Ιnteractions between Beauveria bassiana and Isaria fumosorosea and Their Hosts Sitophilus granarius (L.) and Sitophilus oryzae (L.) (Coleoptera: Curculionidae)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Insect Rearing
2.2. Entomopathogenic Fungi
2.3. Conidial Suspensions
2.4. Bioassays
2.5. Microscopic Fungal Identification Method
2.6. Mathematical Estimation
2.7. Statistical Analysis
3. Results
3.1. Separate and Combined Mortality
3.2. Fungal Sporulation
3.3. Medial Survival Time
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rice, W.C.; Cogburn, R.R. Activity of the entomopathogenic fungus Beauveria bassiana (Deuteromycota: Hyphomycetes) against three coleopteran pests of stored grain. J. Econ. Entomol. 1999, 92, 691–694. [Google Scholar] [CrossRef]
- Padin, S.; Bello, G.D.; Fabrizio, M. Grain loss caused by Tribolium castaneum, Sitophilus oryzae and Acanthoscelides obtectus in stored durum wheat and beans treated with Beauveria bassiana. J. Stored Prod. Res. 2002, 38, 69–74. [Google Scholar] [CrossRef]
- Boush, G.M.; Matsumura, F.; Misato, T. Environmental Toxicology of Pesticides; Academic Press: New York, NY, USA, 1972. [Google Scholar]
- Matsumura, F. Toxicology of Insecticides; Plenum Press: New York, NY, USA, 1980. [Google Scholar]
- Thaung, N.; Collins, P.J. Joint effect of temperature and insecticides on mortality and fecundity of Sitophilus oryzae (Coleoptera: Curculinoidae) in wheat and maize. J. Econ. Entomol. 1986, 79, 909–914. [Google Scholar] [CrossRef]
- Moino, A., Jr.; Alves, S.B.; Pereira, R.M.; Pereira, R. Efficacy of Beauveria bassiana (Balsamo) Vuillemin isolates for control of stored-grain pests. J. Appl. Entomol. 1998, 122, 301–305. [Google Scholar] [CrossRef]
- Benhalima, H.; Chaudhry, M.Q.; Mills, K.A.; Price, N.R. Phosphine resistance in stored-product insects collected from various grain storage facilities in Morocco. J. Stored Prod. Res. 2004, 40, 241–249. [Google Scholar] [CrossRef]
- Moore, D.; Lord, J.C.; Smith, S.M. Pathogens. In Alternatives to Pesticides in Stored Product IPM; Subramanyam, B.H., Hagstrum, D.W., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2000; pp. 193–227. [Google Scholar]
