Nucleic Acid-Based COVID-19 Therapy Targeting Cytokine Storms: Strategies to Quell the Storm
Abstract
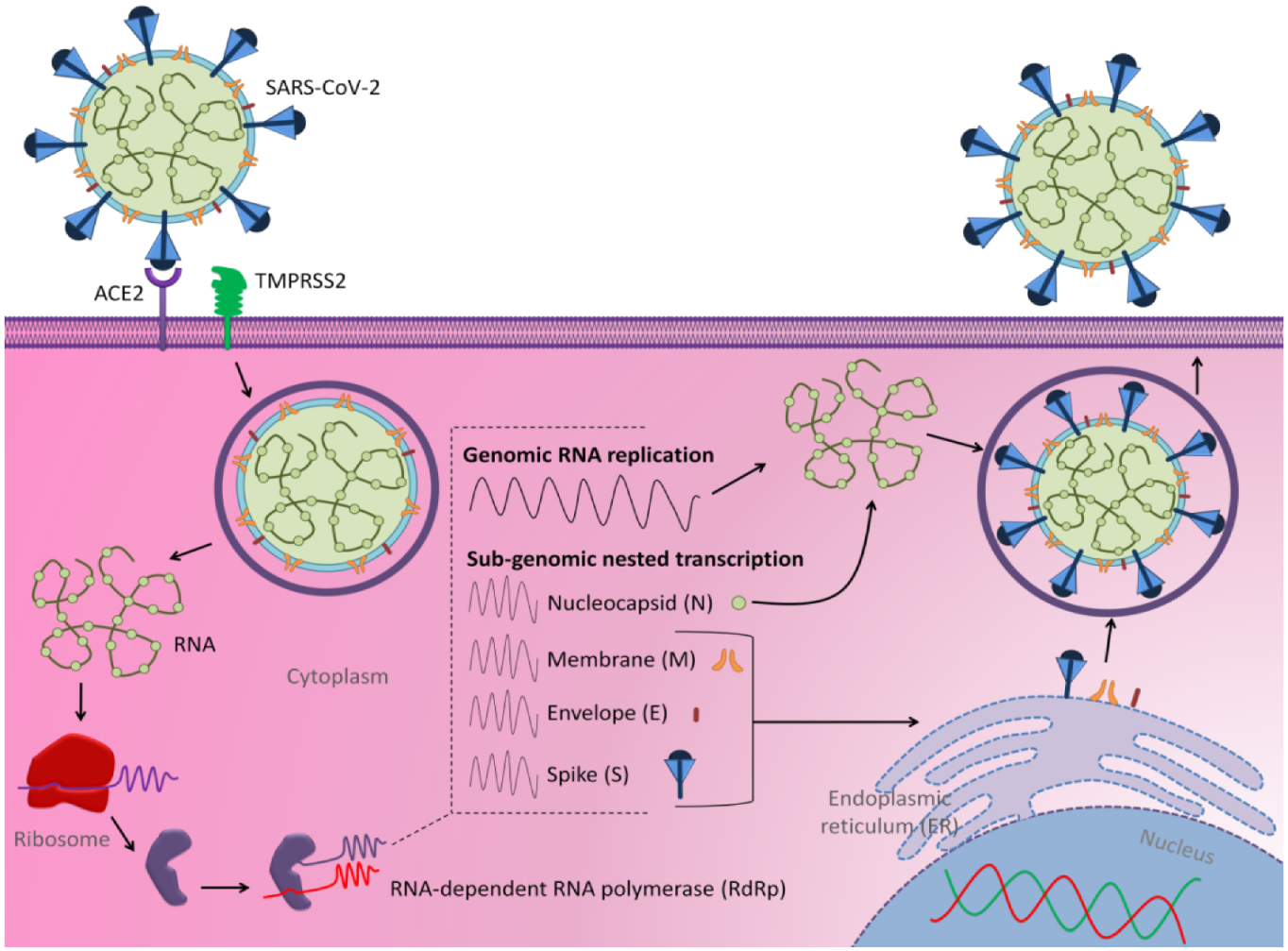
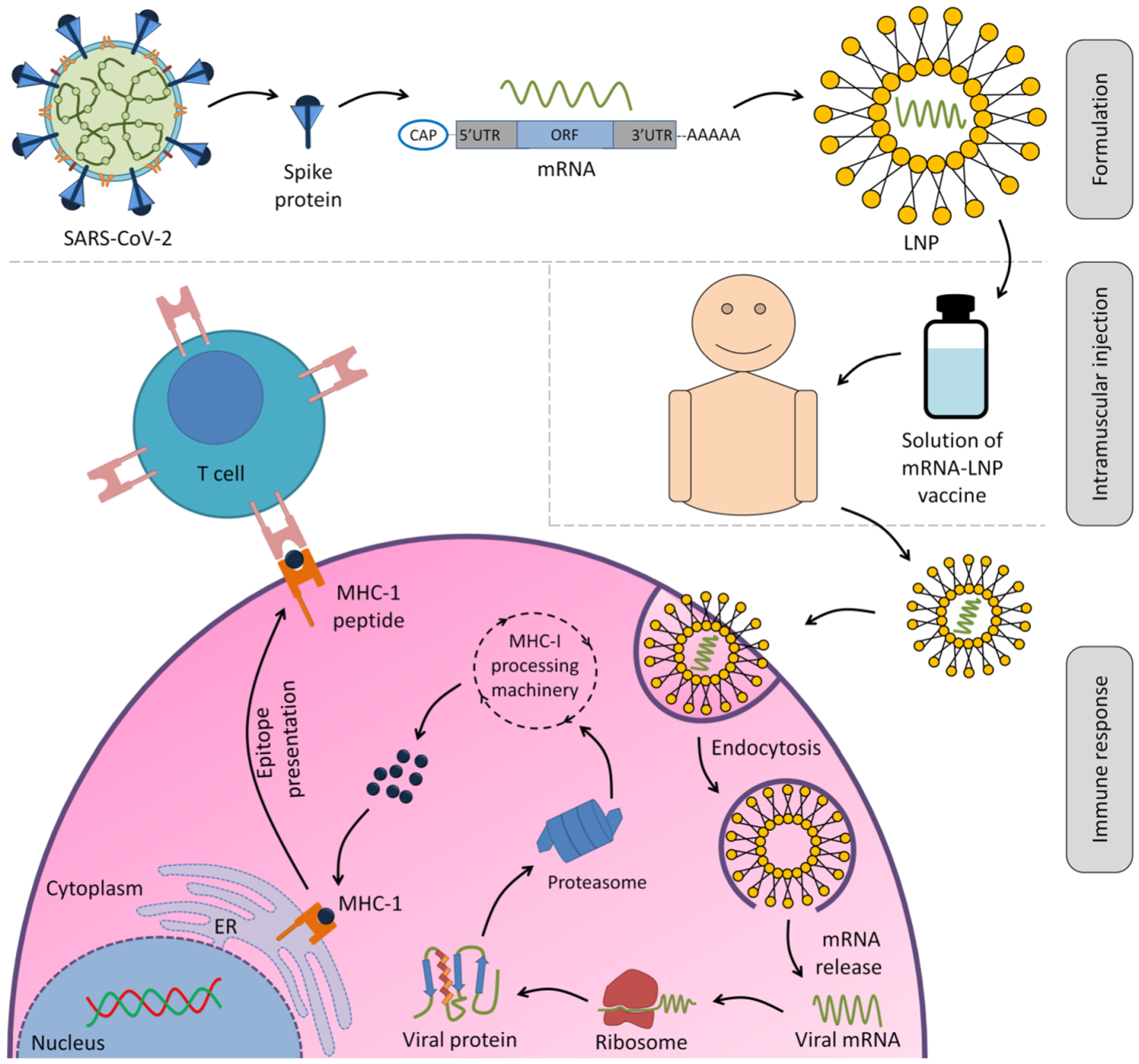
:1. Introduction
2. Current Nucleic Acid-Based Approaches against COVID-19
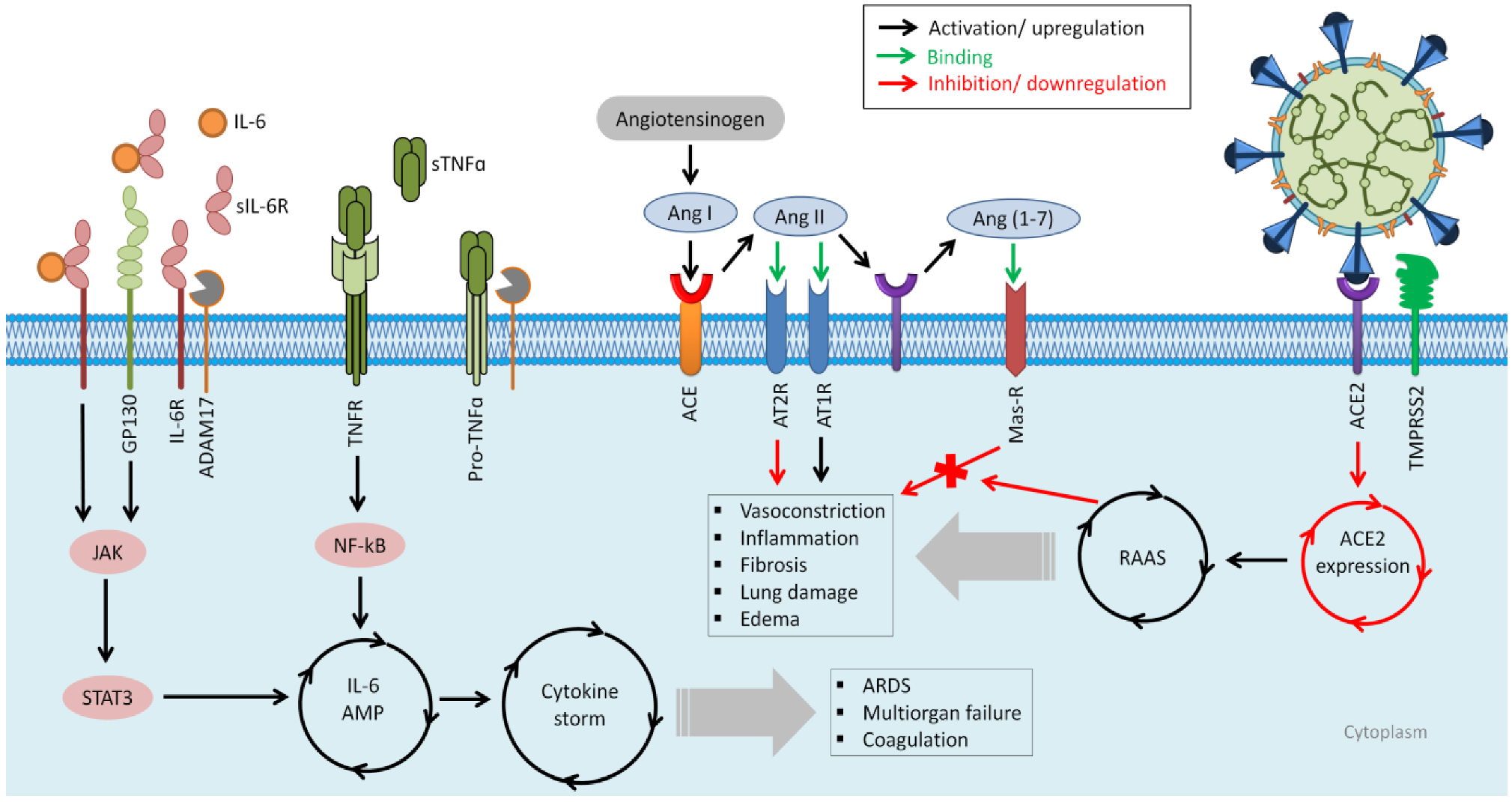
Cytokine Storm
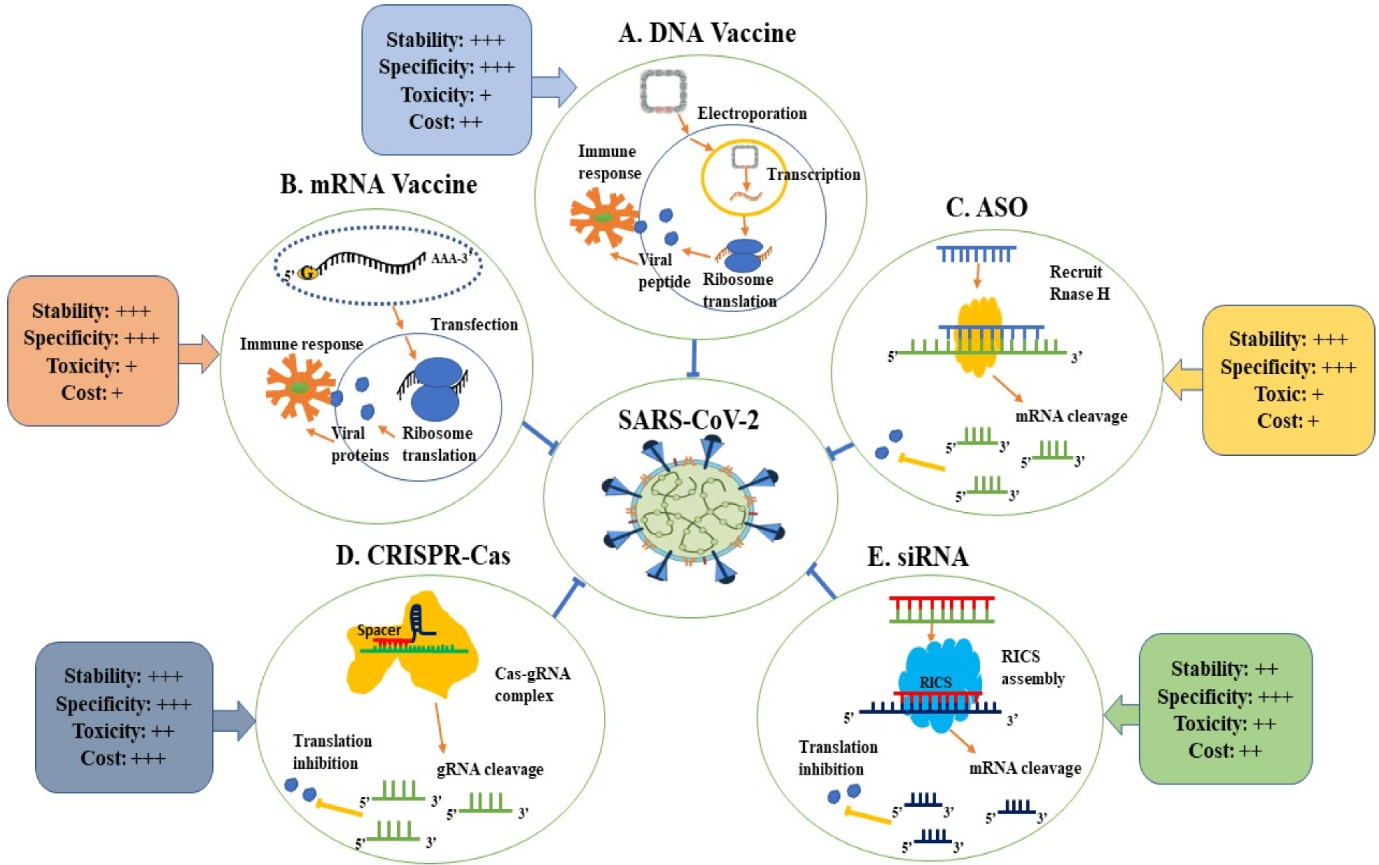
3. COVID-19 Nucleic Acid-Based Therapeutics
4. Targeting the Cytokine Storm via Nucleic Acid-Based Approaches
5. Advantages and Disadvantages of NAT
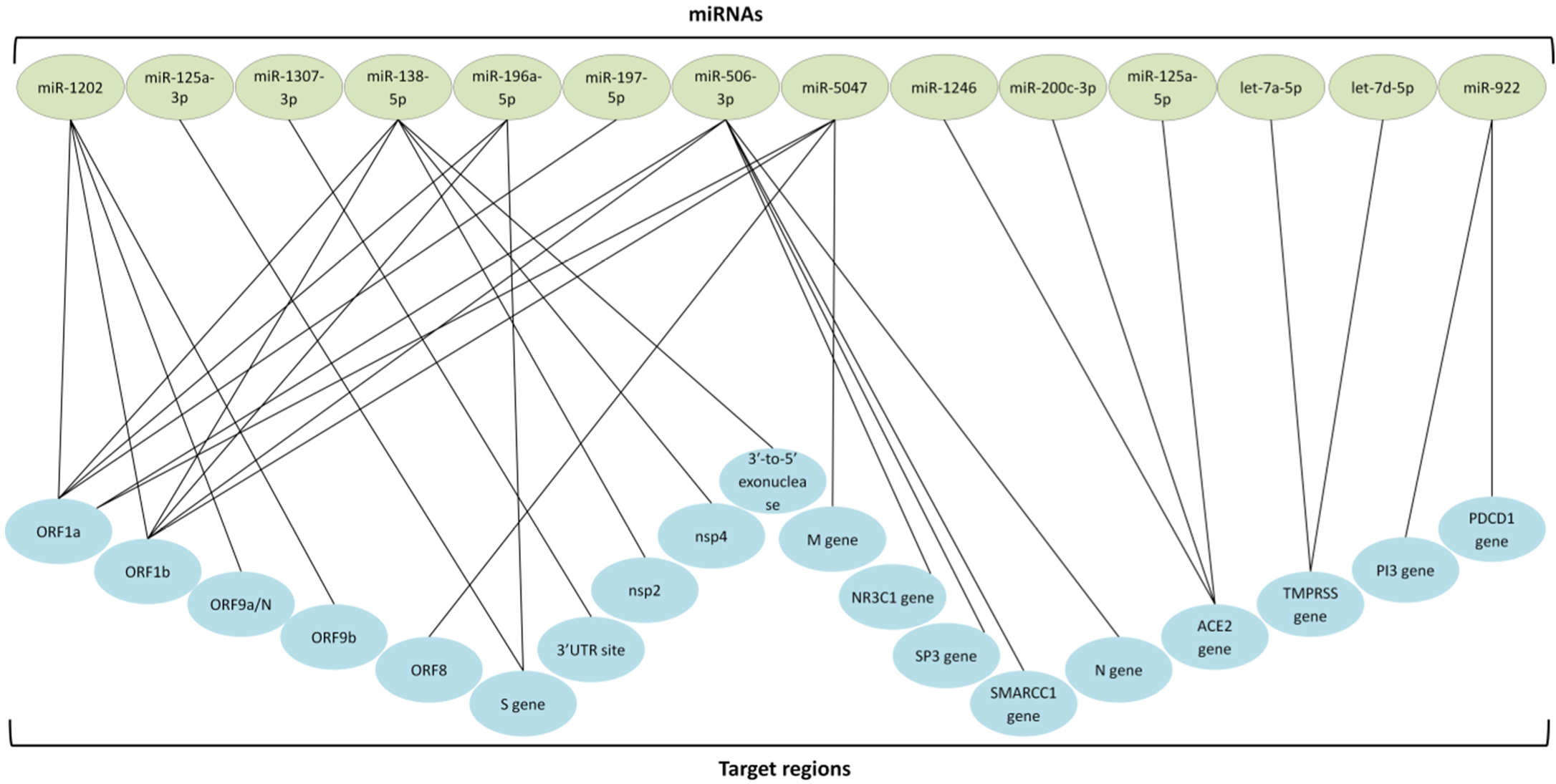
6. Genes Identified as a New Potential Treatment and Protective against COVID-19
7. Future Directions
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Al-Bsheish, M.; Jarrar, M.T.; Scarbrough, A. A Public Safety Compliance Model of Safety Behaviors in the Age of the COVID-19 Pandemic. INQUIRY J. Health Care Organ. Provis. Financ. 2021, 58, 469580211031382. [Google Scholar] [CrossRef]
- Al-Hatamleh, M.A.I.; Hatmal, M.M.; Sattar, K.; Ahmad, S.; Mustafa, M.Z.; Bittencourt, M.C.; Mohamud, R. Antiviral and Immunomodulatory Effects of Phytochemicals from Honey against COVID-19: Potential Mechanisms of Action and Future Directions. Molecules 2020, 25, 5017. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Rizvi, A.; Naeem, A.; Saleem, W.; Ahmed, A.; Parveen, S.; Ilyas, M. COVID-19 and public awareness. Prof. Med. J. 2020, 27, 1710–1716. [Google Scholar] [CrossRef]
- Sahul Hameed, A.S.; Ninawe, A.S.; Nakai, T.; Chi, S.C.; Johnson, K.L. ICTV virus taxonomy profile: Nodaviridae. J. Gen. Virol. 2019, 100, 3–4. [Google Scholar] [CrossRef]
- Xu, R.H.; He, J.F.; Evans, M.R.; Peng, G.W.; Field, H.E.; Yu, D.W.; Lee, C.K.; Luo, H.M.; Lin, W.S.; Lin, P.; et al. Epidemiologic clues to SARS origin in China. Emerg. Infect. Dis. 2004, 10, 1030–1037. [Google Scholar] [CrossRef]
- Zumla, A.I.; Memish, Z.A. Middle East respiratory syndrome coronavirus: Epidemic potential or a storm in a teacup? Eur. Respir. J. 2014, 43, 1243–1248. [Google Scholar] [CrossRef] [Green Version]
- Ali, Z.; Jatoi, M.A.; Al-Wraikat, M.; Ahmed, N.; Li, J. Time to Enhance Immunity via Functional Foods and Supplements: Hope for SARS-CoV-2 Outbreak. Altern. Ther. Health Med. 2021, 27, 30–44. [Google Scholar]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Hatmal, M.M.; Alshaer, W.; Al-Hatamleh, M.A.I.; Hatmal, M.; Smadi, O.; Taha, M.O.; Oweida, A.J.; Boer, J.C.; Mohamud, R.; Plebanski, M. Comprehensive Structural and Molecular Comparison of Spike Proteins of SARS-CoV-2, SARS-CoV and MERS-CoV, and Their Interactions with ACE2. Cells 2020, 9, 2638. [Google Scholar] [CrossRef] [PubMed]
- Fehr, A.R.; Perlman, S. Coronaviruses: An Overview of Their Replication and Pathogenesis; Springer: Berlin, Germany, 2015; Volume 1282, pp. 1–23. [Google Scholar]
- Chen, R.E.; Zhang, X.; Case, J.B.; Winkler, E.S.; Liu, Y.; VanBlargan, L.A.; Liu, J.; Errico, J.M.; Xie, X.; Suryadevara, N.; et al. Resistance of SARS-CoV-2 variants to neutralization by monoclonal and serum-derived polyclonal antibodies. Nat. Med. 2021, 27, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Kubo, H.; Yamada, Y.K.; Taguchi, F. Localization of neutralizing epitopes and the receptor-binding site within the amino-terminal 330 amino acids of the murine coronavirus spike protein. J. Virol. 1994, 68, 5403–5410. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Tenchov, R.; Smoot, J.; Liu, C.; Watkins, S.; Zhou, Q. A Comprehensive Review of the Global Efforts on COVID-19 Vaccine Development. ACS Cent. Sci. 2021, 7, 512–533. [Google Scholar] [CrossRef]
- Anis, S.; Khan, M.M.; Ali, Z.; Khan, A.; Arsalan, H.M.; Naeem, S.; Saleem, I.; Qamar, S.; Khan, M.M.; Ahmad, A. Novel corona virus disease (COVID-19): An updated review on epidemiology, pathogenicity, clinical course, treatments, migrant health concerns and risk factors predictions. Pak. J. Pharm. Sci 2021, 34, 1807–1822. [Google Scholar] [CrossRef]
- Simko, F.; Baka, T. Angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers: Potential allies in the COVID-19 pandemic instead of a threat? Clin. Sci. 2021, 135, 1009–1014. [Google Scholar] [CrossRef]
- Bosch, B.J.; van der Zee, R.; de Haan, C.A.M.; Rottier, P.J.M. The coronavirus spike protein is a class I virus fusion protein: Structural and functional characterization of the fusion core complex. J. Virol. 2003, 77, 8801–8811. [Google Scholar] [CrossRef] [Green Version]
- Gunst, J.D.; Staerke, N.B.; Pahus, M.H.; Kristensen, L.H.; Bodilsen, J.; Lohse, N.; Dalgaard, L.S.; Brønnum, D.; Fröbert, O.; Hønge, B.; et al. Efficacy of the TMPRSS2 inhibitor camostat mesilate in patients hospitalized with COVID-19-a double-blind randomized controlled trial. EClinicalMedicine 2021, 35, 100849. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef]
- Sawicki, S.G.; Sawicki, D.L.; Siddell, S.G. A contemporary view of coronavirus transcription. J. Virol. 2007, 81, 20–29. [Google Scholar] [CrossRef] [Green Version]
- Krijnse-Locker, J.; Ericsson, M.; Rottier, P.J.M.; Griffiths, G. Characterization of the budding compartment of mouse hepatitis virus: Evidence that transport from the RER to the Golgi complex requires only one vesicular transport step. J. Cell Biol. 1994, 124, 55–70. [Google Scholar] [CrossRef] [Green Version]
- de Haan, C.A.M.; Rottier, P.J.M. Molecular interactions in the assembly of coronaviruses. Adv. Virus Res. 2005, 64, 165–230. [Google Scholar] [CrossRef]
