1. Introduction
The search for tribological materials with higher performance requires much experimental work to screen candidates. Furthermore, the overall process lacks clear theoretical guidance, and it is difficult to avoid a certain blindness. Tribologists must determine whether the tribological properties of a substance can be predicted in some way. The progress of all material sciences and technologies has developed from experiential to theoretical, from descriptive to rational, from qualitative to quantitative, and from macroscopic to microscopic studies. The research and development of tribology is no exception.
It has long been recognized that the properties of a substance depend on the molecular structure of the substance. In the 1960s, Hansch et al. expressed the chemical structure in a quantified form, established the correlation between the quantified structural index and the properties of compounds through regression, and thus established a prediction model. The quantitative structure ability relationship (QSAR) model was established and has been used until the present [
1]. Over the years, the QSAR indices (descriptors) representing the structure of compounds have been expanded to be directly computationally available. These indices represent more subtle structural changes. After determining a set of structural descriptors for a given activity, the task of QSAR research is to find the functional relationship between the descriptors and the activity. This function relation is obtained by learning existing data in a typical machine learning task. Common machine learning methods include multiple linear regression (MLR), principal component analysis (PCA), partial least squares (PLS) regression analysis, support vector machine (SVM), and artificial neural network (ANN) methods. At present, there are thousands of structural descriptors and features used in the QSAR method. The use of too many descriptors leads to training difficulties, overfitting, poor model interpretation, and other problems in the modeling process. Therefore, only a small number of descriptors are generally used in the QSAR model. The filtering principle is to remove noise, redundancy, and irrelevant descriptors without losing information. There are usually three selection methods, namely filter, wrapper, and embedded methods.
As tribologists, we must also consider whether there is a certain dependence law between the structure and tribological properties of materials. Edgar et al., introduced a specific QSAR method for traction fluid design in 2004 [
2]. In the same year, Tsubouchi et al., used the quantitative structure property relationship (QSPR) method (having the same basic principle as the QSAR method) to study the high-temperature traction coefficient and low-temperature fluidity of traction fluids [
3]. Minami et al., introduced the molecular design of lubricant additive technology for advanced lubricants in computer-aided design in 2007 [
4]. Koshima et al., used the QSPR method to study factors affecting the high-temperature detergency of alkylated phenyl salicylates in lubricants and studied the antishudder performance and torque capacity of low-molecular-weight alkenylsuccinimides as a transmission oil additive in 2008 [
5,
6]. Koshima et al., also investigated the correlation between the molecular structural characteristics of friction modifiers and the antifriction properties of traction fluids by means of the QSPR method in 2010 [
7]. These were the preliminary attempts to study the tribological properties of lubricants in relation to their structures, but such research accounts for a small and scattered proportion of studies on the properties of tribological materials, and a scientific and systematic method of investigating the relationship between the structures and tribological properties of compounds has not been established.
Gao’s team used the basic principles of QSAR theory to study the basic problems of the tribological quantitative structure activity relationship of lubricants and derived the new concept of the quantitative structure tribo-ability relationship (QSTR) [
8]. The QSTR method has been applied to study the correlation between the digital molecular structure and tribological properties of lubricants, build a model and predict the tribological properties of new compounds, and provide suggestions for finding materials with excellent tribological properties. The tribological properties of compounds are complex physical and chemical properties, and the tribological system is thus also complex. There are many tribological mechanisms and many factors affect the friction process, but the structures of compounds are undoubtedly a decisive factor. Of course, a model is not an accurate description, and some speculations may not be consistent with the specific mechanisms of traditional tribology. However, a model reflects the statistical law hidden behind the uncertain phenomena and provides a means for systematic estimation and extrapolation, and it is thus important for the prediction of the tribological properties of compounds.
In the study of the QSTR, we are faced with another problem in that regression method (a statistical method) is adopted to establish the prediction model. At present, MLR, PLS, and ANN methods are adopted in QSTR modeling. Research on the quantitative relationship between the structure of lubricants and tribological properties is still in its infancy.
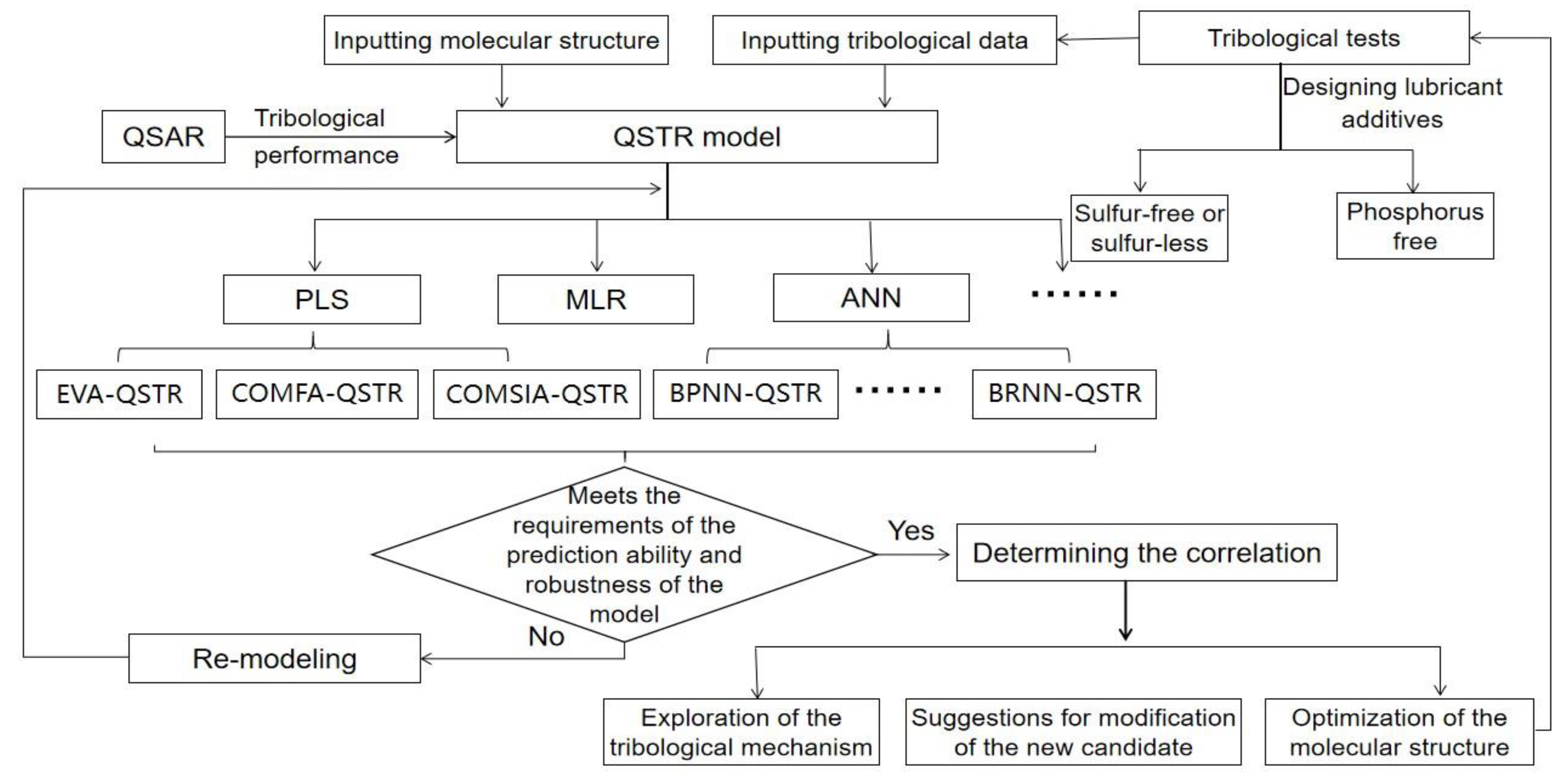
Figure 1 illustrates the basic working idea, which is to conduct the computer-aided design of the molecular structure of the lubricant and predict the tribological properties of the molecules.
