Exploring an Innovative Approach: Integrating Negative-Pressure Wound Therapy with Silver Nanoparticle Dressings in Skin Graft Procedures
Abstract
1. Introduction
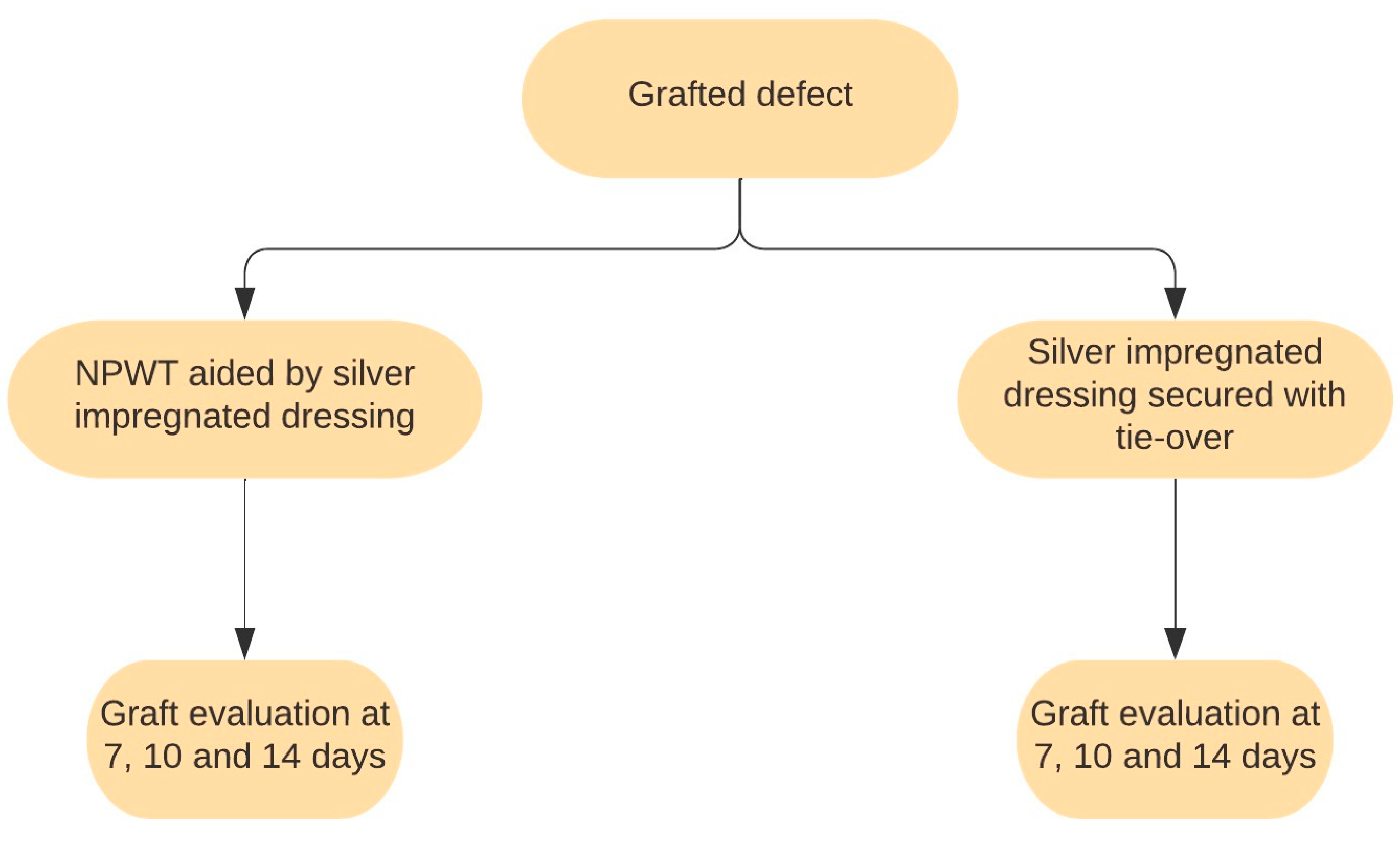
2. Materials and Methods
2.1. Inclusion Criteria
- Patients with lower-limb soft-tissue defects that required grafting;
- Adults.
2.2. Exclusion Criteria
- Children;
- Pregnant women;
- Patients who refused to participate;
- Patients discharged earlier than 14 days;
- Active infections.
2.3. Variables and Definitions
- Graft area smaller than 150 cm2–25 patients;
- Graft area between 150 cm2 and 500 cm2–42 patients;
- Graft area greater than 500 cm2–13 patients.