- AbuSalah, M.A.H.; Gan, S.H.; Al-Hatamleh, M.A.I.; Irekeola, A.A.; Shueb, R.H.; Yean Yean, C. Recent Advances in Diagnostic Approaches for Epstein-Barr Virus. Pathogens 2020, 9, 226. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Xu, Y.; Gao, R.; Lu, R.; Han, K.; Wu, G.; Tan, W. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA 2020, 323, 1843–1844. [Google Scholar] [CrossRef] [Green Version]
- Salah, M.A.H.A.; Binti Hassan, S.A.; Mat Lazim, N.; Abdullah, B.; Binti Wan Sohaimi, W.F.; Husin, A.; Cheng, K.Y.; Yean, C.Y. Design of InnoPrimers-Duplex Real-Time PCR for Detection and Treatment Response Prediction of EBV-Associated Nasopharyngeal Carcinoma Circulating Genetic Biomarker. Diagnostics 2021, 11, 1761. [Google Scholar] [CrossRef]
- Rahimi, A.; Mirzazadeh, A.; Tavakolpour, S. Genetics and genomics of SARS-CoV-2: A review of the literature with the special focus on genetic diversity and SARS-CoV-2 genome detection. Genomics 2021, 113, 1221–1232. [Google Scholar] [CrossRef]
- Al-Hatamleh, M.A.I.; Hatmal, M.M.; Alshaer, W.; Rahman, E.; Mohd-Zahid, M.H.; Alhaj-Qasem, D.M.; Yean, C.Y.; Alias, I.Z.; Jaafar, J.; Ferji, K.; et al. COVID-19 infection and nanomedicine applications for development of vaccines and therapeutics: An overview and future perspectives based on polymersomes. Eur. J. Pharmacol. 2021, 896, 173930. [Google Scholar] [CrossRef]
- Ghosh, S.; Firdous, S.M.; Nath, A. siRNA could be a potential therapy for COVID-19. EXCLI J. 2020, 19, 528–531. [Google Scholar] [CrossRef]
- Beigel, J.H.; Tomashek, K.M.; Dodd, L.E.; Mehta, A.K.; Zingman, B.S.; Kalil, A.C.; Hohmann, E.; Chu, H.Y.; Luetkemeyer, A.; Kline, S.; et al. Remdesivir for the treatment of COVID-19—Final report. N. Engl. J. Med. 2020, 383, 1813–1826. [Google Scholar] [CrossRef]
- Keshavarzi Arshadi, A.; Webb, J.; Salem, M.; Cruz, E.; Calad-Thomson, S.; Ghadirian, N.; Collins, J.; Diez-Cecilia, E.; Kelly, B.; Goodarzi, H.; et al. Artificial Intelligence for COVID-19 Drug Discovery and Vaccine Development. Front. Artif. Intell. 2020, 3, 65. [Google Scholar] [CrossRef]
- Milken Institute. COVID-19 Treatment and Vaccine Tracker. 2020. Available online: https://milkeninstitute.org/sites/default/files/2020-03/Covid19%20Tracker_WEB.pdf (accessed on 29 August 2021).
- Silveira, M.M.; Moreira, G.; Mendonca, M. DNA vaccines against COVID-19: Perspectives and challenges. Life Sci. 2021, 267, 118919. [Google Scholar] [CrossRef]
- Liu, M.A. A Comparison of Plasmid DNA and mRNA as Vaccine Technologies. Vaccines 2019, 7, 37. [Google Scholar] [CrossRef] [Green Version]
- Piyush, R.; Rajarshi, K.; Chatterjee, A.; Khan, R.; Ray, S. Nucleic acid-based therapy for coronavirus disease 2019. Heliyon 2020, 6, e05007. [Google Scholar] [CrossRef]
- Geall, A.J.; Mandl, C.W.; Ulmer, J.B. RNA: The new revolution in nucleic acid vaccines. Semin. Immunol. 2013, 25, 152–159. [Google Scholar] [CrossRef]
- Restifo, N.P.; Ying, H.; Hwang, L.; Leitner, W.W. The promise of nucleic acid vaccines. Gene Ther. 2000, 7, 89–92. [Google Scholar] [CrossRef] [Green Version]
- Rose, A.; Triano, C.; Alatovic, J.; Maas, S. Pfizer and BioNTech Announce Early Positive Data from an Ongoing Phase 1/2 Study of mRNA-Based Vaccine Candidate against SARS-CoV-2. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-announce-early-positive-data-ongoing-0 (accessed on 1 September 2021).
- Mulligan, M.J.; Lyke, K.E.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Raabe, V.; Bailey, R.; Swanson, K.A. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature 2020, 586, 589–593. [Google Scholar] [CrossRef]
- Sahin, U.; Muik, A.; Derhovanessian, E.; Vogler, I.; Kranz, L.M.; Vormehr, M.; Baum, A.; Pascal, K.; Quandt, J.; Maurus, D. Concurrent human antibody and TH1 type T-cell responses elicited by a COVID-19 RNA vaccine. medRxiv 2020, 586, 594–599. [Google Scholar] [CrossRef]
- Walsh, E.E.; Frenck, R.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R. RNA-based COVID-19 vaccine BNT162b2 selected for a pivotal efficacy study. medRxiv 2020. [Google Scholar] [CrossRef]
- Sahin, U.; Muik, A.; Derhovanessian, E.; Vogler, I.; Kranz, L.M.; Vormehr, M.; Baum, A.; Pascal, K.; Quandt, J.; Maurus, D. COVID-19 vaccine BNT162b1 elicits human antibody and TH 1 T cell responses. Nature 2020, 586, 594–599. [Google Scholar] [CrossRef]
- Walsh, E.E.; Frenck, R.W., Jr.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R. Safety and immunogenicity of two RNA-based COVID-19 vaccine candidates. N. Engl. J. Med. 2020, 383, 2439–2450. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Marc, G.P.; Moreira, E.D.; Zerbini, C. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Vogel, A.B.; Kanevsky, I.; Che, Y.; Swanson, K.A.; Muik, A.; Vormehr, M.; Kranz, L.M.; Walzer, K.C.; Hein, S.; Güler, A. BNT162b vaccines are immunogenic and protect non-human primates against SARS-CoV-2. bioRxiv 2020. [Google Scholar] [CrossRef]
- Sahin, U.; Muik, A.; Vogler, I.; Derhovanessian, E.; Kranz, L.M.; Vormehr, M.; Quandt, J.; Bidmon, N.; Ulges, A.; Baum, A. BNT162b2 induces SARS-CoV-2-neutralising antibodies and T cells in humans. medRxiv 2020, 595, 572–577. [Google Scholar] [CrossRef]
- Widger, A.; Triano, C.; Alatovic, J.; Maas, S. Pfizer and BioNTech Provide Data from German Phase 1/2 Study Further Characterizing Immune Response Following Immunization with Lead COVID-19 Vaccine Candidate BNT162b2. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-provide-data-german-phase-12-study (accessed on 1 September 2021).
