Genetic Approaches for the Treatment of Giant Axonal Neuropathy
Abstract
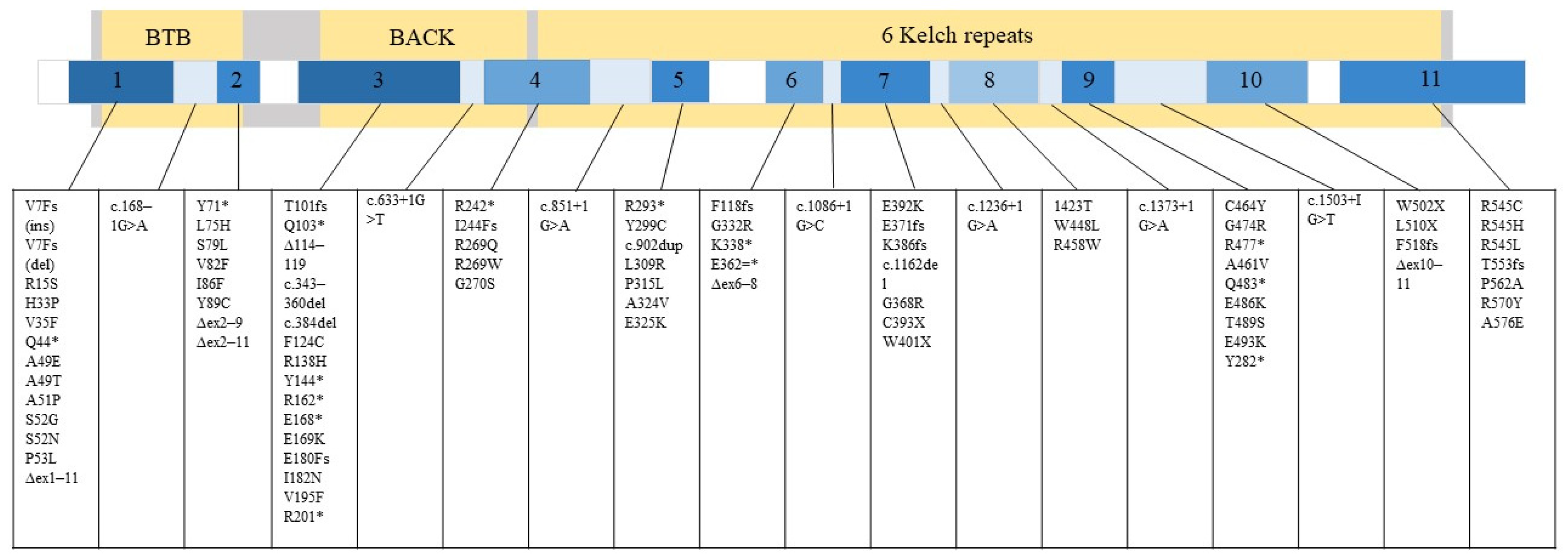
1. Introduction
2. Clinical Heterogeneity in Persons with GAN
3. Gigaxonin Plays an Important Role in IF Homeostasis
4. In Vivo Gene Therapy for GAN Using AAVs
5. Phase I Clinical Trial of GAN
6. Concerns about AAV9-Mediated Gene Replacement Therapy for GAN
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kang, J.J.; Liu, I.Y.; Wang, M.B.; Srivatsan, E.S. A Review of Gigaxonin Mutations in Giant Axonal Neuropathy (GAN) and Cancer. Hum. Genet. 2016, 135, 675–684. [Google Scholar] [CrossRef] [PubMed]
- Asbury, A.K.; Gale, M.K.; Cox, S.C.; Baringer, J.R.; Berg, B.O. Giant Axonal Neuropathy? A Unique Case with Segmental Neurofilamentous Masses. Acta Neuropathol. 1972, 20, 237–247. [Google Scholar] [CrossRef]
- Berg, B.O.; Rosenberg, S.H.; Asbury, A.K. Giant Axonal Neuropathy. Pediatrics 1972, 49, 894–899. [Google Scholar] [CrossRef] [PubMed]
- Johnson-Kerner, B.L.; Roth, L.; Greene, J.P.; Wichterle, H.; Sproule, D.M. Giant Axonal Neuropathy: An Updated Perspective on Its Pathology and Pathogenesis. Muscle Nerve 2014, 50, 467–476. [Google Scholar] [CrossRef]
- Bruno, C.; Bertini, E.; Federico, A.; Tonoli, E.; Lispi, M.L.; Cassandrini, D.; Pedemonte, M.; Santorelli, F.M.; Filocamo, M.; Dotti, M.T.; et al. Clinical and Molecular Findings in Patients with Giant Axonal Neuropathy (GAN). Neurology 2004, 62, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Allen, E.; Ding, J.; Wang, W. Giant Axonal Neuropathy. Cell. Mol. Life Sci. 2007, 64, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Lescouzères, L.; Bomont, P. E3 Ubiquitin Ligases in Neurological Diseases: Focus on Gigaxonin and Autophagy. Front. Physiol. 2020, 11, 1022. [Google Scholar] [CrossRef]
- Fokkema, I.F.A.C.; den Dunnen, J.T.; Taschner, P.E.M. LOVD: Easy Creation of a Locus-Specific Sequence Variation Database Using an “LSDB-in-a-Box” Approach. Hum. Mutat. 2005, 26, 63–68. [Google Scholar] [CrossRef]
- Akagi, M.; Mohri, I.; Iwatani, Y.; Kagitani-Shimono, K.; Okinaga, T.; Sakai, N.; Ozono, K.; Taniike, M. Clinicogenetical Features of a Japanese Patient with Giant Axonal Neuropathy. Brain Dev. 2012, 34, 156–162. [Google Scholar] [CrossRef]
- Abu-Rashid, M.; Mahajnah, M.; Jaber, L.; Kornreich, L.; Bar-On, E.; Basel-Vanagaite, L.; Soffer, D.; Koenig, M.; Straussberg, R. A Novel Mutation in the GAN Gene Causes an Intermediate Form of Giant Axonal Neuropathy in an Arab–Israeli Family. Eur. J. Paediatr. Neurol. 2013, 17, 259–264. [Google Scholar] [CrossRef]
- Bomont, P.; Ioos, C.; Yalcinkaya, C.; Korinthenberg, R.; Vallat, J.; Assami, S.; Munnich, A.; Chabrol, B.; Kurlemann, G.; Tazir, M.; et al. Identification of Seven Novel Mutations in The GAN Gene. Hum. Mutat. 2003, 21, 446. [Google Scholar] [CrossRef] [PubMed]
