Hemipteran Pests of Sugarcane in North America
Abstract
:1. Introduction
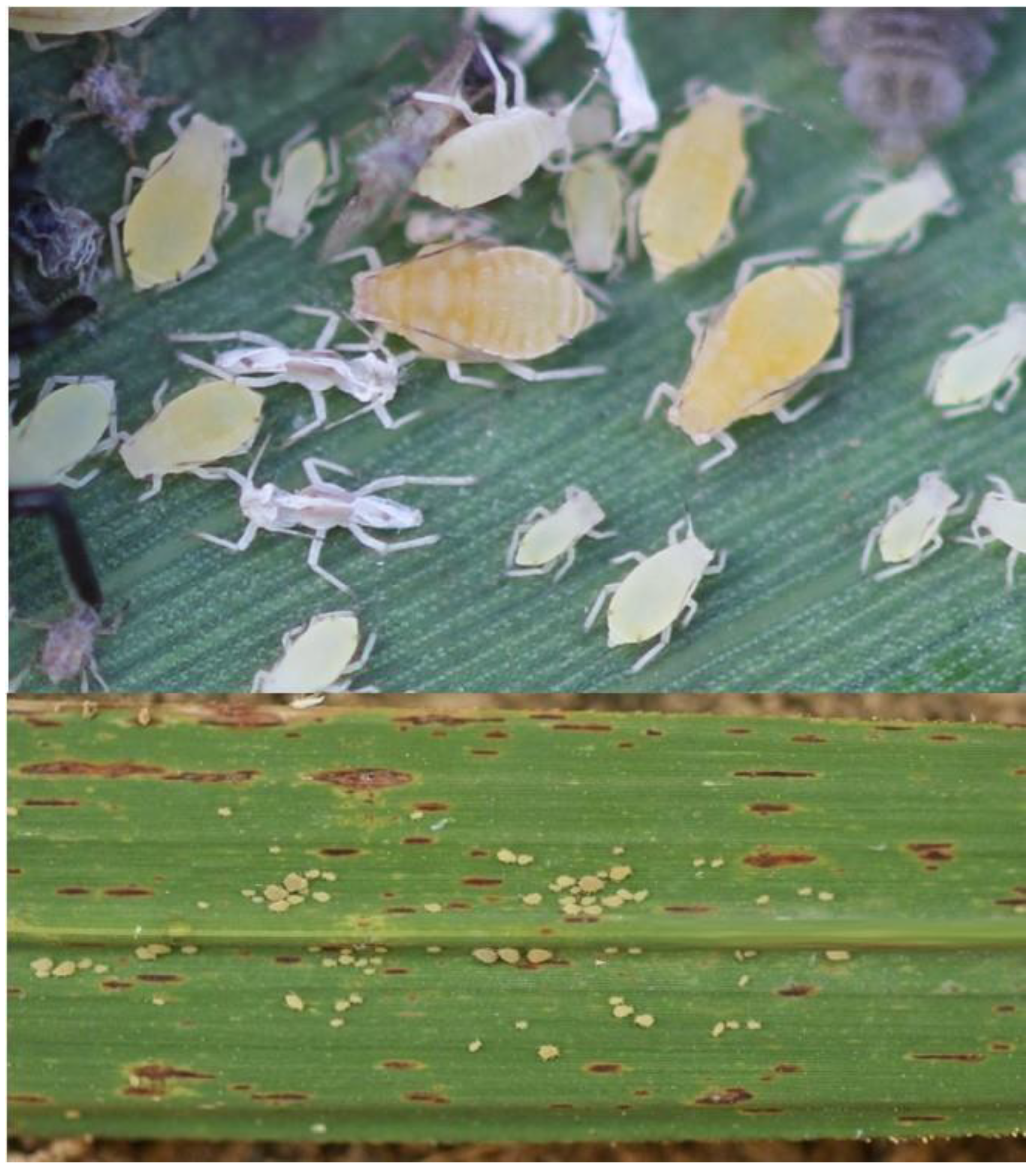
2. The Sugarcane Aphid, Melanaphis sacchari
2.1. Morphology and Biology
2.2. Distribution and Host Plants
2.3. Ecology, Pest Status, and Management in Sugarcane
3. Yellow Sugarcane Aphid, Sipha flava
3.1. Morphology and Biology
3.2. Distribution and Host Plants
3.3. Ecology, Pest Status, and Management in Sugarcane
4. The West Indian Canefly, Saccharosydne saccharivora
4.1. Morphology and Biology
4.2. Distribution and Host Plants
4.3. Ecology, Pest Status, and Management in North American Sugarcane
5. The Sugarcane Leafhopper Perkinsiella saccharicida
5.1. Morphology and Biology
5.2. Distribution and Host Plants
5.3. Ecology, Pest Status, and Management in North American Sugarcane
6. Sugarcane Lace Bug, Leptodictya tabida
6.1. Morphology and Biology
6.2. Distribution and Host Plants
6.3. Ecology, Pest Status, and Management in North American Sugarcane
7. Conclusions
Funding
Conflicts of Interest
References
- USDA. Economic Research Service. Sugarcane Yearbook; 2017. Available online: https://www.ers.usda.gov/data-products/sugar-and-sweeteners-yearbook-tables.aspx (accessed on 16 June 2018).
