Comparison of the Sex Pheromone Composition of Harmonia axyridis Originating from Native and Invaded Areas
Abstract
1. Introduction
2. Methods and Materials
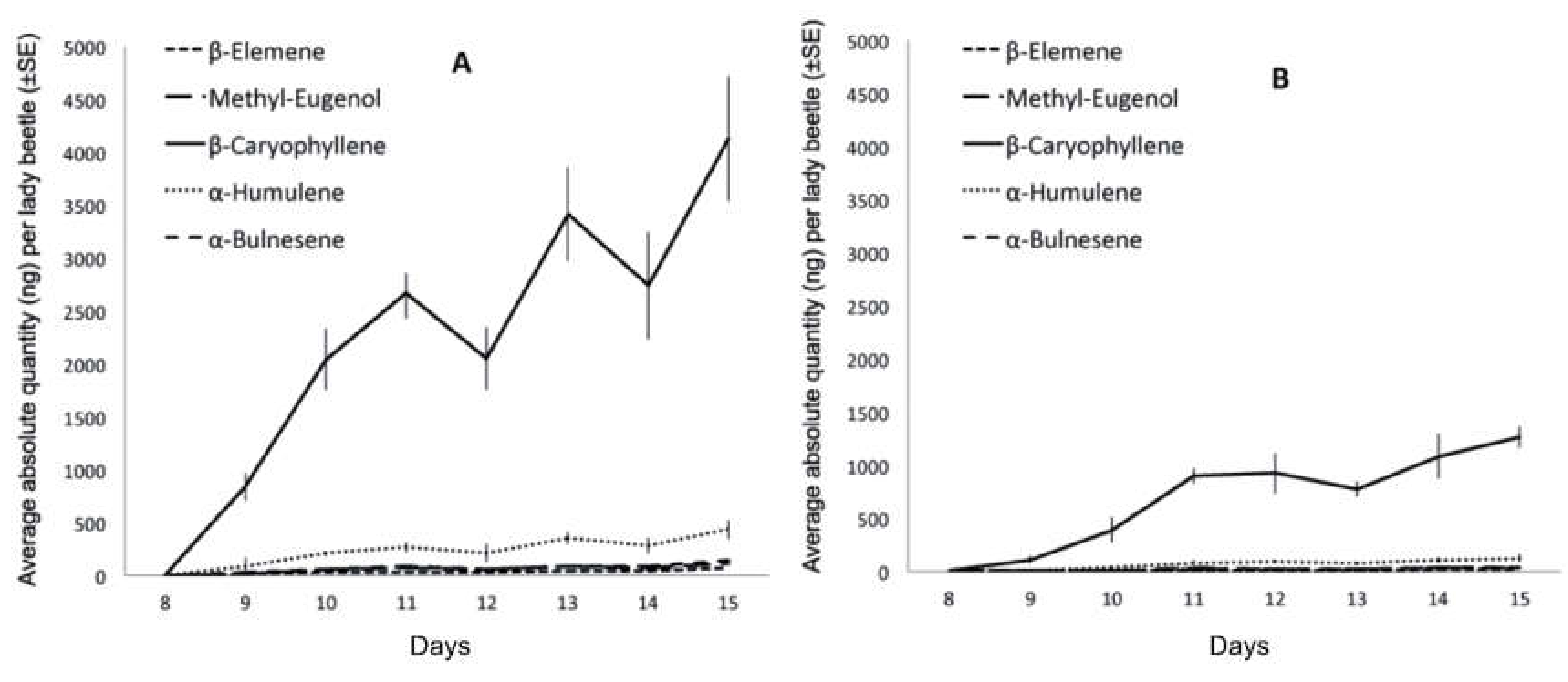
3. Results and Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Gordon, R.D. The Coleoptera (Coccinellidae) of America north of Mexico. J. N. Y. Entomol. Soc. 1985, 93, 1–912. [Google Scholar]
- Brown, P.M.J.; Thomas, C.E.; Lombaert, E.; Jeffries, D.L.; Estoup, A.; Lawson Handley, L.J. The Global Spread of Harmonia Axyridis (Coleoptera: Coccinellidae): Distribution, Dispersal and Routes of Invasion. Biocontrol 2011, 56, 623–641. [Google Scholar] [CrossRef]
- Lombaert, E.; Guillemaud, T.; Cornuet, J.M.; Malausa, T.; Facon, B.; Estoup, A. Bridgehead Effect in the Worldwide Invasion of the Biocontrol Harlequin Ladybird. PLoS ONE 2010, 5, e9743. [Google Scholar] [CrossRef] [PubMed]
- Koch, R.L. The multicolored Asian lady beetle, Harmonia axyridis: A review of its biology, uses in biological control, and non-target impacts. J. Insect Sci. 2003, 3, 32. [Google Scholar] [CrossRef] [PubMed]
- Durieux, D.; Fischer, C.; Brostaux, Y.; Sloggett, J.J.; Deneubourg, J.-L.; Vandereycken, A.; Joie, E.; Wathelet, J.-P.; Lognay, G.; Haubruge, E.; et al. Role of long-chain hydrocarbons in the aggregation behaviour of Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae). J. Insect Physiol. 2012, 58, 801–807. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ingels, B.; Aebi, A.; Hautier, L.; Van Leeuwen, T.; De Clercq, P. Molecular analysis of the gut contents of harmonia axyridis (coleoptera: Coccinellidae) as a method for detecting intra-guild predation by this species on aphidophagous predators other than coccinellids. Eur. J. Entomol. 2013, 110, 567–576. [Google Scholar] [CrossRef]
- Vandereycken, A.; Durieux, D.; Joie, E.; Sloggett, J.J.; Haubruge, E.; Verheggen, F.J. Is the multicolored asian ladybeetle, Harmonia axyridis, the most abundant natural enemy to aphids in agroecosystems? J. Insect Sci. 2013, 13, 158. [Google Scholar] [CrossRef] [PubMed]
- Verheggen, F.J.; Vogel, H.; Vilcinskas, A. Behavioral and immunological features promoting the invasive performance of the harlequin ladybird Harmonia axyridis. Front. Ecol. Evol. 2017. [Google Scholar] [CrossRef]
