Preclinical Application of Reduced Manipulated Processing Strategy to Collect Transplantable Hepatocytes: A Pilot and Feasibility Study
Abstract
1. Introduction
2. Methods
2.1. Animals and Ethics Statement
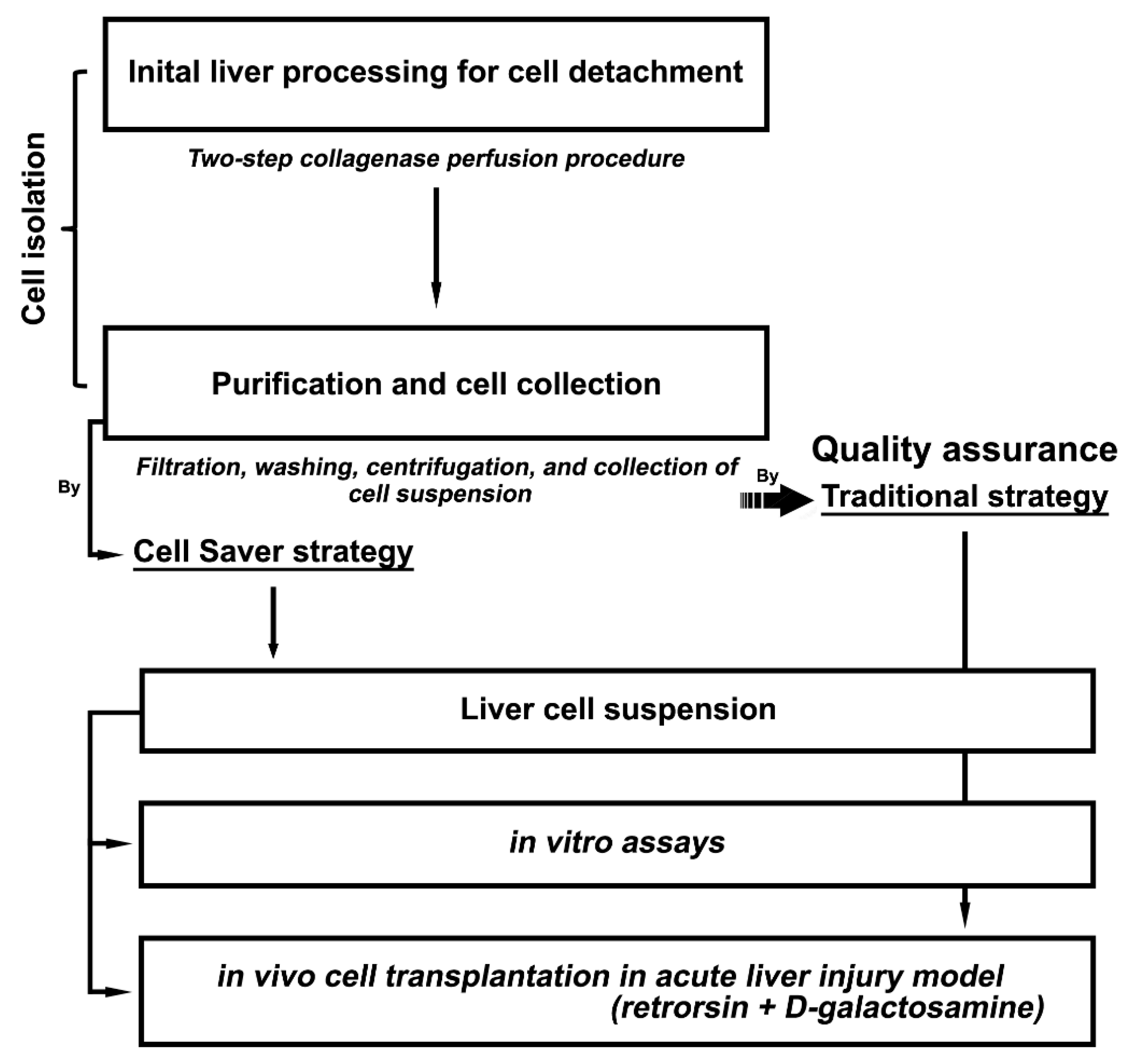
2.2. Experimental Design
2.3. Hepatocyte Isolation and Phenotypic Characterization
2.4. Cell Culture
2.5. Hepatocyte Transplantation
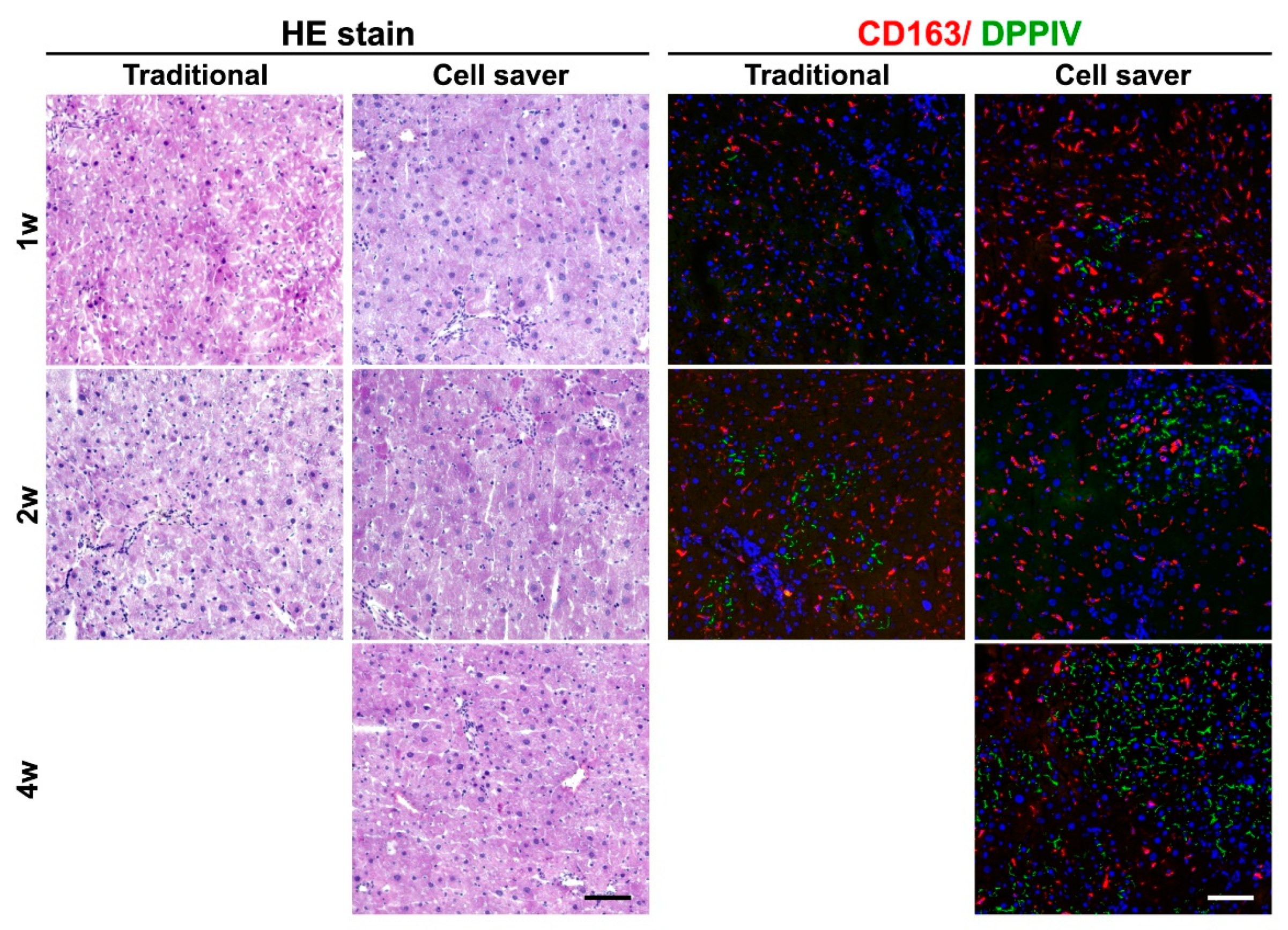
2.6. Hepatic Histology and Determination of Liver Repopulation
2.7. Histochemistry and Immunohistochemistry
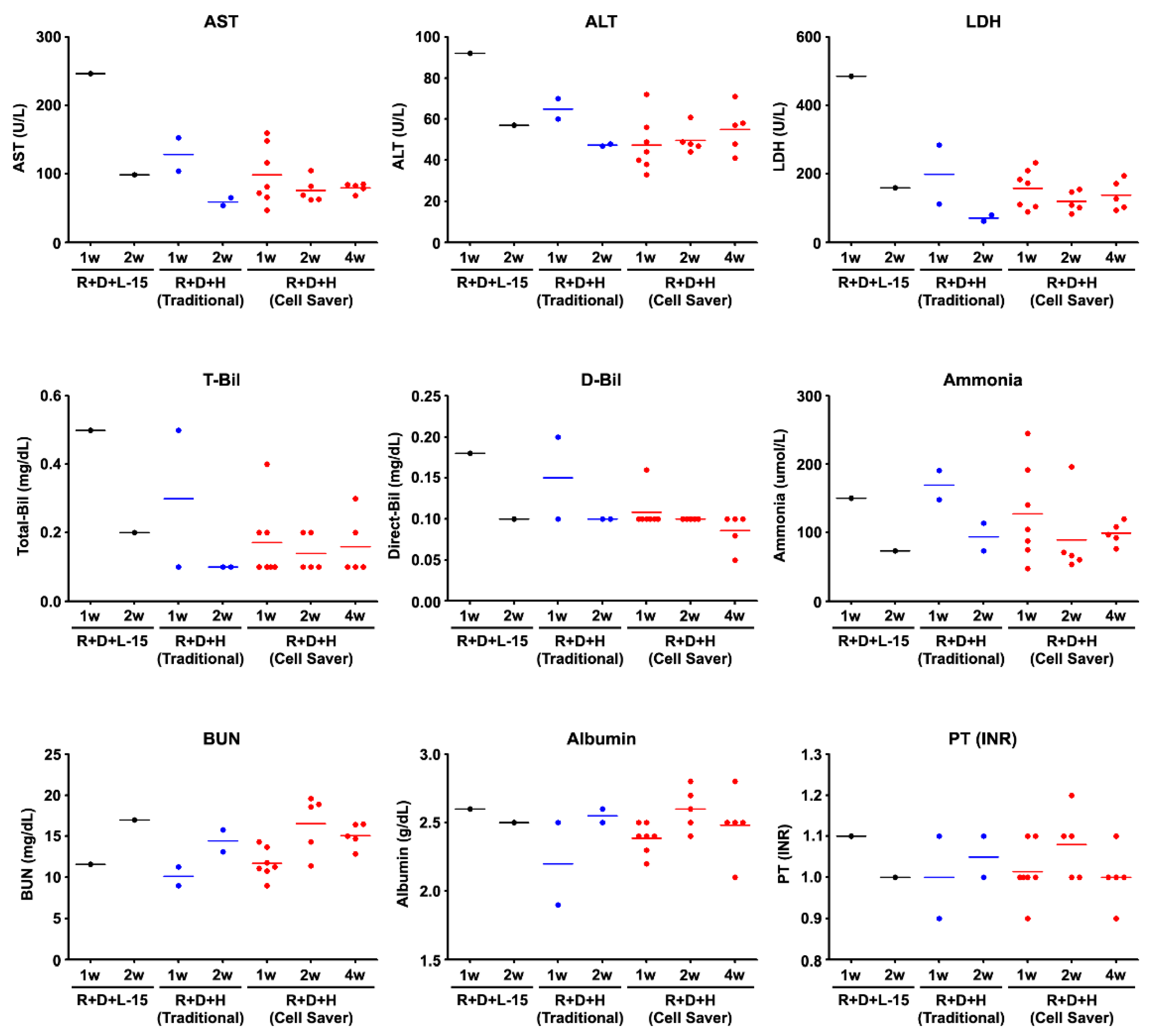
2.8. Serological Assay
2.9. Statistical Analysis
3. Results
3.1. Liver Cell Isolation with Cell Saver® Strategy
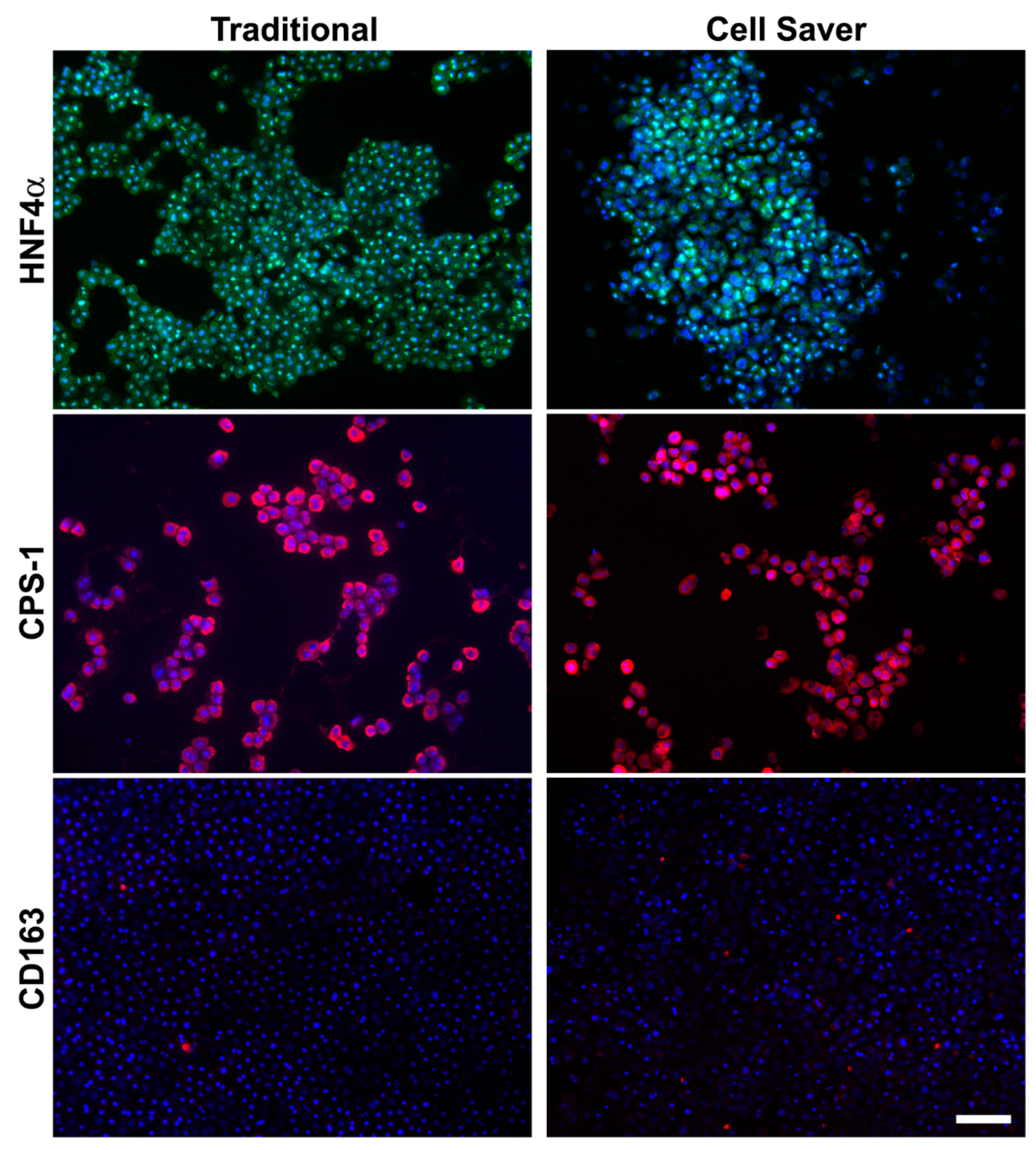
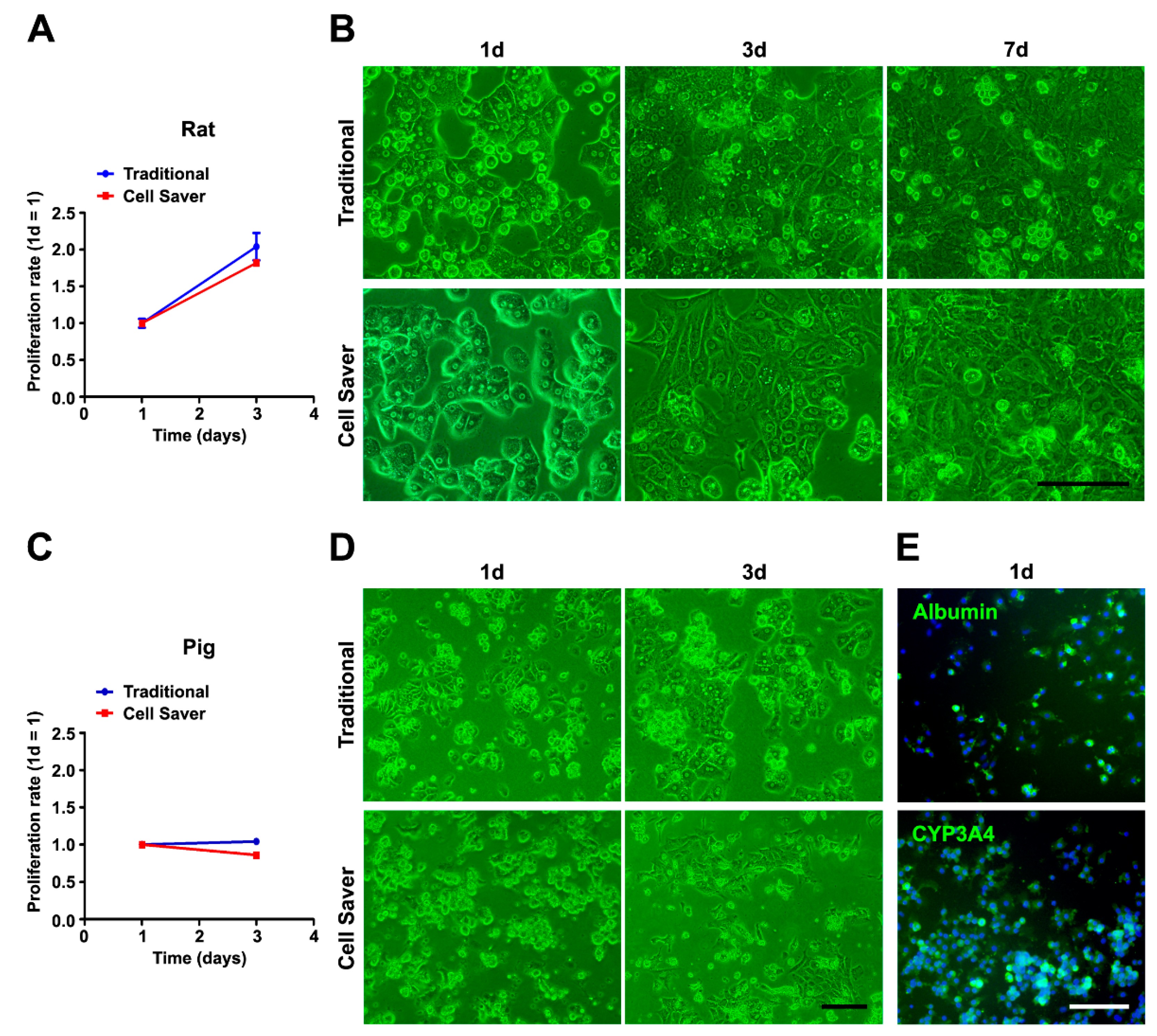
3.2. Characterization of Isolated Cells through In Vitro Assessment
3.3. Engraftment Capability after Cell Transplantation through In Vivo Functional Assessment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviation
References
- Nguyen, M.P.; Jain, V.; Iansante, V.; Mitry, R.R.; Filippi, C.; Dhawan, A. Clinical application of hepatocyte transplantation: Current status, applicability, limitations, and future outlook. Expert Rev. Gastroenterol. Hepatol. 2020, 14, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Weber, A.; Groyer-Picard, M.-T.; Franco, D.; Dagher, I. Hepatocyte transplantation in animal models. Liver Transplant. 2009, 15, 7–14. [Google Scholar] [CrossRef]