- Bourassa, C.; Vincent, C.; Lomer, J.; Borgemeister, C.; Mauffette, Y. Effects of entomopathogenic Hyphomycetes against the larger grain borer, Prostephanus truncatus (Horn) (Coleoptera: Bostrichidae) and its predator, Teretriosoma nigrescens Lewis (Coleoptera: Histeridae). J. Invertebr. Pathol. 2001, 77, 75–77. [Google Scholar] [CrossRef]
- Kavallieratos, N.G.; Athanassiou, C.G.; Michalaki, M.P.; Batta, Y.A.; Rigatos, H.A.; Pashalidou, F.G.; Balotis, G.N.; Tomanovic, Z.; Vayias, B.J. Effect of the combined use of Metarhizium anisopliae (Metschinkoff) Sorokin and diatomaceous earth for the control of three stored-product beetle species. Crop. Prot. 2006, 25, 1087–1094. [Google Scholar] [CrossRef]
- Lemanceau, P.; Alabouvette, C. Suppression of Fusarium-wilts by fluorescent pseudomonads: Mechanisms and applications. Biocontrol Sci. Technol. 1993, 3, 219–234. [Google Scholar] [CrossRef]
- Wakil, W.; Riasat, T.; Ghazanfar, M.U.; Kwon, Y.J.; Shaheen, F.A.; Wakil, W.; Kwon, Y.J. Aptness of Beauveria bassiana and enhanced diatomaceous earth (DEBBM) for control of Rhyzopertha dominica F. Entomol. Res. 2011, 41, 233–241. [Google Scholar] [CrossRef]
- Wakil, W.; Riasat, T.; Ashfaq, M. Residual efficacy of thiamethoxam, Beauveria bassiana (Balsamo) Vuillemin, and diatomaceous earth formulation against Rhyzopertha dominica F. (Coleoptera: Bostrychidae). J. Pest. Sci. 2012, 85, 341–350. [Google Scholar] [CrossRef]
- Mantzoukas, S.; Milonas, P.; Kontodimas, D.; Angelopoulos, K. Interaction between the entomopathogenic bacterium Bacillus thuringiensis subsp. kurstaki and two entomopathogenic fungi in biocontrol of Sesamia nonagrioides (Lefebvre) (Lepidoptera: Noctuidae). Ann. Microbiol. 2013, 63, 1083–1091. [Google Scholar]
- Jacques, R.P.; Morris, O.N. Compatibility of pathogens with other methods of pest control and with different crops. In Microbial Control of Insect and Mites; Burges, H.D., Hussey, N.W., Eds.; Academic Press: New York, NY, USA, 1981; pp. 695–715. [Google Scholar]
- Ansari, M.A.; Shah, F.A.; Tirry, L.; Moens, M. Field trials against Hoplia philanthus (Coleoptera: Scarabaeidae) with a combination of an entomopathogenic nematode and the fungus Metarhizium anisopliae CLO 53. Biol. Control 2006, 39, 453–459. [Google Scholar] [CrossRef]
