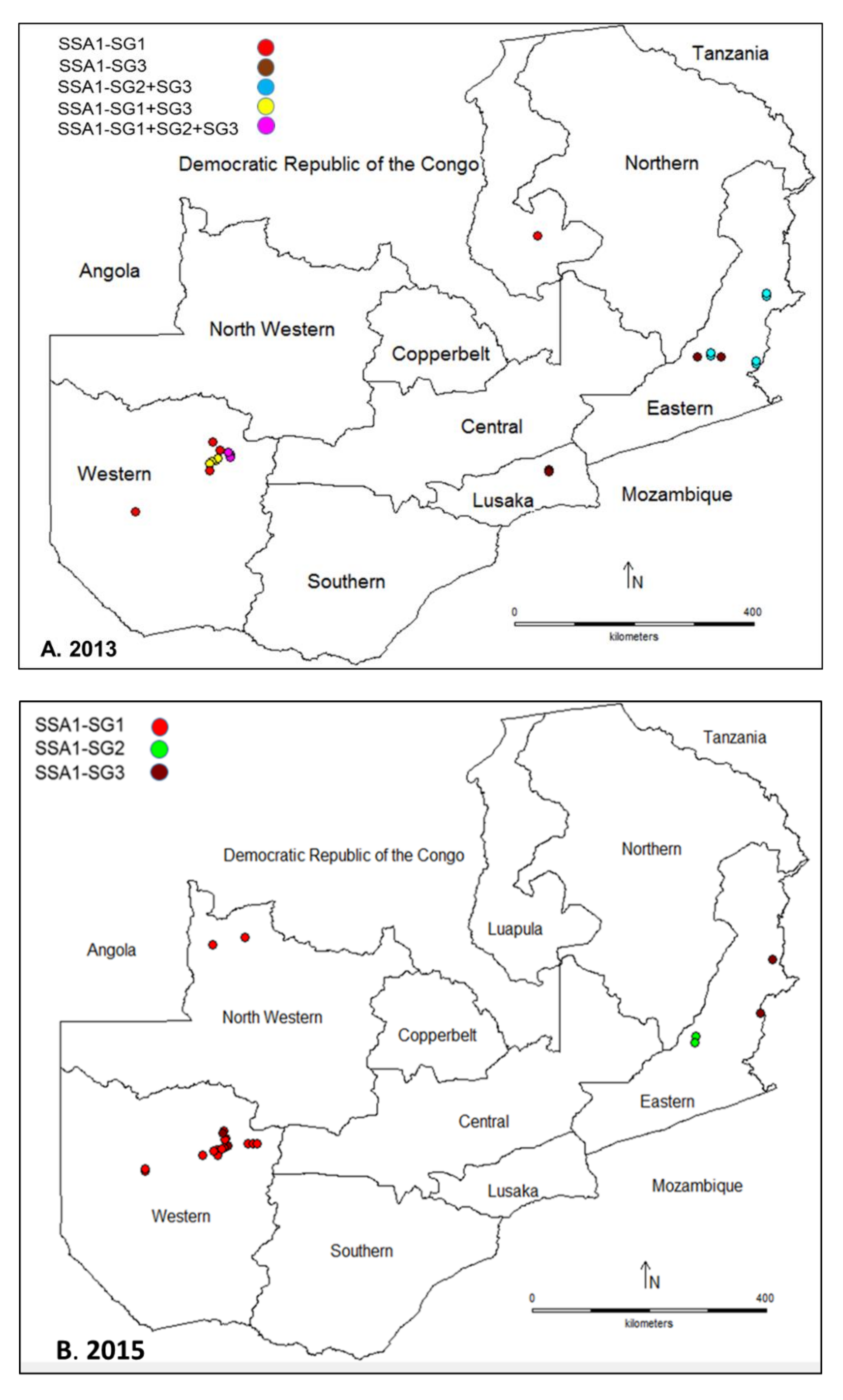
3.2. Phylogenetic Analysis, Evolutionary Divergence and Geographical Distribution of B. tabaci mtCOI
3.3. Diversity Indices
3.4. CMD Incidence, Symptom Severity and Whitefly Abundance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brown, J.; Frohlich, D.; Rosell, R. The sweetpotato or silverleaf whiteflies: Biotypes of Bemisia tabaci (Genn.), or a species complex? Annu. Rev. Entomol. 1995, 40, 511–534. [Google Scholar] [CrossRef]
- Jones, D.R. Plant Viruses Transmitted by Whiteflies. Eur. J. Plant Pathol. 2003, 109, 195–219. [Google Scholar] [CrossRef]
- Polston, J.E.; de Barro, P.; Boykin, L.M. Transmission specificities of plant viruses with the newly identified species of the Bemisia tabaci species complex. Pest Manag. Sci. 2014, 70, 1547–1552. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.K. Molecular markers for the identification and global tracking of whitefly vector–Begomovirus complexes. Virus Res. 2000, 71, 233–260. [Google Scholar] [CrossRef]
- De Barro, P.J.; Driver, F.; Trueman, J.W.; Curran, J. Phylogenetic Relationships of World Populations of Bemisia tabaci (Gennadius) Using Ribosomal ITS1. Mol. Phylogenetics Evol. 2000, 16, 29–36. [Google Scholar] [CrossRef]
- Perring, T.M. The Bemisia tabaci species complex. Crop. Prot. 2001, 20, 725–737. [Google Scholar] [CrossRef]
- Rosell, R.C.; Bedford, I.D.; Frohlich, D.R.; Gill, R.J.; Brown, J.K.; Markham, P.G. Analysis of Morphological Variation in Distinct Populations of Bemisia tabaci (Homoptera: Aleyrodidae). Ann. Entomol. Soc. Am. 1997, 90, 575–589. [Google Scholar] [CrossRef]
- Calvert, L.A.; Cuervo, M.; Arroyave, J.A.; Constantino, L.M.; Bellotti, A.; Frohlich, D. Morphological and Mitochondrial DNA Marker Analyses of Whiteflies (Homoptera: Aleyrodidae) Colonizing Cassava and Beans in Colombia. Ann. Entomol. Soc. Am. 2001, 94, 512–519. [Google Scholar] [CrossRef] [Green Version]
- Maruthi, M.N.; Colvin, J.; Seal, S.; Gibson, G.; Cooper, J. Co-adaptation between cassava mosaic geminiviruses and their local vector populations. Virus Res. 2002, 86, 71–85. [Google Scholar] [CrossRef]
- Berry, S.D.; Fondong, V.N.; Rey, C.; Rogan, D.; Fauquet, C.M.; Brown, J.K. Molecular Evidence for Five Distinct Bemisia tabaci (Homoptera: Aleyrodidae) Geographic Haplotypes Associated with Cassava Plants in Sub-Saharan Africa. Ann. Entomol. Soc. Am. 2004, 97, 852–859. [Google Scholar] [CrossRef]
- Sseruwagi, P.; Legg, J.; Maruthi, M.N.; Colvin, J.; Rey, M.; Brown, J. Genetic diversity of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) populations and presence of the B biotype and a non-B biotype that can induce silverleaf symptoms in squash, in Uganda. Ann. Appl. Biol. 2005, 147, 253–265. [Google Scholar] [CrossRef]