2.4. Statistical analysis
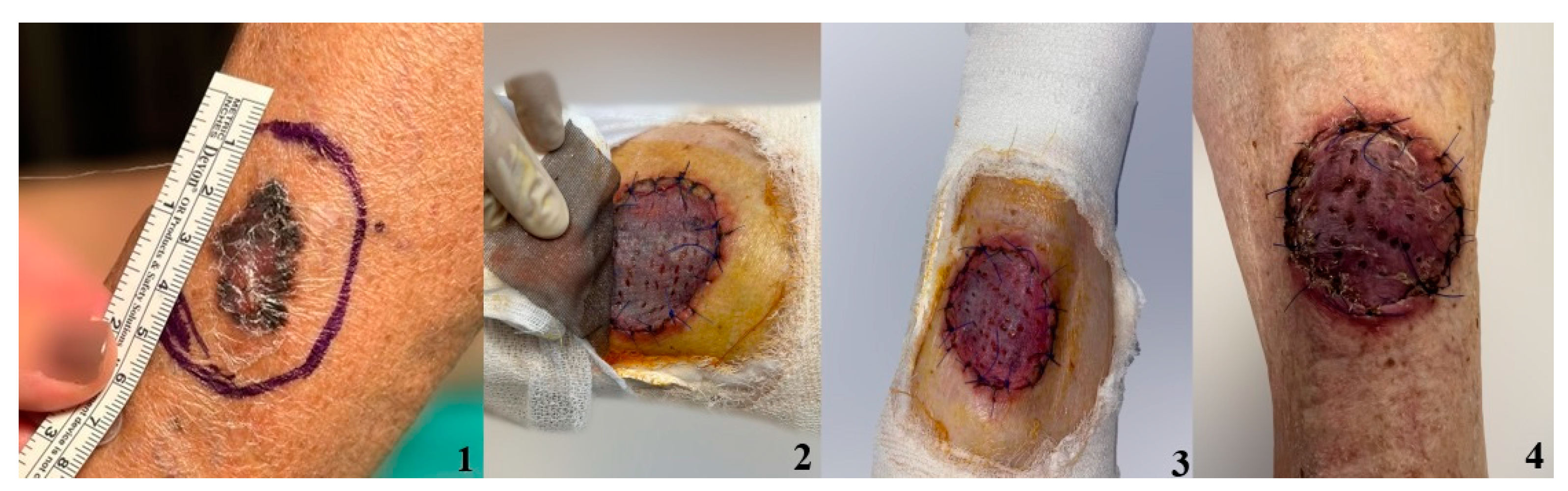
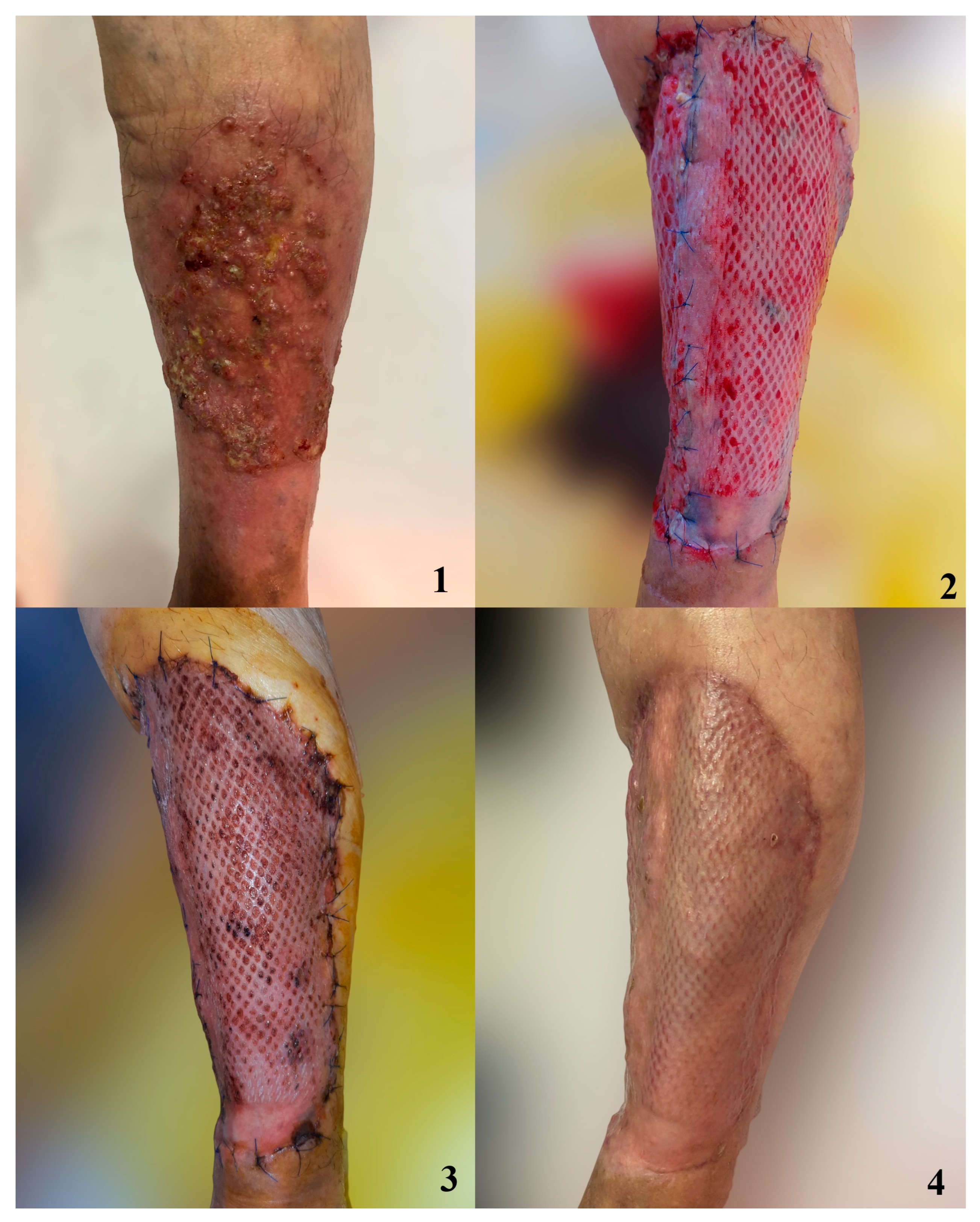
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Herskovitz, I.; Hughes, O.B.; Macquhae, F.; Rakosi, A.; Kirsner, R. Epidermal skin grafting. Int. Wound J. 2016, 13, 52–56. [Google Scholar] [CrossRef]
- Sun, B.K.; Siprashvili, Z.; Khavari, P.A. Advances in skin grafting and treatment of cutaneous wounds. Science 2014, 346, 941–945. [Google Scholar] [CrossRef]
- Khan, A.A.; Khan, I.M.; Nguyen, P.P.; Lo, E.; Chahadeh, H.; Cerniglia, M.; Noriega, J.A. Skin Graft Techniques. Clin. Podiatr. Med. Surg. 2020, 37, 821–835. [Google Scholar] [CrossRef] [PubMed]
- Jarjis, R.D.; Jørgensen, L.; Finnerup, K.; Birk-Sørensen, L. Complications in skin grafts when continuing antithrombotic therapy prior to cutaneous surgery requiring skin grafting: A systematic review. J. Plast. Surg. Hand Surg. 2015, 49, 129–134. [Google Scholar] [CrossRef]
- Paw, E.; Vangaveti, V.; Zonta, M.; Heal, C.; Gunnarsson, R. Effectiveness of fibrin glue in skin graft survival: A systematic review and meta-analysis. Ann. Med. Surg. 2020, 56, 48–55. [Google Scholar] [CrossRef]
- Louiselle, A.E.; Niemiec, S.M.; Zgheib, C.; Liechty, K.W. Macrophage polarization and diabetic wound healing. Transl. Res. 2021, 236, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Ramanujam, C.L.; Han, D.; Fowler, S.; Kilpadi, K.; Zgonis, T. Impact of Diabetes and Comorbidities on Split-Thickness Skin Grafts for Foot Wounds. J. Am. Podiatr. Med. Assoc. 2013, 103, 223–232. [Google Scholar] [CrossRef]
- Patel, B.J.; Asher, C.M.; Bystrzonowski, N.; Healy, C. Safeguarding Skin Grafts: An Evidence-Based Summary of Fixation Techniques. Ann. Plast. Surg. 2021, 87, e180–e188. [Google Scholar] [CrossRef]
- Rădulescu, M.; Andronescu, E.; Cirja, A.; Holban, A.M.; Mogoantă, L.; Bălşeanu, T.A.; Cătălin, B.; Neagu, T.P.; Lascăr, I.; Florea, D.A.; et al. Antimicrobial coatings based on zinc oxide and orange oil for improved bioactive wound dressings and other applications. Rom. J. Morphol. Embryol. 2016, 57, 107–114. [Google Scholar] [PubMed]
- Avino, A.; Jecan, C.R.; Cozma, C.N.; Balcangiu Stroescu, A.E.; Balan, D.G.; Ionescu, D.; Mihai, A.; Tanase, M.; Raducu, L. Negative Pressure Wound Therapy Using Polyurethane Foam in a Patient with Necrotizing Fasciitis. Mater. Plast. 2018, 55, 603–605. [Google Scholar] [CrossRef]
- Ravindhran, B.; Schafer, N.; Howitt, A.; Carradice, D.; Smith, G.; Chetter, I. Molecular mechanisms of action of negative pressure wound therapy: A systematic review. Expert Rev. Mol. Med. 2023, 25, e29. [Google Scholar] [CrossRef]
- Yamashiro, T.; Kushibiki, T.; Mayumi, Y.; Tsuchiya, M.; Ishihara, M.; Azuma, R. Negative-Pressure Wound Therapy: What We Know and What We Need to Know. Adv. Exp. Med. Biol. 2023, 1436, 131–152. [Google Scholar] [CrossRef]
- Normandin, S.; Safran, T.; Winocour, S.; Chu, C.K.; Vorstenbosch, J.; Murphy, A.M.; Davison, P.G. Negative Pressure Wound Therapy: Mechanism of Action and Clinical Applications. Semin. Plast. Surg. 2021, 35, 164–170. [Google Scholar] [CrossRef]
