Assessing Boundary Film Forming Behavior of Phosphonium Ionic Liquids as Engine Lubricant Additives
Abstract
:1. Introduction
2. Materials and Methods
2.1. Lubricants
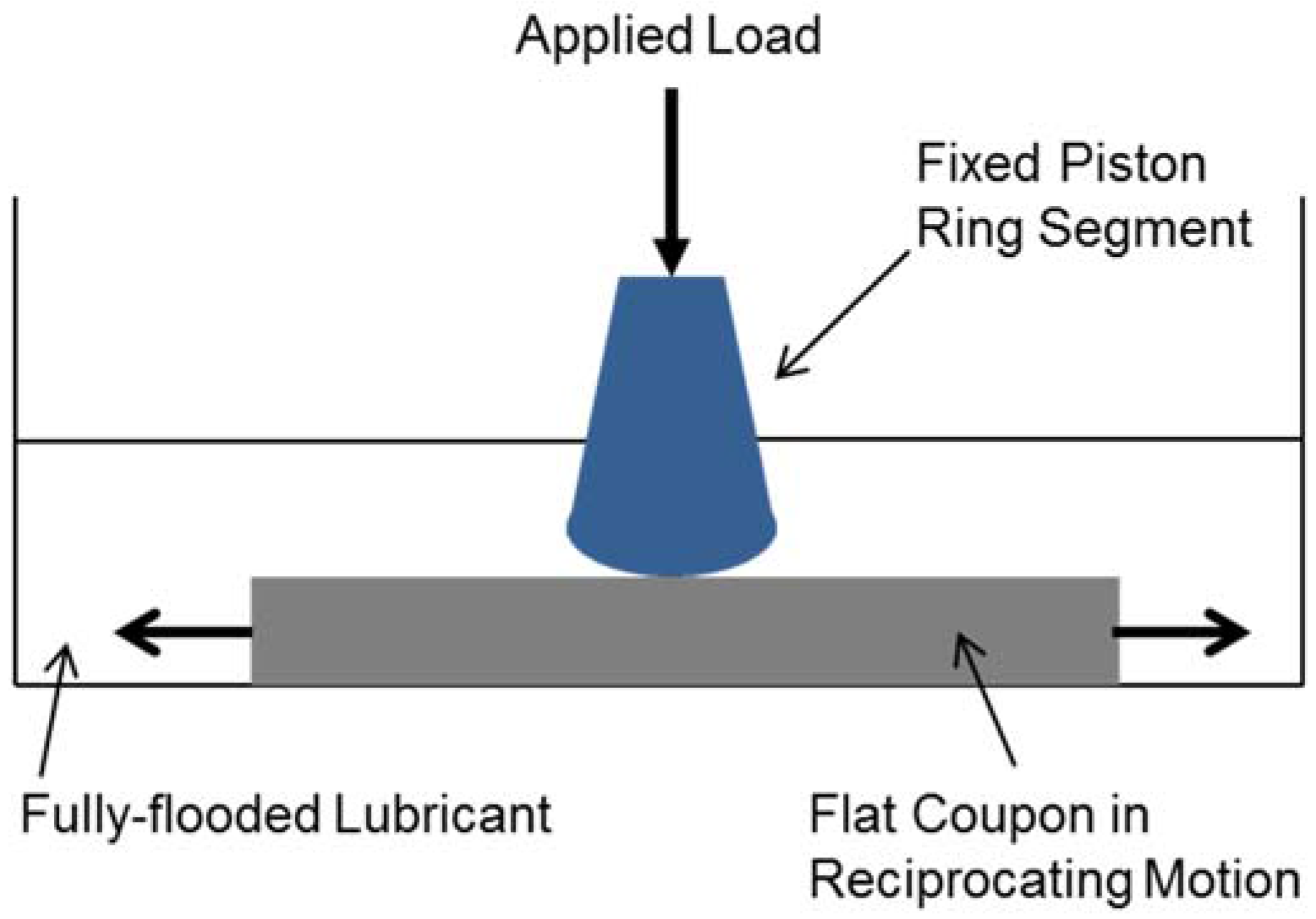
