Development of a Prototype for a Bilingual Patient-Reported Outcome Measure of the Important Health Aspects of Quality of Life in People Living with HIV: The Preference Based HIV Index (PB-HIV)
Abstract
1. Introduction
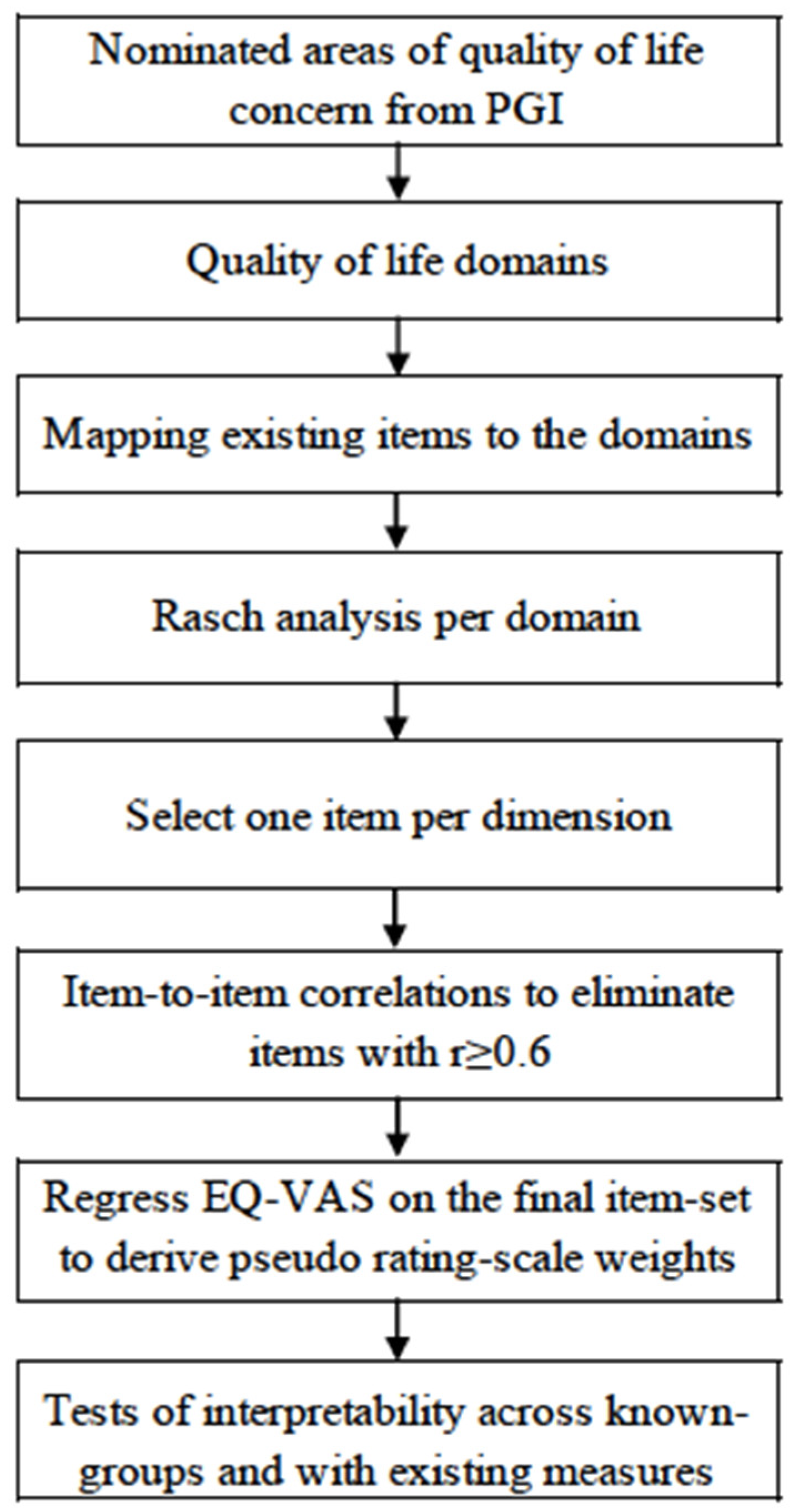
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Items | Levels | Betas (SE) |
|---|---|---|
| Walking | ||
| Vigorous activities, such as running, lifting heavy objects, participating in strenuous sports | Not limited at all (1) | Ref |
| Limited a little (2) | 12.1 (1.3) | |
| Limited a lot (3) | 20.1 (1.4) | |
| Apathy | ||
| Do you have plans and goals for the future? | Not at all (1) | Ref |
| Some (2) | 7.6 (1.8) | |
| A lot (3) | 12.8 (1.8) | |
| Stigma | ||
| To what extent are you bothered by people blaming you for your HIV status? | Not at all (1) | Ref |
| A little (2) | 6.7 (2.9) | |
| A moderate amount (3) | 3.9 (3.1) | |
| Very much (4) | −0.1 (3.6) | |
| An extreme amount (5) | −1.2 (3.5) | |
| Intimacy | ||
| How satisfied are you with your sex life? | Very satisfied (1) | Ref |
| Satisfied (2) | 4.5 (1.7) | |
| Neither satisfied nor dissatisfied (3) | 7.3 (1.6) | |
| Dissatisfied (4) | 9.4 (1.6) | |
| Very dissatisfied (5) | 13.1 (2.3) | |
| Relationships | ||
| About how many times did you talk to someone (friends, relatives, or others) on the telephone in the past week? | Less than once a week (1) | Ref |
| Once a week (2) | 0.1 (2.9) | |
| Almost every day (3) | 8.7 (2.6) | |
| Once a day or more (4) | 10.5 (2.5) | |
| Recreation/Leisure | ||
| To what extent do you have the opportunity for leisure activities? | Completely (1) | Ref |
| Mostly (2) | 7.5 (2.8) | |
| Moderately (3) | 15.3 (2.7) | |
| A little (4) | 18.7 (2.6) | |
| Not at all (5) | 25.1 (2.8) |
References
- Whoqol Group. Development of the World Health Organization WHOQOL-BREF quality of life assessment. Psychol. Med. 1998, 28, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Brazier, J. Health, Health-Related Quality of Life, and Quality of Life: What is the Difference? PharmacoEconomics 2016, 34, 645–649. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, R.M.; Bush, J.W. Health-related quality of life measurement for evaluation research and policy analysis. Health Psychol. 1982, 1, 61. [Google Scholar] [CrossRef]
- Bullinger, M. Indices versus profiles—Advantages and disadvantages. In Quality of Life Assessment: Key Issues in the 1990s; Springer: Berlin/Heidelberg, Germany, 1993; pp. 209–220. [Google Scholar]
- Kannel, W.B.; McGee, D.L. Diabetes and cardiovascular disease: The Framingham study. JAMA 1979, 241, 2035–2038. [Google Scholar] [CrossRef]
- Dawber, T.R. The Framingham Study; Harvard University Press: Cambridge, MA, USA, 2013. [Google Scholar]
- Gordon, T.; B. D'agostino S.R., R. Framingham study. In Encyclopedia of Biostatistics; John Wiley & Sons, Ltd.: New York, NY, USA, 2005; Volume 3. [Google Scholar]
- D’Hoore, W.; Bouckaert, A.; Tilquin, C. Practical considerations on the use of the Charlson comorbidity index with administrative data bases. J. Clin. Epidemiol. 1996, 49, 1429–1433. [Google Scholar] [CrossRef]