2. Establishment of QSTR Model
QSAR method was introduced into the research of tribology simulation. First, some computational chemistry methods are applied to determine the various properties of molecules, which are also called descriptors in subsequent studies. Then, a small number of descriptors are screened, and the functional relationship between tribological properties and descriptors was obtained by machine learning method, so as to obtain the QSTR model. Finally, the tribological properties of untested molecules can be predicted by the QSTR model, and the accuracy of the model is verified by experiments.
At present, in the process of QSTR modeling, molecular structural descriptors are screened by filter and wrapper methods. The filter method sorts the predictive ability of descriptors and selects the descriptors on this basis. The wrapper method uses a genetic function approximation (GFA) to filter descriptors.
2.1. Filter Method
Molecular structural descriptors are screened by filter method for the following models, and the machine learning methods used by these models include multiple linear regression (MLR), partial least square (PLS), and artificial neural network (ANN).
2.1.1. MLR
MLR is the most commonly used statistical method, which uses the linear least square method to establish the linear relationship between independent variable and dependent variable.
Organic Molecule as a Lubricant Additive
Gao’s team calculated the quantum chemistry structural parameters of nitrogen-containing heterocyclic compounds using the Hartree-Fock ab initio quantum chemistry method and established a QSTR model using these compounds as lubricant additives [
9,
10]. They found that both friction-reducing and antiwear properties were related to the dipole moment and total energy of the compound, which provides a way to find lubricant additives with low friction coefficients and high antiwear performance.
Organic Molecule as an Extreme Pressure Additive
In further developing and expanding the research on the QSTR model in the field of tribology, Gao’s team used the QSTR model to study the correlation between the extreme pressure performance of a lubricant and the molecular structures of additives. They used a Hartree-Fock ab initio quantum chemistry method to calculate the quantum chemistry structural parameters of xanthate heterocyclic compounds and established a QSTR model for the antiwear and extreme pressure properties of these compounds as lubricant additives [
11]. Their results showed that when xanthate heterocyclic compounds were used as lubricant additives, entropy was the main quantum-chemistry structural parameter affecting the antiwear property, and the total molecular energy affected the extreme pressure performance.
Ionic Liquid as a Lubricant Additive
Gao’s team expanded the research on the QSTR from organic molecules to ionic liquids, calculated the quantum chemistry structural parameters of ionic liquids using the Hartree-Fock ab initio quantum chemistry method, and studied the QSTR model of ionic liquids as lubrication additives for antiwear performance [
12,
13]. Their results showed that the entropy of the ionic liquid in the system was the main quantum chemistry parameter that affected the antiwear property of the system. A decrease in entropy and an increase in the regularity of the system were conducive to improving the antiwear performance of the lubrication system. This conclusion holds for a system having polyethylene glycol 400 (PEG-400) or dimethyl sulfoxide (DMSO) as the base oil. Gao’s team used density functional theory (DFT) to calculate the structural parameters of the ionic liquid and studied the QSTR model for ionic liquids used as lubricant additives [
14]. Their results showed that for ionic liquids, hydrogen bonding affected the friction-reducing and antiwear properties.
2.1.2. PLS Regression Analysis
PLS regression analysis adopts component extraction to establish a regression model. In the process of component extraction, information in the independent variable data and dependent variable data are considered to maximize the correlation between the information (components) extracted from the independent variable data and dependent variable data, and the extracted components are then used for regression modeling [
15].
EVA
The molecular structural characteristics of lubricant additives were characterized through the evaluation of a novel infrared range vibration-based (EVA) descriptor [
16,
17,
18]. A 3D-QSTR model was established to predict tribological properties according to the three-dimensional structure and conformation of compounds by Gao’s team [
19].
Taking sample data of nitrogen-containing heterocyclic compounds, the friction-reducing and antiwear abilities of a system under the three loading conditions (196N, 294N and 392N) were studied. In the process of establishing the model, it was found that the correlation between the wear scale and EVA index was obviously different under the different loads. This result suggests that we must pay attention to the load in the study of the antiwear mechanism.
The results of the model reflected many interesting structural suggestions for friction-reducing and antiwear compounds. (1) The selection of the optimal carbon chain length was necessary to improve the tribological properties of additives. However, to accurately predict the optimal carbon chain length, in addition to the EVA index, topological parameters describing the geometric properties of molecules may also be used. (2) The aromatic ring and olefin systems can be regarded as rigid systems, and the change in heteroatoms on aromatic rings and the introduction of double bonds thus make little contribution to the improvement of tribological properties. (3) The elements S and P effectively improve the tribological properties of lubricant additives in specific structures.
EVA can be regarded as the simulation of the molecular infrared spectrum, but the real infrared spectrum obviously contains more information, such as the effect of the instrument, the error of the grating, mechanical noise, frequency doubling and other effects. However, we can still distinguish the structural information from the real infrared spectrum. We therefore believe that the EVA model can be used as a useful theoretical reference for the online detection of molecular structural changes of lubricants during motion. There are, of course, also limitations in using the EVA model to study the tribological properties of compounds.
CoMFA and CoMSIA
Comparative molecular field analysis (CoMFA) and comparative molecular similarity index analysis (CoMSIA) are currently the most mature and widely used structure activity relationship analysis methods in pharmacy. The basic assumption of these two methods is that the change in biological activity at the molecular level is closely related to the change in the molecular force field [
20,
21,
22]. CoMFA’s description of molecules includes the analysis of steric and electrostatic fields, whereas CoMSIA includes the analysis of steric fields, electrostatic fields, hydrophobicity, hydrogen-bond donors, and hydrogen-bond acceptors. Small molecules and biomacromolecules are often targeted in the treatment of human diseases. A prerequisite for the realization of drug activity is the high affinity of small drug molecules to binding sites. The binding sites of macromolecules can “feel” the molecular field of small molecules. The CoMFA and CoMSIA models explain the interactions between drug molecules and receptors, which are related to changes in the molecular field around the compound molecule. On this basis, in tribology, Gao’s team hypothesized that the interaction between the lubricant and the friction pair is related to the molecular field brought about by the lubricant. The friction pair will “feel” the appropriate molecular field brought by the lubricant to realize the expected tribological properties [
23,
24,
25,
26,
27,
28,
29].