- Ansari, M.A.; Shah, F.A.; Butt, T.M. Combined use of entomopathogenic nematodes and Metarhizium anisopliae as a new approach for black vine weevil, Otiorhynchus sulcatus, control. Entomol. Exp. Appl. 2008, 129, 340–347. [Google Scholar] [CrossRef]
- Wraight, S.P.; Ramos, M.E. Synergistic interaction between Beauveria bassiana and Bacillus thuringiensis tenebrionis-based biopesticides applied against field populations of Colorado potato beetle larvae. J. Invertebr. Pathol. 2005, 90, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Kryukov, V.Y.; Khodyrev, V.P.; Yaroslavtseva, O.N.; Kamenova, A.S.; Duisembekov, B.A.; Glupov, V.V. Synergistic action of entomopathogenic Hyphomycetes and the bacteria Bacillus thuringiensis ssp. morrisoni in the infection of Colorado potato beetle Leptinotarsa decemlineata. Appl. Biochem. Microbiol. 2009, 45, 511–516. [Google Scholar]
- Ma, X.-M.; Liu, X.-X.; Ning, X.; Zhang, B.; Han, F.; Guan, X.-M.; Tang, Y.-F.; Zhang, Q.-W. Effects of Bacillus thuringiensis toxin Cry1Ac and Beauveria bassiana on Asiatic corn borer (Lepidoptera: Crambidae). J. Invertebr. Pathol. 2008, 99, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Lewis, L.C.; Berry, E.C.; Obrycki, J.J.; Bing, L.A. Aptness of insecticides (Bacillus thuringiensis and carbofuran) with endophytic Beauveria bassiana, in suppressing larval populations of the European corn borer. Agric. Ecosyst. Environ. 1996, 57, 27–34. [Google Scholar] [CrossRef]
- Narciso, J.; Ormskirk, M.; Jones, S.; Rolston, P.; Moran-Diez, M.E.; Hurst, M.; Brookes, J.; Glare, T. Using multiple insecticidal microbial agents against diamondback moth larvae - does it increase toxicity? N. Z. J. Agric. Res. 2019. [Google Scholar] [CrossRef]
- Pauli, G.; Mascarin, G.M.; Eilenberg, J.; Delalibera, I., Jr. Within-Host Competition between Two Entomopathogenic Fungi and a Granulovirus in Diatraea saccharalis (Lepidoptera: Crambidae). Insects 2018, 9, 64. [Google Scholar] [CrossRef]
- Tarasco, E.; Santiago Alvarez, C.; Triggiani, O.; Quesada Moraga, E. Laboratory studies on the competition for insect haemocoel between Beauveria bassiana and Steinernema ichnusae recovered in the same ecological niche. Biocontrol Sci. Technol. 2011, 21, 693–704. [Google Scholar] [CrossRef]