- Soltys, K.A.; Soto-Gutiérrez, A.; Nagaya, M.; Baskin, K.M.; Deutsch, M.; Ito, R.; Shneider, B.L.; Squires, R.; Vockley, J.; Guha, C.; et al. Barriers to the successful treatment of liver disease by hepatocyte transplantation. J. Hepatol. 2010, 53, 769–774. [Google Scholar] [CrossRef]
- Ibars, E.P.; Cortes, M.; Tolosa, L.; Gómez-Lechón, M.J.; López, S.; Castell, J.V.; Mir, J. Hepatocyte transplantation program: Lessons learned and future strategies. World J. Gastroenterol. 2016, 22, 874–886. [Google Scholar] [CrossRef] [PubMed]
- Elkin, K.; Yazer, M.; Kameneva, M.; Waters, J. Variable pressure suction vs. constant suction in order to minimize RBC hemolysis during intraoperative blood salvage. Transfusion 2007, 47, 20A–21A. [Google Scholar]
- Cell Saver Elite. The New Standard of Care in Intraoperative Autotransfusion. Available online: http://www.haemotec.co.za/downloads/Autotransfusion-CellSaverElite.pdf (accessed on 5 April 2020).
- Chisholm, J.; Von Tigerstrom, B.; Bedford, P.; Fradette, J.; Viswanathan, S. Workshop to address gaps in regulation of minimally manipulated autologous cell therapies for homologous use in Canada. Cytotherapy 2017, 19, 1400–1411. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-H.; Chen, H.-L.; Chen, Y.-H.; Chang, M.-F.; Chien, C.-S.; Chang, M.-H. Impaired Hepatocyte Regeneration in Acute Severe Hepatic Injury Enhances Effective Repopulation by Transplanted Hepatocytes. Cell Transplant. 2009, 18, 1081–1092. [Google Scholar] [CrossRef]
- Ho, C.-M.; Dhawan, A.; Hughes, R.D.; Lehec, S.C.; Puppi, J.; Philippeos, C.; Lee, P.-H.; Mitry, R.R. Use of indocyanine green for functional assessment of human hepatocytes for transplantation. Asian J. Surg. 2012, 35, 9–15. [Google Scholar] [CrossRef]
- Ho, C.-M.; Chen, Y.-H.; Chien, C.-S.; Ho, Y.-T.; Ho, S.-L.; Hu, R.-H.; Chen, H.-L.; Lee, P.-H. Transplantation speed offers early hepatocyte engraftment in acute liver injured rats: A translational study with clinical implications. Liver Transplant. 2015, 21, 652–661. [Google Scholar] [CrossRef]
- Chen, Y.-H.; Chen, H.-L.; Chien, C.-S.; Wu, S.-H.; Ho, Y.-T.; Yu, C.-H.; Chang, M.-H. Contribution of Mature Hepatocytes to Biliary Regeneration in Rats with Acute and Chronic Biliary Injury. PLoS ONE 2015, 10, e0134327. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Bishop, G.A.; McCaughan, G.W. Altered zonal expression of the CD26 antigen (dipeptidyl peptidase IV) in human cirrhotic liver. Hepatology 1992, 15, 1048–1053. [Google Scholar] [CrossRef] [PubMed]
