A Clinical Decision Support System for the Prediction of Quality of Life in ALS
Abstract
:1. Introduction
2. Materials and Methods
2.1. C-ALS Model
2.2. Implementation of C-ALS Prototype
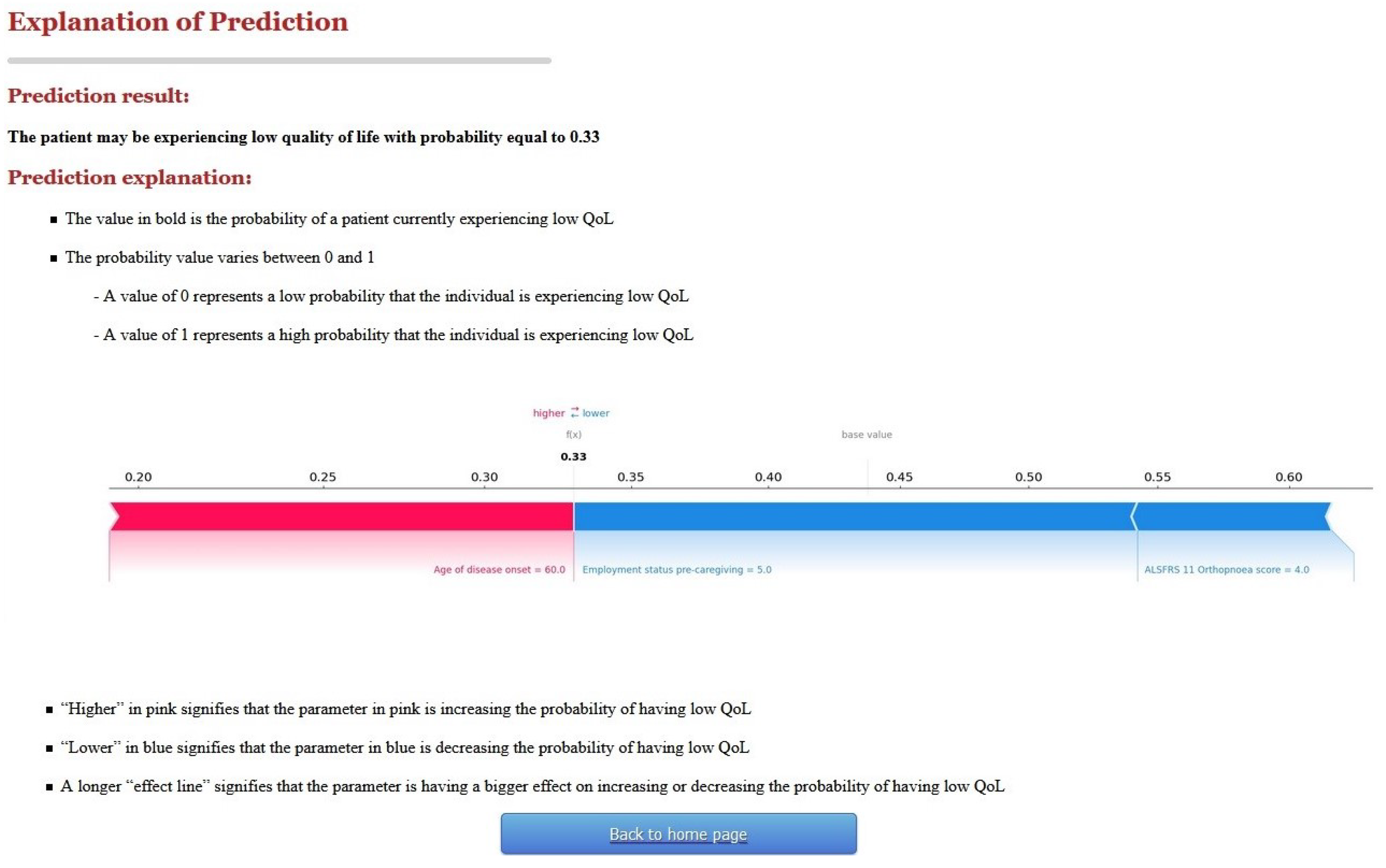
2.2.1. First Version of C-ALS and User Evaluation
2.2.2. User Evaluation
3. Results
3.1. User Evaluation
- Respondent 1: “Maybe another sentence or categorisation after the probability in bold? e.g., mild/moderate/high risk of low QoL? You could also add the three variables into a short narrative to explain their influence on the patient’s QoL, e.g., employment status pre caregiving had the greatest impact etc.”.