- Buysse, K.; Vergult, S.; Mussche, S.; Groote, C.C.; Speleman, F.; Menten, B.; Lissens, W.; Van Coster, R. Giant Axonal Neuropathy Caused by Compound Heterozygosity for a Maternally Inherited Microdeletion and a Paternal Mutation within the GAN Gene. Am. J. Med. Genet. Part A 2010, 152A, 2802–2804. [Google Scholar] [CrossRef]
- Edem, P.; Karakaya, M.; Wirth, B.; Okur, T.D.; Yiş, U. Giant Axonal Neuropathy: A Differential Diagnosis of Consideration. Turk. J. Pediatr. 2019, 61, 275. [Google Scholar] [CrossRef] [PubMed]
- Koop, O.; Schirmacher, A.; Nelis, E.; Timmerman, V.; De Jonghe, P.; Ringelstein, B.; Rasic, V.M.; Evrard, P.; Gärtner, J.; Claeys, K.G.; et al. Genotype–Phenotype Analysis in Patients with Giant Axonal Neuropathy (GAN). Neuromuscul. Disord. 2007, 17, 624–630. [Google Scholar] [CrossRef] [PubMed]
- Kuhlenbäumer, G.; Young, P.; Oberwittler, C.; Hünermund, G.; Schirmacher, A.; Domschke, K.; Ringelstein, B.; Stögbauer, F. Giant Axonal Neuropathy (GAN): Case Report and Two Novel Mutations in the Gigaxonin Gene. Neurology 2002, 58, 1273–1276. [Google Scholar] [CrossRef]
- Miyatake, S.; Tada, H.; Moriya, S.; Takanashi, J.; Hirano, Y.; Hayashi, M.; Oya, Y.; Nakashima, M.; Tsurusaki, Y.; Miyake, N.; et al. Atypical Giant Axonal Neuropathy Arising from a Homozygous Mutation by Uniparental Isodisomy. Clin. Genet. 2015, 87, 395–397. [Google Scholar] [CrossRef]
- Mohammad, S.S.; Lau, C.; Burke, C.; McCallum, N.; Robertson, T. Giant Axonal Neuropathy Diagnosed on Skin Biopsy. J. Clin. Neurosci. 2014, 21, 865–867. [Google Scholar] [CrossRef]
- Nalini, A.; Gayathri, N.; Yasha, T.C.; Ravishankar, S.; Urtizberea, A.; Huehne, K.; Rautenstrauss, B. Clinical, Pathological and Molecular Findings in Two Siblings with Giant Axonal Neuropathy (GAN): Report from India. Eur. J. Med. Genet. 2008, 51, 426–435. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhao, D.; Wang, Z.; Zhang, W.; Lv, H.; Liu, X.; Meng, L.; Yuan, Y. Heterogeneity of Axonal Pathology in Chinese Patients with Giant Axonal Neuropathy. Muscle Nerve 2014, 50, 200–205. [Google Scholar] [CrossRef]
- Xu, X.; Yang, X.; Su, Z.; Wang, H.; Li, X.; Sun, C.; Wang, W.; Chen, Y.; Zhang, C.; Zhang, H.; et al. Identification of Novel Compound Heterozygous Mutations in the GAN Gene of a Chinese Patient Diagnosed With Giant Axonal Neuropathy. Front. Neurosci. 2020, 14, 85. [Google Scholar] [CrossRef]
- Leung, C.L.; Pang, Y.; Shu, C.; Goryunov, D.; Liem, R.K.H. Alterations in Lipid Metabolism Gene Expression and Abnormal Lipid Accumulation in Fibroblast Explants from Giant Axonal Neuropathy Patients. BMC Genet. 2007, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Tazir, M.; Nouioua, S.; Magy, L.; Huehne, K.; Assami, S.; Urtizberea, A.; Grid, D.; Hamadouche, T.; Rautenstrauss, B.; Vallat, J.