- Rajvanshi, P.; Kerr, A.; Bhargava, K.K.; Burk, R.D.; Gupta, S. Studies of liver repopulation using the dipeptidyl peptidase IV-deficient rat and other rodent recipients: Cell size and structure relationships regulate capacity for increased transplanted hepatocyte mass in the liver lobule. Hepatology 1996, 23, 482–496. [Google Scholar]
- Aske, K.C.; Waugh, C.A. Expanding the 3R principles: More rigour and transparency in research using animals. EMBO Rep. 2017, 18, 1490–1492. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Pacher, M.; Balakrishnan, A.; Yuan, Q.; Tsay, H.-C.; Yang, D.; Reetz, J.; Brandes, S.; Dai, Z.; Pützer, B.M.; et al. Direct Reprogramming of Hepatic Myofibroblasts into Hepatocytes In Vivo Attenuates Liver Fibrosis. Cell Stem Cell 2016, 18, 797–808. [Google Scholar] [CrossRef]
- Hamashima, Y.; Harter, J.G.; Coons, A.H. The localization of albumin and fibrinogen in human liver cells. J. Cell Biol. 1964, 20, 271–279. [Google Scholar] [CrossRef]
- Morsiani, E.; Rozga, J.; Scott, H.C.; Lebow, L.T.; Moscioni, A.D.; Kong, L.B.; McGrath, M.F.; Demetriou, A.A. Automated liver cell processing facilitates large scale isolation and purification of porcine hepatocytes. ASAIO J. 1995, 41, 155–161. [Google Scholar] [CrossRef]