The CoMFA-QSTR and CoMSIA-QSTR models visually explain the quantitative structure tribo-ability relationship of the tribological performance of compounds, and the three-dimensional (3D) contour maps of the models directly suggest how to improve the tribological properties of the molecular structure modification. However, the CoMFA and CoMSIA models mainly consider the interaction of the external molecular force field and do not accurately distinguish changes in the internal details of the molecular structure. Therefore, these models cannot accurately describe the internal mechanism of the tribological interaction between a lubricant and friction pairs. The models still provide a new means for the screening, optimization, and design of the molecular structure of a lubricant.
(A) Lubricant additive
- (a)
Nitrogen-containing heterocyclic compound
CoMFA and CoMSIA-QSTR models were used to study the correlation between molecular structural characteristics and antiwear properties of nitrogen-containing heterocyclic lubricant additives. It was found that the molecular electrostatic field had the greatest effect on the antiwear properties of nitrogen-containing heterocyclic lubricant additives [
23]. Additionally, although we have always believed that the presence of heteroatoms in organic molecules, especially atoms of active elements such as S and N, helps improve the antiwear property of a substance, the model suggests that if these atoms that improve the tribological performance appear in inappropriate areas, they will not help to improve the antiwear performance. The analysis results of the model are consistent with the test results.
- (b)
Hydrazide and phosphoric acid
CoMFA and CoMSIA-QSTR modeling studies were conducted on the antiwear properties of hydrazide and phosphoric acid lubricant additives [
24]. The results showed that the molecular electrostatic field had the greatest effect on the antiwear properties of these compounds.
(B) Lubricant base oil
Gao’s team studied the CoMFA and CoMSIA-QSTR models for the antiwear properties of esters as potential lubricant base oils, and provided useful information on structural modifications [
25]. These models can also be used to predict the antiwear properties of potential compounds.
The team also modeled compounds with different parent cores as research samples to study the application range of the models [
26]. CoMFA and CoMSIA-QSTR models were established for antiwear performance, and it was found that the models have good statistical performance in terms of their accuracy and predictive ability. The results show the universality and applicability of the CoMFA and CoMSIA-QSTR models in tribology.
2.1.3. ANN
ANN consists of a large number of simple interconnected neurons that process complex network systems and nonlinear transformations in parallel by simulating the way the human brain processes information. Among many algorithms, back propagation neural network (BPNN) is the most widely used one.
Back Propagation Neural Network (BPNN)
The BPNN is a feedforward neural network. The network is trained through an error backpropagation algorithm and can handle the simulation of nonlinear processes.
Gao’s team chose nitrogen-containing heterocyclic compounds as samples of lubricant additives and established models of antiwear performance under the three loads (196N, 294N, 392N) [
8]. They analyzed the sensitivity of each parameter variable to the antiwear property and speculated and analyzed the antiwear mechanism. They found that the antiwear performance was particularly sensitive to the load. The modeling suggested that the antiwear process involving additive molecules can be divided into four steps, namely the diffusion, adsorption, reaction, and conformational change of additive molecules.
Gao’s team [
30] used the BPNN to study the quantitative structure tribo-ability relationship of the antiwear properties of 2-alkyl xanthate lubricant additives and found that the N and S heteroatoms in the molecules affected the antiwear properties. Additionally, the molecular length, the double-bond of the S atom, the number of aromatic rings and the degree of molecular branching were factors affecting the antiwear performance.
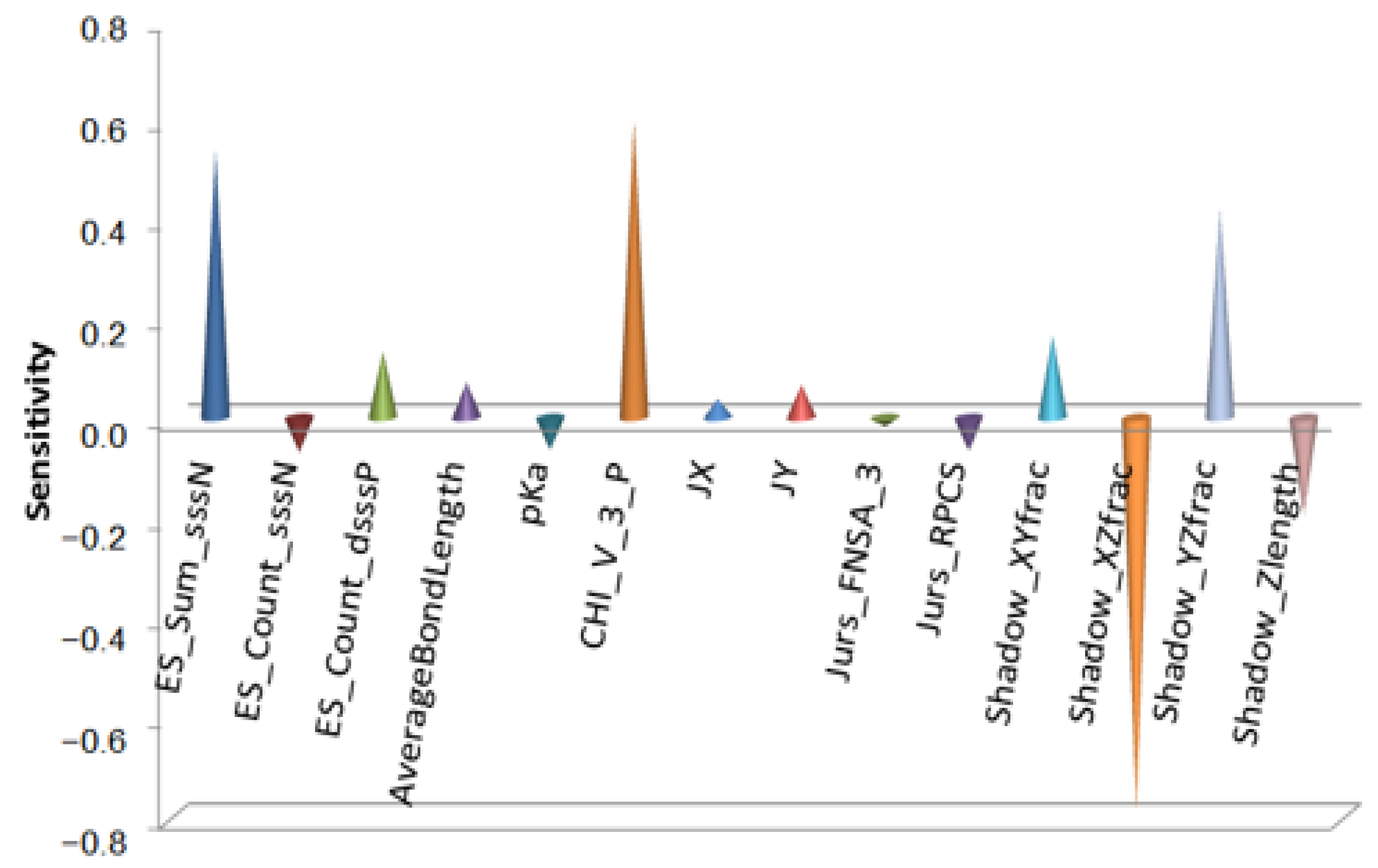
