Comprehensive Survey of Area-Wide Agricultural Pesticide Use in Southern United States Row Crops and Potential Impact on Honey Bee Colonies
Abstract
1. Introduction
2. Methods and Materials
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Visscher, P.K.; Seeley, T.D. Foraging Strategy of Honeybee Colonies in a Temperate Deciduous Forest. Ecology 1982, 63, 1790. [Google Scholar] [CrossRef]
- Hagler, J.R.; Mueller, S.; Teuber, L.R.; Machtley, S.A.; van Deynze, A. Foraging Range of Honey Bees, Apis mellifera, in Alfalfa Seed Production Fields. J. Insect Sci. 2011, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Steffan-Dewenter, I.; Kuhn, A. Honeybee foraging in differentially structured landscapes. Proc. R. Soc. B Boil. Sci. 2003, 270, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Couvillon, M.J.; Pearce, F.C.R.; Accleton, C.; Fensome, K.A.; Quah, S.K.; Taylor, E.L.; Ratnieks, F.L. Honey bee foraging distance depends on month and forage type. Apidologie 2015, 46, 61–70. [Google Scholar] [CrossRef]
- Beekman, M.; Ratnieks, F.L.W. Long-range foraging by the honey-bee, Apis mellifera L. Funct. Ecol. 2000, 14, 490–496. [Google Scholar] [CrossRef]
- Waddington, K.D. Honey bee foraging profitability and round dance correlates. J. Comp. Physiol. A 1982, 148, 297–301. [Google Scholar] [CrossRef]
- Cook, S.M.; Awmack, C.S.; Murray, D.A.; Williams, I.H. Are honey bees’ foraging preferences affected by pollen amino acid composition? Ecol. Entomol. 2003, 28, 622–627. [Google Scholar] [CrossRef]
- Hendriksma, H.P.; Shafir, S. Honey bee foragers balance colony nutritional deficiencies. Behav. Ecol. Sociobiol. 2016, 70, 509–517. [Google Scholar] [CrossRef]
- Schiestl, F.P.; Johnson, S.D. Pollinator-mediated evolution of floral signals. Trends Ecol. Evol. 2013, 28, 307–315. [Google Scholar] [CrossRef]
- Greenleaf, S.S.; Williams, N.M.; Winfree, R.; Kremen, C. Bee foraging ranges and their relationship to body size. Oecologia 2007, 153, 589–596. [Google Scholar] [CrossRef]
- Levin, D.A. Dispersal versus gene flow in plants. Ann. Mo. Bot. Gard. 1981, 68, 233. [Google Scholar] [CrossRef]
- Gillespie, M.A.; Gurr, G.M.; Wratten, S.D. Beyond nectar provision: The other resource requirements of parasitoid biological control agents. Entomol. Exp. Appl. 2016, 159, 207–221. [Google Scholar] [CrossRef]
- Heidel-Baker, T.T.; O’Neal, M.E.; Batzer, J.C.; Gleason, M.L. Attracting Beneficial Insects to Iowa Agricultural Crops through Floral Provisioning; Iowa State University: Ames, IA, USA, 2014. [Google Scholar]
- Ramsden, M.W.; Menéndez, R.; Leather, S.R.; Wäckers, F. Optimizing field margins for biocontrol services: The relative role of aphid abundance, annual floral resources, and overwinter habitat in enhancing aphid natural enemies. Agric. Ecosyst. Environ. 2015, 199, 94–104. [Google Scholar] [CrossRef]
- Calderone, N.W. Insect Pollinated Crops, Insect Pollinators and US Agriculture: Trend Analysis of Aggregate Data for the Period 1992–2009. PLoS ONE 2012, 7, e37235. [Google Scholar] [CrossRef] [PubMed]
- McGregor, S.E. Insect Pollination of Cultivated Crop Plants; Agricultural Research Service: Washington, DC, USA; US Department of Agriculture: Washington, DC, USA, 1976; Volume 496.
- Severson, D.W.; Parry, J.E. A Chronology of Pollen Collection by Honeybees. J. Apic. Res. 1981, 20, 97–103. [Google Scholar] [CrossRef]
- Erickson, E.H.; Berger, G.A.; Shannon, J.G.; Robins, J.M. Honey Bee Pollination Increases Soybean Yields in the Mississippi Delta Region of Arkansas and Missouri. J. Econ. Entomol. 1978, 71, 601–603. [Google Scholar] [CrossRef]
- Chiari, W.C.; Toledo, V.D.A.A.D.; Ruvolo-Takasusuki, M.C.C.; Oliveira, A.J.B.D.; Sakaguti, E.S.; Attencia, V.M.; Costa, F.M.; Mitsui, M.H. Pollination of soybean (Glycine max L. Merril) by honeybees (Apis mellifera L.). Braz. Arch. Biol. Technol. 2005, 48, 31–36. [Google Scholar] [CrossRef]
- Abrol, D.; Shankar, U. Pollination in oil crops: Recent advances and future strategies. In Technological Innovations in Major World Oil Crops; Springer: Berlin, Germany, 2012; Volume 2, pp. 221–267. [Google Scholar]
- Milfont, M.D.O.; Rocha, E.E.M.; Lima, A.O.N.; Freitas, B.M. Higher soybean production using honeybee and wild pollinators, a sustainable alternative to pesticides and autopollination. Environ. Chem. Lett. 2013, 11, 335–341. [Google Scholar] [CrossRef]
- Coy, R.; (Commercial Beekeeper, Jonesboro, AR, USA). Personal communications, 2016.