- De Marchi, B.R.; Marubayashi, J.M.; Favara, G.M.; Yuki, V.A.; Watanabe, L.F.M.; Barbosa, L.F.; Pavan, M.A.; Krause-Sakate, R. Comparative transmission of five viruses by Bemisia tabaci NW2 and MEAM1. Trop. Plant Pathol. 2017, 42, 495–499. [Google Scholar] [CrossRef]
- Sseruwagi, P.; Maruthi, M.N.; Colvin, J.; Rey, M.; Brown, J.; Legg, J.P. Colonization of non-cassava plant species by cassava whiteflies (Bemisia tabaci) in Uganda. Entomol. Exp. Appl. 2006, 119, 145–153. [Google Scholar] [CrossRef]
- Mugerwa, H.; Seal, S.; Wang, H.-L.; Patel, M.V.; Kabaalu, R.; Omongo, C.A.; Alicai, T.; Tairo, F.; Ndunguru, J.; Sseruwagi, P.; et al. African ancestry of New World, Bemisia tabaci-whitefly species. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Costa, A.S.; Russell, L.M. Failure of Bemisia tabaci to breed on cassava plants in Brazil (Homoptera, Aleyrodidae). Ciência. e Cult São Paulo. 1975, 27, 388–390. [Google Scholar]
- Bedford, I.D.; Briddon, R.W.; Brown, J.K.; Rosell, R.C.; Markham, P.G. Geminivirus transmission and biological characterisation of Bemisia tabaci (Gennadius) biotypes from different geographic regions. Ann. Appl. Biol. 1994, 125, 311–325. [Google Scholar] [CrossRef]
- De Barro, P.; Trueman, J.W.H.; Frohlich, D.R. Bemisia argentifolii is a race of B. tabaci (Hemiptera: Aleyrodidae): The molecular genetic differentiation of B. tabaci populations around the world. Bull. Entomol. Res. 2005, 95, 193–203. [Google Scholar] [CrossRef]
- Bosco, D.; Loria, A.; Sartor, C.; Cenis, J.L. PCR-RFLP identification of Bemisia tabaci biotypes in the Mediterranean Basin. Phytoparasitica 2006, 34, 243–251. [Google Scholar] [CrossRef]
- Dinsdale, A.; Cook, L.G.; Riginos, C.; Buckley, Y.M.; De Barro, P. Refined Global Analysis of Bemisia tabaci (Hemiptera: Sternorrhyncha: Aleyrodoidea: Aleyrodidae) Mitochondrial Cytochrome Oxidase 1 to Identify Species Level Genetic Boundaries. Ann. Entomol. Soc. Am. 2010, 103, 196–208. [Google Scholar] [CrossRef]
- Wosula, E.N.; Chen, W.; Fei, Z.; Legg, J.P. Unravelling the Genetic Diversity among Cassava Bemisia tabaci Whiteflies Using NextRAD Sequencing. Genome Biol. Evol. 2017, 9, 2958–2973. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Wosula, E.N.; Hasegawa, D.K.; Casinga, C.; Shirima, R.R.; Fiaboe, K.K.; Hanna, R.; Fosto, A.; Goergen, G.; Tamò, M.; et al. Genome of the African cassava whitefly Bemisia tabaci and distribution and genetic diversity of cassava-colonizing whiteflies in Africa. Insect Biochem. Mol. Biol. 2019, 110, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Storey, H.H. Virus diseases of East African plants. VI. A progress report on studies of disease of cassava. East Afr. Agric J. 1936, 2, 34–39. [Google Scholar]
