Tribological Properties of Borate-Based Protic Ionic Liquids as Neat Lubricants and Biolubricant Additives for Steel-Steel Contact
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of PILs
2.3. Thermal Analysis, Rheology, and Solubility
2.4. Friction and Wear Tests
- = wear volume
- = stroke length
- = contact ball radius
- = wear track width
- = wear track depth
3. Results
3.1. Thermal Analysis, Rheology, and Solubility
3.1.1. Thermogravimetric Analysis
3.1.2. Solubility
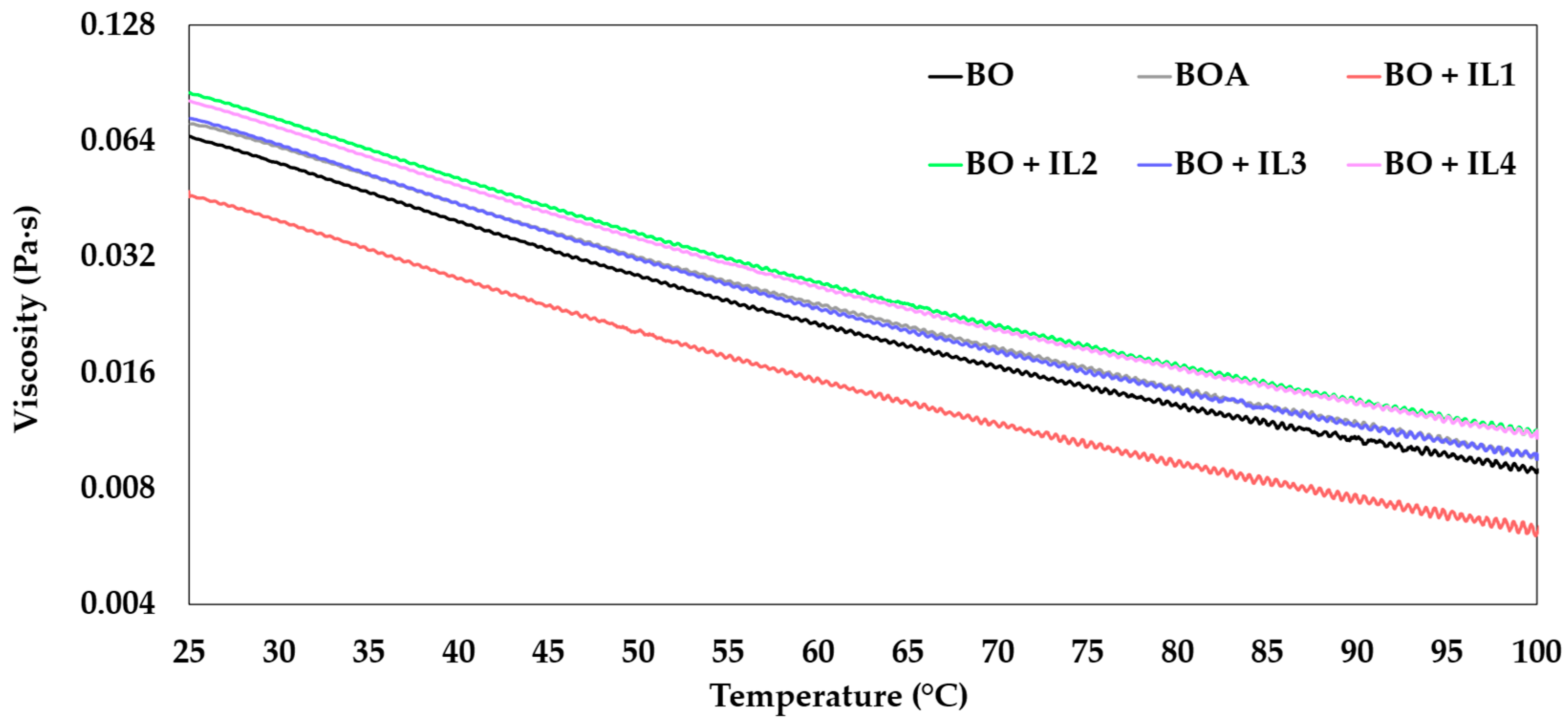
3.1.3. Rheology
3.2. Friction and Wear Tests
3.2.1. Ionic Liquids as Neat Lubricants
3.2.2. Ionic Liquids as Biolubricant Additives
4. Conclusions
- The thermal stability of the PILs increased with the molecular weight, following the series IL1 < IL2 < IL3 < IL4. Thermal stability of the mixtures was slightly reduced by the addition of the PILs.
