Temporal Dynamics of Host Use by Drosophila suzukii in California’s San Joaquin Valley: Implications for Area-Wide Pest Management
Abstract
1. Introduction
2. Materials and Methods
2.1. Fruit Sampling
2.2. Laboratory Tests
2.2.1. Insects
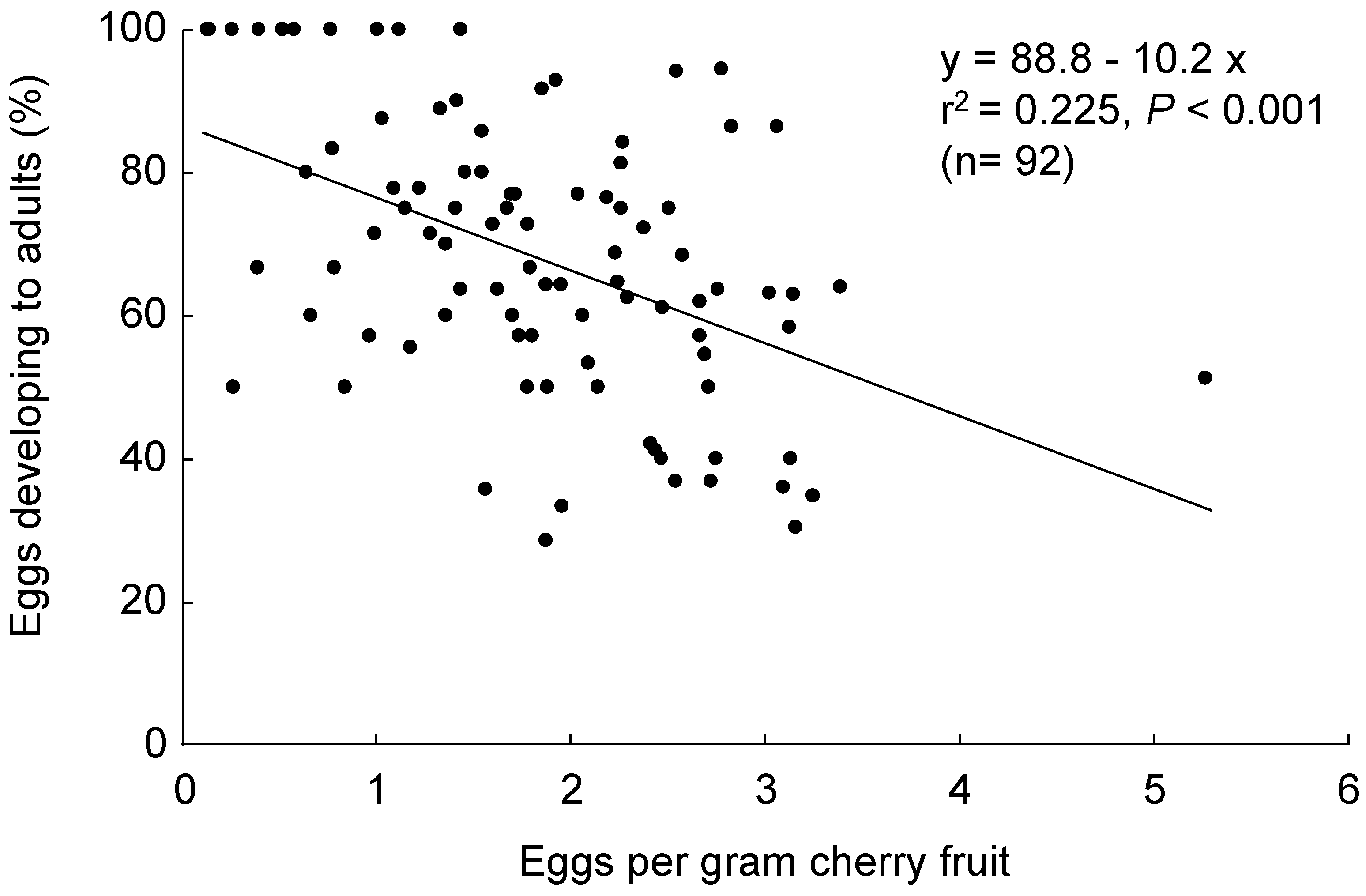
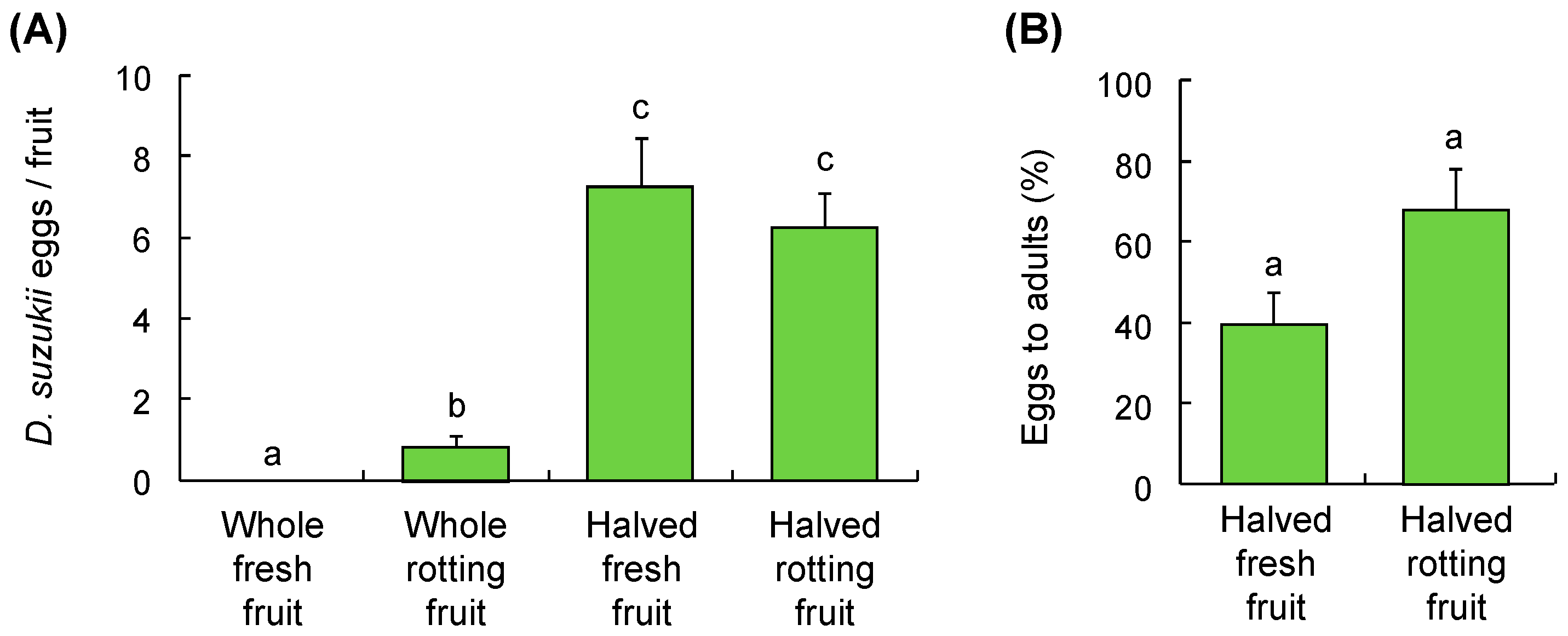
2.2.2. Host Suitability
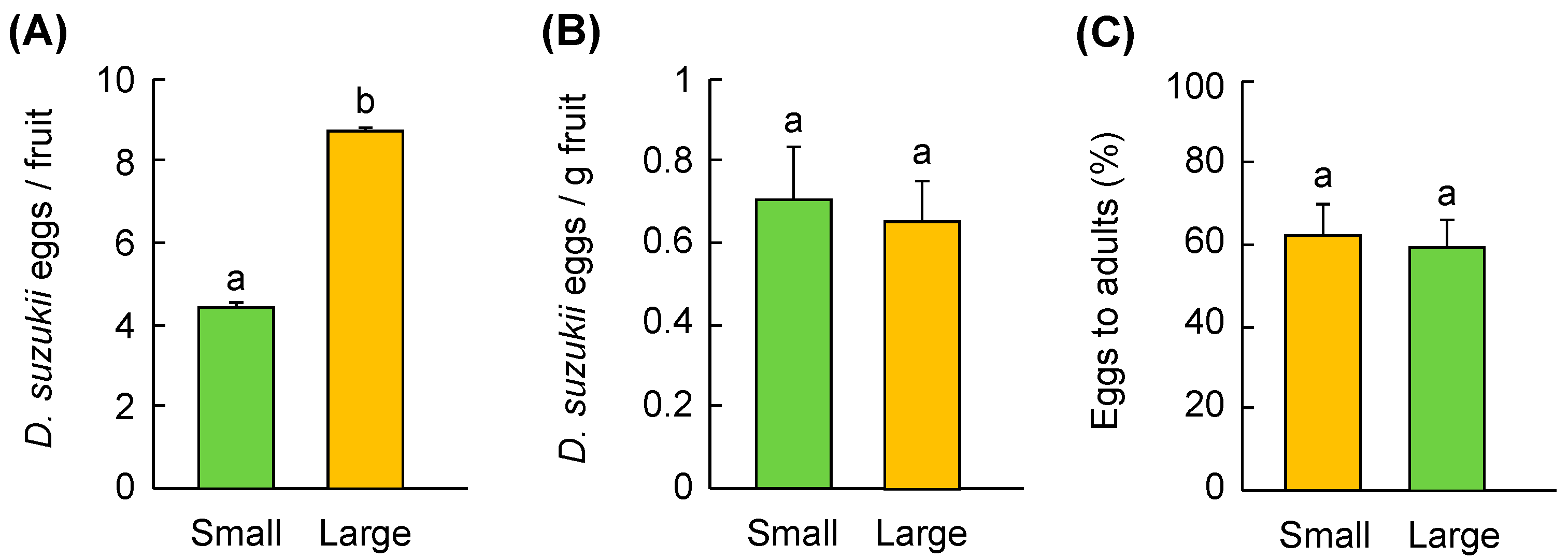
2.2.3. Fruit Size Preference
2.2.4. Egg Density Effect
2.2.5. Citrus Test
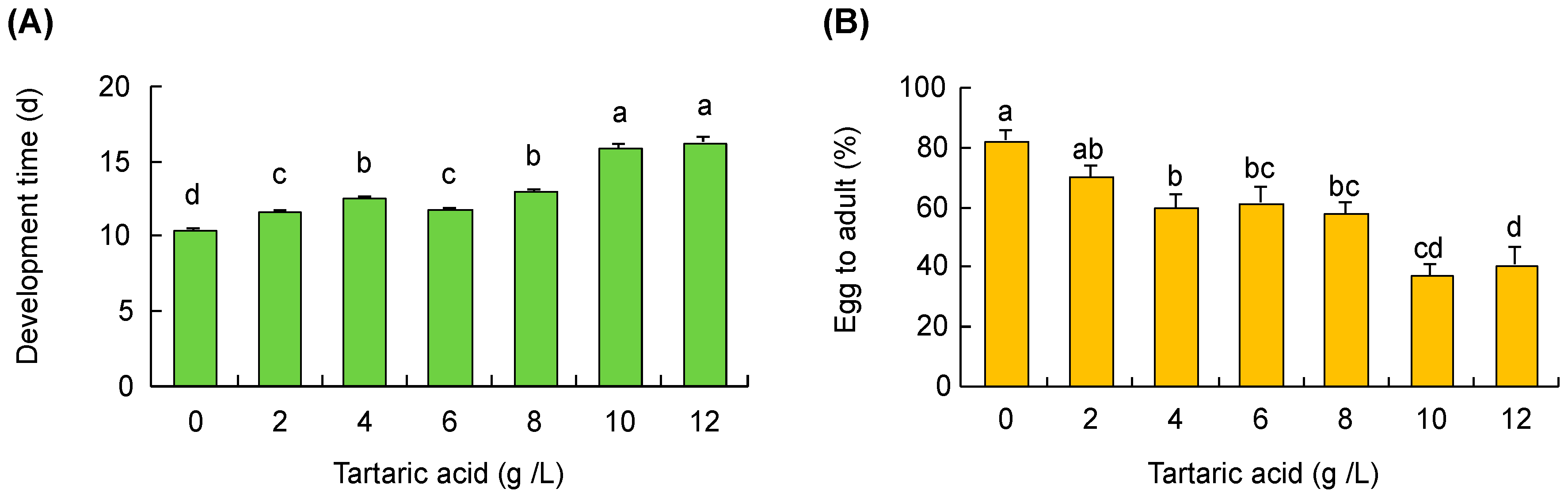
2.2.6. Grape Acidity Effect
2.3. Monitoring of Adult Populations at Different Elevations
2.4. Data Analysis
3. Results
3.1. Fruit Sampling
3.2. Host Suitability
3.3. Fruit Size Preference
3.4. Orange Test
3.5. Grape Acidity
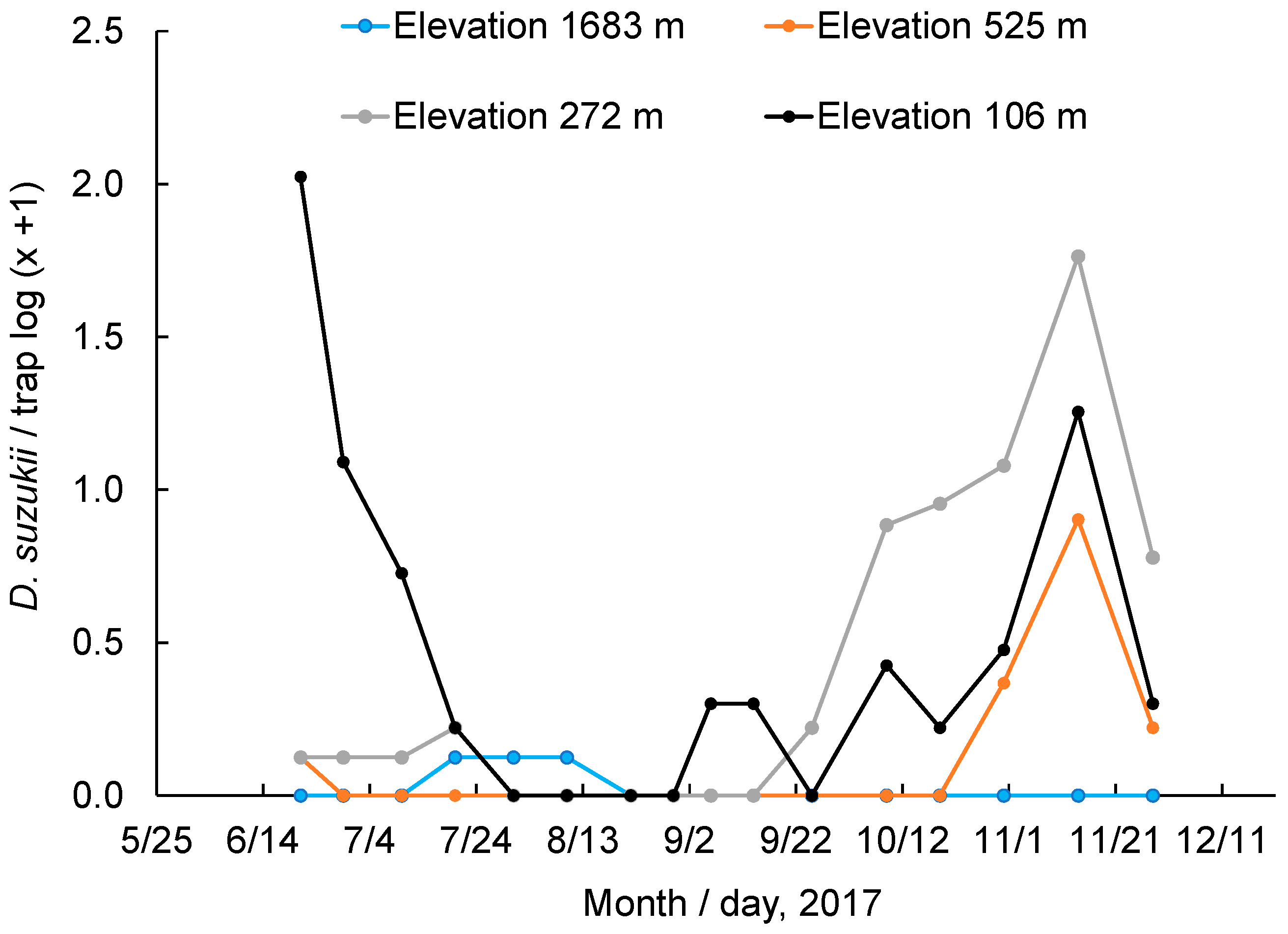
3.6. Monitoring of Adult Populations at Different Elevations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Asplen, M.K.; Anfora, G.; Biondi, A.; Choi, D.S.; Chu, D.; Daane, K.M.; Gibert, P.; Gutierrez, A.P.; Hoelmer, K.A.; Hutchison, W.D.; et al. Invasion biology of spotted wing drosophila (Drosophila suzukii): A global perspective and future priorities. J. Pest Sci. 2015, 88, 469–494. [Google Scholar] [CrossRef]
- Cini, A.; Anfora, G.; Escudero-Colomar, L.A.; Grassi, A.; Santosuosso, U.; Seljak, G.; Papini, A. Tracking the invasion of the alien fruit pest Drosophila suzukii in Europe. J. Pest Sci. 2014, 87, 559–566. [Google Scholar] [CrossRef]
- Walsh, D.B.; Bolda, M.P.; Goodhue, R.E.; Dreves, A.J.; Lee, J.C.; Bruck, D.J.; Walton, V.M.; O’Neal, S.D.; Zalom, F.G. Drosophila suzukii (Diptera: Drosophilidae): Invasive pest of ripening soft fruit expanding its geographic range and damage potential. J. Intergr. Pest Manag. 2011, 2, G1–G7. [Google Scholar] [CrossRef]
- Andreazza, F.; Bernardi, D.; dos Santos, R.S.S.; Garcia, F.R.M.; Oliveira, E.E.; Botton, M.; Nava, D.E. Drosophila suzukii in Southern neotropical region: Current status and future perspectives. Neotrop. Entomol. 2017, 46, 591–605. [Google Scholar] [CrossRef] [PubMed]
- Dagatti, C.V.; Marcucci, B.; Herrera, M.E.; Becerra, V.C. First record of Drosophila suzukii (Diptera: Drosophilidae) associated to blackberry in Mendoza, Argentina. Rev. Soc. Entomol. Argent. 2018, 77, 26–29. [Google Scholar] [CrossRef]
- Burrack, H.J.; Fernandez, G.E.; Spivey, T.; Kraus, D.A. Variation in selection and utilization of host crops in the field and laboratory by Drosophila suzukii Matsumara (Diptera: Drosophilidae), an invasive frugivore. Pest Manag. Sci. 2013, 69, 1173–1180. [Google Scholar] [CrossRef] [PubMed]
- Kanzawa, T. Studies on Drosophila suzukii Mats. Kofu, Yamanashi Agricultural Experimental Station 49. Rev. Appl. Entomol. 1939, 29, 622. [Google Scholar]
- Kenis, M.; Tonina, L.; Eschen, R.; van der Sluis, B.; Sancassani, M.; Mori, N.; Haye, T.; Helsen, H. Non-crop plants used as hosts by Drosophila suzukii in Europe. J. Pest Sci. 2016, 89, 735–748. [Google Scholar] [CrossRef]
