Vitamin D Levels in Asymptomatic Children and Adolescents with Atopy during the COVID-19 Era
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
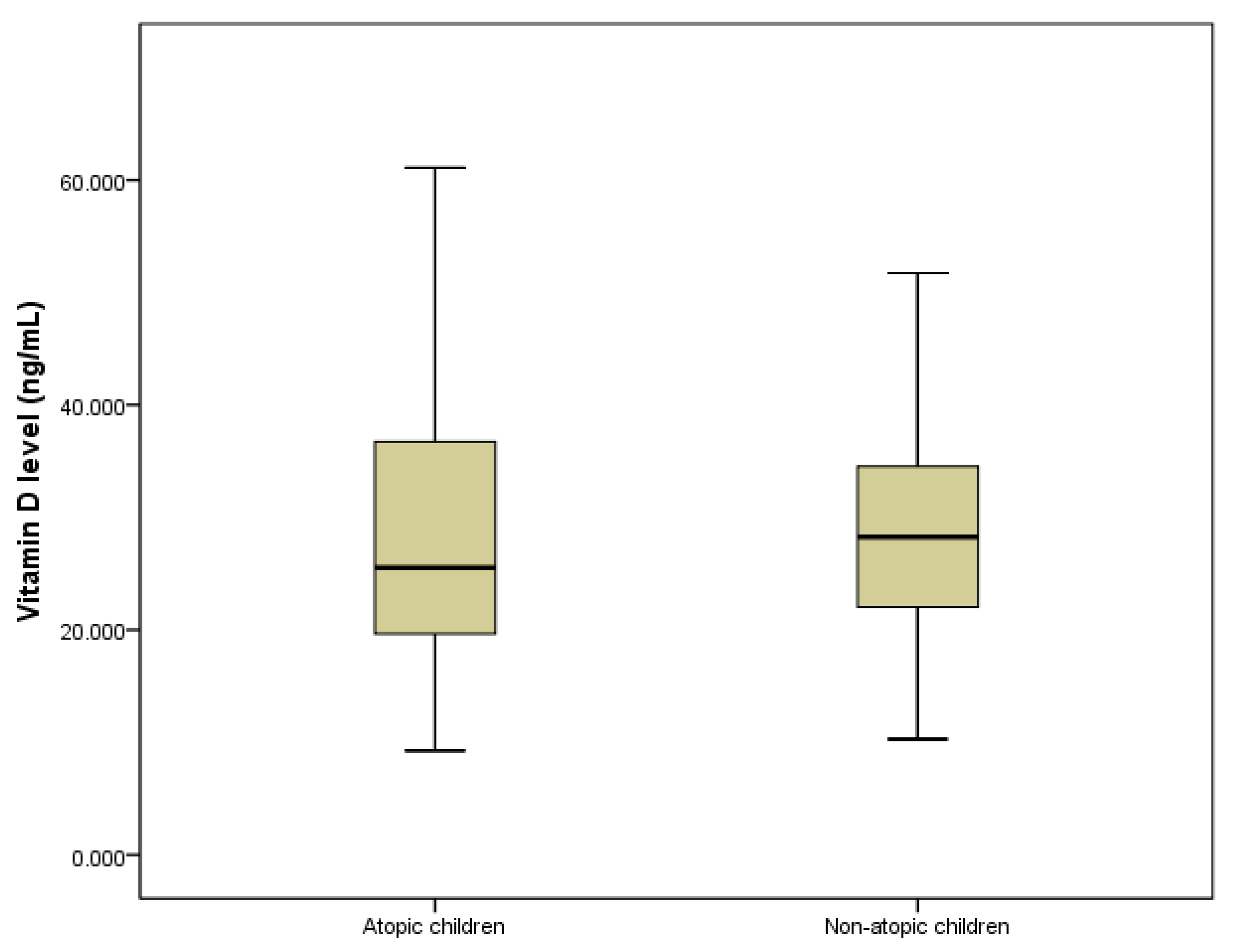
3.1. The Linkage between Vitamin D and Atopy
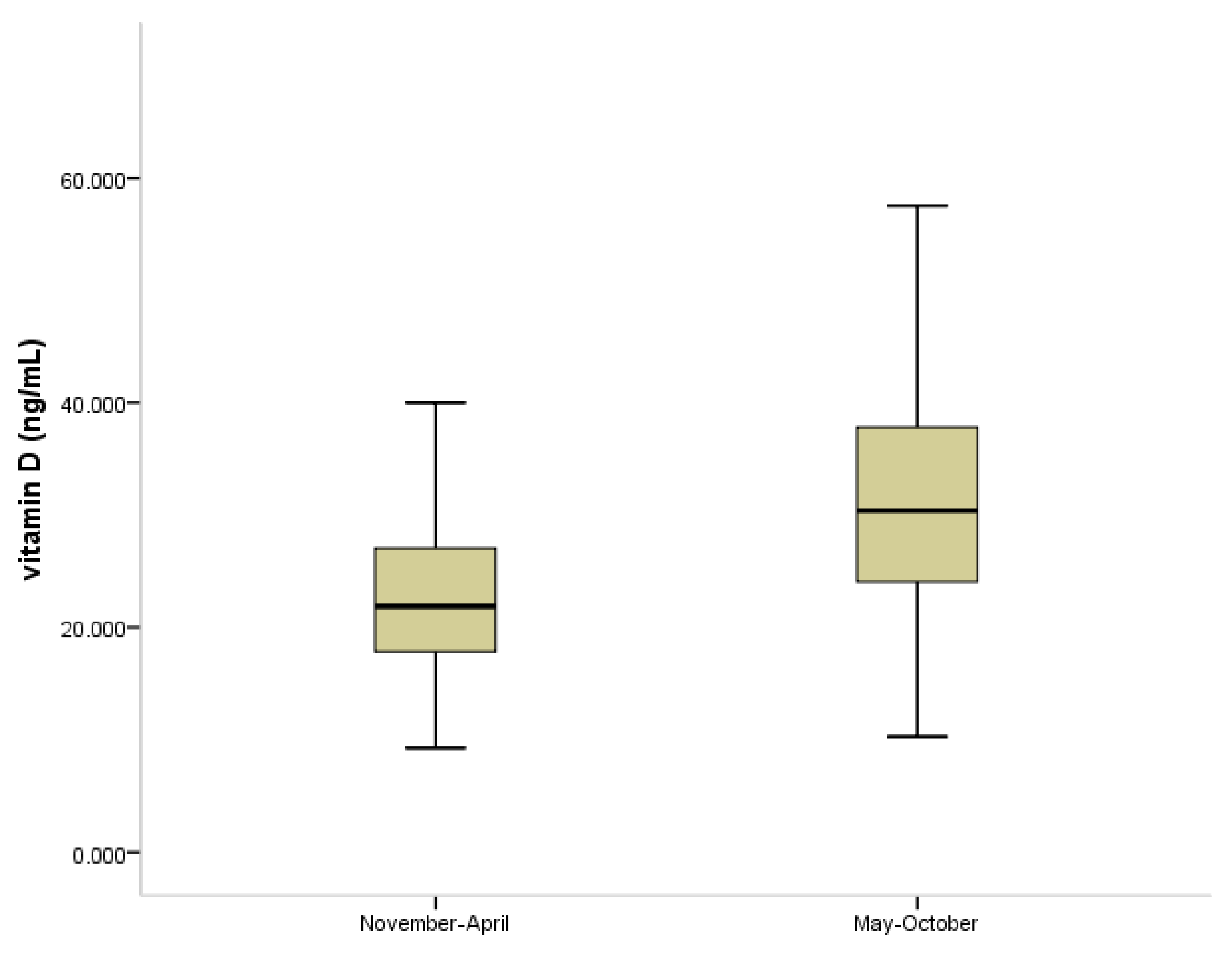
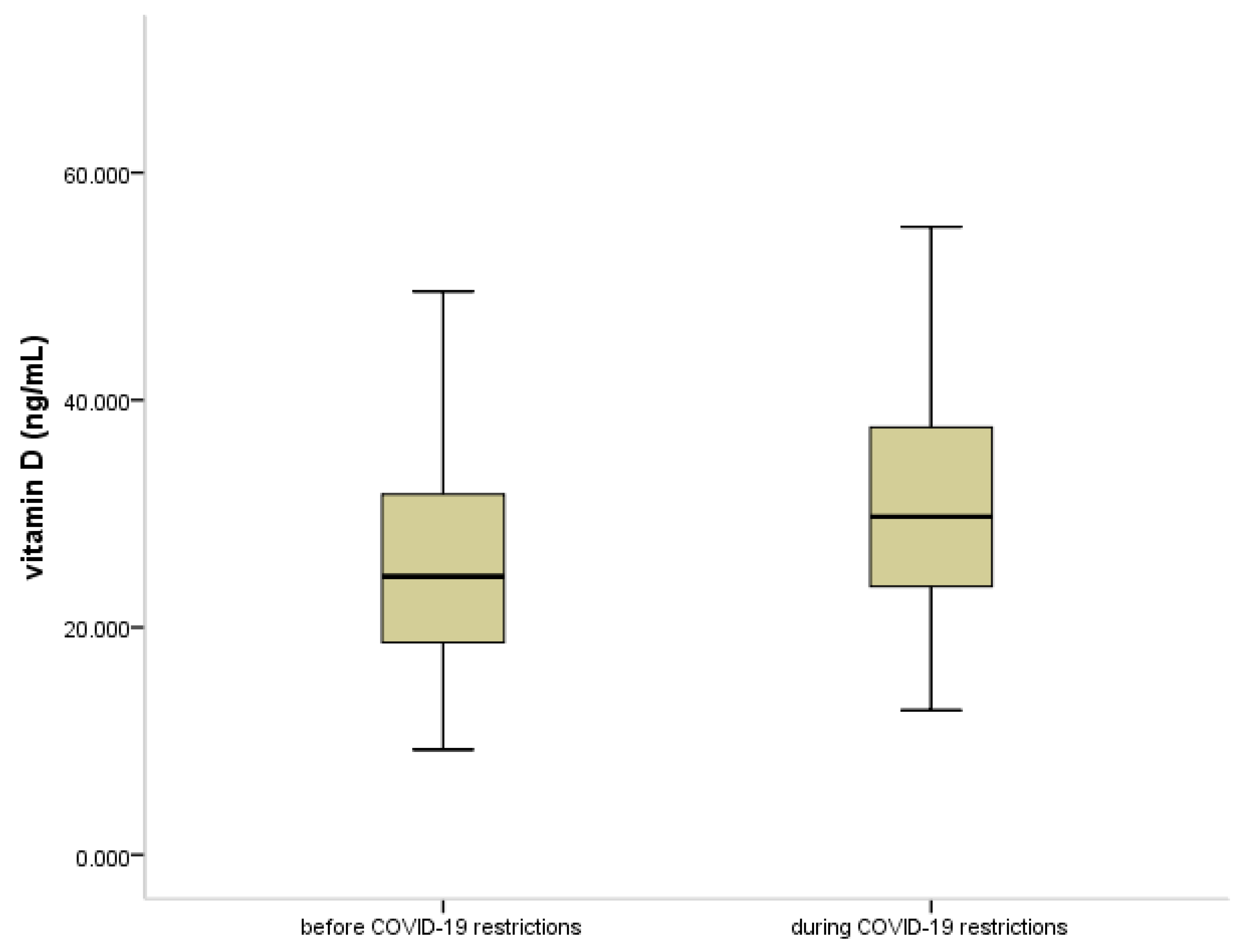
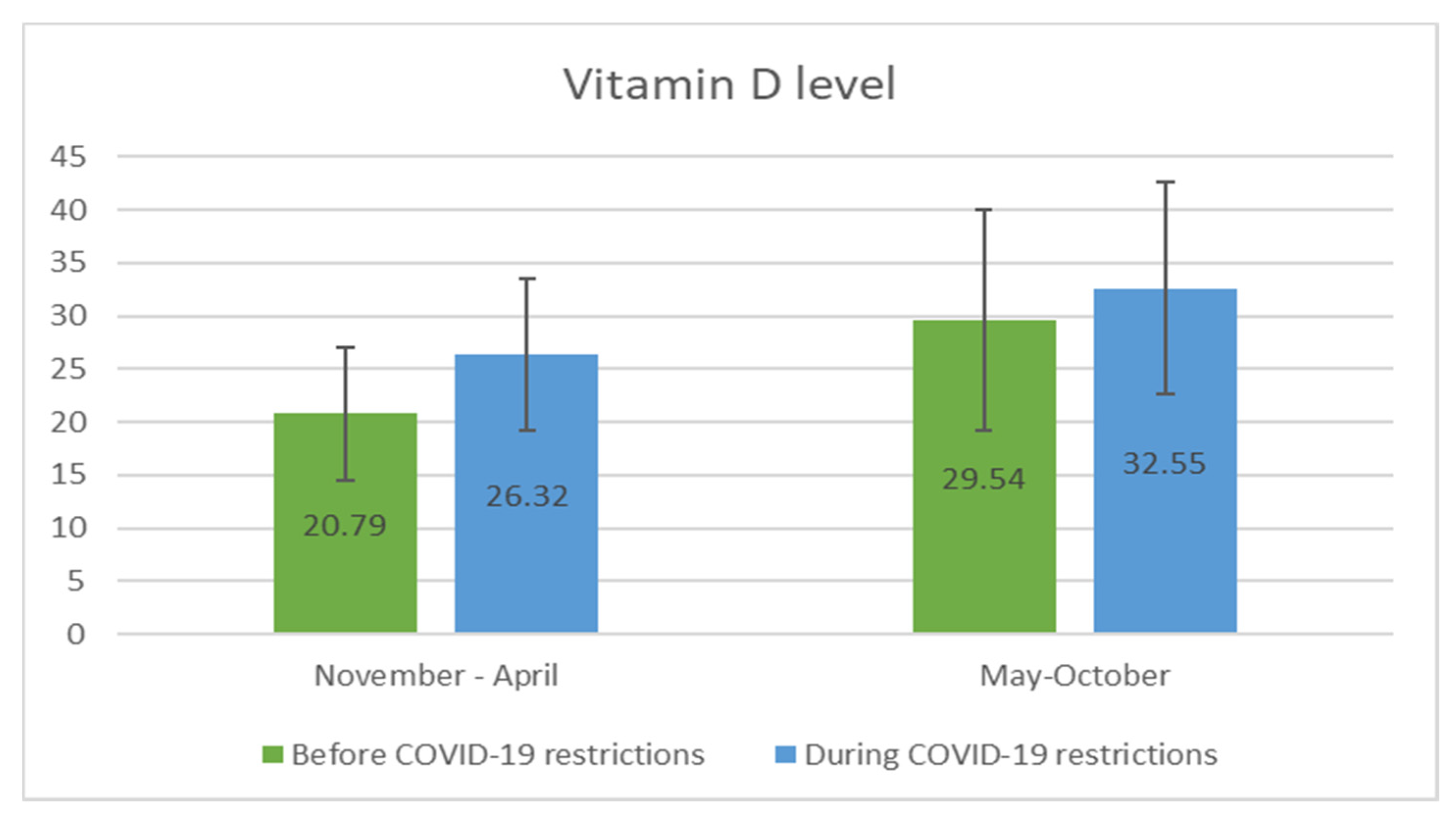
3.2. Vitamin D Status in COVID-19 Pandemia
4. Discussion
Strengths and Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Peebles, R.S.; Church, M.K.; Durham, S.R. Principles of allergy diagnosis. In Allergy; Holgate, S.T., Church, M.K., Broide, D.H., Martinez, F.D., Eds.; W.B. Saunders: Edinburgh, UK, 2012; pp. 129–146. [Google Scholar]
- Justiz Vaillant, A.A.; Modi, P.; Jan, A. “Atopy”. In Statpearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2021. [Google Scholar]
- Wüthrich, B. What is atopy? Condition, disease or a syndrome? Curr. Probl. Dermatol. 1999, 28, 1–8. [Google Scholar] [PubMed]
- Carosso, A.; Bugiani, M.; Migliore, E.; Antò, J.M.; Demarco, R. Reference Values of Total Serum IgE and Their Significance in the Diagnosis of Allergy in Young European Adults. Int. Arch. Allergy Immunol. 2006, 142, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Zihlif, M.; Imraish, A.; Al-Rawashdeh, B.; Qteish, A.; Husami, R.; Husami, R.; Tahboub, F.; Jarrar, Y.; Lee, S.-J. The Association of IgE Levels with ADAM33 Genetic Polymorphisms among Asthmatic Patients. J. Pers. Med. 2021, 11, 329. [Google Scholar] [CrossRef]
- Cardinale, F.; De Benedictis, F.M.; Muggeo, V.; Giordano, P.; Loffredo, M.S.; Iacoviello, G.; Armenio, L. Exhaled nitric oxide, total serum IgE and allergic sensitization in childhood asthma and allergic rhinitis. Pediatr. Allergy Immunol. 2005, 16, 236–242. [Google Scholar] [CrossRef]