- Feeny, D. Preference-based measures: Utility and quality-adjusted life years. Assess. Qual. Life Clin. Trials 2005, 2, 405–431. [Google Scholar]
- Feeny, D.; Furlong, W.; Torrance, G.W.; Goldsmith, C.H.; Zhu, Z.; DePauw, S.; Margaret, D.; Margaret, B. Multiattribute and single-attribute utility functions for the health utilities index mark 3 system. Med. Care 2002, 40, 113–128. [Google Scholar] [CrossRef]
- Balestroni, G.; Bertolotti, G. EuroQol-5D (EQ-5D): An instrument for measuring quality of life. Monaldi Arch. Chest Dis. 2012, 78. [Google Scholar] [CrossRef]
- Kind, P.; Brooks, R.; Rabin, R. EQ-5D concepts and method. A Dev. Hist. 2005, 2005. [Google Scholar]
- Kuspinar, A.; Finch, L.; Pickard, S.; Mayo, N.E. Using existing data to identify candidate items for a health state classification system in multiple sclerosis. Qual. Life Res. 2014, 23, 1445–1457. [Google Scholar] [CrossRef]
- Kuspinar, A.; Mate, K.; Lafontaine, A.-L.; Mayo, N. Evaluating the content validity of generic preference-based measures for use in Parkinson’s disease. Parkinsonism Relat. Disord. 2019, 62, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Poissant, L.; E Mayo, N.; Wood-Dauphinee, S.; E Clarke, A. The development and preliminary validation of a Preference-Based Stroke Index (PBSI). Health Qual. Life Outcomes 2003, 1, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Pickard, A.S.; Wilke, C.; Jung, E.; Patel, S.; Stavem, K.; Lee, T.A. Use of a preference-based measure of health (EQ-5D) in COPD and asthma. Respir. Med. 2008, 102, 519–536. [Google Scholar] [CrossRef] [PubMed]
- Revicki, D.A.; Leidy, N.K.; Brennan-Diemer, F.; Thompson, C.; Togias, A. Development and preliminary validation of the multiattribute Rhinitis Symptom Utility Index. Qual. Life Res. 1998, 7, 693–702. [Google Scholar] [CrossRef]
- Samji, H.; Cescon, A.; Hogg, R.S.; Modur, S.P.; Althoff, K.; Buchacz, K.; Burchell, A.N.; Cohen, M.; Gebo, K.A.; Gill, M.J.; et al. Closing the gap: Increases in life expectancy among treated HIV-positive individuals in the United States and Canada. PLoS ONE 2013, 8, e81355. [Google Scholar] [CrossRef]
- Rodger, A.J.; Cambiano, V.; Bruun, T.; Vernazza, P.; Collins, S.; Van Lunzen, J.; Corbelli, G.M.; Estrada, V.; Geretti, A.M.; Beloukas, A.; et al. Sexual Activity Without Condoms and Risk of HIV Transmission in Serodifferent Couples When the HIV-Positive Partner Is Using Suppressive Antiretroviral Therapy. JAMA 2016, 316, 171–181. [Google Scholar]
- Saag, M.S. HIV Infection—Screening, Diagnosis, and Treatment. N. Engl. J. Med. 2021, 384, 2131–2143. [Google Scholar] [CrossRef]
- Remien, R.H.; Stirratt, M.J.; Nguyen, N.; Robbins, R.N.; Pala, A.N.; Mellins, C.A. Mental health and HIV/AIDS: The need for an integrated response. Aids 2019, 33, 1411–1420. [Google Scholar] [CrossRef]
- Kronfli, N.; Linthwaite, B.; Sheehan, N.; Cox, J.; Hardy, I.; Lebouché, B.; De Pokomandy, A.; Frenette, C.; Roger, M.; Klein, M.B. Delayed linkage to HIV care among asylum seekers in Quebec, Canada. BMC Public Health 2019, 19, 1683. [Google Scholar] [CrossRef]
- Miners, A.; Phillips, A.; Kreif, N.; Rodger, A.; Speakman, A.; Fisher, M.; Anderson, J.; Collins, S.; Hart, G.; Sherr, L.; et al. Health-related quality-of-life of people with HIV in the era of combination antiretroviral treatment: A cross-sectional comparison with the general population. Lancet HIV 2014, 1, e32–e40. [Google Scholar] [CrossRef]
