Pathophysiological, Translational, and Diagnostic Aspects of ME/CFS: A Focus on Skeletal Muscle Involvement
Abstract
1. Prologue
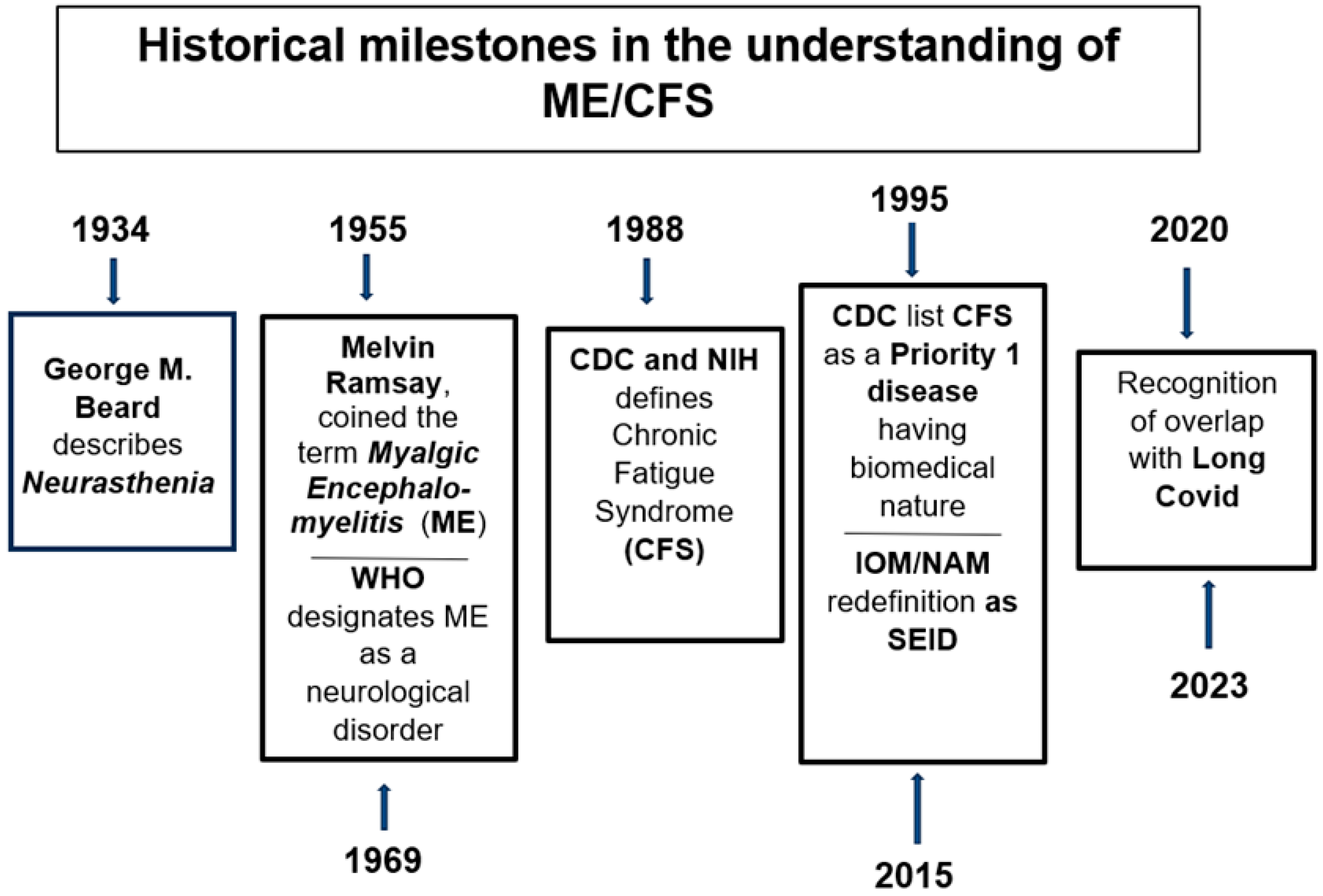
2. The Definition and Management of Myalgic Encephalomyelitis
- Persistent or relapsing fatigue lasting at least six months, not alleviated by rest, resulting in substantial reduction in activity;
- The presence of at least four out of eight associated symptoms, e.g., impaired memory or concentration, sore throat, tender lymph nodes, muscle pain, multi-joint pain, unrefreshing sleep, and post-exertional malaise (PEM).
