Pain Standards for Accredited Healthcare Organizations (ACDON Project): A Mixed Methods Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Review (First Phase)
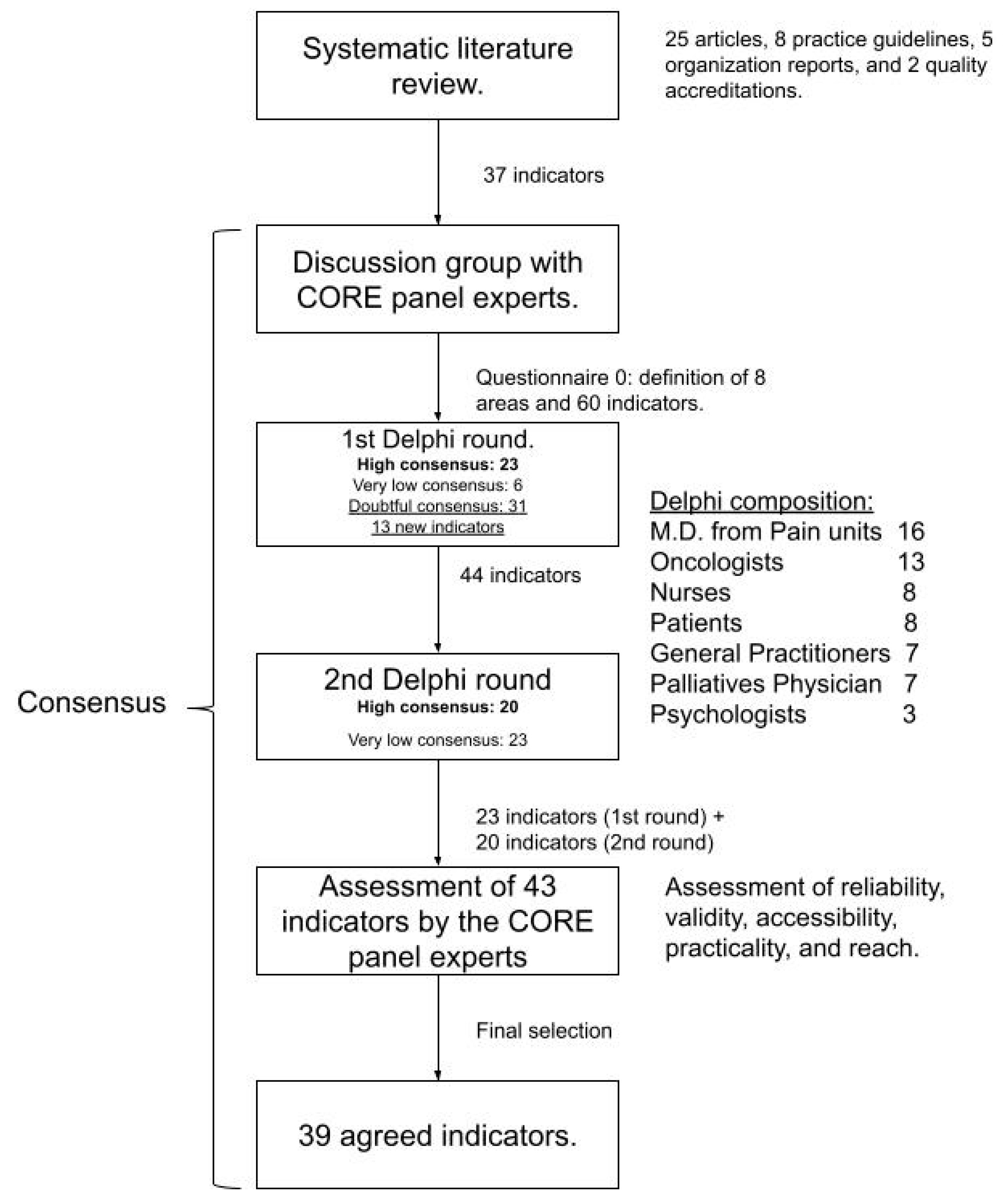
2.2. Nominal Group Technique (Second Phase)
2.3. Delphi Technique (Third Phase)
2.4. Pilot Study (Fourth Phase)
3. Results
3.1. Literature Review (First Phase)
3.2. Nominal Group Technique (Second Phase)
3.3. Delphi Technique (Third Phase)
3.4. Pilot Study (Fourth Phase)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lema, M.J.; Foley, K.M.; Hausheer, F.H. Types and Epidemiology of Cancer-Related Neuropathic Pain: The Intersection of Cancer Pain and Neuropathic Pain. Oncologist 2010, 15, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Shahi, P.K.; Rueda, A.C.; del Pérez Manga, G. Manejo del dolor oncológico. An. Med. Interna 2007, 24, 554–557. [Google Scholar]
- Everdingen, M.V.D.B.-V.; De Rijke, J.M.; Kessels, A.G.; Schouten, H.C.; Van Kleef, M.; Patijn, J. High prevalence of pain in patients with cancer in a large population-based study in The Netherlands. Pain 2007, 132, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Fallon, M.; Giusti, R.; Aielli, F.; Hoskin, P.; Rolke, R.; Sharma, M.; Ripamonti, C. Management of cancer pain in adult patients: ESMO Clinical Practice Guidelines. Ann. Oncol. 2018, 29, iv166–iv191. [Google Scholar] [CrossRef]
- Hui, D.; I Cherny, N.; Wu, J.; Liu, D.; Latino, N.J.; Strasser, F. Indicators of integration at ESMO Designated Centres of Integrated Oncology and Palliative Care. ESMO Open 2018, 3, e000372. [Google Scholar] [CrossRef] [PubMed]
- Hernández, D.C.P.; Babarro, A.A.; Aguerri, A.R.; Estévez, F.V.; Echaburu, J.A.V.; Moncayo, F.G.; Alarcón, D.D.L.; Campos, F.L.; Ferri, C.M. Guía para el Abordaje Interdisciplinar del Dolor Oncológico; Sociedad Española de Oncología Médica: Madrid, Spain, 2018. [Google Scholar]
- Reis-Pina, P.; Lawlor, P.G.; Barbosa, A. Adequacy of cancer-related pain management and predictors of undertreatment at referral to a pain clinic. J. Pain Res. 2017, 10, 2097–2107. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Cancer Pain Relief; WHO: Geneva, Switzerland, 1986. [Google Scholar]
- Bennett, M.; Eisenberg, E.; Ahmedzai, S.H.; Bhaskar, A.; O’Brien, T.; Mercadante, S.; Škvarč, N.K.; Vissers, K.; Wirz, S.; Wells, C.; et al. Standards for the management of cancer-related pain across Europe—A position paper from the EFIC Task Force on Cancer Pain. Eur. J. Pain 2019, 23, 660–668. [Google Scholar] [CrossRef]
- Smith, T.J.; Saiki, C.B. Cancer Pain Management. Mayo Clin. Proc. 2015, 90, 1428–1439. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Everdingen, M.H.V.D.B.-V.; De Rijke, J.; Kessels, A.; Schouten, H.; Van Kleef, M.; Patijn, J. Prevalence of pain in patients with cancer: A systematic review of the past 40 years. Ann. Oncol. 2007, 18, 1437–1449. [Google Scholar] [CrossRef]
- American Society of Clinical Oncology. QOPI Standards Manual—Certificaction Program; American Society of Clinical Oncology: Alexandria, VA, USA, 2018. [Google Scholar]
- Fiset, V.J.; Davies, B.L.; Graham, I.D.; Gifford, W.; Woodend, K. Developing guideline-based quality indicators. Int. J. Evidence Based Health 2019, 17, 92–105. [Google Scholar] [CrossRef]