- Xie, X.; Zou, J.; Fontes-Garfias, C.R.; Xia, H.; Swanson, K.A.; Cutler, M.; Cooper, D.; Menachery, V.D.; Weaver, S.; Dormitzer, P.R. Neutralization of N501Y mutant SARS-CoV-2 by BNT162b2 vaccine-elicited sera. bioRxiv 2021. [Google Scholar] [CrossRef]
- Muik, A.; Wallisch, A.-K.; Sänger, B.; Swanson, K.A.; Mühl, J.; Chen, W.; Cai, H.; Maurus, D.; Sarkar, R.; Türeci, Ö. Neutralization of SARS-CoV-2 lineage B. 1.1. 7 pseudovirus by BNT162b2 vaccine–elicited human sera. Science 2021, 371, 1152–1153. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Li, J.; Hui, A.; Zhang, X.; Yang, Y.; Tang, R.; Ye, H.; Ji, R.; Lin, M.; Zhu, Z. First report demonstrating the safety and immunogenicity of the SARS-COV-2 BNT162b1 mRNA vaccine in younger and older Chinese adults: A randomized, placebo-controlled, observer-blind Phase I study. Nat. Med. 2021, 27, 1062–1070. [Google Scholar] [CrossRef]
- Amy, R.; Triano, C.; Alatovic, J.; Maas, S. Real-World Evidence Confirms High Effectiveness of Pfizer-BioNTech COVID-19 Vaccine and Profound Public Health Impact of Vaccination One Year after Pandemic Declared. Available online: https://investors.biontech.de/news-releases/news-release-details/real-world-evidence-confirms-high-effectiveness-pfizer-biontech (accessed on 1 September 2021).
- Pitts, J.; Triano, C.; Alatovic, J.; Maas, S. Pfizer-BioNTech Announce Positive Topline Results of Pivotal COVID-19 Vaccine Study in Adolescents. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-biontech-announce-positive-topline-results-pivotal (accessed on 1 September 2021).
- Pitts, J.; Triano, C.; Alatovic, J.; Maas, S. Pfizer and BioNTech Confirm High Efficacy and No Serious Safety Concerns Through Up to Six Months Following Second Dose in Updated Topline Analysis of Landmark COVID-19 Vaccine Study. Available online: https://www.businesswire.com/news/home/20210401005365/en/Pfizer-and-BioNTech-Confirm-High-Efficacy-and-No-Serious-Safety-Concerns-Through-Up-to-Six-Months-Following-Second-Dose-in-Updated-Topline-Analysis-of-Landmark-COVID-19-Vaccine-Study (accessed on 1 September 2021).
- Hussey, C.; Budwick, D.; Talukdar, L. Moderna Announces Positive Interim Phase 1 Data for its mRNA Vaccine (mRNA-1273) Against Novel Coronavirus. Available online: https://investors.modernatx.com/news-releases/news-release-details/moderna-announces-positive-interim-phase-1-data-its-mrna-vaccine (accessed on 1 September 2021).
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J. An mRNA vaccine against SARS-CoV-2—Preliminary report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef]
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J. Safety and immunogenicity of SARS-CoV-2 mRNA-1273 vaccine in older adults. N. Engl. J. Med. 2020, 383, 2427–2438. [Google Scholar] [CrossRef]
- Hussey, C.; Talukdar, L. Moderna’s COVID-19 Vaccine Candidate Meets its Primary Efficacy Endpoint in the First Interim Analysis of the Phase 3 COVE Study. Available online: https://www.businesswire.com/news/home/20201116005608/en/Moderna%E2%80%99s-COVID-19-Vaccine-Candidate-Meets-its-Primary-Efficacy-Endpoint-in-the-First-Interim-Analysis-of-the-Phase-3-COVE-Study (accessed on 1 September 2021).
- Widge, A.T.; Rouphael, N.G.; Jackson, L.A.; Anderson, E.J.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J. Durability of responses after SARS-CoV-2 mRNA-1273 vaccination. N. Engl. J. Med. 2021, 384, 80–82. [Google Scholar] [CrossRef]
- Wu, K.; Werner, A.P.; Moliva, J.I.; Koch, M.; Choi, A.; Stewart-Jones, G.B.; Bennett, H.; Boyoglu-Barnum, S.; Shi, W.; Graham, B.S. mRNA-1273 vaccine induces neutralizing antibodies against spike mutants from global SARS-CoV-2 variants. bioRxiv 2021. [Google Scholar] [CrossRef]
- Doria-Rose, N.; Suthar, M.S.; Makowski, M.; O’Connell, S.; McDermott, A.B.; Flach, B.; Ledgerwood, J.E.; Mascola, J.R.; Graham, B.S.; Lin, B.C. Antibody persistence through 6 months after the second dose of mRNA-1273 vaccine for COVID-19. N. Engl. J. Med. 2021, 384, 2259–2261. [Google Scholar] [CrossRef]
- Wu, K.; Choi, A.; Koch, M.; Ma, L.; Hill, A.; Nunna, N.; Huang, W.; Oestreicher, J.; Colpitts, T.; Bennett, H. Preliminary analysis of safety and immunogenicity of a SARS-CoV-2 variant vaccine booster. medRxiv 2021. [Google Scholar] [CrossRef]
- Choi, A.; Koch, M.; Wu, K.; Dixon, G.; Oestreicher, J.; Legault, H.; Stewart, G.B.; Colpitts, T.; Pajon, R.; Bennett, H. Serum neutralizing activity of mRNA-1273 against SARS-CoV-2 variants. bioRxiv 2021. [Google Scholar] [CrossRef] [PubMed]
- Fakih, S.; Kamilli, A.; Jödicke-Braas, B. CureVac Provides Update on Phase 2b/3 Trial of First-Generation COVID-19 Vaccine Candidate, CVnCoV. Available online: https://www.curevac.com/en/2021/06/16/curevac-provides-update-on-phase-2b-3-trial-of-first-generation-covid-19-vaccine-candidate-cvncov/ (accessed on 1 September 2021).
- Fakih, S.; Kamilli, A.; Jödicke-Braas, B. CureVac Final Data from Phase 2b/3 Trial of First-Generation COVID-19 Vaccine Candidate, CVnCoV, Demonstrates Protection in Age Group of 18 to 60. Available online: https://www.curevac.com/en/2021/06/30/curevac-final-data-from-phase-2b-3-trial-of-first-generation-covid-19-vaccine-candidate-cvncov-demonstrates-protection-in-age-group-of-18-to-60/ (accessed on 1 September 2021).
- Zhang, N.N.; Li, X.F.; Deng, Y.Q.; Zhao, H.; Huang, Y.J.; Yang, G.; Huang, W.J.; Gao, P.; Zhou, C.; Zhang, R.R.; et al. A Thermostable mRNA Vaccine against COVID-19. Cell 2020, 182, 1271–1283.e16. [Google Scholar] [CrossRef] [PubMed]
- Holdings, P.T. Providence Therapeutics Announces Very Favorable Interim Phase 1 Trial Data for PTX-COVID19-B, its mRNA Vaccine against COVID-19. Available online: https://www.providencetherapeutics.com/providence-therapeutics-announces-very-favorable-interim-phase-1-trial-data-for-ptx-covid19-b-its-mrna-vaccine-against-covid-19 (accessed on 1 September 2021).
- Liu, J.; Budylowski, P.; Samson, R.; Griffin, B.D.; Babuadze, G.; Rathod, B.; Colwill, K.; Abioye, J.A.; Schwartz, J.A.; Law, R. Preclinical evaluation of a SARS-CoV-2 mRNA vaccine PTX-COVID19-B. bioRxiv 2021, 8, eabj9815. [Google Scholar] [CrossRef] [PubMed]
- Hatmal, M.M.; Al-Hatamleh, M.A.I.; Olaimat, A.N.; Hatmal, M.; Alhaj-Qasem, D.M.; Olaimat, T.M.; Mohamud, R. Side Effects and Perceptions Following COVID-19 Vaccination in Jordan: A Randomized, Cross-Sectional Study Implementing Machine Learning for Predicting Severity of Side Effects. Vaccines 2021, 9, 556. [Google Scholar] [CrossRef]
- Abu-Raddad, L.J.; Chemaitelly, H.; Butt, A.A.; National Study Group for, C.-V. Effectiveness of the BNT162b2 COVID-19 Vaccine against the B.1.1.7 and B.1.351 Variants. N. Engl. J. Med. 2021, 385, 187–189. [Google Scholar] [CrossRef]
- Lubell, M. Healthcare & Pharmaceuticals: Pfizer Says COVID vaccine Is Highly effective against Delta Variant. Available online: https://www.pharmalive.com/pfizer-says-covid-vaccine-highly-effective-against-delta-variant/ (accessed on 31 August 2021).
- U.S. Food and Drug Administration. COVID-19 Vaccines. Available online: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/covid-19-vaccines (accessed on 8 August 2021).
- U.S. Food and Drug Administration. Fact Sheet for Recipients and Caregivers: Emergency Use Authorization (Eua) of the Pfizer-Biontech COVID-19 Vaccine to Prevent Coronavirus Disease 2019 (COVID-19) In Individuals 12 Years of Age and Older. Available online: https://www.fda.gov/media/144414/download (accessed on 8 August 2021).
- Katella, K. Comparing the COVID-19 Vaccines: How Are They Different? Available online: https://www.yalemedicine.org/news/covid-19-vaccine-comparison (accessed on 8 August 2021).
- Kariko, K.; Muramatsu, H.; Welsh, F.A.; Ludwig, J.; Kato, H.; Akira, S.; Weissman, D. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol. Ther. 2008, 16, 1833–1840. [Google Scholar] [CrossRef]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef] [Green Version]
- Ravell, J.C. A Simple Breakdown of the Ingredients in the COVID Vaccines. Available online: https://www.hackensackmeridianhealth.org/HealthU/2021/01/11/a-simple-breakdown-of-the-ingredients-in-the-covid-vaccines/ (accessed on 8 August 2021).
- Lawton, G. Everything you Need to Know about the Pfizer/BioNTech COVID-19 Vaccine. Available online: https://www.newscientist.com/article/2261805-everything-you-need-to-know-about-the-pfizer-biontech-covid-19-vaccine/ (accessed on 8 August 2021).
- Stowe, J.; Andrews, N.; Gower, C.; Gallagher, E.; Utsi, L.; Simmons, R.; Thelwall, S.; Tessier, E.; Groves, N.; Dabrera, G.; et al. Effectiveness of COVID-19 Vaccines against Hospital Admission with the Delta (B.1.617.2) Variant. 2021. Available online: https://media.tghn.org/articles/Effectiveness_of_COVID-19_vaccines_against_hospital_admission_with_the_Delta_B._G6gnnqJ.pdf (accessed on 1 September 2021).
- Bernal, J.L.; Andrews, N.; Gower, C.; Gallagher, E.; Simmons, R.; Thelwall, S.; Stowe, J.; Tessier, E.; Groves, N.; Dabrera, G.; et al. Effectiveness of COVID-19 vaccines against the B.1.617.2 variant. medRvix 2021, 385, e92. [Google Scholar] [CrossRef]
- Thomas, S.J.; Moreira, E.D.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Marc, G.P.; Polack, F.P.; Zerbini, C.; et al. Six Month Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. medRvix 2021. [Google Scholar] [CrossRef]
- Bernal, J.L.; Andrews, N.; Gower, C.; Gallagher, E.; Simmons, R.; Thelwall, S.; Stowe, J.; Tessier, E.; Groves, N.; Dabrera, G.; et al. Effectiveness of COVID-19 Vaccines against the B.1.617.2 (Delta) Variant. N. Engl. J. Med. 2021, 385, 585–594. [Google Scholar] [CrossRef]
- Thompson, M.G.; Burgess, J.L.; Naleway, A.L.; Tyner, H.L.; Yoon, S.K.; Meece, J.; Olsho, L.E.; Caban-Martinez, A.J.; Fowlkes, A.; Lutrick, K.; et al. Interim Estimates of Vaccine Effectiveness of BNT162b2 and mRNA-1273 COVID-19 Vaccines in Preventing SARS-CoV-2 Infection Among Health Care Personnel, First Responders, and Other Essential and Frontline Workers—Eight U.S. Locations, December 2020–March 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Moderna. Moderna Provides Clinical and Supply Updates on COVID-19 Vaccine Program ahead of 2nd Annual Vaccines Day. Available online: https://www.businesswire.com/news/home/20210413006131/en/ (accessed on 9 August 2021).
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Werner, A.P.; Koch, M.; Choi, A.; Narayanan, E.; Stewart-Jones, G.B.E.; Colpitts, T.; Bennett, H.; Boyoglu-Barnum, S.; Shi, W.; et al. Serum Neutralizing Activity Elicited by mRNA-1273 Vaccine. N. Engl. J. Med. 2021, 384, 1468–1470. [Google Scholar] [CrossRef] [PubMed]
- Moderna US, IFact Sheet for Recipients and Caregivers: Emergency Use Authorization (EUA) of the Moderna COVID-19 Vaccine to Prevent Coronavirus Disease 2019 (COVID-19) in Individuals 18 Years of Age and Older. Available online: https://www.fda.gov/media/144638/download (accessed on 9 August 2021).
- Modena. Moderna Provides a Clinical Update on the Neutralizing Activity of its COVID-19 Vaccine on Emerging Variants Including the Delta Variant First Identified in India. Available online: https://www.businesswire.com/news/home/20210629005708/en/ (accessed on 9 August 2021).
- Modena. Moderna Announces First Participants Dosed in Study Evaluating COVID-19 Booster Vaccine Candidates. Available online: https://investors.modernatx.com/news-releases/news-release-details/moderna-announces-first-participants-dosed-study-evaluating (accessed on 9 August 2021).
- Oncotelic Therapeutics. Oncotelic Therapeutics, Inc. Announces Positive Top Line Data for Arti-19 Clinical Trial Evaluating Pulmoheal™ versus COVID-19. Available online: https://www.globenewswire.com/en/news-re-lease/2021/04/20/2213564/0/en/ONCOTELIC-THERAPEUTICS-INC-ANNOUNCES-POSITIVE-TOP-LINE-DATA-FOR-ARTI-19-CLINICAL-TRIAL-EVALUATING-PULMOHEAL-VERSUS-COVID-19.html (accessed on 1 September 2021).
- AIM ImmunoTech. AIM ImmunoTech Announces Positive Safety Data in Second Cohort of Phase 1 Clinical Study Investigating Intranasal Administration of Ampligen as a Potential Prophylaxis or Treatment for COVID-19 and Other Respiratory Viral Diseases. Available online: https://www.biospace.com/article/releases/aim-immunotech-announces-positive-safety-data-in-second-cohort-of-phase-1-clinical-study-investigating-intranasal-administration-of-ampligen-as-a-potential-prophylaxis-or-treatment-for-covid-19-and-other-respiratory-viral-diseases/ (accessed on 1 September 2021).
- AIM ImmunoTech. ImmunoTech Announces Positive Safety Data in First Cohort of Phase 1 Clinical Study Investigating Intranasal Administration of Ampligen as a Potential Prophylaxis or Treatment for COVID-19 and Other Respiratory Viral Diseases. Available online: https://www.biospace.com/article/releases/aim-immunotech-announces-positive-safty-data-in-first-cohort-of-phase-1-clinical-study-investigating-intranasal-administration-of-ampligen-as-a-potential-prophylaxis-or-treatment-for-covid-19-and-other-respiratory-viral-diseases/ (accessed on 1 September 2021).
- Bhattacharyya, P.; Biswas, S.C. Small Non-coding RNAs: Do They Encode Answers for Controlling SARS-CoV-2 in the Future? Front. Microbiol. 2020, 11, 571553. [Google Scholar] [CrossRef] [PubMed]
- Mateon Therapeutics. Mateon Achieves Milestone in its Development of OT-101, a Phase 3 Clinical Drug Candidate, Against COVID-19. Available online: https://www.globenewswire.com/en/news-release/2020/04/06/2012085/10132/en/Mateon-Achieves-Milestone-in-its-Development-of-OT-101-a-Phase-3-Clinical-Drug-Candidate-Against-COVID-19.html (accessed on 11 August 2021).
