Diagnostic Consistency and Morphological Limits of Extraovarian Lesions in Ovarian Serous Tumors: A Comparative Study Between Gynecological and General Pathologists
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Cohort Characteristics
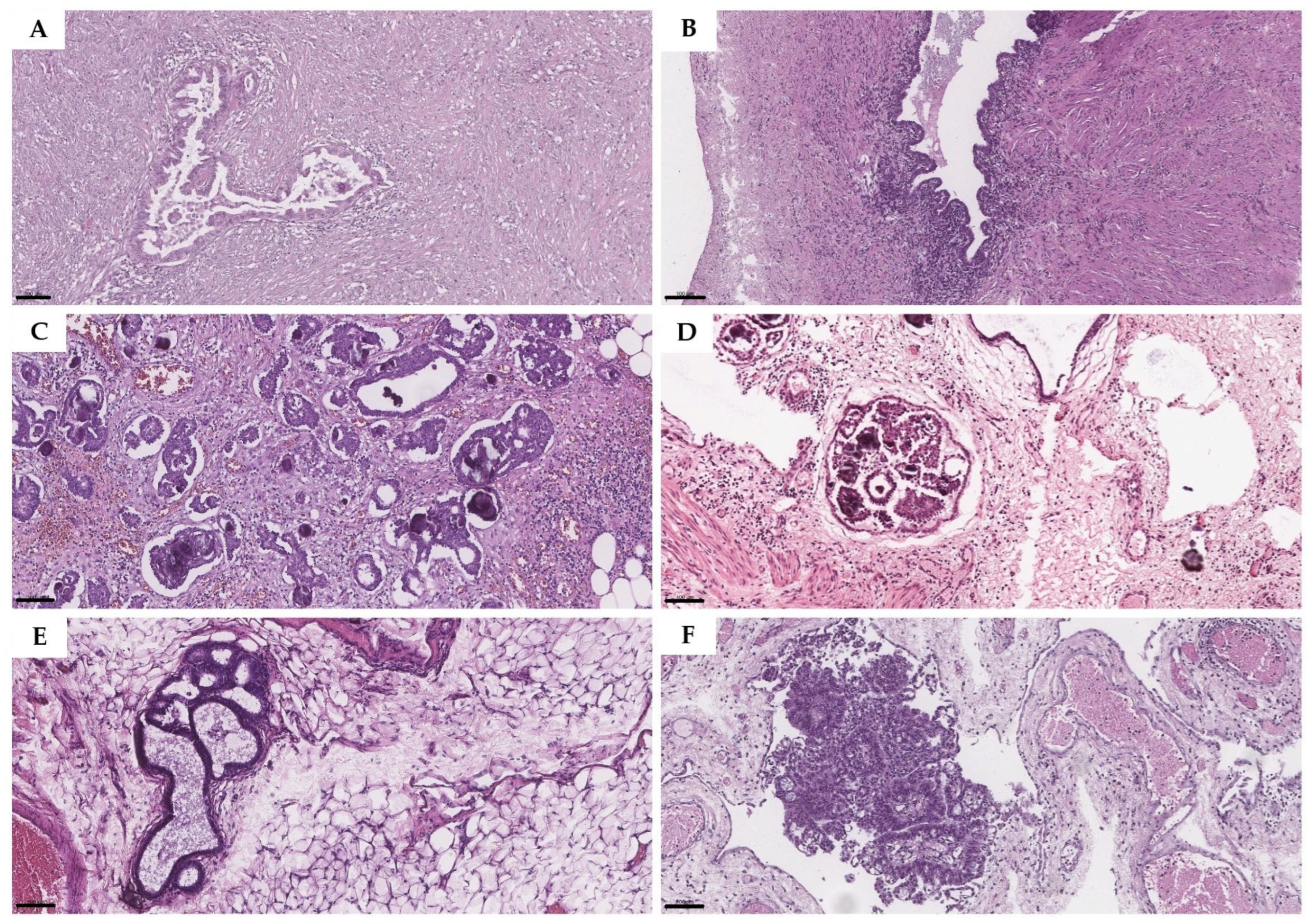
2.2. Histopathological Assessment of Implants
2.3. Statistical Methods
3. Results
3.1. Interobserver Reproducibility of Extraovarian Lesions
3.2. Clinicopathological Characteristics of the Cohort
3.3. Histopathological Assessment of Implants
3.4. Correlation of Tumor Type with Clinical Follow-Up
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SBT | Serous Borderline Tumor |
| LGSC | Low-Grade Serous Carcinoma |
| H&E | Hematoxylin and Eosin |
| WSI | Whole-Slide Image |
| Me | Median |
| Q1 | First Quartile (25th percentile) |
| Q2 | Third Quartile (75th percentile) |
| FIGO | International Federation of Gynecology and Obstetrics |
| N/A | Not Applicable |
| DFS | Disease-Free Survival |
| CI | Confidence Interval |
| Path | Pathologist |
| CD | Consensus Diagnosis |
| NCCN | National Comprehensive Cancer Network |
| HGSC | High-Grade Serous Carcinoma |
| AI | Artificial Intelligence |
Appendix A
| N Case | Path 1 | Path 2 | Path 3 | Path 4 | Path 5 | Path 6 | CD |
| 1 | 1 | 1 | 1 | 1 | 1 | 2 | 1 |
| 2 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 3 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 4 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 5 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 6 | 2 | 2 | 2 | 2 | 1 | 1 | 2 |
| 7 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 8 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 9 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 10 | 1 | 2 | 2 | 2 | 2 | 1 | 2 |
| 11 | 1 | 1 | 1 | 2 | 1 | 2 | 1 |
| 12 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 13 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 14 | 1 | 1 | 2 | 2 | 1 | 2 | 1 |
| 15 | 1 | 1 | 1 | 1 | 2 | 2 | 1 |
| 16 | 2 | 2 | 2 | 2 | 1 | 2 | 2 |
| 17 | 1 | 2 | 1 | 2 | 2 | 1 | 1 |
| 18 | 1 | 1 | 1 | 1 | 1 | 2 | 1 |
| 19 | 1 | 1 | 2 | 1 | 1 | 2 | 1 |
| 20 | 2 | 2 | 2 | 1 | 2 | 2 | 2 |
| 21 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 22 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 23 | 1 | 1 | 1 | 2 | 1 | 2 | 1 |
