Knockdown of β-N-acetylglucosaminidase 2 Impairs Molting and Wing Development in Lasioderma serricorne (Fabricius)
Abstract
1. Introduction
2. Materials and Methods
2.1. Insect Culture
2.2. RNA Extraction and Cloning of LsNAG2 Gene
2.3. Sequence Analysis
2.4. Quantitative Real-Time PCR
2.5. RNA Interference
2.6. Statistical Analysis
3. Results
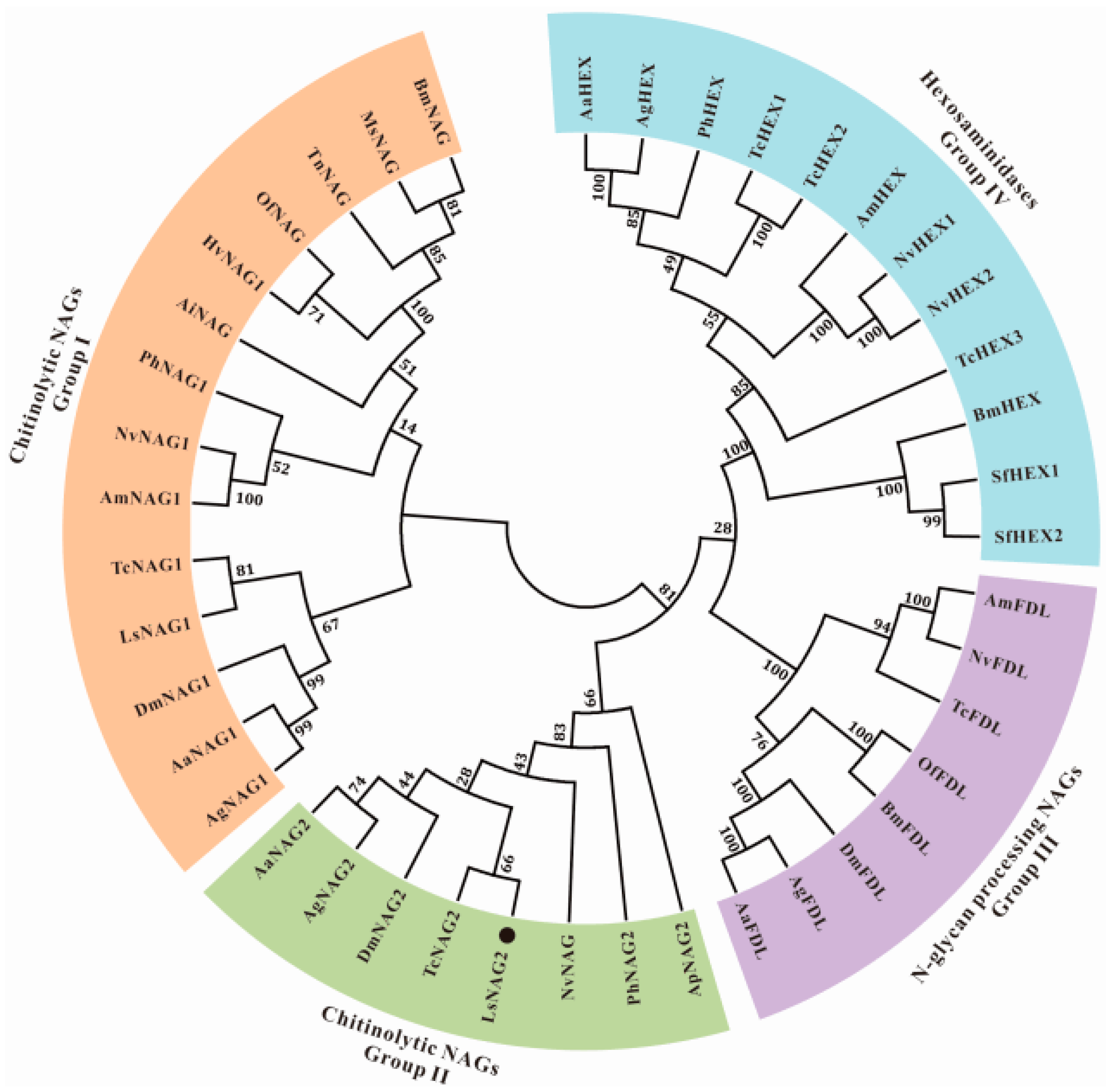
3.1. Identification and Characterization of LsNAG2
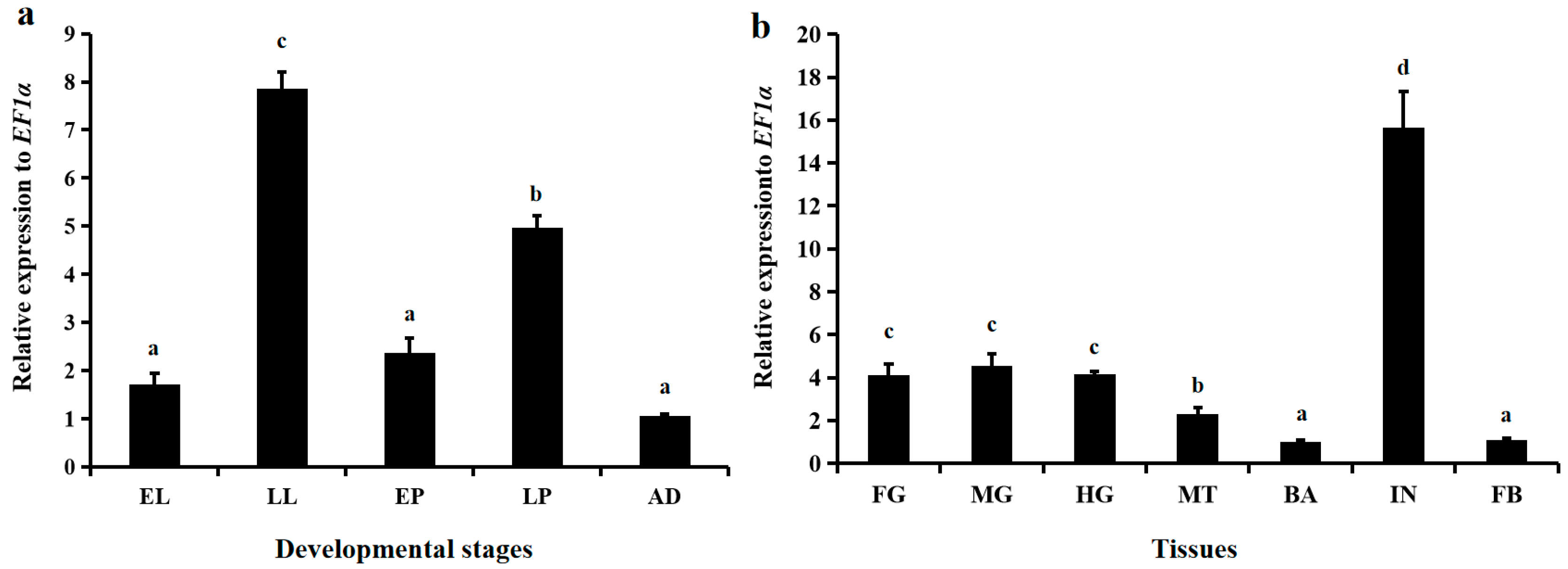
3.2. Spatiotemporal Expression Patterns of LsNAG2
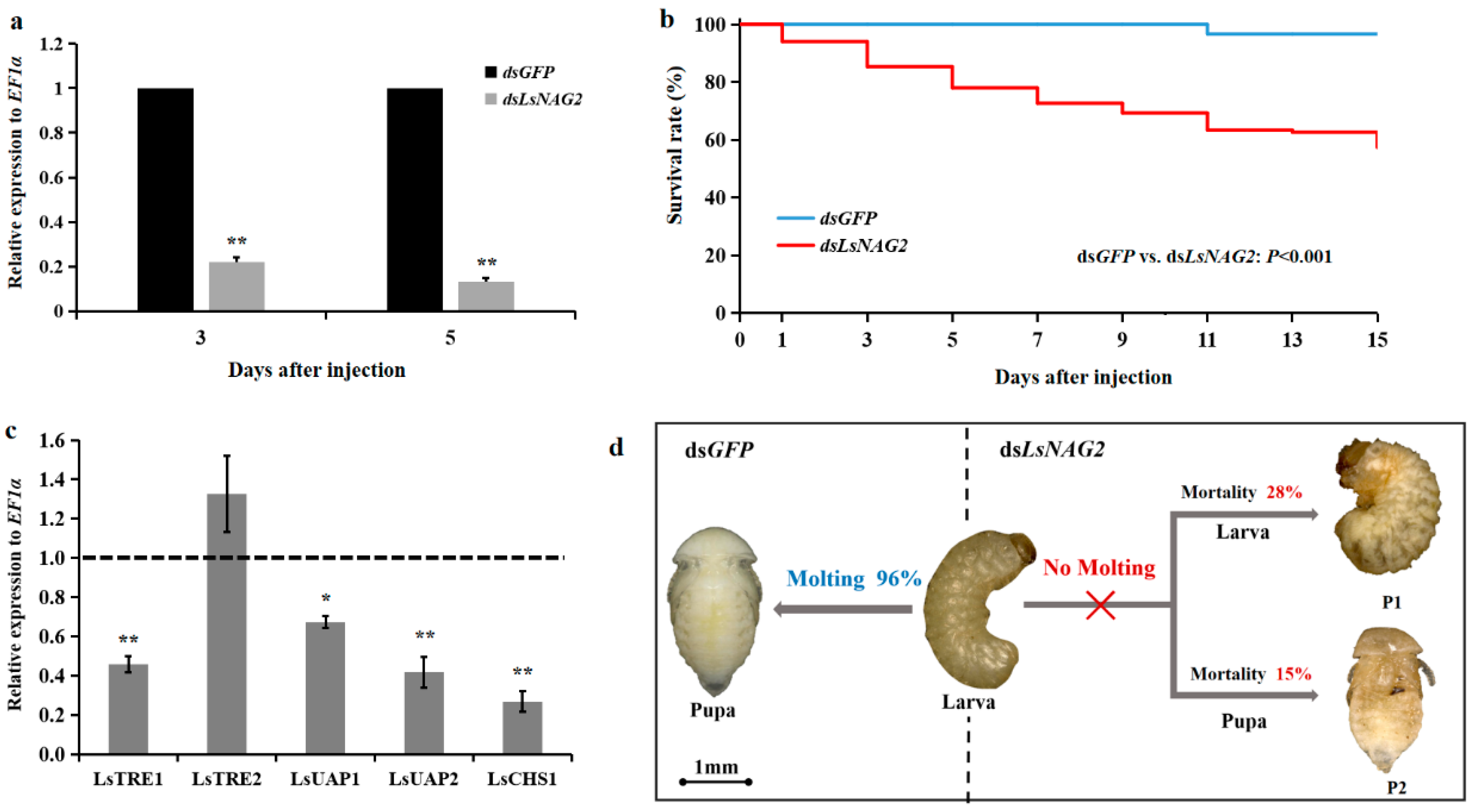
3.3. Effects of LsNAG2 RNAi on the Expressions of Chitin Synthesis Genes
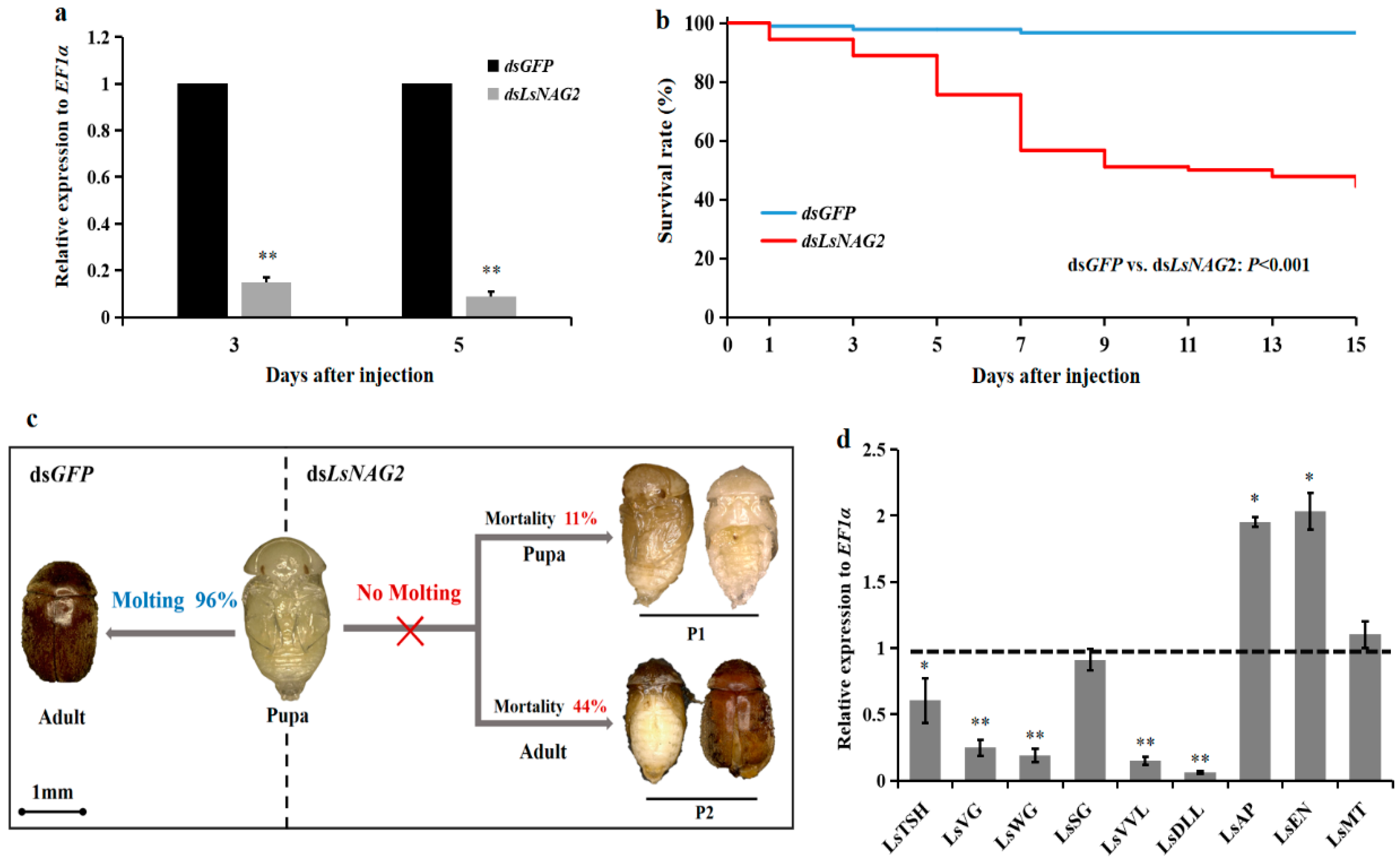
3.4. Effects of LsNAG2 RNAi on the Expressions of Wing Development-Related Genes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kramer, K.J.; Koga, D. Insect chitin: Physical state, synthesis, degradation and metabolic regulation. Insect Biochem. 1986, 16, 851–877. [Google Scholar] [CrossRef]
- Moussian, B. The apical plasma membrane of chitin-synthesizing epithelia. Insect Sci. 2013, 20, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Merzendorfer, H.; Zimoch, L. Chitin metabolism in insects: Structure, function and regulation of chitin synthases and chitinases. J. Exp. Biol. 2003, 206, 4393–4412. [Google Scholar] [CrossRef] [PubMed]
- Moussian, B.; Schwarz, H.; Bartoszewski, S.; Nusslein-Volhard, C. Involvement of chitin in exoskeleton morphogenesis in Drosophila melanogaster. J. Morphol. 2005, 264, 117–130. [Google Scholar] [CrossRef] [PubMed]
- Kramer, K.; Muthukrishnan, S. Chitin metabolism in insects. Compr. Mol. Insect Sci. 2005, 4, 111–144. [Google Scholar]
