1. Introduction
Cardiovascular diseases remains the leading cause of death worldwide, with coronary artery disease (CAD) accounting for approximately 13% of deaths in women and 14% of those in men [
1].
Coronary computed tomography angiography (CCTA) has emerged as a robust noninvasive modality for the evaluation of coronary atherosclerosis in CAD patients. Quantitative CCTA enables comprehensive plaque characterization, including the assessment of coronary lumen volume and total plaque burden, with differentiation of non-calcified (lipid-rich and fibrous–fatty), fibrous, and calcified components [
2,
3].
Coronary atherosclerosis is characterized by vascular inflammation and plaque instability, which may lead to plaque erosion or rupture. Inflammation within the vessel wall is associated with changes in pericoronary adipose tissue (PCAT) attenuation, quantified by the fat attenuation index (FAI). Increased FAI reflects edema and inflammatory cell infiltration in adjacent adipose tissue and may serve as an indirect marker of the intensity of coronary inflammation [
4]. FAI, defined as the mean X-ray attenuation of PCAT within a predefined region of interest on coronary computed tomography angiography, has been reported as an independent predictor of major adverse cardiovascular events (MACEs) [
5].
Over the past decade, advances in image post-processing have expanded the analytical capabilities of cardiovascular imaging. Technologies such as radiomics enable the extraction of considerably more information from medical images than can be obtained through visual analysis by a radiologist [
6,
7,
8]. This approach supports disease phenotyping and characterization of pathological changes through statistical descriptors derived from individual voxels and their spatial relationships, reflecting underlying tissue properties. Radiomic features capture subtle variations in voxel intensity and inter-voxel heterogeneity that may not be apparent on routine qualitative evaluation.
However, the relationship between quantitative CCTA-derived plaque metrics and detailed radiomic PCAT feature in patients with obstructive CAD (OCAD) and nonobstructive CAD (NOCAD) remains insufficiently characterized. Despite the well-known prognostic utility of PFAI and the application of PCAT radiomics to predict plaque progression, it remains unclear whether PCAT radiomic phenotypes reflect the quantitative plaque compositional burden and discriminate OCAD from NOCAD more effectively than conventional attenuation and volume variables [
5,
9,
10,
11,
12,
13]. Establishing associations between PCAT radiomic features, quantitative measurements of coronary atherosclerosis and PCAT in NOCAD and OCAD patients may improve risk stratification for cardiovascular complications and enable a more refined quantitative assessment of pericoronary inflammation associated with atherosclerosis.
The study aimed to evaluate the associations between PCAT characteristics, including radiomic features, and quantitative measures of coronary atherosclerosis in stable CAD patients.
2. Materials and Methods
2.1. Patients and Design
The study included 79 patients with stable CAD who underwent CCTA between 2019 and 2025. All examinations were performed in accordance with contemporary clinical guidelines [
14,
15]. The present study was an observational investigation.
Based on the extent of coronary atherosclerosis and the degree of luminal stenosis, the patients were classified into two groups: NOCAD (stenosis < 50%; n = 61) and OCAD (stenosis ≥ 50%; n = 18). Within the OCAD population, there were 13 (72%) patients with single-vessel disease, 3 (17%) with double-vessel disease, and 2 (11%) with triple-vessel disease.
The inclusion criteria were CAD confirmed by CCTA, high-quality CT images suitable for visual and quantitative assessment of coronary vessels, left ventricular (LV) ejection fraction ≥ 50%, a regular sinus rhythm, and written informed consent to participate in the study. The exclusion criteria were LV hypertrophy or dilation on CCTA; prior myocardial infarction or revascularization; uncontrolled or resistant arterial hypertension; decompensated diabetes mellitus; morbid obesity (body mass index (BMI) ≥ 40 kg/m2); chronic kidney disease of stage > 2; moderate or severe valvular stenosis and/or regurgitation; cardiomyopathies; ventricular extrasystoles of Lown grade ≥ 3; second- or third-degree atrioventricular block or sick sinus syndrome; acute or chronic inflammatory heart disease; severe bronchial asthma and/or chronic obstructive pulmonary disease; systemic inflammatory disease; and active malignant neoplasms.
The research protocol was approved by the Local Ethical Committee of Cardiology Research Institute, Tomsk National Research Medical Center, Russian Academy of Sciences (protocol No. 10, dated 18 February 2021), and all participants provided signed informed consent. The study complied with the Declaration of Helsinki guidelines for all human research.
A total of 237 coronary vessels—left anterior descending artery (LAD), left circumflex artery (LCX), and right coronary artery (RCA) of each patient were analyzed. Of these, 213 (89.9%) had nonobstructive lesions and 24 (10.1%) had obstructive lesions.
CCTA was used to quantify the anatomical degree of luminal stenosis (%) in the left main coronary artery (LMA) and each of the three major epicardial coronary arteries (LAD, LCX, RCA) to classify patients as having NOCAD and OCAD (>50% stenosis in ≥1 coronary artery). Quantitative measures of coronary atherosclerosis, including total plaque volume and plaque burden, pericoronary adipose tissue (PCAT) attenuation parameters (pericoronary fat attenuation index) surrounding proximal vessel segments, and radiomic features of PCAT derived from segmented proximal vessel images were evaluated.