- Aguilar-Rivera, N.; Rodriguez, D.A.; Enriquez, V.; Castillo, R.A.; Herrera, A. The Mexican sugarcane industry: Overview, constraints, current status, and long-term trends. Sugar Tech. 2012, 14, 207–222. [Google Scholar] [CrossRef]
- Wilson, B.E.; VanWeelden, M.T.; Beuzelin, J.M.; Reagan, T.E.; Prado, J.A. Efficacy of insect growth regulators and diamide insecticides for control of stem borers (Lepidoptera: Crambidae) in sugarcane. J. Econ. Entomol. 2017, 110, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.E.; Showler, A.T.; Beuzelin, J.M.; Reagan, T.E. Improved chemical control for the Mexican rice borer (Lepidoptera: Crambidae) in sugarcane: Larval exposure, a novel scouting method, and efficacy of a single aerial insecticide application. J. Econ. Entomol. 2012, 105, 1998–2006. [Google Scholar] [CrossRef]
- Van Zwaluwenburg, R.H. Insect enemies of sugarcane in western Mexico. J. Econ. Entomol. 1926, 19, 664–669. [Google Scholar] [CrossRef]
- Singh, B.U.; Padmaja, P.G.; Seetharama, N. Biology and management of the sugarcane aphid, Melanaphis sacchari (Zehntner) (Homoptera: Aphididae), in sorghum: A review. Crop Prot. 2004, 23, 739–755. [Google Scholar] [CrossRef]
- Bowling, R.D.; Brewer, M.J.; Kerns, D.; Gordy, J.; Seiter, N.; Elliot, N.E.; Buntin, G.D.; Way, M.O.; Royer, T.A.; Biles, S.; et al. Sugarcane aphid (Hemiptera: Aphididae): A new pest of sorghum in North America. J. Integr. Pest Manag. 2016, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Blackman, R.L.; Eastop, V.F. Aphids on the World’s Crops—An Identification Guide; John Wiley and Sons: New York, NY, USA, 1984. [Google Scholar]
- David, S.K.; Sandhu, G.S. New oviparous morph of Melanaphis sacchari (Zehntner) on sorghum. Entomol. Rec. 1976, 88, 28–29. [Google Scholar]
- Yadava, R.L. Oviparity in sugarcane aphid Longiunguis sacchari Zehnt (Aphididae: Homoptera). Curr. Sci. 1966, 1, 18. [Google Scholar]
- Akbar, W.; Showler, A.T.; Reagan, T.E.; White, W.H. Categorizing sugarcane cultivar resistance to the sugarcane aphid and yellow sugarcane aphid (Hemiptera: Aphididae). J. Econ. Entomol. 2010, 103, 1431–1437. [Google Scholar] [CrossRef]
- Lama, L.; Wilson, B.E.; Davis, J.A.; Reagan, T.E. Influence of sorghum cultivar, phenological stage, and fertilization on development and reproduction of Melanaphis sacchari (Hemiptera: Aphididae). Fla. Entomol. 2019, 102. in press. [Google Scholar]
- Wilbrink, G. An investigation on spread of the mosaic disease of sugarcane by aphids. Medid Procfst 1922, 10, 413–456. [Google Scholar]
- White, W.H.; Regan, T.E.; Hall, D.G. Melanaphis sacchari (Homoptera: Aphididae), A sugarcane pest new to Louisiana. Fla. Entomol. 2001, 84, 435–436. [Google Scholar] [CrossRef]
- Nibouche, S.; Costlet, L.; Holt, J.R.; Jacobson, A.; Pekarcik, A.; Sadeyen, J.; Armstrong, J.S.; Peterson, G.C.; McLaren, N.; Medina, R.F. Invasion of sorghum in the Americas by a new sugarcane aphid (Melanaphis sacchari) superclone. PLoS ONE 2018, 13, e0196124. [Google Scholar] [CrossRef] [PubMed]
- Schenck, D.J.; Lehrer, A.T. Factors affecting the transmission and spread of sugarcane yellow leaf virus. Plant Dis. 2000, 84, 1085–1088. [Google Scholar] [CrossRef] [PubMed]