- U. S. Department of Health and Human Services FDA Center for Drug Evaluation and Research; U.S. Department of Health and Human Services FDA Center for Biologics Evaluation and Research; U.S. Department of Health and Human Services FDA Center for Devices and Radiological Health. Guidance for industry: Patient-reported outcome measures: Use in medical product development to support labeling claims: Draft guidance. Health Qual. Life Outcomes 2006, 4, 1–20. [Google Scholar]
- FDA, Guidance for Industry:Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims. Health Qual. Life Outcomes 2006. [CrossRef]
- Brazier, J.E.; Mulhern, B.J.; Bjorner, J.B.; Gandek, B.; Rowen, D.; Alonso, J.; Vilagut, G.; Ware, J.E. Developing a New Version of the SF-6D Health State Classification System From the SF-36v2: SF-6Dv2. Med. Care 2020, 58, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Mayo, N.E.; Positive Brain Health Now Investigators; Brouillette, M.-J.; Fellows, L.K. Understanding and optimizing brain health in HIV now: Protocol for a longitudinal cohort study with multiple randomized controlled trials. BMC Neurol. 2016, 16, 8. [Google Scholar] [CrossRef] [PubMed]
- Mayo, N.E.; Investigators from the Positive Brain Health Now Study; Brouillette, M.-J.; Scott, S.C.; Harris, M.; Smaill, F.; Smith, G.; Thomas, R.; Fellows, L.K. Relationships between cognition, function, and quality of life among HIV+ Canadian men. Qual. Life Res. 2020, 29, 37–55. [Google Scholar] [CrossRef]
- Mayo, N.E.; Brouillette, M.-J.; Nadeau, L.; Dendukuri, N.; Harris, M.; Smaill, F.; Smith, G.; Thomas, R.; Fellows, L.K. A longitudinal view of successful aging with HIV: Role of resilience and environmental factors. Qual. Life Res. 2021, 31, 1135–1145. [Google Scholar] [CrossRef]
- Lam, A.; Mayo, N.E.; Scott, S.; Brouillette, M.-J.; Fellows, L.K. HIV-Related Stigma Affects Cognition in Older Men Living With HIV. J. Acquir. Immune Defic. Syndr. 2019, 80, 198–204. [Google Scholar] [CrossRef]
- Harris, M.; Brouillette, M.-J.; Scott, S.C.; Smaill, F.; Smith, G.; Thomas, R.; Fellows, L.K.; Mayo, N.E. Impact of Loneliness on Brain Health and Quality of Life Among Adults Living With HIV in Canada. J. Acquir. Immune Defic. Syndr. 2020, 84, 336–344. [Google Scholar] [CrossRef]
- Mayo, N.E.; Brouillette, M.J.; Fellows, L.K. Estimates of Prevalence of Cognitive Impairment From Research Studies Can Be Affected by Selection Bias. J. Acquir. Immune Defic. Syndr. 2018, 78, e7–e8. [Google Scholar] [CrossRef]
- Mayo, N.E.; Aburub, A.; Brouillette, M.-J.; Kuspinar, A.; Moriello, C.; Rodriguez, A.M.; Scott, S. In support of an individualized approach to assessing quality of life: Comparison between Patient Generated Index and standardized measures across four health conditions. Qual. Life Res. 2017, 26, 601–609. [Google Scholar] [CrossRef]
- Askari, S.; Fellows, L.K.; Brouillette, M.-J.; Mayo, N.E. Development and validation of a voice-of-the-patient measure of cognitive concerns experienced by people living with HIV. Qual. Life Res. 2021, 30, 921–930. [Google Scholar] [CrossRef] [PubMed]