2.2. Coronary Computed Tomography Angiography
CCTA examinations were performed using standardized protocol on multiple CT systems (Revolution EVO 64, GE Healthcare, Milwaukee, WI, USA; Discovery NM/CT 570c hybrid system, GE Healthcare, Milwaukee, WI, USA). Patient preparation, study protocol, image acquisition, reconstruction, and analysis have been described previously [
16,
17].
Contrast enhancement was achieved via intravenous administration of 80–90 mL iopromide (Ultravist 370 mg I/mL; Bayer, Leverkusen, Germany) at a flow rate of 5 mL/s. The estimated effective radiation doses ranged from 1.5 to 3.0 mSv with prospective ECG synchronization and 15–19 mSv with retrospective ECG synchronization acquisition.
2.3. Quantitative Analysis of Atherosclerosis Based on CCTA Data
CCTA images were reconstructed at 75% of the cardiac cycle using standard reconstruction kernel, with a slice thickness of 0.625 mm and an increment of 0.625 mm. In cases of rhythm-related artifacts, additional reconstructions were performed, including phase 40% of the cardiac cycle. All reconstructed images were transferred to a post-processing workstation (Advantage Workstation VolumeShare 7; GE Healthcare, Milwaukee, WI, USA). Image post-processing was performed at the Core Facility “Medical Genomics”.
Coronary atherosclerosis analysis was restricted to coronary segments free of significant artifacts. The degree of stenosis was assessed on curved and stretched multiplanar reconstructions, as well as cross-sectional images using dedicated software (CardIQ Xpress 2.0 Reveal, GE Healthcare, Milwaukee, WI, USA). Manual correction of the vessel centerline and luminal contour was performed as necessary. No significant artifacts impeding analysis were observed. Short arteries (e.g., left main artery and diagonal arteries) or those with a diameter < 1.5 mm were excluded from processing.
Atherosclerotic plaque components were defined as follows: soft tissue component, with a range of values from −30 to 30 Hounsfield units (HUs); fibrous–fatty component, with a range of values from 31 to 130 HUs; fibrous component, with a range of values from 131 to 350 HUs; calcified component, with values greater than 350 HUs [
18]. The overall quantitative parameters of coronary atherosclerosis were then determined as follows:
- •
TLV—total lumen volume, mm3;
- •
TPV—total plaque volume, mm3;
- •
TPV-ST—total plaque volume of the soft tissue component, mm3;
- •
TPV-FF—total plaque volume of the fibrous–fatty component, mm3;
- •
TPV-F—total plaque volume of the fibrous component, mm3;
- •
TPV-C—total plaque volume of the calcified component, mm3;
- •
TPV-NC—total plaque volume of the non-calcified component, mm3;
- •
TPB—total plaque burden, %;
- •
TPB-ST—total plaque burden of the soft tissue component, %;
- •
TPB-FF—total plaque burden of the fibrous–fatty component, %;
- •
TPB-F—total plaque burden of the fibrous component, %;
- •
TPB-C—total plaque burden of the calcified component, %;
- •
TPB-NC—total plaque burden of the non-calcified component, %.
The LMA and diagonal branches were considered as part of the LAD; the LCX included obtuse marginal branches, ramus intermedius, and left posterior lateral and left posterior descending arteries (in left-dominant coronary circulation); the right posterior lateral and right posterior descending arteries were included in the RCA (in right-dominant circulation). Lateral branches (diagonal, obtuse marginal, posterior descending, and posterior lateral) were analyzed if sufficiently visualized. For vessels with multiple branches, the parameters were summed. Total plaque volumes were calculated by summing volumes across coronary arteries (LAD, LCX, and RCA). Quantitative coronary atherosclerosis analysis followed the current QCCTA guidelines [
19].
2.4. Quantitative Analysis of PCAT Based on CCTA Data
The volume and attenuation of PCAT were assessed on ECG-synchronized post-contrast images reconstructed with a slice thickness of 0.625 mm using the processing workstation (Advanced Workstation, GE Healthcare, Milwaukee, WI, USA). The Auto Coronary Analysis software (v. 4.7) module was used for segmentation. Regions of interest were delineated around the major coronary arteries using the Brush tool on stretched multiplanar reconstructions. For the right coronary artery (RCA), PCAT was evaluated along a 10–50 mm proximal segment, starting 10 mm distal to the ostium, whereas for the left anterior descending (LAD) and left circumflex (LCX) arteries, a 40 mm proximal segment was analyzed [
20]. The radial extent of PCAT was defined as a distance from the outer coronary vessel wall equal to one local luminal diameter. Manual adjustments were applied when necessary to ensure accurate delineation and segmentation. Within each region of interest, the PCAT volume and the pericoronary fat attenuation index (PFAI) were quantified. The PFAI was defined as the mean CT attenuation of PCAT within the predefined region of interest. The volumetric regions of interest were exported in DICOM format for subsequent radiomic analysis.
2.5. Radiomic Analysis of PCAT
Radiomic analysis was performed using the dedicated software (3D Slicer v. 5.7.0, The Slicer Community, Boston, MA, USA). Segmented DICOM images of the PCAT surrounding the major coronary arteries—LAD, LCX, and RCA—were imported into 3D Slicer and processed with the Segment Editor module. Within each region of interest, voxels with attenuation values between −190 and −30 HUs were selected using the Threshold tool (v. 4.7).