- Grisham, M.P.; Pan, Y.B.; Legendre, B.L.; Godshall, M.A.; Eggleston, G. Effect of sugarcane yellow leaf virus on sugarcane yield and juice quality. Proc. Int. Soc. Sugar Cane Technol. 2001, 24, 434–438. [Google Scholar]
- McAllister, C.D.; Hoy, J.W.; Reagan, T.E. Temporal increase and spatial distribution of sugarcane yellow leaf and infestations of Melanaphis sacchari. Plant Dis. 2008, 92, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Comstock, J.C.; Miller, J.D.; Tai, P.Y.P.; Follis, J.E. Incidence of and resistance to sugarcane yellow leaf virus in Florida. Proc. Int. Soc. Sugar Cane Technol. 1999, 23, 366–372. [Google Scholar]
- Flynn, J.; Powell, G.; Perdomo, R.; Montes, G.; Quebedeaux, K.; Comstock, J. Comparison of sugarcane disease incidence and yield of field-run, heat-treated, and tissue-culture based seedcane. J. Am. Soc. Sugar Cane Technol. 2005, 25, 88–100. [Google Scholar]
- Long, W.H.; Hensley, S.D. Insect pests of sugarcane. Ann. Rev. Entomol. 1972, 17, 149–176. [Google Scholar] [CrossRef]
- McAllister, C.D.; Reay-Jones, F.; Posey, F.R.; Flanagan, J.W.; Reagan, T.E. Small plot assessment of insecticides against the sugarcane aphid, 2002. Arthrop. Manag. Tests 2003, 28, F5. [Google Scholar] [CrossRef]
- Akbar, W.; Beuzelin, J.M.; Reagan, T.E.; Coburn, G.E. Small plot assessment of insecticides against the sugarcane aphid, 2006. Arthrop. Manag. Tests 2007, 32, F58. [Google Scholar] [CrossRef]
- Wilson, B.E.; VanWeelden, M.T.; Beuzelin, J.M.; Mészáros, A. Assessment of insecticides against a complex of hemipteran pests in sugarcane. Arthrop. Manag. Tests 2013, 38, F73. [Google Scholar] [CrossRef]
- Wilson, B.E.; Mulcahy, M.; Lama, L.; Richard, R.R. Assessment of insecticides against the West Indian canefly and sugarcane aphid in Louisiana sugarcane, 2017. Arthrop. Manag. Tests 2018, 43, tys011. [Google Scholar] [CrossRef]
- Beuzelin, J.M.; May, D.M. Evaluation of insecticides for control of the sugarcane aphid in grain sorghum, 2014. Arthrop. Manag. Tests 2015, 40, F5. [Google Scholar] [CrossRef]
- Way, M.O.; Pearson, R.A.; Vyavhare, S.; Verret, C.; Voegtlin, D. Foliar treatments for sugarcane aphid control in sorghum, 2013. Arthrop. Manag. Tests 2014, 39, F45. [Google Scholar] [CrossRef]
- LSU AgCenter. Louisiana recommendations for control of sugarcane insects. In 2019 Louisiana Insect Pest Management Guide; publication #1838; LSU AgCenter: Baton Rouge, LA, USA, 2019; pp. 48–50. [Google Scholar]
- Akbar, W.; Showler, A.T.; Beuzelin, J.M.; Reagan, T.E.; Gravois, K.A. Evaluation of aphid resistance among sugarcane cultivars in Louisiana. Ann. Entomol. Soc. Am. 2011, 104, 699–704. [Google Scholar] [CrossRef]
- Akbar, W.; Showler, A.T.; Reagan, T.E.; Davis, J.A.; Beuzelin, J.M. Feeding by sugarcane aphid, Melanaphis sacchari, on sugarcane cultivars with differential susceptibility and potential mechanism of resistance. Entomologia Experimentalis et Applicata 2014, 150, 32–44. [Google Scholar] [CrossRef]