- Nation, J.L. Insect Physiology and Biochemistry, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2008. [Google Scholar]
- Zen, K.C.; Choi, H.K.; Krishnamachary, N.; Muthukrishnan, S.; Kramer, K.J. Cloning, expression, and hormonal regulation of an insect β-N-acetylhexosaminidase gene. Insect Biochem. Mol. Biol. 1996, 26, 435–444. [Google Scholar] [CrossRef]
- Slámová, K.; Bojarová, P.; Petrásková, L.; Kren, V. beta-N-acetylhexosaminidase: What’s in a name? Biotechnol. Adv. 2010, 28, 682–693. [Google Scholar] [CrossRef]
- Qu, M.B.; Liu, T.; Chen, L.; Chen, Q.; Yang, Q. Research progress in insect glycosyl hydrolyase family 20 β-N-acetylhexosaminidase. Sci. Agric. Sin. 2014, 47, 1303–1312. [Google Scholar]
- Hogenkamp, D.G.; Arakane, Y.; Kramer, K.J.; Muthukrishnan, S.; Beeman, R.W. Characterization and expression of the beta-N-acetylhexosaminidase gene family of Tribolium castaneum. Insect Biochem. Mol. Biol. 2008, 38, 478–489. [Google Scholar] [CrossRef]
- Xi, Y.; Pan, P.L.; Zhang, C.X. The β-N-acetylhexosaminidase gene family in the brown planthopper, Nilaparvata lugens. Insect Mol. Biol. 2015, 24, 601–610. [Google Scholar] [CrossRef]
- Cattaneo, F.; Ogiso, M.; Hoshi, M.; Perotti, M.E.; Pasini, M.E. Purification and characterization of the plasma membrane glycosidases of Drosophila melanogaster spermatozoa. Insect Biochem. Mol. Biol. 2002, 32, 929–941. [Google Scholar] [CrossRef]
- Rong, S.; Li, D.Q.; Zhang, X.Y.; Li, S.; Zhu, K.Y.; Guo, Y.P.; Ma, E.B.; Zhang, J.Z. RNA interference to reveal roles of β-N-acetylglucosaminidase gene during molting process in Locusta migratoria. Insect Sci. 2013, 20, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhao, K.J.; Fan, D. Molecular cloning and RNA interference analysis of β-N-acetylglucosaminidase in Mamestra brassicae L. J. Asia Pac. Entomol. 2016, 19, 721–728. [Google Scholar] [CrossRef]