- Respondent 2: “Employ clinicians to advise out terminology and output”.
- Respondent 3: “The areas assessed seems very narrow”.
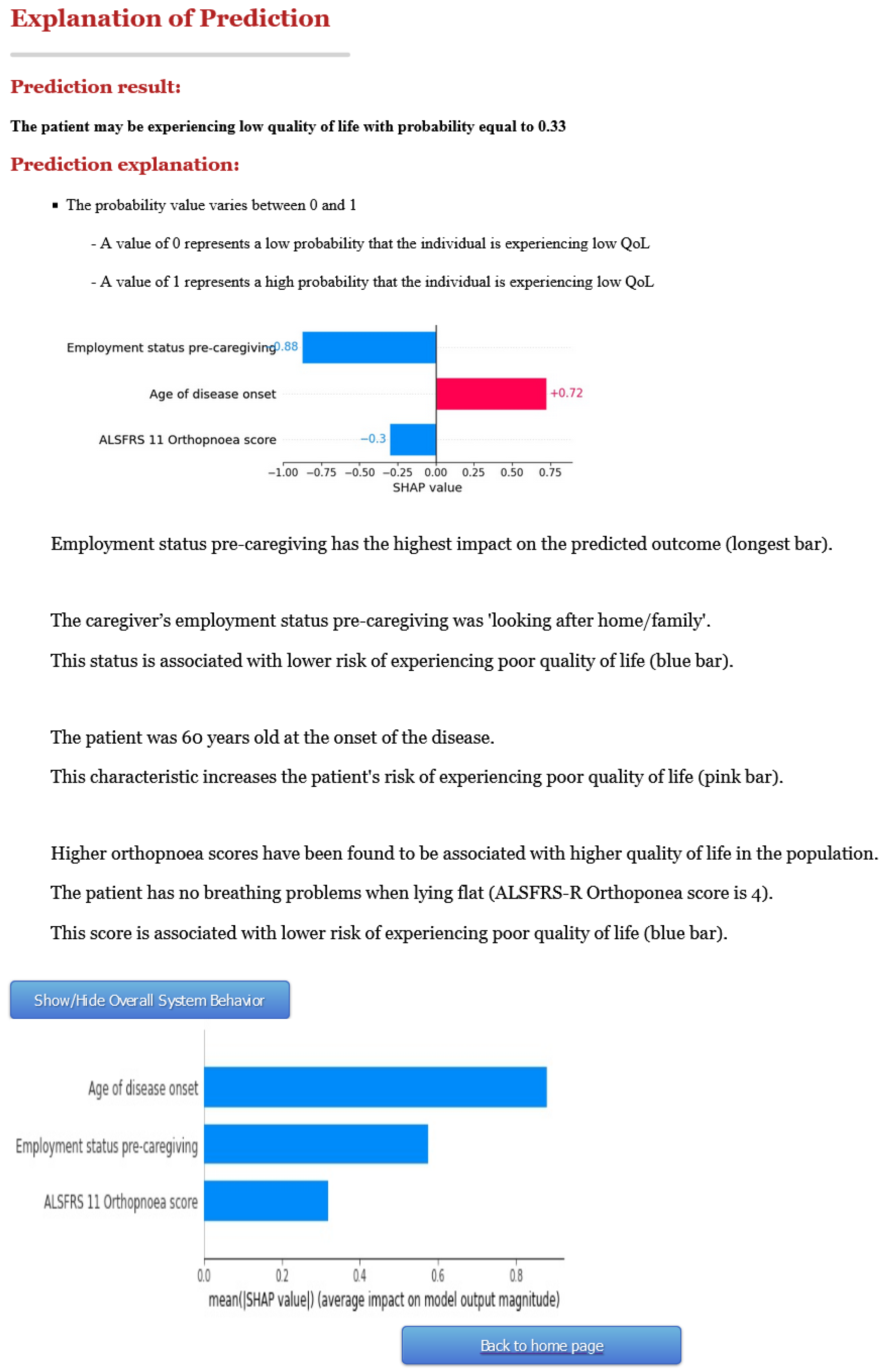
3.2. Second Version of C-ALS
4. Discussion
Limitations and Future Work
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AI | Artificial Intelligence |
| ML | Machine Learning |
| CDSS | Clinical Decision Support Systems |
| XAI | Explainable AI |
| ALS | Amyotrophic Lateral Sclerosis |
| QoL | Quality of Life |
| PALS | People with ALS |
References
- Shortliffe, E.H. Computer programs to support clinical decision making. JAMA 1987, 258, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Sutton, R.T.; Pincock, D.; Baumgart, D.C.; Sadowski, D.C.; Fedorak, R.N.; Kroeker, K.I. An overview of clinical decision support systems: Benefits, risks, and strategies for success. NPJ Digit. Med. 2020, 3, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arrieta, A.B.; Díaz-Rodríguez, N.; Del Ser, J.; Bennetot, A.; Tabik, S.; Barbado, A.; García, S.; Gil-López, S.; Molina, D.; Benjamins, R.; et al. Explainable Artificial Intelligence (XAI): Concepts, taxonomies, opportunities and challenges toward responsible AI. Inf. Fusion 2020, 58, 82–115. [Google Scholar] [CrossRef] [Green Version]
- Belard, A.; Buchman, T.; Forsberg, J.; Potter, B.K.; Dente, C.J.; Kirk, A.; Elster, E. Precision diagnosis: A view of the clinical decision support systems (CDSS) landscape through the lens of critical care. J. Clin. Monit. Comput. 2017, 31, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, K.; Houlihan, C.A.; Balas, E.A.; Lobach, D.F. Improving clinical practice using clinical decision support systems: A systematic review of trials to identify features critical to success. BMJ 2005, 330, 765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bryan, C.; Boren, S. The use and effectiveness of electronic clinical decision support tools in the ambulatory/primary care setting: A systematic review of the literature. J. Innov. Health Inform. 2008, 16, 79–91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagholikar, K.B.; Hankey, R.A.; Decker, L.K.; Cha, S.S.; Greenes, R.A.; Liu, H.; Chaudhry, R. Evaluation of the effect of decision support on the efficiency of primary care providers in the outpatient practice. J. Prim. Care Community Health 2015, 6, 54–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cai, C.J.; Winter, S.; Steiner, D.; Wilcox, L.; Terry, M. “Hello AI”: Uncovering the Onboarding Needs of Medical Practitioners for Human-AI Collaborative Decision-Making. In Proceedings of the ACM on Human-Computer Interaction; ACM: New York, NY, USA, 2019; Volume 3, pp. 1–24. [Google Scholar] [CrossRef] [Green Version]