-M. Phenotypic Variability in Giant Axonal Neuropathy. Neuromuscul. Disord. 2009, 19, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Normendez-Martínez, M.I.; Monterde-Cruz, L.; Martínez, R.; Marquez-Harper, M.; Esquitin-Garduño, N.; Valdes-Flores, M.; Casas-Avila, L.; de Leon-Suarez, V.P.; Romero-Díaz, V.J.; Hidalgo-Bravo, A. Two Novel Mutations in the GAN Gene Causing Giant Axonal Neuropathy. World J. Pediatr. 2018, 14, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.-P.; Zou, L.-P. Clinical and Genetic Studies in a Chinese Family With Giant Axonal Neuropathy. J. Child Neurol. 2009, 24, 1552–1556. [Google Scholar] [CrossRef] [PubMed]
- Echaniz-Laguna, A.; Cuisset, J.-M.; Guyant-Marechal, L.; Aubourg, P.; Kremer, L.; Baaloul, N.; Verloes, A.; Beladgham, K.; Perrot, J.; Francou, B.; et al. Giant Axonal Neuropathy: A Multicenter Retrospective Study with Genotypic Spectrum Expansion. Neurogenetics 2020, 21, 29–37. [Google Scholar] [CrossRef]
- Demir, E. Giant Axonal Neuropathy: Clinical and Genetic Study in Six Cases. J. Neurol. Neurosurg. Psychiatry 2005, 76, 825–832. [Google Scholar] [CrossRef]
- Aharoni, S.; Barwick, K.E.S.; Straussberg, R.; Harlalka, G.V.; Nevo, Y.; Chioza, B.A.; McEntagart, M.M.; Mimouni-Bloch, A.; Weedon, M.; Crosby, A.H. Novel Homozygous Missense Mutation in GAN Associated with Charcot-Marie-Tooth Disease Type 2 in a Large Consanguineous Family from Israel. BMC Med. Genet. 2016, 17, 82. [Google Scholar] [CrossRef]
- Cai, S.; Lin, J.; Liu, Y.-Q.; Lu, J.-H.; Zhao, C.-B. Giant Axonal Neuropathy with Unusual Neuroimagings Caused by Compound Heterozygous Mutations in GAN Gene. Chin. Med. J. (Engl.) 2018, 131, 2371–2372. [Google Scholar] [CrossRef]
- Garg, M.; Kulkarni, S.D.; Hegde, A.U.; Desai, M.; Sayed, R.J. Giant Axonal Neuropathy: Clinical, Radiological, and Genetic Features. Ann. Indian Acad. Neurol. 2018, 21, 304–308. [Google Scholar] [CrossRef]
- Houlden, H.; Groves, M.; Miedzybrodzka, Z.; Roper, H.; Willis, T.; Winer, J.; Cole, G.; Reilly, M.M. New Mutations, Genotype Phenotype Studies and Manifesting Carriers in Giant Axonal Neuropathy. J. Neurol. Neurosurg. Psychiatry 2007, 78, 1267–1270. [Google Scholar] [CrossRef]
- Xu, M.; Da, Y.; Liu, L.; Wang, F.; Jia, J. Giant Axonal Neuropathy Caused by a Novel Compound Heterozygous Mutation in the Gigaxonin Gene. J. Child Neurol. 2013, 28, 1316–1319. [Google Scholar] [CrossRef] [PubMed]
- Roth, L.A.; Johnson-Kerner, B.L.; Marra, J.D.; LaMarca, N.H.; Sproule, D.M. The Absence of Curly Hair Is Associated with a Milder Phenotype in Giant Axonal Neuropathy. Neuromuscul. Disord. 2014, 24, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Koichihara, R.; Saito, T.; Ishiyama, A.; Komaki, H.; Yuasa, S.; Saito, Y.; Nakagawa, E.; Sugai, K.; Shiihara, T.; Shioya, A.; et al. A Mild Case of Giant Axonal Neuropathy without Central Nervous System Manifestation. Brain Dev. 2016, 38, 350–353. [Google Scholar] [CrossRef] [PubMed]