2.2. Test Set-Up and Experimental Conditions
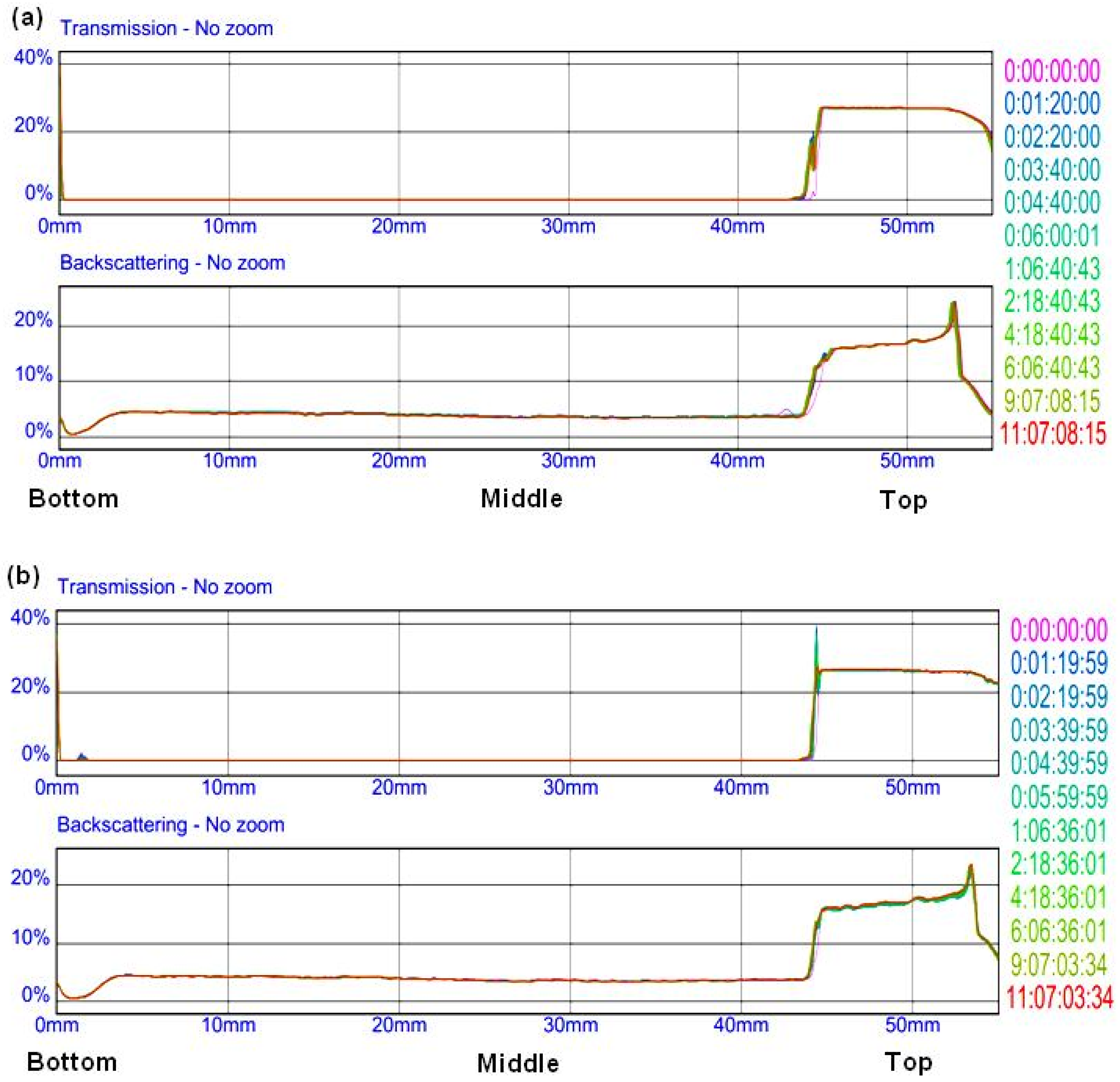
2.3. Surface Analysis
3. Results and Discussion
3.1. Oil-IL Mixture Stability Analysis