- Smith, A.J.; O’Rorke, R.D.; Kale, A.; Rimsa, R.; Tomlinson, M.J.; Kirkham, J.; Davies, A.G.; Wälti, C.; Wood, C.D. Rapid cell separation with minimal manipulation for autologous cell therapies. Sci. Rep. 2017, 7, srep41872. [Google Scholar] [CrossRef]
- Werner, M.; Driftmann, S.; Kleinehr, K.; Kaiser, G.M.; Mathé, Z.; Treckmann, J.-W.; Paul, A.; Skibbe, K.; Timm, J.; Canbay, A.; et al. All-In-One: Advanced preparation of Human Parenchymal and Non-Parenchymal Liver Cells. PLoS ONE 2015, 10, e0138655. [Google Scholar] [CrossRef]
- Gimble, J.M.; Katz, A.J.; Bunnell, B.A. Adipose-Derived Stem Cells for Regenerative Medicine. Circ. Res. 2007, 100, 1249–1260. [Google Scholar] [CrossRef]
- Ho, C.-M.; Lin, Y.-J.; Ho, S.-L.; Chen, H.-L. Ductular reaction in acute liver failure. Liver Int. 2016, 36, 761–762. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.J.; Gupta, S.; Dhawan, A. Cell therapy for liver disease: From liver transplantation to cell factory. J. Hepatol. 2015, 62, S157–S169. [Google Scholar] [CrossRef]
- Lee, C.A.; Sinha, S.; Fitzpatrick, E.; Dhawan, A. Hepatocyte transplantation and advancements in alternative cell sources for liver-based regenerative medicine. J. Mol. Med. 2018, 96, 469–481. [Google Scholar] [CrossRef]