- Shapiro-Ilan, D.I.; Jackson, M.; Reilly, C.C.; Hotchkiss, M.W. Effects of combining an entomopathogenic fungi or bacterium with entomopathogenic nematodes on mortality of Curculio caryae (Coleoptera: Curculionidae). Biol. Control 2004, 30, 119–126. [Google Scholar] [CrossRef]
- Ansari, M.A.; Shah, F.A.; Butt, T.M. The entomopathogenic nematode Steinernema kraussei and Metarhizium anisopliae work synergistically in controlling overwintering larvae of the black vine weevil, Otiorhynchus sulcatus, in strawberry growbags. Biocontrol Sci. Technol. 2010, 20, 99–105. [Google Scholar] [CrossRef]
- Quintela, E.D.; McCoy, C.W. Synergistic effect of imidacloprid and two entomopathogenic fungi on the behavior and survival of larvae of Diaprepes abbreviatus (Coleoptera: Curculionidae) in soil. J. Econ. Entomol. 1998, 91, 110–122. [Google Scholar] [CrossRef]
- Purwar, J.P.; Sachan, G.C. Synergistic effect of entomogenous fungi on some insecticides against Bihar hairy caterpillar Spilarctia obliqua (Lepidoptera: Arctiidae). Microbiol. Res. 2006, 161, 38–42. [Google Scholar] [CrossRef]
- Santos, A.V.; de Oliveira, B.L.; Samuels, R.I. Selection of entomopathogenic fungi for use in combination with sub-lethal doses of imidacloprid: Perspectives for the control of the leaf-cutting ant Atta sexdensrubropilosa Forel (Hymenoptera: Formicidae). Mycopathologia 2007, 163, 233–240. [Google Scholar] [CrossRef]
- Farenhorst, M.; Knols, B.G.; Thomas, M.B.; Howard, A.F.; Takken, W.; Rowland, M.; N’Guessan, R. Synergy in efficacy of fungal entomopathogens and permethrin against West African insecticide-resistant Anopheles gambiae mosquitoes. PLoS ONE 2010, 5, e12081. [Google Scholar] [CrossRef]
- Uma Maheswara Rao, C.; Uma Devi, K.; Akbar Ali Khan, P. Effect of combination treatment with entomopathogenic fungi Beauveria bassiana and Nomuraea rileyi (Hypocreales) on Spodoptera litura (Lepidoptera: Noctuidaeae). Biocontrol Sci. Technol. 2006, 16, 221–232. [Google Scholar] [CrossRef]
- Inglis, G.D.; Johnson, D.L.; Cheng, K.J.; Goettel, M.S. Use of pathogen combinations to overcome the constraints of temperature on entomopathogenic hyphomycetes against grasshoppers. Biol. Control 1997, 8, 143–152. [Google Scholar] [CrossRef]