- Colares, F.; Michaud, J.P.; Bain, C.L.; Torres, J.B. Recruitment of aphidophagous arthropods to sorghum plants infested with Melanaphis sacchari and Schizaphis graminum (Hemiptera: Aphididae). Biol. Control 2015, 90, 16–24. [Google Scholar] [CrossRef]
- Akbar, W.; Carlton, C.; Reagan, T.E. Life cycle and larval morphology of Diomus terminatus (Coleoptera: Coccinellidae), and its potential as a biological control agent of Melanaphis sacchari (Homoptera: Aphididae). Ann. Entomol. Soc. Am. 2009, 102, 96–103. [Google Scholar] [CrossRef]
- Hentz, M.G.; Nuessly, G.S. Morphology and biology of Diomus terminatus (Coleoptera: Coccinellidae), a predator of Sipha flava (Homoptera: Aphididae). Fla. Entomol. 2002, 85, 276–279. [Google Scholar] [CrossRef]
- Haar, P.J.; Bowling, R.; Gardner, W.A.; Bunting, G.D. Epizootics of the entomopathogenic fungus Lecanicillium lecani (Hypocreales: Clavicipitaceae) in sugarcane aphid (Hemiptera: Aphididae) populations infesting grain sorghum in Georgia and Texas. J. Entomol. Sci. 2018, 53, 104–106. [Google Scholar] [CrossRef]
- Nuessly, G.S. Featured Creatures: Yellow Sugarcane Aphid; publication #EENY-354; University of Florida: Gainsville, FL, USA; Available online: http://entnemdept.ufl.edu/creatures/field/bugs/yellow_sugarcane_aphid.htm (accessed on 16 January 2019).
- Hentz, M.G.; Nuessly, G.S. Development, longevity, and fecundity of Sipha flava (Homoptera: Aphididae) feeding on Sorghum bicolor. Environ. Entomol. 2004, 33, 546–553. [Google Scholar] [CrossRef]
- Way, M.J.; Conlong, D.E.; Martin, L.A.; Macfarlane, S.A.; Stranack, R.; Keeping, M.G.; Rutherford, R.S. First record of yellow sugarcane aphid, Sipha flava (Homoptera: Aphididae), in the South African sugarcane industry. Int. Sug. J. 2015, 9, 32–34. [Google Scholar]
- Wilson, B.E. Yellow Sugarcane Aphid, Sipha flava: A Pest of Sugarcane in Louisiana; publication #3630; LSU AgCenter: Baton Rouge, LA, USA, 2018. [Google Scholar]
- Nuessly, G.S.; Hentz, M.G.; Gilbert, R.A. Susceptibility of stage IV Canal Point (CP) sugarcane clones to yellow sugarcane aphid (Sipha flava (Forbes)) feeding damage. J. Am. Soc. Sugar Cane Technol. 2010, 30, 104–114. [Google Scholar]
- Hall, D.G. Notes on the yellow sugarcane aphid Sipha flava (Homoptera: Aphididae) and the lady beetle Diomus terminatus (Coleoptera: Coccinellidae) in Florida. J. Am. Soc. Sugar Cane Technol. 2001, 21, 21–29. [Google Scholar]
- Miskimen, G.W. Population dynamics of the yellow sugarcane aphid, Sipha flava, in Puerto Rico, as affected by heavy rains. Ann. Entomol. Soc. Am. 1970, 63, 642–645. [Google Scholar] [CrossRef]
- Medina-Gaud, S.L.; Martorell, L.F.; Bonilla-obles, R. Notes on the biology and control of the yellow aphid of sugarcane, Sipha flava Forbes in Puerto Rico. In Proceedings of the 12th Congress of the International Society of Sugarcane Technologists, San Juan, Puerto Rico, 28 March–10 April 1965; pp. 1307–1320. [Google Scholar]
- Wilson, B.E.; Gravois, K.A. West Indian Canefly—Biology and Control Measures. LSU AgCenter, 2017. Available online: https://www.lsuagcenter.com/profiles/kgravois/articles/page1491506139044 (accessed on 28 February 2019).
