Duodenal Lymphomas: Comprehensive Evaluation of Endoscopic Features and Clinical Outcomes in a Tertiary Center
Abstract
1. Introduction
- Comprehensive multimodal dataset: We present one of the largest single-center cohorts (N = 50) integrating clinical, endoscopic, and pathologic findings over two decades.
- Novel endoscopic prognosticator: We identify “mucosal granularity” as the first reported significant independent predictor of favorable overall survival in DL, providing a practical visual metric for immediate risk stratification during endoscopy.
- Subtype-specific survival analysis: We provide detailed survival data for aggressive T-cell vs. B-cell DLs, highlighting the unique challenges of managing MEITL in Asian patients.
- The remainder of this manuscript is structured as follows: Section 2 delineates the patient selection criteria, rigorous endoscopic/pathological evaluation protocols (including inter-observer agreement validation), and the statistical framework for survival analysis with Bootstrap resampling. Section 3 details integrated clinicopathological, endoscopic, and survival findings, with a focus on subtype-specific and Asian population-specific patterns. Section 4 provides an analytical comparison of our results with global literature, critically discusses study limitations, and outlines concrete future research directions. Finally, Section 5 summarizes the clinical implications and methodological contributions of this work for DL diagnosis and management.
2. Materials and Methods
2.1. Patients, Study Design, and Ethical Approval
2.2. Endoscopic and Histopathological Examinations
2.3. Outcome
2.4. Statistical Analysis
3. Results
3.1. Clinicopathological Characteristics and Clinical Presentation
3.2. Endoscopic Findings in Patients with Duodenal Lymphoma
3.3. Associations Between Clinical, Pathological, and Endoscopic Features and Overall Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CE | Chromoendoscopy |
| DLBCL | Diffuse Large B-Cell Lymphoma |
| DLs | Duodenal Lymphomas |
| EATLs | Enteropathy-Associated T-Cell Lymphomas |
| GDPL | Gemcitabine, Dexamethasone, Cisplatin, and L-Asparaginase |
| GI | Gastrointestinal |
| IQRs | Interquartile Ranges |
| MALT | Mucosa-Associated Lymphoid Tissue |
| MCL | Mantle-Cell Lymphoma |