- Nolff, M.C. Filling the vacuum: Role of negative pressure wound therapy in open wound management in cats. J. Feline Med. Surg. 2021, 23, 823–833. [Google Scholar] [CrossRef]
- Song, H.; Xu, Y.; Chang, W.; Zhuang, J.; Wu, X. Negative pressure wound therapy promotes wound healing by suppressing macrophage inflammation in diabetic ulcers. Regen. Med. 2020, 15, 2341–2349. [Google Scholar] [CrossRef]
- Kutwin, M.; Woźniacka, A. Interleukins 20 and 8—Less widely known cytokines in psoriasis. Adv. Dermatol. Allergol. 2023, 40, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.-J.; Song, C.-S.; Jiang, N. Single nucleotide variations in the development of diabetic foot ulcer: A narrative review. World J. Diabetes 2022, 13, 1140–1153. [Google Scholar] [CrossRef] [PubMed]
- Ongarora, B.G. Recent technological advances in the management of chronic wounds: A literature review. Heal. Sci. Rep. 2022, 5, e641. [Google Scholar] [CrossRef]
- Fol, M.F.; Hamdi, S.A.-H.; Abdel-Rahman, H.A.; Mostafa, N.A. In vivo efficacy of silver nanoparticles against Syphacia muris infected laboratory Wistar rats. J. Parasit. Dis. 2023, 47, 744–756. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Xu, Z.; Jiang, Q.; Zheng, Y.; Chen, Z.; Chen, X. Characterization and biological evaluation of a novel silver nanoparticle-loaded collagen-chitosan dressing. Regen. Biomater. 2020, 7, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Sabarees, G.; Velmurugan, V.; Tamilarasi, G.P.; Alagarsamy, V.; Solomon, V.R. Recent Advances in Silver Nanoparticles Containing Nanofibers for Chronic Wound Management. Polymers 2022, 14, 3994. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Zhang, R.; Li, S.; Guo, J.; Hou, Z.; Zhang, Y. Negative-pressure therapy versus conventional therapy on split-thickness skin graft: A systematic review and meta-analysis. Int. J. Surg. 2018, 50, 43–48. [Google Scholar] [CrossRef]
- Glass, G.E.; Murphy, G.F.; Esmaeili, A.; Lai, L.; Nanchahal, J. Systematic review of molecular mechanism of action of negative-pressure wound therapy. Br. J. Surg. 2014, 101, 1627–1636. [Google Scholar] [CrossRef]
- Wu, M.M.; Liu, Q.; Yu, Z.M.; Karvar, M.; Aoki, S.M.; Hamaguchi, R.B.; Ma, C.; Orgill, D.P.M.; Panayi, A.C. Negative-Pressure Wound Therapy Induces Lymphangiogenesis in Murine Diabetic Wound Healing. Plast. Reconstr. Surg. 2023, 151, 779–790. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Dumville, J.C.; Hinchliffe, R.J.; Cullum, N.; Game, F.; Stubbs, N.; Sweeting, M.; Peinemann, F. Negative pressure wound therapy for treating foot wounds in people with diabetes mellitus. Cochrane Database Syst. Rev. 2018, 2018, CD010318. [Google Scholar] [CrossRef] [PubMed]
- Cuomo, R.; Grimaldi, L.; Nisi, G.; Zerini, I.; Giardino, F.R.; Brandi, C. Ultraportable Devices for Negative Pressure Wound Therapy: First Comparative Analysis. J. Investig. Surg. 2021, 34, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Shou, K.; Niu, Y.; Zheng, X.; Ma, Z.; Jian, C.; Qi, B.; Hu, X.; Yu, A. Enhancement of Bone-Marrow-Derived Mesenchymal Stem Cell Angiogenic Capacity by NPWT for a Combinatorial Therapy to Promote Wound Healing with Large Defect. BioMed Res. Int. 2017, 2017, 7920265. [Google Scholar] [CrossRef]