- Dakhel, W.H.; Latchininsky, A.V.; Jaronski, S.T. Efficacy of two entomopathogenic fungi, Metarhizium brunneum, strain F52 alone and combined with Paranosema locustae against the migratory grasshopper, Melanoplus sanguinipes, under laboratory and greenhouse conditions. Insects 2019, 10, 94. [Google Scholar] [CrossRef]
- Mora, M.A.E.; Castilho, A.M.C.; Fraga, M.E. Classification and infection mechanism of entomopathogenic fungi. Arq. Inst. Biol. 2017, 84, e0552015. [Google Scholar] [CrossRef]
- Gillespie, A.T.; Claydon, N. The use of entomogenous fungi for pest control and the role of toxins in pathogenesis. Pestic. Sci. 1989, 27, 203–215. [Google Scholar] [CrossRef]
- Valverde-Garcia, P.; Santiago-Alvarez, C.; Thomas, M.B.; Garrido-Jurado, I.; Quesada-Moraga, E. Comparative effects of temperature and thermoregulation on candidate strains of entomopathogenic fungi for Moroccan locust Dociostaurus maroccanus control. BioControl 2018, 63, 819–831. [Google Scholar] [CrossRef]
- Valverde-Garcia, P.; Santiago-Álvarez, C.; Thomas, M.B.; Maranhao, E.A.; Garrido-Jurado, I.; Quesada-Moraga, E. Sublethal effects of mixed fungal infections on the Moroccan locust, Dociostaurus maroccanus. J. Invertebr. Pathol. 2019, 161, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Gurulingappa, P.; Sword, G.A.; Murdoch, G.; McGee, P.A. Colonization of crop plants by fungal entomopathogens and their effects on two insect pests when in planta. Biol. Control 2010, 55, 34–41. [Google Scholar] [CrossRef]
- Goettel, M.S.; Inglis, G.D. Fungi: Hyphomycetes. Manual of Techniques in Insect Pathology; Academic Press: San Diego, CA, USA, 1997. [Google Scholar]
- Robertson, J.L.; Preisler, H.K. Pesticide Bioassays with Arthropods; CRC: Boca Raton, FL, USA, 1992. [Google Scholar]
- Diaz, M.P.; Macias, A.F.; Navarro, S.R.; de la Torres, M. Mechanism of action of entomopathogenic fungi. Interciencia 2006, 31, 856–860. [Google Scholar]
- Adane, K.; Moore, D.; Archer, S.A. Preliminary studies on the use of Beauveria bassiana to control Sitophilus zeamais (Coleoptera: Curculionidae) in the laboratory. J. Stored Prod. Res. 1996, 32, 105–113. [Google Scholar] [CrossRef]