For each coronary segment, 92 radiomic features were extracted using the PyRadiomics package (v. 842cdf 2024-11-13). These included 17 first-order features and 75 texture features: 24 gray-level co-occurrence matrix (GLCM), 14 gray-level dependence matrix (GLDM), 16 gray-level run length matrix (GLRLM), 16 gray-level size zone matrix (GLSZM), and 5 neighborhood gray-tone difference matrix (NGTDM) parameters. Prior to feature extraction, voxel intensities were discretized into 25 bins of equal width (bin width, 6.4 HUs) to reduce image noise while preserving sufficient resolution to capture biologically relevant spatial variations in PCAT attenuation.
2.6. Statistical Analysis
Statistical analysis and graphical images were performed/created using the statistical software R, version 4.5.0 (R Foundation for Statistical Computing, Vienna, Austria). Normality was assessed using the Shapiro–Wilk test. Continuous variables showing a normal distribution were expressed as mean ± standard deviation, whereas non-normally distributed variables are presented as the median with interquartile ranges. Between-group comparisons of independent samples were performed using the Mann–Whitney U test. The associations between continuous variables were evaluated using Spearman rank correlation analysis. Given the exploratory nature of these analyses and the large number of tested variables, no formal correction for multiple comparisons (e.g., Bonferroni adjustment) was applied. Post hoc power analysis demonstrated that the achieved statistical power for the primary between-group comparisons ranged from 0.8 to 0.9, indicating that the sample size was sufficient to detect clinically meaningful differences. All statistical tests were two-sided, and statistical significance was defined as p < 0.05.
3. Results
The study included 79 patients of whom 47 were men (59.5%). Sixty-one patients were classified as having NOCAD and 18 as having OCAD. To address scanner heterogeneity, the patients were well-balanced across CT systems within groups: NOCAD (n = 28 (46%) Revolution EVO 64; n = 33 (54%) Discovery NM/CT 570c hybrid system) and OCAD (n = 10 (56%) Revolution EVO 64; n = 8 (44%) Discovery NM/CT 570c hybrid system).
Clinical and demographic characteristics are presented in
Table 1. Compared with the NOCAD group, the patients with OCAD had a higher prevalence of dyslipidemia, obesity, and diabetes mellitus (all
p < 0.05).
A comparative analysis of quantitative atherosclerosis parameters and PCAT between the groups is presented in
Table 2.
In our prior study, we demonstrated good (0.75 < ICC < 0.90) to excellent (ICC > 0.90) intra- and inter-observer reproducibility for coronary atherosclerosis volume and burden parameters [
16].
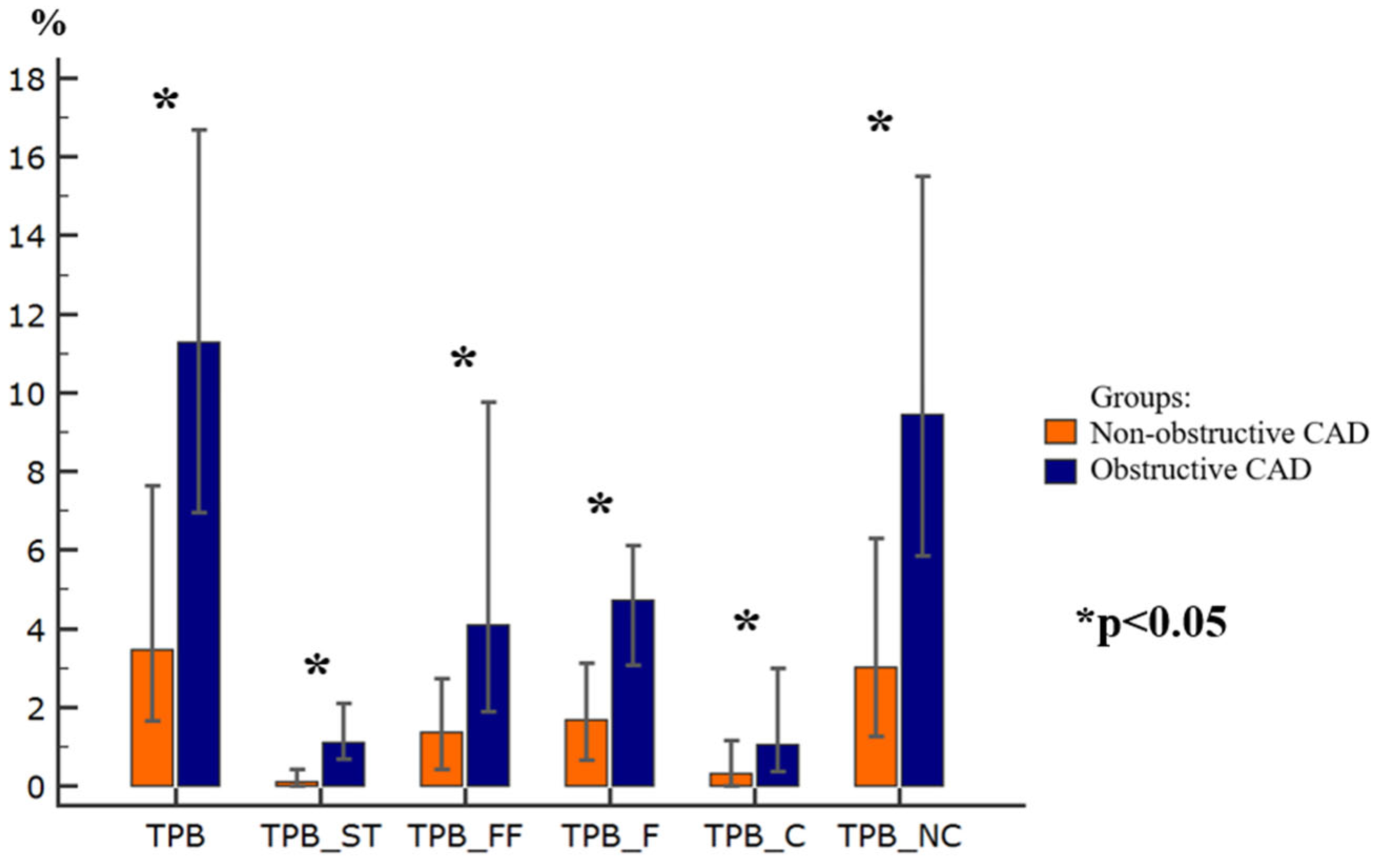
Compared to the NOCAD group, the patients with OCAD demonstrated significantly higher quantitative measures of coronary atherosclerosis (all
p < 0.05). Specifically, TPV, TPV-ST, TPV-FF, TPV-F, TPV-C, TPV-NC, TPB, TPB-ST, TPB-FF, TPB-F, TPB-C, and TPB-NC were increased in the OCAD group. In contrast, total lumen volume (TLV) did not differ significantly between the groups.