| 24 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 25 | 2 | 2 | 2 | 2 | 1 | 2 | 2 |
| 26 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 27 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 28 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 29 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 30 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 31 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| 32 | 2 | 1 | 2 | 2 | 2 | 2 | 2 |
| 33 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| Pathologist (Path); consensus diagnosis (CD); Path 1–3 are gynecological pathologists and Path 4–6 are general pathologists; 1—implant of SBT; 2—metastasis of LGSC. | |||||||
References
- Vang, R.; Davidson, B.; Kong, C.S.; Longacre, T.A.; Malpica, A. WHO Classification of Female Genital Tumors; International Agency for Research on Cancer: Lyon, France, 2021; pp. 43–44. [Google Scholar]
- McKenney, J.K.; Gilks, C.B.; Kalloger, S.; Longacre, T.A. Classification of Extraovarian Implants in Patients with Ovarian Serous Borderline Tumors (Tumors of Low Malignant Potential) Based on Clinical Outcome. Am. J. Surg. Pathol. 2016, 40, 1155–1164. [Google Scholar] [CrossRef]
- Bell, K.A.; Smith Sehdev, A.E.; Kurman, R.J. Refined diagnostic criteria for implants associated with ovarian atypical proliferative serous tumors (borderline) and micropapillary serous carcinomas. Am. J. Surg. Pathol. 2001, 25, 419–432. [Google Scholar] [CrossRef]
- Katzenstein, A.A.; Mazur, M.T.; Morgan, T.E.; Kao, M.S. Proliferative serous tumors of the ovary: Histologic features and prognosis. Am. J. Surg. Pathol. 1978, 2, 339–355. [Google Scholar] [CrossRef]
- Russell, P. The pathological assessment of ovarian neoplasms. I: Introduction to the common “epithelial” tumors and analysis of benign “epithelial” tumors. Pathology 1979, 1, 15–26. [Google Scholar] [CrossRef]
- Sim, J.; Wright, C.C. The kappa statistic in reliability studies: Use, interpretation, and sample size requirements. Phys. Ther. 2005, 85, 257–268. [Google Scholar] [CrossRef]
- NCCN. Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Ovarian Cancer Including Fallopian Tube Cancer and Primary Peritoneal Cancer. Version 1.2025; National Comprehensive Cancer Network: Plymouth Meeting, PA, USA, 2025; Available online: https://www.nccn.org/guidelines/guidelines-detail (accessed on 10 June 2025).