- Incecik, F.; Herguner, O.M.; Ceylaner, S.; Zorludemir, S.; Altunbasak, S. Giant Axonal Disease: Report of Eight Cases. Brain Dev. 2015, 37, 803–807. [Google Scholar] [CrossRef]
- Boizot, A.; Talmat-Amar, Y.; Morrogh, D.; Kuntz, N.L.; Halbert, C.; Chabrol, B.; Houlden, H.; Stojkovic, T.; Schulman, B.A.; Rautenstrauss, B.; et al. The Instability of the BTB-KELCH Protein Gigaxonin Causes Giant Axonal Neuropathy and Constitutes a New Penetrant and Specific Diagnostic Test. Acta Neuropathol. Commun. 2014, 2, 47. [Google Scholar] [CrossRef]
- Bomont, P.; Cavalier, L.; Blondeau, F.; Ben Hamida, C.; Belal, S.; Tazir, M.; Demir, E.; Topaloglu, H.; Korinthenberg, R.; Tüysüz, B.; et al. The Gene Encoding Gigaxonin, a New Member of the Cytoskeletal BTB/Kelch Repeat Family, Is Mutated in Giant Axonal Neuropathy. Nat. Genet. 2000, 26, 370–374. [Google Scholar] [CrossRef]
- Roth, L.A.; Marra, J.D.; LaMarca, N.H.; Sproule, D.M. Measuring Disease Progression in Giant Axonal Neuropathy. J. Child Neurol. 2015, 30, 741–748. [Google Scholar] [CrossRef]
- Guo, Y.; Su, Q.; Zhu, X.; Wang, J.; Lou, Y.; Miao, P.; Wang, Y.; Zhang, B.; Jin, Y.; Gao, L.; et al. Giant Axonal Neuropathy (GAN) in an 8-year-old Girl Caused by a Homozygous Pathogenic Splicing Variant in GAN Gene. Am. J. Med. Genet. Part A 2022, 188, 836–846. [Google Scholar] [CrossRef]
- Bharucha-Goebel, D.X.; Norato, G.; Saade, D.; Paredes, E.; Biancavilla, V.; Donkervoort, S.; Kaur, R.; Lehky, T.; Fink, M.; Armao, D.; et al. Giant Axonal Neuropathy: Cross-Sectional Analysis of a Large Natural History Cohort. Brain 2021, 144, 3239–3250. [Google Scholar] [CrossRef]
- Kretzschmar, H.A.; Berg, B.O.; Davis, R.L. Giant Axonal Neuropathy. Acta Neuropathol. 1987, 73, 138–144. [Google Scholar] [CrossRef]
- Mohri, I.; Taniike, M.; Yoshikawa, H.; Higashiyama, M.; Itami, S.; Okada, S. A Case of Giant Axonal Neuropathy Showing Focal Aggregation and Hypophosphorylation of Intermediate Filaments. Brain Dev. 1998, 20, 594–597. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Hou, B.; Tang, H.; Cai, M.; Zheng, W. Investigation of the Effects of Laminin Present in the Basal Lamina of the Peripheral Nervous System on Axon Regeneration and Remyelination Using the Nerve Acellular Scaffold. J. Biomed. Mater. Res. Part A 2020, 108, 1673–1687. [Google Scholar] [CrossRef] [PubMed]
- Brenner, C.; Speck-Martins, C.E.; Farage, L.; Barker, P.B. 3T MR with Diffusion Tensor Imaging and Single-Voxel Spectroscopy in Giant Axonal Neuropathy. J. Magn. Reson. Imaging 2008, 28, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Majnemer, A.; Rosenblatt, B.; Watters, G.; Andermann, F. Giant Axonal Neuropathy: Central Abnormalities Demonstrated by Evoked Potentials. Ann. Neurol. 1986, 19, 394–396. [Google Scholar] [CrossRef] [PubMed]