- Metcalfe, J.R. Studies on the biology of the sugar-cane pest Saccharosydne saccharivora (Westw.) (Hom. Delphacidae). Bull. Entomol. Res. 1968, 59, 393–408. [Google Scholar] [CrossRef]
- Metcalfe, J.R. Studies on the effect of the nutrient status of sugar-cane on the fecundity of Saccharosydne saccharivora (Westw.) (Hom. Delphacidae). Bull. Entomol. Res. 1970, 60, 73–85. [Google Scholar] [CrossRef]
- Metcalfe, J.R. An analysis of population dynamics of the Jamaican sugar-cane pest Saccharosydne saccharivora (Westw.) (Hom, Delphacidae). Bull. Entomol. Res. 1972, 62, 309–322. [Google Scholar] [CrossRef]
- Wilson, B.E.; Beuzelin, J.M.; White, W.H. The West Indian canefly, Saccharosydne saccharivora (Hemiptera: Delphacidae): An emerging threat to Louisiana sugarcane? Entomology Workshop, 2018; p. 28. Available online: http://www.issct.org/pdf/abstractsprogentowrkusadec18.pdf (accessed on 28 February 2019).
- Hall, D.G. Insects and mites associated with sugarcane in Florida. Fla. Entomol. 1988, 71, 138–150. [Google Scholar] [CrossRef]
- Meagher, R.L., Jr.; Legaspi, J.C. Within-field distribution of three homopteran species in Texas sugarcane. Southwest. Entomol. 2003, 28, 1–9. [Google Scholar]
- Ingram, J.W.; Bynum, E.K. The West Indian sugarcane fulgorid in Louisiana and Mississippi. Sugar Bull. 1945, 23, 101–102. [Google Scholar]
- Charpentier, L.J. The West Indian sugarcane fulgorid in Louisiana. Sugar Bull. 1970, 48, 121–123. [Google Scholar]
- Rodriguez, L.M.; Woolwine, A.E.; Ostheimer, E.A.; Reagan, T.E. Insecticidal control of the West Indian canefly—Small plot insecticide test, 1997. Arthrop. Manag. Tests 1998, 23, 287–288. [Google Scholar] [CrossRef]
- Wilson, B.E.; Beuzelin, J.M.; Reagan, T.E. Field efficacy of insecticides against the West Indian canefly in sugarcane, 2016. Arthrop. Manag. Tests 2017, 42. [Google Scholar] [CrossRef]
- Wilson, B.E.; Beuzelin, J.M.; Reagan, T.E. Assessment of insecticides against the West Indian canefly in sugarcane, 2016. Arthrop. Manag. Tests 2017, 42. [Google Scholar] [CrossRef]
- Wilson, B.E.; Beuzelin, J.M.; Richard, R.; Johnson, R.; Viator, B.; Gravois, K.A.; White, W.H. Material intended for publication. The West Indian canefly (Hemiptera: Delphacidae): An emerging pest of Louisiana sugarcane. J. Econ. Entomol. 2019. in preparation. [Google Scholar]
- Bartlett, C. Sugarcane planthopper Perkinsiella saccharicida (Kirkaldy, 1903) (Hemiptera: Delphacidae). USDA NIFA, 2010. Available online: www.nicpmc.org/sugarcaneplanthopper.pdf (accessed on 9 January 2019).
- Osborn, A.W. Polymorphism in males of the sugarcane leafhopper, Perkinsiella saccharicida. Ann. Entomol. Soc. Am. 1969, 62, 247. [Google Scholar] [CrossRef]
- Bartlett, C. Sugarcane Planthopper Perkinsiella saccharicida. Texas Invasive Species Institute, 2014. Available online: http://www.tsusinvasives.org/home/database/perkinsiella-saccharicida (accessed on 9 January 2019).
- Taniguchi, G.Y.; Ota, A.K.; Chang, V.C.S. Effects of Fiji Disease-resistant sugarcane (Saccharum sp.) on the biology of the sugarcane delphacid. J. Econ. Entomol. 1980, 73, 660–663. [Google Scholar] [CrossRef]
- Fennah, R.G. Damage of sugarcane by fulgoridea and related insects in relation to the metabolic state of the host plant. In Pests of Sugar Cane; Williams, J.R., Mungomery, R.W., Mathes, R., Eds.; Elsevier: Amsterdam, The Netherlands, 1969; pp. 367–389. [Google Scholar]
- Anonymous. Plantwise Technical Factsheet: Sugarcane Planthopper (Perkinsiella saccharicida). Available online: https://www.plantwise.org/KnowledgeBank/Datasheet.aspx?dsID=39680 (accessed on 11 January 2019).
