Genomic Characterization of Host Factors Related to SARS-CoV-2 Infection in People with Dementia and Control Populations: The GR@ACE/DEGESCO Study
Abstract
:1. Introduction
2. Methods
2.1. Study Participants
2.2. Meta-Analyses
2.3. Sample Processing, Genotyping, Quality Control, and Imputation
2.4. Targeted Phenotypes and Statistical Analyses
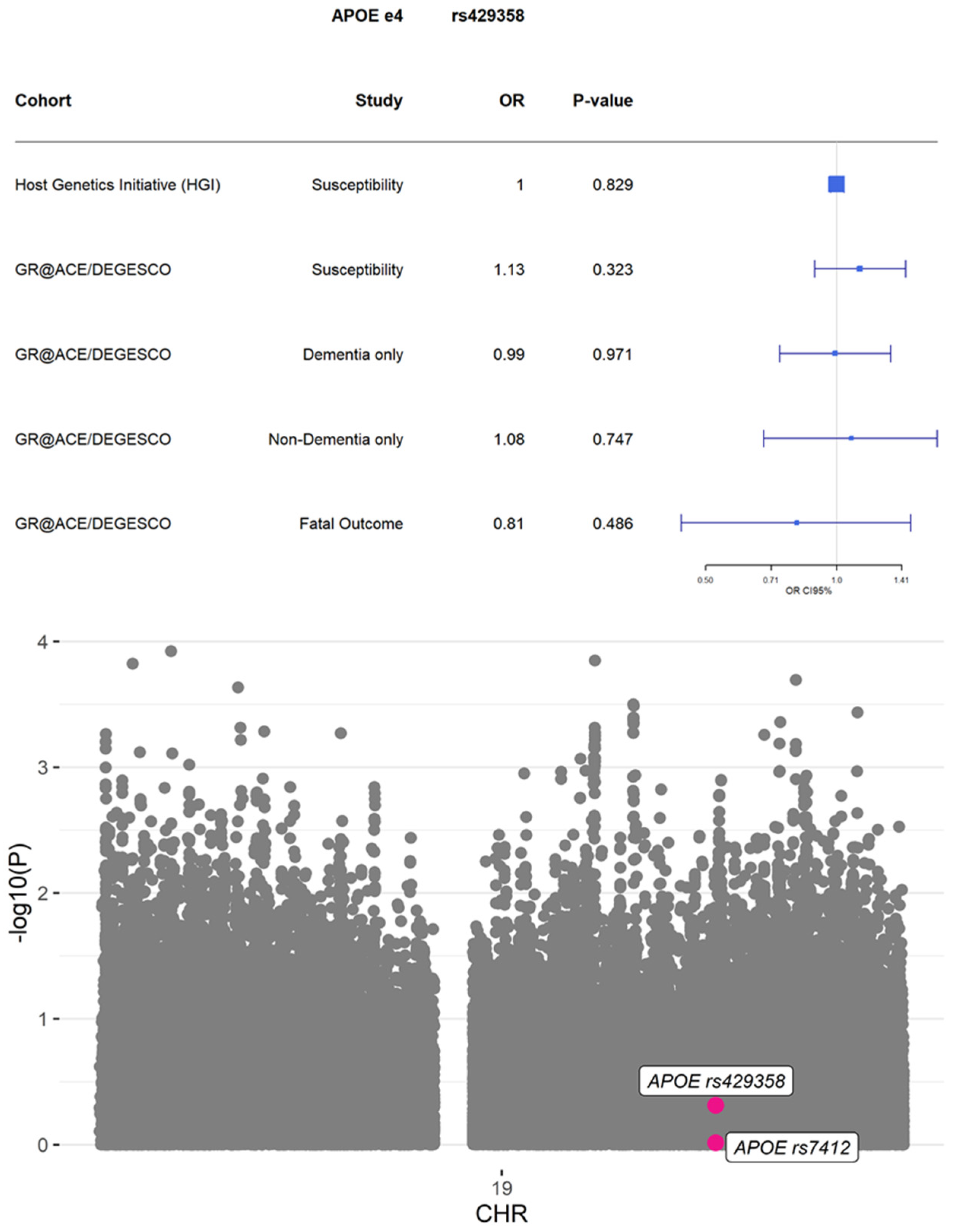
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- COVID-19 Host Genetics Initiative. The COVID-19 Host Genetics Initiative, a global initiative to elucidate the role of host genetic factors in susceptibility and severity of the SARS-CoV-2 virus pandemic. Eur. J. Hum. Genet. 2020, 28, 715–718. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Liu, W.; Liu, K.; Fang, Y.-Y.; Shang, J.; Zhou, L.; Wang, K.; Leng, F.; Wei, P.-F.; Chen, L.; et al. Clinical characteristics of fatal and recovered cases of coronavirus disease 2019 in Wuhan, China: A retrospective study. Chin. Med. J. 2020, 133, 1261–1267. [Google Scholar] [CrossRef] [PubMed]
- Onder, G.; Rezza, G.; Brusaferro, S. Case-Fatality Rate and Characteristics of Patients Dying in Relation to COVID-19 in Italy. JAMA 2020, 323, 1775–1776. [Google Scholar] [CrossRef] [PubMed]
- Severe COVID-19 GWAS Group; Ellinghaus, D.; Degenhardt, F.; Bujanda, L.; Buti, M.; Albillos, A.; Invernizzi, P.; Fernandez, J.; Prati, D.; Baselli, G.; et al. Genomewide Association Study of Severe COVID-19 with Respiratory Failure. N. Engl. J. Med. 2020, 383, 1522–1534. [Google Scholar] [CrossRef] [PubMed]
- Pairo-Castineira, E.; Clohisey, S.; Klaric, L.; Bretherick, A.D.; Rawlik, K.; Pasko, D.; Walker, S.; Parkinson, N.; Fourman, M.H.; Russell, C.D.; et al. Genetic mechanisms of critical illness in COVID-19. Nature 2021, 591, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Torre-Fuentes, L.; Matías-Guiu, J.; Hernández-Lorenzo, L.; Montero-Escribano, P.; Pytel, V.; Porta-Etessam, J.; Gómez-Pinedo, U.; Matías-Guiu, J.A. ACE2, TMPRSS2, and Furin variants and SARS-CoV-2 infection in Madrid, Spain. J. Med. Virol. 2021, 93, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Roberts, G.H.; Park, D.S.; Coignet, M.V.; McCurdy, S.R.; Knight, S.C.; Partha, R.; Rhead, B.; Zhang, M.; Berkowitz, N.; Baltzell, A.K.H.; et al. AncestryDNA COVID-19 Host Genetic Study Identifies Three Novel Loci 1. medRxiv 2020. Available online: https://www.medrxiv.org/content/10.1101/2020.10.06.20205864v1 (accessed on 9 October 2020).