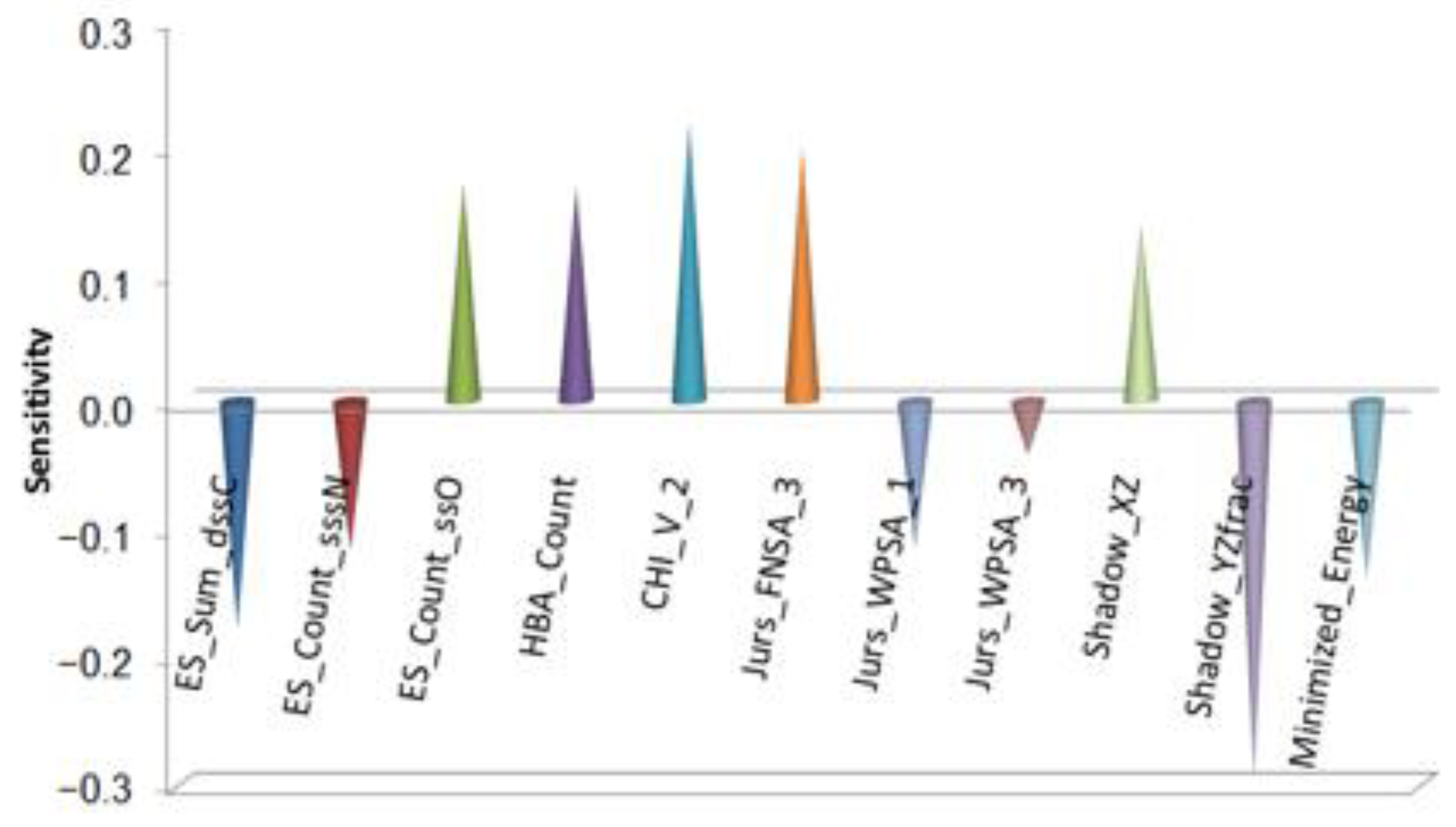
Taking triazine derivatives as samples, Gao’s team conducted tribological quantitative structure activity relationship studies on the extreme pressure performance (
Figure 2) and antiwear performance (
Figure 3) of lubricant additives [
31].
The extreme pressure and antiwear performances are two different tribological properties. In obtaining an additive with good extreme pressure performance and good antiwear performance, there are strict requirements for the structure of the additive. The molecules are limited to being moderate in size in all three dimensions. The composition of substituent groups is important and is related not only to the number of active elements such as N and P atoms but also to the bonding mode and electronic structure of the atoms.
A BPNN-QSTR antiwear model was established for ester lubricant base oil. Additionally, suggestions on the molecular structure to achieve good antiwear performance were put forward; e.g., there should not be too many polar groups in the molecule, the polar group charge should be relatively concentrated, and the molecule should have low hydrophobicity [
32]. Furthermore, a BPNN-QSTR friction-reducing model was established for organic compounds [
33]. It was found that connectivity indices (CHI) contribute a great deal to friction, which means that friction properties are strongly correlated with the molecular structure.
Bayesian Regularization Neural Network (BRNN)
The BPNN is a neural network commonly used for structure activity prediction, but it has disadvantages, such as it being unable to guarantee global optimization and overfitting. The BRNN is a robust model based on Bayesian regularization and has good data fitting and generalization ability.
A BRNN-QSTR antiwear model of nitrogen-containing heterocyclic compounds was established by Gao’s team [
34]. It was found that the prediction and fitting performance of the model using two-dimensional (2D) descriptors as variables was good, which may be due to the specific conformation of molecules. In a study of the 2D QSTR model, it was found that the degree of solvation of additive molecules strongly affected the antiwear performance. It is well-known that during lubrication, additive molecules are diffused in the lubricant and adsorbed on metal surfaces. A study of the sensitivity of quantum descriptors of the model found that the energy of the lowest unoccupied molecular orbital (LUMO) strongly affected the antiwear property, with a lower energy resulting in a better molecular antiwear property. It is thus inferred that there is an electron transfer process similar to a chemical reaction in the additive molecules during the antiwear process. It was also found that the dipole moment, quadrupole moment, and octupole moment were related to the wear process, indicating that the spatial distribution of the molecular charge and the electric field affected the antiwear performance.
It was further found that the molecular topological descriptors representing the structural characteristics of bonded and unbonded atoms in molecules, especially the connectivity indices, were related to the antiwear performance. All these descriptor characteristics showed that the antiwear property of additive molecules was related to the diffusion, adsorption, and reactivity of molecules.
The results of the BRNN and BPNN models [
8] for the same set of samples complement and confirm each other, but the expression of the BRNN model is more specific and refined. In future work, if the sample is large enough, we will use a deep neural network for machine learning.
2.2. Wrapper Method
Chinese tribologists have used the GFA method to screen descriptors and made a useful attempt to study the correlation between the structure of a lubricant and tribological properties. Xiong’s team studied the QSPR model of the friction coefficient and maximum non-seizure load of lubricants on a steel surface [
35]. They also built QSAR models to study the corrosion inhibition efficiency, friction coefficient, and oil film strength of thiadiazole derivatives as lubricant additives [
36]. The results showed that the quantum descriptor is a good choice to predict the tribological properties of lubricant additives.