| MEITL | Monomorphic Epitheliotropic Intestinal T-Cell Lymphoma |
| ME-NBI | Magnifying Endoscopy with Narrow-Band Imaging |
| NHL | Non-Hodgkin’s Lymphoma |
| OS | Overall Survival |
| R-CHOP | Rituximab, Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone |
| SEER | Surveillance, Epidemiology, and End Results |
| WHO | The World Health Organization |
| WLE | White-Light Endoscopy |
References
- Nakamura, S.; Matsumoto, T. Gastrointestinal lymphoma: Recent advances in diagnosis and treatment. Digestion 2013, 87, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Andrews, C.N.; Gill, M.J.; Urbanski, S.J.; Stewart, D.; Perini, R.; Beck, P. Changing epidemiology and risk factors for gastrointestinal non-Hodgkin’s lymphoma in a North American population: Population-based study. Am. J. Gastroenterol. 2008, 103, 1762–1769. [Google Scholar] [CrossRef] [PubMed]
- Howell, J.; Auer-Grzesiak, I.; Zhang, J.; Andrews, C.; Stewart, D.; Urbanski, S. Increasing incidence rates, distribution and histological characteristics of primary gastrointestinal non-Hodgkin lymphoma in a North American population. Can. J. Gastroenterol. Hepatol. 2012, 26, 452–456. [Google Scholar] [CrossRef]
- Ding, W.; Zhao, S.; Wang, J.; Yang, Q.; Sun, H.; Yan, J.; Gao, L.; Yao, W.; Zhang, W.; Liu, W. Gastrointestinal Lymphoma in Southwest China: Subtype Distribution of 1,010 Cases Using the WHO (2008) Classification in a Single Institution. Acta Haematol. 2016, 135, 21–28. [Google Scholar] [CrossRef]
- Nakamura, S.; Matsumoto, T.; Iida, M.; Yao, T.; Tsuneyoshi, M. Primary gastrointestinal lymphoma in Japan: A clinicopathologic analysis of 455 patients with special reference to its time trends. Cancer 2003, 97, 2462–2473. [Google Scholar] [CrossRef]
- Cho, S.J.; Ryu, K.W.; Kim, C.G.; Lee, J.Y.; Kook, M.C.; Min, H.S.; Choi, I.J. Duodenal mucosa-associated lymphoid tissue lymphoma masquerading as an ulcer scar. Endoscopy 2008, 40, E175. [Google Scholar] [CrossRef][Green Version]
- Iwamuro, M.; Kondo, E.; Otsuka, F.; Takata, K.; Yoshino, T.; Kawahara, Y.; Okada, H. Detection of Minute Duodenal Follicular Lymphoma Lesions Using Magnifying Endoscopy. Acta Medica Okayama 2016, 70, 139–144. [Google Scholar]