- IL2 and IL4 showed higher viscosity than IL1 and IL3, which was attributed to a closely packed structure for that particular cationic configuration.
- BO + IL mixtures increased the viscosity of the base oil, except for BO + IL1, due to the poor solvation of the blend illustrated during the solubility study.
- IL1 and IL3 exhibited shear-thinning rheological behavior linked to the breakage of nanostructures when confined with stainless steel plates. This phenomenon was diminished in the case of IL2 and disappeared with the increment of the alkyl chain with IL4.
- When tested as neat lubricants, IL2 and IL3 prevented wear, as only superficial scratches were observed. Additionally, the PILs greatly reduced friction by 20–30% compared to BO. In the case of IL1, a solidification process led to the squeeze-out phenomena of the film.
- The 1 wt.% PILs mixtures reduced friction by 20–25% compared to BO. BO + IL1 reduced wear by 96% compared to BO and by 48% compared to BOA, performing the best among ionic liquids studied as biolubricant additives.
- The proficient performance of these PILs was mainly attributed to an adsorption phenomenon rather than a tribochemical reaction.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Holmberg, K.; Erdemir, A. Global Impact of Friction on Energy Consumption, Economy and Environment. FME Trans. 2015, 43, 181–185. [Google Scholar] [CrossRef]
- Holmberg, K.; Erdemir, A. Influence of Tribology on Global Energy Consumption, Costs and Emissions. Friction 2017, 5, 263–284. [Google Scholar] [CrossRef]
- Schneider, M.P. Plant-Oil-Based Lubricants and Hydraulic Fluids. J. Sci. Food Agric. 2006, 86, 1769–1780. [Google Scholar] [CrossRef]
- Willing, A. Lubricants Based on Renewable Resources—An Environmentally Compatible Alternative to Mineral Oil Products. Chemosphere 2001, 43, 89–98. [Google Scholar] [CrossRef]
- Syahir, A.Z.; Zulkifli, N.W.M.; Masjuki, H.H.; Kalam, M.A.; Alabdulkarem, A.; Gulzar, M.; Khuong, L.S.; Harith, M.H. A Review on Bio-Based Lubricants and Their Applications. J. Clean. Prod. 2017, 168, 997–1016. [Google Scholar] [CrossRef]
- Chan, C.H.; Tang, S.W.; Mohd, N.K.; Lim, W.H.; Yeong, S.K.; Idris, Z. Tribological Behavior of Biolubricant Base Stocks and Additives. Renew. Sustain. Energy Rev. 2018, 93, 145–157. [Google Scholar] [CrossRef]
- Sarma, R.N.; Vinu, R. Current Status and Future Prospects of Biolubricants: Properties and Applications. Lubricants 2022, 10, 70. [Google Scholar] [CrossRef]
- Reeves, C.J.; Siddaiah, A.; Menezes, P.L. Ionic Liquids: A Plausible Future of Bio-Lubricants. J. Bio-Tribo-Corrosion 2017, 3, 18. [Google Scholar] [CrossRef]
- Cai, M.; Guo, R.; Zhou, F.; Liu, W. Lubricating a Bright Future: Lubrication Contribution to Energy Saving and Low Carbon Emission. Sci. China Technol. Sci. 2013, 56, 2888–2913. [Google Scholar] [CrossRef]
- Somers, A.E.; Howlett, P.C.; MacFarlane, D.R.; Forsyth, M. A Review of Ionic Liquid Lubricants. Lubricants 2013, 1, 3–21. [Google Scholar] [CrossRef]
- Stolte, S.; Steudte, S.; Areitioaurtena, O.; Pagano, F.; Thöming, J.; Stepnowski, P.; Igartua, A. Ionic Liquids as Lubricants or Lubrication Additives: An Ecotoxicity and Biodegradability Assessment. Chemosphere 2012, 89, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Adukure, A.R.; Iglesias, P. Effect of Ionicity of Three Protic Ionic Liquids as Neat Lubricants and Lubricant Additives to a Biolubricant. Coatings 2019, 9, 713. [Google Scholar] [CrossRef]