3. Application of the QSTR Model
3.1. Isosteres
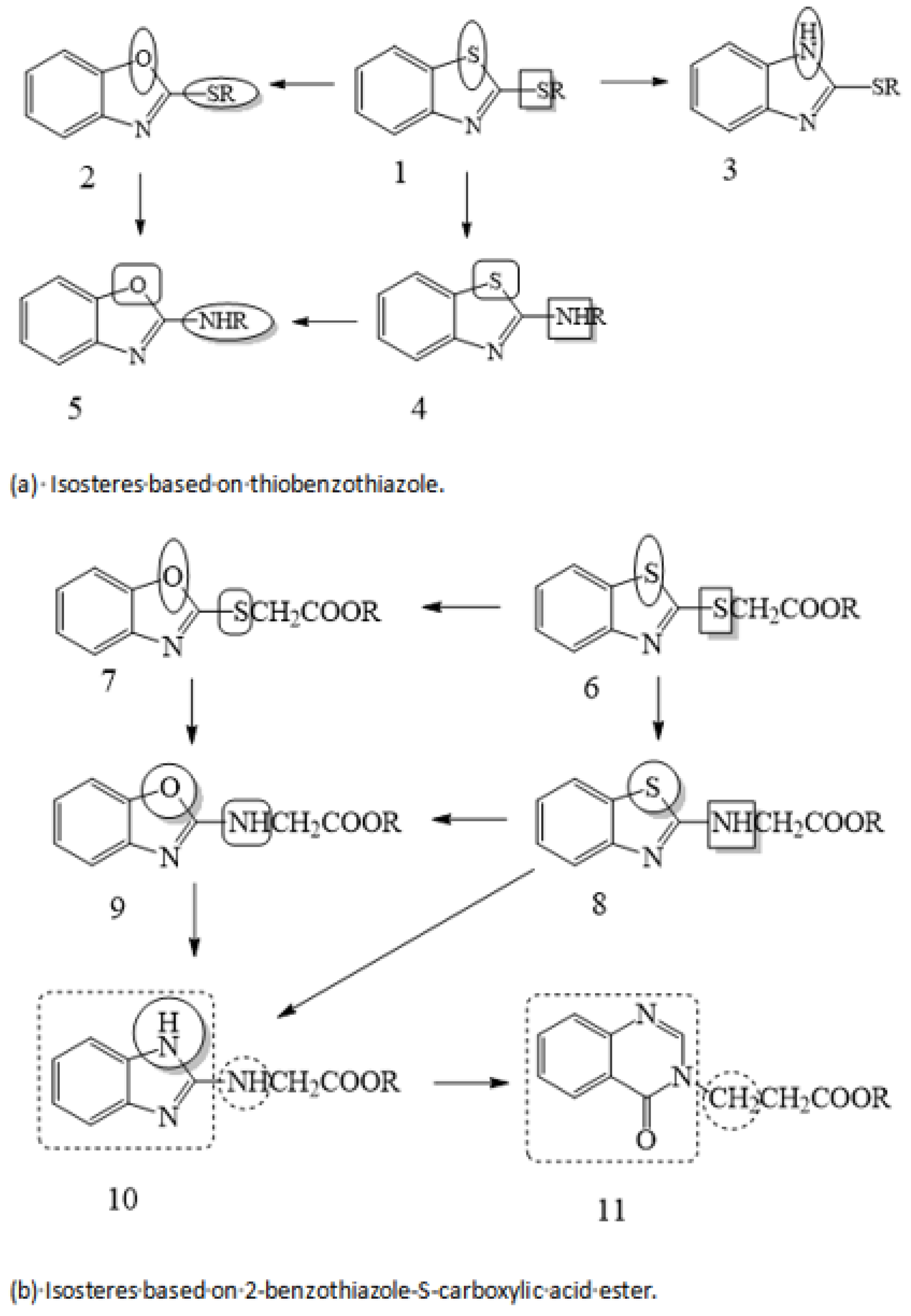
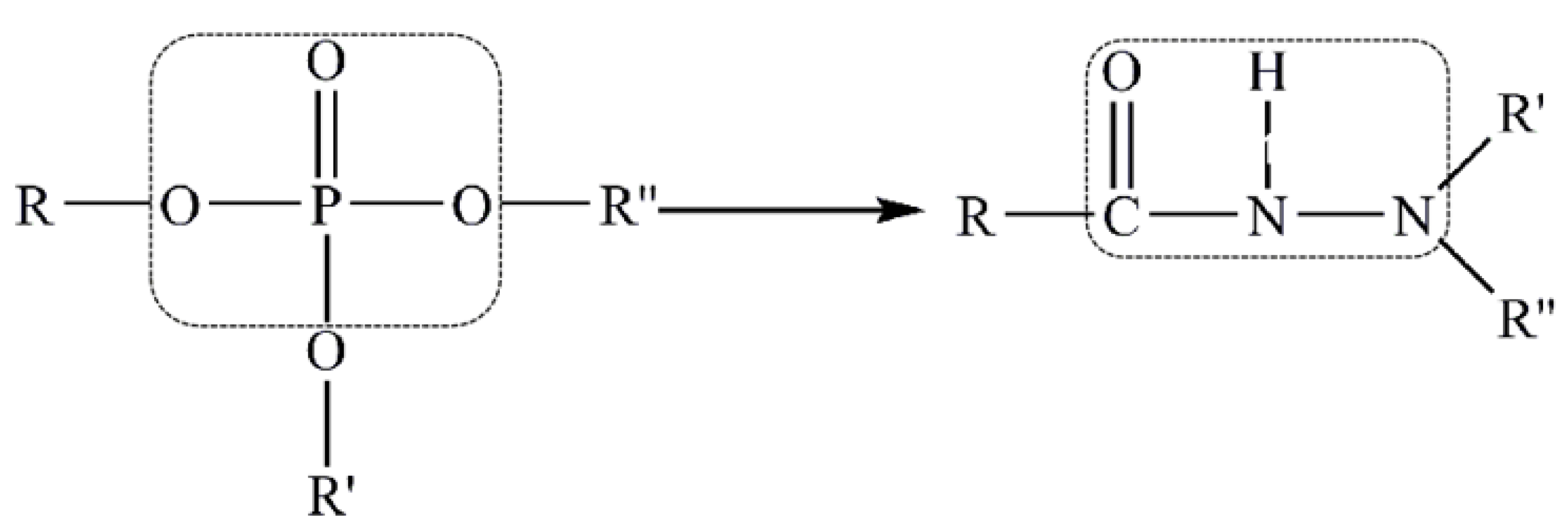
It is generally believed that a lubricant additive containing sulfur or phosphorus improves tribological properties. However, with increasing environmental protection and the promotion of relevant laws and regulations, the restrictions on pollutants are becoming more stringent. The requirements for lubricants have changed from only improving tribological properties to both improving tribological properties and having good environmental compatibility, biodegradability, and renewability and low environmental toxicity. Gao’s team conducted experiments to reduce or even completely replace sulfur and phosphorus in lubricant additives and studied the feasibility of making this change to lubricant additives. The difficulty in developing a new lubricant additive to replace sulfur and phosphorus is maintaining good tribological properties of the compounds. Gao’s team introduced the principle of isosterism to design a low-sulfur/phosphorus or sulfur/phosphorus-free lubricant additive.