- Antoniadi, A.M.; Du, Y.; Guendouz, Y.; Wei, L.; Mazo, C.; Becker, B.A.; Mooney, C. Current Challenges and Future Opportunities for XAI in Machine Learning-Based Clinical Decision Support Systems: A Systematic Review. Appl. Sci. 2021, 11, 5088. [Google Scholar] [CrossRef]
- Topol, E.J. High-performance medicine: The convergence of human and artificial intelligence. Nat. Med. 2019, 25, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Bussone, A.; Stumpf, S.; O’Sullivan, D. The role of explanations on trust and reliance in clinical decision support systems. In Proceedings of the 2015 International Conference on Healthcare Informatics, Dallas, TX, USA, 21–23 October 2015; pp. 160–169. [Google Scholar]
- Matthews, M.; Doherty, G.; Coyle, D.; Sharry, J. Designing mobile applications to support mental health interventions. In Handbook of Research on User Interface Design and Evaluation for Mobile Technology; IGI Global: Hershey, PA, USA, 2008; pp. 635–656. [Google Scholar]
- Gilpin, L.H.; Bau, D.; Yuan, B.Z.; Bajwa, A.; Specter, M.; Kagal, L. Explaining explanations: An overview of interpretability of machine learning. In Proceedings of the 2018 IEEE 5th International Conference on Data Science and Advanced Analytics (DSAA), Turin, Italy, 1–3 October 2018; pp. 80–89. [Google Scholar]
- Holzinger, A.; Langs, G.; Denk, H.; Zatloukal, K.; Müller, H. Causability and explainability of artificial intelligence in medicine. Wiley Interdiscip. Rev. Data Min. Knowl. Discov. 2019, 9, e1312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tonekaboni, S.; Joshi, S.; McCradden, M.D.; Goldenberg, A. What Clinicians Want: Contextualizing Explainable Machine Learning for Clinical End Use. In Proceedings of the Machine Learning for Healthcare Conference, Ann Arbor, MI, USA, 8–10 August 2019; pp. 359–380. [Google Scholar]
- Lundberg, S.; Nair, B.; Vavilala, M.S.; Horibe, M.; Eisses, M.J.; Adams, T.; Liston, D.E.; King-Wai Low, D.; Newman, S.F.; Kim, J.; et al. Explainable machine-learning predictions for the prevention of hypoxaemia during surgery. Nat. Biomed. Eng. 2018, 2, 749–760. [Google Scholar] [CrossRef]
- Kulesza, T.; Stumpf, S.; Burnett, M.; Kwan, I. Tell me more? The effects of mental model soundness on personalizing an intelligent agent. In Proceedings of the SIGCHI Conference on Human Factors in Computing Systems, Austin, TX, USA, 5–10 May 2012; pp. 1–10. [Google Scholar]
- World Health Organization. Ethics and Governance of Artificial Intelligence for Health: WHO Guidance; World Health Organization: Geneva, Switzerland, 2021.