- Batta, Y.A. Efficacy of two species of entomopathogenic fungi against the stored-grain pest, Sitophilus granarius L. (Curculionidae: Coleoptera), via oral ingestion. Egypt. J. Biol. Pest Control 2018, 28, 44. [Google Scholar] [CrossRef]
- Sugandi, R.; Awaknavar, J.S. The pathogenicity of entomopathogenic fungus, Metarhizium anisopliae (Metschinkoff) Sorokin against three major storage grain beetles. J. Exp. Zool. 2014, 17, 775–780. [Google Scholar]
- Mietkiewski, R.; Gorski, R. Growth of selected entomopathogenic fungi species and isolates on media containing insecticides. Acta Mycol. 1995, 30, 27–33. [Google Scholar] [CrossRef]
- Staves, P.A.; Knell, R.J. Virulence and Competitiveness: Testing the relationship between inter- and intraspecific mixed infections. Evolution 2009, 64, 2643–2652. [Google Scholar] [CrossRef]
- Bauer, L.S.; Miller, D.L.; Maddox, J.V.; McManus, M.L. Interactions between a Nosema sp. (Microspora: Nosematidae) and nuclear polyhedrosis virus infecting the gypsy moth, Lymantria dispar (Lepidoptera: Lymantriidae). J. Invertebr. Pathol. 1998, 74, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.B.; Watson, E.L.; Valverde-Garcia, P. Mixed infections and insect pathogen interactions. Ecol. Lett. 2003, 6, 183–188. [Google Scholar] [CrossRef]
- Cedergreen, N. Quantifying synergy: A systematic review of mixture toxicity studies within environmental toxicology. PLoS ONE 2014, 9, e96580. [Google Scholar] [CrossRef] [PubMed]
- Roell, K.R.; Reif, D.M.; Motsinger-Reif, A.A. An introduction to terminology and methodology of chemical synergy—Perspectives from across Disciplines. Front. Pharmacol. 2017, 8, 158. [Google Scholar] [CrossRef]
- Raymond, B.; Sayyed, A.H.; Wright, D.J. The compatibility of a nucleopolyhedrosis virus control with resistance management for Bacillus thuringiensis: Co-infection and cross-resistance studies with the diamondback moth. Plutella xylostella. J. Invertebr. Pathol. 2006, 93, 114–120. [Google Scholar] [CrossRef]
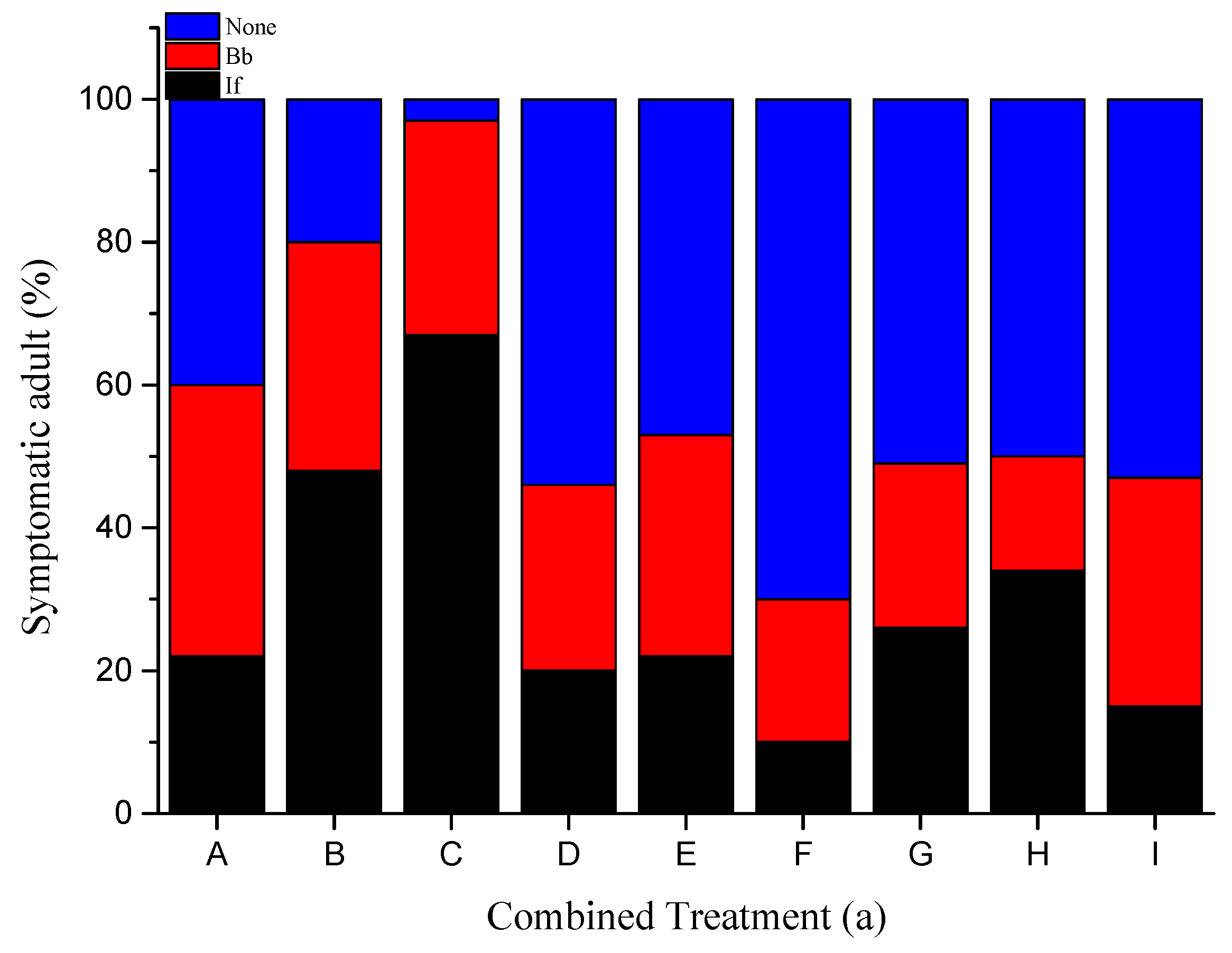
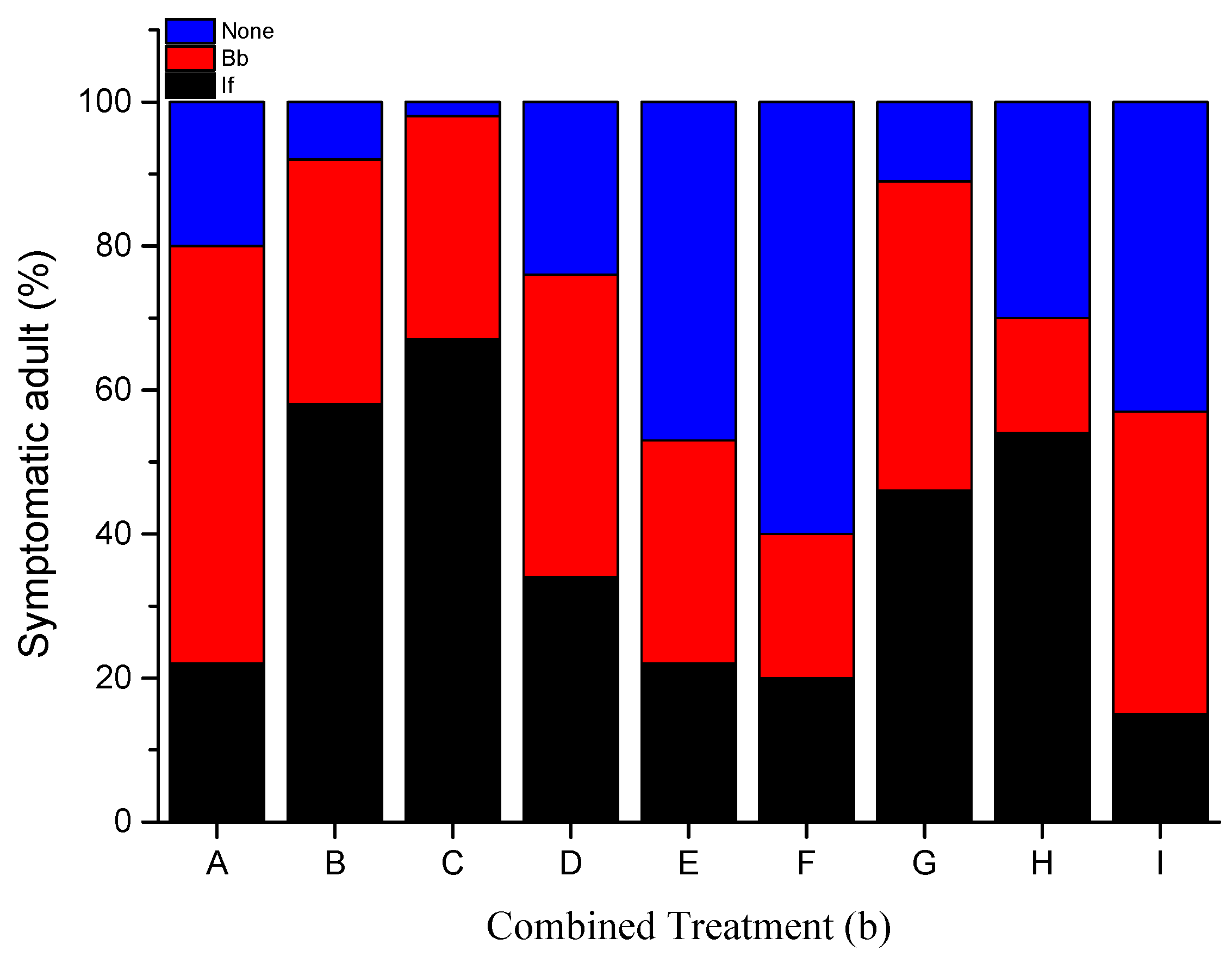
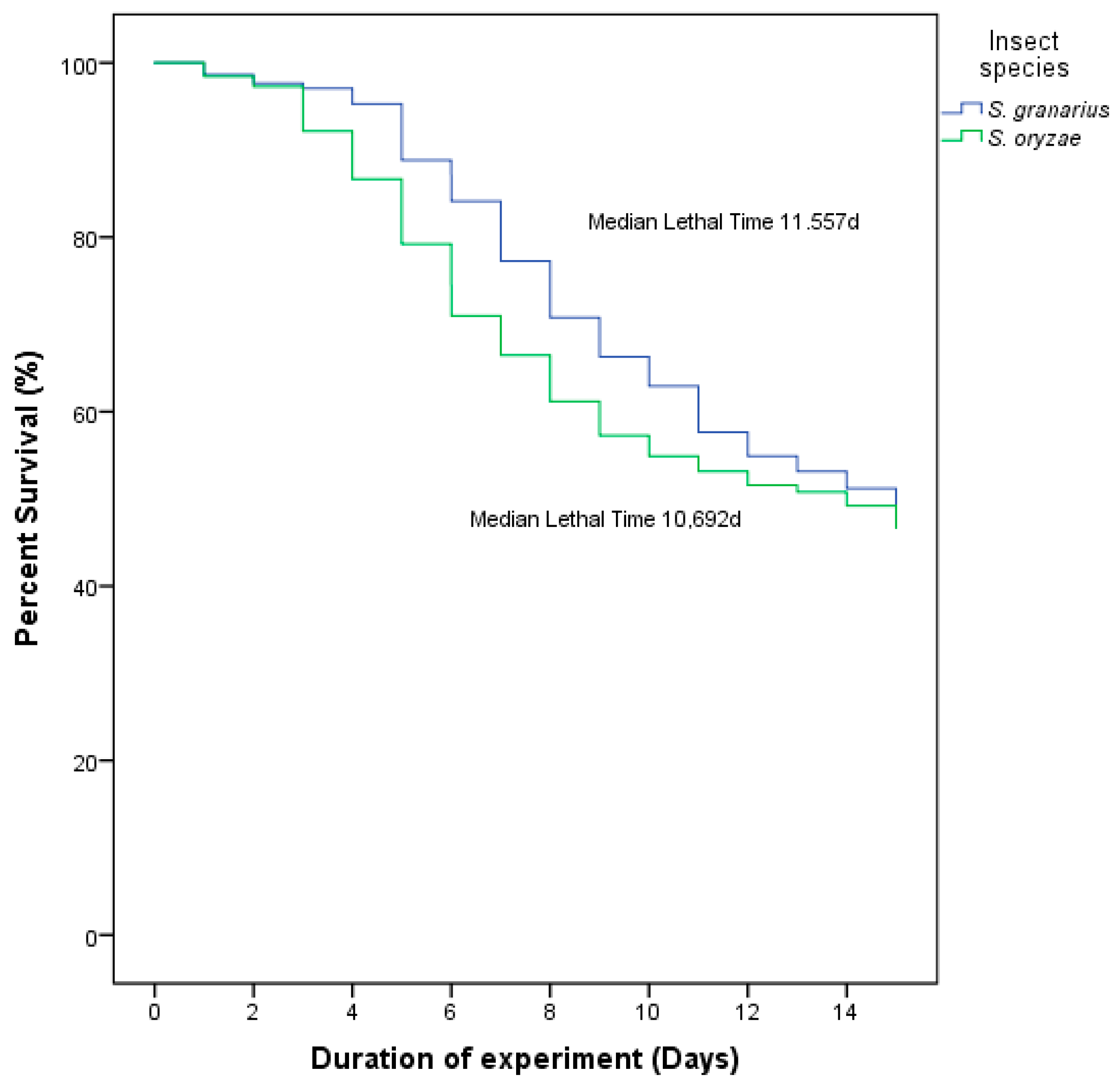
| Insect | Concentration (conidia/mL) | Mortality | Median Survival Time (Days) | |||||
