New Method of Analysis of Lipids in Tribolium castaneum (Herbst) and Rhyzopertha dominica (Fabricius) Insects by Direct Immersion Solid-Phase Microextraction (DI-SPME) Coupled with GC–MS
Abstract
:1. Introduction
2. Material and Methods
2.1. The Insect Culture
2.2. Chemical Reagents and Equipment Used
2.3. GC–MS Instrument and Analytical Conditions
2.4. The Extraction and Analytical Procedures
2.4.1. Preparation of Diluted Standards
2.4.2. Evaluation of the Effect of Different Solvents on DI-SPME for Extraction of Lipids from T. castaneum
2.4.3. Comparison of Lipid Compositions between Two Insect Species in Acetonitrile
2.5. Data Processing and Analysis
3. Results and Discussion
3.1. Effect of Direct Immersion on SPME Extraction in Solvent
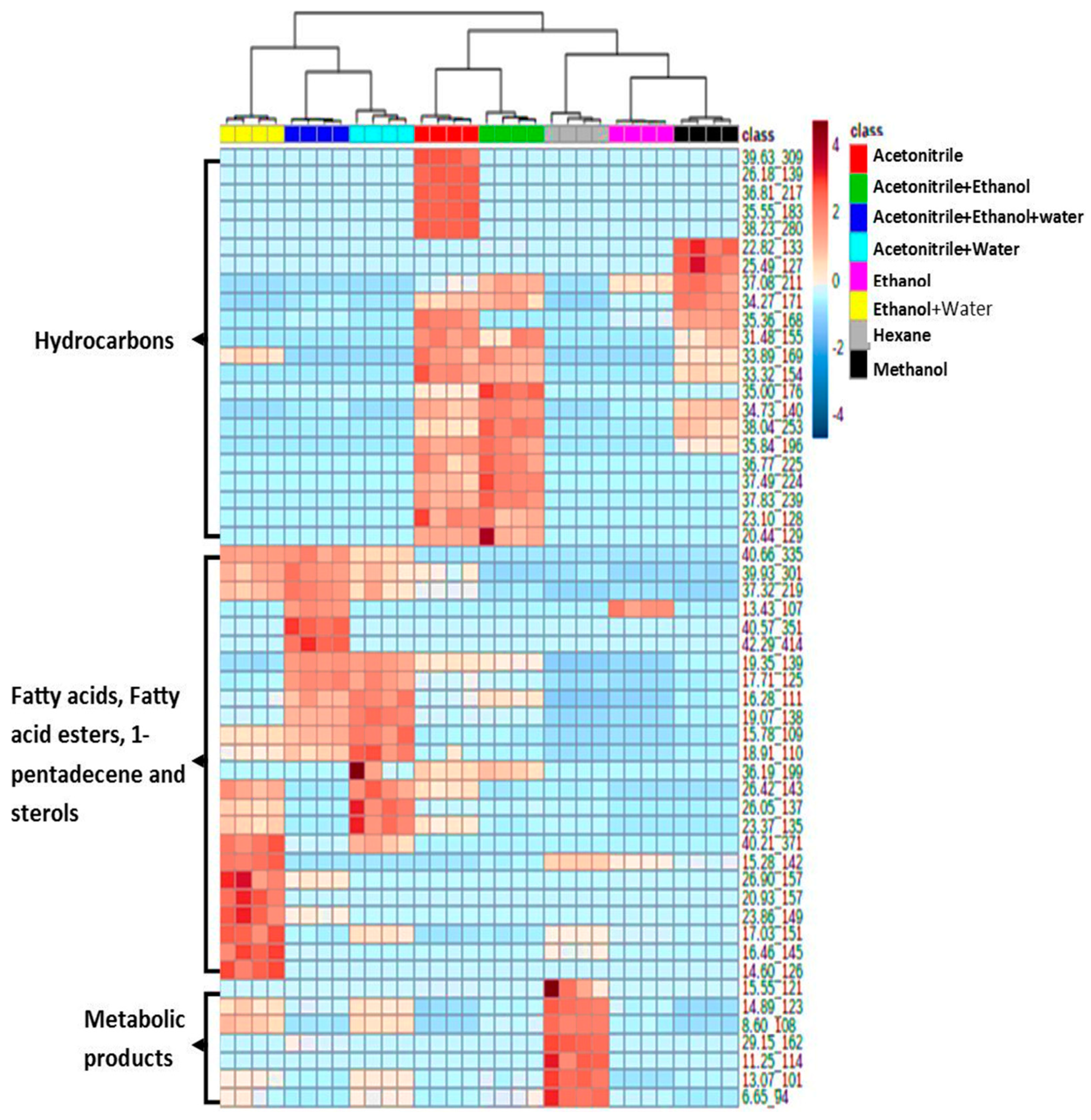
3.2. Effect of Extraction Solvents
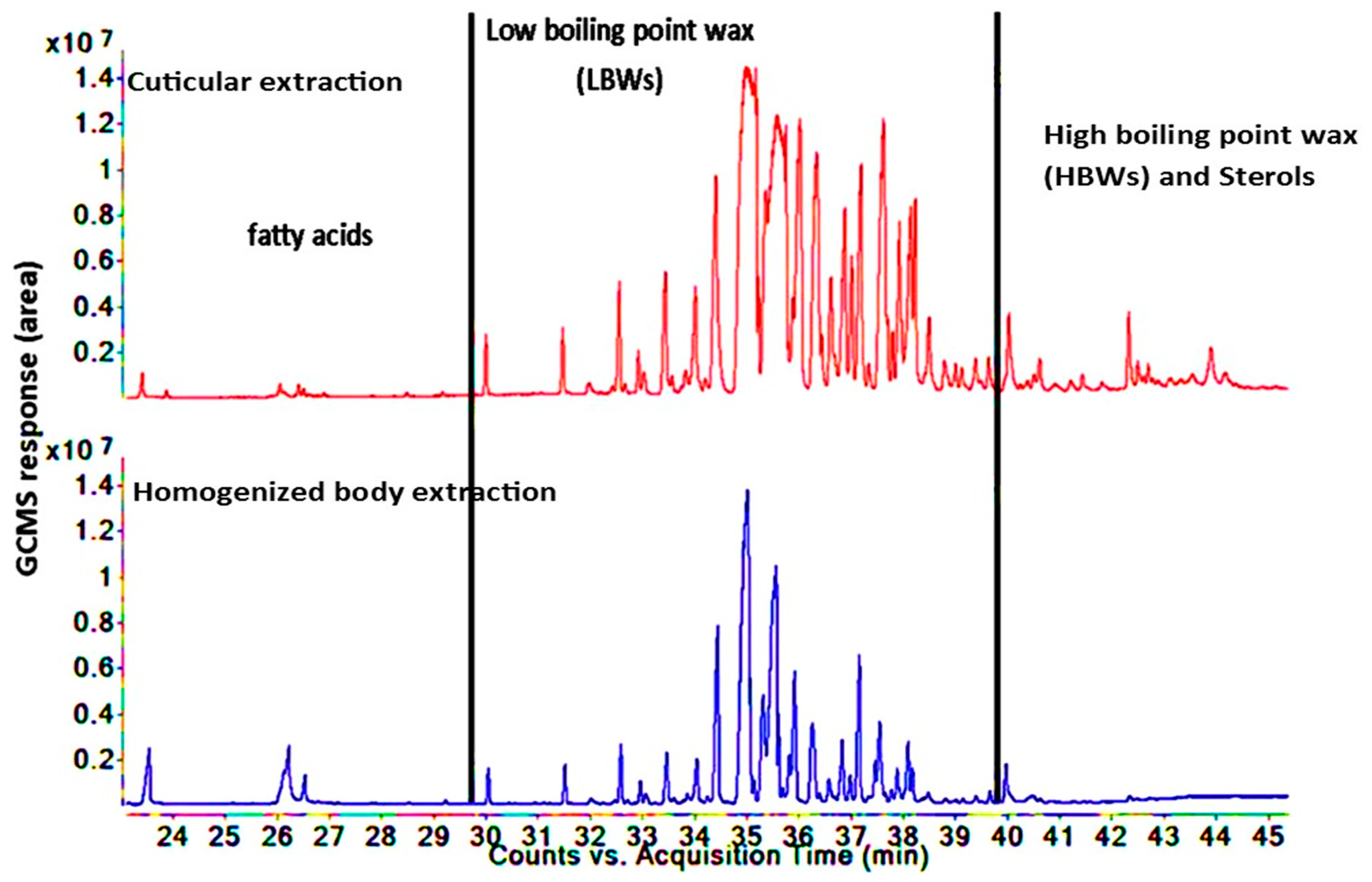
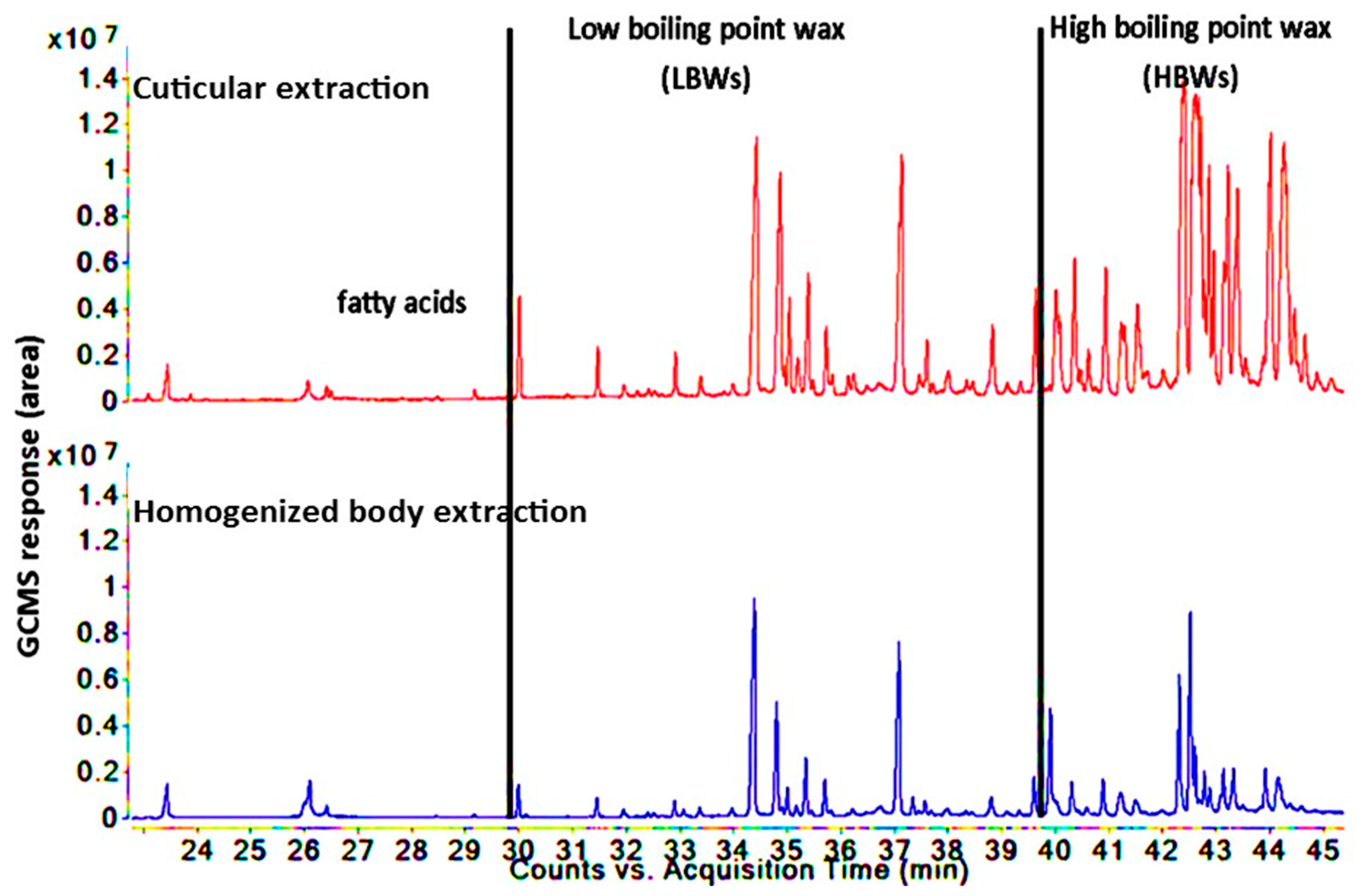
3.3. Comparison of Lipid Compositions between Two Insect Species
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cerkowniak, M.; Puckowski, A.; Stepnowski, P.; Gołębiowski, M. The use of chromatographic techniques for the separation and the identification of insect lipids. J. Chromatogr. B 2013, 937, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Cohen, E.; Moussian, B. Extracellular Composite Matrices in Arthropods; Springer: Cham, Switzerland, 2016; pp. 221–252. [Google Scholar]
- Desbois, A.P.; Smith, V.J. Antibacterial free fatty acids: Activities, mechanisms of action and biotechnological potential. Appl. Microbiol. Biotechnol. 2010, 85, 1629–1642. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, A.G. Water-proofing properties of cuticular lipids. Am. Zool. 1998, 38, 471–482. [Google Scholar] [CrossRef]
- Gołębiowski, M.; Maliński, E.; Nawrot, J.; Stepnowski, P. Identification and characterization of surface lipid components of the dried-bean beetle Acanthoscelides obtectus (Say) (Coleoptera: Bruchidae). J. Stored Prod. Res. 2008, 44, 386–388. [Google Scholar] [CrossRef]
- Gołębiowski, M.; Boguś, M.I.; Paszkiewicz, M.; Stepnowski, P. Cuticular lipids of insects as potential biofungicides: Methods of lipid composition analysis. Anal. Bioanal. Chem. 2011, 399, 3177–3191. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.R.; Charlet, L.D. Cuticular hydrocarbons of the sunflower beetle, Zygogramma exclamationis. Comp. Biochem. Physiol. B: Biochem. Mol. Biol. 2003, 135, 273–284. [Google Scholar]