- Sosa, O., Jr. The sugarcane delphacid, Perkinsiella saccharicida (Homoptera: Delphacidae), a sugarcane pest new to North America detected in Florida. Fla. Entomol. 1985, 68, 357–360. [Google Scholar] [CrossRef]
- Meagher, R., Jr.; Wilson, S.W.; Pfannenstiel, R.S.; Breene, R.G. Documentation of two potential sugarcane pests of south Texas sugarcane. Southwestern Entomol. 1991, 16, 365–366. [Google Scholar]
- White, W.H.; Reagan, T.E.; Sosa, O., Jr. The sugarcane delphacid (Homoptera: Delphacidae) extends its North American range into Louisiana. Fla. Entomol. 1995, 78, 617–619. [Google Scholar] [CrossRef]
- Nguyen, R.U.; Sosa, O., Jr.; Mead, F.W. Sugarcane Delphacid, Perkinsiella saccharicida Kirkaldy 1903 (Homoptera: Delphacidae); Florida Department of Agriculture & Consumer Services, Entomology Circular 265: Tallahassee, FL, USA, 1984. [Google Scholar]
- Allsop, P.G.; Bull, R.M. Sampling distributions and sequential sampling plans for Perkinsiella saccharicida Kirkaldy (Hemiptera: Delphacidae) and Tytthus spp. (Hemiptera: Miridae) on sugarcane. J. Econ. Entomol. 1990, 83, 2284–2289. [Google Scholar] [CrossRef]
- Smith, G.R.; Candy, J.M. Improving Fiji disease resistance screening trials in sugarcane by considering virus transmission class and possible origin of Fiji disease virus. Aust. J. Agric. Res. 2004, 55, 665–672. [Google Scholar] [CrossRef]
- Heidemann, O. The sugar-cane tingid from Mexico. J. Econ. Entomol. 1913, 6, 249–251. [Google Scholar] [CrossRef]
- Nguyen, R.; Hall, D.G. Sugarcane Lace Bug, Leptodictya tabida; Publication EENY-044; Florida Cooperative Extension Service, University of Florida: Gainesville, FL, USA, 1998. [Google Scholar]
- Hall, D.G. Sugarcane lace bug Leptodictya tabida, an insect pest new to Florida. Fla. Entomol. 1991, 74, 148–149. [Google Scholar] [CrossRef]
- Chang, V.C.S. The sugarcane lace bug: A new insect pest in Hawaii. Ann. Confer. Rep. Hawaaii. Sugar Technol. 1985, 44, A27–A29. [Google Scholar]
- Hall, D.G.; Sosa, O., Jr. Population levels of Leptodictya tabida (Hemiptera: Tingidae) in Florida sugarcane. Fla. Entomol. 1994, 77, 91–99. [Google Scholar] [CrossRef]
- Sétamou, M.; Showler, A.T.; Reagan, T.E.; Jones, W.A.; Bernal, J.S. Leptodictya tabida (Hemiptera: Tingidae): A potential threat to sugarcane production in lower Rio Grande Valley of Texas. J. Econ. Entomol. 2005, 98, 1018–1023. [Google Scholar] [CrossRef]
- Smith, D.; Anciso, J. Sugarcane in Texas. Texas AgriLife Crop Briefs. Available online: https://aggie-horticulture.tamu.edu/vegetable/guides/crop-briefs/sugarcane-in-texas/ (accessed on 11 April 2019).





© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilson, B.E. Hemipteran Pests of Sugarcane in North America. Insects 2019, 10, 107. https://doi.org/10.3390/insects10040107
Wilson BE. Hemipteran Pests of Sugarcane in North America. Insects. 2019; 10(4):107. https://doi.org/10.3390/insects10040107
Chicago/Turabian StyleWilson, Blake E. 2019. "Hemipteran Pests of Sugarcane in North America" Insects 10, no. 4: 107. https://doi.org/10.3390/insects10040107
APA StyleWilson, B. E. (2019). Hemipteran Pests of Sugarcane in North America. Insects, 10(4), 107. https://doi.org/10.3390/insects10040107