- Fukuda Criteria (1994 CDC).
- 2.
- Canadian Consensus Criteria (CCC 2003)
- 3.
- IOM Criteria (IOM/SEID, 2015)
- Core Management Strategies (Center Blue Box): Represents the foundational, non-pharmacological interventions required for all patients. Pacing and maintaining the Energy Envelope are the primary defenses against PEM. Sleep hygiene optimization targets the restorative sleep deficit characteristic of the pathology.
- Fatigue & Mitochondrial Support (Left Green Column): Focuses on cellular energy production and neuroinflammation.
- Pain & Inflammation (Center Yellow Column): Outlines a stepped approach to systemic and localized pain.
- Autonomic & Cognitive Support (Right Orange Column): Addresses Orthostatic Intolerance (OI) and POTS (Postural Orthostatic Tachycardia Syndrome), which are highly comorbid with ME/CFS.
3. Part One—Specific Muscle Damage
4. Part Two—Can the Symptomatology of ME/CFS Patients Originate from Muscle?
4.1. The Muscle as a Secretory Organ
4.2. Extracellular Vesicles and miRNAs in Muscle Communication
4.3. Muscle Secretome Dysregulation and Systemic Manifestations
4.4. Immune Dysregulation and the Role of Inflammation
4.5. Muscle as Both Target and Source of Symptoms
5. Part Three—Final Considerations and Future Perspectives
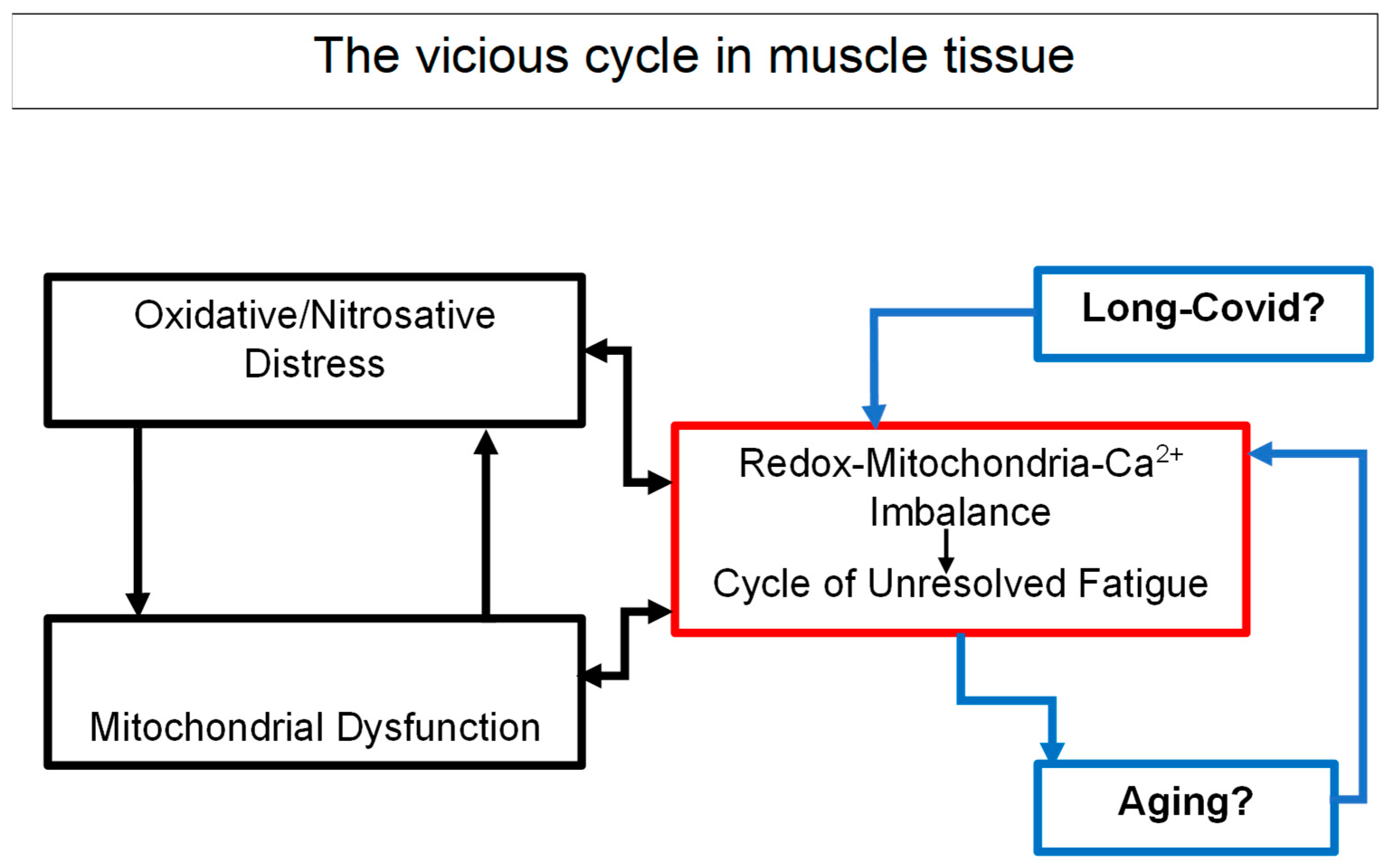
5.1. Oxidative Stress and Mitochondrial Dysfunction as Central Hubs
5.2. A Vicious Cycle of Energetic Collapse
- Mitochondrial inefficiency increases ROS production.
- ROS in turn impair calcium handling and ion transport.
- This leads to further mitochondrial damage and energetic decline.
5.3. Long COVID as a Model for Post-Viral ME/CFS
5.4. Toward a Muscle-Centered Diagnostic and Therapeutic Model
- Blood-based measures of redox imbalance;
- Alterations in specific muscle-related microRNAs;
- EV-associated protein signatures reflecting mitochondrial distress.
5.5. Future Research Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fulle, S.; Mecocci, P.; Fanó, G.; Vecchiet, I.; Vecchini, A.; Racciotti, D.; Cherubini, A.; Pizzigallo, E.; Vecchiet, L.; Senin, U.; et al. Specific Oxidative Alterations in Vastus Lateralis Muscle of Patients with the Diagnosis of Chronic Fatigue Syndrome. Free Radic. Biol. Med. 2000, 29, 1252–1259. [Google Scholar] [CrossRef] [PubMed]
- Fulle, S.; Pietrangelo, T.; Mancinelli, R.; Saggini, R.; Fanò, G. Specific Correlations between Muscle Oxidative Stress and Chronic Fatigue Syndrome: A Working Hypothesis. J. Muscle Res. Cell Motil. 2007, 28, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Fulle, S.; Belia, S.; Di Tano, G. Sarcopenia Is More than a Muscular Deficit. Arch. Ital. Biol. 2005, 143, 229–234. [Google Scholar] [PubMed]