- Lee, J.C.; Bruck, D.J.; Curry, H.; Edwards, D.L.; Haviland, D.R.; van Steenwyk, R.; Yorgey, B. The susceptibility of small fruits and cherries to the spotted wing drosophila, D. suzukii. Pest Manag. Sci. 2011, 67, 1358–1367. [Google Scholar] [CrossRef]
- Lee, J.C.; Dreves, A.J.; Cave, A.M.; Kawai, S.; Isaacs, R.; Miller, J.C.; van Timmeren, S.; Bruck, D.J. Infestation of wild and ornamental noncrop fruits by Drosophila suzukii (Diptera: Drosophilidae). Ann. Entomol. Soc. Am. 2015, 108, 117–129. [Google Scholar] [CrossRef]
- Poyet, M.; Le Roux, V.; Gibert, P.; Meirland, A.; Prevost, G.; Eslin, P.; Chabrerie, O. The wide potential trophic niche of the Asiatic fruit fly Drosophila suzukii: The key of its invasion success in temperate Europe? PLoS ONE 2015, 10, e0142785. [Google Scholar] [CrossRef] [PubMed]
- Tochen, S.; Dalton, D.T.; Wiman, N.G.; Hamm, C.; Shearer, P.W.; Walton, V.M. Temperature-related development and population parameters for Drosophila suzukii (Diptera: Drosophilidae) on cherry and blueberry. Environ. Entomol. 2014, 43, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Wiman, N.W.; Anfora, G.; Burrack, H.J.; Chiu, J.; Daane, K.M.; Dalton, D.T.; Grassi, A.; Anfora, G.; Burrack, H.J.; Chiu, J.C.; et al. Integrating temperature-dependent life table data into a matrix projection model for Drosophila suzukii population estimation. PLoS ONE 2014, 9, e106909. [Google Scholar] [CrossRef] [PubMed]
- Wiman, N.G.; Dalton, D.T.; Anfora, G.; Biondi, A.; Chiu, J.C.; Daane, K.M.; Gerdeman, B.; Gottardello, A.; Hamby, K.A.; Isaacs, R.; et al. Drosophila suzukii population response to environment and management strategies. J. Pest Sci. 2016, 89, 653–665. [Google Scholar] [CrossRef] [PubMed]
- Goodhue, R.E.; Bolda, M.; Farnsworth, D.; Williams, J.C.; Zalom, F.G. Spotted wing drosophila infestation of California strawberries and raspberries: Economic analysis of potential revenue losses and control costs. Pest Manag. Sci. 2011, 67, 1396–1402. [Google Scholar] [CrossRef] [PubMed]
- Mazzi, D.; Bravin, E.; Meraner, M.; Finger, R.; Kuske, S. Economic impact of the introduction and establishment of Drosophila suzukii on sweet cherry production in Switzerland. Insects 2017, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Alnajjar, G.; Collins, J.; Drummond, F.A. Behavioral and preventative management of Drosophila suzukii Matsumura (Diptera: Drosophilidae) in Maine wild blueberry (Vaccinium angustifolium Aiton) through attract and kill trapping and insect exclusion-netting. Intl. J. Entomol. Nematol. 2017, 3, 51–61. [Google Scholar]