- Cigno, E.; Magagnoli, C.; Pierce, M.S.; Iglesias, P. Lubricating Ability of Two Phosphonium-Based Ionic Liquids as Additives of a Bio-Oil for Use in Wind Turbines Gearboxes. Wear 2017, 376–377, 756–765. [Google Scholar] [CrossRef]
- Walden, P. Molecular Weights and Electrical Conductivity of Several Fused Salts. Bull. Acad. Imper. Sci. 1914, 8, 405–422. [Google Scholar]
- Ye, C.; Liu, W.; Chen, Y.; Yu, L. Room-Temperature Ionic Liquids: A Novel Versatile Lubricant. Chem. Commun. 2001, 21, 2244–2245. [Google Scholar] [CrossRef] [PubMed]
- Greaves, T.L.; Drummond, C.J. Protic Ionic Liquids: Properties and Applications. Chem. Rev. 2008, 108, 206–237. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, S.A.; Pringle, J.M.; MacFarlane, D.R. Ionic Liquids—An Overview. Aust. J. Chem. 2004, 57, 113–119. [Google Scholar] [CrossRef]
- Plechkova, N.V.; Seddon, K.R. Applications of Ionic Liquids in the Chemical Industry. Chem. Soc. Rev. 2008, 37, 123–150. [Google Scholar] [CrossRef]
- Cai, M.; Yu, Q.; Liu, W.; Zhou, F. Ionic Liquid Lubricants: When Chemistry Meets Tribology. Chem. Soc. Rev. 2020, 49, 7753–7818. [Google Scholar] [CrossRef] [PubMed]
- Minami, I. Ionic Liquids in Tribology. Molecules 2009, 14, 2286–2305. [Google Scholar] [CrossRef] [PubMed]
- Bermúdez, M.D.; Jiménez, A.E.; Sanes, J.; Carrión, F.J. Ionic Liquids as Advanced Lubricant Fluids. Molecules 2009, 14, 2888–2908. [Google Scholar] [CrossRef]
- Canongia Lopes, J.N.A.; Pádua, A.A.H. Nanostructural Organization in Ionic Liquids. J. Phys. Chem. B 2006, 110, 3330–3335. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Zhang, S. Hydrogen Bonds: A Structural Insight into Ionic Liquids. Chem.-A Eur. J. 2012, 18, 2748–2761. [Google Scholar] [CrossRef] [PubMed]
- Katsyuba, S.A.; Vener, M.V.; Zvereva, E.E.; Fei, Z.; Scopelliti, R.; Laurenczy, G.; Yan, N.; Paunescu, E.; Dyson, P.J. How Strong Is Hydrogen Bonding in Ionic Liquids? Combined x-Ray Crystallographic, Infrared/Raman Spectroscopic, and Density Functional Theory Study. J. Phys. Chem. B 2013, 117, 9094–9105. [Google Scholar] [CrossRef]
- Hunt, P.A.; Ashworth, C.R.; Matthews, R.P. Hydrogen Bonding in Ionic Liquids. Chem. Soc. Rev. 2015, 44, 1257–1288. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Zhang, S.; Wang, J. Understanding the Hydrogen Bonds in Ionic Liquids and Their Roles in Properties and Reactions. Chem. Commun. 2016, 52, 6744–6764. [Google Scholar] [CrossRef]
- Greaves, T.L.; Drummond, C.J. Solvent Nanostructure, the Solvophobic Effect and Amphiphile Self-Assembly in Ionic Liquids. Chem. Soc. Rev. 2013, 42, 1096–1120. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Kozbial, A.; Rose, F.; Li, L. Effect of π-Π+ Stacking on the Layering of Ionic Liquids Confined to an Amorphous Carbon Surface. ACS Appl. Mater. Interfaces 2015, 7, 7078–7081. [Google Scholar] [CrossRef] [PubMed]
- Deetlefs, M.; Hardacre, C.; Nieuwenhuyzen, M.; Padua, A.A.H.; Sheppard, O.; Soper, A.K. Liquid Structure of the Ionic Liquid 1,3-Dimethylimidazolium Bis{(Trifluoromethyl)Sulfonyl}amide. J. Phys. Chem. B 2006, 110, 12055–12061. [Google Scholar] [CrossRef] [PubMed]
- Greaves, T.L.; Drummond, C.J. Protic Ionic Liquids: Evolving Structure-Property Relationships and Expanding Applications. Chem. Rev. 2015, 115, 11379–11448. [Google Scholar] [CrossRef] [PubMed]
- Hayes, R.; Warr, G.G.; Atkin, R. Structure and Nanostructure in Ionic Liquids. Chem. Rev. 2015, 115, 6357–6426. [Google Scholar] [CrossRef] [PubMed]