- Smith, T.; Catchot, A.L.; Harris, J.W.; Gore, J.; Krishnan, N.; Cook, D. Influence of cultural practices on soybean nectar production. In Proceedings of the Entomological Society of America Conference, Denver, CO, USA, 8 November 2017. [Google Scholar]
- Shuel, R.W. Some Factors Affecting Nectar Secretion in Red Clover. Plant. Physiol. 1952, 27, 95–110. [Google Scholar] [CrossRef]
- Requier, F.; Odoux, J.-F.; Tamic, T.; Moreau, N.; Henry, M.; Decourtye, A.; Bretagnolle, V. Honey bee diet in intensive farmland habitats reveals an unexpectedly high flower richness and a major role of weeds. Ecol. Appl. 2015, 25, 881–890. [Google Scholar] [CrossRef]
- Di Pasquale, G.; Salignon, M.; Le Conte, Y.; Belzunces, L.P.; Decourtye, A.; Kretzschmar, A.; Suchail, S.; Brunet, J.L.; Alaux, C. Influence of pollen nutrition on honey bee health: Do pollen quality and diversity matter? PLoS ONE 2013, 8, e72016. [Google Scholar] [CrossRef] [PubMed]
- Sponsler, D.B.; Johnson, R.M. Honey bee success predicted by landscape composition in Ohio, USA. PeerJ 2015, 3, 838. [Google Scholar] [CrossRef] [PubMed]
- Desneux, N.; Decourtye, A.; Delpuech, J.-M. The Sublethal Effects of Pesticides on Beneficial Arthropods. Annu. Rev. Entomol. 2007, 52, 81–106. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.M.; Ellis, M.D.; Mullin, C.A.; Frazier, M. Pesticides and honey bee toxicity—USA. Apidologie 2010, 41, 312–331. [Google Scholar] [CrossRef]
- Blacquière, T.; Smagghe, G.; Van Gestel, C.A.M.; Mommaerts, V. Neonicotinoids in bees: A review on concentrations, side-effects and risk assessment. Ecotoxicology 2012, 21, 973–992. [Google Scholar] [CrossRef] [PubMed]
- Lundin, O.; Rundlöf, M.; Smith, H.G.; Fries, I.; Bommarco, R. Neonicotinoid Insecticides and Their Impacts on Bees: A Systematic Review of Research Approaches and Identification of Knowledge Gaps. PLoS ONE 2015, 10, e0136928. [Google Scholar] [CrossRef] [PubMed]
- Rortais, A.; Arnold, G.; Halm, M.-P.; Touffet-Briens, F. Modes of honeybees exposure to systemic insecticides: Estimated amounts of contaminated pollen and nectar consumed by different categories of bees. Apidologie 2005, 36, 71–83. [Google Scholar] [CrossRef]
- Dively, G.P.; Embrey, M.S.; Kamel, A.; Hawthorne, D.J.; Pettis, J.S. Correction: Assessment of Chronic Sublethal Effects of Imidacloprid on Honey Bee Colony Health. PLoS ONE 2015, 10, e0126043. [Google Scholar] [CrossRef]
- Laycock, I.; Lenthall, K.M.; Barratt, A.T.; Cresswell, J.E. Effects of imidacloprid, a neonicotinoid pesticide, on reproduction in worker bumble bees (Bombus terrestris). Ecotoxicology 2012, 21, 1937–1945. [Google Scholar] [CrossRef]
- Whitehorn, P.; O’Connor, S.; Wäckers, F.L.; Goulson, D.; O’Connor, S. Neonicotinoid Pesticide Reduces Bumble Bee Colony Growth and Queen Production. Science 2012, 336, 351–352. [Google Scholar] [CrossRef]
- Cumming, G.S.; Spiesman, B.J. Regional problems need integrated solutions: Pest management and conservation biology in agroecosystems. Boil. Conserv. 2006, 131, 533–543. [Google Scholar] [CrossRef]
- Chauzat, M.-P.; Faucon, J.-P.; Martel, A.-C.; Lachaize, J.; Cougoule, N.; Aubert, M. A Survey of Pesticide Residues in Pollen Loads Collected by Honey Bees in France. J. Econ. Entomol. 2006, 99, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Bernal, J.L.; Garrido-Bailon, E.; Del Nozal, J.B.; González-Porto, A.V.; Hernández, R.M.; Diego, J.C.; Jimenez, J.J.; Higes, M. Overview of Pesticide Residues in Stored Pollen and Their Potential Effect on Bee Colony (Apis mellifera) Losses in Spain. J. Econ. Entomol. 2010, 103, 1964–1971. [Google Scholar] [CrossRef] [PubMed]
- Mullin, C.A.; Frazier, M.; Frazier, J.L.; Ashcraft, S.; Simonds, R.; Pettis, J.S. High levels of miticides and agrochemicals in North American apiaries: Implications for honey bee health. PLoS ONE 2010, 5, e9754. [Google Scholar] [CrossRef] [PubMed]
- Codling, G.; Al Naggar, Y.; Giesy, J.P.; Robertson, A.J. Concentrations of neonicotinoid insecticides in honey, pollen and honey bees (Apis mellifera L.) in central Saskatchewan, Canada. Chemosphere 2016, 144, 2321–2328. [Google Scholar] [CrossRef]
- Pasture, Fall Armyworm. In Insecticide Recommendations for Arkansas, MP-144; University of Arkansas Systems Division of Agriculture: Little Rock, AR, USA, 2018; Available online: https://www.uaex.edu/publications/pdf/mp144/mp144.pdf (accessed on 18 June 2018).
- Stewart, S.D.; Lorenz, G.M.; Catchot, A.L.; Gore, J.; Cook, D.; Skinner, J.; Mueller, T.C.; Johnson, D.R.; Zawislak, J.; Barber, J. Potential Exposure of Pollinators to Neonicotinoid Insecticides from the Use of Insecticide Seed Treatments in the Mid-Southern United States. Environ. Sci. Technol. 2014, 48, 9762–9769. [Google Scholar] [CrossRef]
- Medici, S.K.; Castro, A.; Sarlo, E.G.; Marioli, J.M.; Eguaras, M.J. The concentration effect of selected acaricides present in beeswax foundation on the survival of Apis mellifera colonies. J. Apic. Res. 2012, 51, 164–168. [Google Scholar] [CrossRef]
- Korta, E.; Bakkali, A.; Berrueta, L.A.; Gallo, B.; Vicente, F.; Kilchenmann, V.; Bogdanov, S. Study of Acaricide Stability in Honey. Characterization of Amitraz Degradation Products in Honey and Beeswax. J. Agric. Food Chem. 2001, 49, 5835–5842. [Google Scholar] [CrossRef]
- Bajuk, B.P.; Babnik, K.; Snoj, T.; Milčinski, L.; Ocepek, M.P.; Škof, M.; Jenčič, V.; Štajnbaher, D.; Kobal, S.; Filazi, A. Coumaphos residues in honey, bee brood, and beeswax after Varroa treatment. Apidologie 2017, 37, 57–598. [Google Scholar]
- Rissato, S.R.; Galhiane, M.S.; De Almeida, M.V.; Gerenutti, M.; Apon, B.M. Multiresidue determination of pesticides in honey samples by gas chromatography–mass spectrometry and application in environmental contamination. Food Chem. 2007, 101, 1719–1726. [Google Scholar] [CrossRef]
- Alburaki, M.; Chen, D.; Skinner, J.A.; Meikle, W.G.; Tarpy, D.R.; Adamczyk, J.; Stewart, S.D. Honey Bee Survival and Pathogen Prevalence: From the Perspective of Landscape and Exposure to Pesticides. Insects 2018, 9, 65. [Google Scholar] [CrossRef] [PubMed]
- Bogdanov, S. Contaminants of bee products. Apidologie 2006, 37, 1–18. [Google Scholar] [CrossRef]
- Thompson, H.M.; Fryday, S.L.; Harkin, S.; Milner, S. Potential impacts of synergism in honeybees (Apis mellifera) of exposure to neonicotinoids and sprayed fungicides in crops. Apidologie 2014, 45, 545–553. [Google Scholar] [CrossRef]
- Johnson, R.M.; Dahlgren, L.; Siegfried, B.D.; Ellis, M.D. Acaricide, Fungicide and Drug Interactions in Honey Bees (Apis mellifera). PLoS ONE 2013, 8, e54092. [Google Scholar] [CrossRef] [PubMed]
- Pettis, J.S.; Lichtenberg, E.M.; Andree, M.; Stitzinger, J.; Rose, R. Crop pollination exposes honey bees to pesticides which alters their susceptibility to the gut pathogen Nosema ceranae. PLoS ONE 2013, 8, e70182. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.C.; Chuang, Y.C.; Chen, Y.L.; Chang, L.H. Abnormal foraging behavior induced by sublethal dosage of imidacloprid in the honey bee (Hymenoptera: Apidae). J. Econ. Entomol. 2008, 101, 1743–1748. [Google Scholar] [CrossRef] [PubMed]
- Bryden, J.; Gill, R.J.; Mitton, R.A.A.; Raine, N.E.; Jansen, V.A.; Hodgson, D. Chronic sublethal stress causes bee colony failure. Ecol. Lett. 2013, 16, 1463–1469. [Google Scholar] [CrossRef]
- Tosi, S.; Burgio, G.; Nieh, J.C. A common neonicotinoid pesticide, thiamethoxam, impairs honey bee flight ability. Sci. Rep. 2017, 7, 303. [Google Scholar] [CrossRef]
- Wu, J.Y.; Anelli, C.M.; Sheppard, W.S. Sub-Lethal Effects of Pesticide Residues in Brood Comb on Worker Honey Bee (Apis mellifera) Development and Longevity. PLoS ONE 2011, 6, e14720. [Google Scholar] [CrossRef]
- Corta, E. Kinetics and mechanism of amitraz hydrolysis in aqueous media by HPLC and GC-MS. Talanta 1999, 48, 189–199. [Google Scholar] [CrossRef]
- Vita Bee Health. Apiguard Product Label. Valdosta, GA 31603. Available online: https://www3.epa.gov/pesticides/chem_search/ppls/079671-00001-20141007.pdf (accessed on 18 June 2018).