- Dubern, J. Transmission of African cassava mosaic geminivirus by the whitefly (Bemisia tabaci). Trop. Sci. 1994, 34, 82–91. [Google Scholar]
- Maruthi, M.N.; Colvin, D.J.; Thwaites, R.M.; Banks, G.K.; Gibson, G.; Seal, S.E. Reproductive incompatibility and cytochrome oxidase I gene sequence variability amongst host-adapted and geographically separate Bemisia tabaci populations (Hemiptera: Aleyrodidae). Syst. Entomol. 2004, 29, 560–568. [Google Scholar] [CrossRef]
- Legg, J.P.; Jeremiah, S.; Obiero, H.; Maruthi, M.; Ndyetabula, I.; Okao-Okuja, G.; Bouwmeester, H.; Bigirimana, S.; Tata-Hangy, W.; Gashaka, G.; et al. Comparing the regional epidemiology of the cassava mosaic and cassava brown streak virus pandemics in Africa. Virus Res. 2011, 159, 161–170. [Google Scholar] [CrossRef]
- Legg, J.; Fauquet, C. Cassava mosaic geminiviruses in Africa. Plant Mol. Biol. 2004, 56, 585–599. [Google Scholar] [CrossRef]
- Legg, J.; Owor, B.; Sseruwagi, P.; Ndunguru, J. Cassava Mosaic Virus Disease in East and Central Africa: Epidemiology and Management of a Regional Pandemic. Adv. Virus Res. 2006, 67, 355–418. [Google Scholar] [CrossRef]
- Tembo, M.; Mataa, M.; Legg, J.; Chikoti, P.C.; Ntawuruhunga, P. Cassava mosaic disease: Incidence and yield performance of cassava cultivars in Zambia. J. Plant Pathol. 2017, 99, 681–689. [Google Scholar]
- Legg, J.P. Emergence, spread and strategies for controlling the pandemic of cassava mosaic virus disease in east and central Africa. Crop. Prot. 1999, 18, 627–637. [Google Scholar] [CrossRef]
- Sseruwagi, P.; Rey, M.E.C.; Brown, J.K.; Legg, J.P. The cassava mosaic geminiviruses occurring in Uganda following the 1990s epidemic of severe cassava mosaic disease. Ann. Appl. Biol. 2004, 145, 113–121. [Google Scholar] [CrossRef]
- Legg, J.P.; French, R.; Rogan, D.; Okao-Okuja, G.; Brown, J.K. A distinct Bemisia tabaci (Gennadius) (Hemiptera: Sternorrhyncha: Aleyrodidae) genotype cluster is associated with the epidemic of severe cassava mosaic virus disease in Uganda. Mol. Ecol. 2002, 11, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- Legg, J.P.; Sseruwagi, P.; Boniface, S.; Okao-Okuja, G.; Shirima, R.; Bigirimana, S.; Gashaka, G.; Herrmann, H.-W.; Jeremiah, S.; Obiero, H.; et al. Spatio-temporal patterns of genetic change amongst populations of cassava Bemisia tabaci whiteflies driving virus pandemics in East and Central Africa. Virus Res. 2014, 186, 61–75. [Google Scholar] [CrossRef]