- Kuo, C.-L.; Pilling, L.C.; Atkins, J.L.; Masoli, J.A.H.; Delgado, J.; Kuchel, G.A.; Melzer, D. APOE e4 Genotype Predicts Severe COVID-19 in the UK Biobank Community Cohort. J. Gerontol. Ser. A Biol. Sci. Med Sci. 2020, 75, 2231–2232. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, M.R.; Poland, G.A.; Graeber, C.W. Does apolipoprotein E genotype predict COVID-19 severity? QJM Int. J. Med. 2020, 113, 529–530. [Google Scholar] [CrossRef] [PubMed]
- Del Ser, T.; Fernández-Blázquez, M.A.; Valentí, M.; Zea-Sevilla, M.A.; Frades, B.; Alfayate, E.; Saiz, L.; Calero, O.; García-López, F.J.; Rábano, A.; et al. Residence, Clinical Features, and Genetic Risk Factors Associated with Symptoms of COVID-19 in a Cohort of Older People in Madrid. Gerontology 2021, 67, 281–289. [Google Scholar] [CrossRef]
- Moreno-Grau, S.; de Rojas, I.; Hernández, I.; Quintela, I.; Montrreal, L.; Alegret, M.; Hernández-Olasagarre, B.; Madrid, L.; González-Perez, A.; Maroñas, O.; et al. Genome-wide association analysis of dementia and its clinical endophenotypes reveal novel loci associated with Alzheimer’s disease and three causality networks: The GR@ACE project. Alzheimer Dement. 2019, 15, 1333–1347. [Google Scholar] [CrossRef]
- De Rojas, I.; Moreno-Grau, S.; Tesi, N.; Grenier-Boley, B.; Andrade, V.; Jansen, I.E.; Pedersen, N.L.; Stringa, N.; Zettergren, A.; Hernández, I.; et al. Common variants in Alzheimer’s disease and risk stratification by polygenic risk scores. Nat. Commun. 2021, 12, 3417. [Google Scholar] [CrossRef] [PubMed]
- Benaque, A.; Gurruchaga, M.J.; Abdelnour, C.; Hernández, I.; Cañabate, P.; Alegret, M.; Rodríguez, I.; Rosende-Roca, M.; Tartari, J.P.; Esteban, E.; et al. Dementia Care in Times of COVID-19: Experience at Fundació ACE in Barcelona, Spain. J. Alzheimer Dis. 2020, 76, 33–40. [Google Scholar] [CrossRef]
- Taliun, D.; NHLBI Trans-Omics for Precision Medicine (TOPMed) Consortium; Harris, D.N.; Kessler, M.D.; Carlson, J.; Szpiech, Z.A.; Torres, R.; Taliun, S.A.G.; Corvelo, A.; Gogarten, S.M.; et al. Sequencing of 53,831 diverse genomes from the NHLBI TOPMed Program. Nat. Cell Biol. 2021, 590, 290–299. [Google Scholar] [CrossRef]