- Tari, A.; Sato, Y.; Asaoku, H.; Kunihiro, M.; Fukumoto, A.; Tanaka, S.; Fujihara, M.; Yoshino, T. A duodenal follicular lymphoma associated with the lesion mimicking MALT lymphoma in terminal ileum and Bauhin valve. Med. Mol. Morphol. 2010, 43, 174–177. [Google Scholar] [CrossRef]
- Trivedi, P.; Gupta, A.; Pasricha, S. Primary diffuse large B-cell lymphoma of ampulla of vater: A rare case report. J. Gastrointest. Cancer 2012, 43, 340–343. [Google Scholar] [CrossRef]
- Zheng, Q.F.; Li, J.Y.; Qin, L.; Wei, H.M.; Cai, L.Y.; Nong, B. Gastrointestinal involvement by mantle cell lymphoma identified by biopsy performed during endoscopy: A case report. Medicine 2018, 97, e9799. [Google Scholar] [CrossRef]
- Zheng, G.; Wang, Y.; Zhao, Y.; Zheng, Z. Clinicopathological Features, Treatment Strategy, and Prognosis of Primary Non-Hodgkin’s Lymphoma of the Duodenum: A SEER Database Analysis. Can. J. Gastroenterol. Hepatol. 2020, 2020, 9327868. [Google Scholar] [CrossRef]
- Karanam, P.K.; Al-Hamadani, M.; Go, R.S. Enteropathy-associated T-cell lymphoma in the US: Higher incidence and poorer survival among Asians. Br. J. Haematol. 2016, 172, 990–992. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.K.; Chan, A.C.; Cheuk, W.; Wan, S.K.; Lee, W.K.; Lui, Y.H.; Chan, W.K. Type II enteropathy-associated T-cell lymphoma: A distinct aggressive lymphoma with frequent gammadelta T-cell receptor expression. Am. J. Surg. Pathol. 2011, 35, 1557–1569. [Google Scholar] [CrossRef] [PubMed]
- Iwamuro, M.; Tanaka, T.; Okada, H. Review of lymphoma in the duodenum: An update of diagnosis and management. World J. Gastroenterol. 2023, 29, 1852–1862. [Google Scholar] [CrossRef]
- Müller, A.M.; Ihorst, G.; Mertelsmann, R.; Engelhardt, M. Epidemiology of non-Hodgkin’s lymphoma (NHL): Trends, geographic distribution, and etiology. Ann. Hematol. 2005, 84, 1–12. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. Int. J. Surg. 2014, 12, 1495–1499. [Google Scholar] [CrossRef]
- Alaggio, R.; Amador, C.; Anagnostopoulos, I.; Attygalle, A.D.; Araujo, I.B.d.O.; Berti, E.; Bhagat, G.; Borges, A.M.; Boyer, D.; Calaminici, M.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia 2022, 36, 1720–1748. [Google Scholar] [CrossRef]
- Bendel, R.B.; Afifi, A.A. Comparison of stopping rules in forward “stepwise” regression. J. Am. Stat. Assoc. 1977, 72, 46–53. [Google Scholar]
