Application of Machine Learning Methods to Analyze Occurrence and Clinical Features of Ascending Aortic Dilatation in Patients with and without Bicuspid Aortic Valve
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Cohort
- Patient from the ECHO database whose treatment started after the 1st of January 2010 and ended before the 30th of November 2018;
- Diameter of the ascending aorta > 40 mm;
- For patients who underwent ECHO examination more than once during this period, only the first results of verified AD were included in the study. ECHO was most commonly performed in the following clinical situations: suspected cardiac etiology based on symptoms, signs or other testing; evaluation and follow-up of subjects with cardiovascular disease;
- Age ≥ 18 years old.
- 5.
- Patients whose treatment started before the 1st of January 2010 or ended after the 30th of November 2018;
- 6.
- Patients who did not have a complete data set.
2.2. Echocardiography
2.3. Statistical Methods
2.4. Data Preprocessing
2.5. Classification Model and Feature Importance
3. Results
4. Discussion
Study Limitation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AA | aortic aneurysm |
| AscAA | ascending aortic aneurysm |
| AS dpmax | antegrade gradient across the narrowed aortic valve, |
| AD | aortic dilatation |
| AR | aortic regurgitation, |
| AS | aortic stenosis, |
| AV | aortic valve |
| BAV | bicuspid aortic valve |
| BMI | body mass index, |
| CAD | coronary artery disease |
| CHD | congenital heart disease |
| COPD | chronic obstructive pulmonary disease, |
| DBP | diastolic blood pressure, |
| ECHO | echocardiographic |
| HF | heart failure |
| HP | hypertension |
| SBP | systolic blood pressure, |
| TAA | thoracic aortic aneurysms |
| VHD | valvular heart disease |
References
- Sampson, U.K.A.; Norman, P.E.; Fowkes, F.G.R.; Aboyans, V.; Song, Y.; Harrell, F.E.; Forouzanfar, M.H.; Naghavi, M.; Denenberg, J.O.; McDermott, M.M.; et al. Global and Regional Burden of Aortic Dissection and Aneurysms: Mortality Trends in 21 World Regions, 1990 to 2010. Glob. Heart 2014, 9, 171–180.e10. [Google Scholar] [CrossRef] [PubMed]
- Devereux, R.B.; De Simone, G.; Arnett, D.K.; Best, L.G.; Boerwinkle, E.; Howard, B.V.; Kitzman, D.; Lee, E.T.; Mosley, T.H.; Weder, A.; et al. Normal Limits in Relation to Age, Body Size and Gender of Two-Dimensional Echocardiographic Aortic Root Dimensions in Persons >15 Years of Age. Am. J. Cardiol. 