- Harbour, R.; Miller, J. A new system for grading recommendations in evidence based guidelines. BMJ 2001, 323, 334–336. [Google Scholar] [CrossRef]
- Babchuk, W.A. Fundamentals of qualitative analysis in family medicine. Family Med Community Health 2019, 7, e000040. [Google Scholar] [CrossRef]
- Weiner, B.J.; Amick, H.R.; Lund, J.L.; Lee, S.-Y.D.; Hoff, T.J. Review: Use of Qualitative Methods in Published Health Services and Management Research: A 10-Year Review. Med. Care Res. Rev. 2010, 68, 3–33. [Google Scholar] [CrossRef] [PubMed]
- Mira, J.J.; Pérez-Jover, V.; Lorenzo, S.; Aranaz, J.; Vitaller, J. Qualitative research is a valid alternative too. Aten. Primaria 2004, 34, 161–166. [Google Scholar] [CrossRef]
- Kavalieratos, D.; Corbelli, J.; Zhang, D.; Dionne-Odom, J.N.; Ernecoff, N.C.; Hanmer, J.; Hoydich, Z.P.; Ikejiani, D.Z.; Klein-Fedyshin, M.; Zimmermann, C.; et al. Association Between Palliative Care and Patient and Caregiver Outcomes. JAMA 2016, 316, 2104–2114. [Google Scholar] [CrossRef]
- Gaertner, J.; Siemens, W.; Meerpohl, J.J.; Antes, G.; Meffert, C.; Xander, C.; Stock, S.; Mueller, D.; Schwarzer, G.; Becker, G. Effect of specialist palliative care services on quality of life in adults with advanced incurable illness in hospital, hospice, or community settings: Systematic review and meta-analysis. BMJ 2017, 357, j2925. [Google Scholar] [CrossRef] [PubMed]
- Haun, M.W.; Estel, S.; Rücker, G.; Friederich, H.-C.; Villalobos, M.; Thomas, M.; Hartmann, M. Early palliative care for adults with advanced cancer. Cochrane Database Syst. Rev. 2017, 6, CD011129. [Google Scholar] [CrossRef]
- I Cherny, N.; Catane, R.; Schrijvers, D.; Kloke, M.; Strasser, F. European Society for Medical Oncology (ESMO) Program for the Integration of Oncology and Palliative Care: A 5-year review of the Designated Centers’ incentive program. Ann. Oncol. 2009, 21, 362–369. [Google Scholar] [CrossRef]
- Janjan, N. Improving Cancer Pain Control with NCCN Guideline-Based Analgesic Administration: A Patient-Centered Outcome. J. Natl. Compr. Cancer Netw. 2014, 12, 1243–1249. [Google Scholar] [CrossRef]
- Scarborough, B.M.; Smith, C.B. Optimal pain management for patients with cancer in the modern era. CA A Cancer J. Clin. 2018, 68, 182–196. [Google Scholar] [CrossRef] [PubMed]
- Mira, J.J.; Guilabert, M.; Pérez-Jover, V.; Lorenzo, S. Barriers for an effective communication around clinical decision making: An analysis of the gaps between doctors’ and patients’ point of view. Health Expect. 2012, 17, 826–839. [Google Scholar] [CrossRef]
- Paley, C.A.; I Johnson, M.; Tashani, O.A.; Bagnall, A.-M. Acupuncture for cancer pain in adults. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Law, E.F.; Fisher, E.; Eccleston, C.; Palermo, T.M. Psychological interventions for parents of children and adolescents with chronic illness. Cochrane Database Syst. Rev. 2019, 3, CD009660. [Google Scholar] [CrossRef] [PubMed]

| Area | Description | Number of Indicators |
|---|---|---|
| Evaluation and assessment of oncological pain | Quality criteria were incorporated with a focus on improving pain assessment using multidimensional scales and patient participation. In addition, assessment of family members and communication between the care team, caregivers, family members, and patients for adequate therapeutic adherence and improvement of clinical practice were included. | 5 |