- Huebert, R.C.; Rakela, J. Cellular therapy for liver disease. Mayo Clin. Proc. 2014, 89, 414–424. [Google Scholar] [CrossRef] [PubMed]
- Stolz, D.B.; Ross, M.A.; Ikeda, A.; Tomiyama, K.; Kaizu, T.; Geller, D.A.; Murase, N. Sinusoidal endothelial cell repopulation following ischemia/reperfusion injury in rat liver transplantation. Hepatology 2007, 46, 1464–1475. [Google Scholar] [CrossRef]
- Starkey Lewis, P.J.; Moroni, F.; Forbes, S.J. Macrophages as a cell-based therapy for liver disease. Semin. Liver Dis. 2019, 39, 442–451. [Google Scholar] [CrossRef]
- Moroni, F.; Dwyer, B.J.; Graham, C.; Pass, C.; Bailey, L.; Ritchie, L.; Mitchell, D.; Glover, A.; Laurie, A.; Doig, S.; et al. Safety profile of autologous macrophage therapy for liver cirrhosis. Nat. Med. 2019, 25, 1560–1565. [Google Scholar] [CrossRef] [PubMed]
- Dusabineza, A.-C.; Najimi, M.; Van Hul, N.; Legry, V.; Khuu, D.N.; Van Grunsven, L.A.; Sokal, E.; Leclercq, I.A. Hepatic Stellate Cells Improve Engraftment of Human Primary Hepatocytes: A Preclinical Transplantation Study in an Animal Model. Cell Transplant. 2015, 24, 2557–2571. [Google Scholar] [CrossRef] [PubMed]