- Mickey, R.M.; Greenland, S. The impact of confounder selection criteria on effect estimation. Am. J. Epidemiol. 1989, 129, 125–137. [Google Scholar] [CrossRef]
- Lagoo, A.S.; Cardona, D.M.; Layne, A. Lymphomas of the gastro-intestinal tract—Pathophysiology, pathology, and differential diagnosis. Indian J. Pathol. Microbiol. 2012, 55, 1–16. [Google Scholar] [CrossRef]
- Fujishima, F.; Katsushima, H.; Fukuhara, N.; Konosu-Fukaya, S.; Nakamura, Y.; Sasano, H.; Ichinohasama, R. Incidence Rate, Subtype Frequency, and Occurrence Site of Malignant Lymphoma in the Gastrointestinal Tract: Population-Based Analysis in Miyagi, Japan. Tohoku J. Exp. Med. 2018, 245, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Ondrejka, S.; Jagadeesh, D. Enteropathy-Associated T-Cell Lymphoma. Curr. Hematol. Malig. Rep. 2016, 11, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Tse, E.; Gill, H.; Loong, F.; Kim, S.J.; Ng, S.; Tang, T.; Ko, Y.; Chng, W.; Lim, S.; Kim, W.S.; et al. Type II enteropathy-associated T-cell lymphoma: A multicenter analysis from the Asia Lymphoma Study Group. Am. J. Hematol. 2012, 87, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Ferreri, A.J.; Zinzani, P.L.; Govi, S.; Pileri, S.A. Enteropathy-associated T-cell lymphoma. Crit. Rev. Oncol. Hematol. 2011, 79, 84–90. [Google Scholar] [CrossRef]
- Veloza, L.; Cavalieri, D.; Missiaglia, E.; Ledoux-Pilon, A.; Bisig, B.; Pereira, B.; Bonnet, C.; Poullot, E.; Quintanilla-Martinez, L.; Dubois, R.; et al. Monomorphic epitheliotropic intestinal T-cell lymphoma comprises morphologic and genomic heterogeneity impacting outcome. Haematologica 2023, 108, 181–195. [Google Scholar] [CrossRef]
- Small, S.; Barnea Slonim, L.; Williams, C.; Karmali, R. B Cell Lymphomas of the GI Tract. Curr. Gastroenterol. Rep. 2021, 23, 9. [Google Scholar] [CrossRef]
- Takata, K.; Miyata-Takata, T.; Sato, Y.; Iwamuro, M.; Okada, H.; Tari, A.; Yoshino, T. Gastrointestinal follicular lymphoma: Current knowledge and future challenges. Pathol. Int. 2018, 68, 1–6. [Google Scholar] [CrossRef]
- Thomas, A.S.; Schwartz, M.; Quigley, E. Gastrointestinal lymphoma: The new mimic. BMJ Open Gastroenterol. 2019, 6, e000320. [Google Scholar] [CrossRef]