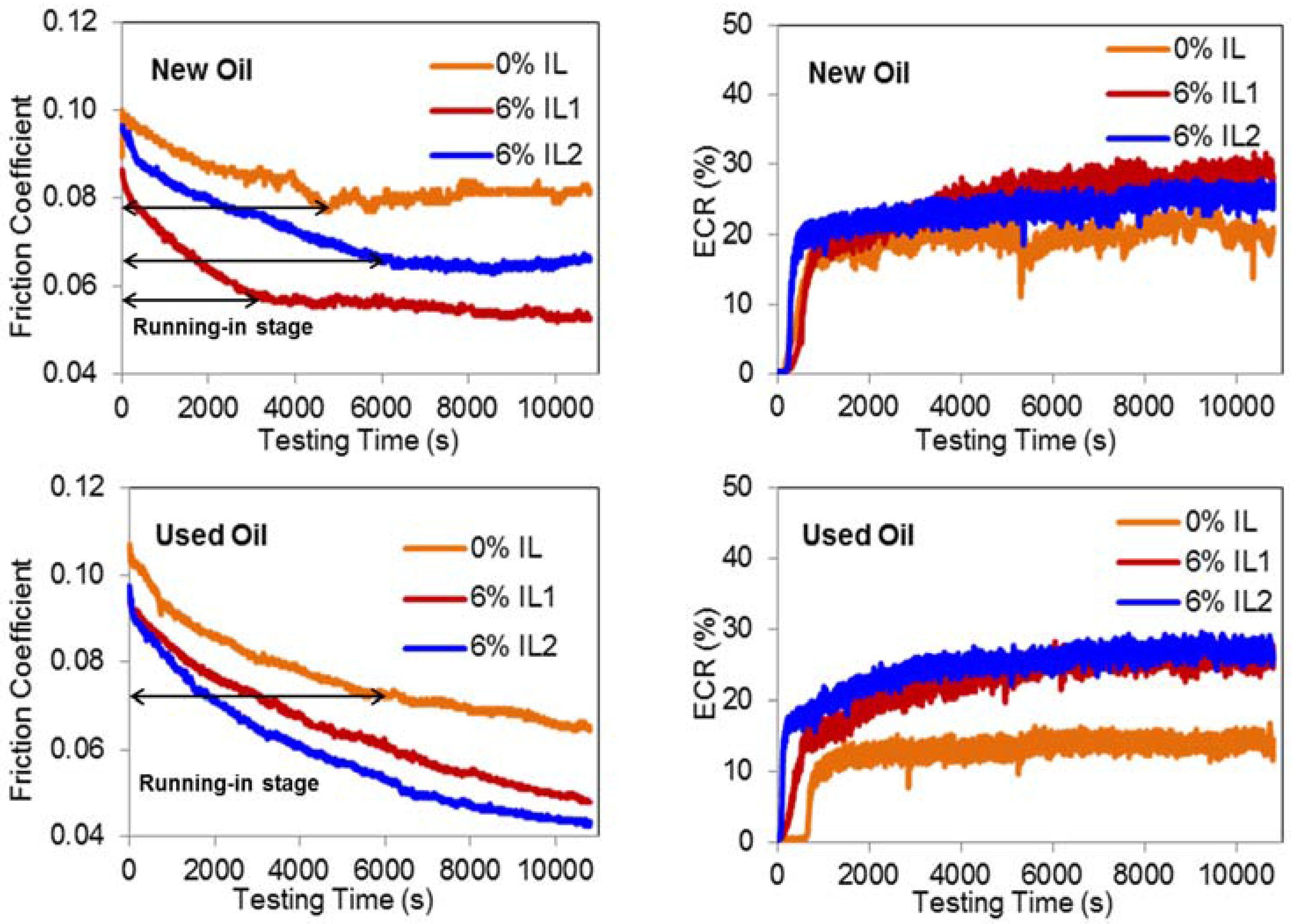
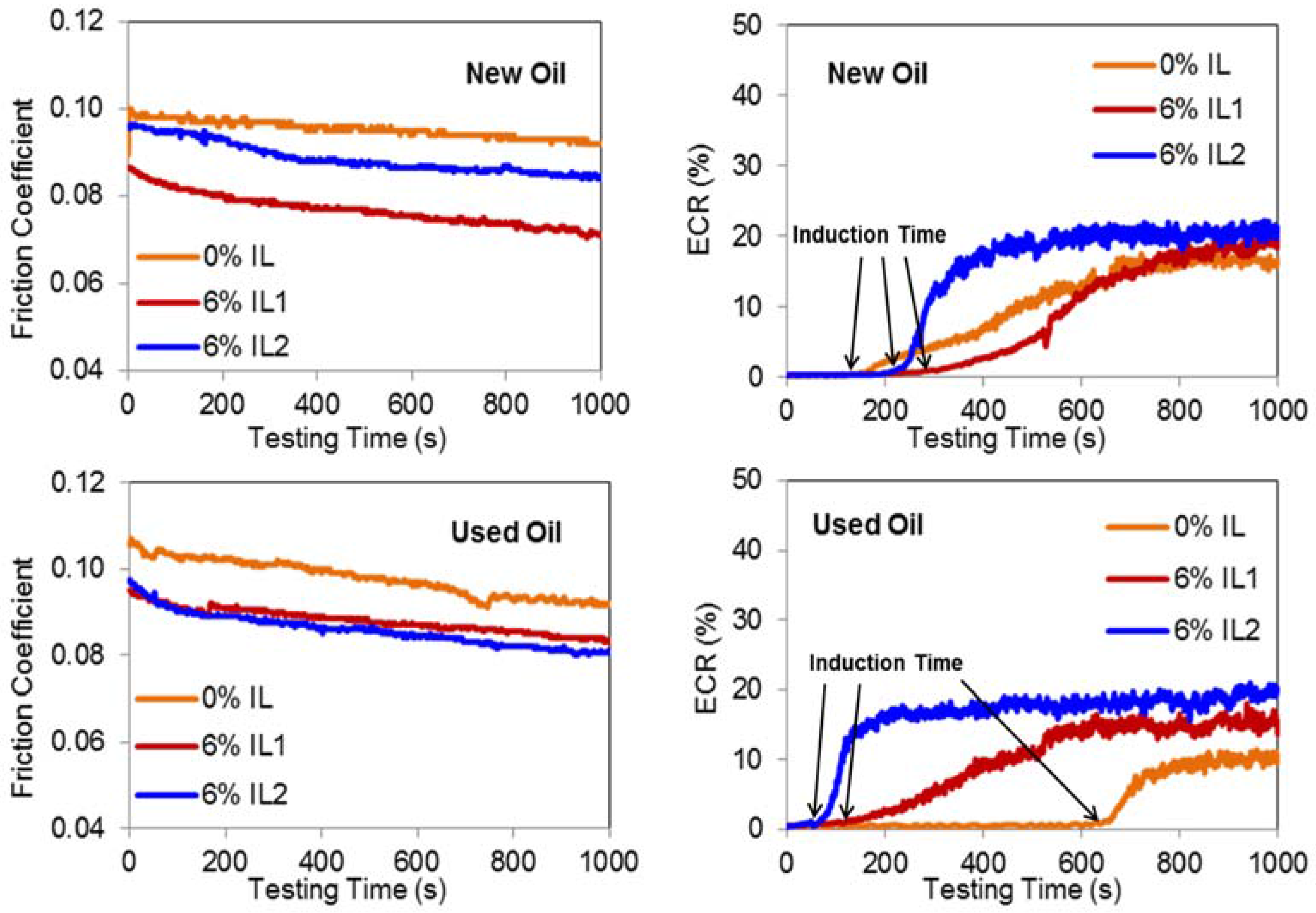
3.2. Friction and Film Forming (ECR) Behavior
3.3. Wear Behavior
3.4. Wear Mechanisms
3.5. Chemical Analysis of Surface Films
4. Conclusions
- The addition of phosphonium ILs to engine-aged lubricant results in a quicker initiation of the boundary film formation process, leading to a stable boundary film.