- Shah, A.; Sumpio, B.J.; Tsay, C.; Swallow, M.; Dash, B.; Thorn, S.L.; Sinusas, A.J.; Koo, A.; Hsia, H.C.; Au, A. Incisional Negative Pressure Wound Therapy Augments Perfusion and Improves Wound Healing in a Swine Model Pilot Study. Ann. Plast. Surg. 2019, 82, S222–S227. [Google Scholar] [CrossRef]
- Notorgiacomo, G.; Klug, J.; Rapp, S.; Boyce, S.T.; Schutte, S.C. A bioreactor for studying negative pressure wound therapy on skin grafts. Int. Wound J. 2022, 19, 633–642. [Google Scholar] [CrossRef]
- Erba, P.; Ogawa, R.; Ackermann, M.; Adini, A.; Miele, L.F.; Dastouri, P.; Helm, D.; Mentzer, S.J.; D’amato, R.J.; Murphy, G.F.; et al. Angiogenesis in Wounds Treated by Microdeformational Wound Therapy. Ann. Surg. 2011, 253, 402–409. [Google Scholar] [CrossRef]
- Wang, X.; Fan, W.; Li, N.; Ma, Y.; Yao, M.; Wang, G.; He, S.; Li, W.; Tan, J.; Lu, Q.; et al. YY1 lactylation in microglia promotes angiogenesis through transcription activation-mediated upregulation of FGF2. Genome Biol. 2023, 24, 87. [Google Scholar] [CrossRef] [PubMed]
- Mascharak, S.; Desjardins-Park, H.E.; Davitt, M.F.; Guardino, N.J.; Gurtner, G.C.; Wan, D.C.; Longaker, M.T. Modulating Cellular Responses to Mechanical Forces to Promote Wound Regeneration. Adv. Wound Care 2022, 11, 479–495. [Google Scholar] [CrossRef] [PubMed]
- Burlec, A.F.; Corciova, A.; Boev, M.; Batir-Marin, D.; Mircea, C.; Cioanca, O.; Danila, G.; Danila, M.; Bucur, A.F.; Hancianu, M. Current Overview of Metal Nanoparticles’ Synthesis, Characterization, and Biomedical Applications, with a Focus on Silver and Gold Nanoparticles. Pharmaceuticals 2023, 16, 1410. [Google Scholar] [CrossRef] [PubMed]
- Avino, A.; Marina, C.; Balcangiu-Stroescu, A.E.; Tanasescu, M.D.; Balan, D.G.; Timofte, D.; Stoicescu, S.; Hariga, C.S.; Ionescu, D. Our Expe-rience in Skin Grafting and Silver Dressing for Venous Leg Ulcers. Rev. Chim. 2019, 70, 742–744. [Google Scholar] [CrossRef]
- Kaiser, K.G.; Delattre, V.; Frost, V.J.; Buck, G.W.; Phu, J.V.; Fernandez, T.G.; Pavel, I.E. Nanosilver: An Old Antibacterial Agent with Great Promise in the Fight against Antibiotic Resistance. Antibiotics 2023, 12, 1264. [Google Scholar] [CrossRef] [PubMed]
- Mihai, M.M.; Dima, M.B.; Dima, B.; Holban, A.M. Nanomaterials for Wound Healing and Infection Control. Materials 2019, 12, 2176. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-C.; Lee, H.-C.; Chen, C.-L.; Kuo, M.-C.; Ramachandran, S.; Chen, R.-F.; Kuo, Y.-R. The Effects of Silver-Releasing Foam Dressings on Diabetic Foot Ulcer Healing. J. Clin. Med. 2021, 10, 1495. [Google Scholar] [CrossRef]
- Varela, P.; Marlinghaus, L.; Sartori, S.; Viebahn, R.; Salber, J.; Ciardelli, G. Response of Human Macrophages to Clinically Applied Wound Dressings Loaded With Silver. Front. Bioeng. Biotechnol. 2020, 8, 124. [Google Scholar] [CrossRef]
- Hesketh, M.; Sahin, K.B.; West, Z.E.; Murray, R.Z. Macrophage Phenotypes Regulate Scar Formation and Chronic Wound Healing. Int. J. Mol. Sci. 2017, 18, 1545. [Google Scholar] [CrossRef]
- You, C.; Zhu, Z.; Wang, S.; Wang, X.; Han, C.; Shao, H. Nanosilver alleviates foreign body reaction and facilitates wound repair by regulating macrophage polarization. J. Zhejiang Univ. Sci. B 2023, 24, 510–523. [Google Scholar] [CrossRef]