2012, 110, 1189–1194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senser, E.M.; Misra, S.; Henkin, S. Thoracic Aortic Aneurysm: A Clinical Review. Cardiol. Clin. 2021, 39, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Gaudry, M.; Barral, P.A.; Blanchard, A.; Palazzolo, S.; Bolomey, S.; Omnes, V.; De Masi, M.; Carcopino-Tusoli, M.; Meyrignac, O.; Rousseau, H.; et al. Prevalence of Thoracic Aortic Aneurysms in Patients with Degenerative Abdominal Aortic Aneurysms: Results from the Prospective ACTA Study. Eur. J. Vasc. Endovasc. Surg. 2021, 61, 930–937. [Google Scholar] [CrossRef]
- Kälsch, H.; Lehmann, N.; Möhlenkamp, S.; Becker, A.; Moebus, S.; Schmermund, A.; Stang, A.; Mahabadi, A.A.; Mann, K.; Jöckel, K.H.; et al. Body-Surface Adjusted Aortic Reference Diameters for Improved Identification of Patients with Thoracic Aortic Aneurysms: Results from the Population-Based Heinz Nixdorf Recall Study. Int. J. Cardiol. 2013, 163, 72–78. [Google Scholar] [CrossRef]
- Rogers, I.S.; Massaro, J.M.; Truong, Q.A.; Mahabadi, A.A.; Kriegel, M.F.; Fox, C.S.; Thanassoulis, G.; Isselbacher, E.M.; Hoffmann, U.; O’Donnell, C.J. Distribution, Determinants, and Normal Reference Values of Thoracic and Abdominal Aortic Diameters by Computed Tomography (from the Framingham Heart Study). Am. J. Cardiol. 2013, 111, 1510–1516. [Google Scholar] [CrossRef] [Green Version]
- Lam, C.S.P.; Xanthakis, V.; Sullivan, L.M.; Lieb, W.; Aragam, J.; Redfield, M.M.; Mitchell, G.F.; Benjamin, E.J.; Vasan, R.S. Aortic Root Remodeling over the Adult Life Course: Longitudinal Data from the Framingham Heart Study. Circulation 2010, 122, 884–890. [Google Scholar] [CrossRef]
- Lenivtceva, I.; Panfilov, D.; Kopanitsa, G.; Kozlov, B. Aortic Risks Prediction Models after Cardiac Surgeries Using Integrated Data. J. Pers. Med. 2022, 12, 637. [Google Scholar] [CrossRef]
- Brady, A.R.; Thompson, S.G.; Fowkes, F.G.R.; Greenhalgh, R.M.; Powell, J.T. Abdominal Aortic Aneurysm Expansion: Risk Factors and Time Intervals for Surveillance. Circulation 2004, 110, 16–21. [Google Scholar] [CrossRef] [Green Version]
- Norman, P.; Spencer, C.A.; Lawrence-Brown, M.M.; Jamrozik, K. C-Reactive Protein Levels and the Expansion of Screen-Detected Abdominal Aortic Aneurysms in Men. Circulation 2004, 110, 862–866. [Google Scholar] [CrossRef] [Green Version]
- Golledge, J.; Muller, J.; Daugherty, A.; Norman, P. Abdominal Aortic Aneurysm: Pathogenesis and Implications for Management. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2605–2613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erbel, R.; Aboyans, V.; Boileau, C.; Bossone, E.; Di Bartolomeo, R.; Eggebrecht, H.; Evangelista, A.; Falk, V.; Frank, H.; Gaemperli, O.; et al. 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases: Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). Eur. Heart J. 2014, 35, 2873–2926. [Google Scholar] [PubMed] [Green Version]