- Hampton, E.; Koski, C.; Barsoian, O.; Faubert, H.; Cowles, R.S.; Alm, S.R. Use of early ripening cultivars to avoid infestation and mass trapping to manage Drosophila suzukii (Diptera: Drosophilidae) in Vaccinium corymbosum (Ericales: Ericaceae). J. Econ. Entomol. 2014, 107, 1849–1857. [Google Scholar] [CrossRef] [PubMed]
- Haye, T.; Girod, P.; Cuthbertson, A.G.S.; Wang, X.G.; Daane, K.M.; Hoelmer, K.A.; Baroffio, C.; Zhang, J.P.; Desneux, N. Current SWD IPM tactics and their practical implementation in fruit crops across different regions around the world. J. Pest Sci. 2016, 89, 643–651. [Google Scholar] [CrossRef]
- Lee, J.C.; Wang, X.G.; Daane, K.M.; Hoelmer, K.A.; Isaacs, R.; Sial, A.A.; Walton, V.M. Biological control of spotted-wing drosophila—Current and pending tactics. J. Integr. Pest Manag. 2019, 10, 13. [Google Scholar] [CrossRef]
- Moses, J.; Hanson, E.; Fanning, P.; Isaacs, R. Rapid harvest schedules and fruit removal as non-chemical approaches for managing spotted wing Drosophila. J. Pest Sci. 2018, 91, 219–226. [Google Scholar]
- Rice, K.B.; Short, B.D.; Leskey, T.C. Development of an attract and kill strategy for Drosophila suzukii (Diptera: Drosophilidae): Evaluation of attracticidal spheres under laboratory and field conditions. Environ. Entomol. 2017, 110, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Schetelig, M.F.; Lee, K.Z.; Otto, S.; Talmann, L.; Stokl, J.; Degenkolb, T.; Vilcinskas, A.; Halitschke, R. Environmentally sustainable pest control options for Drosophila suzukii. J. Appl. Entomol. 2018, 142, 3–17. [Google Scholar] [CrossRef]
- Wallingford, A.K.; Cha, D.H.; Loeb, G.M. Evaluating a push–pull strategy for management of Drosophila suzukii Matsumura in red raspberry. J. Appl. Entomol. 2018, 74, 120–125. [Google Scholar] [CrossRef]
- Van Timmeren, S.; Mota-Sanchez, D.; Wise, J.C.; Isaacs, R. Baseline susceptibility of spotted wing Drosophila (Drosophila suzukii) to four key insecticide classes. Pest Manag. Sci. 2018, 74, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Van Timmeren, S.; Isaacs, R. Control of spotted wing drosophila, Drosophila suzukii, by specific insecticides and by conventional and organic crop protection programs. Crop Prot. 2013, 54, 126–133. [Google Scholar] [CrossRef]
- Arnó, J.; Solà, M.; Riudavets, J.; Gabarra, R. Population dynamics, non-crop hosts and fruit susceptibility of Drosophila suzukii in Northeast Spain. J. Pest Sci. 2016, 89, 713–723. [Google Scholar] [CrossRef]
- Bal, H.K.; Adams, C.; Grieshop, M. Evaluation of off-season potential breeding sources for spotted wing drosophila (Drosophila suzukii Matsumura) in Michigan. J. Econ. Entomol. 2017, 110, 2466–2470. [Google Scholar] [CrossRef] [PubMed]
- Diepenbrock, L.M.; Swoboda-Bhattarai, K.A.; Burrack, H.J. Ovipositional preference, fidelity, and fitness of Drosophila suzukii in a co-occurring crop and non-crop system. J. Pest Sci. 2016, 89, 761–769. [Google Scholar] [CrossRef]