- Mhawech-Fauceglia, P.; Izevbaye, I.; Spindler, T.; Wang, G.; Hwang, H.; Samrao, D.; Elishaev, E.; Maxwell, G.L.; Lawrenson, K.; Darcy, K.M. Genomic heterogeneity in peritoneal implants: A differential analysis of gene expression using nanostring Human Cancer Reference panel identifies a malignant signature. Gynecol. Oncol. 2020, 156, 6–12. [Google Scholar] [CrossRef]
- Varghese, A.; Shih, I.M. Not All Peritoneal Implants Are Created Equal. Gynecol. Oncol. 2020, 156, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, M.; Meylaerts, L.; Govaerts, K. Extrapelvic Endometriosis Mimicking Peritoneal Surface Malignancy: Case Report and a Review of Literature. Indian J. Surg. Oncol. 2023, 14, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.S.; Chin, C.C.; Lin, P.Y. “Pseudocarcinomatosis peritoneii”: Extensive intraperitoneal endometriosis mimic carcinomatosis in morphology—A case report and a survey of the literature. Int. J. Color. Dis. 2007, 22, 89–91. [Google Scholar] [CrossRef] [PubMed]
- Elmohr, M.M.; Elsayes, K.M.; Pickhardt, P.J. Non-neoplastic conditions mimicking peritoneal carcinomatosis at CT imaging. Br. J. Radiol. 2020, 93, 20200401. [Google Scholar] [CrossRef]
- Singh, N.; Murali, S.; Zangmo, R. Florid cystic endosalpingiosis, masquerading as malignancy in a young patient: A brief review. BMJ Case Rep. 2014, 2014, bcr2013201645. [Google Scholar] [CrossRef]
- Zapardiel, I.; Tobias-Gonzalez, P.; de Santiago, J. Endosalpingiosis mimicking recurrent ovarian carcinoma. Taiwan. J. Obstet. Gynecol. 2012, 51, 660–662. [Google Scholar] [CrossRef][Green Version]
- Burla, L.; Kalaitzopoulos, D.R.; Mrozek, A.; Eberhard, M.; Samartzis, N. Intraoperative Appearance of Endosalpingiosis: A Single-Center Experience of Laparoscopic Findings and Systematic Review of Literature. J. Clin. Med. 2022, 11, 7006. [Google Scholar] [CrossRef]
- Cox, H.Y.; Alhatem, A.; Barlog, L.; Heller, D. A Rare Mimic of Malignancy: Papillary Endosalpingiosis. Int. J. Surg. Pathol. 2020, 28, 60–62. [Google Scholar] [CrossRef]
- Huang, L.; Li, Y.; Du, J.; Li, H.; Lu, M.; Wang, Y.; Zhou, W.; Wang, W.; Wu, H. The Prognostic Value of Retraction Clefts in Chinese Invasive Breast Cancer Patients. Pathol. Oncol. Res. 2021, 27, 1609743. [Google Scholar] [CrossRef]
- Lee, E.S.; Leong, A.S.; Kim, Y.S.; Lee, J.H.; Kim, I.; Ahn, G.H.; Kim, H.S.; Chun, Y.K. Calretinin, CD34, and alpha-smooth muscle actin in the identification of peritoneal invasive implants of serous borderline tumors of the ovary. Mod. Pathol. 2006, 19, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Grisham, R.N.; Slomovitz, B.M.; Andrews, N.; Banerjee, S.; Brown, J.; Carey, M.S.; Chui, H.; Coleman, R.L.; Fader, A.N.; Gaillard, S.; et al. Low-grade serous ovarian cancer: Expert consensus report on the state of the science. Int. J. Gynecol. Cancer 2023, 33, 1331–1344. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, L.; Kenny, H.A.; Krishnan, R.; Kelliher, L.; Bilecz, A.J.; Heide, J.; Donle, L.; Shimizu, A.; Metousis, A.; Mendoza, R.; et al. Spatial proteo-transcriptomic profiling reveals the molecular landscape of borderline ovarian tumors and their invasive progression. Cancer Cell 2025, 43, 1495–1511.e7. [Google Scholar] [CrossRef]