- General Data Protection Regulation (GDPR). General Data Protection Regulation (GDPR)—Final Text Neatly Arranged. 2018. Available online: https://gdpr-info.eu/ (accessed on 10 July 2019).
- Haggenmüller, S.; Maron, R.C.; Hekler, A.; Utikal, J.S.; Barata, C.; Barnhill, R.L.; Beltraminelli, H.; Berking, C.; Betz-Stablein, B.; Blum, A.; et al. Skin cancer classification via convolutional neural networks: Systematic review of studies involving human experts. Eur. J. Cancer 2021, 156, 202–216. [Google Scholar] [CrossRef]
- Liu, X.; Faes, L.; Kale, A.U.; Wagner, S.K.; Fu, D.J.; Bruynseels, A.; Mahendiran, T.; Moraes, G.; Shamdas, M.; Kern, C.; et al. A comparison of deep learning performance against health-care professionals in detecting diseases from medical imaging: A systematic review and meta-analysis. Lancet Digit. Health 2019, 1, e271–e297. [Google Scholar] [CrossRef]
- Esteva, A.; Kuprel, B.; Novoa, R.A.; Ko, J.; Swetter, S.M.; Blau, H.M.; Thrun, S. Dermatologist-level classification of skin cancer with deep neural networks. Nature 2017, 542, 115–118. [Google Scholar] [CrossRef]
- Gulshan, V.; Peng, L.; Coram, M.; Stumpe, M.C.; Wu, D.; Narayanaswamy, A.; Venugopalan, S.; Widner, K.; Madams, T.; Cuadros, J.; et al. Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA 2016, 316, 2402–2410. [Google Scholar] [CrossRef]
- Lee, J. Is artificial intelligence better than human clinicians in predicting patient outcomes? J. Med. Internet Res. 2020, 22, e19918. [Google Scholar] [CrossRef]
- Gonçalves, J.J.; Rocha, Á.M. A decision support system for quality of life in head and neck oncology patients. Head Neck Oncol. 2012, 4, 1–9. [Google Scholar] [CrossRef] [Green Version]
- McLeod, J.E.; Clarke, D.M. A review of psychosocial aspects of motor neurone disease. J. Neurol. Sci. 2007, 258, 4–10. [Google Scholar] [CrossRef]
- Wijesekera, L.C.; Leigh, P.N. Amyotrophic lateral sclerosis. Orphanet J. Rare Dis. 2009, 4, 3. [Google Scholar] [CrossRef] [Green Version]
- Galvin, M.; Gavin, T.; Mays, I.; Heverin, M.; Hardiman, O. Individual quality of life in spousal ALS patient-caregiver dyads. Health Qual. Life Outcomes 2020, 18, 1–13. [Google Scholar] [CrossRef]
- Cohen, S.R.; Mount, B.M.; Strobel, M.G.; Bui, F. The McGill Quality of Life Questionnaire: A measure of quality of life appropriate for people with advanced disease. A preliminary study of validity and acceptability. Palliat. Med. 1995, 9, 207–219. [Google Scholar] [CrossRef]
- Antoniadi, A.M.; Galvin, M.; Heverin, M.; Hardiman, O.; Mooney, C. Development of an Explainable Clinical Decision Support System for the Prediction of Patient Quality of Life in Amyotrophic Lateral Sclerosis. In Proceedings of the 36th ACM/SIGAPP Symposium on Applied Computing (SAC ’21), Virtual Event, Korea, 22–26 March 2021; pp. 594–602. [Google Scholar] [CrossRef]
- Antoniadi, A.M.; Galvin, M.; Heverin, M.; Hardiman, O.; Mooney, C. Prediction of quality of life in people with ALS: On the road towards explainable clinical decision support. ACM SIGAPP Appl. Comput. Rev. 2021, 21, 5–17. [Google Scholar] [CrossRef]
- Antoniadi, A.M.; Galvin, M.; Heverin, M.; Hardiman, O.; Mooney, C. Identifying Features That Are Predictive of Quality of Life in People With Amyotrophic Lateral Sclerosis. In Proceedings of the 2020 IEEE International Conference on Healthcare Informatics (ICHI), Oldenburg, Germany, 30 November–3 December 2020; pp. 1–2. [Google Scholar] [CrossRef]
- Mathur, S.; Sutton, J. Personalized medicine could transform healthcare. Biomed. Rep. 2017, 7, 3–5. [Google Scholar] [CrossRef] [Green Version]