- Snellings, Y.; Herrera, B.; Wildemann, B.; Beelen, M.; Zwarts, L.; Wenseleers, T.; Callaerts, P. The role of cuticular hydrocarbons in mate recognition in Drosophila suzukii. Scie. Rep. 2018, 8, 4996. [Google Scholar] [CrossRef]
- Berson, J.D.; Simmons, L.W. Female cuticular hydrocarbons can signal indirect fecundity benefits in an insect. Evolution 2019, 73, 982–989. [Google Scholar] [CrossRef]
- Ferveur, J.-F.; Cortot, J.; Rihani, K.; Cobb, M.; Everaerts, C. Desiccation resistance: effect of cuticular hydrocarbons and water content in Drosophila melanogaster adults. PeerJ 2018, 6, e4318. [Google Scholar] [CrossRef]
- Gołębiowski, M.; Maliński, E.; Boguś, M.I.; Kumirska, J.; Stepnowski, P. The cuticular fatty acids of Calliphora vicina, Dendrolimus pini and Galleria mellonella larvae and their role in resistance to fungal infection. Insect Biochem. Mol. Biol. 2008, 38, 619–627. [Google Scholar] [CrossRef]
- Toolson, E.C.; Kuper-Simbrón, R. Laboratory evolution of epicuticular hydrocarbon composition and cuticular permeability in Drosophila pseudoobscura: effects on sexual dimorphism and thermal-acclimation ability. Evolution 1989, 43, 468–473. [Google Scholar] [PubMed]
- Ye, G.; Li, K.; Zhu, J.; Zhu, G.; Hu, C. Cuticular hydrocarbon composition in pupal exuviae for taxonomic differentiation of six necrophagous flies. J. Med. Entomol. 2007, 44, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Buckner, J.S.; Hagen, M.M.; Nelson, D.R. The composition of the cuticular lipids from nymphs and exuviae of the silverleaf whitefly, Bemisia argentifolii. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1999, 124, 201–207. [Google Scholar] [CrossRef]
- Saïd, I.; Costagliola, G.; Leoncini, I.; Rivault, C. Cuticular hydrocarbon profiles and aggregation in four Periplaneta species (Insecta: Dictyoptera). J. Insect. Physiol. 2005, 51, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Buckner, J.S.; Pitts-Singer, T.L.; Guédot, C.; Hagen, M.M.; Fatland, C.L.; Kemp, W.P. Cuticular lipids of female solitary bees, Osmia lignaria Say and Megachile rotundata (F.)(Hymenoptera: Megachilidae). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2009, 153, 200–205. [Google Scholar] [CrossRef]
- Ginzel, M.D.; Moreira, J.A.; Ray, A.M.; Millar, J.G.; Hanks, L.M. (Z)-9-Nonacosene—major component of the contact sex pheromone of the beetle Megacyllene caryae. J. Chem. Ecol. 2006, 32, 435–451. [Google Scholar] [CrossRef]