- Thomsen, S.F. Epidemiology and natural history of atopic diseases. Eur. Clin. Respir. J. 2015, 2. [Google Scholar] [CrossRef]
- Djukanovic, R.; Lai, C.K.; Wilson, J.W.; Britten, K.M.; Wilson, S.J.; Roche, W.R.; Howarth, P.H.; Holgate, S.T. Bronchial mucosal manifestations of atopy: A comparison of markers of inflammation between atopic asthmatics, atopic nonasthmatics and healthy controls. Eur. Respir. J. 1992, 5, 538–544. [Google Scholar] [PubMed]
- Moody, A.; Fergusson, W.; Wells, A.; Bartley, J.; Kolbe, J. Increased nitric oxide production in the respiratory tract in asymptomatic Pacific Islanders: An association with skin prick reactivity to house dust mite. J. Allergy Clin. Immunol. 2000, 105, 895–899. [Google Scholar] [CrossRef]
- Antonucci, R.; Locci, C.; Clemente, M.G.; Chicconi, E.; Antonucci, L. Vitamin D deficiency in childhood: Old lessons and current challenges. J. Pediatr. Endocrinol. Metab. 2018, 31, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Bountouvi, E.; Douros, K.; Papadopoulou, A. Can Getting Enough Vitamin D during Pregnancy Reduce the Risk of Getting Asthma in Childhood? Front. Pediatr. 2017, 5, 87. [Google Scholar] [CrossRef]
- Baïz, N.; Dargent-Molina, P.; Wark, J.D.; Souberbielle, J.-C.; Annesi-Maesano, I. Cord serum 25-hydroxyvitamin D and risk of early childhood transient wheezing and atopic dermatitis. J. Allergy Clin. Immunol. 2014, 133, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.-Y.; Yao, T.-C.; Chen, S.-H.; Tsai, M.-H.; Tu, Y.-L.; Hua, M.-C.; Yeh, K.-W.; Huang, J.-L. Low cord blood vitamin D levels are associated with increased milk sensitization in early childhood. Pediatr. Allergy Immunol. 2014, 25, 767–772. [Google Scholar] [CrossRef] [PubMed]