- Gambarelli, D.; Hassoun, J.; Pellissier, J.F.; Livet, M.O.; Pinsard, N.; Toga, M. Giant Axonal Neuropathy. Involvement of Peripheral Nerve, Myenteric Plexus and Extra-Neuronal Area. Acta Neuropathol. 1977, 39, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Armao, D.; Bailey, R.M.; Bouldin, T.W.; Kim, Y.; Gray, S.J. Autonomic Nervous System Involvement in the Giant Axonal Neuropathy (GAN) KO Mouse: Implications for Human Disease. Clin. Auton. Res. 2016, 26, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Prineas, J.W.; Ouvrier, R.A.; Wright, R.G.; Walsh, J.C.; McLeod, J.G. Giant Axonal Neuropathy—A Generalized Disorder of Cytoplasmic Microfilament Formation. J. Neuropathol. Exp. Neurol. 1976, 35, 458–470. [Google Scholar] [CrossRef]
- Mahammad, S.; Murthy, S.N.P.; Didonna, A.; Grin, B.; Israeli, E.; Perrot, R.; Bomont, P.; Julien, J.-P.; Kuczmarski, E.; Opal, P.; et al. Giant Axonal Neuropathy–Associated Gigaxonin Mutations Impair Intermediate Filament Protein Degradation. J. Clin. Investig. 2013, 123, 1964–1975. [Google Scholar] [CrossRef]
- Yuan, A.; Rao, M.V.; Veeranna; Nixon, R.A. Neurofilaments and Neurofilament Proteins in Health and Disease. Cold Spring Harb. Perspect. Biol. 2017, 9, a018309. [Google Scholar] [CrossRef]
- Mussche, S.; De Paepe, B.; Smet, J.; Devreese, K.; Lissens, W.; Rasic, V.M.; Murnane, M.; Devreese, B.; Van Coster, R. Proteomic Analysis in Giant Axonal Neuropathy: New Insights into Disease Mechanisms. Muscle Nerve 2012, 46, 246–256. [Google Scholar] [CrossRef]
- Wang, W.; Ding, J.; Allen, E.; Zhu, P.; Zhang, L.; Vogel, H.; Yang, Y. Gigaxonin Interacts with Tubulin Folding Cofactor B and Controls Its Degradation through the Ubiquitin-Proteasome Pathway. Curr. Biol. 2005, 15, 2050–2055. [Google Scholar] [CrossRef] [PubMed]
- Bennett, E.J.; Rush, J.; Gygi, S.P.; Harper, J.W. Dynamics of Cullin-RING Ubiquitin Ligase Network Revealed by Systematic Quantitative Proteomics. Cell 2010, 143, 951–965. [Google Scholar] [CrossRef] [PubMed]
- Johnson-Kerner, B.L.; Garcia Diaz, A.; Ekins, S.; Wichterle, H. Kelch Domain of Gigaxonin Interacts with Intermediate Filament Proteins Affected in Giant Axonal Neuropathy. PLoS ONE 2015, 10, e0140157. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.D.; Lo, S.-C.; Sun, Z.; Habib, G.M.; Lieberman, M.W.; Hannink, M. Ubiquitination of Keap1, a BTB-Kelch Substrate Adaptor Protein for Cul3, Targets Keap1 for Degradation by a Proteasome-Independent Pathway. J. Biol. Chem. 2005, 280, 30091–30099. [Google Scholar] [CrossRef]
- Furukawa, M.; He, Y.J.; Borchers, C.; Xiong, Y. Targeting of Protein Ubiquitination by BTB–Cullin 3–Roc1 Ubiquitin Ligases. Nat. Cell Biol. 2003, 5, 1001–1007. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; Liu, Z.; Yang, Q. The Role of Ubiquitination and Deubiquitination in Cancer Metabolism. Mol. Cancer 2020, 19, 146. [Google Scholar] [CrossRef] [PubMed]