- Véto-pharma, Inc. ApiLifeVar Product Label. Palaiseau, France 91120. Available online: https://www3.epa.gov/pesticides/chem_search/ppls/073291-00001-20190521.pdf (accessed on 18 June 2018).
- Krupke, C.H.; Hunt, G.J.; Eitzer, B.D.; Andino, G.; Given, K. Multiple Routes of Pesticide Exposure for Honey Bees Living Near Agricultural Fields. PLoS ONE 2012, 7, e29268. [Google Scholar] [CrossRef] [PubMed]
- Dively, G.P.; Kamel, A. Insecticide Residues in Pollen and Nectar of a Cucurbit Crop and Their Potential Exposure to Pollinators. J. Agric. Food Chem. 2012, 60, 4449–4456. [Google Scholar] [CrossRef] [PubMed]
- Meikle, W.G.; Adamczyk, J.J.; Weiss, M.; Gregorc, A.; Johnson, D.R.; Stewart, S.D.; Zawislak, J.; Carroll, M.J.; Lorenz, G.M. Sublethal Effects of Imidacloprid on Honey Bee Colony Growth and Activity at Three Sites in the US. PLoS ONE 2016, 11, e0168603. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, E.A.D.; Mulhauser, B.; Mulot, M.; Mutabazi, A.; Glauser, G.; Aebi, A. A worldwide survey of neonicotinoids in honey. Science 2017, 358, 109–111. [Google Scholar] [CrossRef] [PubMed]
- Westphal, C.; Steffan-Dewenter, I.; Tscharntke, T. Mass flowering crops enhance pollinator densities at a landscape scale. Ecol. Lett. 2003, 6, 961–965. [Google Scholar] [CrossRef]
- Le Féon, V.; Schermann-Legionnet, A.; Delettre, Y.; Aviron, S.; Billeter, R.; Bugter, R.; Hendrickx, F.; Burel, F. Intensification of agriculture, landscape composition and wild bee communities: A large scale study in four European countries. Agric. Ecosyst. Environ. 2010, 137, 143–150. [Google Scholar] [CrossRef]
- Holzschuh, A.; Dormann, C.F.; Tscharntke, T.; Steffan-Dewenter, I. Mass-flowering crops enhance wild bee abundance. Oecologia 2013, 172, 477–484. [Google Scholar] [CrossRef]
- Pilling, E.D.; Jepson, P.C. Synergism between EBI fungicides and a pyrethroid insecticide in the honeybee (Apis mellifera). Pestic. Sci. 1993, 39, 293–297. [Google Scholar] [CrossRef]
- Thompson, H.; Wilkins, S. Assessment of the synergy and repellency of pyrethroid/fungicide mixtures. Bull. Insectol. 2003, 56, 131–134. [Google Scholar]
- Haarmann, T.; Spivak, M.; Weaver, D.; Weaver, B.; Glenn, T. Effects of Fluvalinate and Coumaphos on Queen Honey Bees (Hymenoptera: Apidae) in Two Commercial Queen Rearing Operations. J. Econ. Entomol. 2002, 95, 28–35. [Google Scholar] [CrossRef]