Figure 1 and
Figure 2 present a comparative analysis of these coronary atherosclerosis parameters.
No statistically significant intergroup differences were observed in the quantitative PCAT parameters (PFAI and PCAT volume) across the LAD, LCX, and RCA arteries.
No significant differences were observed between the NOCAD and OCAD groups in quantitative PCAT parameters, including the FAI and the PCAT volume, across LAD, LCX, and RCA. In contrast, several radiomic features derived from RCA PCAT differed significantly between patients with obstructive and nonobstructive lesions. These associations are illustrated in the Manhattan plot (
Figure 3).
Significant differences between the groups were identified in 46 of 92 (50%) radiomic features derived from right coronary artery (RCA) pericoronary adipose tissue (PCAT). These included features from first-order, GLCM, GLDM, GLRLM, GLSZM, and NGTDM classes (
Supplementary Table S1).
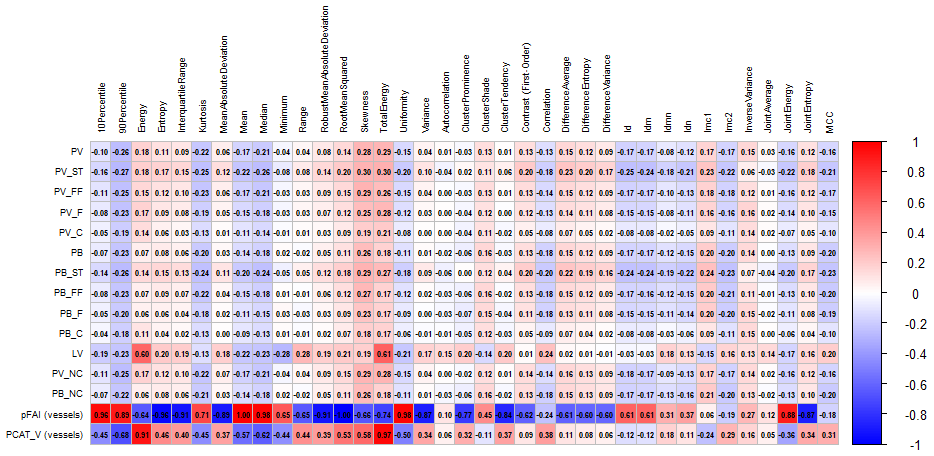
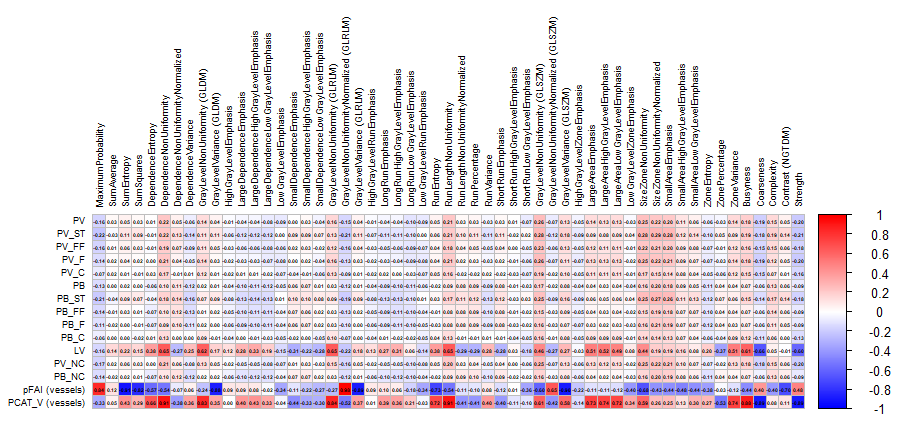
Table 3 presents Spearman’s correlation coefficients (ρ) for statistically significant associations between quantitative parameters of coronary atherosclerosis and PCAT metrics across the evaluated vessels across.