- Singh, M.; Thakur, V.; Kumar, V.; Raj, M.; Gupta, S.; Devi, N.; Upadhyay, S.K.; Macho, M.; Banerjee, A.; Ewe, D.; et al. Silver Nanoparticles and Its Mechanistic Insight for Chronic Wound Healing: Review on Recent Progress. Molecules 2022, 27, 5587. [Google Scholar] [CrossRef] [PubMed]
- Khansa, I.; Schoenbrunner, A.R.; Kraft, C.T.; Janis, J.E.M. Silver in Wound Care—Friend or Foe?: A Comprehensive Review. Plast. Reconstr. Surg. Glob. Open 2019, 7, e2390. [Google Scholar] [CrossRef] [PubMed]
- Troeman, D.P.R.; Hazard, D.; Timbermont, L.; Malhotra-Kumar, S.; van Werkhoven, C.H.; Wolkewitz, M.; Ruzin, A.; Goossens, H.; Bonten, M.J.M.; Harbarth, S.; et al. Postoperative Staphylococcus aureus Infections in Patients With and Without Preoperative Colonization. JAMA Netw. Open 2023, 6, e2339793. [Google Scholar] [CrossRef]
- Shiels, S.M.; Sgromolo, N.M.; Wenke, J.C. Negative pressure wound therapy does not diminish efficacy of topical antibiotic powder in a preclinical contaminated wound model. Bone Jt. Res. 2021, 10, 149–155. [Google Scholar] [CrossRef]
- Hahn, H.M.; Lee, I.J.; Woo, K.-J.; Park, B.Y. Silver-Impregnated Negative-Pressure Wound Therapy for the Treatment of Lower-Extremity Open Wounds: A Prospective Randomized Clinical Study. Adv. Ski. Wound Care 2019, 32, 370–377. [Google Scholar] [CrossRef]
- Vieira, A.L.G.; Stocco, J.G.D.; Ribeiro, A.C.G.; Frantz, C.V. Dressings used to prevent surgical site infection in the postoperative period of cardiac surgery: Integrative review. Rev. Esc. Enferm. USP 2018, 52, e03393. [Google Scholar] [CrossRef]
- Sapino, G.; Lanz, L.; Roesti, A.; Guillier, D.; Deglise, S.; De Santis, G.; Raffoul, W.; di Summa, P. One-Stage Coverage of Leg Region Defects with STSG Combined with VAC Dressing Improves Early Patient Mobilisation and Graft Take: A Comparative Study. J. Clin. Med. 2022, 11, 3305. [Google Scholar] [CrossRef] [PubMed]
- Mo, R.; Ma, Z.; Chen, C.; Meng, X.; Tan, Q. Short- and long-term efficacy of negative-pressure wound therapy in split-thickness skin grafts: A retrospective study. Ann. Palliat. Med. 2021, 10, 2935–2947. [Google Scholar] [CrossRef]
- Blume, P.A.; Key, J.J.; Thakor, P.; Thakor, S.; Sumpio, B. Retrospective evaluation of clinical outcomes in subjects with split-thickness skin graft: Comparing V.A.C.® therapy and conventional therapy in foot and ankle reconstructive surgeries. Int. Wound J. 2010, 7, 480–487. [Google Scholar] [CrossRef]
- Sowers, K.M.; Hayden, M.R. Calcific Uremic Arteriolopathy: Pathophysiology, Reactive Oxygen Species and Therapeutic Approaches. Oxidative Med. Cell. Longev. 2010, 3, 109–121. [Google Scholar] [CrossRef]
- Vinereanu, I.-V.; Peride, I.; Niculae, A.; Tiron, A.T.; Caragheorgheopol, A.; Manda, D.; Checherita, I.A. The Relationship between Advanced Oxidation Protein Products, Vascular Calcifications and Arterial Stiffness in Predialysis Chronic Kidney Disease Patients. Medicina 2021, 57, 452. [Google Scholar] [CrossRef] [PubMed]