- Di Prisco, G.; Cavaliere, V.; Annoscia, D.; Varricchio, P.; Caprio, E.; Nazzi, F.; Gargiulo, G.; Pennacchio, F. Neonicotinoid clothianidin adversely affects insect immunity and promotes replication of a viral pathogen in honey bees. Proc. Natl. Acad. Sci. USA 2013, 110, 18466–18471. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Schmehl, D.R.; Mullin, C.A.; Frazier, J.L. Four Common Pesticides, Their Mixtures and a Formulation Solvent in the Hive Environment Have High Oral Toxicity to Honey Bee Larvae. PLoS ONE 2014, 9, e77547. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.M. Honey Bee Toxicology. Annu. Rev. Entomol. 2015, 60, 415–434. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, P.; Aumeier, P.; Ziegelmann, B. Biology and control of Varroa destructor. J. Invertebr. Pathol. 2010, 103, S96–S119. [Google Scholar] [CrossRef] [PubMed]
- Guzman-Novoa, E.; Eccles, L.; Calvete, Y.; Mcgowan, J.; Kelly, P.G.; Correa-Benítez, A. Varroa destructor is the main culprit for the death and reduced populations of overwintered honey bee (Apis mellifera) colonies in Ontario, Canada. Apidologie 2010, 41, 443–450. [Google Scholar] [CrossRef]
- Chen, Y.; Evans, J.D.; Smith, I.B.; Pettis, J.S. Nosema ceranae is a long-present and wide-spread microsporidian infection of the European honey bee (Apis mellifera) in the United States. J. Invertebr. Pathol. 2008, 97, 186–188. [Google Scholar] [CrossRef] [PubMed]
- Alaux, C.; Ducloz, F.; Crauser, D.; Le Conte, Y. Diet effects on honeybee immunocompetence. Boil. Lett. 2010, 6, 562–565. [Google Scholar] [CrossRef]
- Vanengelsdorp, D.; Evans, J.D.; Saegerman, C.; Mullin, C.; Haubruge, E.; Nguyen, B.K.; Frazier, M.; Frazier, J.; Cox-Foster, D.; Chen, Y.; et al. Colony Collapse Disorder: A Descriptive Study. PLoS ONE 2009, 4, e6481. [Google Scholar] [CrossRef]
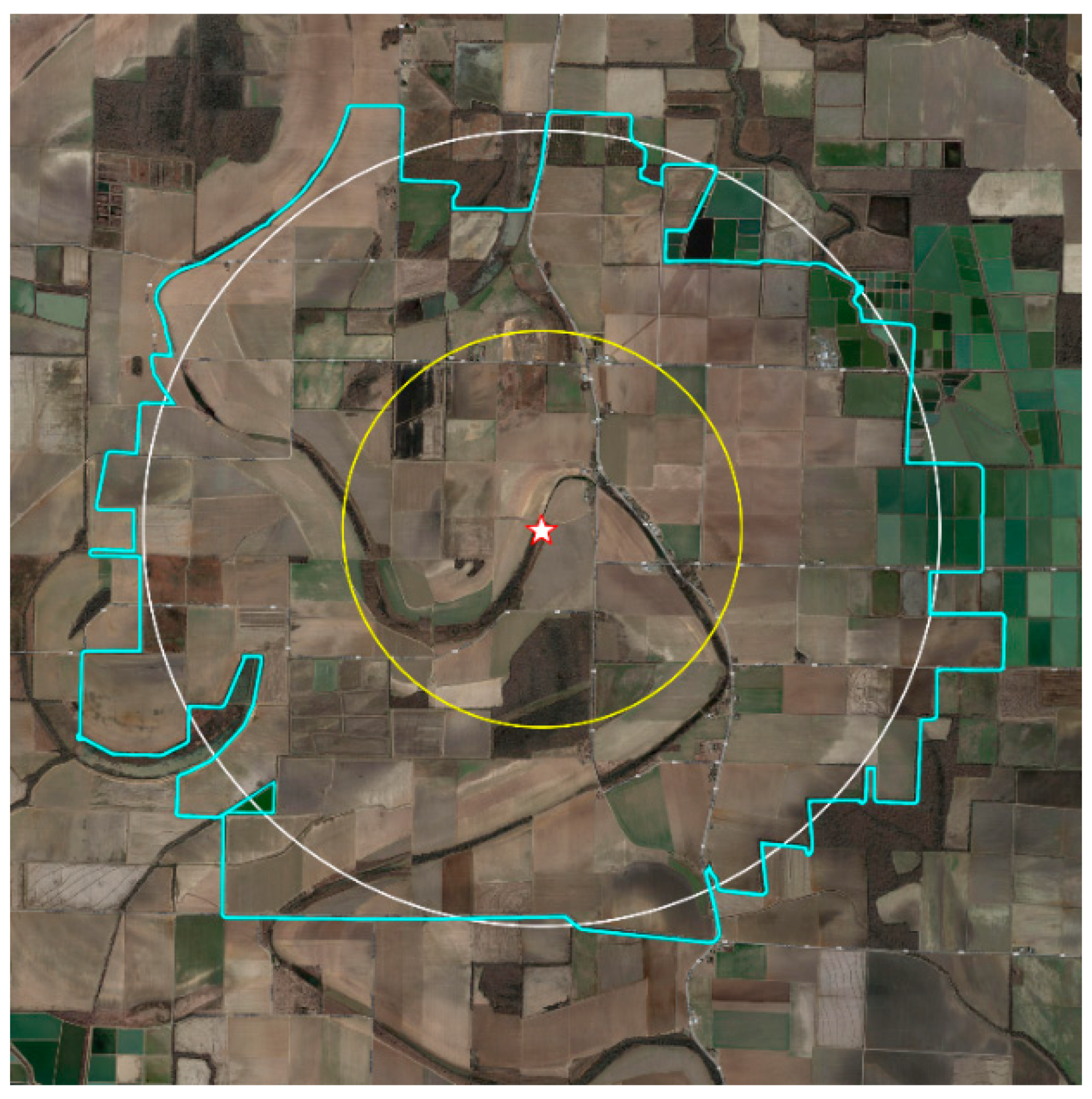
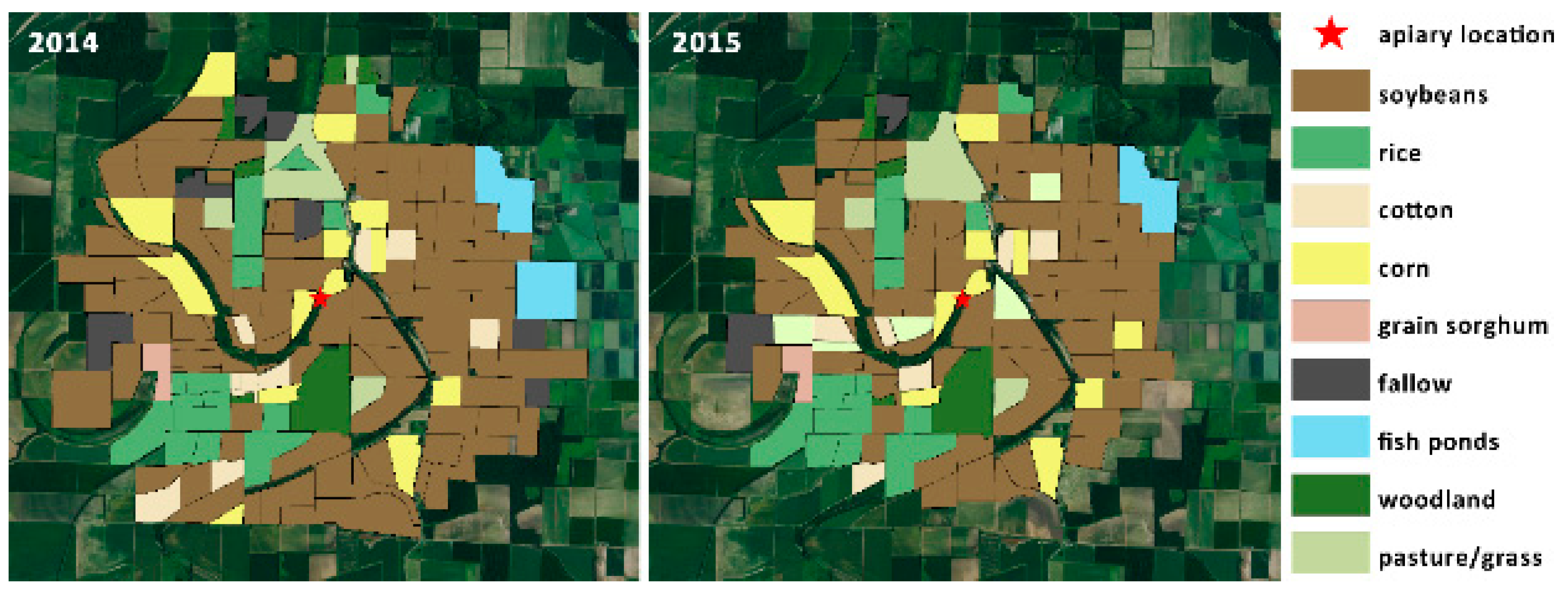
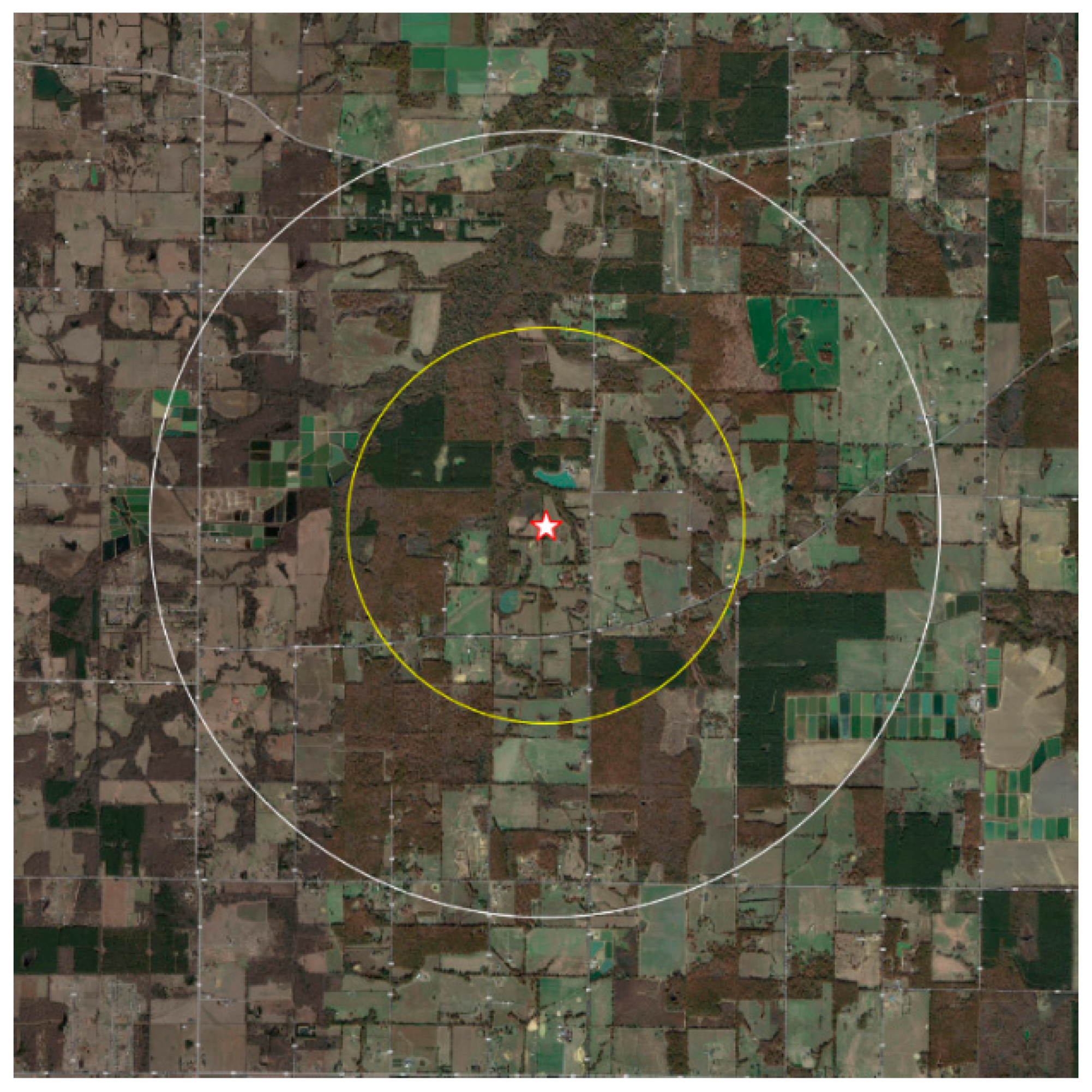
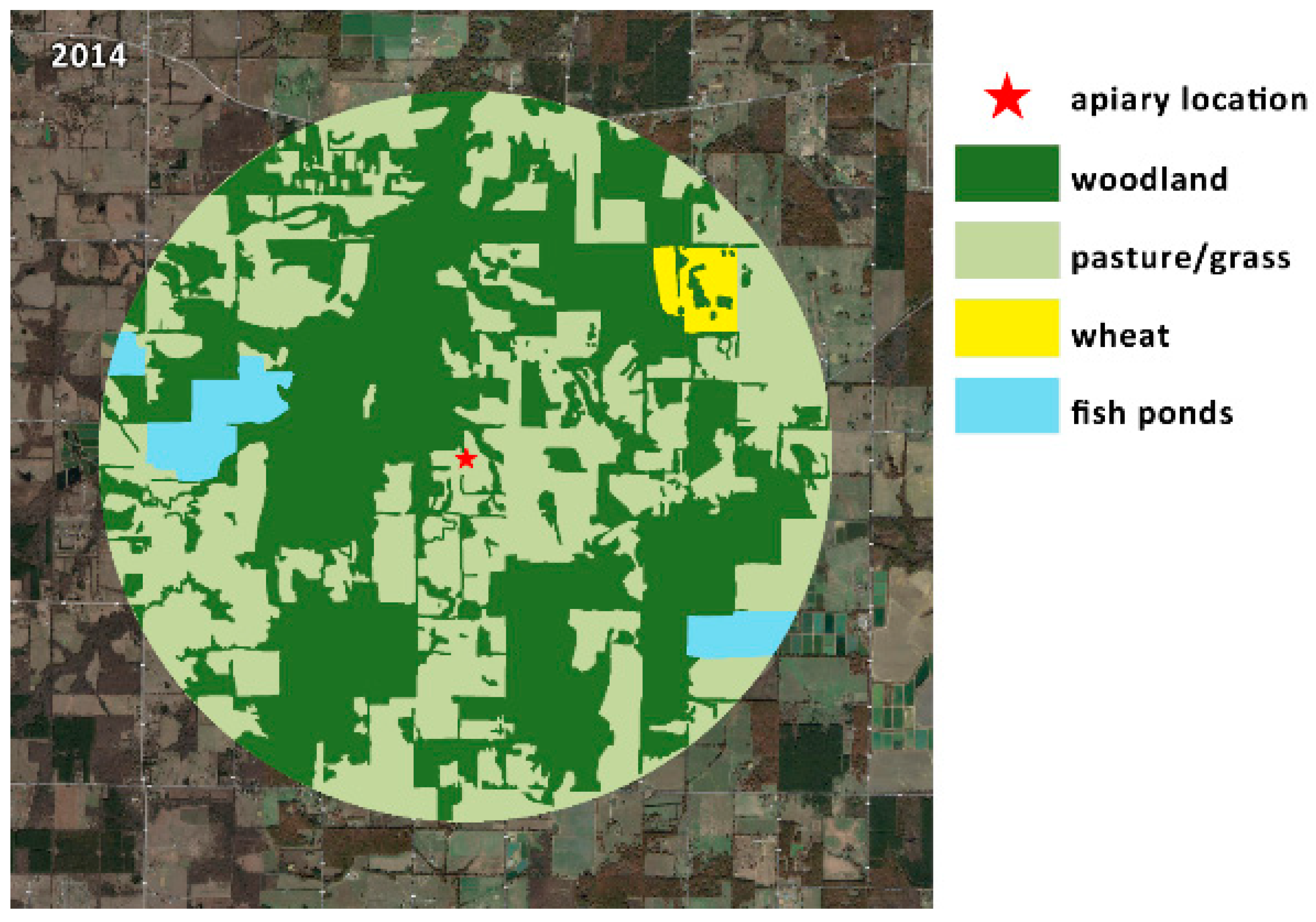
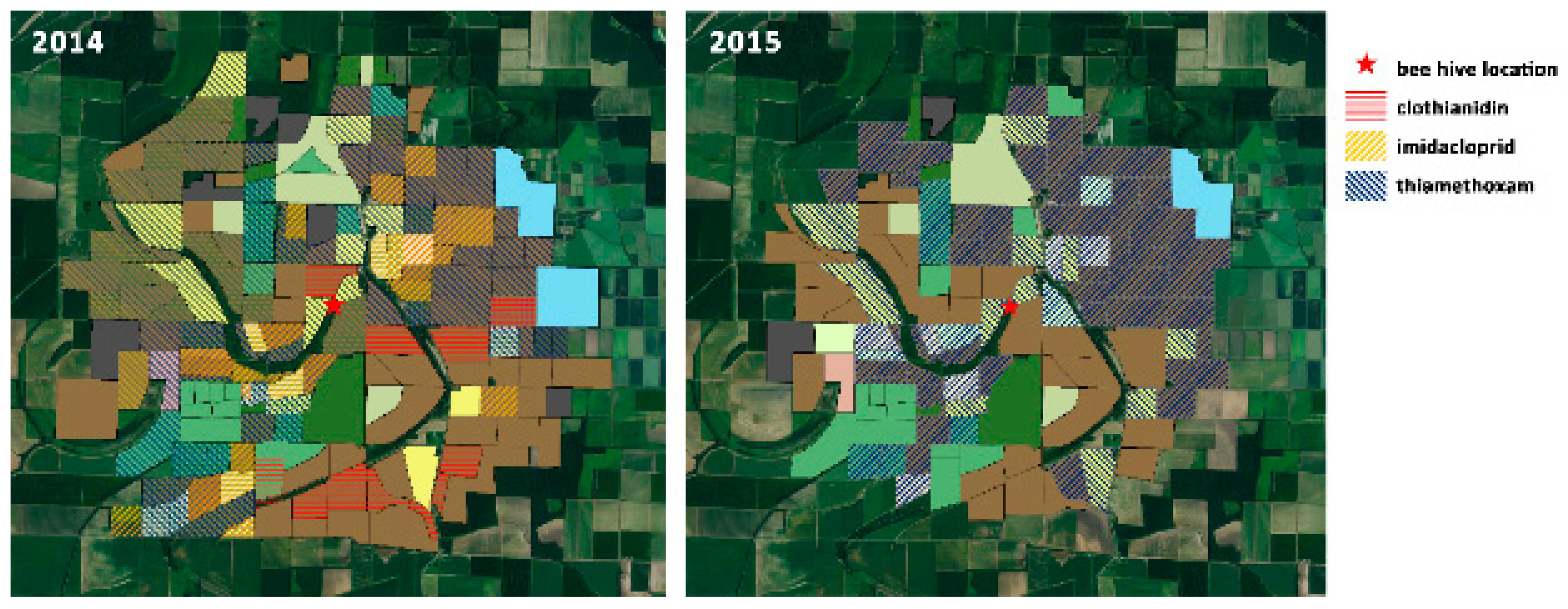
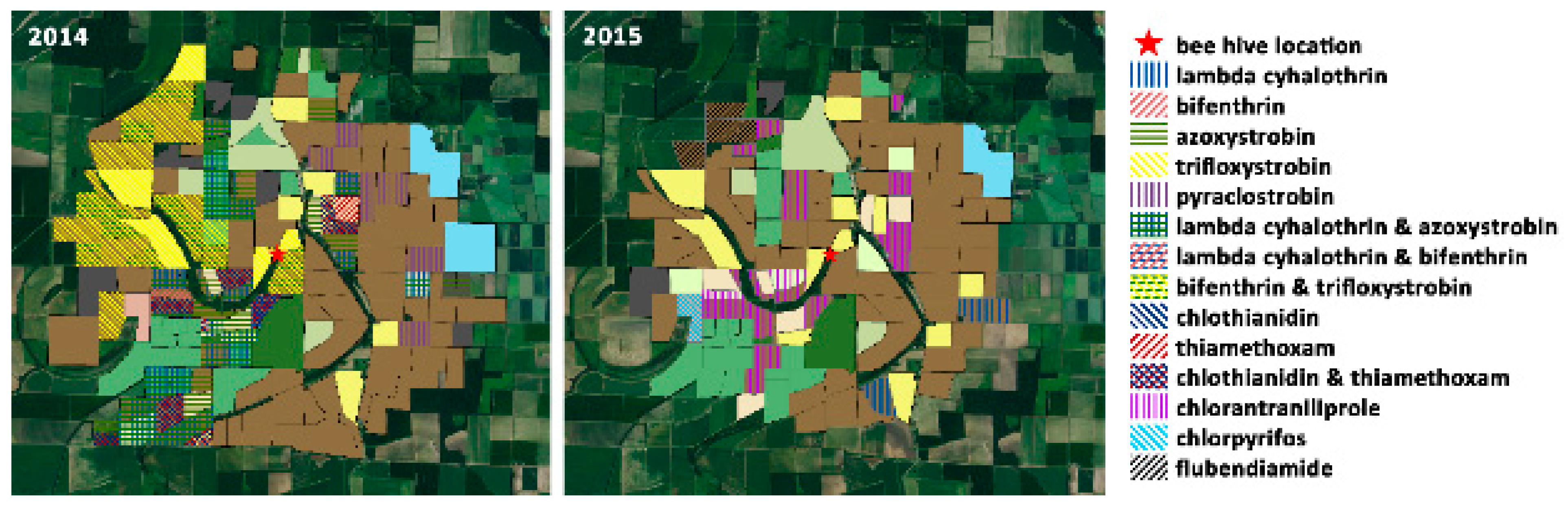
| Land Use | Total Acreage | % Acreage | |||
|---|---|---|---|---|---|
| 2014 | 2015 | 2014 | 2015 | 2-Year Average | |
| Soybean | 7489 | 5285 | 61.6 | 52.5 | 57.1 |
| Rice | 1110 | 1088 | 9.1 | 10.8 | 10 |
| Corn | 1005 | 849 | 8.3 | 8.4 | 8.4 |
| Cotton | 443 | 317 | 3.6 | 3.2 | 3.4 |
| Grain Sorghum | 92 | 91 | 0.8 | 0.9 | 0.9 |
| Green Beans | 0 | 306 | 0 | 3 | 1.5 |
| Total Crop Acreage | 10,139 | 7936 | 83.4 | 78.9 | 81.2 |