- Mitsui, H.; Beppu, K.; Kimura, M.T. Seasonal life cycles and resource uses of flower-and fruit-feeding drosophilid flies (Diptera: Drosophilidae) in central Japan. Entomol. Sci. 2010, 13, 60–67. [Google Scholar] [CrossRef]
- Klick, J.; Yang, W.Q.; Walton, V.M.; Dalton, D.T.; Hagler, J.R.; Dreves, A.J.; Lee, J.C.; Bruck, D.J. Distribution and activity of Drosophila suzukii in cultivated raspberry and surrounding vegetation. J. Appl. Entomol. 2016, 140, 37–46. [Google Scholar] [CrossRef]
- Tait, G.; Grassi, A.; Pfab, F.; Crava, C.M.; Dalton, D.T.; Magarey, R.; Ometto, L.; Vezzulli, S.; Stacconi, M.V.R.; Gottardello, A.; et al. Large-scale spatial dynamics of Drosophila suzukii in Trentino, Italy. J. Pest Sci. 2018, 91, 1213–1224. [Google Scholar] [CrossRef]
- Wong, J.S.; Cave, A.C.; Lightle, D.M.; Mahaffee, W.F.; Naranjo, S.E.; Wiman, N.G.; Woltz, J.M.; Lee, J.C. Drosophila suzukii flight performance reduced by starvation but not affected by humidity. J. Pest Sci. 2019, 91, 1269–1278. [Google Scholar] [CrossRef]
- Hennig, E.I.; Mazzi, D. Spotted wing drosophila in sweet cherry orchards in relation to forest characteristics, bycatch, and resource availability. Insects 2018, 9, 118. [Google Scholar] [CrossRef] [PubMed]
- Pelton, E.; Gratton, C.; Isaacs, R.; van Timmeren, S.; Blanton, A.; Guédot, C. Earlier activity of Drosophila suzukii in high woodland landscapes but relative abundance is unaffected. J. Pest Sci. 2016, 89, 725–733. [Google Scholar] [CrossRef]
- Briem, F.; Dominic, A.R.; Golla, B.; Hoffmann, C.; Englert, C.; Herz, A.; Vogt, H. Explorative data analysis of Drosophila suzukii trap catches from a seven-year monitoring program in southwest Germany. Insects 2018, 9, 125. [Google Scholar] [CrossRef]
- Drummond, F.; Ballman, E.; Collins, J. Population dynamics of spotted wing Drosophila (Drosophila suzukii Matsumara) in Maine wild blueberry (Vaccinium angustifolium Aiton). Insects 2019. [Google Scholar] [CrossRef]
- Grassi, A.; Gottardello, A.; Dalton, D.T.; Tait, G.; Rendon, D.; Ioriatti, C.; Gibeaut, D.; Rossi Stacconi, M.V.; Walton, V.M. Seasonal reproductive biology of Drosophila suzukii (Diptera: Drosophilidae) in temperate climates. Environ. Entomol. 2018, 47, 166–174. [Google Scholar] [CrossRef]
- Wang, X.G.; Stewart, T.J.; Biondi, A.; Chavez, B.A.; Ingels, C.; Caprile, J.; Grant, J.A.; Walton, V.M.; Daane, K.M. Population dynamics and ecology of Drosophila suzukii in Central California. J. Pest Sci. 2016, 89, 701–712. [Google Scholar] [CrossRef]
- Panel, A.D.C.; Zeeman, L.; van der Sluis, B.J.; van Elk, P.; Pannebakker, B.A.; Wertheim, B.; Helsen, H.H.M. Overwintered Drosophila suzukii are the main source for infestations of the first fruit crops of the season. Insects 2018, 9, 145. [Google Scholar] [CrossRef]