- Dong, K.; Liu, X.; Dong, H.; Zhang, X.; Zhang, S. Multiscale Studies on Ionic Liquids. Chem. Rev. 2017, 117, 6636–6695. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, J. Structures and Interactions of Ionic Liquids; Zhang, S., Wang, J., Lu, X., Zhou, Q., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; ISBN 9783642386183. [Google Scholar]
- Zhou, H.; Rouha, M.; Feng, G.; Lee, S.S.; Docherty, H.; Fenter, P.; Cummings, P.T.; Fulvio, P.F.; Dai, S.; Mcdonough, J.; et al. Nanoscale Perturbations of Room Temperature Ionic Liquid Structure at Charged and Uncharged Interfaces. ACS Nano 2012, 6, 9818–9827. [Google Scholar] [CrossRef] [PubMed]
- Perkin, S. Ionic Liquids in Confined Geometries. Phys. Chem. Chem. Phys. 2012, 14, 5052–5062. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, M.; Kemp, T.F.; Howlett, P.C.; Sun, J.; Smith, M.E. A Potential Novel Rapid Screening NMR Approach to Boundary Film Formation at Solid Interfaces in Contact with Ionic Liquids. J. Phys. Chem. C 2008, 112, 13801–13804. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, W.; Voth, G.A. Spatial Heterogeneity in Ionic Liquids. ACS Symp. Ser. 2007, 975, 272–307. [Google Scholar] [CrossRef]
- Hayashi, S.; Ozawa, R.; Hamaguchi, H.O. Raman Spectra, Crystal Polymorphism, and Structure of a Prototype Ionic-Liquid [Bmim]Cl. Chem. Lett. 2003, 32, 498–499. [Google Scholar] [CrossRef]
- Swatloski, R.P.; Holbrey, J.D.; Rogers, R.D. Ionic Liquids Are Not Always Green: Hydrolysis of 1-Butyl-3-Methylimidazolium Hexafluorophosphate. Green Chem. 2003, 5, 361–363. [Google Scholar] [CrossRef]
- Wamser, C.A. Hydrolysis of Fluoboric Acid in Aqueous Solution. J. Am. Chem. Soc. 1948, 70, 1209–1215. [Google Scholar] [CrossRef]
- Shah, F.U.; Glavatskih, S.; Antzutkin, O.N. Boron in Tribology: From Borates to Ionic Liquids. Tribol. Lett. 2013, 51, 281–301. [Google Scholar] [CrossRef]
- Qu, J.; Truhan, J.J. An Efficient Method for Accurately Determining Wear Volumes of Sliders with Non-Flat Wear Scars and Compound Curvatures. Wear 2006, 261, 848–855. [Google Scholar] [CrossRef]
- Cao, Y.; Mu, T. Comprehensive Investigation on the Thermal Stability of 66 Ionic Liquids by Thermogravimetric Analysis. Ind. Eng. Chem. Res. 2014, 53, 8651–8664. [Google Scholar] [CrossRef]
- Atkin, R.; Warr, G.G. The Smallest Amphiphiles: Nanostructure in Protic Room-Temperature Ionic Liquids with Short Alkyl Groups. J. Phys. Chem. B 2008, 112, 4164–4166. [Google Scholar] [CrossRef] [PubMed]
- Sedov, I.A.; Solomonov, B.N. Thermodynamic Description of the Solvophobic Effect in Ionic Liquids. Fluid Phase Equilib. 2016, 425, 9–14. [Google Scholar] [CrossRef]
- Kang, W.; Dong, B.; Gao, Y.; Zheng, L. Aggregation Behavior of Long-Chain Imidazolium Ionic Liquids in Ethylammonium Nitrate. Colloid Polym. Sci. 2010, 288, 1225–1232. [Google Scholar] [CrossRef]
- Philippi, F.; Rauber, D.; Eliasen, K.L.; Bouscharain, N.; Niss, K.; Kay, C.W.M.; Welton, T. Pressing Matter: Why Are Ionic Liquids so Viscous? Chem. Sci. 2022, 13, 2735–2743. [Google Scholar] [CrossRef]
- Rocha, M.A.A.; Neves, C.M.S.S.; Freire, M.G.; Russina, O.; Triolo, A.; Coutinho, J.A.P.; Santos, L.M.N.B.F. Alkylimidazolium Based Ionic Liquids: Impact of Cation Symmetry on Their Nanoscale Structural Organization. J. Phys. Chem. B 2013, 117, 10889–10897. [Google Scholar] [CrossRef]