- Das, S.; Forer, L.; Schönherr, S.; Sidore, C.; Locke, A.E.; Kwong, A.; Vrieze, S.I.; Chew, E.Y.; Levy, S.; McGue, M.; et al. Next-generation genotype imputation service and methods. Nat. Genet. 2016, 48, 1284–1287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuchsberger, C.; Abecasis, G.R.; Hinds, D. minimac2: Faster genotype imputation. Bioinformatics 2015, 31, 782–784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strittmatter, W.J.; Weisgraber, K.H.; Huang, D.Y.; Dong, L.M.; Salvesen, G.S.; Pericak-Vance, M.; Schmechel, D.; Saunders, A.M.; Goldgaber, D.; Roses, A.D. Binding of human apolipoprotein E to synthetic amyloid beta peptide: Isoform-specific effects and implications for late-onset Alzheimer disease. Proc. Natl. Acad. Sci. USA 1993, 90, 8098–8102. [Google Scholar] [CrossRef] [Green Version]
- Pericak-Vance, M.A.; Bebout, J.L.; Gaskell, P.C.; Yamaoka, L.H.; Hung, W.-Y.; Alberts, M.J.; Walker, A.P.; Bartlett, R.J.; Haynes, C.A.; Welsh, K.A.; et al. Linkage studies in familial Alzheimer disease: Evidence for chromosome 19 linkage. Am. J. Hum. Genet. 1991, 48, 1034–1050. [Google Scholar]
- Mahley, R.W. Apolipoprotein E: From cardiovascular disease to neurodegenerative disorders. J. Mol. Med. 2016, 94, 739–746. [Google Scholar] [CrossRef] [Green Version]
- Sebastiani, P.; Gurinovich, A.; Nygaard, M.; Sasaki, T.; Sweigart, B.; Bae, H.; Andersen, S.L.; Villa, F.; Atzmon, G.; Christensen, K.; et al. APOE Alleles and extreme human longevity. J. Gerontol. Ser. A 2019, 74, 44–51. [Google Scholar]
- Deelen, J.; Evans, D.S.; Arking, D.E.; Tesi, N.; Nygaard, M.; Liu, X.; Wojczynski, M.K.; Biggs, M.L.; van der Spek, A.; Atzmon, G.; et al. A meta-analysis of genome-wide association studies identifies multiple longevity genes. Nat. Commun. 2019, 10, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Willer, C.; Li, Y.; Abecasis, G.R. METAL: Fast and efficient meta-analysis of genomewide association scans. Bioinformatics 2010, 26, 2190–2191. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-L.; Pilling, L.C.; Atkins, J.L.; Masoli, J.A.; Delgado, J.; Kuchel, G.A.; Melzer, D. ApoE e4e4 genotype and mortality with COVID-19 in UK Biobank. J. Gerontol. Ser. A 2020, 75, 1801–1803. [Google Scholar] [CrossRef]
- Shelton, J.F.; Shastri, A.J.; Ye, C.; Weldon, C.H.; Filshtein-Somnez, T.; Coker, D.; Symons, A.; Esparza-Gordillo, J.; Aslibekyan, S.; Auton, A. Trans-ethnic analysis reveals genetic and non-genetic associations with COVID-19 susceptibility and severity. medRxiv 2020. [Google Scholar] [CrossRef]