- Siu, S.C.; Silversides, C.K. Bicuspid Aortic Valve Disease. J. Am. Coll. Cardiol. 2010, 55, 2789–2800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agnese, V.; Pasta, S.; Michelena, H.I.; Minà, C.; Romano, G.M.; Carerj, S.; Zito, C.; Maalouf, J.F.; Foley, T.A.; Raffa, G.; et al. Patterns of Ascending Aortic Dilatation and Predictors of Surgical Replacement of the Aorta: A Comparison of Bicuspid and Tricuspid Aortic Valve Patients over Eight Years of Follow-Up. J. Mol. Cell. Cardiol. 2019, 135, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Koechlin, L.; Macius, E.; Kaufmann, J.; Gahl, B.; Reuthebuch, O.; Eckstein, F.; Berdajs, D.A. Aortic Root and Ascending Aorta Dimensions in Acute Aortic Dissection. Perfusion 2020, 35, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Zafar, M.A.; Chen, J.F.; Wu, J.; Li, Y.; Papanikolaou, D.; Abdelbaky, M.; Vinholo, T.F.; Rizzo, J.A.; Ziganshin, B.A.; Mukherjee, S.K.; et al. Natural History of Descending Thoracic and Thoracoabdominal Aortic Aneurysms. J. Thorac. Cardiovasc. Surg. 2021, 161, 498–511.e1. [Google Scholar] [CrossRef]
- Kopanitsa, G. Integration of Hospital Information and Clinical Decision Support Systems to Enable the Reuse of Electronic Health Record Data. Methods Inf. Med. 2017, 56, 238–247. [Google Scholar] [CrossRef]
- Baumgartner, H.; Hung, J.; Bermejo, J.; Chambers, J.B.; Evangelista, A.; Griffin, B.P.; Iung, B.; Otto, C.M.; Pellikka, P.A.; Quiñones, M. Echocardiographic Assessment of Valve Stenosis: EAE/ASE Recommendations for Clinical Practice. J. Am. Soc. Echocardiogr. 2009, 22, 1–23. [Google Scholar] [CrossRef]
- Clouse, W.D.; Hallett, J.W.; Schaff, H.V.; Gayari, M.M.; Ilstrup, D.M.; Melton, L.J. Improved Prognosis of Thoracic Aortic Aneurysms. A Population-Based Study. J. Am. Med. Assoc. 1998, 280, 1926–1929. [Google Scholar] [CrossRef] [Green Version]
- Hiratzka, L.F.; Bakris, G.L.; Beckman, J.A.; Bersin, R.M.; Carr, V.F.; Casey, D.E.; Eagle, K.A.; Hermann, L.K.; Isselbacher, E.M.; Kazerooni, E.A.; et al. 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM Guidelines for the Diagnosis and Management of Patients With Thoracic Aortic Disease. Circulation 2010, 55, e27–e129. [Google Scholar] [CrossRef] [Green Version]
- Lang, R.M.; Bierig, M.; Devereux, R.B.; Flachskampf, F.A.; Foster, E.; Pellikka, P.A.; Picard, M.H.; Roman, M.J.; Seward, J.; Shanewise, J.; et al. Recommendations for Chamber Quantification. Eur. J. Echocardiogr. 2006, 7, 79–108. [Google Scholar] [CrossRef] [PubMed]