- Pintard, L.; Willems, A.; Peter, M. Cullin-Based Ubiquitin Ligases: Cul3–BTB Complexes Join the Family. EMBO J. 2004, 23, 1681–1687. [Google Scholar] [CrossRef]
- Ding, J.; Liu, J.-J.; Kowal, A.S.; Nardine, T.; Bhattacharya, P.; Lee, A.; Yang, Y. Microtubule-Associated Protein 1B. J. Cell Biol. 2002, 158, 427–433. [Google Scholar] [CrossRef]
- Allen, E.; Ding, J.; Wang, W.; Pramanik, S.; Chou, J.; Yau, V.; Yang, Y. Gigaxonin-Controlled Degradation of MAP1B Light Chain Is Critical to Neuronal Survival. Nature 2005, 438, 224–228. [Google Scholar] [CrossRef]
- Ding, J.; Allen, E.; Wang, W.; Valle, A.; Wu, C.; Nardine, T.; Cui, B.; Yi, J.; Taylor, A.; Jeon, N.L.; et al. Gene Targeting of GAN in Mouse Causes a Toxic Accumulation of Microtubule-Associated Protein 8 and Impaired Retrograde Axonal Transport. Hum. Mol. Genet. 2006, 15, 1451–1463. [Google Scholar] [CrossRef][Green Version]
- Johnson-Kerner, B.L.; Ahmad, F.S.; Diaz, A.G.; Greene, J.P.; Gray, S.J.; Samulski, R.J.; Chung, W.K.; Van Coster, R.; Maertens, P.; Noggle, S.A.; et al. Intermediate Filament Protein Accumulation in Motor Neurons Derived from Giant Axonal Neuropathy IPSCs Rescued by Restoration of Gigaxonin. Hum. Mol. Genet. 2015, 24, 1420–1431. [Google Scholar] [CrossRef] [PubMed]
- Scrivo, A.; Codogno, P.; Bomont, P. Gigaxonin E3 Ligase Governs ATG16L1 Turnover to Control Autophagosome Production. Nat. Commun. 2019, 10, 780. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.-H.; Hu, J.; Wu, J.; Huynh, D.T.; Smith, T.J.; Pan, S.; Bisnett, B.J.; Smith, A.B.; Lu, A.; Condon, B.M.; et al. Gigaxonin Glycosylation Regulates Intermediate Filament Turnover and May Impact Giant Axonal Neuropathy Etiology or Treatment. JCI Insight 2020, 5, e127751. [Google Scholar] [CrossRef]
- Bailey, R.M.; Armao, D.; Nagabhushan Kalburgi, S.; Gray, S.J. Development of Intrathecal AAV9 Gene Therapy for Giant Axonal Neuropathy. Mol. Ther. Methods Clin. Dev. 2018, 9, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Cleveland, D.W.; Yamanaka, K.; Bomont, P. Gigaxonin Controls Vimentin Organization through a Tubulin Chaperone-Independent Pathway. Hum. Mol. Genet. 2009, 18, 1384–1394. [Google Scholar] [CrossRef] [PubMed]
- Mussche, S.; Devreese, B.; Nagabhushan Kalburgi, S.; Bachaboina, L.; Fox, J.C.; Shih, H.-J.; Van Coster, R.; Samulski, R.J.; Gray, S.J. Restoration of Cytoskeleton Homeostasis After Gigaxonin Gene Transfer for Giant Axonal Neuropathy. Hum. Gene Ther. 2013, 24, 209–219. [Google Scholar] [CrossRef]
- Intrathecal Administration of ScAAV9/JeT-GAN for the Treatment of Giant Axonal Neuropathy. Available online: https://clinicaltrials.gov/ct2/show/NCT02362438 (accessed on 27 August 2022).