- Litonjua, A.A. Vitamin D deficiency as a risk factor for childhood allergic disease and asthma. Curr. Opin. Allergy Clin. Immunol. 2012, 12, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.T.; Litonjua, A.A. Vitamin D dosing for infectious and immune disorders. Thorax 2015, 70, 919–920. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sabetta, J.R.; DePetrillo, P.; Cipriani, R.J.; Smardin, J.; Burns, L.A.; Landry, M.L. Serum 25-Hydroxyvitamin D and the Incidence of Acute Viral Respiratory Tract Infections in Healthy Adults. PLoS ONE 2010, 5, e11088. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, A.; Infante, M.; Ricordi, C. Editorial—Vitamin D status: A key modulator of innate immunity and natural defense from acute viral respiratory infections. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 4048–4052. [Google Scholar]
- Zdrenghea, M.T.; Makrinioti, H.; Bagacean, C.; Bush, A.; Johnston, S.; Stanciu, L.A. Vitamin D modulation of innate immune responses to respiratory viral infections. Rev. Med. Virol. 2017, 27, e1909. [Google Scholar] [CrossRef]
- Vasiliou, J.; Lui, S.; Walker, S.A.; Chohan, V.; Xystrakis, E.; Bush, A.; Hawrylowicz, C.; Saglani, S.; Lloyd, C.M. Vitamin D deficiency induces T h2 skewing and eosinophilia in neonatal allergic airways disease. Allergy 2014, 69, 1380–1389. [Google Scholar] [CrossRef] [PubMed]
- Johansson, S.G.O.; Hourihane, J.; Bousquet, J.; Bruijnzeel-Koomen, C.; Dreborg, S.; Haahtela, T.; Kowalski, M.L.; Mygind, N.; Ring, J.; Van Cauwenberge, P.; et al. A revised nomenclature for allergy: An EAACI position statement from the EAACI nomenclature task force. Allergy 2008, 56, 813–824. [Google Scholar] [CrossRef]