- Mosteller, R.D. Simplified Calculation of Body-Surface Area. N. Engl. J. Med. 1987, 317, 1098. [Google Scholar] [PubMed]
- Pedregosa FABIANPEDREGOSA, F.; Michel, V.; Grisel OLIVIERGRISEL, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Vanderplas, J.; Cournapeau, D.; Pedregosa, F.; Varoquaux, G.; et al. Scikit-Learn: Machine Learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar]
- Prokhorenkova, L.; Gusev, G.; Vorobev, A.; Dorogush, A.V.; Gulin, A. CatBoost: Unbiased Boosting with Categorical Features. NIPS’18 Adv. Neural Inf. Process. Syst. 2018, 31, 6639–6649. [Google Scholar] [CrossRef]
- Waskom, M.L. Seaborn: Statistical Data Visualization. J. Open Source Softw. 2021, 6, 3021. [Google Scholar] [CrossRef]
- Hunter, J.D. Matplotlib: A 2D Graphics Environment. Comput. Sci. Eng. 2007, 9, 90–95. [Google Scholar] [CrossRef]
- Chawla, N.V.; Bowyer, K.W.; Hall, L.O.; Kegelmeyer, W.P. SMOTE: Synthetic Minority over-Sampling Technique. J. Artif. Intell. Res. 2002, 16, 321–357. [Google Scholar] [CrossRef]
- Lundberg, S.M.; Lee, S.-I. A Unified Approach to Interpreting Model Predictions. NIPS’17 Adv. Neural Inf. Process. Syst. 2017, 30, 4768–4777. [Google Scholar] [CrossRef]
- Wang, S.W.; Huang, Y.B.; Huang, J.W.; Chiu, C.C.; Lai, W.T.; Chen, C.Y. Epidemiology, Clinical Features, and Prescribing Patterns of Aortic Aneurysm in Asian Population from 2005 to 2011. Medicine 2015, 94, e1716. [Google Scholar] [CrossRef]
- McPhee, J.T.; Hill, J.S.; Eslami, M.H. The Impact of Gender on Presentation, Therapy, and Mortality of Abdominal Aortic Aneurysm in the United States, 2001–2004. J. Vasc. Surg. 2007, 45, 891–899. [Google Scholar] [CrossRef] [Green Version]
- Nigam, V.; Srivastava, D. Notch1 Represses Osteogenic Pathways in Aortic Valve Cells. J. Mol. Cell. Cardiol. 2009, 47, 828–834. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boudoulas, K.D.; Wolfe, B.; Ravi, Y.; Lilly, S.; Nagaraja, H.N.; Sai-Sudhakar, C.B. The Aortic Stenosis Complex: Aortic Valve, Atherosclerosis, Aortopathy. J. Cardiol. 2015, 65, 377–382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.L.; Wang, Y.; Liao, M.; Wemmelund, H.; Ren, J.; Fernandes, C.; Zhou, Y.; Sukhova, G.K.; Lindholt, J.S.; Johnsen, S.P.; et al. Allergic Lung Inflammation Aggravates Angiotensin II-Induced Abdominal Aortic Aneurysms in Mice. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 69–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosa, M.; Chignon, A.; Li, Z.; Boulanger, M.C.; Arsenault, B.J.; Bossé, Y.; Thériault, S.; Mathieu, P. A Mendelian Randomization Study of IL6 Signaling in Cardiovascular Diseases, Immune-Related Disorders and Longevity. NPJ Genom. Med. 2019, 4, 23. [Google Scholar] [CrossRef]