- Armao, D.; Bouldin, T.W.; Bailey, R.M.; Hooper, J.E.; Bharucha, D.X.; Gray, S.J. Advancing the Pathologic Phenotype of Giant Axonal Neuropathy: Early Involvement of the Ocular Lens. Orphanet J. Rare Dis. 2019, 14, 27. [Google Scholar] [CrossRef]
- Armao, D.; Bouldin, T.W.; Bailey, R.M.; Gray, S.J. Extensive Rod and Cone Photoreceptor-Cell Degeneration in Rat Models of Giant Axonal Neuropathy: Implications for Gene Therapy of Human Disease. Ophthalmic Genet. 2021, 42, 600–603. [Google Scholar] [CrossRef]
- Mendell, J.R.; Al-Zaidy, S.A.; Rodino-Klapac, L.R.; Goodspeed, K.; Gray, S.J.; Kay, C.N.; Boye, S.L.; Boye, S.E.; George, L.A.; Salabarria, S.; et al. Current Clinical Applications of In Vivo Gene Therapy with AAVs. Mol. Ther. 2021, 29, 464–488. [Google Scholar] [CrossRef]
- Bailey, R.M.; Rozenberg, A.; Gray, S.J. Comparison of High-Dose Intracisterna Magna and Lumbar Puncture Intrathecal Delivery of AAV9 in Mice to Treat Neuropathies. Brain Res. 2020, 1739, 146832. [Google Scholar] [CrossRef]
- Hordeaux, J.; Buza, E.L.; Dyer, C.; Goode, T.; Mitchell, T.W.; Richman, L.; Denton, N.; Hinderer, C.; Katz, N.; Schmid, R.; et al. Adeno-Associated Virus-Induced Dorsal Root Ganglion Pathology. Hum. Gene Ther. 2020, 31, 808–818. [Google Scholar] [CrossRef] [PubMed]
- Mullard, A. Gene Therapy Community Grapples with Toxicity Issues, as Pipeline Matures. Nat. Rev. Drug Discov. 2021, 20, 804–805. [Google Scholar] [CrossRef]
- Opal, P. GAN-Related Neurodegeneration; University of Washington, Seattle: Seattle, WA, USA, 1993. [Google Scholar]
- Jaklevic, M.C. Advice for N-of-1 Drug Developers. JAMA 2021, 325, 613. [Google Scholar] [CrossRef]
- Synofzik, M.; van Roon-Mom, W.M.C.; Marckmann, G.; van Duyvenvoorde, H.A.; Graessner, H.; Schüle, R.; Aartsma-Rus, A. Preparing N-of-1 Antisense Oligonucleotide Treatments for Rare Neurological Diseases in Europe: Genetic, Regulatory, and Ethical Perspectives. Nucleic Acid Ther. 2022, 32, 83–94. [Google Scholar] [CrossRef]
- Jensen, T.L.; Gøtzsche, C.R.; Woldbye, D.P.D. Current and Future Prospects for Gene Therapy for Rare Genetic Diseases Affecting the Brain and Spinal Cord. Front. Mol. Neurosci. 2021, 14, 695937. [Google Scholar] [CrossRef]
- Croci, S.; Carriero, M.L.; Capitani, K.; Daga, S.; Donati, F.; Papa, F.T.; Frullanti, E.; Lopergolo, D.; Lamacchia, V.; Tita, R.; et al. AAV-Mediated FOXG1 Gene Editing in Human Rett Primary Cells. Eur. J. Hum. Genet. 2020, 28, 1446–1458. [Google Scholar] [CrossRef]
- Zielonka, D.; Mielcarek, M.; Landwehrmeyer, G.B. Update on Huntington’s Disease: Advances in Care and Emerging Therapeutic Options. Parkinsonism Relat. Disord. 2015, 21, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Wild, E.J.; Tabrizi, S.J. Targets for Future Clinical Trials in Huntington’s Disease: What’s in the Pipeline? Mov. Disord. 2014, 29, 1434–1445. [Google Scholar] [CrossRef] [PubMed]
- Lubroth, P.; Colasante, G.; Lignani, G. In Vivo Genome Editing Therapeutic Approaches for Neurological Disorders: Where Are We in the Translational Pipeline? Front. Neurosci. 2021, 15, 632522. [Google Scholar] [CrossRef] [PubMed]
- Bennett, C.F.; Krainer, A.R.; Cleveland, D.W. Antisense Oligonucleotide Therapies for Neurodegenerative Diseases. Annu. Rev. Neurosci. 2019, 42, 385–406. [Google Scholar] [CrossRef]
- Aslesh, T.; Yokota, T. Restoring SMN Expression: An Overview of the Therapeutic Developments for the Treatment of Spinal Muscular Atrophy. Cells 2022, 11, 417. [Google Scholar] [CrossRef] [PubMed]
- Goodkey, K.; Aslesh, T.; Maruyama, R.; Yokota, T. Nusinersen in the Treatment of Spinal Muscular Atrophy. Methods Mol. Biol. 2018, 1828, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Finkel, R.S.; Mercuri, E.; Darras, B.T.; Connolly, A.M.; Kuntz, N.L.; Kirschner, J.; Chiriboga, C.A.; Saito, K.; Servais, L.; Tizzano, E.; et al. Nusinersen versus Sham Control in Infantile-Onset Spinal Muscular Atrophy. N. Engl. J. Med. 2017, 377, 1723–1732. [Google Scholar] [CrossRef]