- Askari, S.; Fellows, L.; Brouillette, M.-J.; Moriello, C.; Duracinsky, M.; Mayo, N.E. Development of an item pool reflecting cognitive concerns expressed by people with HIV. Am. J. Occup. Ther. 2018, 72, 7202205070p1–7202205070p9. [Google Scholar] [CrossRef] [PubMed]
- Ruta, D.A.; Garratt, A.M.; Leng, M.; Russell, I.T.; MacDonald, L.M. A new approach to the measurement of quality of life: The Patient-Generated Index. Med. Care 1994, 32, 1109–1126. [Google Scholar] [PubMed]
- Üstün, T.; Chatterji, S.; Bickenbach, J.; Kostanjsek, N.; Schneider, M. The International Classification of Functioning, Disability and Health: A new tool for understanding disability and health. Disabil. Rehabil. 2003, 25, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Hays, R.D.; Morales, L.S. The RAND-36 measure of health-related quality of life. Ann. Med. 2001, 33, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Starkstein, S.E.; Mayberg, H.S.; Preziosi, T.; Andrezejewski, P.; Leiguarda, R.; Robinson, R.G. Reliability, validity, and clinical correlates of apathy in Parkinson’s disease. J. Neuropsychiatry Clin. Neurosci. 1992, 4, 134–139. [Google Scholar] [PubMed]
- Fillenbaum, G.G.; Smyer, M.A. The development, validity, and reliability of the OARS multidimensional functional assessment questionnaire. J. Gerontol. 1981, 36, 428–434. [Google Scholar] [CrossRef]
- Snaith, R.P. The hospital anxiety and depression scale. Health Qual. Life Outcomes 2003, 1, 1–4. [Google Scholar] [CrossRef]
- Bech, P.; Gudex, C.; Johansen, K.S. The WHO (Ten) well-being index: Validation in diabetes. Psychother. Psychosom. 1996, 65, 183–190. [Google Scholar] [CrossRef]
- Sullivan, M.J.; Edgley, K.; Dehoux, E. A survey of multiple sclerosis: I. Perceived cognitive problems and compensatory strategy use. Can. J. Rehabil. 1990, 4, 99–105. [Google Scholar]
- Buysse, D.J.; Reynolds, C.F., III; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef] [PubMed]
- Schulz, P.; Schlotz, W. The Trier Inventory for the Assessment of Chronic Stress (TICS): Scale construction, statistical testing, and validation of the scale work overload. Diagnostica 1999, 45, 8–19. [Google Scholar] [CrossRef]
- Mweemba, P.; Makukula, M.K.; Mukwato, P.K.; Makoleka, M.M. Quality of life and adherence to antiretroviral drugs. Med. J. Zamb. 2010, 37, 31–39. [Google Scholar]
- Katende-Kyenda, L.; Mabindla, B. Quality of life and adherence to HAART in HIV-Infected patients in a primary healthcare setting in South Africa. Int. J. Health Clin. Res. 2020, 3, 25–36. [Google Scholar]
- Balaban, D.J.; Sagi, P.C.; Goldfarb, N.I.; Nettler, S. Weights for scoring the quality of well-being instrument among rheumatoid arthritics: A comparison to general population weights. Med. Care 1986, 24, 973–980. [Google Scholar]
- Hadorn, D.C.; Uebersax, J. Large-scale health outcomes evaluation: How should quality of life be measured? Part I—Calibration of a brief questionnaire and a search for preference subgroups. J. Clin. Epidemiol. 1995, 48, 607–618. [Google Scholar] [CrossRef]
- De Vet, H.C.; Terwee, C.B.; Mokkink, L.B.; Knol, D.L. Measurement in Medicine: A Practical Guide; Cambridge University Press: Cambridge, MA, USA, 2011. [Google Scholar]