- Mulenga, R.M.; Boykin, L.M.; Chikoti, P.C.; Sichilima, S.; Ng’Uni, D.; Alabi, O.J. Cassava Brown Streak Disease and Ugandan cassava brown streak virus Reported for the First Time in Zambia. Plant Dis. 2018, 102, 1410–1418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sseruwagi, P.; Sserubombwe, W.; Legg, J.; Ndunguru, J.; Thresh, J. Methods of surveying the incidence and severity of cassava mosaic disease and whitefly vector populations on cassava in Africa: A review. Virus Res. 2004, 100, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Hahn, S.K.; Terry, E.R.; Leuschner, K. Breeding cassava for resistance to cassava mosaic disease. Euphytica 1980, 29, 673–683. [Google Scholar] [CrossRef]
- Frohlich, D.R.; Torres-Jerez, I.; Bedford, I.D.; Markham, P.G.; Brown, J.K. A phylogeographical analysis of the Bemisia tabaci species complex based on mitochondrial DNA markers. Mol. Ecol. 1999, 8, 1683–1691. [Google Scholar] [CrossRef]
- Simon, C.; Frati, F.; Beckenbach, A.; Crespi, B.; Liu, H.; Flook, P. Evolution, Weighting, and Phylogenetic Utility of Mitochondrial Gene Sequences and a Compilation of Conserved Polymerase Chain Reaction Primers. Ann. Entomol. Soc. Am. 1994, 87, 651–701. [Google Scholar] [CrossRef]
- Walsh, P.S.; A Metzger, D.; Higuchi, R. Chelex 100 as a medium for simple extraction of DNA for PCR-based typing from forensic material. Biotechnology 1991, 10, 506–513. [Google Scholar] [CrossRef] [Green Version]
- Shatters, R.G.; Powell, C.A.; Boykin, L.M.; Liansheng, H.; McKenzie, C.L. Improved DNA Barcoding Method for Bemisia tabaci and Related Aleyrodidae: Development of Universal and Bemisia tabaci Biotype-Specific Mitochondrial Cytochrome c Oxidase I Polymerase Chain Reaction Primers. J. Econ. Entomol. 2009, 102, 750–758. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [Green Version]
- Brown, J.K.; Idris, A.M. Genetic Differentiation of Whitefly Bemisia tabaci Mitochondrial Cytochrome Oxidase I, and Phylogeographic Concordance with the Coat Protein of the Plant Virus Genus Begomovirus. Ann. Entomol. Soc. Am. 2005, 98, 827–837. [Google Scholar] [CrossRef]
- Mugerwa, H.; Rey, M.E.C.; Alicai, T.; Ateka, E.; Atuncha, H.; Ndunguru, J.; Sseruwagi, P. Genetic diversity and geographic distribution of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) genotypes associated with cassava in East Africa. Ecol. Evol. 2012, 2, 2749–2762. [Google Scholar] [CrossRef]
- Tajima, F. Statistical Method for Testing the Neutral Mutation Hypothesis by DNA Polymorphism. Genet 1989, 123, 585–595. [Google Scholar]
- Fu, Y.X.; Li, W.H. Statistical Tests of Neutrality of Mutations. Genet 1993, 133, 693–709. [Google Scholar]
- Rozas, J.; Ferrer-Mata, A.; Sánchez-DelBarrio, J.C.; Guirao-Rico, S.; Librado, P.; Ramos-Onsins, S.E.; Sánchez-Gracia, A. DnaSP 6: DNA Sequence Polymorphism Analysis of Large Data Sets. Mol. Biol. Evol. 2017, 34, 3299–3302. [Google Scholar] [CrossRef]