- Santoiemma, G.; Trivellato, F.; Caloi, V.; Mori, N.; Marini, L. Habitat preference of Drosophila suzukii across heterogeneous landscapes. J. Pest Sci. 2019, 92, 485–494. [Google Scholar] [CrossRef]
- Bolda, M.P.; Goodhue, R.E.; Zalom, F.G. Spotted wing drosophila: Potential economic impact of a newly established pest. Agric. Res. Econ. Update 2010, 13, 5–8. [Google Scholar]
- Hamby, K.A.; Bolda, M.P.; Sheehan, M.E.; Zalom, F.G. Seasonal monitoring for Drosophila suzukii (Diptera: Drosophilidae) in California commercial raspberries. Environ. Entomol. 2014, 43, 1008–1018. [Google Scholar] [CrossRef] [PubMed]
- Wilson, H.E.; Hamby, K.A.; Zalom, F.G. Host susceptibility of “French Prune” Prunus domestica to Drosophila suzukii (Diptera: Drosophilidae). Acta Hortic. 2013, 985, 249–254. [Google Scholar] [CrossRef]
- Yu, D.; Zalom, F.G.; Hamby, K.A. Host status and fruit odor response of Drosophila suzukii (Diptera: Drosophilidae) to figs and mulberries. J. Econ. Entomol. 2013, 106, 1932–1937. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.G.; Levy, K.; Nadel, H.; Johnson, M.W.; Blanchet, A.; Argov, Y.; Pickett, C.H.; Daane, K.M. Overwintering survival of olive fruit fly and two introduced parasitoids in California. Environ. Entomol. 2013, 42, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Harris, D.W.; Hamby, K.A.; Wilson, H.E.; Zalom, F.G. Seasonal monitoring of Drosophila suzukii (Diptera: Drosophilidae) in a mixed fruit production system. J. Appl. Entomol. 2014, 17, 857–864. [Google Scholar] [CrossRef]
- Haviland, D.R.; Caprile, J.L.; Rill, S.M.; Hamby, K.A.; Grant, J.A. Phenology of spotted wing drosophila in the San Joaquin Valley varies by season, crop and nearby vegetation. Calif. Agric. 2016, 70, 24–31. [Google Scholar] [CrossRef]
- Ioriatti, C.; Guzzon, R.; Anfora, G.; Ghidoni, F.; Mazzoni, V.; Villegas, T.R.; Dalton, D.T.; Walton, V.M. Drosophila suzukii (Diptera: Drosophilidae) contributes to the development of sour rot in grape. J. Econ. Entomol. 2018, 111, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Baser, N.; Broutou, O.; Verrastro, V.; Porcelli, F.; Ioriatti, C.; Anfora, G.; Mazzoni, V.; Stacconi, M.V.R. Susceptibility of table grape varieties grown in south-eastern Italy to Drosophila suzukii. J. Appl. Entomol. 2018. [Google Scholar] [CrossRef]
- Entling, W.; Anslinger, S.; Jarausch, B.; Michl, G.; Hoffm, C. Berry skin resistance explains oviposition preferences of Drosophila suzukii at the level of grape cultivars and single berries. J. Pest Sci. 2019, 92, 477–484. [Google Scholar] [CrossRef]
- Kim, M.J.; Kim, J.S.; Park, J.S.; Choi, D.-S.; Park, J.; Kim, I. Oviposition and development potential of the spotted-wing drosophila, Drosophila suzukii (Diptera: Drosophilidae), on uninjured Campbell Early grape. Entomol. Res. 2015, 45, 354–359. [Google Scholar] [CrossRef]