- Roux, E.; Sreng, L.; Provost, E.; Roux, M.; Clement, J.L. Cuticular hydrocarbon profiles of dominant versus subordinate male Nauphoeta cinerea cockroaches. J. Chem. Ecol. 2002, 28, 1221–1235. [Google Scholar] [CrossRef]
- De Pasquale, C.; Guarino, S.; Peri, E.; Alonzo, G.; Colazza, S. Investigation of cuticular hydrocarbons from Bagrada hilaris genders by SPME/GC-MS. Anal. Bioanal. Chem. 2007, 389, 1259–1265. [Google Scholar] [CrossRef]
- Chong, J.; Soufan, O.; Li, C.; Caraus, I.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. MetaboAnalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef]
- Shrivastava, A.; Gupta, V.B. Methods for the determination of limit of detection and limit of quantitation of the analytical methods. Chron. Young sci. 2011, 2, 21–25. [Google Scholar] [CrossRef]
- Alnajim, I.; Agarwal, M.; Liu, T.; Ren, Y. A Novel Method for the Analysis of Volatile Organic Compounds (VOCs) from Red Flour Beetle Tribolium castaneum (H.) Using Headspace-SPME Technology. Curr. Anal. Chem. 2019, 15, 1–9. [Google Scholar] [CrossRef]
- Jeleń, H.H.; Obuchowska, M.; Zawirska-Wojtasiak, R.; Wasowicz, E. Headspace solid-phase microextraction use for the characterization of volatile compounds in vegetable oils of different sensory quality. J. Agric. Food. Chem. 2000, 48, 2360–2367. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, S.; Panigrahi, S. Solid-phase microextraction (SPME) techniques for quality characterization of food products: a review. Food Bioproc. Tech. 2011, 4, 1–26. [Google Scholar] [CrossRef]
- Qiu, R.; Qu, D.; Hardy, G.E.S.J.; Trengove, R.; Agarwal, M.; Ren, Y. Optimization of headspace solid-phase microextraction conditions for the identification of Phytophthora cinnamomi rands. Plant Dis. 2014, 98, 1088–1098. [Google Scholar] [CrossRef]
- Niu, Y.; Hardy, G.; Hua, L.; Trengove, R.; Agarwal, M.; Cheng, H.; Ren, Y. Optimization of HS-SPME-GC method for detection of stored grain insects. In Proceedings of the International Conference on Controlled Atmosphere and Fumigation in Stored Products, Antalya, Turkey, 15–19 October 2012; pp. 15–19. [Google Scholar]
- Lockey, K.H. Hydrocarbons of adult Tribolium castaneum Hbst. and Tribolium confusum Duv.(Coleoptera: Tenebrionidae). Comp. Biochem. Physiol. B Comp. Biochem. 1978, 61, 401–407. [Google Scholar] [CrossRef]
- Aldai, N.; Murray, B.E.; Nájera, A.I.; Troy, D.J.; Osoro, K. Derivatization of fatty acids and its application for conjugated linoleic acid studies in ruminant meat lipids. J. Sci. Food Agric. 2005, 85, 1073–1083. [Google Scholar] [CrossRef] [Green Version]