| Fish Ponds | 396 | 396 | 3.9 | 3.9 | 3.9 |
| Uncultivated Land | 1625 | 1731 | 12.7 | 17.2 | 15 |
| Total Acreage | 12,160 | 10,063 | 100 | 100 | 100 |
| Year | Pesticide | Class * | Number of Acres of Each Crop Treated by Pesticide Listed | Total Acres Treated | Percentage Surveyed Landscape Treated | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Soybean | Corn | Rice | Grain Sorghum | Cotton | Green Bean | |||||
| 2014 | Thiamethoxam | i-neo | 3677 | 789 | 669 | 92 | 264 | 0 | 5491 | 45.2 |
| Imidacloprid | i-neo | 884 | 81 | 0 | 0 | 203 | 0 | 1168 | 9.6 | |
| Clothianidin | i-neo | 1054 | 81 | 0 | 0 | 11 | 0 | 1146 | 9.4 | |
| Dimethoate | I-op | 54 | 0 | 0 | 0 | 0 | 0 | 54 | 0.4 | |
| Cypermethrin | I-py | 33 | 0 | 0 | 0 | 61 | 0 | 94 | 0.8 | |
| Lambda-Cyhalothrin | i-pyr | 685 | 0 | 347 | 0 | 192 | 0 | 1224 | 10.1 | |
| Bifenthrin | i-pyr | 319 | 81 | 0 | 0 | 11 | 0 | 411 | 3.4 | |
| Chlorantraniliprole | i-ry | 319 | 50 | 0 | 0 | 72 | 0 | 441 | 3.6 | |
| Flonicamid | i-u | 175 | 0 | 0 | 0 | 10 | 0 | 185 | 1.5 | |
| Novaluron | igr | 285 | 81 | 0 | 0 | 11 | 0 | 377 | 3.1 | |
| Fludioxonil | f | 3637 | 868 | 669 | 92 | 192 | 0 | 5458 | 44.9 | |
| Mefenoxam | f | 3637 | 868 | 669 | 92 | 192 | 0 | 5458 | 44.9 | |
| Azoxystrobin | f | 1608 | 0 | 347 | 0 | 323 | 0 | 2278 | 18.7 | |
| Prothioconizole | f | 1567 | 509 | 62 | 0 | 0 | 0 | 2138 | 17.6 | |
| Trifloxystrobin | f | 1567 | 509 | 62 | 0 | 0 | 0 | 2138 | 17.6 | |
| Metalaxyl | f | 564 | 0 | 0 | 0 | 131 | 0 | 695 | 5.7 | |
| Tebuconazole | f | 564 | 0 | 0 | 0 | 131 | 0 | 695 | 5.7 | |
| Tiabendazole | f | 519 | 0 | 0 | 0 | 0 | 0 | 519 | 4.3 | |
| Pyraclostrobin | f | 479 | 0 | 0 | 0 | 0 | 0 | 479 | 3.9 | |
| Propiconazole | f | 0 | 0 | 292 | 0 | 0 | 0 | 292 | 2.4 | |
| 2015 | Thiamethoxam | i - neo | 2965 | 0 | 344 | 0 | 317 | 225 | 3851 | 38.3 |
| Clothianidin | i - neo | 0 | 849 | 0 | 0 | 317 | 0 | 1166 | 11.6 | |
| Acephate | i - op | 0 | 0 | 0 | 0 | 317 | 0 | 317 | 3.2 | |
| Chlorpyrifos | i - op | 0 | 0 | 0 | 91 | 0 | 0 | 91 | 0.9 | |
| Bifenthrin | i - pyr | 0 | 0 | 0 | 0 | 317 | 0 | 317 | 3.2 | |
| Lambda-Cyhalothrin | i - pyr | 199 | 0 | 0 | 0 | 0 | 0 | 199 | 2 | |
| Chlorantraniliprole | i - ry | 768 | 0 | 0 | 0 | 317 | 93 | 1178 | 11.7 | |
| Flubendiamide | i - ry | 256 | 0 | 0 | 0 | 0 | 0 | 256 | 2.5 | |
| Novaluron | igr | 0 | 0 | 0 | 0 | 317 | 0 | 317 | 3.2 | |
| Fludioxonil | f | 2197 | 0 | 0 | 0 | 0 | 132 | 2329 | 23.1 | |