Langmuir first proposed the concept of chemical isosteres in 1919. He believed that atoms, functional groups, and molecules can have similar physical and chemical properties due to similar electronic structures [
37]. In the study of structural optimization, bioisosterism is a method that is mainly based on chemical isosterism. That is to say, in the variable part of the basic structure, one isostere is replaced with another to transform the structure of the molecule. The term “isostere” refers to atoms, ions, and molecules having the same number of outer electrons and to chemical groups with similar stereoscopic and electronic configurations. Isosteres with groups or substituents that possess similar physical and chemical properties produce similar, related, or opposite biological activity. According to the principle of isosterism, S-containing molecules with excellent tribological properties were used as template molecules, and atoms or atomic groups similar in molecular volume, electronegativity, and stereochemistry were selected to replace the S element (as shown in
Figure 4), so as to partially or completely replace the S element in the lubricant additive and obtain isosteres [
27,
38].
Research on the S substitution of a lubricant additive suggests that similar methods can be used for P substitution. Using phosphate as a template, a P-free lubricant additive was obtained by replacing the PO
4 group with the CONHN group [
28], as shown in
Figure 5.
3.2. Application of CoMFA and CoMSIA-QSTR Models
Triisodecyl trimellitate (TMT), bis (2-ethylhexyl) adipate (DE), and trimethylolpropane trioleate (TMPTO) were used as the base oil. The template molecules and alternative molecules were dissolved in the base oil in a fixed proportion to form lubricants, and their tribological data were recorded in experiments. The CoMFA and CoMSIA-QSTR models were established with template molecules and isosters as samples, and base oil molecules were embedded in the models [