- Filho, B.P.; Lemos, F.J.; Secundino, N.F.; Pascoa, V.; Pereira, S.T.; Pimenta, P.F. Presence of chitinase and beta-N-acetylglucosaminidase in the Aedes aegypti: A chitinolytic system involving peritrophic matrix formation and degradation. Insect Biochem. Mol. Biol. 2002, 32, 1723–1729. [Google Scholar] [CrossRef]
- Lyu, Z.H.; Chen, J.X.; Li, Z.X.; Cheng, J.; Wang, C.Y.; Lin, T. Knockdown of β-N-acetylglucosaminidase gene disrupts molting process in Heortia vitessoides Moore. Arch. Insect Biochem. Physiol. 2019, 101, e21561. [Google Scholar] [CrossRef]
- Liu, F.Y.; Yang, Q. Investigation of as a potential target with RNA interference in Ostrinia furnacalis. Chin. J. Pestic. Sci. 2013, 15, 145–152. [Google Scholar]
- Chen, X.Y.L.; Xu, K.K.; Yan, X.; Chen, C.X.; Cao, Y.; Wang, Y.W.; Li, C.; Yang, W.J. Characterization of a β-N-acetylglucosaminidase gene and its involvement in the development of Lasioderma serricorne (Fabricius). J. Stored Prod. Res. 2018, 77, 156–165. [Google Scholar] [CrossRef]
- Liu, F.Y.; Liu, T.; Qu, M.B.; Yang, Q. Molecular and biochemical characterization of a novel beta-N-acetyl-D-hexosaminidase with broad substrate-spectrum from the Asian corn borer, Ostrinia furnacalis. Int. J. Biol. Sci. 2012, 8, 1085–1096. [Google Scholar] [CrossRef]
- Leonard, R.; Rendic, D.; Rabouille, C.; Wilson, I.B.H.; Preat, T.; Altmann, F. The Drosophila fused lobes gene encodes an N-acetylglucosaminidase involved in N-glycan processing. J. Biol. Chem. 2006, 281, 4867–4875. [Google Scholar] [CrossRef]
- Huo, Y.M.; Chen, L.; Qu, M.B.; Chen, Q.; Yang, Q. Biochemical characterization of a novel β-N-acetyl-D-hexosaminidase from the insect Ostrinia furnacalis. Arch. Insect Biochem. Physiol. 2013, 83, 115–126. [Google Scholar] [CrossRef]