- Friction and wear performances of engine-aged lubricant outperformed fresh oil after the addition of both ILs.
- The formation of boundary film by ILs in engine-aged lubricant has reduced the effect of both plastic deformation and abrasive wear modes.
- Increase in concentration of phosphorus in boundary film formed by engine-aged lubricant and IL mixtures could suggest the involvement of phosphonium ILs in the boundary film formation process.
Author Contributions
Conflicts of Interest
References
- Tung, S.C.; McMillan, M.L. Automotive tribology overview of current advances and challenges for the future. Tribol. Int. 2004, 37, 517–536. [Google Scholar]
- Qu, J.; Blau, P.J.; Dai, S.; Luo, H.M.; Meyer, H.M. Ionic liquids as novel lubricants and additives for diesel engine applications. Tribol. Lett. 2009, 35, 181–189. [Google Scholar]
- Mordukhovich, G.; Qu, J.; Howe, J.Y.; Bair, S.; Yu, B.; Luo, H.; Smolenski, D.J.; Blau, P.J.; Bunting, B.G.; Dai, S. A low-viscosity ionic liquid demonstrating superior lubricating performance from mixed to boundary lubrication. Wear 2013, 301, 740–746. [Google Scholar] [CrossRef]
- Morris, N.; Rahmani, R.; Rahnejat, H.; King, P.; Fitzsimons, B. Tribology of piston compression ring conjunction under transient thermal mixed regime of lubrication. Tribol. Int. 2013, 59, 248–258. [Google Scholar] [CrossRef]
- Mishra, P.; Balakrishnan, S.; Rahnejat, H. Tribology of compression ring-to-cylinder contact at reversal. J. Eng. Tribol. 2008, 222, 815–826. [Google Scholar] [CrossRef]
- Anand, M.; Hadfield, M.; Viesca, J.L.; Thomas, B.; Hernández Battez, A.; Austen, S. Ionic liquids as tribological performance improving additive for in-service and used fully-formulated diesel engine lubricants. Wear 2015, 334–335, 67–74. [Google Scholar] [CrossRef]
- Qu, J.; Truhan, J.J.; Dai, S.; Luo, H.; Blau, P.J. Ionic liquids with ammonium cations as lubricants or additives. Tribol. Lett. 2006, 22, 207–214. [Google Scholar] [CrossRef]
- Mistry, K.; Fox, M.F.; Priest, M. Lubrication of an electroplated nickel matrix silicon carbide coated eutectic aluminium-silicon alloy automotive cylinder bore with an ionic liquid as a lubricant additive. J. En. Tribol. 2009, 223, 563–569. [Google Scholar] [CrossRef]
- Yu, B.; Bansal, D.G.; Qu, J.; Sun, X.; Luo, H.; Dai, S.; Blau, P.J.; Bunting, B.G.; Mordukhovich, G.; Smolenski, D.J. Oil-miscible and non-corrosive phosphonium-based ionic liquids as candidate lubricant additives. Wear 2012, 289, 58–64. [Google Scholar] [CrossRef]
- Qu, J.; Luo, H.; Chi, M.; Ma, C.; Blau, P.J.; Dai, S.; Viola, M.B. Comparison of an oil-miscible ionic liquid and ZDDP as a lubricant anti-wear additive. Tribol. Int. 2014, 71, 88–97. [Google Scholar] [CrossRef]
- Predel, T.; Pohrer, B.; Schlücker, E. Ionic Liquids as alternative lubricants for special applications. Chem. Eng. Technol. 2010, 33, 132–136. [Google Scholar] [CrossRef]
- Qu, J.; Blau, P.J.; Dai, S.; Luo, H.; Meyer, H.M., III; Truhan, J.J. Tribological characteristics of aluminum alloys sliding against steel lubricated by ammonium and imidazolium ionic liquids. Wear 2009, 267, 1226–1231. [Google Scholar] [CrossRef]
- Young, W.C.; Budynas, R.G. Bodies in contact undergoing direct bearing and shear stress. In Roark’s Formulas for Stress and Strain, 7th ed.; McGraw-Hill: New York, NY, USA, 2002; pp. 689–707. [Google Scholar]
- Furey, M. Metallic contact and friction between sliding surfaces. ASLE Trans. 1961, 4, 1–11. [Google Scholar] [CrossRef]