- Galperin, T.A.; Cronin, A.J.; Leslie, K.S. Cutaneous Manifestations of ESRD. Clin. J. Am. Soc. Nephrol. 2014, 9, 201–218. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Shen, G.; Hao, C. Negative pressure wound therapy (NPWT) is superior to conventional moist dressings in wound bed preparation for diabetic foot ulcers: A randomized controlled trial. SciVee 2023, 44, 1020–1029. [Google Scholar] [CrossRef] [PubMed]
- Bordianu, A.; Bobircă, F.; Pătraşcu, T. Skin Grafting in the Treatment of Diabetic Foot Soft Tissue Defects. Chirurgia 2018, 113, 644–650. [Google Scholar] [CrossRef]
- Turissini, J.D.; Elmarsafi, T.; Evans, K.K.; Kim, P.J. Major Risk Factors Contributing to Split Thickness Skin Graft Failure. Georget. Med. Rev. 2019, 3, 7755. [Google Scholar] [CrossRef]
- Lim, K.; Lim, X.; Hong, Q.; Yong, E.; Chandrasekar, S.; Tan, G.W.L.; Lo, Z.J. Use of home negative pressure wound therapy in peripheral artery disease and diabetic limb salvage. Int. Wound J. 2020, 17, 531–539. [Google Scholar] [CrossRef]
- Zhao, C.X.; Scher, M.; Hanks, J.E.; McLean, S.A. Full-thickness versus split-thickness skin graft reconstruction of scalp defects with and without calvarium exposure. JPRA Surg. 2023. [Google Scholar] [CrossRef]
- Liao, C.D.; Deccico, J.-B.; Cuzzone, D.; George, F.; Xipoleas, G.D. Interrupting Negative-pressure Wound Therapy with Dakin’s Solution Is Associated with Increased Skin Graft Survival. Plast. Reconstr. Surg. Glob. Open 2022, 10, e4724. [Google Scholar] [CrossRef]
- Nakamura, Y.; Fujisawa, Y.; Ishitsuka, Y.; Tanaka, R.; Maruyama, H.; Okiyama, N.; Watanabe, R.; Fujimoto, M. Negative-pressure closure was superior to tie-over technique for stabilization of split-thickness skin graft in large or muscle-exposing defects: A retrospective study. J. Dermatol. 2018, 45, 1207–1210. [Google Scholar] [CrossRef]
- Raffetto, J.D.; Ligi, D.; Maniscalco, R.; Khalil, R.A.; Mannello, F. Why Venous Leg Ulcers Have Difficulty Healing: Overview on Pathophysiology, Clinical Consequences, and Treatment. J. Clin. Med. 2020, 10, 29. [Google Scholar] [CrossRef]
- Maduba, C.C.; Nnadozie, U.U.; Modekwe, V.I.; Onah, I.I. Split Skin Graft Take in Leg Ulcers: Conventional Dressing Versus Locally Adapted Negative Pressure Dressing. J. Surg. Res. 2020, 251, 296–302. [Google Scholar] [CrossRef]
- Kantak, N.A.; Mistry, R.; Varon, D.E.; Halvorson, E.G. Negative Pressure Wound Therapy for Burns. Clin. Plast. Surg. 2017, 44, 671–677. [Google Scholar] [CrossRef]
- Salvo, P.; Dini, V.; Kirchhain, A.; Janowska, A.; Oranges, T.; Chiricozzi, A.; Lomonaco, T.; Di Francesco, F.; Romanelli, M. Sensors and Biosensors for C-Reactive Protein, Temperature and pH, and Their Applications for Monitoring Wound Healing: A Review. Sensors 2017, 17, 2952. [Google Scholar] [CrossRef] [PubMed]
- Beiner, E.; Miguel, S.B.; Friederich, H.-C.; Tesarz, J. PerPAIN Consortium Elevated high sensitive C-reactive protein in fibromyalgia. Front. Psychiatry 2023, 14, 1237518. [Google Scholar] [CrossRef] [PubMed]