Quantitative PCAT parameters demonstrated significant weak to moderate negative correlations for the FAI and positive correlations of the PCAT volume with measures of coronary atherosclerosis. The soft tissue plaque component, expressed as both volume and burden, exhibited the strongest inverse correlations with vessel FAI. The PCAT volume correlated positively with TPV, as well as with the soft tissue, fibro-fatty, fibrous, calcified, and non-calcified plaque components and the soft tissue plaque burden. In addition, coronary lumen volume showed an inverse correlation with the FAI and a direct correlation with the PCAT volume. Significant associations were also observed between PCAT radiomic features and plaque volume and burden, as well as between PCAT density and volume. The magnitude and direction of these correlations are illustrated in heat maps (
Figure 4 and
Figure 5).
4. Discussion
According to CCTA, all quantitative measures of coronary atherosclerosis, including plaque volumes (TPV, TPV-ST, TPV-FF, TPV-F, TPV-C, and TPV-NC), as well as plaque burdens (TPB, TPB-ST, TPB-FF, TPB-F, TPB-C, and TPB-NC) were higher in the OCAD patients than in those with NOCAD. Notably, the differences between the groups were driven predominantly by the soft tissue plaque component.
4.1. Quantitative Plaque Volume and Burden: Prognostic Value in NOCAD and OCAD Patients
Contemporary investigations of quantitative coronary atherosclerosis measures focus on their diagnostic and prognostic implications, particularly in relation to adverse cardiovascular events. In the SCOT-HEART trial, Williams et al. demonstrated that the burden of low-attenuation plaque (<30 HU) was associated with an increased risk of fatal or nonfatal acute myocardial infarction (AMI) in patients with both NOCAD and OCAD [
21]. It was found that a low-density (soft tissue) plaque burden >4% was identified as an independent predictor of subsequent fatal or nonfatal AMI in NOCAD and OCAD patients. Our findings support the concept that quantitative CCTA-derived assessment of atherosclerosis may contribute to risk stratification for AMI. Volumetric plaque parameters, particularly those reflecting the low-attenuation components, may provide incremental information in patients with both NOCAD and OCAD. In the present study, soft tissue plaque volume was significantly higher in patients with obstructive atherosclerosis than in those with nonobstructive lesions (median 30.5 vs. 2.6 mm
3), suggesting a more adverse plaque phenotype in the obstructive group. These observations are consistent with prior evidence linking increased low-attenuation plaque burden to a higher risk of subsequent AMI and may indicate greater vulnerability in this population [
21].
4.2. Pericoronary Adipose Tissue Radiomic Features in NOCAD and OCAD Patients
Despite the absence of significant differences in PCAT density and volume between the groups, radiomic analysis of RCA PCAT identified significant intergroup differences in half of the extracted features. These findings suggest that PCAT radiomic features may capture aspects of tissue remodeling not reflected by conventional metrics such as the fat attenuation index (FAI) or the PCAT volume. Prior studies have demonstrated associations between PCAT radiomic signatures and vascular inflammation and adverse plaque characteristics [
4,
9,
10]. In this context, the radiomic profile may provide more information regarding the inflammatory and structural heterogeneity of PCAT in both NOCAD and OCAD compared to PCAT attenuation and volume. The groups differed by texture features (GLCM, GLDM, GLRLM, GLSZM, and NGTDM), which describe the spatial relationships between voxel characteristics, and thus consider the inflammatory phenotype of PCAT and the heterogeneity of pathophysiological processes.
Weak but statistically significant negative correlations were found between the quantitative measures of coronary atherosclerosis and the PFAI. This finding differs from prior reports, demonstrating a positive association between increasing plaque burden and higher (less negative) PCAT attenuation values [
2]. Although the coronary artery calcium score is a valuable prognostic tool, it does not reflect the presence of non-calcified plaques or the degree of vascular inflammation. In contrast, the FAI obtained from CCTA serves as a surrogate marker of coronary inflammation and has demonstrated predictive value for adverse cardiac outcomes [
22]. The discrepancy between our findings and previous data may be attributable to the predominance of the NOCAD patients in the present cohort. In this subgroup, lower-grade or more diffuse inflammatory activity may modify the relationship between plaque burden and the FAI compared with the patients with OCAD. These observations suggest that the association between coronary atherosclerosis and PCAT attenuation may vary according to disease phenotype and inflammatory intensity. Some studies have demonstrated that increased PCAT attenuation on CT robustly predicts cardiovascular risk and local inflammation in stable patients [
11,
23]; however, more recent investigations report inconsistent associations with long-term adverse events after adjustment for stenosis severity and metabolic confounders, underscoring heterogeneity in PCAT prognostic utility across cohorts [
24,
25]. The inverse correlations observed between the PFAI and plaque metrics in our study should be interpreted in light of emerging evidence that the relationship between RCAT and coronary atherosclerosis is composition-dependent rather than purely burden-dependent. Guo et al. (2025) reported that while the FAI values were higher in vessels with vulnerable plaque phenotypes, total plaque burden showed an inverse association with the FAI in multivariable analysis, suggesting a non-monotonic relationship [
26]. Similarly, Jing et al. (2024) demonstrated that the periplaque FAI was positively associated with lipid plaque fraction but negatively associated with calcified plaque fraction [
27]. Zhang et al. (2025) further showed that increases in the FAI were driven primarily by necrotic core components, whereas fibrous plaque components exerted an opposing effect [
28]. Studies on acute coronary syndrome populations have used lesion-specific FAI measurements and demonstrated strong associations with vulnerable plaque characteristics, which may not be directly comparable to patient-level or proximal-segment measurements in stable CAD cohorts [
29].