- Iwamuro, M.; Okada, H.; Takata, K.; Nose, S.; Miyatani, K.; Yoshino, T.; Yamamoto, K. Diagnostic accuracy of endoscopic biopsies for the diagnosis of gastrointestinal follicular lymphoma: A clinicopathologic study of 48 patients. Ann. Diagn. Pathol. 2014, 18, 99–103. [Google Scholar] [CrossRef]
- Vetro, C.; Romano, A.; Amico, I.; Conticello, C.; Motta, G.; Figuera, A.; Chiarenza, A.; Di Raimondo, C.; Giulietti, G.; Bonanno, G.; et al. Endoscopic features of gastro-intestinal lymphomas: From diagnosis to follow-up. World J. Gastroenterol. 2014, 20, 12993–13005. [Google Scholar] [CrossRef]
- Na, H.K.; Won, S.H.; Ahn, J.Y.; Kim, G.H.; Jung, K.W.; Lee, J.H.; Kim, D.H.; Choi, K.D.; Song, H.J.; Lee, G.H.; et al. Clinical course of duodenal mucosa-associated lymphoid tissue lymphoma: Comparison with gastric mucosa-associated lymphoid tissue lymphoma. J. Gastroenterol. Hepatol. 2021, 36, 406–412. [Google Scholar] [CrossRef] [PubMed]
- Tran, Q.T.; Nguyen Duy, T.; Nguyen-Tran, B.S.; Nguyen-Thanh, T.; Ngo, Q.T.; Tran Thi, N.P.; Le, V.; Dang-Cong, T. Endoscopic and Histopathological Characteristics of Gastrointestinal Lymphoma: A Multicentric Study. Diagnostics 2023, 13, 2767. [Google Scholar] [CrossRef]
- Ghimire, P.; Wu, G.Y.; Zhu, L. Primary gastrointestinal lymphoma. World J. Gastroenterol. 2011, 17, 697–707. [Google Scholar] [CrossRef] [PubMed]
- Miike, T.; Kawakami, H.; Kameda, T.; Yamamoto, S.; Tahara, Y.; Hidaka, T.; Kubuki, Y.; Yorita, K.; Akiyama, Y.; Arimura, Y.; et al. Clinical characteristics of adult T-cell leukemia/lymphoma infiltration in the gastrointestinal tract. BMC Gastroenterol. 2020, 20, 298. [Google Scholar] [CrossRef] [PubMed]

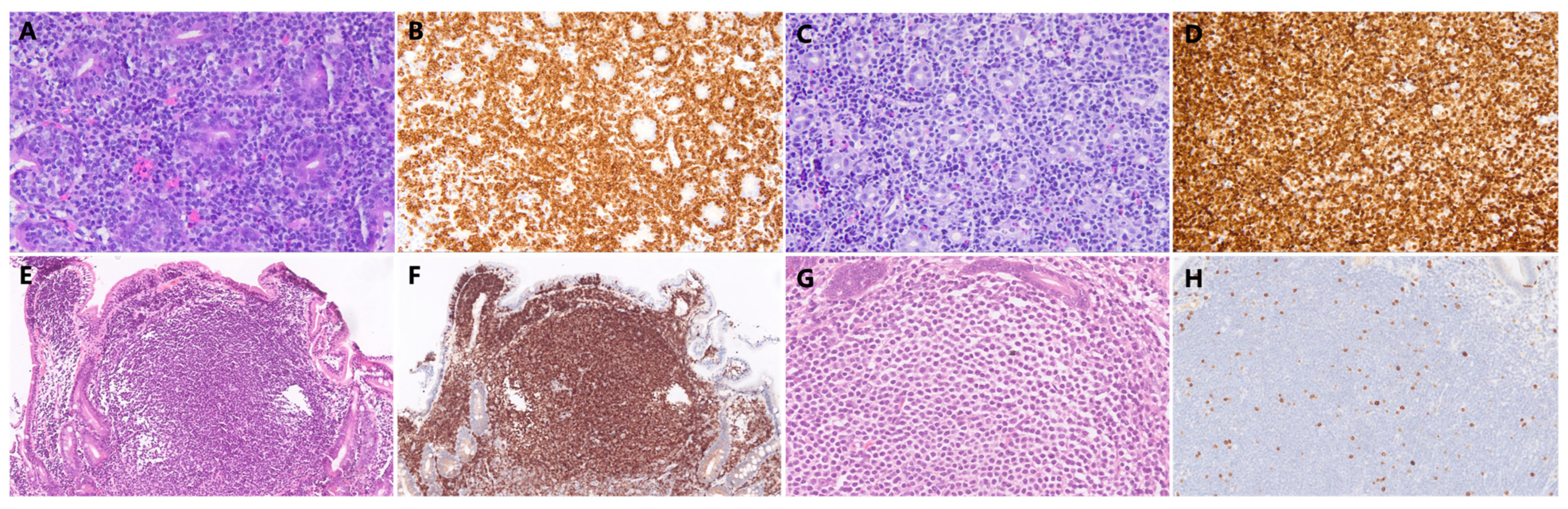

| Characteristic | N = 50 |
|---|---|
| Sex | |
| Female | 22 (44%) |
| Male | 28 (56%) |
| Age, years (IQR) | 56 (46, 70) |
| Lymphoma type | |
| B-cell, indolent | 16 (32%) |
| B-cell, aggressive | 20 (40%) |
| T-cell | 14 (28%) |
| Histopathological subtype | |
| DLBCL | 18 (36%) |
| Mantle-cell lymphoma | 2 (4.0%) |
| MALT | 8 (16%) |
| Follicular lymphoma | 8 (16%) |
| Peripheral T-cell lymphoma | 1 (2.0%) |
| T-cell lymphoblastic lymphoma | 1 (2.0%) |
| Enteropathy-type T-cell lymphoma | 4 (8.0%) |
| T-cell unclassified | 8 (16%) |
| Ann Arbor stage simplified | |
| II | 10 (20%) |
| III | 5 (10%) |