- Ioriatti, C.; Walton, V.; Dalton, D.; Anfora, G.; Grassi, A.; Maistri, S.; Mazzoni, V. Drosophila suzukii (Diptera: Drosophilidae) and its potential impact to wine grapes during harvest in two cool climate wine grape production regions. J. Econ. Entomol. 2015, 108, 1148–1155. [Google Scholar] [CrossRef] [PubMed]
- Pelton, E.; Gratton, C.; Guédot, C. Susceptibility of cold hardy grapes to Drosophila suzukii (Diptera: Drosophilidae). J. Appl. Entomol. 2017, 141, 644–652. [Google Scholar] [CrossRef]
- Rezazadeh, A.; Sampson, B.J.; Stafne, E.T.; Marshall-Shaw, D.; Stringer, S.J.; Hummer, K. Susceptibility of bunch grape and muscadine cultivars to berry splitting and spotted-wing Drosophila oviposition. Am. J. Enol. Vitic. 2018. [Google Scholar] [CrossRef]
- Sial, A.A.; Grant, J.A. Potential of muscadine grapes as a viable host of Drosophila suzukii (Diptera: Drosophilidae) in blueberry-producing regions of the Southeastern United States. J. Econ. Entomol. 2016, 109, 1261–1266. [Google Scholar]
- Van Timmeren, S.; Isaacs, R. Drosophila suzukii in Michigan vineyards, and the first report of Zaprionus indianus from this region. J. Appl. Entomol. 2014, 138, 519–527. [Google Scholar] [CrossRef]
- Sasaki, M.; Sato, R. Bionomics of the cherry drosophila, Drosophila suzukii Matsumura (Diptera: Drosophilidae) in Futeushima prefecture (Japan). Ann. Rep. Soc. Plant Prot. North Jpn. 1995, 46, 164–172. [Google Scholar]
- Stewart, T.E.; Wang, X.G.; Molinar, A.; Daane, K.M. Factors limiting peach as a potential host for spotted wing drosophila. J. Econ. Entomol. 2014, 107, 1771–1779. [Google Scholar] [CrossRef]
- Lasa, R.; Tadeo, E.; Dinorín, L.A.; Lima, I.; Williams, T. Fruit firmness, superficial damage and location modulate infestation by Drosophila suzukii and Zaprionus indianus (Diptera: Drosophilidae): The case of guava Psidium guajava L., in Veracruz, Mexico. Entomol. Exp. Appl. 2017, 162, 4–12. [Google Scholar] [CrossRef]
- Little, C.M.; Chapman, T.W.; Moreau, D.L.; Hillier, N.K. Susceptibility of selected boreal fruits and berries to the invasive pest Drosophila suzukii (Diptera: Drosophilidae). Pest Manag. Sci. 2017, 73, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J. The Oxford Companion to Wine, 3rd ed.; Oxford University Press: Oxford, UK, 2006. [Google Scholar]
- Kliewer, W.M.; Howarth, L.; Omori, M. Concentrations of tartaric acid and malic acids and their salts in Vitis vinifera grapes. Am. J. Enol. Vitic. 1967, 18, 42–54. [Google Scholar]
- McCarthy, B.; Gohil, H.; Gu, S. Organic acids and pH of Cabernet Sauvignon fruit forced to ripen under cooler conditions in a warm region. Am. J. Enol. Vitic. 2012, 63, 460–461. [Google Scholar]
- Calflora. Information on California Plants for Education, Research and Conservation, Berkeley, California. Available online: http://www.calflora.org (accessed on 13 February 2018).