In the present study, PCAT volume was positively associated with quantitative measures of the coronary atherosclerosis burden. To our knowledge, no prior publications have specifically evaluated PCAT volume within a predefined region of interest. The observed association may reflect two complementary mechanisms. First, progressive atherosclerosis may lead to an increase in vessel diameter through positive remodeling mechanism, thereby expanding the anatomical region of interest and the measurable PCAT volume. Second, increasing plaque burden may be accompanied by a more extensive inflammatory response within PCAT, resulting in greater infiltration of the vessel wall and adjacent adipose tissue.
Significant correlations were observed between quantitative coronary atherosclerosis measures and radiomic features of PCAT. These findings support the potential role of PCAT radiomic phenotyping in improving risk stratification among stable CAD patients.
Future investigations should extend this approach to epicardial adipose tissue (EAT), given prior evidence that EAT radiomic phenotypes are associated with the risk of major adverse cardiovascular events (MACEs) [
30]. Integrating PCAT and EAT radiomic characterization may enhance the prediction of adverse outcomes in patients with CAD.
5. Limitations
This study has several limitations. This single-center observational study design, relatively small study sample, an imbalanced distribution between the NOCAD and OCAD groups may limit statistical power and generalizability, as well as increase the risk of type II error. Multiple radiomic features were analyzed without external validation, increasing the risk of overfitting. Variability related to CT acquisition parameters and segmentation methodology may also have influenced radiomic measurements. Furthermore, radiomic analysis was limited to RCA PCAT, as this segment is best described in the literature. We recognize that vessel-level or plaque-level analyses may offer more detailed insights into local inflammatory processes and should be explored in future investigations. Therefore, these results should be regarded as preliminary and hypothesis-generating. Future multicenter investigations involving more diverse patient cohorts are warranted to confirm and extend these observations.
6. Conclusions
In this study, quantitative CCTA-derived measures of coronary atherosclerosis showed significant associations with radiomic features of pericoronary adipose tissue (PCAT) in NOCAD and OCAD patients, although the correlation coefficients were weak. Conventional PCAT metrics, including density and volume, showed no significant intergroup differences and were not associated with other imaging parameters.
Radiomic features of PCAT, predominantly texture-based metrics, differed between obstructive and nonobstructive lesions and were strongly associated with the atherosclerosis burden. These findings suggest that selected PCAT radiomic features might be considered as promising imaging biomarkers of pericoronary inflammation and that they require future investigation to elucidate their potential contribution to risk stratification for cardiovascular complications in NOCAD and OCAD patients.
Author Contributions
K.V.Z., O.V.M., and A.N.M. designed the research study. K.V.Z., A.V.K., A.N.M., K.V.K., O.V.M., A.S.D., A.V.M., and E.V.G. performed the research. K.V.Z., A.V.K., and A.N.M. analyzed the data. K.V.Z., A.V.K., A.N.M., and K.V.K. wrote the manuscript. All authors contributed to editorial changes in the manuscript. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The research protocol was approved by the Local Ethical Committee of Cardiology Research Institute, Tomsk National Research Medical Center, Russian Academy of Sciences (protocol No. 10, dated 18 February 2021) . The study was conducted in accordance with the Declaration of Helsinki for all human research.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy considerations.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| AMI | acute myocardial infarction |
| CAD | coronary artery disease |
| CCTA | coronary computed tomography angiography |
| CT | computed tomography |
| GFR | glomerular filtration rate |
| GLCM | gray-level co-occurrence matrix |
| GLDM | gray-level dependence matrix |
| GLRLM | gray-level run length matrix |
| GLSZM | gray-level size zone matrix |
| HU | Hounsfield unit |
| LAD | left anterior descending artery |
| LCX | left circumflex artery |
| MACE | major adverse cardiovascular event |
| NGTDM | neighborhood gray-tone difference matrix |
| NOCAD | nonobstructive coronary artery disease |
| OCAD | obstructive coronary artery disease |
| PCAT | pericoronary adipose tissue |