| Mefenoxam | f | 2197 | 0 | 0 | 0 | 0 | 132 | 2329 | 23.1 | |
| Azoxystrobin | f | 877 | 312 | 745 | 0 | 0 | 306 | 2240 | 22.3 | |
| Propiconazole | f | 0 | 312 | 344 | 0 | 0 | 0 | 656 | 6.5 | |
| Land Use | Total Acreage | % Acreage |
|---|---|---|
| Woodland | 7489 | 54.0 |
| Grass/Pasture | 1110 | 42.5 |
| Fish Ponds | 1005 | 3.5 |
| Wheat | 443 | 1.2 |
| Total Acreage | 8043 | 100 |
| Compound | Class * | Level of Detection (ppb) | Beeswax Foundation | Package Bees |
|---|---|---|---|---|
| coumaphos | a | 5 | 323.5 | 59 |
| fluvalinate | a | 1 | 273 | 136.5 |
| chlorpyriphos | i | 1 | 2.6 | 0 |
| hexythiazox | igr | 30 | trace | 0 |
| vinclozolin | f | 1 | trace | 0 |
| atrazine | h | 6 | 0 | 96.9 |
| Pesticide | Class * | Level of Detection (ppb) | 2014 | 2015 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Low-Ag | High-Ag | High-Ag | ||||||||
| New Wax | Honey | Pollen | New Wax | Honey | Pollen | New Wax | Bees | |||
| Coumaphos | a | 5 | 158.85 (95.38) | 0 | 0 | 103.75 (73.08) | 0 | 0 | 0 | 0 |
| Coumaphos Oxon *** | a | 5 | 1.28 (2.55) | 0 | 0 | trace | 0 | 0 | 0 | 0 |
| Fluvalinate | a | 1 | 128.53 (61.1) | 0 | 0 | 63 (73.52) | 0 | 0 | 0 | 0 |
| Amitraz | a | 4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| DMA ** | a | 50 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 297.5 (595) |
| DMPF ** | a | 10 | 0 | 0 | 0 | 0 | 13.05 (15.66) | 0.38 (0.25) | 769.75 (373.05) | trace |
| Thymol | a | 50 | trace | 0 | 0 | 0 | 0 | 0 | 0 | 747.5 (1495) |
| Bifenthrin | i | 2 | 37 (30.2) | 0 | 4.98 (9.95) | 3.75 (4.37) | 0 | 2.05 (4.1) | 14.3 (3.03) | 0 |
| Chlorpyrifos | i | 1 | 0.68 (1.35) | 0 | 0 | 0.55 (1.1) | 0 | 0 | 0 | 0 |
| Cyhalothrin | i | 1 | 0.55 (1.1) | 0 | 3.78 (0.79) | 0 | 0 | 2.48 (2.94) | 0 | 0 |
| Dimethoate | i | 50 | 0.25 (0.5) | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Flubendiamide | i | 25 | 0 | 48.7 (68.87) | 0 | 0 | 0 | 0 | 0 | 0 |
| Methyl Parathion | i | 2 | 0.25 (0.5) | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Hexythiazox | igr | 30 | 0.25 (0.5) | 0 | 0 | 0.5 (0.58) | 0 | 0 | 0 | 0 |
| Azoxystrobin | f | 2 | 1.13 (2.25) | 0 | 30.25 (36.07) | 2.13 (4.25) | 0 | 0 | 0 | 0 |
| Carbendazim | f | 5 | 0 | 0 | 0 | 0 | 0 | 0 | 0.25 (0.29) | 0 |
| Chlorothalonil | f | 30 | 0 | 0 | 0 | 0.5 (0.58) | 0 | 0 | 0 | 0 |