- Kataoka, H.; Lord, H.L.; Pawliszyn, J. Applications of solid-phase microextraction in food analysis. J. Chrmatogr. A 2000, 880, 35–62. [Google Scholar] [CrossRef]
- Brondz, I.; Ekeberg, D.; Høiland, K.; Bell, D.S.; Annino, A.R. The real nature of the indole alkaloids in Cortinarius infractus: Evaluation of artifact formation through solvent extraction method development. J. Chromatogr. A 2007, 1148, 1–7. [Google Scholar] [CrossRef]
- Caputo, B.; Dani, F.; Horne, G.; N’fale, S.; Diabate, A.; Turillazzi, S.; Coluzzi, M.; Costantini, C.; Priestman, A.; Petrarca, V. Comparative analysis of epicuticular lipid profiles of sympatric and allopatric field populations of Anopheles gambiae ss molecular forms and An. arabiensis from Burkina Faso (West Africa). Insect Biochem. Mol. Biol. 2007, 37, 389–398. [Google Scholar] [CrossRef]
- Buckner, J.S.; Mardaus, M.C.; Nelson, D.R. Cuticular lipid composition of Heliothis virescens and Helicoverpa zea pupae. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 1996, 114, 207–216. [Google Scholar] [CrossRef]
- Gołębiowski, M.; Boguś, M.I.; Paszkiewicz, M.; Stepnowski, P. The composition of the free fatty acids from Dendrolimus pini exuviae. J. Insect. Physiol. 2010, 56, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Gołębiowski, M.; Cerkowniak, M.; Dawgul, M.; Kamysz, W.; BOGUŚ, M.I.; Stepnowski, P. The antifungal activity of the cuticular and internal fatty acid methyl esters and alcohols in Calliphora vomitoria. Parasitology 2013, 140, 972–985. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.R.; Freeman, T.P.; Buckner, J.S. Waxes and lipids associated with the external waxy structures of nymphs and pupae of the giant whitefly, Aleurodicus dugesii. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2000, 125, 265–278. [Google Scholar] [CrossRef]
- Barbosa, R.R.; Braga, M.V.; Blomquist, G.J.; Queiroz, M.M.d.C. Cuticular hydrocarbon profiles as a chemotaxonomic tool for three blowfly species (Diptera: Calliphoridae) of forensic interest. J. Nat. Hist. 2017, 51, 1491–1498. [Google Scholar] [CrossRef]
- Braga, M.V.; Pinto, Z.T.; de Carvalho Queiroz, M.M.; Matsumoto, N.; Blomquist, G.J. Cuticular hydrocarbons as a tool for the identification of insect species: Puparial cases from Sarcophagidae. Acta Tropica 2013, 128, 479–485. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Leger, R.J.S. Developmental and transcriptional responses to host and non-host cuticles by the specific locust pathogen Metarhizium anisopliae var. acridum. Eukaryot. Cell. 2005, 4, 937–947. [Google Scholar] [CrossRef]