27]. The models indicated that a higher structural similarity between the base oil and additive results in a stronger synergistic effect of the molecular forces of the lubrication system. A synergistic molecular force field will improve tribological properties.
Hydrazine compounds were selected as lubricant additives and dissolved in three base oils, namely TMT, DE, and TMPTO, to form lubricant [
28]. CoMFA and CoMSIA-QSTR models showed that a higher structural similarity between the base oil and additive results in a greater synergistic effect of the molecular force field in the lubricant.
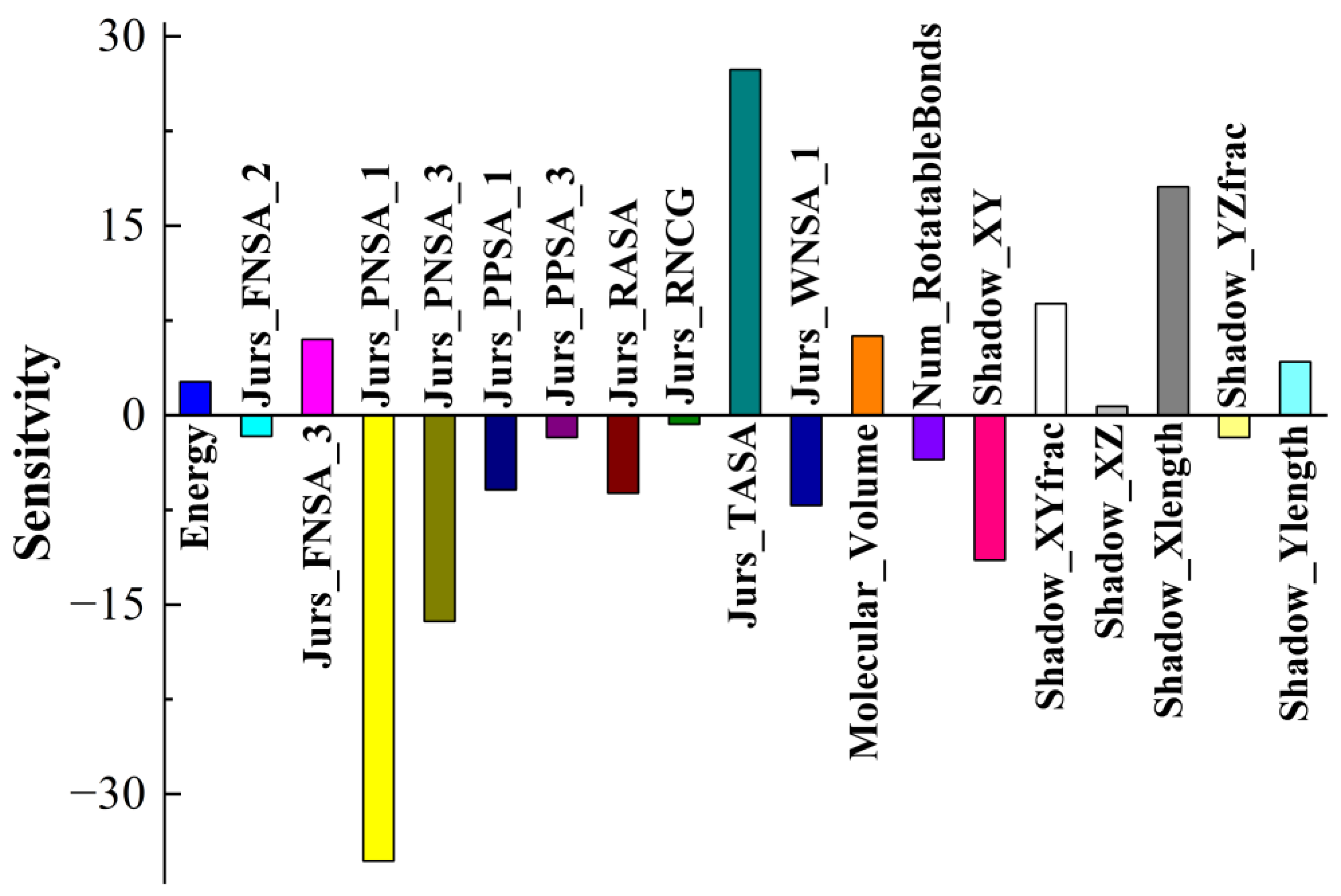
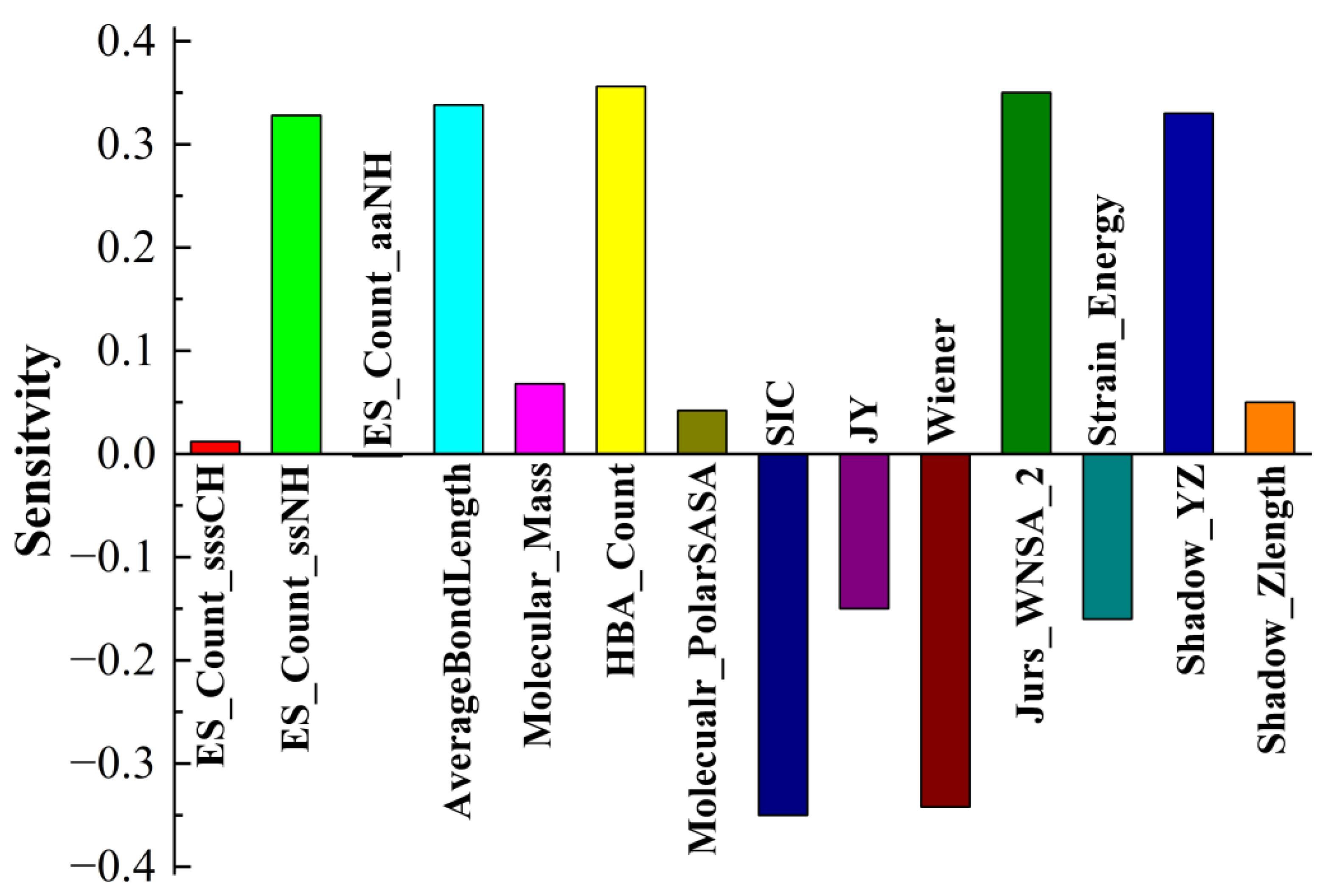
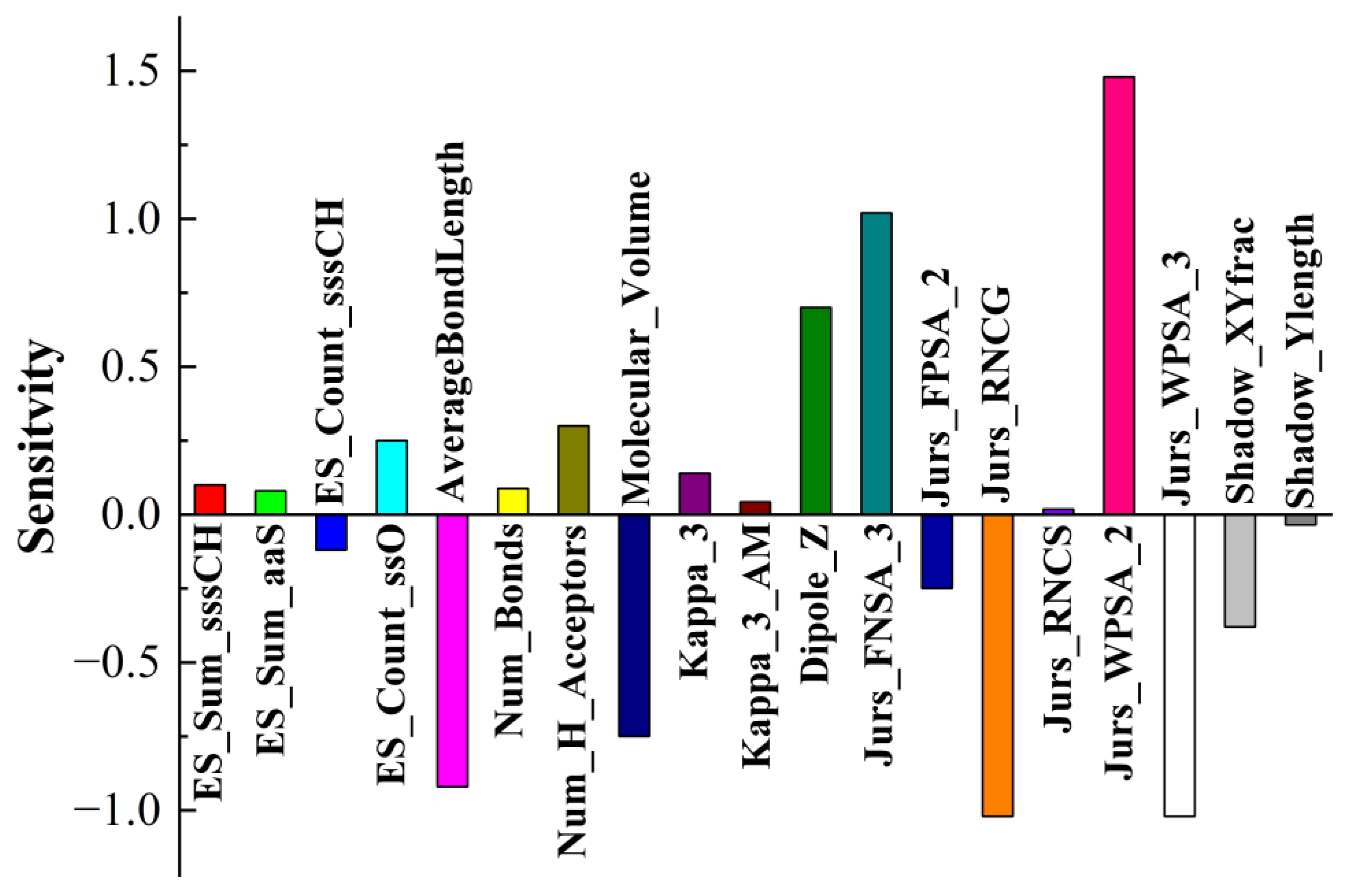
3.3. Application of the BPNN-QSTR Model
Tribological tests were carried out, where S-substituted molecules and template molecules were used as lubricant additives and TMT, DE, and TMPTO were used as base oils [
38]. BPNN-QSTR modeling and descriptor sensitivity analysis were conducted with template molecules and substitute molecules as samples, as shown in
Figure 6,
Figure 7 and
Figure 8. The rules for filtering QSTR descriptors are based on their levels of importance. The model selects the most important descriptors and removes those that are less important until selecting or removing more descriptors does not improve the model further. During the modeling, structural descriptors related to sulfur are introduced and filtered. However, almost all sulfur-related structural descriptors were subsequently removed. The only exception is sulfur-related ES_Sum_aaS in the TMPTO model, but its contribution to the antiwear performance is small. The modeling thus shows that the contribution of sulfur to the antiwear performance is small, which confirms that the substitution of sulfur in additive molecules is reasonable and feasible.