- Dalton, D.T.; Walton, V.M.; Shearer, P.W.; Walsh, D.B.; Caprile, J.; Isaacs, R. Laboratory survival of Drosophila suzukii under simulated winter conditions of the Pacific Northwest and seasonal field trapping in five primary regions of small and stone fruit production in the United States. Pest Manag. Sci. 2011, 67, 1368–1374. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.G.; Nadel, H.; Johnson, M.W.; Daane, K.M.; Hoelmer, K.; Walton, V.M.; Pickett, C.P.; Sime, K.R. Crop domestication relaxes both bottom-up and top-down effects on a specialist herbivore. Basic Appl. Ecol. 2009, 10, 216–227. [Google Scholar] [CrossRef]
- Kaçar, G.; Wang, X.G.; Stewart, T.J.; Daane, K.M. Overwintering survival of Drosophila suzukii (Diptera: Drosophilidae) and the effect of food on adult survival in California’s San Joaquin Valley. Environ. Entomol. 2016, 45, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, D.E.; Sisterson, M.S.; Walse, S.S. Quantifying host potentials: Indexing postharvest fresh fruits for spotted wing drosophila, Drosophila suzukii. PLoS ONE 2013, 8, e61227. [Google Scholar] [CrossRef] [PubMed]
- Silva-Soares, N.F.; Nogueira-Alves, A.; Beldade, P.; Mirth, C.K. Adaptation to new nutritional environments: Larval performance, foraging decisions, and adult oviposition choices in Drosophila suzukii. BMC Ecol. 2017, 17, 21. [Google Scholar] [CrossRef]
- Hamby, K.A.; Bellamy, D.; Chiu, J.C.; Lee, J.C.; Walton, V.M.; Wiman, N.G.; York, R.M.; Biondi, A. Biotic and abiotic factors impacting development, behavior, phenology, and reproductive biology of Drosophila suzukii. J. Pest Sci. 2016, 89, 605–619. [Google Scholar] [CrossRef]
- Jesse, A.; Hardin, J.A.; Kraus, D.A.; Burrack, H.J. Diet quality mitigates intraspecific larval competition in Drosophila suzukii. Entomol. Exp. Appl. 2015, 156, 59–65. [Google Scholar]
- Kinjo, H.; Kunimi, Y.; Ban, T.; Nakai, M. Oviposition efficacy of Drosophila suzukii (Diptera: Drosophilidae) on different cultivars of blueberry. J. Econ. Entomol. 2013, 106, 1767–1771. [Google Scholar] [CrossRef] [PubMed]
- Woltz, J.M.; Walton, V.M. Characterization and manipulation of fruit susceptibility to Drosophila suzukii. J. Pest Sci. 2016, 89, 771–780. [Google Scholar]
- Vursavus, K.; Kelebek, H.; Selli, S. A study on some chemical and physicomechanical properties of three sweet cherry varieties (Prunus avium L.) in Turkey. J. Food Eng. 2006, 74, 568–575. [Google Scholar] [CrossRef]
- Prokopy, R.J.; Roitberg, B.D. Fruit fly foraging behavior. In Fruit Flies: Their Biology, Natural Enemies and Control, World Crop Pests; Robinson, A.S., Hooper, G., Eds.; Elsevier Science Publications: Amsterdam, The Netherlands, 1989; Volume 3a, pp. 293–306. [Google Scholar]
- Daane, K.M.; Wang, X.G.; Biondi, A.; Miller, B.E.; Miller, J.C.; Riedl, H.; Shearer, P.W.; Guerrieri, E.; Giorgini, M.; Buffington, M.; et al. First exploration of parasitoids of Drosophila suzukii in South Korea as potential classical biological agents. J. Pest Sci. 2016, 89, 823–835. [Google Scholar] [CrossRef]
- Pfab, F.; Rossi Stacconi, M.V.; Anfora, G.; Grassi, A.; Walton, V.M.; Pugliese, A. Optimized timing of parasitoid release: A mathematical model for biological control of Drosophila suzukii. Theor. Ecol. 2018, 11, 489–501. [Google Scholar] [CrossRef]
- Rossi Stacconi, M.V.; Amiresmaeili, N.; Biondi, A.; Carli, C.; Caruso, S.; Dindo, M.L.; Francati, S.; Gottardello, A.; Grassi, A.; Lupi, D.; et al. Host location and dispersal ability of the cosmopolitan parasitoid Trichopria drosophilae released to control the invasive spotted-wing drosophila. Biol. Control 2018, 117, 188–196. [Google Scholar] [CrossRef]
| Family | Host Species | May | Jun | Jul | Aug | Sept | Oct | Nov | Dec to Apr |
|---|---|---|---|---|---|---|---|---|---|
| Rosaceae | Cherry (Prunus avium) | ||||||||
| Rosaceae | Peach (Prunus persica) | ||||||||
| Rosaceae | Nectarine (Prunus armeniaca) | ||||||||
| Rosaceae | Plum (Prunus domestica) | ||||||||
| Rosaceae | Pear (Pyrus domestica) | ||||||||
| Vitaceae | Raisin grape (Vitis vinifera) | ||||||||
| Moraceae | Fig (Ficus carica) | ||||||||
| Lythraceae | Pomegranate (Punica granatum) | ||||||||
| Rosaceae | Apple (Pyrus malus) | ||||||||
| Ebenaceae | Persimmon (Diospyros kaki) | ||||||||
| Rutaceae | Naval orange (Citrus C. × sinensis) | ||||||||
| Rutaceae | Mandarin orange (Citrus reticulata) | ||||||||
| Rosaceae | Loquat (Eriobotrya japonica) | ||||||||
| Rosaceae | Wild plum (Prunus subcordata) | ||||||||
| Loquat | Cactus (Optunia streptacantha) | ||||||||
| Rosaceae | Bitter cherry (Prunus emarginata) | ||||||||
| Rhamnaceae | Buckthorn (Frangula purshiana) |
| Fruit | Collection Years | Months Sampled | Intact Fruit | Damaged Fruit | ||
|---|---|---|---|---|---|---|
| No. of Fruit Collected | No. of D. suzukii Emerged | No. of Fruit Collected | No. of D. suzukii Emerged | |||
| Cherry | 2013–2015 | May–Jun | 582 | 372 | 1372 | 366 |
| Peach | 2013–2014 | Jun–Aug | 50 | 0 | 41 | 11 |
| Nectarine | 2013–2014 | Jun–Jul | 50 | 0 | 40 | 2 |
| Plum | 2013–2014 | Jul–Aug | 50 | 0 | 60 | 5 |
| Pear | 2013–2014 | Jul–Sept | 30 | 0 | 23 | 104 |
| Fig | 2013–2014 | Sept–Nov | 50 | 0 | 119 | 115 |
| Apple | 2013–2014 | Sept–Oct | 50 | 0 | 106 | 4 |
| Pomegranate | 2013–2014 | Sept–Nov | - | - | 100 | 12 |
| Raisin grape | 2013–2014 | Oct–Nov | 1020 | 0 | 20 | 0 |
| Persimmon | 2013–2014 | Oct–Dec | 50 | 0 | 15 | 1 |
| Mandarin | 2013–2014 | Oct–Apr | - | - | 55 | 2 |
| Naval orange | 2013–2014 | Oct–Apr | - | - | 20 | 1 |
| Loquat | 2014 | May–Jun | 50 | 0 | 15 | 5 |
| Wild plum | 2014 | Jul | 50 | 0 | - | - |
| Cherry plum | 2014 | Jul | 50 | 0 | - | - |
| Cactus | 2014 | Sept | 30 | 0 | 23 | 1 |
| Buckthorn | 2016 | Oct | 150 | 0 | - | - |
| Bitter cherry | 2016–2017 | Sept–Oct | 4,800 | 0 | - | - |
| Fruit | Fruit Firmness (gmm−1) 2 | Brix2 | n | Fruit Size (g) 2 | Eggs per Gram Fruit 2 | Eggs Developed to Adults (%) 2 |
|---|---|---|---|---|---|---|
| Cherry ‘Bing’ 1 | 76.8 ± 7.8 (15) c | 21.5 ± 0.4 (10) d | 25 | 8.56 ± 0.17 a | 1.22 ± 0.18 c | 63.4 ± 6.3 a |
| Pomegranate seed | 45.8 ± 3.2 (20) d | 17.0 ± 0.2 (20) b | 49 | 0.45 ± 0.01 g | 4.11 ± 0.26 b | 68.7 ± 5.3 a |
| Mandarin orange | - | 10.6 ± 0.4 (20) de | 53 | 2.37 ± 0.08 e | 1.80 ± 0.19 c | 51.2 ± 5.5 ab |
| Raisin grape | 84.8 ± 6.7 (20) c | 17.9 ± 0.2 (20) c | 52 | 1.76 ± 0.05 f | 2.17 ± 0.25 c | 26.5 ± 4.5 c |
| Table grape | 181.5 ± 10.0 (20) b | 21.2 ± 0.3 (20) c | 33 | 3.50 ± 0.09 d | 1.12 ± 0.25 c | 31.4 ± 6.1 c |
| Wine grape | 102.3 ± 6.3 (15) c | 23.6 ± 0.5 (20) b | 22 | 0.70 ± 0.03 g | 4.84 ± 1.17 ab | 4.5 ± 3.1 d |
| Wild plum | 295.4 ± 7.9 (15) a | 10.5 ± 0.2 (10) e | 25 | 9.71 ± 0.11 a | 1.10 ± 0.16 c | 53.1 ± 6.6 ab |
| Cactus | - | 11.5 ± 0.1 (20) a | 22 | 7.21 ± 0.45 c | 1.56 ± 0.12 c | 64.1 ± 4.9 a |
| Buckthorn | - | - | 53 | 0.69 ± 0.02 g | 5.10 ± 0.39 ab | 68.8 ± 5.4 a |
| Bitter cherry | - | - | 51 | 0.32 ± 0.01 g | 6.46 ± 0.46 a | 33.2 ± 5.0 c |
| Cultivar 1 | Fruit Color | Fruit Size (g) (n = 10) 2 | Brix (n = 10) 2 | Fruit Firmness (g mm−1) (n = 15) 2 | Eggs per Gram Fruit (n= 25) 2 | Eggs Developed to Adults (%) 2 (n= 25) |
|---|---|---|---|---|---|---|
| Cultivar 1 | Purple | 8.7 ± 0.4 a | 21.5 ± 0.6 c | 76.9 ± 7.9 d | 1.23 ± 0.18 a | 63.4 ± 6.3 abc |
| Cultivar 2 | Purple | 7.4 ± 0.4 b | 24.0 ± 0.8 bc | 99.6 ± 3.0 c | 1.19 ± 0.17 a | 80.3 ± 4.9 ab |
| Cultivar 3 | Pink | 6.5 ± 0.1 bc | 24.6 ± 0.3 ab | 99.7 ± 3.9 c | 1.36 ± 0.19 a | 69.1 ± 6.4 abc |
| Cultivar 4 | Pink | 4.4 ± 0.2 e | 27.3 ± 0.5 a | 128.7 ± 4.2 b | 2.71 ± 0.55 a | 53.1 ± 6.0 abc |
| Cultivar 5 | Red | 4.3 ± 0.1 e | 22.6 ± 0.7 bc | 156.8 ± 3.9 a | 2.18 ± 0.31 a | 68.4 ± 4.6 abc |
| Cultivar 6 | Yellow | 2.5 ± 0.1f | 16.0 ± 0.9 d | 114.4 ± 3.5 bc | 4.44 ± 0.69 b | 52.9 ± 5.7 c |
| Cultivar 7 | Yellow | 5.6 ± 0.3 cd | 18.3 ± 0.9 d | 97.6 ± 3.5 c | 1.88 ± 0.26 a | 77.8 ± 4.8 abc |
| Cultivar 8 | Yellow | 4.8 ± 0.2 de | 22.5 ± 0.4 bc | 132.8 ± 2.4 b | 1.98 ± 0.29 a | 61.0 ± 6.7 abc |
| Cultivar 9 | Black | 3.6 ± 0.3 ef | 15.6 ± 0.8 d | 100.8 ± 4.0 c | 2.03 ± 0.30 a | 80.2 ± 4.6 a |
| Cultivar 10 | Black | 4.4 ± 0.1 e | 24.2 ± 0.6 bc | 52.0 ± 4.1 e | 2.35 ± 0.27 a | 57.1 ± 5.6 abc |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Kaçar, G.; Daane, K.M. Temporal Dynamics of Host Use by Drosophila suzukii in California’s San Joaquin Valley: Implications for Area-Wide Pest Management. Insects 2019, 10, 206. https://doi.org/10.3390/insects10070206
Wang X, Kaçar G, Daane KM. Temporal Dynamics of Host Use by Drosophila suzukii in California’s San Joaquin Valley: Implications for Area-Wide Pest Management. Insects. 2019; 10(7):206. https://doi.org/10.3390/insects10070206
Chicago/Turabian StyleWang, Xingeng, Gülay Kaçar, and Kent M. Daane. 2019. "Temporal Dynamics of Host Use by Drosophila suzukii in California’s San Joaquin Valley: Implications for Area-Wide Pest Management" Insects 10, no. 7: 206. https://doi.org/10.3390/insects10070206
APA StyleWang, X., Kaçar, G., & Daane, K. M. (2019). Temporal Dynamics of Host Use by Drosophila suzukii in California’s San Joaquin Valley: Implications for Area-Wide Pest Management. Insects, 10(7), 206. https://doi.org/10.3390/insects10070206