| PFAI | perivascular fat attenuation index |
| RCA | right coronary artery |
References
- Timmis, A.; Aboyans, V.; Vardas, P.; Townsend, N.; Torbica, A.; Kavousi, M.; Boriani, G.; Huculeci, R.; Kazakiewicz, D.; Scherr, D.; et al. European Society of Cardiology: The 2023 Atlas of Cardiovascular Disease Statistics. Eur. Heart J. 2024, 45, 4019–4062. [Google Scholar] [CrossRef]
- Cundari, G.; Marchitelli, L.; Pambianchi, G.; Catapano, F.; Conia, L.; Stancanelli, G.; Catalano, C.; Galea, N. Imaging biomarkers in cardiac CT: Moving beyond simple coronary anatomical assessment. Radiol. Med. 2024, 129, 380–400. [Google Scholar] [CrossRef]
- Williams, M.C.; Earls, J.P.; Hecht, H. Quantitative assessment of atherosclerotic plaque, recent progress and current limitations. J. Cardiovasc. Comput. Tomogr. 2022, 16, 124–137. [Google Scholar] [CrossRef]
- Feng, Y.; Xu, Z.; Zhang, L.; Zhang, Y.; Xu, H.; Zhuang, X.; Zhang, H.; Xie, X. Machine-learning-derived radiomics signature of pericoronary tissue in coronary CT angiography associates with functional ischemia. Front. Physiol. 2022, 13, 980996. [Google Scholar] [CrossRef] [PubMed]
- Oikonomou, E.K.; Williams, M.C.; Kotanidis, C.P.; Desai, M.Y.; Marwan, M.; Antonopoulos, A.S.; Thomas, K.E.; Thomas, S.; Akoumianakis, I.; Fan, L.M.; et al. A novel machine learning-derived radiotranscriptomic signature of perivascular fat improves cardiac risk prediction using coronary CT angiography. Eur. Heart J. 2019, 40, 3529–3543. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Pan, T.; Yin, X.; Xu, H.; Gao, X.; Tao, X.; Zhou, L.; Xie, G.; Kong, X.; Huang, X.; et al. CT texture analysis of vulnerable plaques on optical coherence tomography. Eur. J. Radiol. 2021, 136, 109551. [Google Scholar] [CrossRef]
- Gillies, R.J.; Kinahan, P.E.; Hricak, H. Radiomics: Images Are More than Pictures, They Are Data. Radiology 2016, 278, 563–577. [Google Scholar] [CrossRef]
- Kolossváry, M.; De Cecco, C.N.; Feuchtner, G.; Maurovich-Horvat, P. Advanced atherosclerosis imaging by CT: Radiomics, machine learning and deep learning. J. Cardiovasc. Comput. Tomogr. 2019, 13, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Hao, G.; Xu, J.; Liu, Y.; Yu, Y.; Hu, S.; Hu, C. Radiomics analysis of lesion-specific pericoronary adipose tissue to predict major adverse cardiovascular events in coronary artery disease. BMC Med. Imaging 2024, 24, 150. [Google Scholar] [CrossRef]
- Lin, A.; Kolossváry, M.; Yuvaraj, J.; Cadet, S.; McElhinney, P.A.; Jiang, C.; Nerlekar, N.; Nicholls, S.J.; Slomka, P.J.; Maurovich-Horvat, P.; et al. Myocardial Infarction Associates With a Distinct Pericoronary Adipose Tissue Radiomic Phenotype: A Prospective Case-Control Study. JACC Cardiovasc. Imaging 2020, 13, 2371–2383. [Google Scholar] [CrossRef]
- Oikonomou, E.K.; Marwan, M.; Desai, M.Y.; Mancio, J.; Alashi, A.; Hutt Centeno, E.; Thomas, S.; Herdman, L.; Kotanidis, C.P.; Thomas, K.E.; et al. Non-invasive detection of coronary inflammation using computed tomography and prediction of residual cardiovascular risk (the CRISP CT study): A post-hoc analysis of prospective outcome data. Lancet 2018, 392, 929–939. [Google Scholar] [CrossRef]
- Li, Y.; Huo, H.; Liu, H.; Zheng, Y.; Tian, Z.; Jiang, X.; Jin, S.; Hou, Y.; Yang, Q.; Teng, F.; et al. Coronary CTA-based radiomic signature of pericoronary adipose tissue predict rapid plaque progression. Insights Imaging 2024, 15, 151. [Google Scholar] [CrossRef]
- Chen, R.; Li, X.; Jia, H.; Feng, C.; Dong, S.; Liu, W.; Lin, S.; Zhu, X.; Xu, Y.; Zhu, Y. Radiomics Analysis of Pericoronary Adipose Tissue From Baseline Coronary Computed Tomography Angiography Enables Prediction of Coronary Plaque Progression. J. Thorac. Imaging 2024, 39, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Barbarash, O.L.; Karpov, Y.A.; Panov, A.V.; Akchurin, R.S.; Alekyan, B.G.; Alekhin, M.N.; Aronov, D.M.; Harutyunyan, G.K.; Belenkov, Y.N.; Boytsov, S.A.; et al. 2024 Clinical practice guidelines for Stable coronary artery disease. Russ. J. Cardiol. 2024, 29, 6110. (In Russian) [Google Scholar] [CrossRef]
- Vrints, C.; Andreotti, F.; Koskinas, K.C.; Rossello, X.; Adamo, M.; Ainslie, J.; Banning, A.P.; Budaj, A.; Buechel, R.R.; Chiariello, G.A.; et al. 2024 ESC Guidelines for the management of chronic coronary syndromes. Eur. Heart J. 2024, 45, 3415–3537. [Google Scholar] [CrossRef]
- Maltseva, A.N.; Dorzhieva, R.V.; Kopeva, K.V.; Dasheeva, A.S.; Mochula, A.V.; Grakova, E.V.; Zavadovsky, K.V. Coronary Computed Tomography Angiography Atherosclerotic Plaque Volume as A Predictor of Myocardial Blood Flow Impairment in Nonobstructive Coronary Artery Disease. Rev. Cardiovasc. Med. 2025, 26, 39291. [Google Scholar] [CrossRef]