- Chang, Y.-C.; Lee, T.-J.; Huang, C.-C.; Chang, P.-H.; Chen, Y.-W.; Fu, C.-H. The Role of Phadiatop Tests and Total Immunoglobulin E Levels in Screening Aeroallergens: A Hospital-Based Cohort Study. J. Asthma Allergy 2021, 14, 135–140. [Google Scholar] [CrossRef]
- Bousquet, J.; Anto, J.M.; Bachert, C.; Bousquet, P.J.; Colombo, P.; Crameri, R.; Daeron, M.; Fokkens, W.; Leynaert, B.; Lahoz, C.; et al. Factors responsible for differences between asymptomatic subjects and patients presenting an IgE sensitization to allergens. A GA2LEN project. Allergy 2006, 61, 671–680. [Google Scholar] [CrossRef] [PubMed]
- Nowak, S.; Wang, H.; Schmidt, B.; Jarvinen, K.M. Vitamin D and iron status in children with food allergy. Ann. Allergy Asthma Immunol. 2021, 127, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Mirzakhani, H.; Al-Garawi, A.; Weiss, S.T.; Litonjua, A.A. Vitamin D and the development of allergic disease: How important is it? Clin. Exp. Allergy 2014, 45, 114–125. [Google Scholar] [CrossRef]
- Mirzakhani, H.; Carey, V.J.; Zeiger, R.; Bacharier, L.B.; O’Connor, G.T.; Schatz, M.X.; Laranjo, N.; Weiss, S.T.; Litonjua, A.A. Impact of parental asthma, prenatal maternal asthma control, and vitamin D status on risk of asthma and recurrent wheeze in 3-year-old children. Clin. Exp. Allergy 2018, 49, 419–429. [Google Scholar] [CrossRef]
- Alyasin, S.; Momen, T.; Kashef, S.; Alipour, A.; Amin, R. The Relationship Between Serum 25 Hydroxy Vitamin D Levels and Asthma in Children. Allergy Asthma Immunol. Res. 2011, 3, 251–255. [Google Scholar] [CrossRef]