- Viesca, J.L.; Hernandez Battez, A.; González, R.; Reddyhoff, T.; Pérez, A.T.; Spikes, H.A. Assessing boundary film formation of lubricant additivised with 1-hexyl-3-methylimidazolium tetrafluoroborate using ECR as qualitative indicator. Wear 2010, 269, 112–117. [Google Scholar] [CrossRef]
- Cambiella, A.; Benito, J.; Pazos, C.; Coca, J.; Ratoi, M.; Spikes, H.A. The effect of emulsifier concentration on the lubricating properties of oil-in-water emulsions. Tribol. Lett. 2006, 22, 53–65. [Google Scholar] [CrossRef]
- Blanco, D.; Hernandez Battez, A.; Viesca, J.L.; González, R.; Fernández-González, A. Lubrication of CrN coating with ethyl-dimethyl-2-methoxyethylammonium tris (pentafluoroethyl) trifluorophosphate ionic liquid as additive to PAO 6. Tribol. Lett. 2011, 41, 295–302. [Google Scholar] [CrossRef]
- Blanco, D.; González, R.; Hernández Battez, A.; Viesca, J.L.; Fernández-González, A. Use of ethyl-dimethyl-2-methoxyethylammonium tris (pentafluoroethyl) trifluorophosphate as base oil additive in the lubrication of TiN PVD coating. Tribol. Int. 2011, 44, 645–650. [Google Scholar] [CrossRef]
- González, R.; Hernandez Battez, A.; Blanco, D.; Viesca, J.L.; Fernández-González, A. Lubrication of TiN, CrN and DLC PVD coatings with 1-butyl-1-methylpyrrolidinium tris (pentafluoroethyl) trifluorophosphate. Tribol. Lett. 2010, 40, 269–277. [Google Scholar] [CrossRef]
- Hernandez Battez, A.; González, R.; Viesca, J.L.; Fernández-González, A.; Hadfield, M. Lubrication of PVD coatings with ethyl-dimethyl-2-methoxyethylammonium tris (pentafluoroethyl) trifluorophosphate. Tribol. Int. 2013, 58, 71–78. [Google Scholar] [CrossRef]
- González, R.; Hernandez Battez, A.; Viesca, J.L.; Higuera-Garrido, A.; Fernández-González, A. Lubrication of DLC coatings with two tris(pentafluoroethyl)trifluorophosphate anion-based ionic liquids. Tribol. Trans. 2013, 56, 887–895. [Google Scholar] [CrossRef]
- Viesca, J.L.; García, A.; Hernandez Battez, A.; González, R.; Monge, R.; Fernández-González, A.; Hadfield, M. FAP-anion ionic liquids used in the lubrication of a steel-steel contact. Tribol. Lett. 2013, 52, 431–437. [Google Scholar] [CrossRef]
- Monge, R.; González, R.; Hernandez Battez, A.; Fernández-González, A.; Viesca, J.L.; García, A.; Hadfield, M. Ionic liquids as an additive in fully formulated wind turbine gearbox oils. Wear 2015, 328–329, 50–63. [Google Scholar] [CrossRef]
- Yamaguchi, E.; Ryason, P.; Yeh, S.; Hansen, T. Boundary film formation by ZnDTPs and detergents using ECR. Tribol. Trans. 1998, 41, 262–272. [Google Scholar] [CrossRef]
- Blau, P.J. Friction Science and Technology: From Concepts to Applications, 2nd ed.; CRC Press: Florida, FL, USA, 2012. [Google Scholar]
- Greenall, A.; Neville, A.; Morina, A.; Sutton, M. Investigation of the interactions between a novel, organic anti-wear additive, ZDDP and overbased calcium sulphonate. Tribol. Int. 2012, 46, 52–61. [Google Scholar] [CrossRef]
- Woydt, M.; Kelling, N. Testing the tribological properties of lubricants and materials for the system “piston ring/cylinder liner” outside of engines. Ind. Lubr. Tribol. 2003, 55, 213–222. [Google Scholar] [CrossRef]
- Radil, K.C. Test Method to Evaluate Cylinder Liner-Piston Ring Coatings for Advanced Heat Engines; Memorandum Report ARL-MR-362; National Aeronautics and Space Administration Cleveland OH Lewis Research Center: Ohio, OH, USA, 1997.