| IV | 35 (70%) |
| Clinical presentation | |
| Helicobacter pylori (H. pylori) status | |
| H. pylori-negative | 35 (70%) |
| H. pylori-positive | 15 (30%) |
| Fever | 11 (22%) |
| Abdominal distension | 12 (24%) |
| Abdominal pain | 25 (50%) |
| Diarrhea | 15 (30%) |
| Difficulty eating | 1 (2.0%) |
| Abdominal mass | 1 (2.0%) |
| Poor appetite | 9 (18%) |
| Vomiting | 12 (24%) |
| Rectal bleeding | |
| No hematochezia | 41 (82%) |
| Melena | 6 (12%) |
| Hematochezia | 3 (6.0%) |
| Difficulty defecating | 3 (6.0%) |
| >5 kg weight loss | 33 (66%) |
| Chemotherapy | 31 (62%) |
| Surgery | 3 (6.0%) |
| Survival | |
| Died | 32 (64%) |
| Alive | 18 (36%) |
| Follow-up, months (IQR) | 20 (3, 38) |
| Survival, months (IQR) | 24.1 (13, 117) |
| Characteristic | N = 50 |
|---|---|
| Lesion location | |
| Duodenal bulb | 9 (18%) |
| Descending duodenum | 17 (34%) |
| Descending duodenum and bulb | 19 (38%) |
| Horizontal duodenum | 3 (6.0%) |
| Descending and horizontal duodenum | 2 (4.0%) |
| Other sites of gastrointestinal involvement | |
| Esophageal involvement | 1 (2.0%) |
| Stomach involvement | 11 (22%) |
| Duodenal involvement alone | 36 (72%) |
| Jejunum/ileum involvement | 2 (4.0%) |
| End of ileum involvement | 5 (10%) |
| Granularity | 24 (48%) |
| Villous shortening | 11 (22%) |
| Shallow folds | 12 (24%) |
| Infiltrative change | 13 (26%) |
| Polyp-like protrusion | 5 (10%) |
| Mass-forming | 20 (40%) |
| Lymphangiectasia | 16 (32%) |
| Mucosal erosion | 12 (24%) |
| Ulceration | |
| No ulceration | 29 (58%) |
| Superficial ulceration | 16 (32%) |
| Deep ulceration | 5 (10%) |
| Ulcer number | |
| No ulceration | 29 (58%) |
| Single ulceration | 7 (14%) |
| Multiple ulcerations | 14 (28%) |
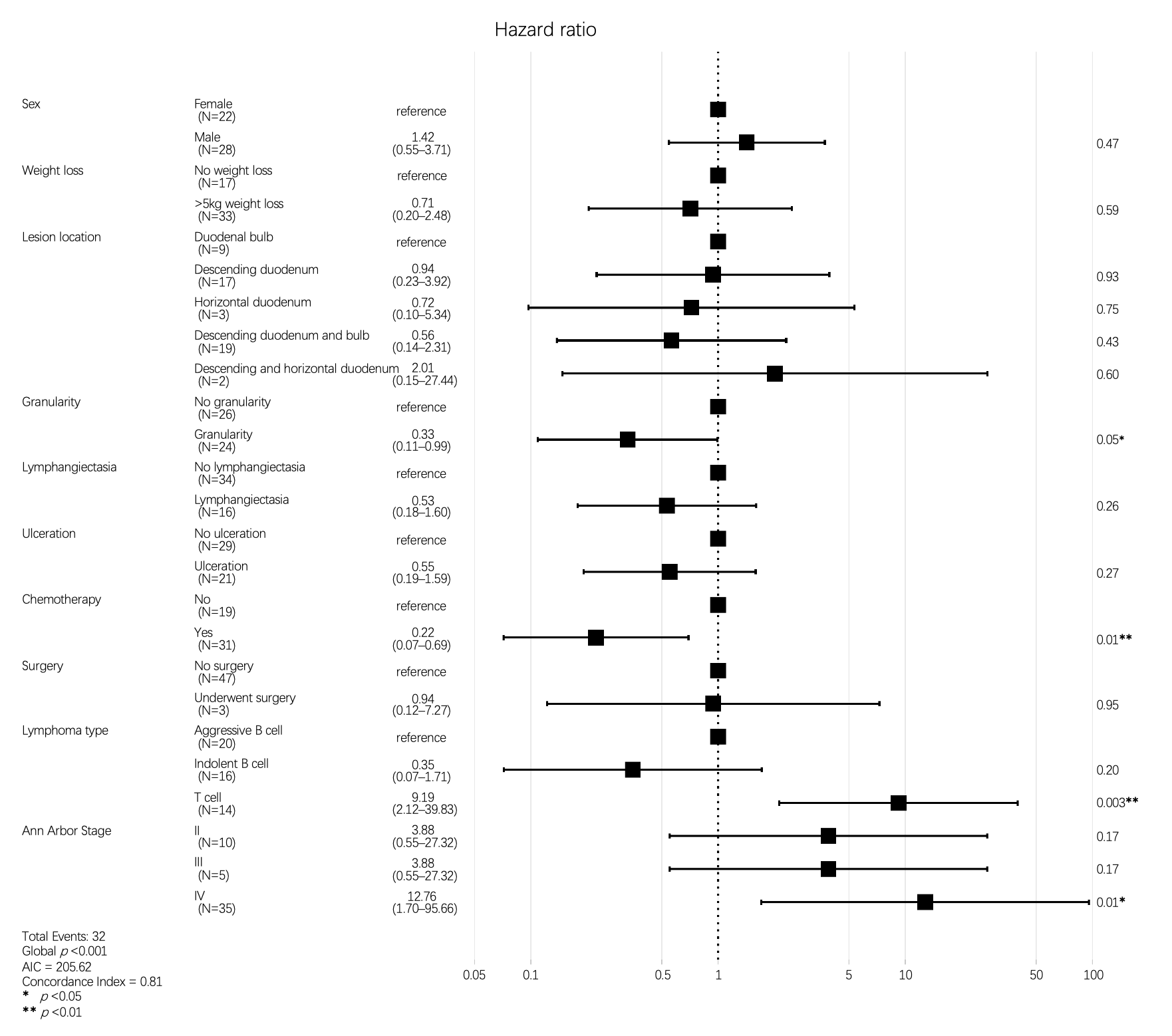
| Variable | HR (Univariable) | HR (Multivariable) | |
|---|---|---|---|
| Sex | Female | - | - |
| Male | 1.81 (0.87–3.77, p = 0.111) | 1.42 (0.55–3.71, p = 0.470) | |
| Weight loss | No weight loss | - | - |
| >5 kg weight loss | 2.02 (0.93–4.41, p = 0.076) | 0.71 (0.20–2.48, p = 0.591) | |
| Lesion location | Duodenal bulb | - | - |
| Descending duodenum | 0.54 (0.20–1.47, p = 0.229) | 0.94 (0.22–3.92, p = 0.930) | |
| Horizontal duodenum | 0.89 (0.18–4.33, p = 0.887) | 0.72 (0.10–5.34, p = 0.748) | |