- Graves, B.S.; Patel, M.; Newgent, H.; Parvathy, G.; Nasri, A.; Moxam, J.; Gill, G.S.; Sawhney, V.; Gupta, M. Chronic Fatigue Syndrome: Diagnosis, Treatment, and Future Direction. Cureus 2024, 16, e70616. [Google Scholar] [CrossRef]
- Beard, G. Neurasthenia, or Nervous Exhaustion. Boston Med. Surg. J. 1869, 80, 217–221. [Google Scholar] [CrossRef]
- Gilliam, A.G. Epidemiological study of an epidemic, Los Angeles County Hospital, 1934. Public Health Rep. 1938, 53, 1683–1689. [Google Scholar]
- Ramsay, A.M. Myalgic Encephalomyelitis and Postviral Fatigue States: The Saga of Royal Free Disease, 2nd ed.; Produced by Gower Medical for the Myalgic Encephalomyelitis Association: London, UK, 1988; ISBN 978-0-906923-99-3. [Google Scholar]
- World Health Organization (WHO). International Classification of Diseases; 8th Revision; WHO: Geneva, Switzerland, 1969. [Google Scholar]
- Holmes, G.P.; Kaplan, J.E.; Gantz, N.M.; Komaroff, A.L.; Schonberger, L.B.; Straus, S.E.; Jones, J.F.; Dubois, R.E.; Cunningham-Rundles, C.; Pahwa, S.; et al. Chronic Fatigue Syndrome: A Working Case Definition. Ann. Intern. Med. 1988, 108, 387–389. [Google Scholar] [CrossRef]
- Straus, S. The Incline Village Outbreak; Mythic Pictures: Incline Village, NV, USA, 2014. [Google Scholar]
- Institute of Medicine Committee on the Diagnostic Criteria for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Beyond Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Redefining an Illness; National Academies Press: Washington, DC, USA, 2015; ISBN 978-0-309-31689-7. [Google Scholar]
- Fukuda, K.; Straus, S.E.; Hickie, I.; Sharpe, M.C.; Dobbins, J.G.; Komaroff, A. International Chronic Fatigue Syndrome Study Group. The Chronic Fatigue Syndrome: A Comprehensive Approach to Its Definition and Study. Ann. Intern. Med. 1994, 121, 953–959. [Google Scholar] [CrossRef]
- Bateman, L.; Bested, A.C.; Bonilla, H.F.; Chheda, B.V.; Chu, L.; Curtin, J.M.; Dempsey, T.T.; Dimmock, M.E.; Dowell, T.G.; Felsenstein, D.; et al. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Essentials of Diagnosis and Management. Mayo Clin. Proc. 2021, 96, 2861–2878. [Google Scholar] [CrossRef]
- Komaroff, A.L.; Bateman, L. Will COVID-19 Lead to Myalgic Encephalomyelitis/Chronic Fatigue Syndrome? Front. Med. 2021, 7, 606824. [Google Scholar] [CrossRef]
- Proal, A.D.; VanElzakker, M.B. Long COVID or Post-Acute Sequelae of COVID-19 (PASC): An Overview of Biological Factors That May Contribute to Persistent Symptoms. Front. Microbiol. 2021, 12, 698169. [Google Scholar] [CrossRef]