- Mayr, D.; Hirschmann, A.; Löhrs, U.; Diebold, J. KRAS and BRAF mutations in ovarian tumors: A comprehensive study of invasive carcinomas, borderline tumors and extraovarian implants. Gynecol. Oncol. 2006, 103, 883–887. [Google Scholar] [CrossRef]
- Sciallis, A.; Zhang, G. Epithelial tumors of the ovary: A review focusing on carcinoma and recent advances. Hum. Pathol. Rep. 2024, 37, 300749. [Google Scholar] [CrossRef]
- Zuo, T.; Wong, S.; Buza, N.; Hui, P. KRAS mutation of extraovarian implants of serous borderline tumor: Prognostic indicator for adverse clinical outcome. Mod. Pathol. 2018, 31, 350–357. [Google Scholar] [CrossRef]
- Tsang, Y.T.; Deavers, M.T.; Sun, C.C.; Kwan, S.Y.; Kuo, E.; Malpica, A.; Mok, S.C.; Gershenson, D.M.; Wong, K.K. KRAS (but not BRAF) mutations in ovarian serous borderline tumour are associated with recurrent low-grade serous carcinoma. J. Pathol. 2013, 231, 449–456. [Google Scholar] [CrossRef]
- McHenry, A.; Rottmann, D.A.; Buza, N.; Hui, P. KRAS mutation in primary ovarian serous borderline tumors correlates with tumor recurrence. Virchows Arch. 2023, 483, 71–79. [Google Scholar] [CrossRef]
- Li, N.; Ming, X.; Li, Z. Unilateral cystectomy and serous histology are associated with relapse in borderline ovarian tumor patients with fertility-sparing surgery: A systematic review and meta-analysis. Arch. Gynecol. Obstet. 2020, 302, 1063–1074. [Google Scholar] [CrossRef]
- Ko, M.E.; Lin, Y.H.; Huang, K.J.; Chang, W.C.; Sheu, B.C. Fertility and Pregnancy Outcomes after Fertility-Sparing Surgery for Early-Stage Borderline Ovarian Tumors and Epithelial Ovarian Cancer: A Single-Center Study. Cancers 2023, 15, 5327. [Google Scholar] [CrossRef] [PubMed]
- Renz, M.; Friedlander, M.; Berek, J.S. Cancer of the ovary, fallopian tube, and peritoneum: 2025 update. Int. J. Gynecol. Obstet. 2025, 171, 6–35. [Google Scholar] [CrossRef] [PubMed]
- Arcieri, M.; Tius, V.; Filippin, S.; Aletti, G.; Lorusso, D.; Fagotti, A.; Sehouli, J.; Zapardiel, I.; De Iaco, P.; Scollo, P.; et al. Management of Patients with Epithelial Ovarian Cancer: A Systematic Comparison of International Guidelines from Scientific Societies (AIOM-BGCS-ESGO-ESMO-JGSO-NCCN-NICE). Cancers 2025, 17, 3915. [Google Scholar] [CrossRef] [PubMed]
- Morice, P.; Scambia, G.; Abu-Rustum, N.R.; Acien, M.; Arena, A.; Brucker, S.; Cheong, Y.; Collinet, P.; Fanfani, F.; Filippi, F.; et al. Fertility-sparing treatment and follow-up in patients with cervical cancer, ovarian cancer, and borderline ovarian tumours: Guidelines from ESGO, ESHRE, and ESGE. Lancet Oncol. 2024, 25, e602–e610. [Google Scholar] [CrossRef]