- Kuspinar, A.; Bouchard, V.; Moriello, C.; Mayo, N.E. Development of a bilingual MS-specific health classification system: The preference-based multiple sclerosis index. Int. J. MS Care 2016, 18, 63–70. [Google Scholar]
- Jenkins, M.; Almeida, Q.; Spaulding, S.; van Oostveen, R.; Holmes, J.; Johnson, A.; Perry, S. Plantar cutaneous sensory stimulation improves single-limb support time, and EMG activation patterns among individuals with Parkinson’s disease. Park. Relat. Disord. 2009, 15, 697–702. [Google Scholar] [CrossRef]
- Devlin, N.J.; Brooks, R. EQ-5D and the EuroQol group: Past, present and future. Appl. Health Econ. Health Policy 2017, 15, 127–137. [Google Scholar] [CrossRef]
- Shaw, J.W.; Johnson, J.A.; Coons, S.J. US valuation of the EQ-5D health states: Development and testing of the D1 valuation model. Med. Care 2005, 43, 203–220. [Google Scholar] [CrossRef]
- Janssen, M.F.; Pickard, A.S.; Shaw, J.W. General population normative data for the EQ-5D-3L in the five largest European economies. Eur. J. Health Econ. 2021, 22, 1467–1475. [Google Scholar] [CrossRef] [PubMed]
- Poder, T.G.; Carrier, N.; Kouakou, C.R.C. Quebec Health-Related Quality-of-Life Population Norms Using the EQ-5D-5L: Decomposition by Sociodemographic Data and Health Problems. Value Health 2020, 23, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Duracinsky, M.; Duracinsky, M.; Herrmann, S.; Berzins, B.; Armstrong, A.R.; Kohli, R.; Le Coeur, S.; Assane, D.; Isabelle, F.; Mauro, S.; et al. The development of PROQOL-HIV: An international instrument to assess the health-related quality of life of persons living with HIV/AIDS. J. Acquir. Immune Defic. Syndr. 2012, 59, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Duran, S.; Savès, M.; Spire, B.; Cailleton, V.; Sobel, A.; Carrieri, P.; Salmon, D.; Moatti, J.-P.; Leport, C. Failure to maintain long-term adherence to highly active antiretroviral therapy: The role of lipodystrophy. Aids 2001, 15, 2441–2444. [Google Scholar] [CrossRef][Green Version]
- Torrance, G.W. Measurement of health state utilities for economic appraisal. J. Health Econ. 1986, 5, 1–30. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Feeny, D.H.; Patrick, D.L. Measuring health-related quality of life. Ann. Intern. Med. 1993, 118, 622–629. [Google Scholar] [CrossRef]
- Torrance, G.W. Utility approach to measuring health-related quality of life. J. Chronic Dis. 1987, 40, 593–603. [Google Scholar] [CrossRef]
- Kind, P.; Macran, S. Eliciting social preference weights for Functional Assessment of Cancer Therapy-Lung health states. Pharmacoeconomics 2005, 23, 1143–1153. [Google Scholar] [CrossRef]
- Brazier, J.; Akehurst, R.; Brennan, A.; Dolan, P.; Claxton, K.; McCabe, C.; Sculpher, M.; Tsuchyia, A. Should patients have a greater role in valuing health states? Appl. Health Econ. Health Policy 2005, 4, 201–208. [Google Scholar]
- Wilson, I.B.; Cleary, P.D. Linking clinical variables with health-related quality of life. A conceptual model of patient outcomes. JAMA 1995, 273, 59–65. [Google Scholar] [CrossRef]