- Cattaneo, F.; Pasini, M.E.; Intra, J.; Matsumoto, M.; Briani, F.; Hoshi, M.; Perotti, M.E. Identification and expression analysis of Drosophila melanogaster genes encoding beta-hexosaminidases of the sperm plasma membrane. Glycobiology 2006, 16, 786–800. [Google Scholar] [CrossRef] [PubMed]
- Pasini, M.E.; Intra, J.; Gomulski, L.M.; Calvenzani, V.; Petroni, K.; Briani, F.; Perotti, M.E. Identification and expression profiling of Ceratitis capitata genes coding for β-hexosaminidases. Gene 2011, 473, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, J.R. The biology of Lasioderma serricorne. J. Stored Prod. Res. 1993, 29, 291–303. [Google Scholar] [CrossRef]
- Mahroof, R.M.; Phillips, T.W. Life history parameters of Lasioderma serricorne (F.) as influenced by food sources. J. Stored Prod. Res. 2008, 44, 219–226. [Google Scholar] [CrossRef]
- Li, C.; Li, Z.Z.; Cao, Y.; Zhou, B.; Zheng, X.W. Partial characterization of stress-induced carboxylesterase from adults of Stegobium paniceum and Lasioderma serricorne (Coleoptera: Anobiidae) subjected to CO2-enriched atmosphere. J. Pest Sci. 2009, 82, 7–11. [Google Scholar] [CrossRef]
- Kim, S.; Park, C.; Ohh, M.H.; Cho, H.C.; Ahn, Y.J. Contact and fumigant activity of aromatic plant extracts and essential oils against Lasioderma serricorne (Coleoptera: Anobiidae). J. Stored Prod. Res. 2003, 39, 11–19. [Google Scholar] [CrossRef]
- Allahvaisi, S. Controlling Lasioderma serricorne F. (Col.: Anobiidae) by fumigation and packaging. World Appl. Sci. J. 2013, 28, 1983–1988. [Google Scholar]