- Yang, P. Maximizing quality of life remains an ultimate goal in the era of precision medicine: Exemplified by lung cancer. Precis. Clin. Med. 2019, 2, 8–12. [Google Scholar] [CrossRef]
- Neumann, U.; Genze, N.; Heider, D. EFS: An ensemble feature selection tool implemented as R-package and web-application. BioData Min. 2017, 10, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Chen, T.; Guestrin, C. XGBoost: A Scalable Tree Boosting System. In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining (KDD ’16), San Francisco, CA, USA, 13–17 August 2016; Association for Computing Machinery: New York, NY, USA, 2016; pp. 785–794. [Google Scholar] [CrossRef] [Green Version]
- Friedman, J. Greedy Function Approximation: A Gradient Boosting Machine. Ann. Stat. 2001, 29, 1189–1232. [Google Scholar] [CrossRef]
- Lundberg, S.M.; Lee, S.I. A Unified Approach to Interpreting Model Predictions. In Advances in Neural Information Processing Systems 30; Guyon, I., Luxburg, U.V., Bengio, S., Wallach, H., Fergus, R., Vishwanathan, S., Garnett, R., Eds.; Curran Associates, Inc.: Red Hook, NY, USA, 2017; pp. 4765–4774. [Google Scholar]
- De Koning, R.; Egiz, A.; Kotecha, J.; Ciuculete, A.C.; Ooi, S.Z.Y.; Bankole, N.D.A.; Erhabor, J.; Higginbotham, G.; Khan, M.; Dalle, D.U.; et al. Survey Fatigue During the COVID-19 Pandemic: An Analysis of Neurosurgery Survey Response Rates. Front. Surg. 2021, 8, 326. [Google Scholar] [CrossRef]
- Mitchell, M.; Wu, S.; Zaldivar, A.; Barnes, P.; Vasserman, L.; Hutchinson, B.; Spitzer, E.; Raji, I.D.; Gebru, T. Model cards for model reporting. In Proceedings of the Conference on Fairness, Accountability, and Transparency; ACM: New York, NY, USA, 2019; pp. 220–229. [Google Scholar]
- Zhang, B.; Nilsson, M.E.; Prigerson, H.G. Factors important to patients’ quality of life at the end of life. Arch. Intern. Med. 2012, 172, 1133–1142. [Google Scholar] [CrossRef] [Green Version]
- Byock, I.R.; Merriman, M.P. Measuring quality of life for patients with terminal illness: The Missoula–VITAS® quality of life index. Palliat. Med. 1998, 12, 231–244. [Google Scholar] [CrossRef]
- Epton, J.; Harris, R.; Jenkinson, C. Quality of life in amyotrophic lateral sclerosis/motor neuron disease: A structured review. Amyotroph. Lateral Scler. 2009, 10, 15–26. [Google Scholar] [CrossRef]
- Kolachalama, V.B.; Garg, P.S. Machine learning and medical education. NPJ Digit. Med. 2018, 1, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Schoonderwoerd, T.A.; Jorritsma, W.; Neerincx, M.A.; van den Bosch, K. Human-centered XAI: Developing design patterns for explanations of clinical decision support systems. Int. J. Hum.-Comput. Stud. 2021, 154, 102684. [Google Scholar] [CrossRef]
- Müller, J.; Stoehr, M.; Oeser, A.; Gaebel, J.; Streit, M.; Dietz, A.; Oeltze-Jafra, S. A visual approach to explainable computerized clinical decision support. Comput. Graph. 2020, 91, 1–11. [Google Scholar] [CrossRef]
- Lamy, J.B.; Sekar, B.; Guezennec, G.; Bouaud, J.; Séroussi, B. Explainable artificial intelligence for breast cancer: A visual case-based reasoning approach. Artif. Intell. Med. 2019, 94, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.X.; Adhikari, N.K.; McDonald, H.; Rosas-Arellano, M.P.; Devereaux, P.J.; Beyene, J.; Sam, J.; Haynes, R.B. Effects of computerized clinical decision support systems on practitioner performance and patient outcomes: A systematic review. JAMA 2005, 293, 1223–1238. [Google Scholar] [CrossRef] [PubMed]