| No. | Solvent | Compound Numbers | The Rate of Total Expected Compounds (%) | Number of Compounds Only Detected in Solvent |
|---|---|---|---|---|
| 1 | Acetonitrile | 41 | 77.4 | 5 |
| 2 | Hexane | 23 | 43.4 | 1 |
| 3 | Ethanol | 25 | 47.2 | 0 |
| 4 | Methanol | 28 | 52.8 | 1 |
| 5 | Acetonitrile and ethanol (1:1 v/v) | 34 | 64.2 | 0 |
| 6 | Acetonitrile and water (1:1 v/v) | 22 | 41.5 | 0 |
| 7 | Ethanol and water (1:1 v/v) | 27 | 50.9 | 0 |
| 8 | Ethanol and acetonitrile and water (2:2:1 v/v/v) | 30 | 56.6 | 1 |
| Chemical Standards | Formula | RT (min) | R2 | LOD (µg/g) | |
|---|---|---|---|---|---|
| 1 | p-benzoquinone | C6H4O2 | 4.84 | 0.995 | 0.47 |
| 2 | p-benzoquinone, 2-methyl- | C7H6O2 | 6.72 | 0.976 | 0.36 |
| 3 | 1-pentadecene | C15H30 | 16.17 | 0.999 | 0.22 |
| 4 | Palmitic acid | C16H32O2 | 23.44 | 0.990 | 0.84 |
| 5 | Henicosane | C21H44 | 25.35 | 0.939 | 0.21 |
| 6 | Linolenic acid | C18H30O2 | 25.99 | 0.937 | 0.87 |
| 7 | Linoleic acid | C18H32O2 | 26.13 | 0.989 | 0.87 |
| 8 | Oleic acid | C18H34O2 | 26.43 | 0.993 | 0.89 |
| 9 | Docosane | C22H46 | 26.60 | 0.964 | 0.13 |
| 10 | Arachidic acid | C20H40O2 | 28.21 | 0.963 | 0.65 |
| 11 | Tricosane | C23H48 | 31.30 | 0.961 | 0.13 |
| 12 | Tetracosane | C24H50 | 32.24 | 0.920 | 0.21 |
| 13 | Pentacosane | C25H52 | 33.06 | 0.923 | 0.24 |
| 14 | Hexacosane | C26H54 | 34.41 | 0.961 | 0.14 |
| 15 | Heptacosane | C27H56 | 35.27 | 0.988 | 0.08 |
| 16 | Octacosane | C28H58 | 36.10 | 0.979 | 0.10 |
| 17 | Nonacosane | C29H60 | 37.40 | 0.987 | 0.08 |
| 18 | Triacontane | C30H62 | 39.52 | 0.988 | 0.08 |
| 19 | Hentriacontane | C31H64 | 41.48 | 0.982 | 0.09 |
| 20 | Cholesterol | C27H46O | 39.98 | 0.948 | 0.34 |
| 21 | Dotriacontane | C32H66 | 42.45 | 0.935 | 0.26 |
| Compounds | RT (min) | NIST RI | Calculated RI | Qualitative m/z | GC Response (105) ± SD, n = 4 | Relative GC Response | Distribution Coefficient B/(A + B) × 100 | ||
|---|---|---|---|---|---|---|---|---|---|
| Cuticular Extraction (a) | Homogenized Body Extraction (b) | Cuticular | Homogenized Body | ||||||
| 2-methylbenzoquinone | 6.73 | 1116 | 1117 | 122 | 14 ± 1 | 29 ± 39 | 0.13 | 0.49 | 66.64 |
| 2-ethyl-p-benzoquinone | 8.66 | 1215 | 1212 | 108 | 47 ± 8 | 46 ± 4 | 0.41 | 0.78 | 49.27 |
| 1,4-benzenediol, 2-methyl- | 13.55 | 1223 | 1234 | 124 | 16 ± 3 | 65 ± 2 | 0.14 | 1.1 | 80.52 |
| 1,2-benzenediol, 4-ethyl- | 15.07 | 1392 | 1388 | 138 | 30 ± 3 | 105 ± 2 | 0.26 | 1.78 | 77.65 |