- Doolittle, A.K. Studies in Newtonian Flow. II. the Dependence of the Viscosity of Liquids on Free-Space. J. Appl. Phys. 1951, 22, 1471–1475. [Google Scholar] [CrossRef]
- Harrap, B.S.; Heymann, E. Theories of Viscosity Applied to Ionic Liquids. Chem. Rev. 1951, 48, 45–67. [Google Scholar] [CrossRef]
- Silva, W.; Zanatta, M.; Ferreira, A.S.; Corvo, M.C.; Cabrita, E.J. Revisiting Ionic Liquid Structure-property Relationship: A Critical Analysis. Int. J. Mol. Sci. 2020, 21, 7745. [Google Scholar] [CrossRef]
- Burrell, G.L.; Dunlop, N.F.; Separovic, F. Non-Newtonian Viscous Shear Thinning in Ionic Liquids. Soft Matter 2010, 6, 2080–2086. [Google Scholar] [CrossRef]
- Smith, J.A.; Webber, G.B.; Warr, G.G.; Atkin, R. Rheology of Protic Ionic Liquids and Their Mixtures. J. Phys. Chem. B 2013, 117, 13930–13935. [Google Scholar] [CrossRef] [PubMed]
- Shakeel, A.; Mahmood, H.; Farooq, U.; Ullah, Z.; Yasin, S.; Iqbal, T.; Chassagne, C.; Moniruzzaman, M. Rheology of Pure Ionic Liquids and Their Complex Fluids: A Review. ACS Sustain. Chem. Eng. 2019, 7, 13586–13626. [Google Scholar] [CrossRef]
- Yamaguchi, T. Shear Thinning and Nonlinear Structural Deformation of Ionic Liquids with Long Alkyl Chains Studied by Molecular Dynamics Simulation. J. Phys. Chem. B 2019, 123, 6260–6265. [Google Scholar] [CrossRef]
- Capozza, R.; Vanossi, A.; Benassi, A.; Tosatti, E. Squeezout Phenomena and Boundary Layer Formation of a Model Ionic Liquid under Confinement and Charging. J. Chem. Phys. 2015, 142, 64707. [Google Scholar] [CrossRef]
- Han, M.; Espinosa-Marzal, R.M. Electroviscous Retardation of the Squeeze out of Nanoconfined Ionic Liquids. J. Phys. Chem. C 2018, 122, 21344–21355. [Google Scholar] [CrossRef]
- Han, M.; Espinosa-Marzal, R.M. Molecular Mechanisms Underlying Lubrication by Ionic Liquids: Activated Slip and Flow. Lubricants 2018, 6, 64. [Google Scholar] [CrossRef]
- Klein, J.; Kumacheva, E. Simple Liquids Confined to Molecularly Thin Layers. I. Confinement-Induced Liquid-to-Solid Phase Transitions. J. Chem. Phys. 1998, 108, 6996–7009. [Google Scholar] [CrossRef]
- Kumacheva, E.; Klein, J. Simple Liquids Confined to Molecularly Thin Layers. II. Shear and Frictional Behavior of Solidified Films. J. Chem. Phys. 1998, 108, 7010–7022. [Google Scholar] [CrossRef]
- Perkin, S.; Albrecht, T.; Klein, J. Layering and Shear Properties of an Ionic Liquid, 1-Ethyl-3- Methylimidazolium Ethylsulfate, Confined to Nano-Films between Mica Surfaces. Phys. Chem. Chem. Phys. 2010, 12, 1243–1247. [Google Scholar] [CrossRef]
- Smith, A.M.; Lovelock, K.R.J.; Perkin, S. Monolayer and Bilayer Structures in Ionic Liquids and Their Mixtures Confined to Nano-Films. Faraday Discuss. 2013, 167, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Calandra, P.; Szerb, E.I.; Lombardo, D.; Algieri, V.; De Nino, A.; Maiuolo, L. A Presentation of Ionic Liquids as Lubricants: Some Critical Comments. Appl. Sci. 2021, 11, 5677. [Google Scholar] [CrossRef]
- Li, Z.; Mangolini, F. Recent Advances in Nanotribology of Ionic Liquids. Exp. Mech. 2021, 61, 1093–1107. [Google Scholar] [CrossRef]













 | |||||
| Code | R1 | R2 | Cation * | Anion * | Name |
| IL1 |  |  |  |  | Ethanolamine 1,2-dodecanediol borate |
| IL2 |  |  |  |  | N-methylethanolamine 1,2-dodecanediol borate |
| IL3 |  |  |  |  | N’N-dimethylethanolamine 1,2-dodecanediol borate |
| IL4 |  |  |  |  | N-butylethanolamine 1,2-dodecanediol borate |
 Boron
Boron  Nitrogen
Nitrogen  Oxygen
Oxygen  Carbon
Carbon  Hydrogen.