- Brainard, W.A.; Wheeler, D.R. An XPS study of the adherence of refractory carbide silicide and boride rf-sputtered wear-resistant coatings. J. Vac. Sci. Technol. 1978, 15, 1800–1805. [Google Scholar] [CrossRef]
- Mathieu, H.J.; Landolt, D. An investigation of thin oxide films thermally grown in situ on Fe24Cr and Fe24Cr11Mo by auger electron spectroscopy and X-ray photoelectron spectroscopy. Corros. Sci. 1986, 26, 547–559. [Google Scholar] [CrossRef]
- Tan, B.J.; Klabunde, K.J.; Sherwood, P.M. X-Ray photoelectron spectroscopy studies of solvated metal atom dispersed catalysts. Monometallic iron and bimetallic iron-cobalt particles on alumina. Chem. Mater. 1990, 2, 186–191. [Google Scholar] [CrossRef]
- Wagner, C.; Riggs, W.; Davis, L.; Moulder, J.; Muilenberg, G. Handbook of X-Ray Photoelectron Spectroscopy; Perkin-Elmer: Eden Prairie, MN, USA, 1979. [Google Scholar]
- Paparazzo, E. X-Ray photo-emission and Auger spectra of damage induced by Ar+-ion etching at SiO2 surfaces. J. Phys. D Appl. Phys. 1987, 20. [Google Scholar] [CrossRef]
- Seyama, H.; Soma, M. Fe 2p spectra of silicate minerals. J. Electron. Spectrosc. 1987, 42, 97–101. [Google Scholar] [CrossRef]


| Additive/Lubricant | Description | Purity (%) | Density (g/mL) | Viscosity (cSt) | |
|---|---|---|---|---|---|
| 23 °C | 40 °C | 100 °C | |||
| IL1 * | Trihexyltetradecyl phosphonium bis(2,4,4-tri-methylpentyl) phosphinate | >95 | 0.90 | 388.8 | 35.4 |
| IL2 * | Trihexyltetradecyl phosphonium bis(2-ethylhexyl) phosphate | >98 | 0.91 | 429.0 | 49.5 |
| New Oil ‡ | Fresh SAE 15W40 | - | 0.886 | 106.1 | 14.3 |
| Used Oil ‡ | Engine-aged SAE 15W40 | - | 0.884 | 91.5 | 12.7 |
| Test Parameter | Value | Unit |
|---|---|---|
| Contact Pressure | 285 | MPa |
| Applied Load | 50 | N |
| Sliding Frequency | 4.4 | Hz |
| Stroke Length | 5 | mm |
| Oil Temperature | 25 | °C |
| Test Duration | 3 | h |
| Test Sample | Fe (%) | C (%) | Si (%) | Mn (%) | P (%) | S (%) | Cr (%) | O (%) |
|---|---|---|---|---|---|---|---|---|
| Flat coupon 1 | Rem. | 3.0–3.3 | 2.4–2.6 | 0.7–1.0 | 0.4–1.1 | 0.1–1.1 | - | - |
| Piston ring (coating) 2 | - | 4.92 | - | - | - | - | 93.53 | 1.55 |
| Flat Coupon Surface Lubricated with | Wear Volume, V (mm3) | Specific Wear Rate, K (×10−7 mm3/Nm) | |
|---|---|---|---|
| Mean | Std. Dev. | ||
| New Oil | 0.0095 | 0.0009 | 3.98 |
| New Oil + 6% IL1 | 0.0085 | 0.0014 | 3.56 |
| New Oil + 6% IL2 | 0.0121 | 0.0014 | 5.07 |
| Used Oil | 0.0217 | 0.0079 | 9.11 |