- Boumaza, A.; Gay, L.; Mezouar, S.; Bestion, E.; Diallo, A.B.; Michel, M.; Desnues, B.; Raoult, D.; La Scola, B.; Halfon, P.; et al. Monocytes and Macrophages, Targets of Severe Acute Respiratory Syndrome Coronavirus 2: The Clue for Coronavirus Disease 2019 Immunoparalysis. J. Infect. Dis. 2021, 224, 395–406. [Google Scholar] [CrossRef]
- Carvacho, I.; Piesche, M. RGD-binding integrins and TGF-beta in SARS-CoV-2 infections—Novel targets to treat COVID-19 patients? Clin. Transl. Immunol. 2021, 10, e1240. [Google Scholar] [CrossRef]
- Uckun, F.M.; Qazi, S.; Hwang, L.; Trieu, V.N. Recurrent or Refractory High-Grade Gliomas Treated by Convection-Enhanced Delivery of a TGFbeta2-Targeting RNA Therapeutic: A Post-Hoc Analysis with Long-Term Follow-Up. Cancers 2019, 11, 1892. [Google Scholar] [CrossRef] [Green Version]
- Mateon Therapeutics. Mateon Report Positive Results for Multiple COVID-19 Drug CandidatesOT-101 and Two Additional Candidates Demonstrated Viral Inhibition Activity against Coronavirus. Available online: https://www.biospace.com/article/releases/mateon-report-positive-results-for-multiple-covid-19-drug-candidatesot-101-and-two-additional-candidates-demonstrated-viral-inhibition-activity-against-coronavirus/ (accessed on 11 August 2021).
- Chan, J.F.; Yao, Y.; Yeung, M.L.; Deng, W.; Bao, L.; Jia, L.; Li, F.; Xiao, C.; Gao, H.; Yu, P.; et al. Treatment with Lopinavir/Ritonavir or Interferon-beta1b Improves Outcome of MERS-CoV Infection in a Nonhuman Primate Model of Common Marmoset. J. Infect. Dis. 2015, 212, 1904–1913. [Google Scholar] [CrossRef]
- Prokunina-Olsson, L.; Alphonse, N.; Dickenson, R.E.; Durbin, J.E.; Glenn, J.S.; Hartmann, R.; Kotenko, S.V.; Lazear, H.M.; O’Brien, T.R.; Odendall, C.; et al. COVID-19 and emerging viral infections: The case for interferon lambda. J. Exp. Med. 2020, 217, e20200653. [Google Scholar] [CrossRef] [Green Version]
- Simabuco, F.M.; Tamura, R.E.; Pavan, I.C.B.; Morale, M.G.; Ventura, A.M. Molecular mechanisms and pharmacological interventions in the replication cycle of human coronaviruses. Genet. Mol. Biol. 2020, 44, e20200212. [Google Scholar] [CrossRef]
- AIM ImmunoTech. Ampligen’s Update in COVID-19, ME/CFS, COVID-19 Induced Fatigue and Immuno-Oncology Alferon Manufacturing Update. Available online: https://d2ghdaxqb194v2.cloudfront.net/2265/181798.pdf (accessed on 11 August 2021).
- Barnard, D.L.; Day, C.W.; Bailey, K.; Heiner, M.; Montgomery, R.; Lauridsen, L.; Chan, P.K.; Sidwell, R.W. Evaluation of immunomodulators, interferons and known in vitro SARS-coV inhibitors for inhibition of SARS-coV replication in BALB/c mice. Antivir. Chem. Chemother. 2006, 17, 275–284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Day, C.W.; Baric, R.; Cai, S.X.; Frieman, M.; Kumaki, Y.; Morrey, J.D.; Smee, D.F.; Barnard, D.L. A new mouse-adapted strain of SARS-CoV as a lethal model for evaluating antiviral agents in vitro and in vivo. Virology 2009, 395, 210–222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frieman, M.; Heise, M.; Baric, R. SARS coronavirus and innate immunity. Virus Res. 2008, 133, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Sadler, A.J.; Williams, B.R. Interferon-inducible antiviral effectors. Nat. Rev. Immunol. 2008, 8, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Soppe, J.A.; Lebbink, R.J. Antiviral goes viral: Harnessing CRISPR/Cas9 to combat viruses in humans. Trends Microbiol. 2017, 25, 833–850. [Google Scholar] [CrossRef]
- INOVIO Pharmaceuticals. INOVIO Announces Positive Interim Phase 1 Data for INO-4800 Vaccine for COVID-19. Available online: http://ir.inovio.com/news-releases/news-releases-details/2020/INOVIO-Announces-Positive-Interim-Phase-1-Data-For-INO-4800-Vaccine-for-COVID-19/default.aspx (accessed on 1 September 2021).
- Fomsgaard, A. The Statens Serum Institut is Developing a New Promising. Available online: https://www.ssi.dk/aktuelt/nyheder/2020/statens-serum-institut-udvikler-en-ny-lovende-vaccine-mod-covid-19 (accessed on 1 September 2021).
- Clinical Trials Arena. University of Cambridge to Trial COVID-19 Vaccine Candidate. Available online: https://www.clinicaltrialsarena.com/news/cambridge-uni-covid-vaccine/ (accessed on 1 September 2021).
- Rego, G.N.A.; Nucci, M.P.; Alves, A.H.; Oliveira, F.A.; Marti, L.C.; Nucci, L.P.; Mamani, J.B.; Gamarra, L.F. Current Clinical Trials Protocols and the Global Effort for Immunization against SARS-CoV-2. Vaccines 2020, 8, 474. [Google Scholar] [CrossRef]
- Zydus Cadila. Zydus Cadila Announces Completion of Dosing in Phase I Clinical Trial of ZyCoV-D. Available online: https://pipelinereview.com/index.php/2020080575496/Vaccines/Zydus-Cadila-Announces-Completion-of-Dosing-in-Phase-I-Clinical-Trial-of-ZyCoV-D.html (accessed on 16 August 2021).
- Kumar, V.M.; Pandi-Perumal, S.R.; Trakht, I.; Thyagarajan, S.P. Strategy for COVID-19 vaccination in India: The country with the second highest population and number of cases. Npj Vaccines 2021, 6, 60. [Google Scholar] [CrossRef]
- Precision Vaccinations. ZyCoV-D COVID-19 Vaccine. Available online: https://www.precisionvaccinations.com/vaccines/zycov-d-covid-19-vaccine (accessed on 19 August 2021).
- Kaur, S.P.; Gupta, V. COVID-19 Vaccine: A comprehensive status report. Virus. Res. 2020, 288, 198114. [Google Scholar] [CrossRef]
- Zydus Cadila. ZyCoV-D: Zydus Cadila to Begin Phase II Human Trials of COVID-19 Vaccine from Tomorrow. Available online: https://www.indiatvnews.com/business/news-zycov-d-coronavirus-vaccine-zydus-cadila-phase-ii-clinical-trials-covid-19-vaccine-august-6-639700 (accessed on 16 August 2021).
- Zydus Cadila. Zydus Applies to the DCGI for EUA to Launch ZyCoV-D, the World’s First Plasmid DNA Vaccine for COVID-19. 2021. Available online: https://www.zyduscadila.com/public/pdf/pressrelease/ZyCoV_D_Press_Release_1_7_2021.pdf (accessed on 18 August 2021).
- Brisse, M.; Vrba, S.M.; Kirk, N.; Liang, Y.; Ly, H. Emerging Concepts and Technologies in Vaccine Development. Front. Immunol. 2020, 11, 583077. [Google Scholar] [CrossRef]
- Rawat, K.; Kumari, P.; Saha, L. COVID-19 vaccine: A recent update in pipeline vaccines, their design and development strategies. Eur. J. Pharmacol. 2021, 892, 173751. [Google Scholar] [CrossRef]
- Precision Vaccinations. AG0301 COVID-19 Vaccine Description. Available online: https://www.precisionvaccinations.com/vaccines/ag0301-covid-19-vaccine (accessed on 19 August 2021).
- AnGes, I. Study of COVID-19 DNA Vaccine (AG0301-COVID19). Available online: https://clinicaltrials.gov/ct2/show/NCT04463472 (accessed on 19 August 2021).
- AnGes, I. Phase II/III Study of COVID-19 DNA Vaccine (AG0302-COVID19). Available online: https://clinicaltrials.gov/ct2/show/NCT04655625 (accessed on 19 August 2021).
- Pharmaceuticals, I. Safety, Tolerability and Immunogenicity of INO-4800 for COVID-19 in Healthy Volunteers. Available online: https://clinicaltrials.gov/ct2/show/NCT04336410 (accessed on 19 August 2021).
- Precision Vaccinations. INO-4800 COVID-19 Vaccine. Available online: https://www.precisionvaccinations.com/vaccines/ino-4800-covid-19-vaccine (accessed on 23 August 2021).
- Smith, T.R.; Patel, A.; Ramos, S.; Elwood, D.; Zhu, X.; Yan, J.; Gary, E.N.; Walker, S.N.; Schultheis, K.; Purwar, M. Immunogenicity of a DNA vaccine candidate for COVID-19. Nat. Commun. 2020, 11, 2601. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Walters, J.; Reuschel, E.L.; Schultheis, K.; Parzych, E.; Gary, E.N.; Maricic, I.; Purwar, M.; Eblimit, Z.; Walker, S.N. Intradermal-delivered DNA vaccine provides anamnestic protection in a rhesus macaque SARS-CoV-2 challenge model. BioRxiv 2020. [Google Scholar] [CrossRef]
- INOVIO. Reports Positive Interim Phase I Data for COVID-19 DNA Vaccine, Joins “Warp Speed” Primate Study. Available online: https://www.genengnews.com/news/inovio-reports-positive-interim-phase-i-data-for-covid-19-dna-vaccine-joins-warp-speed-primate-study/ (accessed on 23 August 2021).
- Tebas, P.; Yang, S.; Boyer, J.D.; Reuschel, E.L.; Patel, A.; Christensen-Quick, A.; Andrade, V.M.; Morrow, M.P.; Kraynyak, K.; Agnes, J. Safety and immunogenicity of INO-4800 DNA vaccine against SARS-CoV-2: A preliminary report of an open-label, Phase 1 clinical trial. EClinicalMedicine 2021, 31, 100689. [Google Scholar] [CrossRef] [PubMed]
- Andrade, V.M.; Christensen-Quick, A.; Agnes, J.; Tur, J.; Reed, C.; Kalia, R.; Marrero, I.; Elwood, D.; Schultheis, K.; Purwar, M. INO-4800 DNA vaccine induces neutralizing antibodies and T cell Activity against global SARS-CoV-2 variants. bioRxiv 2021, 6, 121. [Google Scholar] [CrossRef]
- Mammen, M.P.; Tebas, P.; Agnes, J.; Giffear, M.; Kraynyak, K.A.; Blackwood, E.; Amante, D.; Reuschel, E.L.; Purwar, M.; Christensen-Quick, A. Safety and immunogenicity of INO-4800 DNA vaccine against SARS-CoV-2: A preliminary report of a randomized, blinded, placebo-controlled, Phase 2 clinical trial in adults at high risk of viral exposure. medRxiv 2021. [Google Scholar] [CrossRef]
- INOVIO. INOVIO Announces Initiation of Phase 2 Segment of its Phase 2/3 Clinical Trial for its COVID-19 DNA Vaccine Candidate, INO-4800. Trial Will Be Funded by the U.S. Department of Defense. Available online: https://ir.inovio.com/news-releases/news-releases-details/2020/INOVIO-Announces-Initiation-of-Phase-2-Segment-of-its-Phase-23-Clinical-Trial-for-its-COVID-19-DNA-Vaccine-Candidate-INO-4800-Trial-Will-Be-Funded-by-the-U.S.-Department-of-Defense/default.aspx (accessed on 23 August 2021).
- INOVIO. INOVIO’s Pan-COVID-19 Vaccine Candidate (INO4802) Induces Broad Immunity Against Major Viral Variants in Preclinical Studies. Available online: https://ir.inovio.com/news-releases/news-releases-details/2021/INOVIOs-Pan-COVID-19-Vaccine-Candidate-INO-4802-Induces-Broad-Immunity-Against-Major-Viral-Variants-in-Preclinical-Studies/default.aspx (accessed on 23 August 2021).
- Ma, H.; Zeng, W.; He, H.; Zhao, D.; Jiang, D.; Zhou, P.; Cheng, L.; Li, Y.; Ma, X.; Jin, T. Serum IgA, IgM, and IgG responses in COVID-19. Cell. Mol. Immunol. 2020, 17, 773–775. [Google Scholar] [CrossRef]
- Han, H.; Ma, Q.; Li, C.; Liu, R.; Zhao, L.; Wang, W.; Zhang, P.; Liu, X.; Gao, G.; Liu, F.; et al. Profiling serum cytokines in COVID-19 patients reveals IL-6 and IL-10 are disease severity predictors. Emerg. Microbes Infect. 2020, 9, 1123–1130. [Google Scholar] [CrossRef]
- Liu, B.M.; Martins, T.B.; Peterson, L.K.; Hill, H.R. Clinical significance of measuring serum cytokine levels as inflammatory biomarkers in adult and pediatric COVID-19 cases: A review. Cytokine 2021, 142, 155478. [Google Scholar] [CrossRef]
- McElvaney, O.J.; McEvoy, N.L.; McElvaney, O.F.; Carroll, T.P.; Murphy, M.P.; Dunlea, D.M.; Ní Choileáin, O.; Clarke, J.; O’Connor, E.; Hogan, G.; et al. Characterization of the inflammatory response to severe COVID-19 illness. Am. J. Respir. Crit. Care Med. 2020, 202, 812–821. [Google Scholar] [CrossRef]
- Ragab, D.; Salah Eldin, H.; Taeimah, M.; Khattab, R.; Salem, R. The COVID-19 cytokine storm; what we know so far. Front. Immunol. 2020, 11, 1446. [Google Scholar] [CrossRef]
- Panigrahy, D.; Gilligan, M.M.; Huang, S.; Gartung, A.; Cortés-Puch, I.; Sime, P.J.; Phipps, R.P.; Serhan, C.N.; Hammock, B.D. Inflammation resolution: A dual-pronged approach to averting cytokine storms in COVID-19? Cancer Metastasis Rev. 2020, 39, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, S.; Sinha, A.; Banach, M.; Mittoo, S.; Weissert, R.; Kass, J.S.; Rajagopal, S.; Pai, A.R.; Kutty, S. Cytokine storm in COVID-19-immunopathological mechanisms, clinical considerations, and therapeutic approaches: The REPROGRAM consortium position paper. Front. Immunol. 2020, 11, 1648. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Wu, D.; Guo, W.; Cao, Y.; Huang, D.; Wang, H.; Wang, T.; Zhang, X.; Chen, H.; Yu, H.; et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Investig. 2020, 130, 2620–2629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Hirano, T.; Murakami, M. COVID-19: A new virus, but a familiar receptor and cytokine release syndrome. Immunity 2020, 52, 731–733. [Google Scholar] [CrossRef]
- Liu, B.; Li, M.; Zhou, Z.; Guan, X.; Xiang, Y. Can we use interleukin-6 (IL-6) blockade for coronavirus disease 2019 (COVID-19)-induced cytokine release syndrome (CRS)? J. Autoimmun. 2020, 111, 102452. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Q.; Guo, D. Emerging coronaviruses: Genome structure, replication, and pathogenesis. J. Med. Virol. 2020, 92, 418–423. [Google Scholar] [CrossRef]
- Challen, R.; Brooks-Pollock, E.; Read, J.M.; Dyson, L.; Tsaneva-Atanasova, K.; Danon, L. Risk of mortality in patients infected with SARS-CoV-2 variant of concern 202012/1: Matched cohort study. BMJ 2021, 372, n579. [Google Scholar] [CrossRef]
- Zhou, D.; Dejnirattisai, W.; Supasa, P.; Liu, C.; Mentzer, A.J.; Ginn, H.M.; Zhao, Y.; Duyvesteyn, H.M.E.; Tuekprakhon, A.; Nutalai, R.; et al. Evidence of escape of SARS-CoV-2 variant B.1.351 from natural and vaccine-induced sera. Cell 2021, 184, 2348–2361.e6. [Google Scholar] [CrossRef]
- Mayo Clinic. COVID-19 Variants: What’s the Concern? Available online: https://www.mayoclinic.org/diseases-conditions/coronavirus/expert-answers/covid-variant/faq-20505779 (accessed on 31 August 2021).
