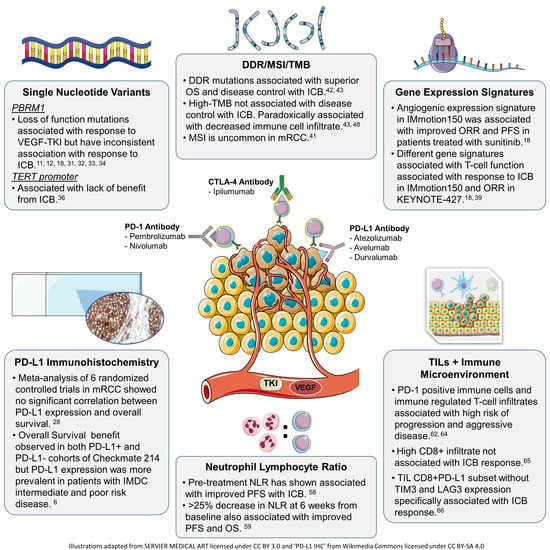
Biomarker Development for Metastatic Renal Cell Carcinoma: Omics, Antigens, T-cells, and Beyond
Abstract
Share and Cite
Miron, B.; Xu, D.; Zibelman, M. Biomarker Development for Metastatic Renal Cell Carcinoma: Omics, Antigens, T-cells, and Beyond. J. Pers. Med. 2020, 10, 225. https://doi.org/10.3390/jpm10040225
Miron B, Xu D, Zibelman M. Biomarker Development for Metastatic Renal Cell Carcinoma: Omics, Antigens, T-cells, and Beyond. Journal of Personalized Medicine. 2020; 10(4):225. https://doi.org/10.3390/jpm10040225
Chicago/Turabian StyleMiron, Benjamin, David Xu, and Matthew Zibelman. 2020. "Biomarker Development for Metastatic Renal Cell Carcinoma: Omics, Antigens, T-cells, and Beyond" Journal of Personalized Medicine 10, no. 4: 225. https://doi.org/10.3390/jpm10040225
APA StyleMiron, B., Xu, D., & Zibelman, M. (2020). Biomarker Development for Metastatic Renal Cell Carcinoma: Omics, Antigens, T-cells, and Beyond. Journal of Personalized Medicine, 10(4), 225. https://doi.org/10.3390/jpm10040225