- Dudova, D.; Bozhkova, M.; Petrov, S.; Nikolova, R.; Kalfova, T.; Ivanovska, M.; Vaseva, K.; Nikolova, M.; Ivanov, I.N. Insights into the Complex Biological Network Underlying Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Int. J. Mol. Sci. 2026, 27, 268. [Google Scholar] [CrossRef]
- Myalgic Encephalomyelitis (or Encephalopathy)/Chronic Fatigue Syndrome: Diag-nosis and Management; National Institute for Health and Care Excellence (NICE): London, UK, 2021; Guideline NG206 N.
- Jason, L.A.; Sunnquist, M. The Development of the DePaul Symptom Ques-tionnaire: Original, Expanded, Brief, and Pediatric Versions. Front. Pediatr. 2018, 6, 330. [Google Scholar] [CrossRef]
- Sommerfelt, K.; Schei, T.; Seton, K.A.; Carding, S.R. Assessing functional capac-ity in myalgic encephalopathy/chronic fatigue syndrome: A Patient-Informed Ques-tionnaire. J. Clin. Med. 2024, 13, 3486. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Vøllestad, N.K.; Mengshoel, A.M. Post-exertional malaise in daily life and experimental exercise models in patients with myalgic encephalomyelitis/chronic fatigue syndrome. Front. Physiol. 2023, 14, 1257557. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sanal-Hayes, N.E.M.; Mclaughlin, M.; Hayes, L.D.; Mair, J.L.; Ormerod, J.; Carless, D.; Hilliard, N.; Meach, R.; Ingram, J.; Sculthorpe, N.F. A scoping review of ‘Pacing’ for management of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): Lessons learned for the long COVID pandemic. J. Transl. Med. 2023, 21, 720. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Slavin, M.D.; Bailey, H.M.; Hickey, E.J.; Vasudevan, A.; Ledingham, A.; Tannenbaum, L.; Bateman, L.; Kaufman, D.L.; Peterson, D.L.; Ruhoy, I.S.; et al. Myalgic Encephalomyelitis-Chronic Fatigue Syndrome Common Data Element item content analysis. PLoS ONE 2023, 18, e0291364. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mecocci, P.; Fanó, G.; Fulle, S.; MacGarvey, U.; Shinobu, L.; Polidori, M.C.; Cherubini, A.; Vecchiet, J.; Senin, U.; Beal, M.F. Age-Dependent Increases in Oxidative Damage to DNA, Lipids, and Proteins in Human Skeletal Muscle. Free Radic. Biol. Med. 1999, 26, 303–308. [Google Scholar] [CrossRef]
- Pansarasa, O.; Castagna, L.; Colombi, B.; Vecchiet, J.; Felzani, G.; Marzatico, F. Age and Sex Differences in Human Skeletal Muscle: Role of Reactive Oxygen Species. Free Radic. Res. 2000, 33, 287–293. [Google Scholar] [CrossRef]
- Fanò, G.; Mecocci, P.; Vecchiet, J.; Belia, S.; Fulle, S.; Polidori, M.C.; Felzani, G.; Senin, U.; Vecchiet, L.; Beal, M.F. Age and Sex Influence on Oxidative Damage and Functional Status in Human Skeletal Muscle. J. Muscle Res. Cell Motil. 2001, 22, 345–351. [Google Scholar] [CrossRef]