- Hodcroft, E. Variant: 21A (Delta). Available online: https://covariants.org/variants/21A.Delta (accessed on 31 August 2021).
- WHO. Tracking SARS-CoV-2 Variants. Available online: https://www.who.int/en/activities/tracking-SARS-CoV-2-variants/ (accessed on 31 August 2021).
- Hagen, A. How Dangerous Is the Delta Variant (B.1.617.2)? Available online: https://asm.org/Articles/2021/July/How-Dangerous-is-the-Delta-Variant-B-1-617-2 (accessed on 31 August 2021).
- Abbott, B. Delta Variant Accounts for 83% of Known U.S. Covid-19 Cases. Available online: https://www.wsj.com/articles/delta-variant-accounts-for-83-of-known-covid-19-cases-11626807019 (accessed on 31 August 2021).
- WHO. Update on Omicron. Available online: https://www.who.int/news/item/28-11-2021-update-on-omicron (accessed on 19 January 2022).
- Geddes, L. What Do We Know about the New B.1.1.529 Coronavirus Variant and Should We Be Worried? Available online: https://www.gavi.org/vaccineswork/what-we-know-about-new-b11529-coronavirus-variant-so-far?gclid=Cj0KCQiAip-PBhDVARIsAPP2xc3Q_oSb2Vr7HHnELNZmKy2-vrIG_qeiugLkRy3k3mjodpSQfcg_T78aAiVVEALw_wcB (accessed on 19 January 2022).
- Sinha, A. ‘IHU’ Variant of COVID-19 Explained: Few Cases, Limited Spread. Available online: https://indianexpress.com/article/explained/ihu-variant-few-cases-limited-spread-7706641/ (accessed on 19 January 2022).
- Le, T.K.; Paris, C.; Khan, K.S.; Robson, F.; Ng, W.-L.; Rocchi, P. Nucleic acid-based technologies targeting coronaviruses. Trends Biochem. Sci. 2021, 46, 351–365. [Google Scholar] [CrossRef]
- Abbott, T.R.; Dhamdhere, G.; Liu, Y.; Lin, X.; Goudy, L.; Zeng, L.; Chemparathy, A.; Chmura, S.; Heaton, N.S.; Debs, R.; et al. Development of CRISPR as an antiviral strategy to combat SARS-CoV-2 and influenza. Cell 2020, 181, 865–876.e12. [Google Scholar] [CrossRef] [PubMed]
- Sridharan, K.; Gogtay, N.J. Therapeutic nucleic acids: Current clinical status. Br. J. Clin. Pharmacol. 2016, 82, 659–672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gewirtz, A.M.; Sokol, D.L.; Ratajczak, M.Z. Nucleic acid therapeutics: State of the art and future prospects. Blood 1998, 92, 712–736. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.P.; Yang, M.; Lai, C.L. COVID-19 Vaccines: A Review of the Safety and Efficacy of Current Clinical Trials. Pharmaceuticals 2021, 14, 406. [Google Scholar] [CrossRef]
- Dash, P.; Mohapatra, S.; Ghosh, S.; Nayak, B. A Scoping Insight on Potential Prophylactics, Vaccines and Therapeutic Weaponry for the Ongoing Novel Coronavirus (COVID-19) Pandemic- A Comprehensive Review. Front. Pharmacol. 2020, 11, 590154. [Google Scholar] [CrossRef]
- Flanagan, K.L.; Best, E.; Crawford, N.W.; Giles, M.; Koirala, A.; Macartney, K.; Russell, F.; Teh, B.W.; Wen, S.C. Progress and Pitfalls in the Quest for Effective SARS-CoV-2 (COVID-19) Vaccines. Front. Immunol. 2020, 11, 579250. [Google Scholar] [CrossRef]
- Berber, B.; Aydin, C.; Kocabas, F.; Guney-Esken, G.; Yilancioglu, K.; Karadag-Alpaslan, M.; Caliseki, M.; Yuce, M.; Demir, S.; Tastan, C. Gene editing and RNAi approaches for COVID-19 diagnostics and therapeutics. Gene Ther. 2021, 28, 290–305. [Google Scholar] [CrossRef]
- Kim, D.; Rossi, J. RNAi mechanisms and applications. BioTechniques 2008, 44, 613–616. [Google Scholar] [CrossRef]
- Lee, Y.; Hur, I.; Park, S.-Y.; Kim, Y.-K.; Suh, M.R.; Kim, V.N. The role of PACT in the RNA silencing pathway. EMBO J. 2006, 25, 522–532. [Google Scholar] [CrossRef]
- Settleman, J.; Sawyers, C.L.; Hunter, T. Challenges in validating candidate therapeutic targets in cancer. eLife 2018, 7, e32402. [Google Scholar] [CrossRef] [Green Version]
- Deng, Y.; Wang, C.C.; Choy, K.W.; Du, Q.; Chen, J.; Wang, Q.; Li, L.; Chung, T.K.H.; Tang, T. Therapeutic potentials of gene silencing by RNA interference: Principles, challenges, and new strategies. Gene 2014, 538, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Elbashir, S.M.; Harborth, J.; Lendeckel, W.; Yalcin, A.; Weber, K.; Tuschl, T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 2001, 411, 494–498. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, I.G.; Khayat, A.S.; Stransky, B.; Santos, S.; Assumpção, P.; de Souza, J.E.S. A small interfering RNA (siRNA) database for SARS-CoV-2. Sci. Rep. 2021, 11, 8849. [Google Scholar] [CrossRef]
- Chen, W.; Feng, P.; Liu, K.; Wu, M.; Lin, H. Computational identification of small interfering RNA targets in SARS-CoV-2. Virol. Sin. 2020, 35, 359–361. [Google Scholar] [CrossRef] [Green Version]
- Idris, A.; Davis, A.; Supramaniam, A.; Acharya, D.; Kelly, G.; Tayyar, Y.; West, N.; Zhang, P.; McMillan, C.L.; Soemardy, C. A SARS-CoV-2 targeted siRNA-nanoparticle therapy for COVID-19. Mol. Ther. 2021, 29, 2219–2226. [Google Scholar] [CrossRef]
- Niktab, I.; Haghparast, M.; Beigi, M.-H.; Megraw, T.L.; Kiani, A.; Ghaedi, K. Design of advanced siRNA therapeutics for the treatment of COVID-19. Meta Gene 2021, 29, 100910. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhou, Q.; Li, Y.; Garner, L.V.; Watkins, S.P.; Carter, L.J.; Smoot, J.; Gregg, A.C.; Daniels, A.D.; Jervey, S.; et al. Research and development on therapeutic agents and vaccines for COVID-19 and related human coronavirus diseases. ACS Cent. Sci. 2020, 6, 315–331. [Google Scholar] [CrossRef]
- Ullah, A.; Qazi, J.; Rahman, L.; Kanaras, A.G.; Khan, W.S.; Hussain, I.; Rehman, A. Nanoparticles-assisted delivery of antiviral-siRNA as inhalable treatment for human respiratory viruses: A candidate approach against SARS-COV-2. Nano Sel. 2020, 1, 612–621. [Google Scholar] [CrossRef]
- Farr, R.; Rootes, C.; Rowntree, L.; Nguyen, T.; Hensen, L.; Kedzierski, L.; Cheng, A.; Kedzierska, K.; Au, G.; Marsh, G. Altered microRNA expression in COVID-19 patients enables identification of SARS-CoV-2 infection. PLoS Pathog. 2021, 17, e1009759. [Google Scholar] [CrossRef]
- Friedman, R.C.; Farh, K.K.-H.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105. [Google Scholar] [CrossRef] [Green Version]
- Hum, C.; Loiselle, J.; Ahmed, N.; Shaw, T.A.; Toudic, C.; Pezacki, J.P. MicroRNA mimics or inhibitors as antiviral therapeutic approaches against COVID-19. Drugs 2021, 81, 517–531. [Google Scholar] [CrossRef]
- Balmeh, N.; Mahmoudi, S.; Mohammadi, N.; Karabedianhajiabadi, A. Predicted therapeutic targets for COVID-19 disease by inhibiting SARS-CoV-2 and its related receptors. Inform. Med. Unlocked 2020, 20, 100407. [Google Scholar] [CrossRef] [PubMed]
- Narozna, M.; Rubis, B. Anti-SARS-CoV-2 Strategies and the Potential Role of miRNA in the Assessment of COVID-19 Morbidity, Recurrence, and Therapy. Int. J. Mol. Sci. 2021, 22, 8663. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Lee, Y.; Yeom, K.-H.; Nam, J.-W.; Heo, I.; Rhee, J.-K.; Sohn, S.Y.; Cho, Y.; Zhang, B.-T.; Kim, V.N. Molecular basis for the recognition of primary microRNAs by the Drosha-DGCR8 complex. Cell 2006, 125, 887–901. [Google Scholar] [CrossRef] [Green Version]
- Catalanotto, C.; Cogoni, C.; Zardo, G. MicroRNA in control of gene expression: An overview of nuclear functions. Int. J. Mol. Sci. 2016, 17, 1712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, J.; Stojanovic, J.; Yasamineh, S.; Yasamineh, P.; Karuppannan, S.K.; Hussain Dowlath, M.J.; Serati-Nouri, H. The potential use of microRNAs as a therapeutic strategy for SARS-CoV-2 infection. Arch. Virol. 2021, 166, 2649–2672. [Google Scholar] [CrossRef]
- Gallant-Behm, C.L.; Piper, J.; Lynch, J.M.; Seto, A.G.; Hong, S.J.; Mustoe, T.A.; Maari, C.; Pestano, L.A.; Dalby, C.M.; Jackson, A.L. A microRNA-29 mimic (Remlarsen) represses extracellular matrix expression and fibroplasia in the skin. J. Investig. Dermatol. 2019, 139, 1073–1081. [Google Scholar] [CrossRef]
- Seto, A.G.; Beatty, X.; Lynch, J.M.; Hermreck, M.; Tetzlaff, M.; Duvic, M.; Jackson, A.L. Cobomarsen, an oligonucleotide inhibitor of miR-155, co-ordinately regulates multiple survival pathways to reduce cellular proliferation and survival in cutaneous T-cell lymphoma. Br. J. Haematol. 2018, 183, 428–444. [Google Scholar] [CrossRef] [Green Version]
- Lam, J.K.W.; Chow, M.Y.T.; Zhang, Y.; Leung, S.W.S. siRNA Versus miRNA as therapeutics for gene silencing. Mol. Ther. Nucleic Acids 2015, 4, e252. [Google Scholar] [CrossRef] [Green Version]
- Iorio, M.V.; Croce, C.M. MicroRNA dysregulation in cancer: Diagnostics, monitoring and therapeutics. A comprehensive review. EMBO Mol. Med. 2012, 4, 143–159. [Google Scholar] [CrossRef]
- Hosseini Rad Sm, A.; McLellan, A.D. Implications of SARS-CoV-2 mutations for genomic RNA structure and host microRNA targeting. Int. J. Mol. Sci. 2020, 21, 4807. [Google Scholar] [CrossRef] [PubMed]
- Maitra, A.; Sarkar, M.C.; Raheja, H.; Biswas, N.K.; Chakraborti, S.; Singh, A.K.; Ghosh, S.; Sarkar, S.; Patra, S.; Mondal, R.K.; et al. Mutations in SARS-CoV-2 viral RNA identified in Eastern India: Possible implications for the ongoing outbreak in India and impact on viral structure and host susceptibility. J. Biosci. 2020, 45, 76. [Google Scholar] [CrossRef]
- Lu, D.; Chatterjee, S.; Xiao, K.; Riedel, I.; Wang, Y.; Foo, R.; Bär, C.; Thum, T. MicroRNAs targeting the SARS-CoV-2 entry receptor ACE2 in cardiomyocytes. J. Mol. Cell. Cardiol. 2020, 148, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Matarese, A.; Gambardella, J.; Sardu, C.; Santulli, G. miR-98 regulates TMPRSS2 expression in human endothelial cells: Key implications for COVID-19. Biomedicines 2020, 8, 462. [Google Scholar] [CrossRef] [PubMed]
- Kaur, T.; Kapila, S.; Kapila, R.; Kumar, S.; Upadhyay, D.; Kaur, M.; Sharma, C. Tmprss2 specific miRNAs as promising regulators for SARS-CoV-2 entry checkpoint. Virus Res. 2021, 294, 198275. [Google Scholar] [CrossRef] [PubMed]
- Sardar, R.; Satish, D.; Birla, S.; Gupta, D. Dataset of mutational analysis, miRNAs targeting SARS-CoV-2 genes and host gene expression in SARS-CoV and SARS-CoV-2 infections. Data Brief. 2020, 32, 106207. [Google Scholar] [CrossRef]
- Chow, J.T.-S.; Salmena, L. Prediction and analysis of SARS-CoV-2-targeting microRNA in human lung epithelium. Genes 2020, 11, 1002. [Google Scholar] [CrossRef]
- Ge, J.; Li, J.; Na, S.; Wang, P.; Zhao, G.; Zhang, X. miR-548c-5p inhibits colorectal cancer cell proliferation by targeting PGK1. J. Cell. Physiol. 2019, 234, 18872–18878. [Google Scholar] [CrossRef]
- Fulzele, S.; Sahay, B.; Yusufu, I.; Lee, T.J.; Sharma, A.; Kolhe, R.; Isales, C.M. COVID-19 virulence in aged patients might be impacted by the host cellular microRNAs abundance/profile. Aging Dis. 2020, 11, 509–522. [Google Scholar] [CrossRef]
- El-Nabi, S.H.; Elhiti, M.; El-Sheekh, M. A new approach for COVID-19 treatment by micro-RNA. Med. Hypotheses 2020, 143, 110203. [Google Scholar] [CrossRef]
- Khan, M.A.-A.-K.; Sany, M.R.U.; Islam, M.S.; Islam, A.B.M.M.K. Epigenetic regulator miRNA pattern differences among SARS-CoV, SARS-CoV-2, and SARS-CoV-2 world-wide isolates delineated the mystery behind the epic pathogenicity and distinct clinical characteristics of pandemic COVID-19. Front. Genet. 2020, 11, 765. [Google Scholar] [CrossRef] [PubMed]
- Saçar Demirci, M.D.; Adan, A. Computational analysis of microRNA-mediated interactions in SARS-CoV-2 infection. PeerJ 2020, 8, e9369. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.S.S.J.; Paramasivam, P.; Raj, K.; Kumar, V.; Murugesan, R.; Ramakrishnan, V. Regulatory cross talk between SARS-CoV-2 receptor binding and replication machinery in the human host. Front. Physiol. 2020, 11, 802. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.T.-A.; Khalid, Z.; Zahid, H.; Yousaf, M.A.; Shakoori, A.R. A computational and bioinformatic analysis of ACE2: An elucidation of its dual role in COVID-19 pathology and finding its associated partners as potential therapeutic targets. J. Biomol. Struct. Dyn. 2020, 40, 1813–1829. [Google Scholar] [CrossRef]
- Zhang, H.; Rostami, M.R.; Leopold, P.L.; Mezey, J.G.; O’Beirne, S.L.; Strulovici-Barel, Y.; Crystal, R.G. Expression of the SARS-CoV-2 ACE2 receptor in the human airway epithelium. Am. J. Respir. Crit. Care Med. 2020, 202, 219–229. [Google Scholar] [CrossRef]
- Nersisyan, S.; Shkurnikov, M.; Turchinovich, A.; Knyazev, E.; Tonevitsky, A. Integrative analysis of miRNA and mRNA sequencing data reveals potential regulatory mechanisms of ACE2 and TMPRSS2. PLoS ONE 2020, 15, e0235987. [Google Scholar] [CrossRef]
- Nersisyan, S.A.; Shkurnikov, M.Y.; Osipyants, A.I.; Vechorko, V.I. Role of ACE2/TMPRSS2 genes regulation by intestinal microRNA isoforms in the COVID-19 pathogenesis. Bull. Russ. State Med. Univ. 2020, 16–18. [Google Scholar] [CrossRef]
- Arisan, E.D.; Dart, A.; Grant, G.H.; Arisan, S.; Cuhadaroglu, S.; Lange, S.; Uysal-Onganer, P. The prediction of miRNAs in SARS-CoV-2 genomes: Hsa-miR databases identify 7 key miRs linked to host responses and virus pathogenicity-related KEGG pathways significant for comorbidities. Viruses 2020, 12, 614. [Google Scholar] [CrossRef]
- Chen, L.; Zhong, L. Genomics functional analysis and drug screening of SARS-CoV-2. Genes Dis. 2020, 7, 542–550. [Google Scholar] [CrossRef]
- Sardar, R.; Satish, D.; Birla, S.; Gupta, D. Integrative analyses of SARS-CoV-2 genomes from different geographical locations reveal unique features potentially consequential to host-virus interaction, pathogenesis and clues for novel therapies. Heliyon 2020, 6, e04658. [Google Scholar] [CrossRef]
- Mukhopadhyay, D.; Mussa, B.M. Identification of novel hypothalamic microRNAs as promising therapeutics for SARS-CoV-2 by regulating ACE2 and TMPRSS2 expression: An in silico analysis. Brain Sci. 2020, 10, 666. [Google Scholar] [CrossRef] [PubMed]
- Taz, T.A.; Ahmed, K.; Paul, B.K.; Kawsar, M.; Aktar, N.; Mahmud, S.M.H.; Moni, M.A. Network-based identification genetic effect of SARS-CoV-2 infections to Idiopathic pulmonary fibrosis (IPF) patients. Brief. Bioinform. 2021, 22, 1254–1266. [Google Scholar] [CrossRef] [PubMed]
- Vastrad, B.; Vastrad, C.; Tengli, A. Identification of potential mRNA panels for severe acute respiratory syndrome coronavirus 2 (COVID-19) diagnosis and treatment using microarray dataset and bioinformatics methods. 3 Biotech 2020, 10, 422. [Google Scholar] [CrossRef] [PubMed]