- Nienaber, C.A. Diabetes Mellitus and Thoracic Aortic Disease: Are People With Diabetes Mellitus Protected From Acute Aortic Dissection? J. Am. Heart Assoc. 2012, 1, e001404. [Google Scholar] [CrossRef] [Green Version]
- Jiménez-Trujillo, I.; González-Pascual, M.; Jiménez-Garća, R.; Hernández-Barrera, V.; Miguel-Yanes, J.M.; Méndez-Bailón, M.; De Miguel-DIez, J.; Ángel Salinero-Fort, M.; Perez-Farinos, N.; Carrasco-Garrido, P.; et al. Type 2 Diabetes Mellitus and Thoracic Aortic Aneurysm and Dissection an Observational Population-Based Study in Spain from 2001 to 2012. Medicine 2016, 95, e3618. [Google Scholar] [CrossRef]
- Liu, H.; Shi, L.; Zeng, T.; Ji, Q.; Shi, Y.; Huang, Y.; Zhang, L.; Xiao, T.; Ye, J.; Lin, Y.; et al. Type 2 Diabetes Mellitus Reduces Clinical Complications and Mortality in Stanford Type B Aortic Dissection after Thoracic Endovascular Aortic Repair: A 3-Year Follow-up Study. Life Sci. 2019, 230, 104–110. [Google Scholar] [CrossRef]
- Takagi, H.; Umemoto, T. Negative Association of Diabetes with Thoracic Aortic Dissection and Aneurysm. Angiology 2017, 68, 216–224. [Google Scholar] [CrossRef]
- Avdic, T.; Franzeán, S.; Zarrouk, M.; Acosta, S.; Nilsson, P.; Gottsäter, A.; Svensson, A.M.; Gudbjörnsdottir, S.; Eliasson, B. Reduced Long-Term Risk of Aortic Aneurysm and Aortic Dissection Among Individuals With Type 2 Diabetes Mellitus: A Nationwide Observational Study. J. Am. Heart. Assoc. 2018, 7, e007618. [Google Scholar] [CrossRef] [Green Version]
- Ortega, R.; Collado, A.; Selles, F.; Gonzalez-Navarro, H.; Sanz, M.-J.; Real, J.T.; Piqueras, L. SGLT-2 (Sodium-Glucose Cotransporter 2) Inhibition Reduces Ang II (Angiotensin II)-Induced Dissecting Abdominal Aortic Aneurysm in ApoE (Apolipoprotein E) Knockout Mice. Arter. Thromb. Vasc. Biol. 2019, 39, 1614–1628. [Google Scholar] [CrossRef] [Green Version]
- Michelena, H.I.; Khanna, A.D.; Mahoney, D.; Margaryan, E.; Topilsky, Y.; Suri, R.M.; Eidem, B.; Edwards, W.D.; Sundt, T.M.; Enriquez-Sarano, M. Incidence of Aortic Complications in Patients with Bicuspid Aortic Valves. JAMA—J. Am. Med. Assoc. 2011, 306, 1104–1112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guirguis-Blake, J.M.; Beil, T.L.; Senger, C.A.; Whitlock, E.P. Ultrasonography Screening for Abdominal Aortic Aneurysms: A Systematic Evidence Review for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2014, 160, 321–329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sidloff, D.; Stather, P.; Dattani, N.; Bown, M.; Thompson, J.; Sayers, R.; Choke, E. Aneurysm Global Epidemiology Study Public Health Measures Can Further Reduce Abdominal Aortic Aneurysm Mortality. Circulation 2014, 129, 747–753. [Google Scholar] [CrossRef] [PubMed] [Green Version]