|---|---|---|---|---|---|---|---|---|
| Bb | If | (%) | Sd | Estimate | Sd | 95% Confidence Interval | ||
| Lower Bound | Upper Bound | |||||||
| S. granarius | 0 | 0 | 0.00 | 0.00 | 15.000a | |||
| 104 | 0 | 26.67aA | 5.77 | 12.800bc | 0.772 | 11.286 | 14.314 | |
| 106 | 0 | 46.67bA | 10.00 | 11.867b | 0.779 | 10.341 | 13.393 | |
| 108 | 0 | 53.33bA | 5.77 | 12.000b | 0.721 | 10.587 | 13.413 | |
| 0 | 104 | 20.00aA | 0.00 | 13.700c | 0.517 | 12.687 | 14.713 | |
| 0 | 106 | 36.67bA | 5.77 | 12.500bc | 0.679 | 11.168 | 13.832 | |
| 0 | 108 | 53.33bA | 11.55 | 10.767d | 0.831 | 9.138 | 12.395 | |
| S. oryzae | 0 | 0 | 3.33 | 0.00 | 14.855a | 0.145 | 14.710 | 15.000 |
| 104 | 0 | 36.60aA | 11.55 | 13.567c | 0.466 | 12.654 | 14.479 | |
| 106 | 0 | 43.33aA | 11.55 | 12.100b | 0.836 | 10.461 | 13.739 | |
| 108 | 0 | 63.33aA | 5.77 | 10.467d | 0.895 | 8.712 | 12.221 | |
| 0 | 104 | 46.67aB | 5.77 | 10.433d | 1.017 | 8.439 | 12.427 | |
| 0 | 106 | 55.00aB | 7.07 | 10.800d | 0.918 | 9.000 | 12.600 | |
| 0 | 108 | 66.67aA | 15.28 | 9.700d | 0.880 | 7.975 | 11.425 | |
| Insect | Concentration (conidia/mL) | Mortality | Median Survival Time (Days) * | |||||
|---|---|---|---|---|---|---|---|---|
| Bb | If | (%) | Sd | Estimate | Sd | 95% Confidence Interval | ||
| Lower Bound | Upper Bound | |||||||
| S. granarius | 108 | 104 | 66.67bcA | 11.55 | 10.100a | 0.712 | 8.704 | 11.496 |
| 106 | 106 | 56.67bA | 5.77 | 11.367b | 0.796 | 9.806 | 12.927 | |
| 104 | 108 | 63.33bcA | 11.55 | 11.067b | 0.689 | 9.715 | 12.418 | |
| 108 | 106 | 70.00bcA | 10.00 | 9.900a | 0.753 | 8.425 | 11.375 | |
| 106 | 108 | 66.67bcA | 7.07 | 9.800a | 0.807 | 8.217 | 11.383 | |
| 104 | 104 | 30.00aA | 0.00 | 13.000c | 0.609 | 11.806 | 14.194 | |
| 108 | 108 | 86.67cA | 7.07 | 9.300a | 0.661 | 8.005 | 10.595 | |
| 104 | 106 | 46.67bA | 5.77 | 11.267b | 0.786 | 9.726 | 12.807 | |
| 106 | 104 | 56.67bA | 5.77 | 10.367ab | 0.802 | 9.710 | 11.939 | |
| S. oryzae | 108 | 104 | 46.67abA | 5.77 | 11.767a | 0.803 | 10.192 | 13.341 |
| 106 | 106 | 50.00bA | 0.00 | 11.433a | 0.721 | 10.021 | 12.846 | |
| 104 | 108 | 46.67abA | 5.77 | 11.200a | 0.918 | 9.400 | 13.000 | |
| 108 | 106 | 80.00cA | 5.77 | 8.300b | 0.752 | 6.827 | 9.773 | |