- Douros, K.; Loukou, I.; Boutopoulou, B.; Fouzas, S. Does Vitamin D Deficiency Epidemic Parallel with Allergy and Asthma Epidemic? Mini Rev. Med. Chem. 2015, 15, 967–973. [Google Scholar] [CrossRef]
- Douros, K.; Boutopoulou, B.; Fouzas, S.; Loukou, I. Asthma and Allergy “Epidemic” and the Role of Vitamin D Deficiency. Adv. Exp. Med. Biol. 2017, 996, 169–183. [Google Scholar] [CrossRef] [PubMed]
- Loukou, I.; Moustaki, M.; Sardeli, O.; Plyta, M.; Douros, K. Association of vitamin D status with lung function measurements in children and adolescents with cystic fibrosis. Pediatr. Pulmonol. 2020, 55, 1375–1380. [Google Scholar] [CrossRef]
- Feketea, G.; Bocsan, I.; Tsiros, G.; Voila, P.; Stanciu, L.; Zdrenghea, M. Vitamin D Status in Children in Greece and Its Relationship with Sunscreen Application. Children 2021, 8, 111. [Google Scholar] [CrossRef]
- Kalmpourtzidou, A.; Xinias, I.; Agakidis, C.; Mavroudi, A.; Mouselimis, D.; Tsarouchas, A.; Agakidou, E.; Karagiozoglou-Lampoudi, T. Diet Quality: A Neglected Parameter in Children with Food Allergies. A Cross–Sectional Study. Front. Pediatr. 2021, 9, 344. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Shin, Y.H.; Chung, I.H.; Kim, H.J.; Yoo, E.-G.; Yoon, J.W.; Jee, H.M.; Chang, Y.E.; Han, M.Y. The Link between Serum Vitamin D Level, Sensitization to Food Allergens, and the Severity of Atopic Dermatitis in Infancy. J. Pediatr. 2014, 165, 849–854.e1. [Google Scholar] [CrossRef] [PubMed]
- Brehm, J.M.; Celedón, J.C.; Soto-Quiros, M.E.; Avila, L.; Hunninghake, G.M.; Forno, E.; Laskey, D.; Sylvia, J.S.; Hollis, B.W.; Weiss, S.T.; et al. Serum Vitamin D Levels and Markers of Severity of Childhood Asthma in Costa Rica. Am. J. Respir. Crit. Care Med. 2009, 179, 765–771. [Google Scholar] [CrossRef]