The substitution of S and P elements in lubricant additive molecules is based on the study of the correlation between the lubricant molecular structure and tribological properties. The structural groups or atoms of some variable parts of the molecular structure are replaced by searching for the similarity characteristics of structures of the compound. This work can lead to better tribological performance and environmentally friendly additive molecules. Additionally, the work is an important step in the evolution of QSTR modeling and prediction to specific molecular design.
4. Development of the QSTR Model
In the QSTR modeling of a lubricant additive, the molecular state of additive molecules in a vacuum was directly simulated without considering the effect of the molecular structure of the base oil [
23], yet the presence of the base oil will affect the molecular force field in the system. Therefore, a new solvation model was established using a base oil as the additive solvent. The solvated model was compared with the non-solvated model generated using the same data [
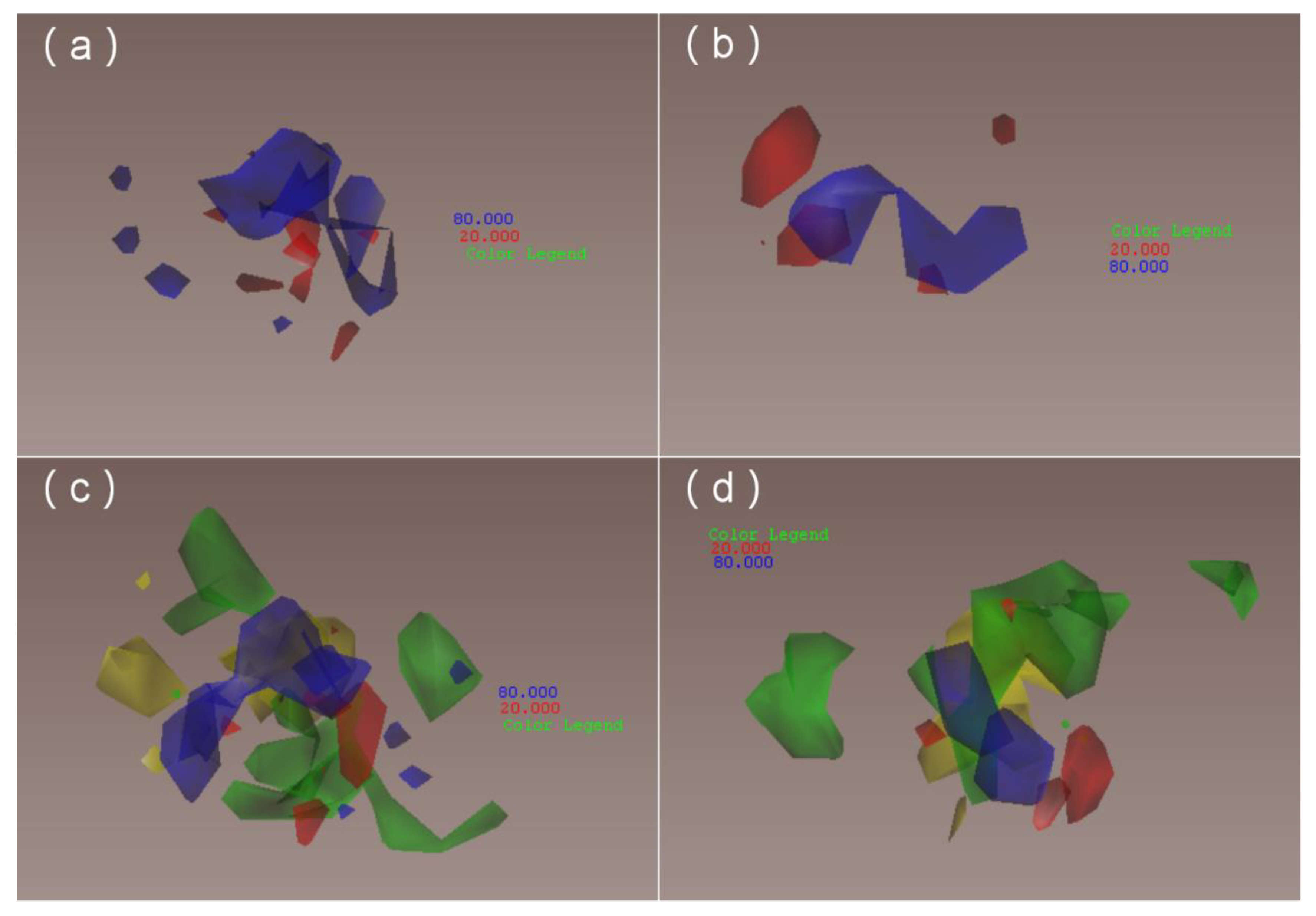
29]. In fact, both the solvated and non-solvated models meet accuracy and prediction requirements. Interestingly, the contour maps obtained using the solvated and non-solvated models are similar, although there are more small colored blocks in the former, as shown in
Figure 9. These small fragment fields provide more precise and specific predictions and provide more information relevant for modifying molecular structures.
The purpose of the work on the solvated model is to further expand research on the application of the QSTR in tribology. The structure of the base oil is included in the modeling, which allows a more comprehensive study of the effect and change rule of the QSTR of a system comprising lubricant additives and a base oil.
5. Conclusions and Outlook
In summary, QSTR method reveals the quantitative relationship between the structure and tribological properties of a material and has a simple, non-experimental, and acceptable prediction model. At present, the QSTR model is not sufficiently accurate but it provides a new means for discovering new lubricants and exploring the specific mechanisms of tribological processes. Gao’s team adopted different QSTR models to study the same data set [
8,
10,
19,
23,
29,
34]. Different QSTR models emphasize different molecular structural characteristics and further clarify the relationship between the molecular structure and tribological properties.
The QSTR model naturally has drawbacks. A series of descriptors are used to describe the tribological process of compounds, and the real tribological process must be much more complex than the process described by the model. Indeed, at the present stage, no descriptor fully and accurately reflects the structural characteristics of compounds. Therefore, the QSTR method is unable to accurately describe the specific mechanism in the friction process and only describes the mechanism or process in a general way. Tribological systems have system dependence and time variability. The system dependence can be resolved by limiting the tribological test conditions. The solution to time variability is selecting descriptors that reflect the transient structure of molecules during friction, such as conformational change, energy change, and polar change. Therefore, even though the QSTR model cannot reflect instantaneous changes of the system, the structural descriptors of the optimal state can be calculated, which provides useful data for selecting the tribological materials with the best structures. However, the introduction of these descriptors is still not comprehensive. Therefore, in future work, molecular dynamics (MD) will be included to study and explain the factors and characteristics of the tribological properties of the compound structure from a microscopic perspective and study the specific tribological mechanism more comprehensively.
This review may have limitations in that there are many issues related to lubricant research and the research on the QSTR of lubricants covered in this paper was mentioned mainly to introduce the methods of modeling. Chinese scholars may have conducted important similar work that is not covered in this review.