- Feng, Y.; Parkin, D.; Devlin, N.J. Assessing the performance of the EQ-VAS in the NHS PROMs programme. Qual. Life Res. 2014, 23, 977–989. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Mean (SD) or N [%] | |
|---|---|---|
| Men | Women | |
| Participants | 728 [84] | 136 [16] |
| Age (years) | 53.4 (8.3) | 50.7 (7.4) |
| Education Primary school High school College/vocational diploma Bachelor MSc, PhD, or professional degree | 28 [4] 179 [25] 238 [34] 180 [26] 76 [11] | 11 [9] 47 [37] 46 [36] 15 [12] 7 [6] |
| Years since HIV | 17.1 (8.1) | 15.5 (6.7) |
| Time of diagnosis (1996) Pre cART Post cART | 355 [49] 373 [51] | 54 [40] 82 [60] |
| Living situation Alone Spouse or partner Family member Friends Other Missing | 345 [47] 231 [33] 32 [4] 68 [9] 29 [4] 23 [3] | 51 [38] 28 [21] 35 [26] 4 [3] 9 [6] 9 [6] |
| Days did you spend in bed due to an illness | 4.3 (12) | 6.8 (29) |
| Nadir CD4 < 200 cells/µL/% < 200 | 212.1 (165.8)/52 | 241.1(184.5)/48 |
| Item (Scale) | Betas (SE) | Weights |
|---|---|---|
| Pain/discomfort (EQ-5D-3L) | ||
| I have no pain or discomfort | Referent | 0 |
| I have moderate pain or discomfort | 9.4 (2.1) | 9 |
| I have extreme pain or discomfort | 13.9 (2.2) | 14 |
| Fatigue Did you have a lot of energy? (RAND-36) | ||
| A little of the time/None of the time | Referent | 0 |
| Some of the time/A good bit of the time | 4.2 (1.3) | 4 |
| Most of the time/All of the time | 8.6 (1.5) | 9 |
| Memory/concentration Miss appointments and meetings you had scheduled (PDQ-20) a | ||
| Never | Referent | 0 |
| Rarely/Sometimes | 4.8 (2.4) | 5 |
| Often/Almost always | 7.3 (2.5) | 7 |
| Physical appearance/Body image Are you able to accept your bodily appearance? (WHOQOL BREF) | ||
| Completely | Referent | 0 |
| Mostly/Moderately | 3.9 (1.4) | 4 |
| A little/Not at all | 5.9 (1.7) | 6 |
| Sleep Do you feel rested when you wake up? (Sleep questionnaire) | ||
| Always | Referent | 0 |
| Often | 2.3 (1.1) | 2 |
| Never | 3.1 (1.8) | 3 |
| Anxiety/Depression (EQ-5D-3L) | ||
| I am not anxious or depressed | Referent | 0 |
| I am moderately anxious or depressed | 10.9 (2.2) | 11 |
| I am extremely anxious or depressed | 14.6 (2.2) | 15 |
| Motivation Do you have plans and goals for the future (Motivation questionnaire) | ||
| A lot | Referent | 0 |
| Some | 2.3 (1.5) | 2 |
| Not at all | 3.2 (1.6) | 3 |
| Mean (SD); Range (Higher Is Better) | Pearson’s Correlation (95% CI) | |
|---|---|---|
| PB-HIV Index | 64.4 (21.5); 0, 100 | --- |
| B-CAM | 56.4 (14.4); 11.8, 97.1 | 0.14 (0.07, 0.21) |
| EQ-5D-3L Utility | 0.8 (0.2); 0.2, 1 | 0.75 (0.72, 0.78) |
| SF-6D Index | 0.7 (0.1); 0.3, 1 | 0.72 (0.69, 0.75) |
| Physical Component Summary score—Oblique | 45.2 (10.3); 14.9, 61.5 | 0.72 (0.69, 0.75) |
| Physical Component Summary score—Orthogonal | 46.7 (9.8); 14.1, 69.5 | 0.55 (0.51, 0.59) |
| Mental Component Summary score—Oblique | 43.3 (12.4); 9.5, 67.2 | 0.74 (0.71, 0.77) |
| Mental Component Summary score—Orthogonal | 43.9 (12.4); 11.8, 72.4 | 0.65 (0.61, 0.69) |
| WHO-QOL BREF | ||
| Physical | 68.5 (19.8); 0, 100 | 0.74 (0.71, 0.78) |
| Psychological | 63.4 (18.5); 0, 100 | 0.78 (0.75, 0.81) |
| Level of Independence | 69.4 (20.5); 6.3, 100 | 0.72 (0.68, 0.75) |