| Metalaxyl | f | 2 | 1.55 (3.1) | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Trifloxystrobin | f | 1 | 0.5 (0.58) | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Vinclozolin | f | 1 | 0 | 0 | 0 | 0.25 (0.5) | 0 | 0 | 0 | 0 |
| Atrazine | h | 6 | 2.35 (4.7) | 0 | 0 | 0 | 0 | 0 | 0.25 (0.29) | 0 |
| Metolachlor | h | 6 | 0 | 0 | 0 | 0 | 0 | 0 | 241.25 (311.42) | 0 |
| Metribuzin | h | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 10.9 (5.01) | 0 |
| Pendimethalin | h | 6 | 8.8 (16.94) | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Tribufos | d | 2 | 0 | 0 | 3.9 (7.8) | 0 | 0 | 0 | 8.48 (16.95) | 0 |
| Compound | LOD | Compound | LOD | Compound | LOD |
|---|---|---|---|---|---|
| 1-Naphthol | 10 | Dinotefuran | 2 | Parathion methyl | 2 |
| 3-Hydroxycarbofuran | 10 | Diphenamid | 20 | Permethrin total | 10 |
| 4,4 dibromobenzophenone | 4 | Endosulfan I | 2 | Phenothrin | 10 |
| 4-Hydroxychlorothalonil | 50 | Endosulfan II | 2 | Phorate | 50 |
| Acephate | 50 | Endosulfan sulfate | 2 | Phosalone | 10 |
| Acetamiprid | 2 | Endrin | 10 | Phosmet | 10 |
| Acetochlor | 50 | Epoxiconazole | 1 | Piperonyl butoxide | 50 |
| Alachlor | 10 | Esfenvalerate | 2 | Pirimiphos methyl | 20 |
| Aldicarb | 4 | Ethion | 10 | Prallethrin | 4 |
| Aldicarb sulfone | 2 | Ethofumesate | 10 | Profenofos | 10 |
| Aldicarb sulfoxide | 20 | Etoxazole | 1 | Pronamide | 1 |
| Aldrin | 10 | Etridiazole | 50 | Propachlor | 10 |
| Allethrin | 10 | Famoxadone | 20 | Propanil | 10 |
| Amicarbazone | 30 | Fenamidone | 10 | Propargite | 10 |
| Azinphos methyl | 6 | Fenbuconazole | 10 | Propazine | 20 |
| Bendiocarb | 10 | Fenhexamid | 6 | Propetamphos | 4 |
| Benoxacor | 20 | Fenoxaprop-ethyl | 20 | Propham | 20 |
| BHC alpha | 4 | Fenpropathrin | 10 | Propiconazole | 20 |
| Bifenazate | 20 | Fenpyroximate | 5 | Pymetrozine | 20 |
| Boscalid | 4 | Fenthion | 10 | Pyraclostrobin | 15 |
| Bromuconazole | 20 | Fipronil | 10 | Pyrethrins | 50 |
| Buprofezin | 20 | Flonicamid | 8 | Pyridaben | 10 |
| Captan | 10 | Fludioxonil | 20 | Pyrimethanil | 20 |
| Carbaryl | 30 | Fluoxastrobin | 4 | Pyriproxyfen | 10 |
| Carbofuran | 10 | Fluridone | 10 | Quinoxyfen | 10 |
| Carboxin | 4 | Flutolanil | 4 | Quintozene (PCNB) | 1 |
| Carfentrazone ethyl | 1 | Heptachlor epoxide | 10 | Resmethrin total | 5 |
| Chlorfenopyr | 1 | Heptachlor | 4 | Sethoxydim | 2 |
| Chlorfenvinphos | 6 | Hexachlorobenzene (HCB) | 1 | Simazine | 50 |
| Chlorferone | 50 | Hydroprene | 20 | Spinosad | 50 |
| Chlorpropham (CIPC) | 40 | Imazalil | 20 | Spirodiclofen | 2 |