- Marchi, R.; Sugita, B.; Centa, A.; Fonseca, A.S.; Bortoletto, S.; Fiorentin, K.; Ferreira, S.; Cavalli, L.R. The role of microRNAs in modulating SARS-CoV-2 infection in human cells: A systematic review. Infection, genetics and evolution. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2021, 91, 104832. [Google Scholar] [CrossRef]
- Zhang, S.; Amahong, K.; Sun, X.; Lian, X.; Liu, J.; Sun, H.; Lou, Y.; Zhu, F.; Qiu, Y. The miRNA: A small but powerful RNA for COVID-19. Brief. Bioinform. 2021, 22, 1137–1149. [Google Scholar] [CrossRef]
- Fani, M.; Zandi, M.; Ebrahimi, S.; Soltani, S.; Abbasi, S. The role of miRNAs in COVID-19 disease. Future Virol. 2021, 16, 301–306. [Google Scholar] [CrossRef]
- Gambardella, J.; Sardu, C.; Morelli, M.B.; Messina, V.; Castellanos, V.; Marfella, R.; Maggi, P.; Paolisso, G.; Wang, X.; Santulli, G. Exosomal microRNAs drive thrombosis in COVID-19. medRxiv 2020, 142. [Google Scholar] [CrossRef]
- Gallagher, T.M.; Buchmeier, M.J. Coronavirus spike proteins in viral entry and pathogenesis. Virology 2001, 279, 371–374. [Google Scholar] [CrossRef] [Green Version]
- Alam, T.; Lipovich, L. miRCOVID-19: Potential targets of human miRNAs in SARS-CoV-2 for RNA-based drug discovery. Non-Coding RNA 2021, 7, 18. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, S.; Li, F.; Zhou, Y.; Zhang, Y.; Wang, Z.; Zhang, R.; Zhu, J.; Ren, Y.; Tan, Y.; et al. Therapeutic target database 2020: Enriched resource for facilitating research and early development of targeted therapeutics. Nucleic Acids Res. 2020, 48, D1031–D1041. [Google Scholar] [CrossRef] [Green Version]
- Mann, M.; Wright, P.R.; Backofen, R. IntaRNA 2.0: Enhanced and customizable prediction of RNA-RNA interactions. Nucleic Acids Res. 2017, 45, W435–W439. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, W.; Flamant, S.; Rasko, J.E.J. mimiRNA: A microRNA expression profiler and classification resource designed to identify functional correlations between microRNAs and their targets. Bioinformatics 2010, 26, 223–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dai, X.; Zhao, P.X. psRNATarget: A plant small RNA target analysis server. Nucleic Acids Res. 2011, 39, W155–W159. [Google Scholar] [CrossRef] [Green Version]
- Betel, D.; Wilson, M.; Gabow, A.; Marks, D.S.; Sander, C. The microRNA.org resource: Targets and expression. Nucleic Acids Res. 2008, 36, D149–D153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chou, C.-H.; Shrestha, S.; Yang, C.-D.; Chang, N.-W.; Lin, Y.-L.; Liao, K.-W.; Huang, W.-C.; Sun, T.-H.; Tu, S.-J.; Lee, W.-H.; et al. miRTarBase update 2018: A resource for experimentally validated microRNA-target interactions. Nucleic Acids Res. 2018, 46, D296–D302. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-Y.; Lin, Y.-C.-D.; Li, J.; Huang, K.-Y.; Shrestha, S.; Hong, H.-C.; Tang, Y.; Chen, Y.-G.; Jin, C.-N.; Yu, Y.; et al. miRTarBase 2020: Updates to the experimentally validated microRNA-target interaction database. Nucleic Acids Res. 2020, 48, D148–D154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pardi, N.; Hogan, M.J.; Porter, F.W.; Weissman, D. mRNA vaccines—a new era in vaccinology. Nat. Rev. Drug Discov. 2018, 17, 261–279. [Google Scholar] [CrossRef] [Green Version]
- Yi, C.; Yi, Y.; Li, J. mRNA vaccines: Possible tools to combat SARS-CoV-2. Virol. Sin. 2020, 35, 259–262. [Google Scholar] [CrossRef]
- McNamara, M.A.; Nair, S.K.; Holl, E.K. RNA-based vaccines in cancer immunotherapy. J. Immunol. Res. 2015, 2015, 794528. [Google Scholar] [CrossRef] [Green Version]
- Rosenberg, Y.; Sack, M.; Montefiori, D.; Labranche, C.; Lewis, M.; Urban, L.; Mao, L.; Fischer, R.; Jiang, X. Pharmacokinetics and immunogenicity of broadly neutralizing HIV monoclonal antibodies in macaques. PLoS ONE 2015, 10, e0120451. [Google Scholar] [CrossRef]
- Kamboj, M.; Sepkowitz, K.A. Risk of transmission associated with live attenuated vaccines given to healthy persons caring for or residing with an immunocompromised patient. Infect. Control. Hosp. Epidemiol. 2007, 28, 702–707. [Google Scholar] [CrossRef]
- Lim, B.; Lee, K. Stability of the osmoregulated promoter-derived prop mRNA is posttranscriptionally regulated by RNase III in Escherichia coli. J. Bacteriol. 2015, 197, 1297–1305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pardi, N.; Weissman, D. Nucleoside modified mRNA vaccines for infectious diseases. Methods Mol. Biol. 2017, 1499, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Schlake, T.; Thess, A.; Thran, M.; Jordan, I. mRNA as novel technology for passive immunotherapy. Cell. Mol. Life Sci. 2019, 76, 301–328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jackson, N.A.C.; Kester, K.E.; Casimiro, D.; Gurunathan, S.; DeRosa, F. The promise of mRNA vaccines: A biotech and industrial perspective. Npj Vaccines 2020, 5, 11. [Google Scholar] [CrossRef]
- Wang, F.; Kream, R.M.; Stefano, G.B. An evidence based perspective on mRNA-SARS-CoV-2 vaccine development. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2020, 26, e924700. [Google Scholar] [CrossRef] [Green Version]
- Crommelin, D.J.A.; Anchordoquy, T.J.; Volkin, D.B.; Jiskoot, W.; Mastrobattista, E. Addressing the cold reality of mRNA vaccine stability. J. Pharm. Sci. 2021, 110, 997–1001. [Google Scholar] [CrossRef]
- Reichmuth, A.M.; Oberli, M.A.; Jaklenec, A.; Langer, R.; Blankschtein, D. mRNA vaccine delivery using lipid nanoparticles. Ther. Deliv. 2016, 7, 319–334. [Google Scholar] [CrossRef] [Green Version]
- Vigerust, D.J.; Shepherd, V.L. Virus glycosylation: Role in virulence and immune interactions. Trends Microbiol. 2007, 15, 211–218. [Google Scholar] [CrossRef]
- Corbett, K.S.; Edwards, D.K.; Leist, S.R.; Abiona, O.M.; Boyoglu-Barnum, S.; Gillespie, R.A.; Himansu, S.; Schäfer, A.; Ziwawo, C.T.; DiPiazza, A.T.; et al. SARS-CoV-2 mRNA vaccine design enabled by prototype pathogen preparedness. Nature 2020, 586, 567–571. [Google Scholar] [CrossRef]
- Xie, X.; Liu, Y.; Liu, J.; Zhang, X.; Zou, J.; Fontes-Garfias, C.R.; Xia, H.; Swanson, K.A.; Cutler, M.; Cooper, D.; et al. Neutralization of SARS-CoV-2 spike 69/70 deletion, E484K and N501Y variants by BNT162b2 vaccine-elicited sera. Nat. Med. 2021, 27, 620–621. [Google Scholar] [CrossRef]
- Skowronski, D.M.; Serres, G.D. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N. Engl. J. Med. 2021, 384, 1576–1578. [Google Scholar] [CrossRef] [PubMed]
- Haas, E.J.; Angulo, F.J.; McLaughlin, J.M.; Anis, E.; Singer, S.R.; Khan, F.; Brooks, N.; Smaja, M.; Mircus, G.; Pan, K.; et al. Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: An observational study using national surveillance data. Lancet 2021, 397, 1819–1829. [Google Scholar] [CrossRef]
- Simpson, C.R.; Shi, T.; Vasileiou, E.; Katikireddi, S.V.; Kerr, S.; Moore, E.; McCowan, C.; Agrawal, U.; Shah, S.A.; Ritchie, L.D.; et al. First-dose ChAdOx1 and BNT162b2 COVID-19 vaccines and thrombocytopenic, thromboembolic and hemorrhagic events in Scotland. Nat. Med. 2021, 27, 1290–1297. [Google Scholar] [CrossRef] [PubMed]
- Rauch, S.; Jasny, E.; Schmidt, K.E.; Petsch, B. New vaccine technologies to combat outbreak situations. Front. Immunol. 2018, 9, 1963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahimi, H.; Salehiabar, M.; Barsbay, M.; Ghaffarlou, M.; Kavetskyy, T.; Sharafi, A.; Davaran, S.; Chauhan, S.C.; Danafar, H.; Kaboli, S.; et al. CRISPR systems for COVID-19 diagnosis. ACS Sens. 2021, 6, 1430–1445. [Google Scholar] [CrossRef]
- Freije, C.A.; Myhrvold, C.; Boehm, C.K.; Lin, A.E.; Welch, N.L.; Carter, A.; Metsky, H.C.; Luo, C.Y.; Abudayyeh, O.O.; Gootenberg, J.S.; et al. Programmable inhibition and detection of RNA viruses using Cas13. Mol. Cell 2019, 76, 826–837.e11. [Google Scholar] [CrossRef] [Green Version]
- Barrey, E.; Burzio, V.; Dhorne-Pollet, S.; Eléouët, J.-F.; Delmas, B. Think Different with RNA Therapy: Can Antisense Oligonucleotides Be Used to Inhibit Replication and Transcription of SARS-Cov-2? Preprints 2020, 2020040412. [Google Scholar] [CrossRef] [Green Version]
- Gasparello, J.; Finotti, A.; Gambari, R. Tackling the COVID-19 “cytokine storm” with microRNA mimics directly targeting the 3′UTR of pro-inflammatory mRNAs. Med. Hypotheses 2021, 146, 110415. [Google Scholar] [CrossRef]
- Centa, A.; Fonseca, A.S.; da Silva Ferreira, S.G.; Azevedo, M.L.V.; Vaz de Paula, C.B.; Nagashima, S.; Machado-Souza, C.; dos Santos Miggiolaro, A.F.R.; Baena, C.P.; de Noronha, L.; et al. Deregulated miRNA expression is associated with endothelial dysfunction in post-mortem lung biopsies of COVID-19 patients. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 320, L405–L412. [Google Scholar] [CrossRef]
- Wicik, Z.; Eyileten, C.; Jakubik, D.; Simões, S.N.; Martins, D.C.; Pavão, R.; Siller-Matula, J.M.; Postula, M. ACE2 interaction networks in COVID-19: A physiological framework for prediction of outcome in patients with cardiovascular risk factors. J. Clin. Med. 2020, 9, 3743. [Google Scholar] [CrossRef] [PubMed]
- Nersisyan, S.; Engibaryan, N.; Gorbonos, A.; Kirdey, K.; Makhonin, A.; Tonevitsky, A. Potential role of cellular miRNAs in coronavirus-host interplay. PeerJ 2020, 8, e9994. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Takizawa, S.; Ohgaku, Y.; Asami, T.; Furuya, K.; Yamamoto, K.; Takahashi, F.; Hamajima, C.; Inaba, C.; Endo, K.; et al. MicroRNA 16-5p is upregulated in calorie-restricted mice and modulates inflammatory cytokines of macrophages. Gene 2020, 725, 144191. [Google Scholar] [CrossRef]
- Ye, E.-A.; Liu, L.; Jiang, Y.; Jan, J.; Gaddipati, S.; Suvas, S.; Steinle, J.J. miR-15a/16 reduces retinal leukostasis through decreased pro-inflammatory signaling. J. Neuroinflamm. 2016, 13, 305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, C.-M.; Wu, J.; Zhang, H.; Shi, G.; Chen, Z.-T. Circulating miR-125a but not miR-125b is decreased in active disease status and negatively correlates with disease severity as well as inflammatory cytokines in patients with Crohn’s disease. World J. Gastroenterol. 2017, 23, 7888–7898. [Google Scholar] [CrossRef] [PubMed]
- Uludağ, H.; Parent, K.; Aliabadi, H.M.; Haddadi, A. Prospects for RNAi therapy of COVID-19. Front. Bioeng. Biotechnol. 2020, 8, 916. [Google Scholar] [CrossRef]
- Mai, J.; Virtue, A.; Maley, E.; Tran, T.; Yin, Y.; Meng, S.; Pansuria, M.; Jiang, X.; Wang, H.; Yang, X.-F. MicroRNAs and other mechanisms regulate interleukin-17 cytokines and receptors. Front. Biosci. (Elite Ed.) 2012, 4, 1478–1495. [Google Scholar] [CrossRef]
- Chen, X.; Zhou, L.; Peng, N.; Yu, H.; Li, M.; Cao, Z.; Lin, Y.; Wang, X.; Li, Q.; Wang, J.; et al. MicroRNA-302a suppresses influenza A virus-stimulated interferon regulatory factor-5 expression and cytokine storm induction. J. Biol. Chem. 2017, 292, 21291–21303. [Google Scholar] [CrossRef] [Green Version]
- Desjarlais, M.; Wirth, M.; Lahaie, I.; Ruknudin, P.; Hardy, P.; Rivard, A.; Chemtob, S. Nutraceutical targeting of inflammation-modulating microRNAs in severe forms of COVID-19: A novel approach to prevent the cytokine storm. Front. Pharmacol. 2020, 11, 602999. [Google Scholar] [CrossRef]
- Gangemi, S.; Tonacci, A. AntagomiRs: A novel therapeutic strategy for challenging COVID-19 cytokine storm. Cytokine Growth Factor Rev. 2021, 58, 111–113. [Google Scholar] [CrossRef]
- Chen, B.-B.; Li, Z.-H.; Gao, S. Circulating miR-146a/b correlates with inflammatory cytokines in COPD and could predict the risk of acute exacerbation COPD. Medicine 2018, 97, e9820. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Guan, H.; Zhang, J.-L.; Zheng, Z.; Wang, H.-T.; Tao, K.; Han, S.-C.; Su, L.-L.; Hu, D. Acute downregulation of miR-199a attenuates sepsis-induced acute lung injury by targeting SIRT1. Am. J. Physiol. Cell Physiol. 2018, 314, C449–C455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, H.-L.; Nie, Z.-Q.; Lu, Y.; Yang, X.; Song, C.; Chen, H.; Zhu, S.; Chen, B.-B.; Huang, J.; Geng, S.; et al. Circulating miR-125b but not miR-125a correlates with acute exacerbations of chronic obstructive pulmonary disease and the expressions of inflammatory cytokines. Medicine 2017, 96, e9059. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Seeliger, B.; Derda, A.A.; Xiao, K.; Gietz, A.; Scherf, K.; Sonnenschein, K.; Pink, I.; Hoeper, M.M.; Welte, T.; et al. Circulating cardiovascular microRNAs in critically ill COVID-19 patients. Eur. J. Heart Fail. 2021, 23, 468–475. [Google Scholar] [CrossRef]
- Sabbatinelli, J.; Giuliani, A.; Matacchione, G.; Latini, S.; Laprovitera, N.; Pomponio, G.; Ferrarini, A.; Svegliati Baroni, S.; Pavani, M.; Moretti, M.; et al. Decreased serum levels of the inflammaging marker miR-146a are associated with clinical non-response to tocilizumab in COVID-19 patients. Mech. Ageing Dev. 2021, 193, 111413. [Google Scholar] [CrossRef]
- Kalhori, M.R.; Saadatpour, F.; Arefian, E.; Soleimani, M.; Farzaei, M.H.; Aneva, I.Y.; Echeverria, J. The Potential Therapeutic Effect of RNA Interference and Natural Products on COVID-19: A Review of the Coronaviruses Infection. Front. Pharmacol. 2021, 12, 616993. [Google Scholar] [CrossRef]
- Chen, Z.-M.; Fu, J.-F.; Shu, Q.; Chen, Y.-H.; Hua, C.-Z.; Li, F.-B.; Lin, R.; Tang, L.-F.; Wang, T.-L.; Wang, W. Diagnosis and treatment recommendations for pediatric respiratory infection caused by the 2019 novel coronavirus. World J. Pediatr. 2020, 16, 240–246. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Zhu, M.; Ye, L.; Chen, C.; She, J.; Song, Y. MiR-29b-3p promotes particulate matter-induced inflammatory responses by regulating the C1QTNF6/AMPK pathway. Aging 2020, 12, 1141–1158. [Google Scholar] [CrossRef]
- Plowman, T.; Lagos, D. Non-Coding RNAs in COVID-19: Emerging Insights and Current Questions. Non-Coding RNA 2021, 7, 54. [Google Scholar] [CrossRef]
- OECD. Access to COVID-19 Vaccines: Global Approaches in a Global Crisis. Available online: https://www.oecd.org/coronavirus/policy-responses/access-to-covid-19-vaccines-global-approaches-in-a-global-crisis-c6a18370/ (accessed on 31 August 2021).