- Rajendran, S.; Narasimhan, K.S. Phosphine resistance in the cigarette beetle Lasioderma serricorne (Coleoptera: Anobiidae) and overcoming control failures during fumigation of stored tobacco. Int. J. Pest Manag. 1994, 40, 207–210. [Google Scholar] [CrossRef]
- Sağlam, Ö.; Edde, P.A.; Phillips, T.W. Resistance of Lasioderma serricorne (Coleoptera: Anobiidae) to fumigation with phosphine. J. Econ. Entomol. 2015, 108, 2489–2495. [Google Scholar] [CrossRef]
- Huvenne, H.; Smagghe, G. Mechanisms of dsRNA uptake in insects and potential of RNAi for pest control: A review. J. Insect Physiol. 2010, 56, 227–235. [Google Scholar] [CrossRef]
- Khan, A.M.; Ashfaq, M.; Khan, A.A.; Naseem, M.T.; Mansoor, S. Evaluation of potential RNA-interference-target genes to control cotton mealybug, Phenacoccus solenopsis (Hemiptera: Pseudococcuidae). Insect Sci. 2018, 25, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Knorr, E.; Fishilevich, E.; Tenbusch, L.; Frey, M.L.F.; Rangasamy, M.; Billion, A.; Worden, S.E.; Gandra, P.; Arora, K.; Lo, W.; et al. Gene silencing in Tribolium castaneum as a tool for the targeted identification of candidate RNAi targets in crop pests. Sci. Rep. 2018, 8, 2061. [Google Scholar] [CrossRef] [PubMed]
- Kola, V.S.R.; Renuka, P.; Madhav, M.S.; Mangrauthia, S.K. Key enzymes and proteins of crop insects as candidate for RNAi based gene silencing. Front. Physiol. 2015, 6, 119. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Jiang, W.H.; Zhang, Z.; Xing, Y.R.; Li, F. Transcriptome analysis and screening for potential target genes for RNAi-mediated pest control of the beet armyworm, Spodoptera exigua. PLoS ONE 2013, 8, e65931. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Khan, S.A.; Hasse, C.; Ruf, S.; Heckel, D.G.; Bock, R. Full crop protection from an insect pest by expression of long double-stranded rnas in plastids. Science 2015, 347, 991–994. [Google Scholar] [CrossRef] [PubMed]