- Thomas, G.O.; Tutar, E.; Tokuc, G.; Oktem, S. 25-hydroxy Vitamin D Levels in Pediatric Asthma Patients and its Link with Asthma Severity. Cureus 2019, 11, e4302. [Google Scholar] [CrossRef]
- Telcian, A.G.; Zdrenghea, M.T.; Edwards, M.R.; Laza-Stanca, V.; Mallia, P.; Johnston, S.L.; Stanciu, L.A. Vitamin D increases the antiviral activity of bronchial epithelial cells in vitro. Antivir. Res. 2017, 137, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-Y.; Forno, E.; Boutaoui, N.; Canino, G.; Celedón, J.C. Vitamin D insufficiency, TH2 cytokines, and allergy markers in Puerto Rican children with asthma. Ann. Allergy Asthma Immunol. 2018, 121, 497–498.e1. [Google Scholar] [CrossRef]
- Toelen, J.; Ritz, N.; de Winter, J.P. Changes in pediatric infections during the COVID-19 pandemic: ‘a quarantrend for coronials’? Eur. J. Nucl. Med. Mol. Imaging 2021, 180, 1965–1967. [Google Scholar] [CrossRef]
- Cauchemez, S.; Valleron, A.-J.; Boëlle, P.-Y.; Flahault, A.; Ferguson, N.M. Estimating the impact of school closure on influenza transmission from Sentinel data. Nature 2008, 452, 750–754. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, T.; Nicholson, E.; Conlon, C.; Barrett, M.; Cummins, F.; Hensey, C.; McAuliffe, E. Assessing the Impact of COVID-19 Public Health Stages on Paediatric Emergency Attendance. Int. J. Environ. Res. Public Health 2020, 17, 6719. [Google Scholar] [CrossRef]
- Polcwiartek, L.B.; Polcwiartek, C.; Andersen, M.P.; Østergaard, L.; Broccia, M.D.; Gislason, G.H.; Køber, L.; Torp-Pedersen, C.; Schou, M.; Fosbøl, E.; et al. Consequences of coronavirus disease-2019 (COVID-19) lockdown on infection-related hospitalizations among the pediatric population in Denmark. Eur. J. Nucl. Med. Mol. Imaging 2021, 180, 1955–1963. [Google Scholar] [CrossRef]