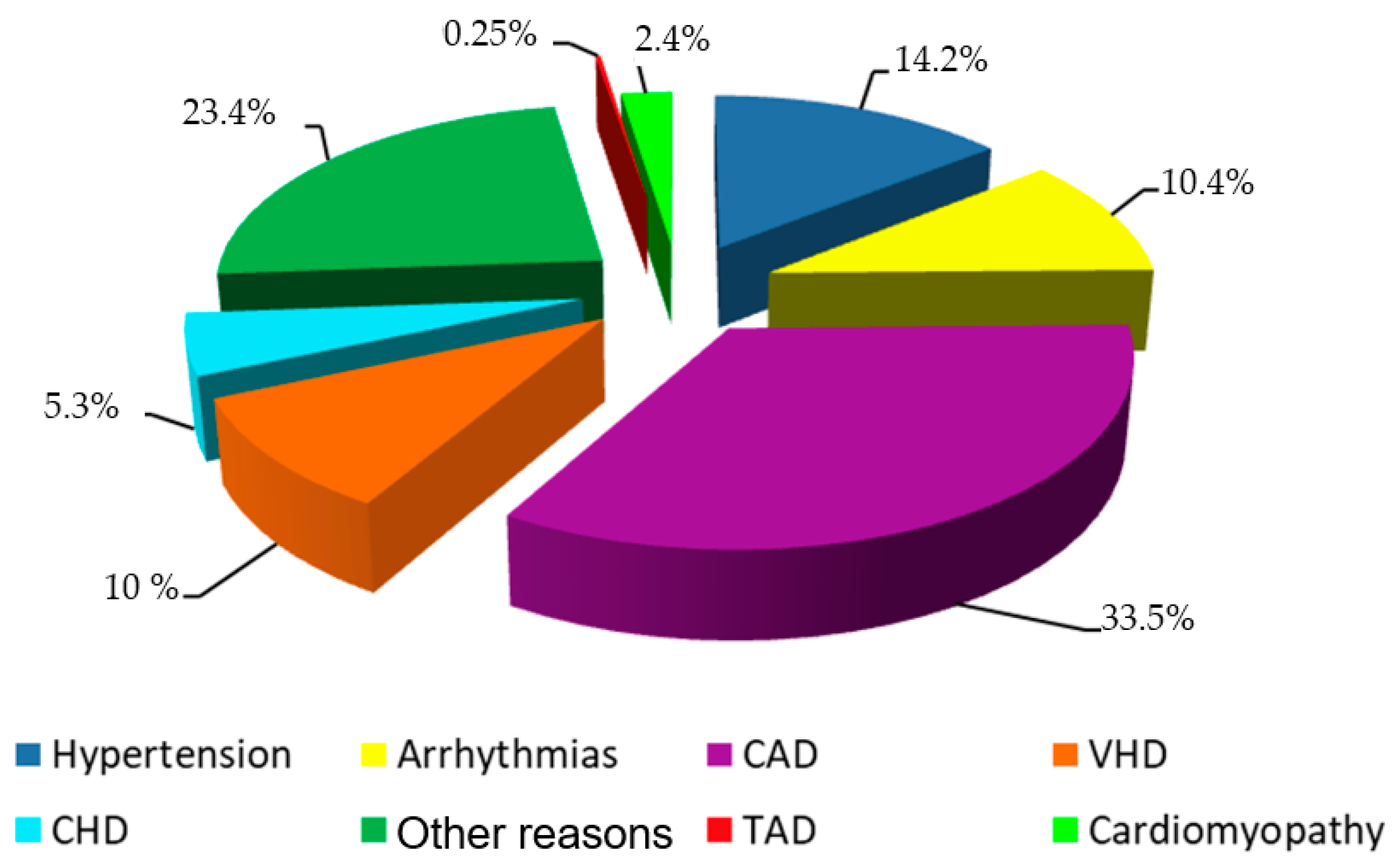
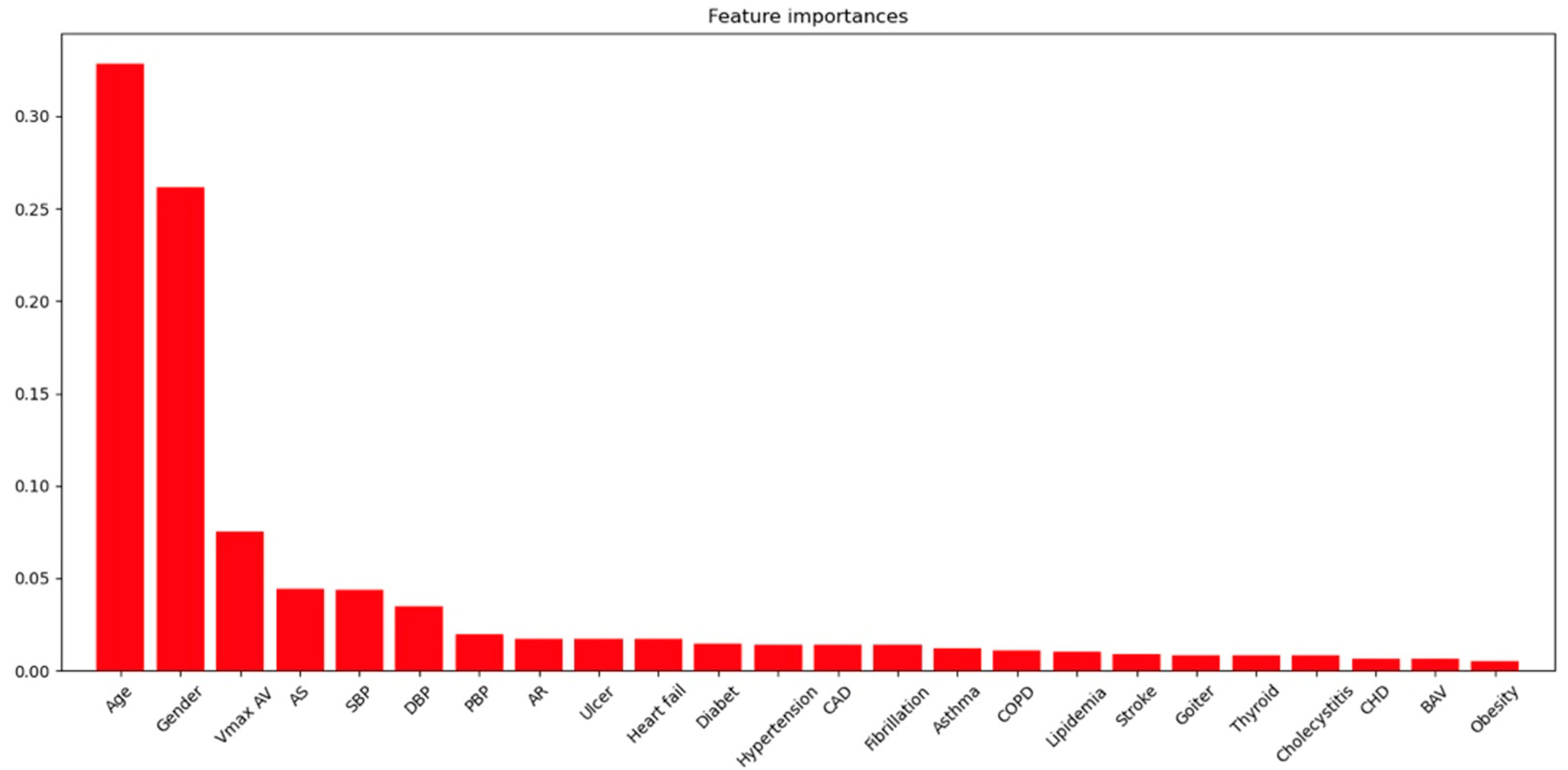
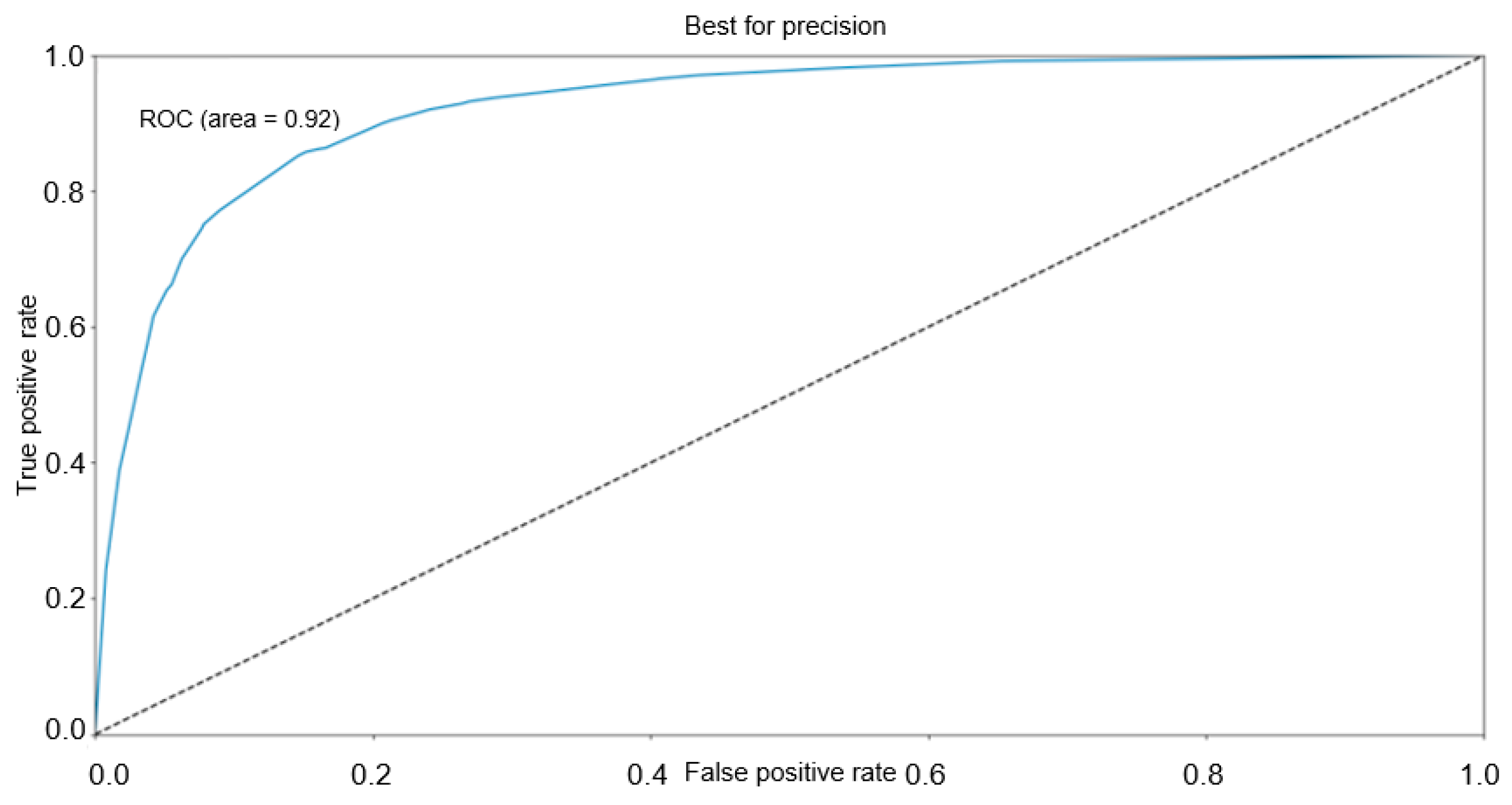
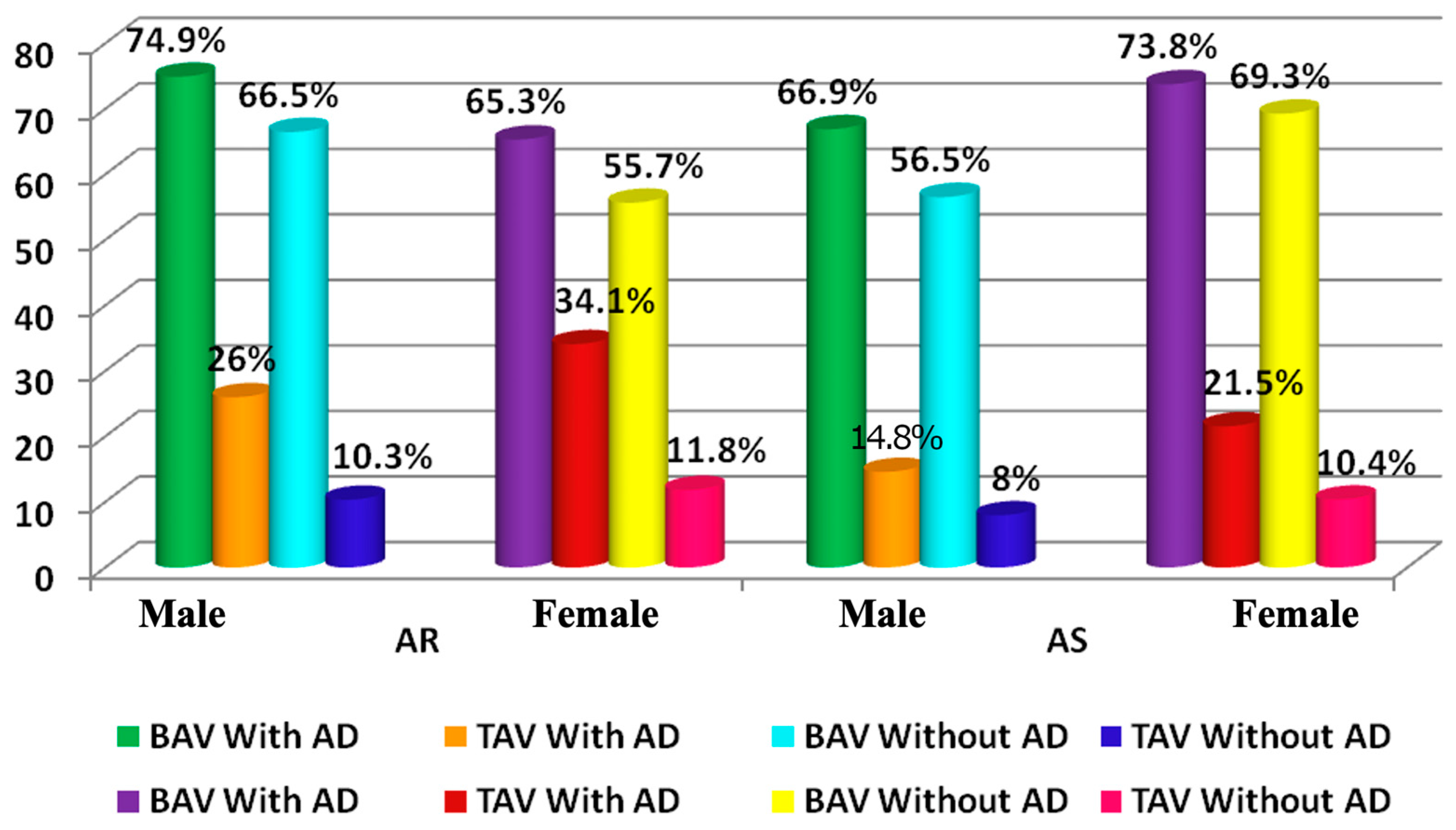
| Variables | N | All Patients | Min/Max |
|---|---|---|---|
| Age, years (median; quartiles) | 84,851 | 59 (34; 68) | 18; 107 |
| Aortic diameter at the sinus of the Valsalva, mm, median; quartiles | 84,851 | 34 (31; 37) | 8; 90 |
| Aortic diameter at the proximal ascending aorta, mm, median; quartiles | 84,851 | 33 (30; 36) | 12; 98 |
| BMI, kg/m2, median; quartiles | 27,362 | 27.3 (21.4; 31.0) | 12.5; 97.7 |
| AS dpmax, mmHg, median; quartiles | 84,851 | 7.0 (5.0; 10.0) | 0.36; 424 |
| EF LV (%), median; quartiles | 76,800 | 63.9 (56.9; 68.9) | 7.0; 91.5 |
| SBP office, mmHg, median; quartiles | 84,851 | 130 (120; 142) | 55; 270 |
| DBP office, mmHg, median; quartiles | 84,851 | 80 (80; 87) | 20; 140 |
| AR, n (%) | 84,757 | 4460 (5.26) | - |
| AS, n (%) | 84,851 | 11,252 (13.26) | - |
| Diabetes mellitus, n (%) | 84,851 | 8426 (9.93) | - |
| Hypertension, n (%) | 84,851 | 59,711 (70.37) | - |
| CAD, n (%) | 84,851 | 28,440 (33.52) | - |
| COPD, n (%) | 84,851 | 6818 (8.04) | - |
| Asthma, n (%) | 84,851 | 2207 (2.60) | - |
| Obesity, (BMI > 30), n (%) | 27,362 | 8420 (30.77) | - |
| Hyperlipidemia, n (%) | 84,851 | 21,087(24.85) | - |
| Heart failure, n (%) | 84,851 | 35,194 (41.48) | - |
| Variables | BAV, n = 1544 N; Median; Quartiles | TAV, n = 83,317 N; Median; Quartiles | p |
|---|---|---|---|
| Age, years (median and bounds) | 40.5 (18; 104) | 59 (18; 88) | <0.0001 |
| Aortic diameter at the sinus of the Valsalva, mm | 35 (32; 39) | 34 (31; 37) | <0.0001 |
| Aortic diameter at the proximal ascending aorta, mm | 36 (32; 42) | 33 (30; 36) | <0.0001 |
| BMI, kg/m2 | 25.5 (22.8; 28.6) | 27.3 (24.2; 31.1) | <0.0001 |
| AS dpmax, mmHg | 18 (11; 34) | 7 (5; 10) | <0.0001 |
| EF LV (%), ΦB | 65.0 (59.7; 70.1) | 63.9 (56.9; 68.9) | <0.0001 |
| SBP office, mmHg | 130 (120; 140) | 130 (120; 143) | 0.008 |
| DBP office, mmHg | 80 (73.5; 83.5) | 80 (80; 88) | 0.0006 |
| AR, n (%) | 333 (21.72) | 4127 (4.96) | <0.0001 |
| AS, n (%) | 901 (58.77) | 10,351 (12.42) | <0.0001 |
| Diabetes mellitus, n (%) | 77 (5.02) | 8349 (10.02) | <0.0001 |
| Hypertension, n (%) | 861 (56.13) | 58,850 (70.63) | <0.0001 |