| Descending duodenum and bulb | 0.75 (0.30–1.89, p = 0.543) | 0.56 (0.14–2.31, p = 0.425) | |
| Descending and horizontal duodenum | 0.45 (0.06–3.67, p = 0.456) | 2.01 (0.15–27.44, p = 0.599) | |
| Granularity | No granularity | - | - |
| Granularity | 0.45 (0.22–0.92, p = 0.029) | 0.33 (0.11–0.99, p = 0.049) | |
| Lymphangiectasia | No lymphangiectasia | - | - |
| Lymphangiectasia | 0.61 (0.28–1.37, p = 0.235) | 0.53 (0.18–1.60, p = 0.261) | |
| Ulceration present/absent | No ulceration | - | - |
| Ulceration | 1.59 (0.79–3.22, p = 0.197) | 0.55 (0.19–1.59, p = 0.269) | |
| Chemotherapy | No | - | - |
| Yes | 0.68 (0.33–1.38, p = 0.282) | 0.22 (0.07–0.69, p = 0.010) | |
| Surgery | No surgery | - | - |
| Underwent surgery | 2.75 (0.81–9.36, p = 0.105) | 0.94 (0.12–7.27, p = 0.954) | |
| Lymphoma type | Aggressive B-cell | - | - |
| Indolent B-cell | 0.23 (0.07–0.68, p = 0.008) | 0.35 (0.07–1.71, p = 0.195) | |
| T-cell | 2.27 (1.05–4.90, p = 0.037) | 9.19 (2.12–39.83, p = 0.003) | |
| Ann Arbor stage simplified | II | - | - |
| III | 2.16 (0.44–10.71, p = 0.346) | 3.88 (0.55–27.32, p = 0.173) | |
| IV | 3.49 (1.05–11.57, p = 0.041) | 12.76 (1.70–95.66, p = 0.013) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yan, Z.; Zhang, Y.; Jia, C.; Zhang, Y.; Zhang, S.; Yang, A. Duodenal Lymphomas: Comprehensive Evaluation of Endoscopic Features and Clinical Outcomes in a Tertiary Center. Diagnostics 2026, 16, 1173. https://doi.org/10.3390/diagnostics16081173
Yan Z, Zhang Y, Jia C, Zhang Y, Zhang S, Yang A. Duodenal Lymphomas: Comprehensive Evaluation of Endoscopic Features and Clinical Outcomes in a Tertiary Center. Diagnostics. 2026; 16(8):1173. https://doi.org/10.3390/diagnostics16081173
Chicago/Turabian StyleYan, Zhiyu, Yuheng Zhang, Congwei Jia, Yan Zhang, Shengyu Zhang, and Aiming Yang. 2026. "Duodenal Lymphomas: Comprehensive Evaluation of Endoscopic Features and Clinical Outcomes in a Tertiary Center" Diagnostics 16, no. 8: 1173. https://doi.org/10.3390/diagnostics16081173
APA StyleYan, Z., Zhang, Y., Jia, C., Zhang, Y., Zhang, S., & Yang, A. (2026). Duodenal Lymphomas: Comprehensive Evaluation of Endoscopic Features and Clinical Outcomes in a Tertiary Center. Diagnostics, 16(8), 1173. https://doi.org/10.3390/diagnostics16081173