- Scalabrin, M.; Turco, E.; Davigo, I.; Filadi, R.; Nogara, L.; Gherardi, G.; Barazzuol, L.; Armani, A.; Trani, G.; Negro, S.; et al. Alterations in Peroxisome-Mitochondria Interplay in Skeletal Muscle Accelerate Muscle Dysfunction. Nat. Commun. 2025, 16, 9868. [Google Scholar] [CrossRef] [PubMed]
- Fulle, S.; Didonna, S.; Puglielli, C.; Pietrangelo, T.; Beccafico, S.; Bellomo, R.; Protasi, F.; Fano, G. Age-Dependent Imbalance of the Antioxidative System in Human Satellite Cells. Exp. Gerontol. 2005, 40, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Beccafico, S.; Puglielli, C.; Pietrangelo, T.; Bellomo, R.; Fanò, G.; Fulle, S. Age-Dependent Effects on Functional Aspects in Human Satellite Cells. Ann. N. Y. Acad. Sci. 2007, 1100, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Fulle, S.; Belia, S.; Vecchiet, J.; Morabito, C.; Vecchiet, L.; Fanò, G. Modification of the Functional Capacity of Sarcoplasmic Reticulum Membranes in Patients Suffering from Chronic Fatigue Syndrome. Neuromuscul. Disord. 2003, 13, 479–484. [Google Scholar] [CrossRef]
- Pietrangelo, T.; D’Amelio, L.; Doria, C.; Mancinelli, R.; Fulle, S.; Fanò, G. Tiny Percutaneous Needle Biopsy: An Efficient Method for Studying Cellular and Molecular Aspects of Skeletal Muscle in Humans. Int. J. Mol. Med. 2011, 27, 361–367. [Google Scholar] [CrossRef][Green Version]
- Smirnova, I.V.; Pall, M.L. Elevated Levels of Protein Carbonyls in Sera of Chronic Fatigue Syndrome Patients. Mol. Cell Biochem. 2003, 248, 93–95. [Google Scholar] [CrossRef]
- Thambirajah, A.A.; Sleigh, K.; Stiver, H.G.; Chow, A.W. Differential Heat Shock Protein Responses to Strenuous Standardized Exercise in Chronic Fatigue Syndrome Patients and Matched Healthy Controls. Clin. Investig. Med. 2008, 31, E319–E327. [Google Scholar] [CrossRef]
- Manuel y Keenoy, B.; Moorkens, G.; Vertommen, J.; De Leeuw, I. Antioxidant Status and Lipoprotein Peroxidation in Chronic Fatigue Syndrome. Life Sci. 2001, 68, 2037–2049. [Google Scholar] [CrossRef]
- Shankar, V.; Wilhelmy, J.; Curtis, E.J.; Michael, B.; Cervantes, L.; Mallajosyula, V.; Davis, R.W.; Snyder, M.; Younis, S.; Robinson, W.H.; et al. Oxidative Stress Is a Shared Characteristic of ME/CFS and Long COVID. Proc. Natl. Acad. Sci. USA 2025, 122, e2426564122. [Google Scholar] [CrossRef]
- Pietrangelo, T.; Fulle, S.; Coscia, F.; Gigliotti, P.V.; Fanò-Illic, G. Old Muscle in Young Body: An Aphorism Describing the Chronic Fatigue Syndrome. Eur. J. Transl. Myol. 2018, 28, 7688. [Google Scholar] [CrossRef]