- Zhang, C.; Maruggi, G.; Shan, H.; Li, J. Advances in mRNA Vaccines for Infectious Diseases. Front. Immunol. 2019, 10, 594. [Google Scholar] [CrossRef] [Green Version]
- Kulkarni, J.A.; Witzigmann, D.; Thomson, S.B.; Chen, S.; Leavitt, B.R.; Cullis, P.R.; van der Meel, R. The current landscape of nucleic acid therapeutics. Nat. Nanotechnol. 2021, 16, 630–643. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Arun Kumar, S.; Jhan, Y.Y.; Bishop, C.J. Engineering DNA vaccines against infectious diseases. Acta Biomater. 2018, 80, 31–47. [Google Scholar] [CrossRef] [PubMed]
- Scoles, D.R.; Minikel, E.V.; Pulst, S.M. Antisense oligonucleotides: A primer. Neurol. Genet. 2019, 5, e323. [Google Scholar] [CrossRef] [Green Version]
- Duharte, A.B. Antisense Oligonucleotides for Vaccine Improvement. Available online: https://encyclopedia.pub/11540 (accessed on 24 August 2021).
- Ying, H.; Ebrahimi, M.; Keivan, M.; Khoshnam, S.E.; Salahi, S.; Farzaneh, M. miRNAs; a novel strategy for the treatment of COVID-19. Cell Biol. Int. 2021, 45, 2045–2053. [Google Scholar] [CrossRef]
- Mehta, A.; Michler, T.; Merkel, O.M. siRNA Therapeutics against Respiratory Viral Infections-What Have We Learned for Potential COVID-19 Therapies? Adv. Health Mater. 2021, 10, e2001650. [Google Scholar] [CrossRef] [PubMed]
- Karolinska Institutet. Protective Gene Variant against COVID-19 Identified. Available online: https://www.sciencedaily.com/releases/2022/01/220113120742.htm (accessed on 19 January 2022).
- Huffman, J.E.; Butler-Laporte, G.; Khan, A.; Pairo-Castineira, E.; Drivas, T.G.; Peloso, G.M.; Nakanishi, T.; Ganna, A.; Verma, A.; Baillie, J.K. Multi-ancestry fine mapping implicates OAS1 splicing in risk of severe COVID-19. Nature Genet. 2022, 54, 125–127. [Google Scholar] [CrossRef]
- Pairo-Castineira, E.; Clohisey, S.; Klaric, L.; Bretherick, A.D.; Rawlik, K.; Pasko, D.; Walker, S.; Parkinson, N.; Fourman, M.H.; Russell, C.D. Genetic mechanisms of critical illness in COVID-19. Nature 2021, 591, 92–98. [Google Scholar] [CrossRef]
- Niemi, M.E.; Karjalainen, J.; Liao, R.G.; Neale, B.M.; Daly, M.; Ganna, A.; Pathak, G.A.; Andrews, S.J.; Kanai, M.; Veerapen, K. Mapping the human genetic architecture of COVID-19. Nature 2021, 600, 472–477. [Google Scholar]
- Zeberg, H.; Pääbo, S. A genomic region associated with protection against severe COVID-19 is inherited from Neandertals. Proc. Natl. Acad. Sci. USA 2021, 118, e2026309118. [Google Scholar] [CrossRef]
- Bonnevie-Nielsen, V.; Field, L.L.; Lu, S.; Zheng, D.-J.; Li, M.; Martensen, P.M.; Nielsen, T.B.; Beck-Nielsen, H.; Lau, Y.-L.; Pociot, F. Variation in antiviral 2′, 5′-oligoadenylate synthetase (2′ 5′ AS) enzyme activity is controlled by a single-nucleotide polymorphism at a splice-acceptor site in the OAS1 gene. Am. J. Hum. Genet. 2005, 76, 623–633. [Google Scholar] [CrossRef] [Green Version]
- UK Research and Inovation. Five Genes Identified That Could Be Key to New COVID-19 Treatments. Available online: https://www.ukri.org/news/five-genes-identified-that-could-be-key-to-new-covid-19-treatments/ (accessed on 22 January 2022).
- Meara, K. Genes May Be Key to New COVID-19 Treatments. Available online: https://www.contagionlive.com/view/genes-may-be-key-to-new-covid-19-treatments (accessed on 22 January 2022).
| Patent ID | Description | Publication Number/ Application Number | CAS RN | Target Region | Reference |
|---|---|---|---|---|---|
| CN101173275 | Two dsRNAs were designed to target two regions of the SARS protein M mRNA | 101173275/200610114168.0 | 1023405-01-7 1023405-02-8 | 220–241 region of M1 protein | [159] |
| 1023405-03-9 1023405-04-0 | 460–480 region of M2 protein | [159] | |||
| US20050004063 | Six siRNAs were designed to target mRNA sequences of replicase A1 gene region | 20050004063/10848737 | 821121-38-1 | 512–531 bp of replicase A1 | [159,169] |
| 821121-38-2 | 586–604 bp of replicase A1 | ||||
| 821121-38-3 | 916–934 bp of replicase A1 | ||||
| 821121-38-4 | 1194–1213 bp of replicase A1 | ||||
| 821121-38-5 | 3028–3046 bp of replicase A1 | ||||
| 821121-38-6 | 5024–5042 bp of replicase A1 | ||||
| CN1569233 | siRNAs were designed to target RdRp, helicase, nucleoprotein N, and proteolytic enzymes | 1569233/03147398.9 | 872062-80-1 | RdRp | [159,169] |
| 872062-81-2 | Helicase | ||||
| 872062-82-3 | Nucleoprotein N | ||||
| 872067-98-6 | Proteolytic enzyme | ||||
| CN1648249 | siRNAs were specifically designed to target the M, N, and E genes of SARS | 1648249/200410016001.1 | 874840-18-3 874840-32-1 | M protein | [169,170] |
| 874840-19-4 874840-47-8 | N protein | ||||
| 874840-20-7 874840-46-7 | E protein | ||||
| CN101113158 | Two anti-SARS-CoV siRNA were designed to disturb RdRp gene function of SARS virus | 101113158/200610022519.5 | - | RdRp | [33,169] |
| CN101085986 | A SARS coronavirus disturbance RNA, which is aimed to disturb RNA for SARS coronavirus and its application, and also to suppress the release of SARS virus by inhibiting the expression of ORF3a gene | 101085986/200610027475.5 | - | ORF3a gene | [33,169] |
| WO2017044507 | siRNA/nanoparticle formulations for treatment MERS-CoV infection, which was designed to target S protein, RdRp and PLpro | WO/2017/044507/PCT/US2016/050590 | - | S protein, RdRp and PLpro | [33,169] |
| CN1548054 | Two siRNAs or it can be RNAi medicine were used in preventing and treating SARS coronavirus. It is anti-SARS coronavirus transcription and replication polymerases. | 1548054/03125172.2 | - | Viral mRNA, viral N protein, RNA transcriptase, virion RNA, complementary and RNA polymerase. | [27] |
| WO2005019410 | siRNA molecules and their analogs were designed to target respiratory infections, including SARS coronavirus | WO/2005/019410/PCT/US2004/012730 | - | nsp-PP1ab, nsp-PP1a and S protein | [27] |
| US20070191294 | Novel double-stranded siRNA analogs comprising LNA monomers, which induces sequence-specific post-transcriptional gene silencing in many organisms by a process known as RNA interference | 20070191294/10550152 | - | mRNA, pre-mRNA, or a variety of structural RNAs (such as tRNA, snRNA, scRNA and rRNA) or even regulatory RNAs (like miRNAs). | [27] |
| mRNA | miRNA Binding SITES | Sequences of miRNA Binding Sites * | Recent Findings | References |
|---|---|---|---|---|
| IL-1ß | miR-376c-3p | 43-AACAUAGAGGAAAUUCCACGU-63 | miR-7 and miR-429 target RPS6KB1 mRNA and inhibit the viral replication.miR-101 and miR-7 target the mTOR mRNA and inhibit the viral replication.miR-21, miR-155 and miR-126 were reported as potential prognostic factor of COVID-19 based on in vivo study miR126-3p and miR-21-5p were reported as potential biomarkers based on in vivo study | [175,178,256,257] |
| miR-155-5p | 4-UUAAUGCUAAUCGUGAUAGGGGUU-27 | |||
| miR-181c-3p | 65-AACCAUCGACCGUUGAGUGGAC-86 | |||
| miR-587 | 16-UUUCCAUAGGUGAUGAGUCAC-36 | |||
| miR-101-3p | 47-UACAGUACUGUGAUAACUGAA-67 | |||
| miR-10b-5p | 27-UACCCUGUAGAACCGAAUUUGUG-49 | |||
| miR-126-3p | 52-UCGUACCGUGAGUAAUAAUGCG-73 | |||
| miR-128-3p | 50-UCACAGUGAACCGGUCUCUUU-70 | |||
| miR-129–2-3p | 57-AAGCCCUUACCCCAAAAAGCAU-78 | |||
| miR-203a-3p | 65-GUGAAAUGUUUAGGACCACUAG-86 | |||
| miR-34a-5p | 22-UGGCAGUGUCUUAGCUGGUUGU-43 | |||
| miR-34c-5p | 13-AGGCAGUGUAGUUAGCUGAUUGC-35 | |||
| miR-375-5p | 5-GCGACGAGCCCCUCGCACAAACC-27 | |||
| miR-375-3p | 40-UUUGUUCGUUCGGCUCGCGUGA-61 | |||
| miR-429 | 51-UAAUACUGUCUGGUAAAACCGU-72 | |||
| miR-449a | 16-UGGCAGUGUAUUGUUAGCUGGU-37 | |||
| miR-7-5p | 24-UGGAAGACUAGUGAUUUUGUUGUU-47 | |||
| miR-21-5p | 8-UAGCUUAUCAGACUGAUGUUGA-29 | |||
| miR-204-5p | 33-UUCCCUUUGUCAUCCUAUGCCU-54 | |||
| IL-6 | miR-155-5p | 4-UUAAUGCUAAUCGUGAUAGGGGUU-27 | miR-98-5p targets and inhibits IL-6 gene expression, in turn influencing several proinflammatory cytokines, including TNF-α, IL-1β, and IL-10. miR-7 and miR-16 target RPS6KB1 mRNA and inhibit the viral replication.miR-100, let-7, miR-7 and miR-99a target the mTOR mRNA and inhibit the viral replication. Upregulation of miR-124-3p causes the degradation of Ddx58, thereby leading to a decrease in viral replication. miR-125a-3p inhibits the cleavage of the S gene miR-138-5p inhibits the cleavage of the ORF1a/b polyprotein gene miR-21-3p expressed in respiratory epithelial cells in the trachea and lung tissues, which targets the binding site of 6 different coronavirus, including SARS-CoV-2 and SARS miR-21 and miR-155 were reported as potential prognostic factor of COVID-19 based on in vivo study | [175,178,206,256] |
| miR-125a-3p | 53-ACAGGUGAGGUUCUUGGGAGCC-74 | |||
| miR-149-5p | 15-UCUGGCUCCGUGUCUUCACUCCC-37 | |||
| miR-192-5p | 24-CUGACCUAUGAAUUGACAGCC-44 | |||
| miR-590-3p | 56-UAAUUUUAUGUAUAAGCUAGU-76 | |||
| miR-100-5p | 13-AACCCGUAGAUCCGAACUUGUG-34 | |||
| miR-671-5p | 29-AGGAAGCCCUGGAGGGGCUGGAG-51 | |||
| miR-20a-5p | 8-UAAAGUGCUUAUAGUGCAGGUAG-30 | |||
| let-7b-5p | 6-UGAGGUAGUAGGUUGUGUGGUU-27 | |||
| miR-16-5p | 14-UAGCAGCACGUAAAUAUUGGCG-35 | |||
| miR-376a-5p | 7-GUAGAUUCUCCUUCUAUGAGUA-28 | |||
| miR-335-5p | 16-UCAAGAGCAAUAACGAAAAAUGU-38 | |||
| miR-98-5p | 22-UGAGGUAGUAAGUUGUAUUGUU-43 | |||
| miR-124-3p | 53-UAAGGCACGCGGUGAAUGCCAA-74 | |||
| miR-1-3p | 53-UGGAAUGUAAAGAAGUAUGUAU-74 | |||
| miR-34a-5p | 22-UGGCAGUGUCUUAGCUGGUUGU-43 | |||
| miR-99a-5p | 13-AACCCGUAGAUCCGAUCUUGUG-34 | |||
| miR-191-5p | 16-CAACGGAAUCCCAAAAGCAGCUG-38 | |||
| miR-128-3p | 50-UCACAGUGAACCGGUCUCUUU-70 | |||
| miR-138-5p | 10-AGCUGGUGUUGUGAAUCAGGCCG-32 | |||
| miR-182-5p | 23-UUUGGCAAUGGUAGAACUCACACU-46 | |||
| miR-195-5p | 15-UAGCAGCACAGAAAUAUUGGC-35 | |||
| miR-203a-3p | 65-GUGAAAUGUUUAGGACCACUAG-86 | |||
| miR-205-5p | 34-UCCUUCAUUCCACCGGAGUCUG-55 | |||
| miR-21-3p | 46-CAACACCAGUCGAUGGGCUGU-66 | |||
| miR-21-5p | 8-UAGCUUAUCAGACUGAUGUUGA-29 | |||
| miR-221-3p | 65-AGCUACAUUGUCUGCUGGGUUUC-87 | |||
| miR-27a-3p | 51-UUCACAGUGGCUAAGUUCCGC-71 | |||
| miR-27a-5p | 10-AGGGCUUAGCUGCUUGUGAGCA-31 | |||
| miR-330-3p | 57-GCAAAGCACACGGCCUGCAGAGA-79 | |||
| miR-34b-5p | 13-UAGGCAGUGUCAUUAGCUGAUUG-35 | |||
| miR-375-5p | 5-GCGACGAGCCCCUCGCACAAACC-27 | |||
| miR-375-3p | 40-UUUGUUCGUUCGGCUCGCGUGA-61 | |||
| miR-429 | 51-UAAUACUGUCUGGUAAAACCGU-72 | |||
| miR-7-5p | 24-UGGAAGACUAGUGAUUUUGUUGUU-47 | |||
| miR-373-3p | 44-GAAGUGCUUCGAUUUUGGGGUGU-66 | |||
| miR-372-3p | 42-AAAGUGCUGCGACAUUUGAGCGU-64 | |||
| miR-302a-3p | 44-UAAGUGCUUCCAUGUUUUGGUGA-66 | |||
| miR-148b-3p | 63-UCAGUGCAUCACAGAACUUUGU-84 | |||
| miR-133a-3p | 53-UUUGGUCCCCUUCAACCAGCUG-74 | |||
| miR-122-5p | 15-UGGAGUGUGACAAUGGUGUUUG-36 | |||
| IL-8 | miR-195-5p | 15-UAGCAGCACAGAAAUAUUGGC-35 | Upregulation of miR-17 and miR-214 have an antiviral effect by binding to S-protein-encoding mRNA, hence cause inhibition of the viral replication. miRNA-145 downregulates ADAM17, which is a target of Jagged1/Notch1 signaling in vascular smooth muscle cells. miR-7, miR-17, miR-16 and miR-107 target RPS6KB1 mRNA and inhibit the viral replication. miR-101, let-7, miR-107, miR-7 and miR-99b target the mTOR mRNA and inhibit the viral replication. Upregulation of miR-124-3p causes the degradation of Ddx58, thereby leading to a decrease in viral replication. miR-99b-5p regulates immune reactions. miR-130a predicts the targets of QFPD, and QFPD, which may exert anti-SARS-CoV-2 activity miR-17-5p have antiviral roles against SARS-CoV-2 by targeting the ORF1ab and the S region miR-23a-3p has prognostic and therapeutic effects based on in vivo study miR-146a-5p was reported as potential biomarker based on in vivo study miR-29a-3p promote IL-8 and other pro-inflammatory cytokine expression, despite being inversely correlated with IL-8 in COVID-19 | [175,178,193,242,257,258,259,260,261] |