| Path 1 | Path 2 | Path 3 | Path 4 | Path 5 | Path 6 | |
|---|---|---|---|---|---|---|
| Path 1 | 0.812 | 0.817 | 0.639 | 0.619 | 0.530 | |
| Path 2 | 0.756 | 0.698 | 0.687 | 0.348 | ||
| Path 3 | 0.697 | 0.573 | 0.578 | |||
| Path 4 | 0.518 | 0.513 | ||||
| Path 5 | 0.413 | |||||
| CD | 0.821 | 0.745 | 0.781 | 0.698 | 0.687 | 0.467 |
| Micropapillary | Clefts | Destructive Pattern | % of Agreement |
|---|---|---|---|
| 1 | 0 | 0 | 66.7 |
| 1 | 0 | 1 | 100 |
| 0 | 1 | 0 | 100 |
| 0 | 0 | 1 | 66.7 |
| 0 | 1 | 0 | 83.3 |
| 0 | 0 | 1 | 83.3 |
| 0 | 1 | 0 | 100 |
| 1 | 0 | 1 | 83.3 |
| 1 | 0 | 1 | 100 |
| 0 | 0 | 1 | 100 |
| 1 | 1 | 0 | 100 |
| 1 | 1 | 0 | 100 |
| 0 | 1 | 0 | 83.3 |
| 0 | 0 | 1 | 100 |
| Characteristics | SBT (n = 13) | LGSC (n = 11) | p-Value |
|---|---|---|---|
| Median (Q1; Q3) | |||
| Age (yr) | 31 (27; 38) | 33 (29; 33) | 0.57 |
| Follow-up (mn) | 36 (23; 65.5) | 27 (12; 64.2) | 0.16 |
| FIGO stage: | n (%) | ||
| II B | 2 (15) | 1 (9) | 0.58 |
| III A | 8 (62) | 5 (46) | |
| III B | 3 (23) | 3 (27) | |
| III C | 0 | 1 (9) | |
| IV | 0 | 1 (9) | |
| Pattern of invasion: | n (%) | * | |
| micropapillary architecture | N/A | 1 (7) | N/A |
| retraction artifact | N/A | 4 (28.5) | |
| destructive pattern | N/A | 4 (28.5) | |
| mixed | N/A | 5 (36) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Badlaeva, A.; Tregubova, A.; Arzhanukhina, N.; Chamorovskaya, A.; Borzunov, V.; Sheshko, P.; Litvinova, V.; Ezhova, L.; Asaturova, A. Diagnostic Consistency and Morphological Limits of Extraovarian Lesions in Ovarian Serous Tumors: A Comparative Study Between Gynecological and General Pathologists. Diagnostics 2026, 16, 1136. https://doi.org/10.3390/diagnostics16081136
Badlaeva A, Tregubova A, Arzhanukhina N, Chamorovskaya A, Borzunov V, Sheshko P, Litvinova V, Ezhova L, Asaturova A. Diagnostic Consistency and Morphological Limits of Extraovarian Lesions in Ovarian Serous Tumors: A Comparative Study Between Gynecological and General Pathologists. Diagnostics. 2026; 16(8):1136. https://doi.org/10.3390/diagnostics16081136
Chicago/Turabian StyleBadlaeva, Alina, Anna Tregubova, Natalia Arzhanukhina, Alevtina Chamorovskaya, Vladimir Borzunov, Polina Sheshko, Valentina Litvinova, Larisa Ezhova, and Aleksandra Asaturova. 2026. "Diagnostic Consistency and Morphological Limits of Extraovarian Lesions in Ovarian Serous Tumors: A Comparative Study Between Gynecological and General Pathologists" Diagnostics 16, no. 8: 1136. https://doi.org/10.3390/diagnostics16081136
APA StyleBadlaeva, A., Tregubova, A., Arzhanukhina, N., Chamorovskaya, A., Borzunov, V., Sheshko, P., Litvinova, V., Ezhova, L., & Asaturova, A. (2026). Diagnostic Consistency and Morphological Limits of Extraovarian Lesions in Ovarian Serous Tumors: A Comparative Study Between Gynecological and General Pathologists. Diagnostics, 16(8), 1136. https://doi.org/10.3390/diagnostics16081136