- Tocko-Marabena, B.K.; Silla, S.; Simiand, C.; Zinga, I.; Legg, J.; Reynaud, B.; Delatte, H. Genetic diversity of Bemisia tabaci species colonizing cassava in Central African Republic characterized by analysis of cytochrome c oxidase subunit I. PLoS ONE 2017, 12, e0182749. [Google Scholar] [CrossRef] [Green Version]
- Nwezeobi, J.; Onyegbule, O.; Nkere, C.; Onyeka, J.; Van Brunschot, S.; Seal, S.; Colvin, J. Cassava whitefly species in eastern Nigeria and the threat of vector-borne pandemics from East and Central Africa. PLoS ONE 2020, 15, e0232616. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Bouvaine, S.; Maruthi, M.N. Prevalence and genetic diversity of endosymbiotic bacteria infecting cassava whiteflies in Africa. BMC Microbiol. 2015, 15, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Muimba-Kankolongo, A.; Chalwe, A.; Sisupo, P.; Kang, M.S. Distribution, prevalence and outlook for control of cassava mosaic disease in Zambia. Roots 1997, 4, 2–7. [Google Scholar]
- Chikoti, P.C.; Ndunguru, J.; Melis, R.; Tairo, F.; Shanahan, P.; Sseruwagi, P. Cassava mosaic disease and associated viruses in Zambia: Occurrence and distribution. Int. J. Pest Manag. 2013, 59, 63–72. [Google Scholar] [CrossRef]
- Chikoti, P.C.; Tembo, M.; Chisola, M.; Ntawuruhunga, P.; Ndunguru, J. Status of cassava mosaic disease and whitefly population in Zambia. Afr. J. Biotechnol. 2015, 14, 2539–2546. [Google Scholar] [CrossRef] [Green Version]
- Legg, J.P. Epidemiology of a Whitefly-Transmitted Cassava Mosaic Geminivirus Pandemic in Africa. In Bemisia: Bionomics and Management of a Global Pest; Springer Science and Business Media LLC: Berlin, Germany, 2009; pp. 233–257. [Google Scholar]
- Tembo, M. Epidemiological and Molecular Characterization of Cassava Mosaic Geminiviruses and Their Effect on Yield of Cassava. Master’s Thesis, University of Zambia, Lusaka, Zambia, 2016. [Google Scholar]
- Ally, H.M. Genetic Diversity and Structure of the Superabundant Whitefly Populations, Vectors of Viruses Causing Diseases of Cassava in Three East African Countries (Malawi, Tanzania, and Uganda). Ph.D. Thesis, Université de la Réunion, Réunion, France, 2019. [Google Scholar]
- Tajebe, L.S.; Boni, S.B.; Guastella, D.; Cavalieri, V.; Lund, O.S.; Rugumamu, C.P.; Rapisarda, C.; Legg, J.P. Abundance, diversity and geographic distribution of cassava mosaic disease pandemic-associated Bemisia tabaci in Tanzania. J. Appl. Entomol. 2014, 139, 627–637. [Google Scholar] [CrossRef]