- Dasheeva, A.; Vorobeva, D.; Kopeva, K.; Maltseva, A.; Mochula, A.; Vorozhtsova, I.; Grakova, E.; Zavadovsky, K. Quantitative Coronary CT Angiography and Pericoronary Adipose Tissue in Acute Myocardial Infarction: Relationship with Dynamic Myocardial Perfusion SPECT. Diagnostics 2025, 15, 2840. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.J.; Blankstein, R.; Bax, J.J.; Ferencik, M.; Bittencourt, M.S.; Min, J.K.; Berman, D.S.; Leipsic, J.; Villines, T.C.; Dey, D.; et al. Society of Cardiovascular Computed Tomography / North American Society of Cardiovascular Imaging—Expert Consensus Document on Coronary CT Imaging of Atherosclerotic Plaque. J. Cardiovasc. Comput. Tomogr. 2021, 15, 93–109. [Google Scholar] [CrossRef]
- Nieman, K.; García-García, H.M.; Hideo-Kajita, A.; Collet, C.; Dey, D.; Pugliese, F.; Weissman, G.; Tijssen, J.G.P.; Leipsic, J.; Opolski, M.P.; et al. Standards for quantitative assessments by coronary computed tomography angiography (CCTA): An expert consensus document of the society of cardiovascular computed tomography (SCCT). J. Cardiovasc. Comput. Tomogr. 2024, 18, 429–443. [Google Scholar] [CrossRef]
- Sagris, M.; Antonopoulos, A.S.; Simantiris, S.; Oikonomou, E.; Siasos, G.; Tsioufis, K.; Tousoulis, D. Pericoronary fat attenuation index-a new imaging biomarker and its diagnostic and prognostic utility: A systematic review and meta-analysis. Eur. Heart J. Cardiovasc. Imaging 2022, 23, e526–e536. [Google Scholar] [CrossRef]
- Williams, M.C.; Kwiecinski, J.; Doris, M.; McElhinney, P.; D’Souza, M.S.; Cadet, S.; Adamson, P.D.; Moss, A.J.; Alam, S.; Hunter, A.; et al. Low-Attenuation Noncalcified Plaque on Coronary Computed Tomography Angiography Predicts Myocardial Infarction: Results From the Multicenter SCOT-HEART Trial (Scottish Computed Tomography of the HEART). Circulation 2020, 141, 1452–1462. [Google Scholar] [CrossRef]
- Savo, M.T.; De Amicis, M.; Cozac, D.A.; Cordoni, G.; Corradin, S.; Cozza, E.; Amato, F.; Lassandro, E.; Da Pozzo, S.; Tansella, D.; et al. Comparative Prognostic Value of Coronary Calcium Score and Perivascular Fat Attenuation Index in Coronary Artery Disease. J. Clin. Med. 2024, 13, 5205. [Google Scholar] [CrossRef]
- Antonopoulos, A.S.; Sanna, F.; Sabharwal, N.; Thomas, S.; Oikonomou, E.K.; Herdman, L.; Margaritis, M.; Shirodaria, C.; Kampoli, A.M.; Akoumianakis, I.; et al. Detecting human coronary inflammation by imaging perivascular fat. Sci. Transl. Med. 2017, 9, eaal2658. [Google Scholar] [CrossRef]
- van Rosendael, S.E.; Kamperidis, V.; Maaniitty, T.; de Graaf, M.A.; Saraste, A.; McKay-Goodall, G.E.; Jukema, J.W.; Knuuti, J.; Bax, J.J. Pericoronary adipose tissue for predicting long-term outcomes. Eur. Heart J. Cardiovasc. Imaging 2024, 25, 1351–1359. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, D.; Shou, B.L.; Matheson, M.B.; Ostovaneh, M.R.; Rochitte, C.; Chen, M.Y.; Dewey, M.; Ortman, J.; Cox, C.; Lima, J.A.C.; et al. Perivascular fat attenuation for predicting adverse cardiac events in stable patients undergoing invasive coronary angiography. J. Cardiovasc. Comput. Tomogr. 2022, 16, 483–490. [Google Scholar] [CrossRef]
- Guo, T.; Wang, X.P.; Xia, R.; Gu, Z.; Dou, X.F. Correlation Between Fat Attenuation Index and Plaque Parameters in Coronary CT Angiography: An Observational Study in Stable Coronary Artery Disease. Clin. Appl. Thromb. Hemost. 2025, 31, 10760296241313459. [Google Scholar] [CrossRef]
- Jing, M.; Xi, H.; Wang, Y.; Zhu, H.; Sun, Q.; Zhang, Y.; Ren, W.; Xu, Z.; Deng, L.; Zhang, B.; et al. Association between Pericoronary Fat Attenuation Index Values and Plaque Composition Volume Fraction Measured by Coronary Computed Tomography Angiography. Acad. Radiol. 2024, 31, 3579–3589. [Google Scholar] [CrossRef]
- Zhang, R.; Zhao, W.; Tang, Z.; Xu, Y.; Xie, H.; Wei, C.; Liu, D.; Dong, W.; Liu, J.; Xu, L.; et al. Relationship between pericoronary fat attenuation index and quantitative plaque components in newly identified coronary plaques. Eur. J. Radiol. 2025, 190, 112206. [Google Scholar] [CrossRef]
- Sun, J.T.; Sheng, X.C.; Feng, Q.; Yin, Y.; Li, Z.; Ding, S.; Pu, J. Pericoronary Fat Attenuation Index Is Associated with Vulnerable Plaque Components and Local Immune-Inflammatory Activation in Patients with Non-ST Elevation Acute Coronary Syndrome. J. Am. Heart Assoc. 2022, 11, e022879. [Google Scholar] [CrossRef] [PubMed]
- Popov, E.V.; Ilyushenkova, Y.N.; Repin, A.N.; Zavadovsky, K.V.; Sazonova, S.I. The potential role of radiochemical analysis of CT images of epicardial adipose tissue in the prognosis of acute myocardial infarction. Sib. J. Clin. Exp. Med. 2023, 38, 143–152. [Google Scholar] [CrossRef]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |