Effects of Diet Quality and Temperature on Stable Fly (Diptera: Muscidae) Development
Abstract
1. Introduction
2. Materials and Methods
3. Results
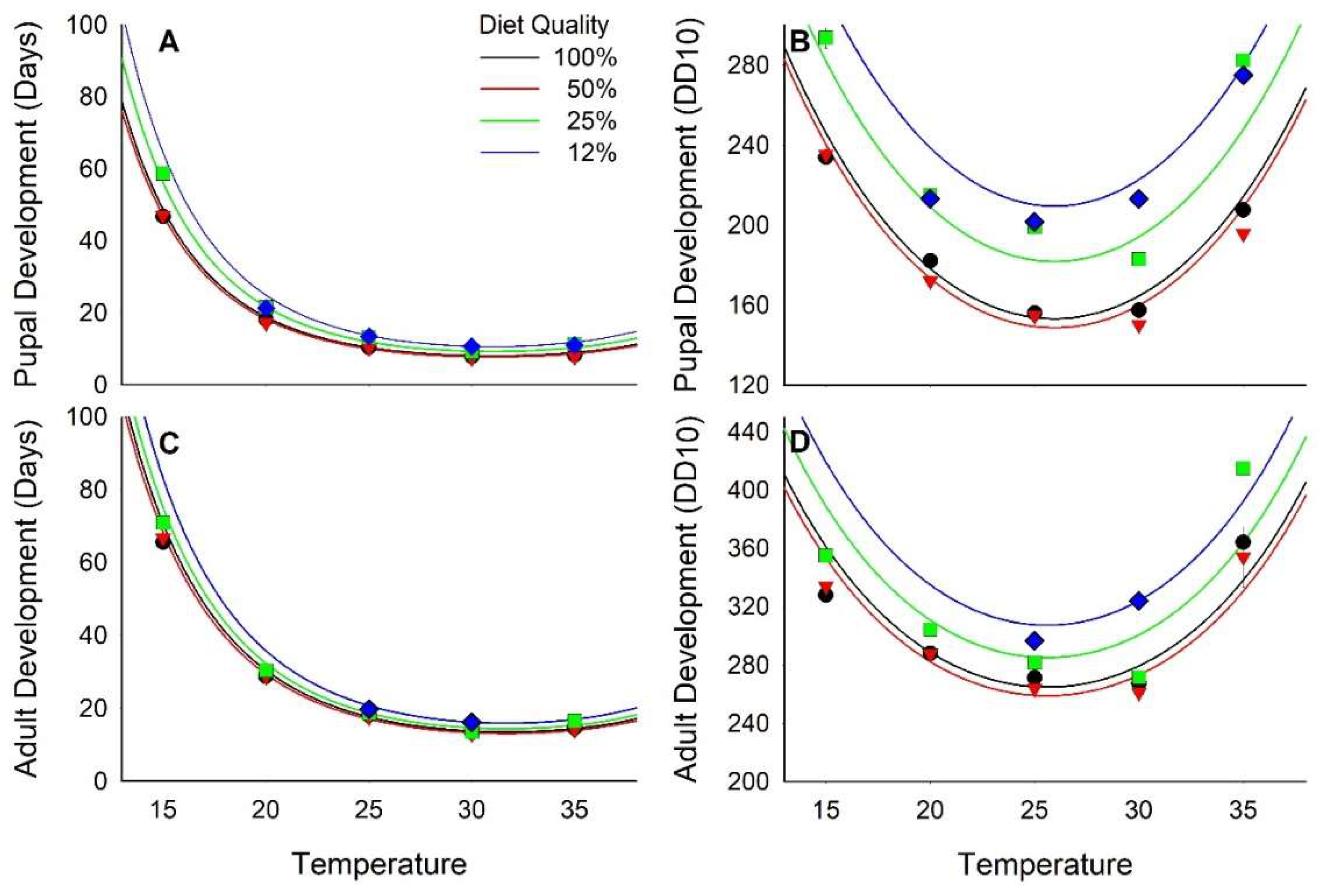
3.1. Development Time
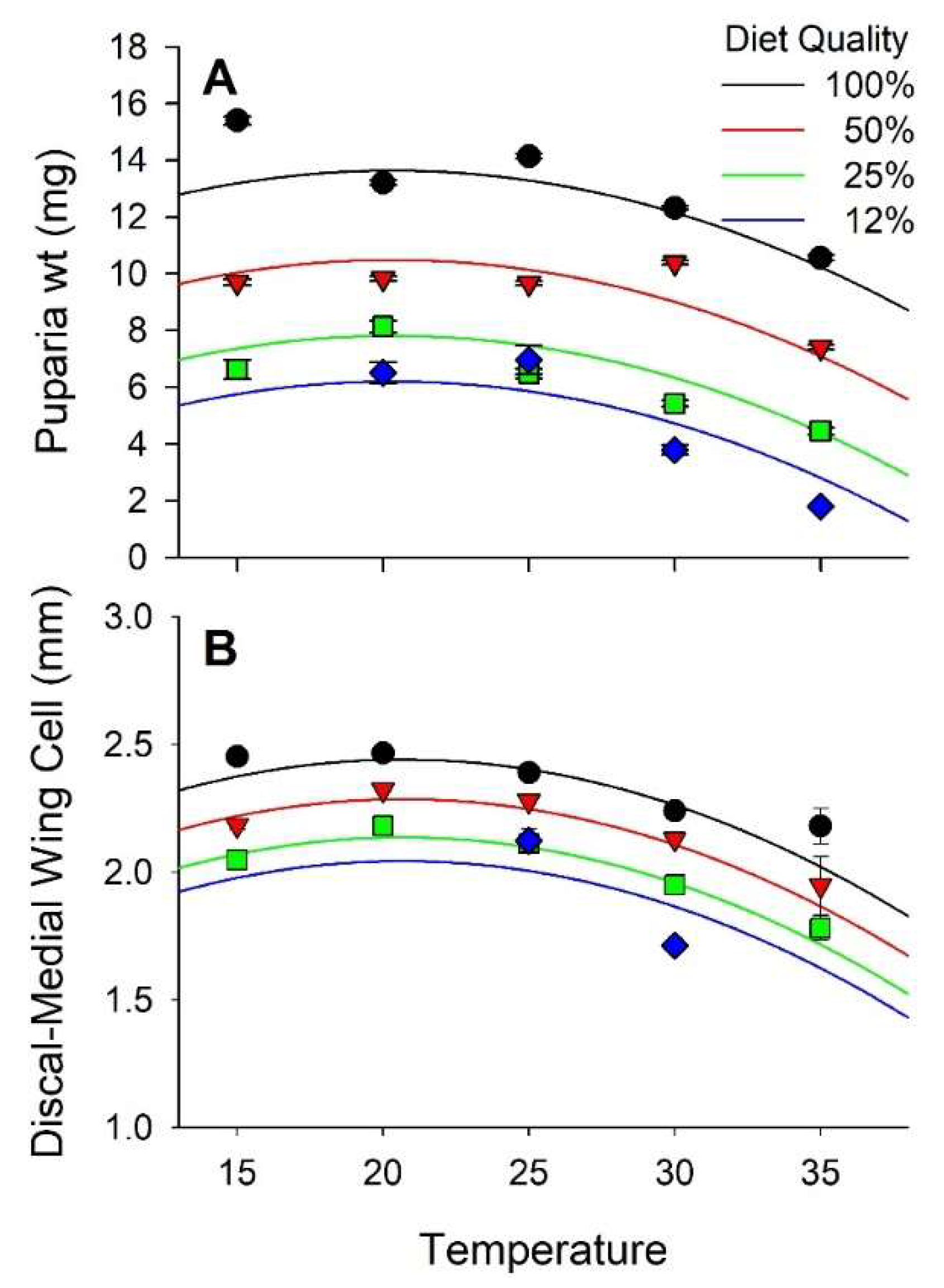
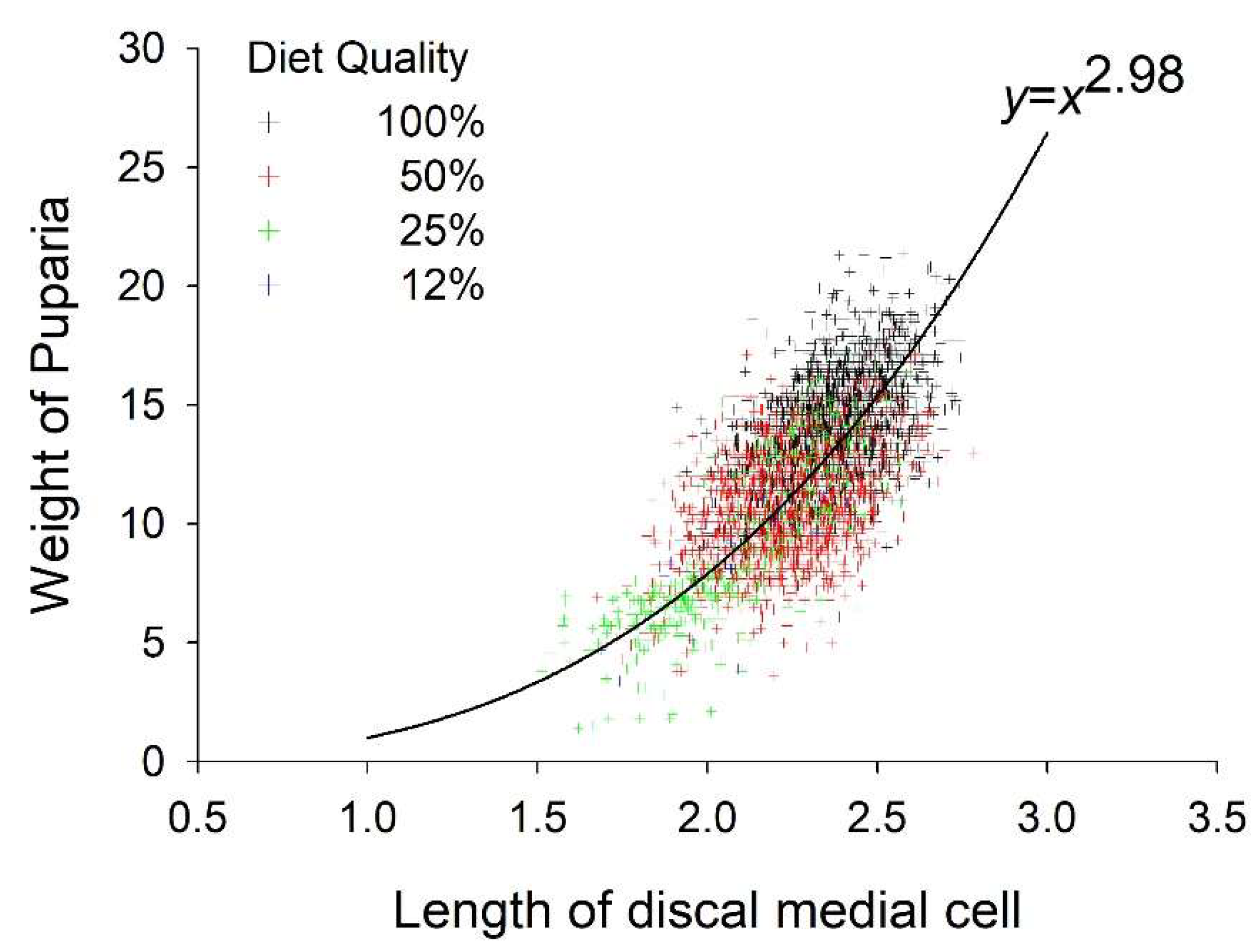
3.2. Weight and Size
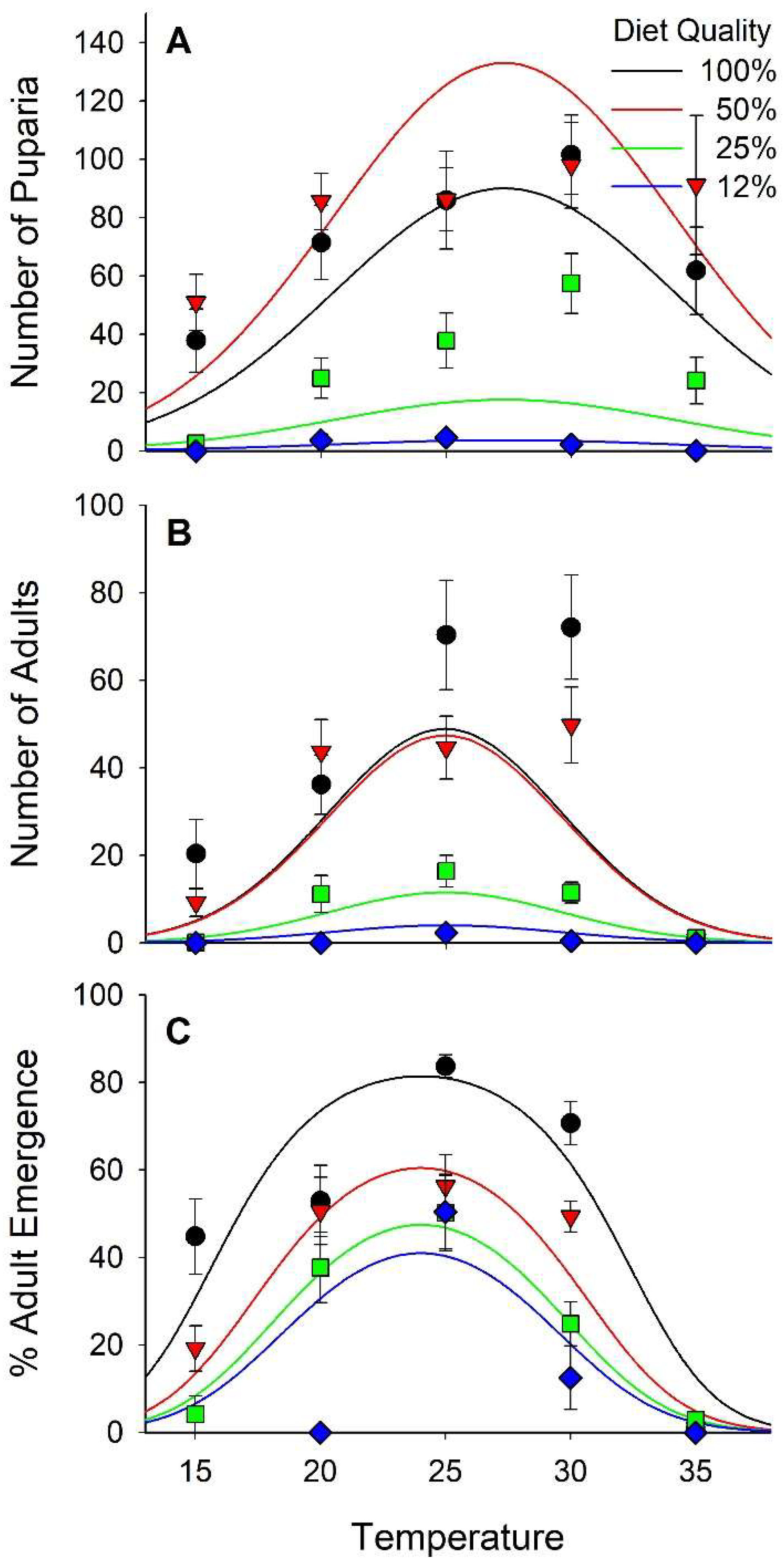
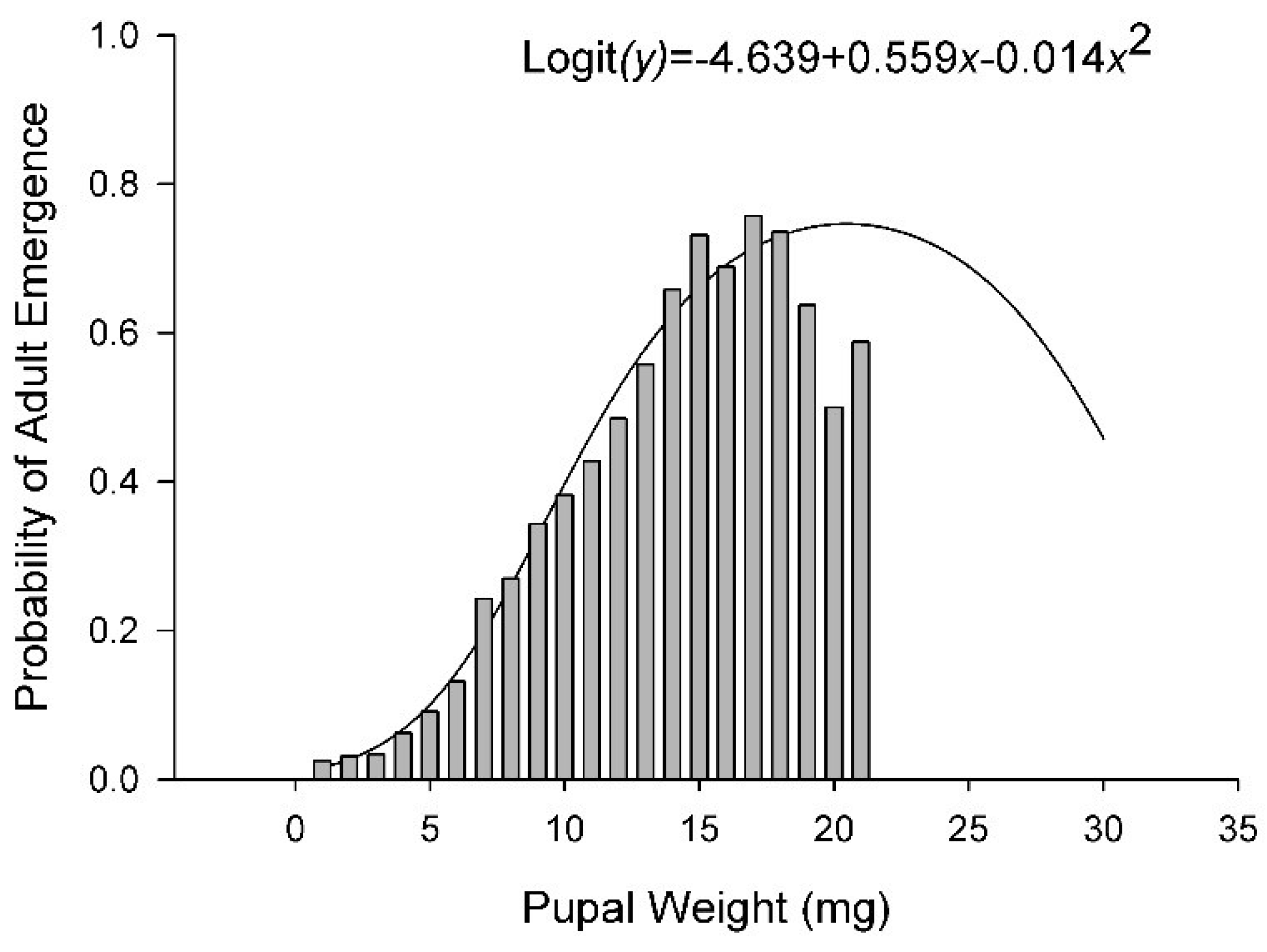
3.3. Survival
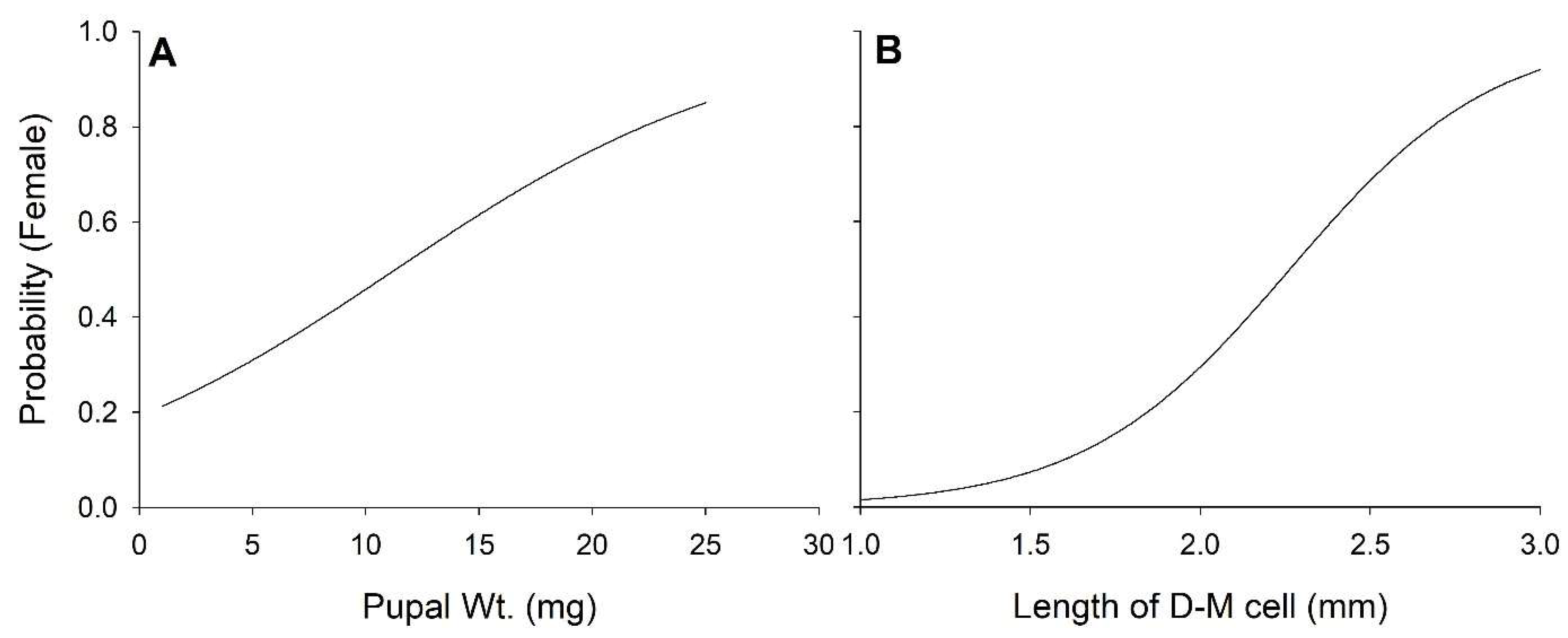
3.4. Sex
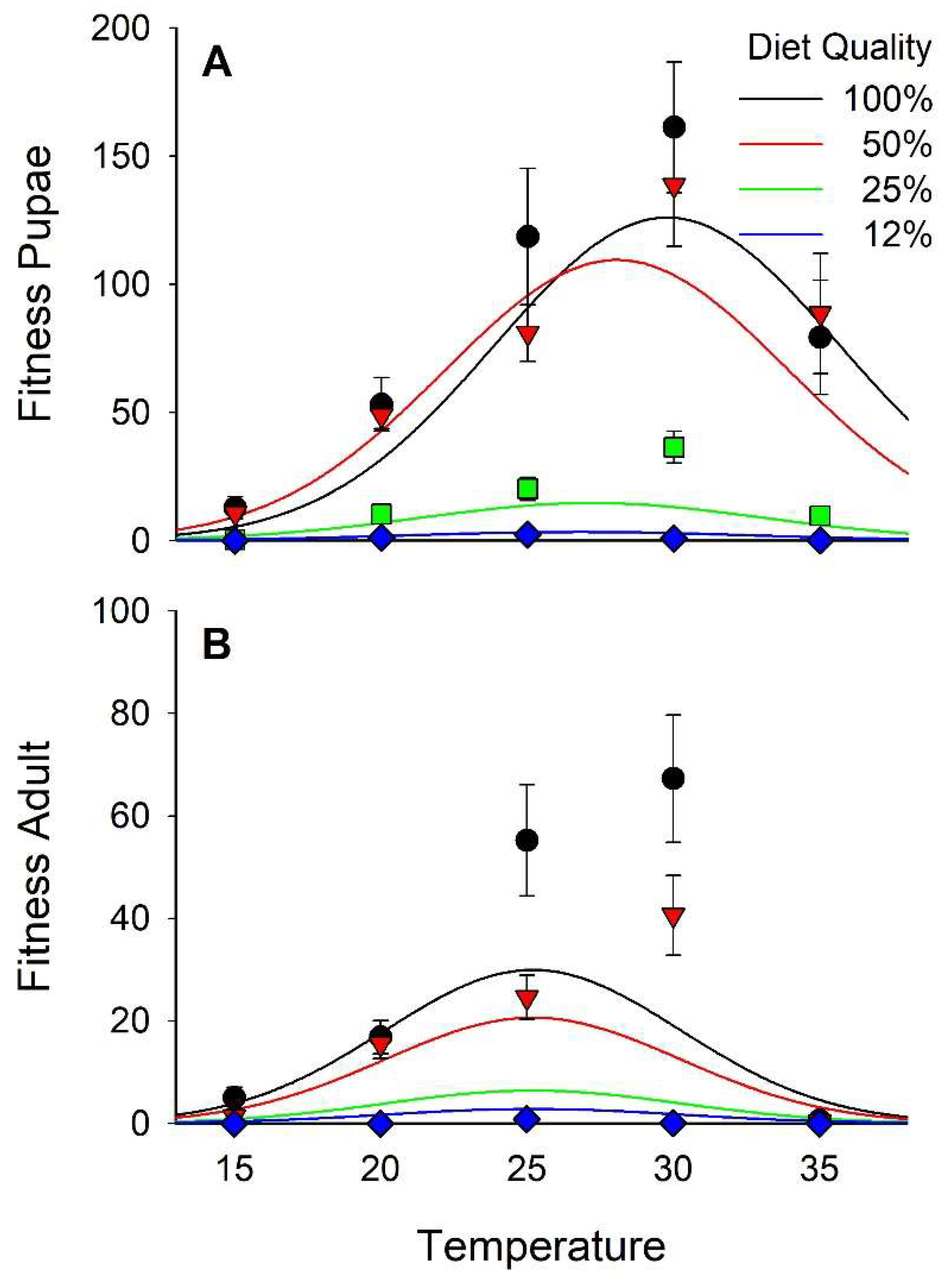
3.5. Fitness
4. Discussion
4.1. Development Time and Size
4.2. Survival
4.3. Sex
4.4. Fitness
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Blanckenhorn, W.U. Different growth responses to temperature and resource limitation in three species with similar life histories. Evol. Ecol. 1999, 13, 395–409. [Google Scholar] [CrossRef]
- Evans, E.W. Consequences of body size for fecundity in the predatory stinkbug, Podisus maculiventris (Hemiptera: Pentatomidae). Ann. Entomol. Soc. Am. 1982, 75, 418–420. [Google Scholar] [CrossRef]
- Gilbert, N. Control of fecundity in Pieris rapae: I. The problem. J. Anim. Ecol. 1984, 53, 581–588. [Google Scholar] [CrossRef]
- Honěk, A. Intraspecific variation in body size and fecundity in insects: A general relationship. Oikos 1993, 66, 483–492. [Google Scholar] [CrossRef]
- Abrams, P.A.; Leimar, O.; Nylin, S.; Wiklund, C. The effect of flexible growth rates on optimal sizes and development times in a seasonal environment. Am. Nat. 1996, 147, 381–395. [Google Scholar] [CrossRef]
- Atkinson, D. Temperature and organism size: A biological law for ectotherms? Adv. Ecol. Res. 1994, 25, 1–58. [Google Scholar]
- Wilbur, H.M.; Collins, J.P. Ecological aspects of amphibian metamorphosis. Science 1973, 182, 1305–1314. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.R. Mating behavior of Stomoxys calcitrans: Effects of a blood meal on the mating drive of males and its necessity as a prerequisite for proper insemination of females. J. Econ. Entomol. 1978, 71, 379–386. [Google Scholar] [CrossRef]
- Baldacchino, F.; Muenworn, V.; Desquesnes, M.; Desoli, F.; Charoenviriyaphap, T.; Duvallet, G. Transmission of pathogens by Stomoxys flies (Diptera, Muscidae): A review. Parasite 2013, 20, 26. [Google Scholar] [CrossRef]
- Larsen, E.B.; Thomsen, M. The influence of temperature on the development of some species of Diptera. Vidensk Medd fra Dansk Naturh Foren 1941, 104, 1–75. [Google Scholar]
- Berry, I.L.; Foerster, K.W.; Ilcken, E.H. Prediction model for development time of stable flies. Trans. Am. Soc. Agric. Eng. 1976, 19, 123–127. [Google Scholar] [CrossRef]
- Kunz, S.E.; Berry, I.L.; Foerster, K.W. The development of the immature forms of Stomoxys calcitrans. Ann. Entomol. Soc. Am. 1977, 70, 169–172. [Google Scholar] [CrossRef]
- Lysyk, T.J. Relationships between temperature and life-history parameters of Stomoxys calcitrans (Diptera: Muscidae). J. Med. Entomol. 1998, 35, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Gilles, J.; David, J.F.; Duvallet, G. Temperature effects on development and survival of two stable flies, Stomoxys calcitrans and Stomoxys niger niger (Diptera: Muscidae), in La Réunion Island. J. Med. Entomol. 2005, 42, 260–265. [Google Scholar] [CrossRef]
- Beresford, D.V.; Sutcliffe, J.F. Field measurements of stable fly (Diptera: Muscidae) demography, fecundity, and survival based on daily trap catches at a beef farm in southern Ontario Over a 5-yr Period. J. Med. Entomol. 2012, 49, 1262–1269. [Google Scholar] [CrossRef]
- Solórzano, J.-A.; Gilles, J.; Bravo, O.; Vargas, C.; Gomez-Bonilla, Y.; Bingham, G.V.; Taylor, D.B. Biology and trapping of stable flies (Diptera: Muscidae) developing in pineapple residues (Ananas comosus) in Costa Rica. J. Insect Sci. 2015, 15, 145. [Google Scholar] [CrossRef] [PubMed]
- Skovgärd, H.; Nachman, G. Modeling the temperature- and age-dependent survival, development, and oviposition rates of stable flies (Stomoxys calcitrans) (Diptera: Muscidae). Environ. Entomol. 2017, 46, 1130–1142. [Google Scholar] [CrossRef]
- Cook, D.F.; Telfer, D.V.; Lindsey, J.B.; Deyl, R.A. Substrates across horticultural and livestock industries that support the development of stable fly, Stomoxys calcitrans (Diptera: Muscidae). Austral Entomol. 2017, 57, 344–348. [Google Scholar] [CrossRef]
- Broce, A.B.; Haas, M.S. Relation of cattle manure age to colonization by stable fly and house fly (Diptera: Muscidae). J. Kans. Entomol. Soc. 1999, 72, 60–72. [Google Scholar]
- Taylor, D.B.; Berkebile, D.R. Phenology of stable fly (Diptera: Muscidae) larvae in round bale hay feeding sites in eastern Nebraska. Environ. Entomol. 2011, 40, 184–193. [Google Scholar] [CrossRef]
- Albuquerque, T.A.; Zurek, L. Temporal changes in the bacterial community of animal feces and their correlation with stable fly oviposition, larval development, and adult fitness. Front. Microbiol. 2014, 5, 590. [Google Scholar] [CrossRef] [PubMed]
- Friesen, K.; Berkebile, D.R.; Zhu, J.J.; Taylor, D.B. Laboratory rearing of stable flies and other muscoid Diptera. J. Vis. Exp. 2018, 138, e57341. [Google Scholar] [CrossRef] [PubMed]
- Arnold, C.Y. The determination and significance of the base temperature in a linear heat unit system. J. Am. Soc. Hortic. Sci. 1959, 74, 430–445. [Google Scholar]
- Lysyk, T.J. Seasonal abundance of stable flies and house flies (Diptera: Muscidae) in dairies in Alberta, Canada. J. Med. Entomol. 1993, 30, 888–895. [Google Scholar] [CrossRef] [PubMed]
- Pruess, K.P. Day-degree methods for pest management. Environ. Entomol. 1983, 12, 613–619. [Google Scholar] [CrossRef]
- Hogsette, J.A.; Ruff, J.P. Stable fly (Diptera: Muscidae) migration in northwest Florida. Environ. Entomol. 1985, 14, 170–175. [Google Scholar] [CrossRef]
- Taylor, D.B.; Moon, R.D.; Campbell, J.B.; Berkebile, D.R.; Scholl, P.J.; Broce, A.B.; Hogsette, J.A. Dispersal of stable flies (Diptera: Muscidae) from larval developmental sites. Environ. Entomol. 2010, 39, 1101–1110. [Google Scholar] [CrossRef]
- Parsons, P.A. Fluctuating asymmetry: An epigenetic measure of stress. Biol. Rev. Camb. Philos. Soc. 1990, 65, 131–145. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.; Mateus, R.P.; Moura, M.O. Constant fluctuating asymmetry but not directional asymmetry along the geographic distribution of Drosophila antonietae (Diptera, Drosophilidae). Rev. Bras. Entomol. 2015, 59, 337–342. [Google Scholar] [CrossRef]
| Ingredient (%) | |||
|---|---|---|---|
| % Nutrients | Wheat Bran | Fish Meal | Vermiculite |
| 100 | 59 | 13 | 28 |
| 50 | 30 | 8 | 63 |
| 25 | 15 | 3 | 82 |
| 12 | 7 | 2 | 91 |
| Transformation | df | Diet | Diet 2 | Temp | Temp 2 | Diet × Temp | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| t | p | t | p | t | p | t | p | t | p | |||
| Pupal Dev. (d) | Logn | 161 | −7.18 | <0.01 | 6.35 | <0.01 | −30.38 | <0.01 | 23.55 | <0.01 | −1.13 | 0.26 |
| Pupal Dev. (DD10) | Square Root | 161 | −7.72 | <0.01 | 6.68 | <0.01 | −14.56 | <0.01 | 14.24 | <0.01 | −0.86 | 0.39 |
| Adult Dev. (d) | Logn | 135 | −5.07 | <0.01 | 4.79 | <0.01 | −29.95 | <0.01 | 22.07 | <0.01 | −0.68 | 0.50 |
| Adult Dev. (DD10) | Logn | 135 | −5.11 | <0.01 | 4.59 | <0.01 | −12.14 | <0.01 | 11.69 | <0.01 | −0.10 | 0.92 |
| Pupal wt. | 161 | 4.86 | <0.01 | −2.39 | 0.02 | 2.86 | <0.01 | −3.57 | <0.01 | −1.31 | 0.19 | |
| Adult D−M cell | 130 | 4.12 | <0.01 | −2.31 | <0.01 | 4.95 | <0.01 | −5.78 | <0.01 | −1.07 | 0.29 | |
| No. Pupae | Logn (N+1) | 71 | 11.18 | <0.01 | −9.24 | <0.01 | 4.81 | <0.01 | −4.38 | <0.01 | 0.28 | 0.78 |
| No. Adults | Logn (N+1) | 71 | 6.77 | <0.01 | −5.20 | <0.01 | 7.96 | <0.01 | −8.02 | <0.01 | −1.26 | 0.21 |
| % Adult Emer. | Logit | 58 | 7.74 | <0.01 | −1.87 | 0.07 | 10.03 | <0.01 | −10.25 | <0.01 | −1.24 | 0.22 |
| Fitness Puparia | Logn (N+1) | 70 | 8.01 | <0.01 | −8.92 | <0.01 | 6.21 | <0.01 | −5.94 | <0.01 | 2.17 | 0.03 |
| Fitness Adult | Logn (N+1) | 71 | 5.59 | <0.01 | −4.02 | <0.01 | 8.12 | <0.01 | −8.01 | <0.01 | 0.17 | 0.86 |
| Parameter | Intercept | Diet | Diet 2 | Temp | Temp 2 | Diet × Temp |
|---|---|---|---|---|---|---|
| b ± SEM | b ± SEM | b ± SEM | b ± SEM | b ± SEM | b ± SEM | |
| Pupal Dev. (d) | 9.33 ± 0.18 | −1.489 ± 0.207 | 1.033 ± 0.163 | −0.439 ± 0.014 | 0.007 ± 0.0003 | |
| Pupal Dev. (DD10) | 34.55 ± 1.30 | −10.65 ± 1.38 | 7.28 ± 1.09 | −1.46 ± 0.10 | 0.028 ± 0.002 | |
| Adult Dev. (d) | 8.84 ± 0.16 | −0.947 ± 0.187 | 0.677 ± 0.141 | −0.377 ± 0.013 | 0.0059 ± 0.0003 | |
| Adult Dev. (DD10) | 7.68 ± 0.16 | −0.822 ± 0.161 | 0.572 ± 0.125 | −0.146 ± 0.012 | 0.0028 ± 0.0002 | |
| Pupal wt. | −2.09 ± 2.92 | 15.09 ± 3.10 | −5.86 ± 2.45 | 0.639 ± 0.224 | −0.0157 ± 0.0044 | |
| Adult D−M cell | 1.07 ± 0.22 | 0.884 ± 0.215 | −0.383 ± 0.165 | 0.084 ± 0.017 | −0.002 ± 0.0004 | |
| No. Pupae | −8.75 ± 1.47 | 17.31 ± 1.55 | −12.12 ± 1.31 | 0.589 ± 0.122 | −0.011 ± 0.0025 | |
| No. Adults | −13.08 ± 1.59 | 11.35 ± 1.68 | −7.39 ± 1.42 | 1.06 ± 0.13 | −0.021 ± 0.0027 | |
| % Adult Emer. | −16.86 ± 1.64 | 2.10 ± 0.27 | 1.35 ± 0.13 | −0.028 ± 0.0027 | ||
| Fitness Pupal | −10.08 ± 1.42 | 13.84 ± 1.73 | −10.73 ± 1.20 | 0.706 ± 0.114 | −0.013 ± 0.0023 | 0.087 ± 0.040 |
| Fitness Adult | −12.42 ± 1.46 | 8.61 ± 1.54 | −5.24 ± 1.31 | 0.988 ± 0.122 | −0.020 ± 0.0025 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Florez-Cuadros, M.; Berkebile, D.; Brewer, G.; Taylor, D.B. Effects of Diet Quality and Temperature on Stable Fly (Diptera: Muscidae) Development. Insects 2019, 10, 207. https://doi.org/10.3390/insects10070207
Florez-Cuadros M, Berkebile D, Brewer G, Taylor DB. Effects of Diet Quality and Temperature on Stable Fly (Diptera: Muscidae) Development. Insects. 2019; 10(7):207. https://doi.org/10.3390/insects10070207
Chicago/Turabian StyleFlorez-Cuadros, Melina, Dennis Berkebile, Gary Brewer, and David B. Taylor. 2019. "Effects of Diet Quality and Temperature on Stable Fly (Diptera: Muscidae) Development" Insects 10, no. 7: 207. https://doi.org/10.3390/insects10070207
APA StyleFlorez-Cuadros, M., Berkebile, D., Brewer, G., & Taylor, D. B. (2019). Effects of Diet Quality and Temperature on Stable Fly (Diptera: Muscidae) Development. Insects, 10(7), 207. https://doi.org/10.3390/insects10070207