- Alcolea, D.; Clarimón, J.; Carmona-Iragui, M.; Illán-Gala, I.; Morenas-Rodríguez, E.; Barroeta, I.; Ribosa-Nogué, R.; Sala, I.; Sánchez-Saudinós, M.B.; Videla, L.; et al. The Sant Pau Initiative on Neurodegeneration (SPIN) cohort: A data set for biomarker discovery and validation in neurodegenerative disorders. Alzheimer Dement. 2019, 5, 597–609. [Google Scholar] [CrossRef]
- Lonsdale, J.; Thomas, J.; Salvatore, M.; Phillips, R.; Lo, E.; Shad, S.; Hasz, R.; Walters, G.; Garcia, F.; Young, N.; et al. The Genotype-Tissue Expression (GTEx) project. Nat. Genet. 2013, 45, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Kleine-Weber, H.; Krüger, N.; Mueller, M.A.; Drosten, C.; Pöhlmann, S. The novel coronavirus 2019 (2019-nCoV) uses the SARS-coronavirus receptor ACE2 and the cellular protease TMPRSS2 for entry into target cells. Cell 2020. [Google Scholar] [CrossRef]
- O’Driscoll, M.; Dos Santos, G.R.; Wang, L.; Cummings, D.A.T.; Azman, A.S.; Paireau, J.; Fontanet, A.; Cauchemez, S.; Salje, H. Age-specific mortality and immunity patterns of SARS-CoV-2. Nature 2021, 590, 140–145. [Google Scholar] [CrossRef] [PubMed]
- Corder, E.H.; Saunders, A.M.; Strittmatter, W.J.; Schmechel, D.E.; Gaskell, P.C.; Small, G.W.; Roses, A.D.; Haines, J.L.; Pericak-Vance, M.A. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science 1993, 261, 921–923. [Google Scholar] [CrossRef]
- Daniloski, Z.; Jordan, T.X.; Wessels, H.H.; Hoagland, D.A.; Kasela, S.; Legut, M.; Maniatis, S.; Mimitou, E.P.; Lu, L.; Geller, E.; et al. Identification of Required Host Factors for SARS-CoV-2 Infection in Human Cells. Cell 2021, 184, 92–105.e16. [Google Scholar] [CrossRef]
- Nuriel, T.; Peng, K.Y.; Ashok, A.; Dillman, A.A.; Figueroa, H.Y.; Apuzzo, J.; Ambat, J.; Levy, E.; Cookson, M.R.; Mathews, P.M.; et al. The Endosomal–Lysosomal Pathway Is Dysregulated by APOE4 Expression in Vivo. Front. Neurosci. 2017, 11, 702. [Google Scholar] [CrossRef] [PubMed]
- Bellenguez, C.; Küçükali, F.; Jansen, I.; Andrade, V.; Moreno-grau, S. New insights on the genetic etiology of Alzheimer’s and related dementia. MedRxiv 2020. [Google Scholar] [CrossRef]
- Wood, H. Elevated risk of COVID-19 in people with dementia. Nat. Rev. Neurol. 2021, 17, 194. [Google Scholar] [CrossRef] [PubMed]
- Peterson, A.; Largent, E.A.; Karlawish, J. Ethics of reallocating ventilators in the COVID-19 pandemic. BMJ 2020, 369, m1828. [Google Scholar]
- Triage Decisions during COVID-19 Pandemic—Our Opinion on Policy—Alzheimer Europe. Available online: https://www.alzheimer-europe.org/Policy/Our-opinion-on/Triage-decisions-during-COVID-19-pandemic (accessed on 8 April 2020).