| Social Relationships | 62.4 (20.3); 0, 100 | 0.51 (0.45, 0.55) |
| Environment | 70.4 (16.6); 21.9, 100 | 0.61 (0.56, 0.65) |
| Spirituality/Religion/Personal Beliefs | 70.5 (19.2); 7.3, 100 | 0.53 (0.47, 0.57) |
| Pain | Fatigue | Memory | Self-Image | Sleep | Motivation | Depression | |
|---|---|---|---|---|---|---|---|
| Pain | 1 | ||||||
| Fatigue | 0.41 | 1 | |||||
| Memory | 0.33 | 0.34 | 1 | ||||
| Self-image | 0.32 | 0.46 | 0.32 | 1 | |||
| Sleep | 0.38 | 0.55 | 0.28 | 0.31 | 1 | ||
| Motivation | 0.19 | 0.39 | 0.16 | 0.26 | 0.19 | 1 | |
| Depression | 0.38 | 0.48 | 0.29 | 0.35 | 0.37 | 0.29 | 1 |
| Forget ART Medication | Odds Ratio | 95% Confidence Interval |
|---|---|---|
| EQ-VAS | 1.08 | 0.99, 1.18 |
| PB-HIV Index | 1.13 | 1.05, 1.21 |
| N | PB-HIV Index | SF Index | EQ Utility | |
|---|---|---|---|---|
| Sex | ||||
| Men | 728 | 65.2 (20.7) | 69.4 (12.4) | 81.5 (16.4) |
| Women | 136 | 60.5 (24.5) | 69.2 (12.5) | 80.8 (17.4) |
| Age (years) | ||||
| <45 | 136 | 65.3 (22.1) | 69.6 (12.4) | 82.1 (16.2) |
| 45 to 55 | 405 | 62.6 (21.7) | 68.5 (12.1) | 80.4 (17.2) |
| 55 to 65 | 252 | 64.2 (21.3) | 69.5 (12.7) | 81.3 (16.1) |
| >65 | 72 | 73.3 (17.5) | 73.7 (12.1) | 85.9 (13.7) |
| Time of diagnosis | ||||
| Before 1996 | 410 | 62.6 (21.9) | 68.3 (12.6) | 80.2 (16.9) |
| After 1996 | 455 | 66.1 (21.1) | 70.4 (12.1) | 82.5 (16.1) |
| CD4 | ||||
| Nadir <200 | 457 | 63.8 (22.1) | 68.9 (12.9) | 80.8 (17.3) |
| ≥200 | 408 | 65.1 (20.9) | 69.9 (11.7) | 82.1 (15.6) |
| Number of Comorbidities | ||||
| 0 | 404 | 66.2 (22.4) | 70.9 (12.3) | 82.9 (15.8) |
| 1 | 213 | 63.6 (23.1) | 69.4 (12.6) | 81.5 (17.2) |
| 2 | 133 | 62.6 (19.1) | 67.9 (12.5) | 79.4 (16.6) |
| 3 | 63 | 61.1 (18.8) | 66.3 (12.1) | 77.5 (19.1) |
| 4 | 52 | 62.8 (15.8) | 66.3 (11.5) | 79.3 (14.9) |
| Variables | Patient Experts (n) | Healthcare Professionals (n) |
|---|---|---|
| Sex (women/men) | 1/4 | 4/4 |
| Age range | 20–50 | 20–45 |
| Ethnicity | Asian (1), African (1), European (2), Middle Eastern (1) | Asian (1), European (1), North American (4), Middle Eastern (1) |
| Time since HIV diagnosis | >5 years (3), <2 years (2) | --- |
| Languages | Bilingual (English and French n = 3) English only (n = 2) | Bilingual (English and French n = 7) English only (n = 1) |
| Education Bachelor Master PhD or Professional | 1 2 1 | 2 2 4 |
| English | French |
|---|---|
| Select the option that best represents your health status in the last month | Sélectionnez l’option qui représente le mieux votre état de santé au cours du dernier mois |
| Pain/discomfort | Douleur/inconfort |
| I rarely have pain or discomfort | J’ai rarement de la douleur ou de l’inconfort |
| I have pain or discomfort some days | J’ai de la douleur ou de l’inconfort certains jours |
| I have pain or discomfort most days | J’ai de la douleur ou de l’inconfort la plupart des jours |
| Fatigue | Fatigué |
| I am rarely tired | Je suis rarement fatigué(e) |
| I am tired some of the days | Je suis fatigué(e) certains jours |
| I am tired most of the days | Je suis fatigué(e) la plupart des jours |
| Memory/concentration | Mémoire/concentration |
| I have no memory or concentration difficulties | Je n’ai pas de troubles de mémoire ou de concentration |