| 7-dodecenol | 15.83 | 1465 | 1468 | 165 | 72 ± 12 | 84 ± 4 | 0.63 | 1.43 | 53.86 |
| 1-pentadecene | 16.26 | 1502 | 1504 | 154 | 1250 ± 172 | 1336 ± 4 | 10.98 | 22.70 | 51.66 |
| Benzene, 1-ethoxy-4-isothiocyanato- | 16.54 | 1527 | 1528 | 166 | 2 ± 0.3 | 2 ± 0.1 | 0.02 | 0.03 | 43.78 |
| 1-(2-hydroxy-4-methoxyphenyl)propan-1-one | 17.07 | 1538 * | 1558 | 151 | 47 ± 2 | 33 ± 3 | 0.40 | 0.56 | 41.42 |
| 7-hexadecene, (Z)- | 17.82 | 1620 | 1605 | 152 | 39 ± 1 | 28 ± 3 | 0.34 | 0.47 | 41.75 |
| 1,8,11-heptadecatriene, (Z,Z)- | 18.98 | 1655 | 1653 | 149 | ND | 11 ± 0.4 | ND | 0.19 | 100 |
| cis-7-tetradecen-1-ol | 19.14 | 1660 | 1661 | 179 | 515 ± 60 | 357 ± 31 | 4.52 | 6.06 | 40.9 |
| 2-hexadecanol | 19.50 | 1702 | 1705 | 182 | 453 ± 36 | 374 ± 17 | 3.98 | 6.37 | 45.23 |
| Myristic acid | 20.44 | 1752 | 1755 | 185 | ND | 0.82 ± 0.09 | ND | 0.01 | 100 |
| Palmitoleic acid | 23.10 | 1936 | 1938 | 192 | 4 ± 0.3 | ND | 0.04 | ND | 0 |
| Palmitic acid | 23.60 | 1954 | 1956 | 199 | 17 ± 2 | 143 ± 12 | 0.15 | 2.44 | 89.54 |
| Linolenic acid | 26.15 | 2115 | 2119 | 222 | 3 ± 0.2 | 72 ± 5 | 0.03 | 1.23 | 96.01 |
| Oleic acid | 26.25 | 2134 | 2125 | 220 | ND | 92 ± 4 | ND | 1.57 | 100 |
| Stearic acid | 26.57 | 2153 | 2157 | 227 | 5 ± 0.6 | 45 ± 5 | 0.05 | 0.77 | 89.68 |
| Unknown | 31.52 | - | 2505 | - | 12 ± 2 | 4 ± 0.4 | 0.10 | 0.08 | 26.71 |
| Pentacosane | 33.30 | 2500 | 2515 | 238 | 33 ± 3 | 14 ± 0.7 | 0.29 | 0.24 | 30.49 |
| Hexacosane | 33.85 | 2600 | 2612 | 266 | 174 ± 27 | 59 ± 3 | 1.53 | 1.00 | 25.3 |
| Unknown | 34.06 | - | 2618 | - | 27 ± 2 | 38 ± 4 | 0.23 | 0.65 | 58.52 |
| 2-methylhexacosane | 34.28 | 2661 | 2684 | 294 | 105 ± 10 | 5 ± 0.7 | 0.92 | 0.08 | 4.418 |
| 13-methylheptacosane | 34.41 | 2731 | 2741 | 296 | 289 ± 22 | 274 ± 19 | 2.53 | 4.66 | 48.72 |
| 11-methylheptacosane | 34.82 | 2734 | 2750 | 309 | 2358 ± 186 | 969 ± 85 | 20.71 | 16.5 | 29.11 |
| 2-methylheptacosane | 35.02 | 2762 | 2766 | 336 | 635 ± 37 | 218 ± 14 | 5.57 | 3.71 | 25.57 |
| 3-methylheptacosane | 35.58 | 2773 | 2771 | 337 | 1409 ± 147 | 449 ± 17 | 12.37 | 7.64 | 24.17 |
| Octacosane | 35.87 | 2800 | 2815 | 323 | 540 ± 35 | 118 ± 10 | 4.74 | 2.00 | 17.9 |
| 3-methyloctacosane | 36.28 | 2872 | 2849 | 351 | 531 ± 76 | 133 ± 10 | 4.66 | 2.27 | 20.05 |
| Nonacosane | 36.60 | 2900 | 2902 | 365 | 177 ± 29 | 30 ± 3 | 1.55 | 0.51 | 14.58 |
| Unknown | 36.85 | - | 2908 | - | 165 ± 18 | 41 ± 3 | 1.44 | 0.70 | 19.99 |
| Unknown | 37.08 | - | 2911 | - | 437 ± 15 | 393 ± 31 | 3.83 | 6.68 | 47.36 |