- Fiks, A.G. Designing computerized decision support that works for clinicians and families. Curr. Probl. Pediatr. Adolesc. Health Care 2011, 41, 60–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liberati, E.G.; Ruggiero, F.; Galuppo, L.; Gorli, M.; González-Lorenzo, M.; Maraldi, M.; Ruggieri, P.; Friz, H.P.; Scaratti, G.; Kwag, K.H.; et al. What hinders the uptake of computerized decision support systems in hospitals? A qualitative study and framework for implementation. Implement. Sci. 2017, 12, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Rafferty, A.R.; McAuliffe, F.M.; Wei, L.; Mooney, C. An explainable machine learning-based clinical decision support system for prediction of gestational diabetes mellitus. Sci. Rep. 2022, 12, 1170. [Google Scholar] [CrossRef] [PubMed]
- Antoniadi, A.M.; Galvin, M.; Heverin, M.; Hardiman, O.; Mooney, C. Prediction of caregiver quality of life in amyotrophic lateral sclerosis using explainable machine learning. Sci. Rep. 2021, 11, 12237. [Google Scholar] [CrossRef]
- Antoniadi, A.M.; Galvin, M.; Heverin, M.; Hardiman, O.; Mooney, C. Prediction of caregiver burden in amyotrophic lateral sclerosis: A machine learning approach using random forests applied to a cohort study. BMJ Open 2020, 10, e033109. [Google Scholar] [CrossRef] [Green Version]


| Question | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| Would you use a CDSS that may fall short in accuracy (i.e., sometimes make a wrong prediction) provided that an explanation is provided? | 0 (0%) | 2 (25%) | 6 (75%) | 0 (0%) | 0 (0%) |
| Would you find a CDSS that assesses the QoL of a patient with ALS useful for your decision-making regarding the patient’s and caregiver’s support provision? | 0 (0%) | 1 (12.5%) | 4 (50%) | 2 (25%) | 1 (12.5%) |
| Regarding our CDSS, would the provided output and explanation help you justify your clinical decision-making (e.g., to patients and colleagues)? | 0 (0%) | 1 (12.5%) | 4 (50%) | 3 (37.5%) | 0 (0%) |
| Does the visual representation of the CDSS output help you understand the predictions? | 0 (0%) | 0 (0%) | 3 (37.5%) | 5 (62.5%) | 0 (0%) |
| Does the visual representation of the CDSS output help you rationalise the predictions? | 1 (12.5%) | 0 (0%) | 2 (25%) | 5 (62.5%) | 0 (0%) |
| Does the explanation provided add towards your trust of model predictions? | 0 (0%) | 0 (0%) | 5 (62.5%) | 3 (37.5%) | 0 (0%) |
| Does the explanation provided help you decide on actionable steps you can undertake? | 0 (0%) | 1 (12.5%) | 5 (62.5%) | 2 (25%) | 0 (0%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antoniadi, A.M.; Galvin, M.; Heverin, M.; Wei, L.; Hardiman, O.; Mooney, C. A Clinical Decision Support System for the Prediction of Quality of Life in ALS. J. Pers. Med. 2022, 12, 435. https://doi.org/10.3390/jpm12030435
Antoniadi AM, Galvin M, Heverin M, Wei L, Hardiman O, Mooney C. A Clinical Decision Support System for the Prediction of Quality of Life in ALS. Journal of Personalized Medicine. 2022; 12(3):435. https://doi.org/10.3390/jpm12030435
Chicago/Turabian StyleAntoniadi, Anna Markella, Miriam Galvin, Mark Heverin, Lan Wei, Orla Hardiman, and Catherine Mooney. 2022. "A Clinical Decision Support System for the Prediction of Quality of Life in ALS" Journal of Personalized Medicine 12, no. 3: 435. https://doi.org/10.3390/jpm12030435
APA StyleAntoniadi, A. M., Galvin, M., Heverin, M., Wei, L., Hardiman, O., & Mooney, C. (2022). A Clinical Decision Support System for the Prediction of Quality of Life in ALS. Journal of Personalized Medicine, 12(3), 435. https://doi.org/10.3390/jpm12030435