- Vandereycken, A.; Durieux, D.; Joie, E.; Haubruge, E.; Verheggen, F.J. Habitat diversity of the multicolored asian ladybeetle Harmonia axyridis pallas (Coleoptera: Coccinellidae) in agricultural and arboreal ecosystems: A review. Biotechnol. Agron. Soc. Environ. 2012, 16, 553–563. [Google Scholar]
- Facon, B.; Hufbauer, R.A.; Tayeh, A.; Loiseau, A.; Lombaert, E.; Vitalis, R.; Guillemaud, T.; Lundgren, J.G.; Estoup, A. Inbreeding Depression Is Purged in the Invasive Insect Harmonia axyridis. Curr. Biol. 2011, 21, 424–427. [Google Scholar] [CrossRef] [PubMed]
- Laugier, G.J.M.; Le Moguédec, G.; Tayeh, A.; Loiseau, A.; Osawa, N.; Estoup, A.; Facon, B. Increase in Male Reproductive Success and Female Reproductive Investment in Invasive Populations of the Harlequin Ladybird Harmonia Axyridis. PLoS ONE 2013, 8, e77083. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tayeh, A.; Estoup, A.; Lombaert, E.; Guillemaud, T.; Kirichenko, N.; Lawson-Handley, L.; De Clercq, P.; Facon, B. Cannibalism in Invasive, Native and Biocontrol Populations of the Harlequin Ladybird. BMC Evol. Biol. 2014, 14, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tayeh, A.; Hufbauer, R.A.; Estoup, A.; Ravigné, V.; Frachon, L.; Facon, B. Biological invasion and biological control select for different life histories. Nat. Commun. 2015, 6, 7268. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yang, X.; Zang, L.; Wang, Z.; Ruan, C.; Liu, X. Effect of geographic variation on biology and cold tolerance of Harmonia axyridis in China. Entomol. Gen. 2017, 36, 239–250. [Google Scholar] [CrossRef]
- Fassotte, B.; Fischer, C.; Durieux, D.; Lognay, G.; Haubruge, E.; Francis, F.; Verheggen, F.J. First Evidence of a Volatile Sex Pheromone in Lady Beetles. PLoS ONE 2014, 9, e115011. [Google Scholar] [CrossRef] [PubMed]
- Obata, S. The Influence of Aphids on the Behaviour of Adults of the Ladybird Beetle, Harmonia axyridis (Col.: Coccinellidae). Entomophaga 1997, 42, 103–106. [Google Scholar] [CrossRef]
- Fassotte, B.; Francis, F.; Verheggen, F.J. The Scent of Love: How Important Are Semiochemicals in the Sexual Behavior of Lady Beetles? J. Pest Sci. 2016, 89, 347–358. [Google Scholar] [CrossRef]
- Brown, A.E.; Riddick, E.W.; Aldrich, J.R.; Holmes, W.E. Identification of (-) B-caryophyllene as a gender specific terpene produced by the multicolored Asian lady beetle. J. Chem. Ecol. 2006, 32, 2489–2499. [Google Scholar] [CrossRef] [PubMed]