| My memory or concentration difficulties sometimes affects my daily life | Mes troubles de mémoire ou de concentration interfèrent parfois avec ma vie quotidienne |
| My memory or concentration difficulties frequently affects my daily life | Mes troubles de mémoire ou de concentration interfèrent fréquemment avec ma vie quotidienne |
| Physical appearance/Body image | Apparence Physique/l’image corporelle |
| I am satisfied with the way I look | Je suis satisfait(e) de mon apparence physique |
| I am somewhat satisfied with the way I look | Je suis plus ou moins satisfait(e) de mon apparence physique |
| I am not satisfied with the way I look | Je ne suis pas satisfait(e) de mon apparence physique |
| Sleep | Sommeil |
| I feel well rested when I wake up on most days | Je me sens reposé(e) quand je me reveille la plupart des jours |
| I feel well rested when I wake up on some days | Je me sens reposé(e) quand je me reveille certains jours |
| I rarely feel well rested when I wake up | Je me sens rarement reposé(e) quand je me réveille |
| Depression | Dépression |
| I rarely feel depressed | Je me sens rarement déprimé |
| I feel depressed some of the days | Je me sens déprimé certains jours |
| I feel depressed most of the days | Je me sens déprimé la plupart des jours |
| Motivation | Motivation |
| I often plan or set goals for my future | Je planifie ou me fixe souvent des objectifs pour mon avenir |
| I sometimes plan or set goals for my future | Je planifie ou me fixe parfois des objectifs pour mon avenir |
| I rarely plan or set goals for my future | Je planifie ou me fixe rarement des objectifs pour mon avenir |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mate, K.K.V.; Lebouché, B.; Brouillette, M.-J.; Fellows, L.K.; Mayo, N.E. Development of a Prototype for a Bilingual Patient-Reported Outcome Measure of the Important Health Aspects of Quality of Life in People Living with HIV: The Preference Based HIV Index (PB-HIV). J. Pers. Med. 2022, 12, 2080. https://doi.org/10.3390/jpm12122080
Mate KKV, Lebouché B, Brouillette M-J, Fellows LK, Mayo NE. Development of a Prototype for a Bilingual Patient-Reported Outcome Measure of the Important Health Aspects of Quality of Life in People Living with HIV: The Preference Based HIV Index (PB-HIV). Journal of Personalized Medicine. 2022; 12(12):2080. https://doi.org/10.3390/jpm12122080
Chicago/Turabian StyleMate, Kedar K. V., Bertrand Lebouché, Marie-Josée Brouillette, Lesley K. Fellows, and Nancy E. Mayo. 2022. "Development of a Prototype for a Bilingual Patient-Reported Outcome Measure of the Important Health Aspects of Quality of Life in People Living with HIV: The Preference Based HIV Index (PB-HIV)" Journal of Personalized Medicine 12, no. 12: 2080. https://doi.org/10.3390/jpm12122080
APA StyleMate, K. K. V., Lebouché, B., Brouillette, M.-J., Fellows, L. K., & Mayo, N. E. (2022). Development of a Prototype for a Bilingual Patient-Reported Outcome Measure of the Important Health Aspects of Quality of Life in People Living with HIV: The Preference Based HIV Index (PB-HIV). Journal of Personalized Medicine, 12(12), 2080. https://doi.org/10.3390/jpm12122080