- Yan, S.; Qian, J.; Cai, C.; Ma, Z.Z.; Li, H.J.; Yin, M.Z.; Ren, B.Y.; Shen, J. Spray method application of transdermal dsRNA delivery system for efficient gene silencing and pest control on soybean aphid Aphis glycines. J. Pest Sci. 2019, 1–11. [Google Scholar] [CrossRef]
- Khajuria, C.; Ivashuta, S.; Wiggins, E.; Flagel, L.; Moar, W.; Pleau, M.; Miller, K.; Zhang, Y.J.; Ramaseshadri, P.; Jiang, C.J.; et al. Development and characterization of the first dsRNA-resistant insect population from western corn rootworm, Diabrotica virgifera virgifera LeConte. PLoS ONE 2018, 13, e0197059. [Google Scholar] [CrossRef]
- Cohen, E. Chitin synthesis and degradation as targets for pesticide action. Arch. Insect Biochem. Physiol. 1993, 22, 245–261. [Google Scholar] [CrossRef]
- Kramer, K.J.; Muthukrishnan, S. Insect chitinases: Molecular biology and potential use as biopesticides. Insect Biochem. Mol. Biol. 1997, 27, 887–900. [Google Scholar] [CrossRef]
- Zhao, X.X.; Situ, G.M.; He, K.; Xiao, H.M.; Su, C.C.; Li, F. Function analysis of eight chitinase genes in rice stem borer and their potential application in pest control. Insect Mol. Biol. 2018, 27, 835–846. [Google Scholar] [CrossRef]
- Yang, W.J.; Xu, K.K.; Cao, Y.; Meng, Y.L.; Liu, Y.; Li, C. Identification and expression analysis of four small heat shock protein genes in cigarette beetle, Lasioderma serricorne (Fabricius). Insects 2019, 10, 139. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Zhai, Y.F.; Huang, M.X.; Wu, Y.; Zhao, G.D.; Du, J.; Li, B.; Shen, W.D.; Wei, Z.G. The expression profile and promoter analysis of β-N-acetylglucosaminidases in the silkworm Bombyx mori. Mol. Biol. Rep. 2014, 41, 6667–6678. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhang, J.; Xiang, J. Molecular characterization and function of β-N-acetylglucosaminidase from ridgetail white prawn Exopalaemon carinicauda. Gene 2018, 648, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, H.T.; Liu, F.Y.; Wu, Q.Y.; Shen, X.; Yang, Q. Structural determinants of an insect β-N-acetyl-D-hexosaminidase specialized as a chitinolytic enzyme. J. Biol. Chem. 2011, 286, 4049–4058. [Google Scholar] [CrossRef]
- Zheng, Y.P.; Krell, P.J.; Doucet, D.; Arif, B.M.; Feng, Q.L. Cloning, expression, and localization of a molt-related β-N-acetylglucosaminidase in the spruce budworm, Choristoneura fumiferana. Arch. Insect Biochem. Physiol. 2008, 68, 49–59. [Google Scholar] [CrossRef]
- Yang, W.J.; Xu, K.K.; Yan, X.; Chen, C.X.; Cao, Y.; Meng, Y.L.; Li, C. Functional characterization of chitin deacetylase 1 gene disrupting larval–pupal transition in the drugstore beetle using RNA interference. Comp. Biochem. Physiol. Part B 2018, 219, 10–16. [Google Scholar] [CrossRef]
- Wu, Q.; Liu, T.; Yang, Q. Cloning, expression and biocharacterization of OfCht5, the chitinase from the insect Ostrinia furnacalis. Insect Sci. 2013, 20, 147–157. [Google Scholar] [CrossRef]
- Zhu, K.Y.; Merzendorfer, H.; Zhang, W.; Zhang, J.Z.; Muthukrishnan, S. Biosynthesis, turnover, and functions of chitin in insects. Annu. Rev. Entomol. 2016, 61, 177–196. [Google Scholar] [CrossRef]