| Pharmacological treatment | Treatment should be multimodal, through combining individualized strategies, treatment of underlying cause, modification of the underlying treatment, and the use of rescue treatment therapies and interventional techniques were included. Quality criteria to ensure pharmacological treatment has a continuous pattern that has to be agreed between professional and patient, including additional therapeutic demand (rescue) in the event of breakthrough pain were included. | 6 |
| Non-pharmacological treatment | Patients with chronic oncological pain requires a multidimensional approach. Quality criteria have been included to optimize pain management by considering proposals pertaining non-pharmacological measures that were aimed at improving the outcomes of cancer pain management with the ultimate goal of alleviating patient suffering. | 5 |
| Palliative care | Access to palliative care services for patients with chronic cancer pain is fundamental. Palliative care should be included in the routine management of patients to ensure that there is a continuous assessment, and that the patient can be duly informed in a timely manner. | 4 |
| Coordination | Effective coordination was highlighted for the optimal functioning of multidisciplinary teams. This coordination should be accompanied by information about the pain unit and other services that are available to the patient. | 4 |
| Training, teaching, and research | It would be necessary for the service to include the training for professionals to achieve adequate communication of bad news to patients and families as well as for activities to prevent burnout among physicians. Research has been highlighted as a necessary activity for improving care. | 5 |
| Patient safety | Quality criteria related to safe medication use, the prevention and control of drug interactions and adverse events as well as their timely reporting were included to ensure patient safety. | 6 |
| Patient satisfaction | All clinical efforts should be aimed at improving the patient’s quality of life and experience with health services. Results related to analgesia should be obtained from patients and relatives as well as the degree of dependence of the patient and the care and multimodal attention that was received. | 4 |
| Hospital | Essential Standards (Fulfilled/Total) | Recommended Standards (Fulfilled/Total) | Overall Compliance |
|---|---|---|---|
| Hospital 1 | 8/10 | 20/29 | 71.8% |
| Hospital 2 | 10/10 | 29/29 | 100% |
| Hospital 3 | 7/10 | 15/29 | 56.4% |
| Hospital 4 | 9/10 | 27/29 | 92.3% |
| Hospital 5 | 10/10 | 22/29 | 82.1% |
| Hospital 6 | 8/10 | 19/29 | 69.2% |
| Hospital 7 | 10/10 | 21/29 | 79.5% |
| Hospital 8 | 10/10 | 29/29 | 100% |
| Areas | Number of Standards | Overall Compliance |
|---|---|---|
| Assessment and counselling for cancer pain | 5 | 85% |
| Pharmacological treatment | 6 | 92.5% |
| Non-pharmacological treatment | 5 | 80% |
| Palliative Care | 4 | 94.7% |
| Coordination | 4 | 65.6% |
| Training, education, and research | 5 | 62.5% |
| Patient Safety | 6 | 87.5% |
| Patient Satisfaction | 4 | 78.1% |
| Areas | Standards | Overall Compliance |
|---|---|---|