- Poole, S.; Brendish, N.J.; Tanner, A.R.; Clark, T.W. Physical distancing in schools for SARS-CoV-2 and the resurgence of rhinovirus. Lancet Respir. Med. 2020, 8, e92–e93. [Google Scholar] [CrossRef]
- Yeoh, D.K.; A Foley, D.; A Minney-Smith, C.; Martin, A.C.; Mace, A.O.; Sikazwe, C.T.; Le, H.; Levy, A.; Blyth, C.C.; Moore, H.C. Impact of Coronavirus Disease 2019 Public Health Measures on Detections of Influenza and Respiratory Syncytial Virus in Children During the 2020 Australian Winter. Clin. Infect. Dis. 2021, 72, 2199–2202. [Google Scholar] [CrossRef] [PubMed]
- Dogru, M.; Seren, L. Serum 25-hydroxyvitamin D levels in children with recurrent wheezing and relation to the phenotypes and frequency of wheezing. Eur. Ann. Allergy Clin. Immunol. 2017, 49, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Feketea, G.; Bocsan, C.I.; Stanciu, L.A.; Buzoianu, A.D.; Zdrenghea, M.T. The Role of Vitamin D Deficiency in Children With Recurrent Wheezing—Clinical Significance. Front. Pediatr. 2020, 8, 344. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, F. Vitamin-D and COVID-19: Do deficient risk a poorer outcome? Lancet Diabetes Endocrinol. 2020, 8, 570. [Google Scholar] [CrossRef]
- Sassi, F.; Tamone, C.; D’Amelio, P. Vitamin D: Nutrient, Hormone, and Immunomodulator. Nutrients 2018, 10, 1656. [Google Scholar] [CrossRef] [PubMed]
- NICE Guideline. Covid-19 Rapid Guideline: Vitamin D. 2020. Available online: https://www.nice.org.uk/guidance/ng187/resources/covid19-rapid-guideline-vitamin-d-pdf-66142026720709 (accessed on 28 May 2021).