- Tajebe, L.; Guastella, D.; Cavalieri, V.; Kelly, S.; Hunter, M.; Lund, O.; Legg, J.P.; Rapisarda, C. Diversity of symbiotic bacteria associated with Bemisia tabaci (Homoptera: Aleyrodidae) in cassava mosaic disease pandemic areas of Tanzania. Ann. Appl. Biol. 2014, 166, 297–310. [Google Scholar] [CrossRef]


| Sequence Name | Country | GenBank Reference | Code (mtCOI) | Author |
|---|---|---|---|---|
| Texas B type | USA | AF164675 | - | Unpublished |
| Bur2-2 | Burundi | KF425621 | SSA1-SG2 | [32] |
| Tz10 | Tanzania | KF425607 | SSA1-SG1 | [32] |
| 21Malaw | Malawi | AY057162 | SSA1-SG3 | [31] |
| CAMW13 | Cameroon | AF344254 | SSA4 | [10] |
| CAMW16 | Cameroon | AF344257 | SSA3 | [10] |
| ChinaCQAY686091NB | China | AY686091 | - | [41] |
| DRC-KICKAL1 | DRC | MF417582 | SSA1-SG1 | [20] |
| Kenyz23a | Kenya | JQ286408 | SSA1-SG1 | [42] |
| Tz62a | Tanzania | JQ286461 | SSA1-SG3 | [42] |
| ZAMB1 | Zambia | AF344280 | SSA1-SG1 | [10] |
| ZAMB2 | Zambia | AF344281 | SSA1-SG3 | [10] |
| ZAMB6 | Zambia | AF344284 | SSA1-SG1 | [10] |
| Bemisia afer | Uganda | AF418673 | Bemisia afer | [24] |
| Sequence Name | Province a | Subgroup Name | Closest Relative | Sequence from GenBank | Country b |
|---|---|---|---|---|---|
| (Nt Identity%) | |||||
| MT542001 c | Eastern | SSA1-SG3 | 99.6 | AY057162 | Malawi |
| MT542025 c | Western | SSA1-SG3 | 99.6 | AY057162 | Malawi |
| MT434853 d | Eastern | SSA1-SG3 | 99.6 | AY057162 | Malawi |
| MT434852 d | Eastern | SSA1-SG3 | 99.6 | AY057162 | Malawi |
| MT434835 d | Western | SSA1-SG3 | 99.6 | AY057162 | Malawi |
| MT542019 c | Western | SSA1-SG1 | 100 | KF425607 | Tanzania |
| MT542031 c | Western | SSA1-SG1 | 100 | KF425607 | Tanzania |
| MT542017 c | Western | SSA1-SG1 | 100 | KF425607 | Tanzania |
| MT434844 d | Western | SSA1-SG1 | 100 | MF417582 | DRC |
| MT434855 d | North Western | SSA1-SG1 | 100 | MF417582 | DRC |
| MT434830 d | Western | SSA1-SG1 | 100 | MF417582 | DRC |
| MT434846 d | Western | SSA1-SG1 | 100 | MF417582 | DRC |
| MT434827 d | Western | SSA1-SG1 | 100 | MF417582 | DRC |
| MT542012 c | Eastern | SSA1-SG2 | 99.1 | KF425621 | Burundi |
| MT542015 c | Western | SSA1-SG2 | 99.1 | KF425621 | Burundi |
| MT434851 d | Eastern | SSA1-SG2 | 99.1 | KF425621 | Burundi |
| MT434850 d | Eastern | SSA1-SG2 | 99.1 | KF425621 | Burundi |
| Parameter | All | SSA1-SG1 | SSA1-SG2 | SSA1-SG3 |
|---|---|---|---|---|
| Sample size | 68 | 38 | 8 | 22 |
| Number of haplotypes | 7 | 4 | 1 | 2 |
| Polymorphic sites (S) | 15 | 3 | 0 | 1 |
| Average number of nucleotide differences (k) | 5.52941 | 0.25462 | 0 | 0.09091 |
| Nucleotide diversity (Pi) | 0.00744 | 0.00034 | 0 | 0.00012 |
| Haplotype diversity (Hd) | 0.662 | 0.245 | - | 0.091 |
| Variance of Hd | 0.00154 | 0.00796 | - | 0.00655 |