- Li, Q. Nusinersen as a Therapeutic Agent for Spinal Muscular Atrophy. Yonsei Med. J. 2020, 61, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Aslesh, T.; Yokota, T. Development of Antisense Oligonucleotide Gapmers for the Treatment of Huntington’s Disease. Methods Mol. Biol. 2020, 2176, 57–67. [Google Scholar] [CrossRef] [PubMed]
| Diagnostic Findings and Testing | Comments |
|---|---|
| Clinical findings |
|
| Electron Microscopy | Abnormally large axons in their paranodal junction and decreased axonal myelination in sural nerve and skin/muscle fiber biopsy |
| Electrophysiology | NCS: Generally normal to moderately reduced nerve conduction velocity. Can prolong, even to demyelinated range EMG: Severely reduced compound motor action potentials and absent sensory nerve action potentials of distal limbs. Sensory evoked responses (EEG): Latency to deficit in auditory brainstem evoked, visual evoked, and somatosensory evoked responses Spikes and sharp waves (EEG): Epileptiform transient discharges in the form of focal spikes and sharp waves (regardless of presence or absence of a history of seizures) |
| Neuroimaging | MRI: Classical GAN phenotype Hyperintense signal in cerebellar white matter surrounding the dentate nucleus. High T2-weighted sequences (and decreased T1 signal) in anterior and posterior periventricular regions and cerebellar white matter. Increased T2-weighted signals also occurs in the internal capsule, thalamus, and brainstem Milder phenotype Normal (most cases) to mild cerebral and cerebellar atrophy MRS: Increased Cho compounds and osmolyte myoinositol (Ins). Decreased N-acetylaspartate (NAA) |
| System/Concern | Feature | |
|---|---|---|
| Pyramidal signs (e.g., spasticity, Babinski signs) | ||
| Romberg sign | ||
| Cerebellar signs (e.g., ataxia, nystagmus, dysarthria) | ||
| Facial weakness | ||
| Bulbar weakness | ||
| Cranial nerve involvement | Ptosis | |
| Ophthalmoplegia | ||
| CNS | Optic atrophy | |
| Dysarthria | ||
| Hearing loss | ||
| Vision loss | ||
| Vertigo | ||
| Epilepsy | ||
| Intellectual disability/developmental delay | ||
| Cognitive decline | ||
| Dementia | ||
| Decreased muscle strength/tone | ||
| Motor neuropathy | Diffused muscle atrophy | |
| Distal motor weakness | ||
| PNS | Proximal motor weakness | |
| Sensory neuropathy | ||
| Flaccid paralysis | ||
| Areflexia | ||
| Constipation | ||
| Reflux | ||
| ANS | Regurgitation | |
| Diabetes | ||
| Renal tubular acidosis | ||
| Lactose intolerance | ||
| Kinky hair/ long eyelashes | ||
| Pale skin | ||
| High forehead | ||
| Body-wide/miscellaneous | Short stature | |
| Scoliosis (often associated with lumbar hyper-lordosis) | ||
| Tendon contracture | ||
| Foot deformity/arched feet | ||
| Precocious puberty | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shirakaki, S.; Roshmi, R.R.; Yokota, T. Genetic Approaches for the Treatment of Giant Axonal Neuropathy. J. Pers. Med. 2023, 13, 91. https://doi.org/10.3390/jpm13010091
Shirakaki S, Roshmi RR, Yokota T. Genetic Approaches for the Treatment of Giant Axonal Neuropathy. Journal of Personalized Medicine. 2023; 13(1):91. https://doi.org/10.3390/jpm13010091
Chicago/Turabian StyleShirakaki, Satomi, Rohini Roy Roshmi, and Toshifumi Yokota. 2023. "Genetic Approaches for the Treatment of Giant Axonal Neuropathy" Journal of Personalized Medicine 13, no. 1: 91. https://doi.org/10.3390/jpm13010091
APA StyleShirakaki, S., Roshmi, R. R., & Yokota, T. (2023). Genetic Approaches for the Treatment of Giant Axonal Neuropathy. Journal of Personalized Medicine, 13(1), 91. https://doi.org/10.3390/jpm13010091