| Unknown | 37.49 | - | 2917 | - | 457 ± 40 | 23 ± 2 | 4.01 | 0.40 | 4.86 |
| 13-methylnonacosane | 37.58 | 2930 | 2927 | 379 | 766 ± 74 | 71 ± 7 | 6.73 | 1.21 | 8.52 |
| 11-methylnonacosane | 37.92 | 2939 | 2950 | 393 | 31 ± 2 | ND | 0.27 | ND | 0 |
| Nonacosane, 2-methyl- | 38.13 | 2962 | 2961 | 421 | 229 ± 16 | 79 ± 2 | 2.42 | 1.34 | 25.56 |
| 3-methylnonacosane | 38.44 | 2974 | 2973 | 395 | 275 ± 17 | 10 ± 1 | 2.41 | 0.16 | 3.351 |
| Triacontane | 39.64 | 3000 | 3003 | 239 | 141 ± 17 | 13 ± 1 | 1.24 | 0.22 | 8.425 |
| Cholesterol | 40.20 | 3087 | 3060 | 386 | 29 ± 1 | 75 ± 6 | 025 | 1.29 | 73.01 |
| Desmosterol | 40.51 | 3125 | 3133 | 364 | 15 ± 2 | 29 ± 1 | 0.13 | 0.49 | 66.06 |
| Dotriacontane | 42.37 | 3200 | 3203 | 449 | 36 ± 1 | 13 ± 1 | 0.32 | 0.22 | 26.27 |
| Compounds | RT (min) | NIST RI | Calculated RI | Qualitative M/Z | GC Response (105) ± SD, n = 4 | Relative GC Response | Distribution Coefficient B/(A + B) × 100 | ||
|---|---|---|---|---|---|---|---|---|---|
| Cuticular Extraction (A) | Homogenized Body Extraction (B) | Cuticular | Homogenized Body | ||||||
| Palmitic acid | 23.53 | 1954 | 1956 | 199 | 21 ± 3 | ND | 0.53 | ND | 0 |
| Linolenic acid | 26.06 | 2115 | 2119 | 222 | 6 ± 0.7 | 51 ± 5 | 0.17 | 4.39 | 88.72 |
| Stearic acid | 26.42 | 2153 | 2157 | 227 | 3 ± 0.3 | 13 ± 3 | 0.07 | 1.15 | 83.79 |
| Octadecanamide, N-(2-hydroxyethyl)- | 29.16 | 2347 | 2347 | 280 | 17 ± 0.7 | 9 ± 1 | 0.43 | 0.75 | 34.31 |
| Unknown | 31.46 | - | 2515 | - | 17 ± 3 | ND | 0.45 | ND | 0 |
| 11-methylpentacosane | 31.94 | 2535 | 2555 | 281 | 5 ± 0.8 | 3 ± 0.2 | 0.12 | 0.28 | 41.64 |
| Unknown | 32.91 | - | 2628 | - | 17 ± 2 | ND | 0.45 | ND | 0 |
| 13-methylheptacosane | 34.41 | 2731 | 2741 | 296 | 291 ± 56 | 209 ± 26 | 7.52 | 18.10 | 41.77 |
| 2-methylheptacosane | 34.98 | 2762 | 2766 | 336 | 166 ± 13 | 31 ± 6 | 4.28 | 2.66 | 15.64 |
| 3-methylheptacosane | 35.34 | 2773 | 2771 | 337 | 154 ± 28 | 7 ± 1 | 3.98 | 0.59 | 4.20 |
| Octacosane | 35.82 | 2800 | 2815 | 323 | 45 ± 4 | 33 ± 7 | 1.16 | 2.89 | 42.77 |
| Unknown | 37.10 | - | 2912 | - | 321 ± 25 | 15 ± 2 | 8.30 | 1.32 | 4.54 |
| 13-methylnonacosane | 37.58 | 2930 | 2927 | 379 | 33 ± 3 | 234 ± 21 | 0.84 | 20.3 | 87.79 |
| Triacontane | 39.64 | 3000 | 3003 | 239 | 156 ± 30 | 16 ± 2 | 4.04 | 1.44 | 9.59 |
| Cholesterol | 40.00 | 3087 | 3060 | 386 | 12 ± 1 | 48 ± 8 | 0.32 | 4.21 | 79.67 |
| Hentriacontane | 41.22 | 3100 | 3117 | 435 | 65 ± 3 | 23 ± 3 | 1.69 | 1.98 | 25.93 |