| CAD, n (%) | 249 (16.23) | 28,191 (33.84) | <0.0001 |
| COPD, n (%) | 101 (6.58) | 6717 (8.06) | 0.03 |
| Asthma, n (%) | 46 (3.00) | 2161 (2.59) | 0.32 |
| Obesity, (BMI > 30), n (%) | 133 (18.68) | 8287 (31.10) | <0.0001 |
| Hyperlipidemia, n (%) | 305 (19.88) | 20,782 (24.94) | <0.0001 |
| Heart failure, n (%) | 750 (48.89) | 34,444 (41.34) | <0.0001 |
| Model | Parameters |
|---|---|
| LR * (imp. feat.) | ‘C’: 2.83, ‘solver’: ‘newton-cg’ |
| LR + SMOTE (imp. feat.) | ‘C’: 0.5, ‘solver’: ‘newton-cg’ |
| LR + SMOTE (all feat.) | ‘C’: 4.0, ‘solver’: ‘liblinear’ |
| RF (imp. feat.) | ‘criterion’: ‘gini’, ‘max_features’: ‘auto’ |
| RF + SMOTE (imp. feat.) | ‘criterion’: ‘gini’, ‘max_features’: ‘auto’ |
| RF + SMOTE (all feat.) | ‘criterion’: ‘gini’, ‘max_features’: ‘log2’ |
| CC * (all. feat.) | ‘depth’: 4, ‘l2_leaf_reg’: 3, ‘learning_rate’: 0.6 |
| CC + SMOTE (imp. feat.) | ‘depth’: 5, ‘l2_leaf_reg’: 2, ‘learning_rate’: 0.9 |
| CC + SMOTE (all feat.) | ‘depth’: 4, ‘l2_leaf_reg’: 1, ‘learning_rate’: 0.2 |
| Variables | BAV | TAV | ||
|---|---|---|---|---|
| With AD, n = 150 | Without AD, n = 400 | With AD, n = 2688 | Without AD, n = 40,925 | |
| Age, years | 54.5 | 35 | 67 | 59 |
| Aortic diameter (sinus Valsalva) | 36 | 31 | 38 | 32 |
| Aortic diameter proximal ascending aorta | 44 (41; 47) | 33 (30; 4) | 41 (40; 44) | 31 (29; 34) |
| BMI, kg/m2 | 26.7 | 24.5 | 28.7 | 27.2 |
| ASdpmax, mm Hg. | 22 (14; 44) | 22 (12; 42.5) | 10 (6; 19) | 7 (6; 10) |
| EF LV (%), | 66.2 | 66.9 | 64.2 | 65.8 |
| SBP office, mm Hg. | 122.5 | 120 | 135 | 130 |
| DBPoffice, mm Hg. | 80 | 80 | 80 | 80 |
| AR, n (%) | 24 (16.0) | 67 (16.75) | 372 (13.9) | 1691 (4.1) |
| AS, n (%) | 105 (70.0) | 260 (65.0) | 824 (30.7) | 5104 (12.5) |
| Diabetes mellitus, n (%) | 8 (5.33) | 21 (5.25) | 305 (11.4) | 4389 (10.7) |
| CAD, n (%) | 25 (16.67) | 51 (12.75) | 1041 (38.7) | 11,110 (27.2) |
| COPD, n (%) | 5 (3.33) | 15 (3.75) | 205 (7.6) | 2365 (5.8) |
| Asthma, n (%) | 5 (3.33) | 10 (2.50) | 122 (4.5) | 1219 (2.9) |
| Obesity, n (%) | 24 (30.77) | 28 (13.93) | 392 (42.2) | 4496 (32.2) |
| Hyperlipidemia, n (%) | 51 (34.0) | 62 (15.50) | 856 (31.9) | 9695 (23.7) |
| Heart failure, n (%) | 26.7 (24.5; 32.1) | 24.5 (21.5; 27.5) | 28.7 (25.1; 32.9) | 27.2 (23.7; 31.2) |
| Variables | BAV | TAV | ||
|---|---|---|---|---|
| With AD, n = 495 | Without AD, n = 499 | With AD, n = 9717 | Without AD, n = 29,987 | |
| Age, years, | 50 | 29 | 63 | 56 |
| Aortic diameter (sinus Valsalva) | 41 | 34 | 41 | 35 |
| Aortic diameter proximal ascending aorta | 42 (40; 46) | 33 (30; 36) | 40 (37; 42) | 33 (31; 36) |