Hydrogen.| Parameter | Value |
|---|---|
| Normal load (N) | 3 |
| Max. Hertz contact pressure (GPa) | 2.36 |
| Stroke length (mm) | 3 |
| Frequency (Hz) | 3 |
| Duration (s) | 3600 |
| Sliding speed (m/s) | 0.03 |
| Sliding distance (m) | 64.8 |
| Lubricant | Tonset (°C) |
|---|---|
| BO | 348.7 |
| BOA | 348.5 |
| IL1 | 268.0 |
| IL2 | 273.6 |
| IL3 | 293.7 |
| IL4 | 263.1 |
| BO + IL1 | 330.5 |
| BO + IL2 | 324.5 |
| BO + IL3 | 344.0 |
| BO + IL4 | 349.2 |
| Dynamic Viscosity (Pa s) | Arrhenius Parameters | |||||
|---|---|---|---|---|---|---|
| Lubricant | 25 °C | 40 °C | 100 °C | A (106) (Pa·s) | Ea (kJ/mol) | r2 |
| BO | 0.0644 | 0.0392 | 0.0088 | 1.82 | 26.02 | 0.997 |
| BOA | 0.0708 | 0.0427 | 0.0096 | 2.30 | 25.68 | 0.998 |
| IL1 | 0.5294 | 0.2280 | 0.0260 | 2.27 × 10−2 | 42.02 | 0.998 |
| IL2 | 97.48 | 24.42 | 0.1607 | 1.75 × 10−5 | 72.48 | 0.998 |
| IL3 | 2.723 | 0.8661 | 0.0194 | 1.11 × 10−5 | 65.20 | 0.999 |
| IL4 | 13.65 | 4.938 | 0.0556 | 2.86 × 10−4 | 61.34 | 0.992 |
| BO + IL1 | 0.0461 | 0.0281 | 0.0060 | 1.32 | 25.95 | 0.995 |
| BO + IL2 | 0.0862 | 0.0512 | 0.0113 | 2.00 | 26.44 | 0.997 |
| BO + IL3 | 0.0735 | 0.0439 | 0.0096 | 1.70 | 26.47 | 0.997 |
| BO + IL4 | 0.0811 | 0.0491 | 0.0109 | 2.42 | 25.84 | 0.998 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sierra, A.; Coleman, M.G.; Iglesias, P. Tribological Properties of Borate-Based Protic Ionic Liquids as Neat Lubricants and Biolubricant Additives for Steel-Steel Contact. Lubricants 2022, 10, 269. https://doi.org/10.3390/lubricants10100269
Sierra A, Coleman MG, Iglesias P. Tribological Properties of Borate-Based Protic Ionic Liquids as Neat Lubricants and Biolubricant Additives for Steel-Steel Contact. Lubricants. 2022; 10(10):269. https://doi.org/10.3390/lubricants10100269
Chicago/Turabian StyleSierra, Alfonso, Michael G. Coleman, and Patricia Iglesias. 2022. "Tribological Properties of Borate-Based Protic Ionic Liquids as Neat Lubricants and Biolubricant Additives for Steel-Steel Contact" Lubricants 10, no. 10: 269. https://doi.org/10.3390/lubricants10100269
APA StyleSierra, A., Coleman, M. G., & Iglesias, P. (2022). Tribological Properties of Borate-Based Protic Ionic Liquids as Neat Lubricants and Biolubricant Additives for Steel-Steel Contact. Lubricants, 10(10), 269. https://doi.org/10.3390/lubricants10100269