- Wang, Q.; Davis, P.B.; Gurney, M.E.; Xu, R. COVID-19 and dementia: Analyses of risk, disparity, and outcomes from electronic health records in the US. Alzheimer Dement. 2021, 17, 1297–1306. [Google Scholar] [CrossRef] [PubMed]
| Whole Study | |||
|---|---|---|---|
| Cases with COVID-19 | Population | Total | |
| Participants (n) | 221 | 17,035 | 17,256 |
| Gender (% Female) | 69.68 | 58.78 | 58.92 |
| Age, years (SD) | 71.08 (17.55) | 70.45 (16.49) | 70.46 (16.51) |
| APOE4 carriers (%) | 34.39 | 30.13 | 30.19 |
| Dementia (%) | 57.92 | 45.79 | 45.95 |
| Dementia individuals | |||
| Cases with COVID-19 | Population | Total | |
| Participants (n) | 128 | 7801 | 7929 |
| Gender (% Female) | 78.12 | 68.43 | 68.58 |
| Age, years (SD) | 75.4 (17.29) | 72.86 (14.94) | 72.93 (15.01) |
| APOE4 carriers (%) | 42.19 | 41.35 | 41.37 |
| Non-Dementia individuals | |||
| Cases with COVID-19 | Population | Total | |
| Participants (n) | 93 | 9223 | 9316 |
| Gender (% Female) | 58.06 | 50.61 | 50.69 |
| Age, years (SD) | 65.16 (16.21) | 69.05 (17.17) | 69 (17.16) |
| APOE4 carriers (%) | 23.66 | 20.64 | 20.67 |
| Individuals with COVID-19 | |||
| Cases with COVID-19 | Population | Total | |
| Participants (n) | 55 | 166 | 221 |
| Gender (% Female) | 61.82 | 72.29 | 69.68 |
| Age, years (SD) | 83.44 (7.83) | 76.68 (15.35) | 78.42 (14.11) |
| APOE4 carriers (%) | 36.36 | 33.73 | 34.39 |
| Dementia (%) | 87.27 | 50.60 | 59.73 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Rojas, I.; Hernández, I.; Montrreal, L.; Quintela, I.; Calero, M.; Royo, J.L.; Huerto Vilas, R.; González-Pérez, A.; Franco-Macías, E.; Macías, J.; et al. Genomic Characterization of Host Factors Related to SARS-CoV-2 Infection in People with Dementia and Control Populations: The GR@ACE/DEGESCO Study. J. Pers. Med. 2021, 11, 1318. https://doi.org/10.3390/jpm11121318
de Rojas I, Hernández I, Montrreal L, Quintela I, Calero M, Royo JL, Huerto Vilas R, González-Pérez A, Franco-Macías E, Macías J, et al. Genomic Characterization of Host Factors Related to SARS-CoV-2 Infection in People with Dementia and Control Populations: The GR@ACE/DEGESCO Study. Journal of Personalized Medicine. 2021; 11(12):1318. https://doi.org/10.3390/jpm11121318
Chicago/Turabian Stylede Rojas, Itziar, Isabel Hernández, Laura Montrreal, Inés Quintela, Miguel Calero, Jose Luís Royo, Raquel Huerto Vilas, Antonio González-Pérez, Emilio Franco-Macías, Juan Macías, and et al. 2021. "Genomic Characterization of Host Factors Related to SARS-CoV-2 Infection in People with Dementia and Control Populations: The GR@ACE/DEGESCO Study" Journal of Personalized Medicine 11, no. 12: 1318. https://doi.org/10.3390/jpm11121318
APA Stylede Rojas, I., Hernández, I., Montrreal, L., Quintela, I., Calero, M., Royo, J. L., Huerto Vilas, R., González-Pérez, A., Franco-Macías, E., Macías, J., Menéndez-González, M., Frank-García, A., Diez-Fairen, M., Lage, C., García-Madrona, S., Aguilera, N., García-González, P., Puerta, R., Sotolongo-Grau, O., ... Ruiz, A. (2021). Genomic Characterization of Host Factors Related to SARS-CoV-2 Infection in People with Dementia and Control Populations: The GR@ACE/DEGESCO Study. Journal of Personalized Medicine, 11(12), 1318. https://doi.org/10.3390/jpm11121318