- Verheggen, F.J.; Fagel, Q.; Heuskin, S.; Lognay, G.; Francis, F.; Haubruge, E. Electrophysiological and behavioral responses of the multicolored asian lady beetle, Harmonia axyridis Pallas, to sesquiterpene semiochemicals. J. Chem. Ecol. 2007, 33, 2148–2155. [Google Scholar] [CrossRef] [PubMed]
- Vandermoten, S.; Mescher, M.C.; Francis, F.; Haubruge, E.; Verheggen, F.J. Aphid alarm pheromone: An overview of current knowledge on biosynthesis and functions. Insect Biochem. Mol. Biol. 2012, 42, 155–163. [Google Scholar] [CrossRef] [PubMed]

| Sex Pheromone Components | ANCOVA Table (Absolute Abundance) | ANCOVA Table (Relative Proportions) | ||||||
|---|---|---|---|---|---|---|---|---|
| Population | Population × Time | Population | Population × Time | |||||
| F | p | F | p | F | p | F | p | |
| β-Elemene | 32.630 | <0.001 | 3.610 | 0.067 | 13.22 | 0.001 | 0.400 | 0.534 |
| Methyl-eugenol | 30.460 | <0.001 | 4.260 | 0.048 | 12.60 | 0.001 | 20.99 | <0.001 |
| β-Caryophyllene | 28.040 | <0.001 | 2.360 | 0.135 | 0.300 | 0.588 | 1.740 | 0.198 |
| α-Humulene | 31.540 | <0.001 | 2.880 | 0.100 | 0.330 | 0.567 | 0.640 | 0.432 |
| α-Bulnesene | 28.390 | <0.001 | 3.000 | 0.094 | 4.810 | 0.036 | 2.100 | 0.158 |
| Total | 28.600 | <0.001 | 2.480 | 0.126 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Legrand, P.; Vanderplanck, M.; Verheggen, F.J. Comparison of the Sex Pheromone Composition of Harmonia axyridis Originating from Native and Invaded Areas. Insects 2019, 10, 326. https://doi.org/10.3390/insects10100326
Legrand P, Vanderplanck M, Verheggen FJ. Comparison of the Sex Pheromone Composition of Harmonia axyridis Originating from Native and Invaded Areas. Insects. 2019; 10(10):326. https://doi.org/10.3390/insects10100326
Chicago/Turabian StyleLegrand, Pauline, Maryse Vanderplanck, and Francois J. Verheggen. 2019. "Comparison of the Sex Pheromone Composition of Harmonia axyridis Originating from Native and Invaded Areas" Insects 10, no. 10: 326. https://doi.org/10.3390/insects10100326
APA StyleLegrand, P., Vanderplanck, M., & Verheggen, F. J. (2019). Comparison of the Sex Pheromone Composition of Harmonia axyridis Originating from Native and Invaded Areas. Insects, 10(10), 326. https://doi.org/10.3390/insects10100326