| Used Oil + 6% IL1 | 0.0082 | 0.0001 | 3.45 |
| Used Oil + 6% IL2 | 0.0077 | 0.0007 | 3.22 |
| Flat Coupon Surface Lubricated with | Element Concentration (in wt. %) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| C | O | Si | S | Ca | Zn | P | Mn | Fe | Cr | |
| New Oil | 8.41 | 3.88 | 2.37 | 0.30 | 0.36 | 0.34 | 0.26 | 0.71 | 83.27 | 0.10 |
| New Oil + 6% IL1 | 19.44 | 5.17 | 2.01 | 0.11 | 0.34 | 0.31 | 0.22 | nd | 72.27 | 0.12 |
| New Oil + 6% IL2 | 18.49 | 6.24 | 1.53 | 0.05 | 0.42 | 0.29 | 0.27 | 0.50 | 72.08 | 0.13 |
| Used Oil | 13.16 | 5.70 | 2.09 | 0.04 | 0.06 | 0.49 | 0.17 | 0.58 | 77.69 | 0.04 |
| Used Oil + 6% IL1 | 10.22 | 4.30 | 2.28 | 0.12 | 0.19 | nd | 0.29 | 0.64 | 81.97 | nd |
| Used Oil + 6% IL2 | 7.52 | 5.30 | 2.21 | 0.09 | 0.18 | nd | 0.30 | 0.65 | 83.78 | nd |
| Flat Coupon Surface Lubricated with | Binding Energy (eV) | Assigned Chemical Compounds | References |
|---|---|---|---|
| New Oil | 708.8 | Fe3O4 | [29] |
| 710.6 | Fe2O3 | [30] | |
| New Oil + 6% IL1 | nd | - | - |
| New Oil + 6% IL2 | 708.4 | Fe3O4 | [31] |
| 710.2 | Fe3O4 | [31] | |
| Used Oil | 708.8 | Fe3O4 | [29] |
| 710.1 | Fe3O4 | [31] | |
| Used Oil + 6% IL1 | 710.0 | Fe3O4 | [31] |
| Used Oil + 6% IL2 | 709.7 | Fe3O4 | [31] |
| 711.3 | Fe2O3 | [32,33,34] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anand, M.; Hadfield, M.; Viesca, J.-L.; Thomas, B.; González, R.; Cantrill, R.; Hernández Battez, A. Assessing Boundary Film Forming Behavior of Phosphonium Ionic Liquids as Engine Lubricant Additives. Lubricants 2016, 4, 17. https://doi.org/10.3390/lubricants4020017
Anand M, Hadfield M, Viesca J-L, Thomas B, González R, Cantrill R, Hernández Battez A. Assessing Boundary Film Forming Behavior of Phosphonium Ionic Liquids as Engine Lubricant Additives. Lubricants. 2016; 4(2):17. https://doi.org/10.3390/lubricants4020017
Chicago/Turabian StyleAnand, Mayank, Mark Hadfield, Jose-Luis Viesca, Ben Thomas, Ruben González, Rob Cantrill, and Antolin Hernández Battez. 2016. "Assessing Boundary Film Forming Behavior of Phosphonium Ionic Liquids as Engine Lubricant Additives" Lubricants 4, no. 2: 17. https://doi.org/10.3390/lubricants4020017
APA StyleAnand, M., Hadfield, M., Viesca, J.-L., Thomas, B., González, R., Cantrill, R., & Hernández Battez, A. (2016). Assessing Boundary Film Forming Behavior of Phosphonium Ionic Liquids as Engine Lubricant Additives. Lubricants, 4(2), 17. https://doi.org/10.3390/lubricants4020017