| miR-20a-5p | 8-UAAAGUGCUUAUAGUGCAGGUAG-30 | |||
| miR-106a-5p | 13-AAAAGUGCUUACAGUGCAGGUAG-35 | |||
| miR-17-5p | 14-CAAAGUGCUUACAGUGCAGGUAG-36 | |||
| miR-30c-1-3p | 56-CUGGGAGAGGGUUGUUUACUCC-77 | |||
| miR-93-5p | 11-CAAAGUGCUGUUCGUGCAGGUAG-33 | |||
| miR-373-3p | 44-GAAGUGCUUCGAUUUUGGGGUGU-66 | |||
| miR-520c-3p | 54-AAAGUGCUUCCUUUUAGAGGGU-75 | |||
| miR-10a-3p | 63-CAAAUUCGUAUCUAGGGGAAUA-84 | |||
| miR-1225-5p | 1-GUGGGUACGGCCCAGUGGGGGG-22 | |||
| miR-23a-3p | 45-AUCACAUUGCCAGGGAUUUCC-65 | |||
| miR-23b-3p | 58-AUCACAUUGCCAGGGAUUACCAC-80 | |||
| miR-296-3p | 48-GAGGGUUGGGUGGAGGCUCUCC-69 | |||
| miR-302c-5p | 8-UUUAACAUGGGGGUACCUGCUG-29 | |||
| miR-302d-5p | 6-ACUUUAACAUGGAGGCACUUGC-27 | |||
| miR-450a-5p | 18-UUUUGCGAUGUGUUCCUAAUAU-39 | |||
| miR-493-5p | 16-UUGUACAUGGUAGGCUUUCAUU-37 | |||
| miR-499a-3p | 70-AACAUCACAGCAAGUCUGUGCU-91 | |||
| miR-519d-3p | 54-CAAAGUGCCUCCCUUUAGAGUG-75 | |||
| miR-520a-3p | 53-AAAGUGCUUCCCUUUGGACUGU-74 | |||
| miR-526b-3p | 51-GAAAGUGCUUCCUUUUAGAGGC-72 | |||
| miR-5582-3p | 47-UAAAACUUUAAGUGUGCCUAGG-68 | |||
| miR-587 | 16-UUUCCAUAGGUGAUGAGUCAC-36 | |||
| miR-664a-3p | 49-UAUUCAUUUAUCCCCAGCCUACA-71 | |||
| miR-1-3p | 53-UGGAAUGUAAAGAAGUAUGUAU-74 | |||
| miR-429 | 51-UAAUACUGUCUGGUAAAACCGU-72 | |||
| miR-34a-5p | 22-UGGCAGUGUCUUAGCUGGUUGU-43 | |||
| miR-155-5p | 4-UUAAUGCUAAUCGUGAUAGGGGUU-27 | |||
| let-7b-5p | 6-UGAGGUAGUAGGUUGUGUGGUU-27 | |||
| miR-124-3p | 53-UAAGGCACGCGGUGAAUGCCAA-74 | |||
| miR-126-3p | 52-UCGUACCGUGAGUAAUAAUGCG-73 | |||
| miR-16-5p | 14-UAGCAGCACGUAAAUAUUGGCG-35 | |||
| miR-27a-3p | 51-UUCACAGUGGCUAAGUUCCGC-71 | |||
| miR-335-5p | 16-UCAAGAGCAAUAACGAAAAAUGU-38 | |||
| miR-1291 | 14-UGGCCCUGACUGAAGACCAGCAGU-37 | |||
| miR-138-5p | 10-AGCUGGUGUUGUGAAUCAGGCCG-32 | |||
| miR-101-3p | 47-UACAGUACUGUGAUAACUGAA-67 | |||
| miR-107 | 50-AGCAGCAUUGUACAGGGCUAUCA-72 | |||
| miR-129–2-3p | 57-AAGCCCUUACCCCAAAAAGCAU-78 | |||
| miR-130a-3p | 55-CAGUGCAAUGUUAAAAGGGCAU-76 | |||
| miR-146a-5p | 21-UGAGAACUGAAUUCCAUGGGUU-42 | |||
| miR-147a | 47-GUGUGUGGAAAUGCUUCUGC-66 | |||
| miR-194-5p | 15-UGUAACAGCAACUCCAUGUGGA-36 | |||
| miR-203a-3p | 65-GUGAAAUGUUUAGGACCACUAG-86 | |||
| miR-21-3p | 46-CAACACCAGUCGAUGGGCUGU-66 | |||
| miR-21-5p | 8-UAGCUUAUCAGACUGAUGUUGA-29 | |||
| miR-210-3p | 66-CUGUGCGUGUGACAGCGGCUGA-87 | |||
| miR-212-3p | 71-UAACAGUCUCCAGUCACGGCC-91 | |||
| miR-214-3p | 71-ACAGCAGGCACAGACAGGCAGU-92 | |||
| miR-221-3p | 65-AGCUACAUUGUCUGCUGGGUUUC-87 | |||
| miR-29a-5p | 4-ACUGAUUUCUUUUGGUGUUCAG-25 | |||
| miR-29a-3p | 42-UAGCACCAUCUGAAAUCGGUUA-63 | |||
| miR-30d-5p | 6-UGUAAACAUCCCCGACUGGAAG-27 | |||
| miR-376a-5p | 7-GUAGAUUCUCCUUCUAUGAGUA-28 | |||
| miR-671-5p | 29-AGGAAGCCCUGGAGGGGCUGGAG-51 | |||
| miR-7-5p | 24-UGGAAGACUAGUGAUUUUGUUGUU-47 | |||
| miR-941 | 47-CACCCGGCUGUGUGCACAUGUGC-69 | |||
| miR-99b-5p | 7-CACCCGUAGAACCGACCUUGCG-28 | |||
| miR-520f-3p | 55-AAGUGCUUCCUUUUAGAGGGUU-76 | |||
| miR-372-3p | 42-AAAGUGCUGCGACAUUUGAGCGU-64 | |||
| miR-148b-3p | 63-UCAGUGCAUCACAGAACUUUGU-84 | |||
| miR-133a-3p | 53-UUUGGUCCCCUUCAACCAGCUG-74 | |||
| miR-9-5p | 16-UCUUUGGUUAUCUAGCUGUAUGA-38 | |||
| miR-30a-5p | 6-UGUAAACAUCCUCGACUGGAAG-27 |
| Nucleic Acid Therapy | Advantages | Disadvantages | References |
|---|---|---|---|
| mRNA vaccine | Noninfectious, no genome integration risk, reusable platform, simple formulations, rapid and scalable production. It is flexible and can mimic the antigen structure and expression as seen in the course of a natural infection. The anti-vector immunity is avoided. The immunogenicity of the mRNA can be minimized. Alterations can be made to increase the stability of these vaccines. The translation of mRNA occurs in the host cell’s cytosol, averting the risk of any sort of integration into the host genome. Small size, simpler constructs, and lack any extra encoded proteins. The potential risk of infection and insertion induced mutagenesis are minimized by mRNA-based vaccines. Stimulate innate immune response, induction of T and B cell immune response. The self-amplifying mRNA-based vaccines (SAM) vaccine technology is capable of swift and cost-effective vaccines production. These can be used for both therapeutic and prophylactic purposes, as shown by various preclinical and clinical projects. | It needs an efficient delivery and expression into the cytoplasm, due to the plasma membrane hinders the entry of mRNA. Safety issues with reactogenicity have been reported for various RNA based vaccines. Local and systemic inflammatory responses, possible development of autoreactive antibodies, persistence, and bio-distribution of induced immunogenic responses. Toxic effect of delivery system components and non-native nucleotide May need extremely low temperatures for storage and transportation. Mutation in the S protein increases the possibility that the vaccine will not be very effective in the long term. The price of mRNA vaccines is higher (US$30−40 per shot) than conventional or viral vector-based vaccines (US$2−5). | [13,33,112,116,263,264] |
| DNA vaccine | Fast to produce, scalable, noninfectious and reusable platform. DNA vaccine encodes the antigen and an adjuvant, will induces the adaptive immune response. Stimulating effective humoral as well as cell-mediated immune responses. The synthetic DNA is temperature stable and cold-chain free. It can be developed at an accelerated pace. It does not require the handling of the infectious viral particle. Stable at room temperature. DNA vaccines have low production cost when compared to protein vaccines. Enhanced stability for transportation and storage and can be administered to immunocompromised patients. | It may need special delivery devices. Though it elicits both Cytotoxic and humoral immunity, the titer remains low. Insertion of foreign DNA into the host genome may cause abnormalities in the cell. May induce the antibody production against itself. In human and large animals, DNA vaccine causes lower immunogenicity in comparison to inactivated vaccines. Autoimmune responses and DNA integration in the host genome may occur. Require multiple booster doses. | [13,33,112,263,265] |
| ASO-based therapy | Acquisition of the target sequence provides immediate knowledge of putative complementary oligonucleotide therapeutics. They can be used for antigen modification of whole-cell immunogens or as a vaccine adjuvant by enhancing the host immune response. Block the expression of specific target genes via complementary hybridization to mRNA. Have high specificity and a well-known mechanism of action. | Insufficient stability and low cellular delivery have not been sufficiently resolved to achieve effective and safe ASO-based vaccines. ASOs bioavailability and the occurrence of potential off-target effects. The possibility of toxicity by immune overstimulation needs to be deeply studied. | [240,266,267] |
| miRNAs-based therapy | Promising approach against heart failure or cardiovascular diseases, neurological disorders, tumorigenesis, and viral infection. Promising therapeutic avenue for future antiviral applications, where it will facilitate the diagnosis and improve the treatment of COVID-19 infection. Potent epigenetic regulators which stimulate the innate and adaptive immune system. miRNAs-based therapeutics could be used in the nanovaccines that are specific with minimal off-target effects. | Less specific due to miRNAs- mediated therapeutics as miRNAs can hybridize with mRNA having partially complementary sequences Can target an entire immune pathway. Various miRNAs with multi-step maturation need several strategies to enhance miRNA’s function and develop miRNAs-based therapeutics against COVID-19. May cause adverse effects on the host genome. May target several signaling pathways and critical cellular processes. Unstable nature of RNA molecules The requirement for targeted delivery based on its site of action. | [33,173,208,268] |
| siRNAs-based therapy | It does not require translation of mRNA, is programmable, scalable, stable, and able to repress coronaviruses potently. siRNAs are more specific than miRNAs- mediated therapeutics as miRNAs have then the ability to hybridize with mRNA having partially complementary sequences. It has also been shown to be an effective adjuvant for DNA vaccines. Therapeutically effective ASOs are heavily modified, so they do not require a delivery carrier. This limited downstream processing during manufacturing decreases production-associated costs. Allows for addressing the root cause of the infection rather than palliating only the symptoms of the disease both in prophylactic or curative settings Low cost of production to manufacture and easy to design | A siRNA against the Leader sequence was relatively less effective (∼50%, estimated). Limited effectiveness and potential toxicity effects in the early applications. Their efficient delivery to relevant cells in the lung is the next hurdle. | [33,165,167,240,248,264,265,269] |
| CRISPR–Cas based therapy | Strong antiviral and diagnostic technology platform for a wide variety of ssRNA viruses. Can be used as therapeutic tools for SARS-CoV-2 infection. Can target different regions of a virus or different coronavirus strains simultaneously with the crRNA pool, preventing possible viral escapes. Promising strategy to combat pan-coronaviruses that will occur in pandemics. Can successfully target and cleave the RNA sequences of SARS-CoV-2 fragments and IAV in lung epithelial cell cultures. It is a genetic strategy that can target conserved regions, hence prevent the virus to escape inhibition through mutation. RNA-targeting CRISPR effectors could offer a future promising possible antiviral approach. Cas13 could be programmed to target and destroy a wide variety of mammalian viruses. | The presence of pre-existing antibodies to CRISPRs33. The need to translate the packaged CRISPR mRNA and gRNA in virus infected cells will hinder the clinical translation of this approach. CRISPR systems may be more costly and perhaps toxic than all other systems. Further validation in animal models earlier than clinical checks in humans is required. | [153,159,167,239] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abusalah, M.A.H.; Khalifa, M.; Al-Hatamleh, M.A.I.; Jarrar, M.; Mohamud, R.; Chan, Y.Y. Nucleic Acid-Based COVID-19 Therapy Targeting Cytokine Storms: Strategies to Quell the Storm. J. Pers. Med. 2022, 12, 386. https://doi.org/10.3390/jpm12030386
Abusalah MAH, Khalifa M, Al-Hatamleh MAI, Jarrar M, Mohamud R, Chan YY. Nucleic Acid-Based COVID-19 Therapy Targeting Cytokine Storms: Strategies to Quell the Storm. Journal of Personalized Medicine. 2022; 12(3):386. https://doi.org/10.3390/jpm12030386
Chicago/Turabian StyleAbusalah, Mai Abdel Haleem, Moad Khalifa, Mohammad A. I. Al-Hatamleh, Mu’taman Jarrar, Rohimah Mohamud, and Yean Yean Chan. 2022. "Nucleic Acid-Based COVID-19 Therapy Targeting Cytokine Storms: Strategies to Quell the Storm" Journal of Personalized Medicine 12, no. 3: 386. https://doi.org/10.3390/jpm12030386
APA StyleAbusalah, M. A. H., Khalifa, M., Al-Hatamleh, M. A. I., Jarrar, M., Mohamud, R., & Chan, Y. Y. (2022). Nucleic Acid-Based COVID-19 Therapy Targeting Cytokine Storms: Strategies to Quell the Storm. Journal of Personalized Medicine, 12(3), 386. https://doi.org/10.3390/jpm12030386