| Standard deviation of Hd | 0.039 | 0.089 | - | 0.081 |
| Theta per sequence | 3.13195 | 0.71402 | - | 0.27432 |
| Theta per site | 0.00422 | 0.00096 | - | 0.00037 |
| Fu’s Fs statistic | 1.00136 | −1.79006 | - | −1.67803 |
| Tajima’s D p for Tajima’s D | 2.23673 p < 0.05 | −1.42080 p > 0.10 | - | −1.16240 p > 0.10 |
| 2013 | 2015 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Province | Number of Fields | CMD Incidence (%) | SE | Mean CMD Severity (1–5) | SE | Mean Whitefly Population | SE | Number of Fields | CMD Incidence (%) | SE | Mean CMD Severity (1–5) | SE | Mean Whitefly Population | SE |
| Central | 36 | 57.9 | 4.027 | 3.3 | 0.25 | 0.7 | 0.035 | 29 | 70.2 | 3.211 | 3.8 | 0.026 | 1.3 | 0.084 |
| Eastern | 51 | 26.6 | 4.324 | 2.8 | 0.29 | 1.0 | 0.085 | 44 | 29.3 | 4.527 | 3.1 | 0.037 | 0.6 | 0.079 |
| Luapula | 47 | 49.9 | 4.102 | 3.2 | 0.19 | 0.5 | 0.034 | 28 | 46.2 | 4.915 | 3.2 | 0.037 | 0.3 | 0.035 |
| Lusaka | 24 | 78 | 4.914 | 3.4 | 0.42 | 6.6 | 0.628 | 14 | 83.6 | 7.759 | 3.9 | 0.038 | 3.7 | 0.463 |
| North Western | 16 | 55.8 | 8.271 | 3.3 | 0.33 | 2.2 | 0.311 | 17 | 63.6 | 7.765 | 3.4 | 0.04 | 2.2 | 0.408 |
| Northern | 40 | 39.1 | 4.571 | 3.1 | 0.25 | 0.2 | 0.045 | 43 | 33.2 | 4.698 | 3.4 | 0.027 | 0.2 | 0.038 |
| Western | 31 | 73.1 | 3.529 | 3.1 | 11.1 | 0.451 | 25 | 58.8 | 5.688 | 3.3 | 0.028 | 10.8 | 0.599 | |
| Mean | 50.5 | 3.2 | 3.3 | 48.9 | 3.5 | 2.3 | ||||||||
| SE | 2.045 | 0.11 | 0.117 | 2.32 | 0.013 | 0.104 | ||||||||
| p-value (5%) | p < 0.0001 | p < 0.0001 | p < 0.0001 | p < 0.0001 | p < 0.0001 | p < 0.0001 | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiza Chikoti, P.; Tembo, M.; Peter Legg, J.; Rufini Shirima, R.; Mugerwa, H.; Sseruwagi, P. Genetic Diversity of Mitochondrial DNA of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) Associated with Cassava and the Occurrence of Cassava Mosaic Disease in Zambia. Insects 2020, 11, 761. https://doi.org/10.3390/insects11110761
Chiza Chikoti P, Tembo M, Peter Legg J, Rufini Shirima R, Mugerwa H, Sseruwagi P. Genetic Diversity of Mitochondrial DNA of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) Associated with Cassava and the Occurrence of Cassava Mosaic Disease in Zambia. Insects. 2020; 11(11):761. https://doi.org/10.3390/insects11110761
Chicago/Turabian StyleChiza Chikoti, Patrick, Mathias Tembo, James Peter Legg, Rudolph Rufini Shirima, Habibu Mugerwa, and Peter Sseruwagi. 2020. "Genetic Diversity of Mitochondrial DNA of Bemisia tabaci (Gennadius) (Hemiptera: Aleyrodidae) Associated with Cassava and the Occurrence of Cassava Mosaic Disease in Zambia" Insects 11, no. 11: 761. https://doi.org/10.3390/insects11110761