- Legendre, C.; Debure, C.; Meaume, S.; Lok, C.; Golmard, J.L.; Senet, P. Impact of protein deficiency on venous ulcer healing. J. Vasc. Surg. 2008, 48, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Rashid, H.U.; Rashid, M.; Sarwar, S.U.R.; Khan, I.; Khan, N.; Bibi, N. Negative Pressure Wound Therapy (NPWT): Our Experience in Pakistan With Locally Made Dressing. Cureus 2020, 12, e9464. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Yang, F.; Li, Z.; Yi, C.; Bai, X. A prospective pilot study to evaluate wound outcomes and levels of serum C-reactive protein and interleukin-6 in the wound fluid of patients with trauma-related chronic wounds. Ostomy Wound Manag. 2014, 60, 30–37. [Google Scholar]


| Silver Nanoparticle Sheet | NPWT+AgNPs | Total | ||
|---|---|---|---|---|
| Defect origin | Postexcisional defect | 17 | 11 | 28 |
| Ulceration debridement | 24 | 28 | 52 | |
| Graft area (cm2) | <150 cm2 | 14 | 11 | 25 |
| 150–500 cm2 | 22 | 20 | 42 | |
| >500 cm2 | 5 | 8 | 13 | |
| Cardiovascular comorbidities | No | 8 | 14 | 22 |
| Yes | 33 | 25 | 58 | |
| Diabetes mellitus | No | 10 | 9 | 19 |
| Yes | 31 | 30 | 61 |
| Mean | Std. Deviation | p-Value | ||
|---|---|---|---|---|
| Graft take after 7 days (%) | Silver nanoparticle sheet | 74.88 | 11.374 | <0.001 |
| NPWT+AgNPs | 85.00 | 8.959 | ||
| Graft take after 10 days (%) | Silver nanoparticle sheet | 87.20 | 9.686 | <0.001 |
| NPWT+AgNPs | 97.56 | 4.115 | ||
| Graft take after 14 days (%) | Silver nanoparticle sheet | 93.41 | 8.905 | <0.001 |
| NPWT+AgNPs | 99.62 | 1.771 |
| No CV Disease | CV Disease | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | Std. Deviation | p Value | Effect Size | Mean | Std. Deviation | p Value | Effect Size | ||
| Graft take after 7 days (%) | Silver nanoparticle sheet | 79.38 | 10.501 | 0.718 | −0.16246 | 73.79 | 11.459 | <0.001 | −1.35267 |
| NPWT+AgNPs | 81.07 | 10.411 | 87.20 | 7.371 | |||||
| Graft take after 10 days (%) | Silver nanoparticle sheet | 90.63 | 9.039 | 0.064 | −0.86838 | 86.36 | 9.785 | <0.001 | −1.52861 |
| NPWT+AgNPs | 96.43 | 4.972 | 98.20 | 3.500 | |||||
| Graft take after 14 days (%) | Silver nanoparticle sheet | 93.75 | 7.906 | 0.092 | −1.07499 | 93.33 | 9.242 | <0.001 | −0.92157 |
| NPWT+AgNPs | 99.29 | 2.673 | 99.80 | 1.000 | |||||
| No Diabetes | Diabetes Mellitus | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | Std. Deviation | p Value | Effect Size | Mean | Std. Deviation | p Value | Effect Size | ||
| Graft take after 7 days (%) | Silver nanoparticle sheet | 84.00 | 8.756 | 0.098 | −0.8043 | 71.94 | 10.621 | <0.001 | −1.17855 |
| NPWT+AgNPs | 91.11 | 8.937 | 83.17 | 8.251 | |||||
| Graft take after 10 days (%) | Silver nanoparticle sheet | 96.00 | 4.595 | 0.304 | −0.4871 | 84.35 | 9.196 | <0.001 | −1.82809 |
| NPWT+AgNPs | 98.33 | 5.000 | 97.33 | 3.880 | |||||
| Graft take after 14 days (%) | Silver nanoparticle sheet | 100.00 | 0.000 | 0.347 | 0.4859 | 91.29 | 9.307 | <0.001 | −1.23936 |
| NPWT+AgNPs | 99.44 | 1.667 | 99.67 | 1.826 | |||||
| <150 cm2 | 150–500 cm2 | >500 cm2 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | p Value | Effect Size | Mean | p Value | Effect Size | Mean | p Value | Effect Size | ||
| Graft take after 7 days (%) | Silver nanoparticle sheet | 84.29 | 0.068 | −0.77274 | 72.05 | 0.000 | −1.49177 | 61.00 | 0.000 | −3.50173 |
| NPWT+AgNPs | 89.55 | 86.25 | 75.63 | |||||||
| Graft take after 10 days (%) | Silver nanoparticle sheet | 95.00 | 0.004 | −1.21345 | 85.23 | 0.000 | −1.81813 | 74.00 | 0.000 | −4.34509 |