- Sampaziotis, F.; Justin, A.W.; Tysoe, O.C.; Sawiak, S.; Godfrey, E.M.; Upponi, S.S.; Gieseck, R.L.; De Brito, M.C.; Berntsen, N.L.; Gómez-Vázquez, M.J.; et al. Reconstruction of the mouse extrahepatic biliary tree using primary human extrahepatic cholangiocyte organoids. Nat. Med. 2017, 23, 954–963. [Google Scholar] [CrossRef]
- Camp, J.G.; Sekine, K.; Gerber, T.; Loeffler-Wirth, H.; Binder, H.L.-W.H.; Gac, M.; Kanton, S.; Kageyama, J.; Damm, G.; Seehofer, G.D.D.; et al. Multilineage communication regulates human liver bud development from pluripotency. Nat. Cell Biol. 2017, 546, 533–538. [Google Scholar] [CrossRef]
- Prior, N.; Inacio, P.; Huch, M. Liver organoids: From basic research to therapeutic applications. Gut 2019, 68, 2228–2237. [Google Scholar] [CrossRef]


| Antibodies | Company/Producer | Cat. Number | Dilution |
|---|---|---|---|
| Albumin | Bethyl Laboratories | A90-234A | 1:100 |
| CD163 | Serotec | MCA342R | 1:100 |
| CK-19 | Novacastra, Newcastle | NCL-CK19 | 1:100 |
| CPS-1 | Santa Cruz | SC-10516 | 1:100 |
| CYP3A4 | Santa Cruz | SC-27639 | 1:200 |
| DPPIV | R&D Systems | AF954 | 1:100 |
| HNF4α | Cell Signaling | #3113 | 1:100 |