- Zhu, Q.; Arakane, Y.; Beeman, R.W.; Kramer, K.J.; Muthukrishnan, S. Functional specialization among insect chitinase family genes revealed by RNA interference. Proc. Natl. Acad. Sci. USA 2008, 105, 6650–6655. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Qiu, L.Y.; Yang, H.L.; Wang, H.J.; Zhou, M.; Wang, S.G.; Tang, B. Study on the effect of wing bud chitin metabolism and its developmental network genes in the brown planthopper, Nilaparvata lugens, by knockdown of TRE gene. Front. Physiol. 2017, 8, 750. [Google Scholar] [CrossRef] [PubMed]
- Brook, W.J.; Sm, D.B.F. Organizing spatial pattern in limb development. Annu. Rev. Cell Dev. Biol. 1996, 12, 161–180. [Google Scholar] [CrossRef] [PubMed]
- Clark-Hachtel, C.M.; Linz, D.M.; Tomoyasu, Y. Insights into insect wing origin provided by functional analysis of vestigial in the red flour beetle, Tribolium castaneum. Proc. Natl. Acad. Sci. USA 2013, 110, 16951–16956. [Google Scholar] [CrossRef] [PubMed]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, W.-J.; Xu, K.-K.; Yan, X.; Li, C. Knockdown of β-N-acetylglucosaminidase 2 Impairs Molting and Wing Development in Lasioderma serricorne (Fabricius). Insects 2019, 10, 396. https://doi.org/10.3390/insects10110396
Yang W-J, Xu K-K, Yan X, Li C. Knockdown of β-N-acetylglucosaminidase 2 Impairs Molting and Wing Development in Lasioderma serricorne (Fabricius). Insects. 2019; 10(11):396. https://doi.org/10.3390/insects10110396
Chicago/Turabian StyleYang, Wen-Jia, Kang-Kang Xu, Xin Yan, and Can Li. 2019. "Knockdown of β-N-acetylglucosaminidase 2 Impairs Molting and Wing Development in Lasioderma serricorne (Fabricius)" Insects 10, no. 11: 396. https://doi.org/10.3390/insects10110396
APA StyleYang, W.-J., Xu, K.-K., Yan, X., & Li, C. (2019). Knockdown of β-N-acetylglucosaminidase 2 Impairs Molting and Wing Development in Lasioderma serricorne (Fabricius). Insects, 10(11), 396. https://doi.org/10.3390/insects10110396