| Pharmacological treatment | A specific record should be kept in the digital medical record of the “itinerary” through the WHO analgesic ladder, as well as the reasons for it (at the discretion of your responsible physician), to facilitate the best coordination between the responsible team and the inter-current (emergency) teams. | 62.5% |
| Non-pharmacological treatment | In case of pain that does not subside with non-invasive treatment, a non-pharmacological interventional technique should be proposed to the patient. | 62.5% |
| Coordination | Patient information should be provided about the Pain Unit, its portfolio of services, accessibility and the strategy designed for the care of patients with chronic oncological pain in a proactive manner. | 62.5% |
| High resolution consultations should be implemented to reduce the discomfort associated with in-between-consultations. | 50% | |
| A telephone follow-up plan should be established by calling the home of patients with cancer pain. | 62.5% | |
| Training, education, and research | Actions should be taken to prevent burnout among professionals. For example: Increased psychological support to reduce burnout among professionals. | 25% |
| The Pain Unit should participate in the development of new treatments and advances in the improvement of therapeutic effectiveness in the management of oncological pain, participating in research projects and clinical trials with direct translation to clinical practice. | 50% | |
| Patient Safety | The reasons for deciding not to follow the WHO pain management ladder should be recorded in the digital medical record. | 62.5% |
| Patient Satisfaction | There must be a procedure for assessing the patient’s experience and using this information to improve the care process. | 50% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pérez, C.; Martin-Delgado, J.; Vinuesa, M.; Ibor, P.J.; Guilabert, M.; Gomez, J.; Beato, C.; Sánchez-Jiménez, J.; Velázquez, I.; Calvo-Espinos, C.; et al. Pain Standards for Accredited Healthcare Organizations (ACDON Project): A Mixed Methods Study. J. Pers. Med. 2021, 11, 102. https://doi.org/10.3390/jpm11020102
Pérez C, Martin-Delgado J, Vinuesa M, Ibor PJ, Guilabert M, Gomez J, Beato C, Sánchez-Jiménez J, Velázquez I, Calvo-Espinos C, et al. Pain Standards for Accredited Healthcare Organizations (ACDON Project): A Mixed Methods Study. Journal of Personalized Medicine. 2021; 11(2):102. https://doi.org/10.3390/jpm11020102
Chicago/Turabian StylePérez, Concepción, Jimmy Martin-Delgado, Mercedes Vinuesa, Pedro J. Ibor, Mercedes Guilabert, José Gomez, Carmen Beato, Juana Sánchez-Jiménez, Ignacio Velázquez, Claudio Calvo-Espinos, and et al. 2021. "Pain Standards for Accredited Healthcare Organizations (ACDON Project): A Mixed Methods Study" Journal of Personalized Medicine 11, no. 2: 102. https://doi.org/10.3390/jpm11020102
APA StylePérez, C., Martin-Delgado, J., Vinuesa, M., Ibor, P. J., Guilabert, M., Gomez, J., Beato, C., Sánchez-Jiménez, J., Velázquez, I., Calvo-Espinos, C., Cánovas, M. L., Yáñez, J. A., Rodríguez, M., Baquero, J. L., Gallach, E., Folch, E., Tuca, A., Santiña, M., & Mira, J. J. (2021). Pain Standards for Accredited Healthcare Organizations (ACDON Project): A Mixed Methods Study. Journal of Personalized Medicine, 11(2), 102. https://doi.org/10.3390/jpm11020102