| SE-1 | Immuno-Biological Laboratories | 10078 | 1:100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.-H.; Chen, H.-L.; Ho, C.-M.; Chen, H.-Y.; Ho, S.-L.; Hu, R.-H.; Lee, P.-H.; Chang, M.-H. Preclinical Application of Reduced Manipulated Processing Strategy to Collect Transplantable Hepatocytes: A Pilot and Feasibility Study. J. Pers. Med. 2021, 11, 326. https://doi.org/10.3390/jpm11050326
Chen Y-H, Chen H-L, Ho C-M, Chen H-Y, Ho S-L, Hu R-H, Lee P-H, Chang M-H. Preclinical Application of Reduced Manipulated Processing Strategy to Collect Transplantable Hepatocytes: A Pilot and Feasibility Study. Journal of Personalized Medicine. 2021; 11(5):326. https://doi.org/10.3390/jpm11050326
Chicago/Turabian StyleChen, Ya-Hui, Hui-Ling Chen, Cheng-Maw Ho, Hung-Yen Chen, Shu-Li Ho, Rey-Heng Hu, Po-Huang Lee, and Mei-Hwei Chang. 2021. "Preclinical Application of Reduced Manipulated Processing Strategy to Collect Transplantable Hepatocytes: A Pilot and Feasibility Study" Journal of Personalized Medicine 11, no. 5: 326. https://doi.org/10.3390/jpm11050326
APA StyleChen, Y.-H., Chen, H.-L., Ho, C.-M., Chen, H.-Y., Ho, S.-L., Hu, R.-H., Lee, P.-H., & Chang, M.-H. (2021). Preclinical Application of Reduced Manipulated Processing Strategy to Collect Transplantable Hepatocytes: A Pilot and Feasibility Study. Journal of Personalized Medicine, 11(5), 326. https://doi.org/10.3390/jpm11050326