| NPWT+AgNPs | 99.55 | 97.75 | 94.38 | |||||||
| Graft take after 14 days (%) | Silver nanoparticle sheet | 98.57 | 0.218 | −0.46041 | 93.18 | 0.003 | −0.99211 | 80.00 | 0.002 | −4.90153 |
| NPWT+AgNPs | 100.00 | 99.50 | 99.38 | |||||||
| Postexcisional | Ulceration Debridement | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | Std. Deviation | p Value | Effect Size | Mean | Std. Deviation | p Value | Effect Size | ||
| Graft take after 7 days (%) | Silver nanoparticle sheet | 82.65 | 7.729 | 0.000 | −1.60545 | 69.38 | 10.354 | 0.000 | −1.3342 |
| NPWT+AgNPs | 93.18 | 4.045 | 81.79 | 8.302 | |||||
| Graft take after 10 days (%) | Silver nanoparticle sheet | 93.53 | 5.524 | 0.000 | −1.49319 | 82.71 | 9.553 | 0.000 | −1.90856 |
| NPWT+AgNPs | 100.00 | 0.000 | 96.61 | 4.524 | |||||
| Graft take after 14 days (%) | Silver nanoparticle sheet | 98.82 | 2.811 | 0.104 | −0.53342 | 89.58 | 9.771 | 0.000 | −1.45283 |
| NPWT+AgNPs | 100.00 | 0.000 | 99.46 | 2.081 | |||||
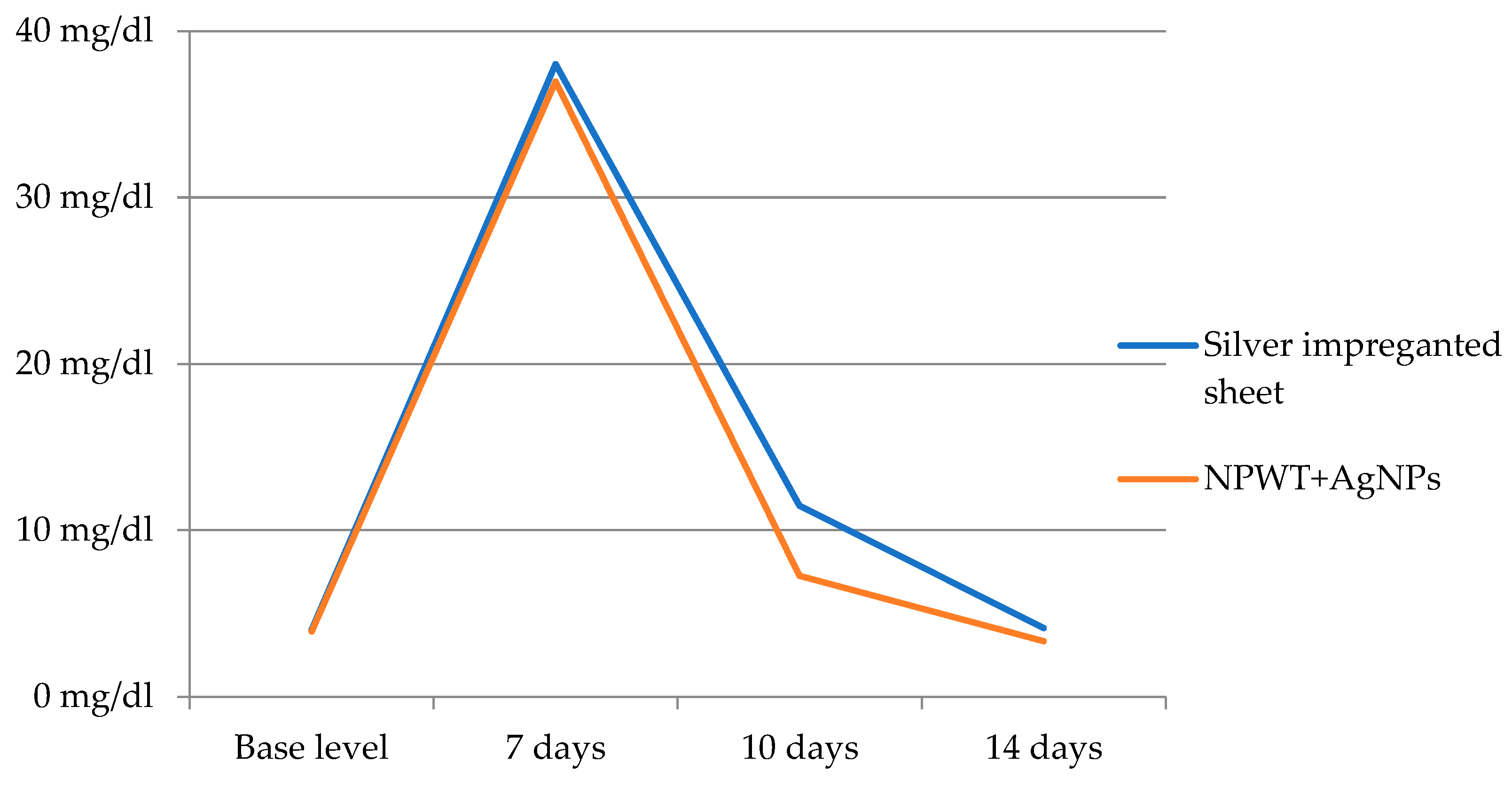
| Mean | Std. Deviation | p-Value | ||
|---|---|---|---|---|
| CRP before graft (mg/dL) | Silver nanoparticle sheet | 4.020 | 2.8149 | 0.868 |
| NPWT+AgNPs | 3.915 | 2.7547 | ||
| CRP after 7 days (mg/dL) | Silver nanoparticle sheet | 38.02 | 5.985 | 0.481 |
| NPWT+AgNPs | 36.97 | 7.235 | ||
| CRP after 10 days (mg/dL) | Silver nanoparticle sheet | 11.473 | 6.5328 | 0.001 |
| NPWT+AgNPs | 7.267 | 3.9482 | ||
| CRP after 14 days (mg/dL) | Silver nanoparticle sheet | 4.129 | 2.4921 | 0.145 |
| NPWT+AgNPs | 3.338 | 2.3040 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abu-Baker, A.; Țigăran, A.-E.; Peligrad, T.; Ion, D.-E.; Gheoca-Mutu, D.-E.; Avino, A.; Hariga, C.-S.; Moraru, O.E.; Răducu, L.; Jecan, R.-C. Exploring an Innovative Approach: Integrating Negative-Pressure Wound Therapy with Silver Nanoparticle Dressings in Skin Graft Procedures. J. Pers. Med. 2024, 14, 206. https://doi.org/10.3390/jpm14020206
Abu-Baker A, Țigăran A-E, Peligrad T, Ion D-E, Gheoca-Mutu D-E, Avino A, Hariga C-S, Moraru OE, Răducu L, Jecan R-C. Exploring an Innovative Approach: Integrating Negative-Pressure Wound Therapy with Silver Nanoparticle Dressings in Skin Graft Procedures. Journal of Personalized Medicine. 2024; 14(2):206. https://doi.org/10.3390/jpm14020206
Chicago/Turabian StyleAbu-Baker, Abdalah, Andrada-Elena Țigăran, Teodora Peligrad, Daniela-Elena Ion, Daniela-Elena Gheoca-Mutu, Adelaida Avino, Cristian-Sorin Hariga, Oriana Elena Moraru, Laura Răducu, and Radu-Cristian Jecan. 2024. "Exploring an Innovative Approach: Integrating Negative-Pressure Wound Therapy with Silver Nanoparticle Dressings in Skin Graft Procedures" Journal of Personalized Medicine 14, no. 2: 206. https://doi.org/10.3390/jpm14020206
APA StyleAbu-Baker, A., Țigăran, A.-E., Peligrad, T., Ion, D.-E., Gheoca-Mutu, D.-E., Avino, A., Hariga, C.-S., Moraru, O. E., Răducu, L., & Jecan, R.-C. (2024). Exploring an Innovative Approach: Integrating Negative-Pressure Wound Therapy with Silver Nanoparticle Dressings in Skin Graft Procedures. Journal of Personalized Medicine, 14(2), 206. https://doi.org/10.3390/jpm14020206