- Norman, A.W. From vitamin D to hormone D: Fundamentals of the vitamin D endocrine system essential for good health. Am. J. Clin. Nutr. 2008, 88, 491S–499S. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Children with Atopy * (n = 137) | Children without Atopy (n = 203) | p | |
|---|---|---|---|---|
| Age (years) ^ | 8 (5–13) | 8 (5–11) | 0.212 | |
| Sex | M | 76 (55.5%) | 79 (38.9%) | 0.03 |
| F | 61 (44.5%) | 124 (61.1%) | ||
| Vitamin D status | Deficient | 37 (27%) | 32 (15,8%) | 0.041 |
| Insufficient | 50 (36.5%) | 85 (41.9%) | ||
| Normal | 50 (36.5%) | 86 (42.3%) | ||
| Eosinophils | Hypereosinophilia ** | 35 (25.5%) | 32 (15.8%) | 0.037 |
| Normal | 102 (74.5%) | 171 (84.2%) | ||
| Parameter | HyperEo * (n = 67) | Normal Eosinophil Count (n = 273) | p | |
|---|---|---|---|---|
| Age ^ (years) | 7 (5–11) | 9 (5–12.5) | 0.187 | |
| Sex | M | 33 (49.3%) | 122 (44.7%) | 0.584 |
| F | 34 (50.7%) | 151 (55.3%) | ||
| Vitamin D status | Deficient | 14 (20.9%) | 55 (20.1%) | 0.723 |
| Insufficient | 29 (43.3%) | 106 (38.8) | ||
| Normal | 24 (35.8%) | 112 (41.1%) | ||
| Vitamin D Status | Age Group 1–4 Years (n = 76) | Age Group 5–18 Years (n = 264) | ||||
|---|---|---|---|---|---|---|
| before Pandemic | during Pandemic | p Value | before Pandemic | during Pandemic | p Value | |
| Deficient (25(OH)D < 20 ng/mL) | 6 (16.2%) | 2 (5.1%) | <0.05 | 44 (33.6%) | 17 (12.8%) | <0.001 |
| Insufficient (25(OH)D 20–30 ng/mL) | 15 (40.5%) | 12 (30.8%) | 52 (39.7%) | 56 (42.1%) | ||
| Sufficient (25(OH)D > 30 ng/mL) | 16 (43.3%) | 25 (64.1%) | 35 (26.7%) | 60 (45.1%) | ||
| Total | 37 | 39 | 131 | 133 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feketea, G.; Vlacha, V.; Tsiros, G.; Voila, P.; Pop, R.M.; Bocsan, I.C.; Stanciu, L.A.; Zdrenghea, M. Vitamin D Levels in Asymptomatic Children and Adolescents with Atopy during the COVID-19 Era. J. Pers. Med. 2021, 11, 712. https://doi.org/10.3390/jpm11080712
Feketea G, Vlacha V, Tsiros G, Voila P, Pop RM, Bocsan IC, Stanciu LA, Zdrenghea M. Vitamin D Levels in Asymptomatic Children and Adolescents with Atopy during the COVID-19 Era. Journal of Personalized Medicine. 2021; 11(8):712. https://doi.org/10.3390/jpm11080712
Chicago/Turabian StyleFeketea, Gavriela, Vasiliki Vlacha, Georgios Tsiros, Panagiota Voila, Raluca Maria Pop, Ioana Corina Bocsan, Luminita Aurelia Stanciu, and Mihnea Zdrenghea. 2021. "Vitamin D Levels in Asymptomatic Children and Adolescents with Atopy during the COVID-19 Era" Journal of Personalized Medicine 11, no. 8: 712. https://doi.org/10.3390/jpm11080712
APA StyleFeketea, G., Vlacha, V., Tsiros, G., Voila, P., Pop, R. M., Bocsan, I. C., Stanciu, L. A., & Zdrenghea, M. (2021). Vitamin D Levels in Asymptomatic Children and Adolescents with Atopy during the COVID-19 Era. Journal of Personalized Medicine, 11(8), 712. https://doi.org/10.3390/jpm11080712