| 106 | 108 | 73.33bcA | 5.77 | 8.500b | 0.928 | 6.682 | 10.318 | |
| 104 | 104 | 36.67aA | 5.77 | 12.033a | 0.846 | 10.376 | 13.691 | |
| 108 | 108 | 83.33cA | 5.77 | 7.767c | 0.718 | 6.360 | 9.173 | |
| 104 | 106 | 56.67bA | 11.55 | 10.187a | 0.805 | 8.610 | 11.765 | |
| 106 | 104 | 60.00bcA | 10.00 | 10.133a | 0.824 | 8.518 | 11.749 | |
| Combined Concentration (conidia/mL) | Mortality (%) * | χ2 (1 df; p = 0.05) | Interaction | ||
|---|---|---|---|---|---|
| Bb | If | Observed | Expected | ||
| S. granarius | |||||
| 108 | 104 | 67 | 66 | −0.012 | A |
| 106 | 106 | 57 | 66 | 0.138 | A |
| 104 | 108 | 63 | 63 | 0.034 | A |
| 108 | 106 | 67- | 80 | 0.520 | A |
| 106 | 108 | 70 | 70 | 0.008 | A |
| 104 | 104 | 30 | 41 | −0.161 | A |
| 108 | 108 | 80 | 78 | −0.280 | A |
| 104 | 106 | 46 | 54 | 0.019 | A |
| 106 | 104 | 57 | 57 | 0.004 | A |
| S. oryzae | |||||
| 108 | 104 | 77 | 81 | 0.366 | A |
| 106 | 106 | 51 | 75 | 7.917 | C |
| 104 | 108 | 78 | 80 | 0.070 | A |
| 108 | 106 | 73 | 83 | 1.986 | A |
| 106 | 108 | 67 | 84 | 2.607 | A |
| 104 | 104 | 37 | 62 | 8.172 | C |
| 108 | 108 | 89 | 83 | 0.984 | A |
| 104 | 106 | 60 | 72 | 2.375 | A |
| 106 | 104 | 57 | 66 | 1.164 | A |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mantzoukas, S.; Zikou, A.; Triantafillou, V.; Lagogiannis, I.; Eliopoulos, P.A. Ιnteractions between Beauveria bassiana and Isaria fumosorosea and Their Hosts Sitophilus granarius (L.) and Sitophilus oryzae (L.) (Coleoptera: Curculionidae). Insects 2019, 10, 362. https://doi.org/10.3390/insects10100362
Mantzoukas S, Zikou A, Triantafillou V, Lagogiannis I, Eliopoulos PA. Ιnteractions between Beauveria bassiana and Isaria fumosorosea and Their Hosts Sitophilus granarius (L.) and Sitophilus oryzae (L.) (Coleoptera: Curculionidae). Insects. 2019; 10(10):362. https://doi.org/10.3390/insects10100362
Chicago/Turabian StyleMantzoukas, Spiridon, Athanasia Zikou, Vasw Triantafillou, Ioannis Lagogiannis, and Panagiotis A. Eliopoulos. 2019. "Ιnteractions between Beauveria bassiana and Isaria fumosorosea and Their Hosts Sitophilus granarius (L.) and Sitophilus oryzae (L.) (Coleoptera: Curculionidae)" Insects 10, no. 10: 362. https://doi.org/10.3390/insects10100362