| Clofentezine | 100 | Imidacloprid 5-hydroxy | 25 | Spiromesifen | 10 |
| Clothianidin | 1 | Imidacloprid | 1 | Tebuconazole | 8 |
| Cyfluthrin | 4 | Imidacloprid olefin | 10 | Tebufenozide | 10 |
| Cypermethrin | 4 | Indoxacarb | 3 | Tebuthiuron | 2 |
| Cyphenothrin | 20 | Iprodione | 50 | Tefluthrin | 1 |
| Cyprodinil | 1 | Lindane | 4 | Tetrachlorvinphos | 4 |
| DDD p,p’ | 4 | Linuron | 20 | Tetraconazole | 6 |
| DDE p,p’ | 2 | Malathion | 4 | Tetradifon | 1 |
| DDT p,p’ | 4 | Methamidophos | 4 | Tetramethrin | 10 |
| Deltamethrin | 50 | Methidathion | 10 | Thiabendazole | 1 |
| Diazinon | 5 | Methomyl | 10 | Thiacloprid | 1 |
| Dichlorvos (DDVP) | 50 | Methoxyfenozide | 10 | Thiamethoxam | 1 |
| Dicloran | 1 | MGK-264 | 50 | THPI | 50 |
| Dicofol | 1 | MGK-326 | 10 | Triadimefon | 2 |
| Dieldrin | 10 | Myclobutanil | 15 | Triadimenol | 45 |
| Difenoconazole | 10 | Norflurazon | 6 | Triflumizole | 50 |
| Diflubenzuron | 10 | Oxamyl | 5 | Triticonazole | 10 |
| Dimethenamid | 10 | Oxyfluorfen | 1 | ||
| Dimethomorph | 20 | Paradichlorobenzene | 10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zawislak, J.; Adamczyk, J.; Johnson, D.R.; Lorenz, G.; Black, J.; Hornsby, Q.; Stewart, S.D.; Joshi, N. Comprehensive Survey of Area-Wide Agricultural Pesticide Use in Southern United States Row Crops and Potential Impact on Honey Bee Colonies. Insects 2019, 10, 280. https://doi.org/10.3390/insects10090280
Zawislak J, Adamczyk J, Johnson DR, Lorenz G, Black J, Hornsby Q, Stewart SD, Joshi N. Comprehensive Survey of Area-Wide Agricultural Pesticide Use in Southern United States Row Crops and Potential Impact on Honey Bee Colonies. Insects. 2019; 10(9):280. https://doi.org/10.3390/insects10090280
Chicago/Turabian StyleZawislak, Jon, John Adamczyk, Donald R. Johnson, Gus Lorenz, Joe Black, Quinton Hornsby, Scott D. Stewart, and Neelendra Joshi. 2019. "Comprehensive Survey of Area-Wide Agricultural Pesticide Use in Southern United States Row Crops and Potential Impact on Honey Bee Colonies" Insects 10, no. 9: 280. https://doi.org/10.3390/insects10090280
APA StyleZawislak, J., Adamczyk, J., Johnson, D. R., Lorenz, G., Black, J., Hornsby, Q., Stewart, S. D., & Joshi, N. (2019). Comprehensive Survey of Area-Wide Agricultural Pesticide Use in Southern United States Row Crops and Potential Impact on Honey Bee Colonies. Insects, 10(9), 280. https://doi.org/10.3390/insects10090280