| 2-methylhentriacontane | 41.53 | 3162 | 3152 | 436 | 68 ± 5 | 28 ± 5 | 1.77 | 2.46 | 29.39 |
| 3-methylhentriacontane | 42.01 | 3172 | 3182 | 424 | 29 ± 6 | 14 ± 3 | 0.75 | 1.21 | 32.50 |
| Dotriacontane | 42.37 | 3200 | 3203 | 449 | 271 ± 51 | 6 ± 0.9 | 7.00 | 0.52 | 2.16 |
| 10-methyldotriacontane | 42.50 | 3235 | 3218 | 477 | 540 ± 57 | 64 ± 7 | 14.00 | 5.55 | 10.61 |
| 8-methyldotriacontane | 42.60 | 3240 | 3221 | 450 | 92 ± 6 | 118 ± 9 | 2.37 | 10.20 | 56.17 |
| Unknown | 42.78 | - | 3231 | - | 181 ± 12 | 17 ± 2 | 4.69 | 1.46 | 8.53 |
| Unknown | 42.87 | - | 3237 | - | 66 ± 12 | ND | 1.70 | ND | 0 |
| Unknown | 42.96 | - | 3249 | - | 243 ± 36 | 24 ± 4 | 6.29 | 2.08 | 8.98 |
| Dotriacontane, 2-methyl- | 43.22 | 3263 | 3266 | 481 | 260 ± 38 | 30 ± 5 | 6.29 | 2.64 | 10.47 |
| Unknown | 43.40 | - | 3276 | - | 240 ± 20 | 42 ± 5 | 6.22 | 3.66 | 14.95 |
| 15-methyltritriacontane | 44.03 | 3333 | 3323 | 463 | 384 ± 60 | 29 ± 5 | 9.93 | 2.55 | 7.11 |
| Unknown | 44.47 | - | 3351 | - | 58 ± 6 | 63 ± 12 | 1.50 | 5.47 | 52.17 |
| Tetratriacontane | 44.88 | 3400 | 3387 | 492 | 65 ± 12 | 11 ± 0.7 | 1.68 | 0.95 | 14.40 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alnajim, I.; Du, X.; Lee, B.; Agarwal, M.; Liu, T.; Ren, Y. New Method of Analysis of Lipids in Tribolium castaneum (Herbst) and Rhyzopertha dominica (Fabricius) Insects by Direct Immersion Solid-Phase Microextraction (DI-SPME) Coupled with GC–MS. Insects 2019, 10, 363. https://doi.org/10.3390/insects10100363
Alnajim I, Du X, Lee B, Agarwal M, Liu T, Ren Y. New Method of Analysis of Lipids in Tribolium castaneum (Herbst) and Rhyzopertha dominica (Fabricius) Insects by Direct Immersion Solid-Phase Microextraction (DI-SPME) Coupled with GC–MS. Insects. 2019; 10(10):363. https://doi.org/10.3390/insects10100363
Chicago/Turabian StyleAlnajim, Ihab, Xin Du, Byungho Lee, Manjree Agarwal, Tao Liu, and Yonglin Ren. 2019. "New Method of Analysis of Lipids in Tribolium castaneum (Herbst) and Rhyzopertha dominica (Fabricius) Insects by Direct Immersion Solid-Phase Microextraction (DI-SPME) Coupled with GC–MS" Insects 10, no. 10: 363. https://doi.org/10.3390/insects10100363
APA StyleAlnajim, I., Du, X., Lee, B., Agarwal, M., Liu, T., & Ren, Y. (2019). New Method of Analysis of Lipids in Tribolium castaneum (Herbst) and Rhyzopertha dominica (Fabricius) Insects by Direct Immersion Solid-Phase Microextraction (DI-SPME) Coupled with GC–MS. Insects, 10(10), 363. https://doi.org/10.3390/insects10100363