| BMI, kg/m2, | 27.1 | 24.3 | 28.4 | 27.0 |
| ASdpmax, mm Hg. | 20 (12; 39) | 14,5 (9; 26) | 7 (5; 11) | 6 (5; 8) |
| EF LV (%), | 62.9 | 64.9 | 59.4 | 61.6 |
| SBP office, mm Hg. | 135 | 130 | 130 | 130 |
| DBPoffice, mm Hg. | 80 | 80 | 80 | 80 |
| AR, n (%) | 129 (26.6) | 113 (22.7) | 1029 (10.6) | 1035(3.4) |
| AS, n (%) | 302 (62.3) | 234 (46.9) | 1661 (17.1) | 2762 (9.2) |
| Diabetes mellitus, n (%) | 24 (4.95) | 24 (4.8) | 953 (9.8) | 2702 (9.0) |
| CAD, n (%) | 119 (24.54) | 54 (10.8) | 4454 (45.8) | 11,586 (38.6) |
| COPD, n (%) | 52 (10.7) | 29 (5.8) | 1045 (10.8) | 3102 (10.3) |
| Asthma, n (%) | 16 (3.3) | 15 (3.0) | 230 (2.4) | 590 (1.9) |
| Obesity, n (%) | 53 (24.31) | 28 (13.0) | 1046 (36.9) | 2353 (26.5) |
| Hyperlipidemia, n (%) | 122 (25.2) | 70 (14.0) | 2901 (29.9) | 7330 (24.4) |
| Heart failure, n (%) | 282 (58.1) | 195 (39.1) | 4854 (49.9) | 12248 (40.8) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Irtyuga, O.; Kopanitsa, G.; Kostareva, A.; Metsker, O.; Uspensky, V.; Mikhail, G.; Faggian, G.; Sefieva, G.; Derevitskii, I.; Malashicheva, A.; et al. Application of Machine Learning Methods to Analyze Occurrence and Clinical Features of Ascending Aortic Dilatation in Patients with and without Bicuspid Aortic Valve. J. Pers. Med. 2022, 12, 794. https://doi.org/10.3390/jpm12050794
Irtyuga O, Kopanitsa G, Kostareva A, Metsker O, Uspensky V, Mikhail G, Faggian G, Sefieva G, Derevitskii I, Malashicheva A, et al. Application of Machine Learning Methods to Analyze Occurrence and Clinical Features of Ascending Aortic Dilatation in Patients with and without Bicuspid Aortic Valve. Journal of Personalized Medicine. 2022; 12(5):794. https://doi.org/10.3390/jpm12050794
Chicago/Turabian StyleIrtyuga, Olga, Georgy Kopanitsa, Anna Kostareva, Oleg Metsker, Vladimir Uspensky, Gordeev Mikhail, Giuseppe Faggian, Giunai Sefieva, Ilia Derevitskii, Anna Malashicheva, and et al. 2022. "Application of Machine Learning Methods to Analyze Occurrence and Clinical Features of Ascending Aortic Dilatation in Patients with and without Bicuspid Aortic Valve" Journal of Personalized Medicine 12, no. 5: 794. https://doi.org/10.3390/jpm12050794
APA StyleIrtyuga, O., Kopanitsa, G., Kostareva, A., Metsker, O., Uspensky, V., Mikhail, G., Faggian, G., Sefieva, G., Derevitskii, I., Malashicheva, A., & Shlyakhto, E. (2022). Application of Machine Learning Methods to Analyze Occurrence and Clinical Features of Ascending Aortic Dilatation in Patients with and without Bicuspid Aortic Valve. Journal of Personalized Medicine, 12(5), 794. https://doi.org/10.3390/jpm12050794