- Pietrangelo, T.; Cagnin, S.; Bondi, D.; Santangelo, C.; Marramiero, L.; Purcaro, C.; Bonadio, R.S.; Di Filippo, E.S.; Mancinelli, R.; Fulle, S.; et al. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome from Current Evidence to New Diagnostic Perspectives through Skeletal Muscle and Metabolic Disturbances. Acta Physiol. 2024, 240, e14122. [Google Scholar] [CrossRef] [PubMed]
- Tomas, C.; Brown, A.; Strassheim, V.; Elson, J.; Newton, J.; Manning, P. Cellular Bioenergetics Is Impaired in Patients with Chronic Fatigue Syndrome. PLoS ONE 2017, 12, e0186802. [Google Scholar] [CrossRef] [PubMed]
- Myhill, S.; Booth, N.E.; McLaren-Howard, J. Chronic Fatigue Syndrome and Mitochondrial Dysfunction. Int. J. Clin. Exp. Med. 2009, 2, 1–16. [Google Scholar] [PubMed]
- Bizjak, D.A.; Ohmayer, B.; Buhl, J.L.; Schneider, E.M.; Walther, P.; Calzia, E.; Jerg, A.; Matits, L.; Steinacker, J.M. Functional and Morphological Differences of Muscle Mitochondria in Chronic Fatigue Syndrome and Post-COVID Syndrome. Int. J. Mol. Sci. 2024, 25, 1675. [Google Scholar] [CrossRef]
- Plioplys, A.V.; Plioplys, S. Electron-Microscopic Investigation of Muscle Mitochondria in Chronic Fatigue Syndrome. Neuropsychobiology 1995, 32, 175–181. [Google Scholar] [CrossRef]
- Scheibenbogen, C.; Wirth, K.J. Key Pathophysiological Role of Skeletal Muscle Disturbance in Post COVID and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): Accumulated Evidence. J. Cachexia Sarcopenia Muscle 2025, 16, e13669. [Google Scholar] [CrossRef]
- Jäkel, B.; Kedor, C.; Grabowski, P.; Wittke, K.; Thiel, S.; Scherbakov, N.; Doehner, W.; Scheibenbogen, C.; Freitag, H. Hand Grip Strength and Fatigability: Correlation with Clinical Parameters and Diagnostic Suitability in ME/CFS. J. Transl. Med. 2021, 19, 159. [Google Scholar] [CrossRef]
- Pietrangelo, T.; Mancinelli, R.; Toniolo, L.; Montanari, G.; Vecchiet, J.; Fanò, G.; Fulle, S. Transcription Profile Analysis of Vastus Lateralis Muscle from Patients with Chronic Fatigue Syndrome. Int. J. Immunopathol. Pharmacol. 2009, 22, 795–807. [Google Scholar] [CrossRef]
- Pietrangelo, T.; Toniolo, L.; Paoli, A.; Fulle, S.; Puglielli, C.; Fanò, G.; Reggiani, C. Functional Characterization of Muscle Fibres from Patients with Chronic Fatigue Syndrome: Case-Control Study. Int. J. Immunopathol. Pharmacol. 2009, 22, 427–436. [Google Scholar] [CrossRef]
- Sies, H.; Jones, D.P. Reactive Oxygen Species (ROS) as Pleiotropic Physiological Signalling Agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef]
- Madreiter-Sokolowski, C.T.; Ramadani-Muja, J.; Ziomek, G.; Burgstaller, S.; Bischof, H.; Koshenov, Z.; Gottschalk, B.; Malli, R.; Graier, W.F. Tracking Intra- and Inter-Organelle Signaling of Mitochondria. FEBS J. 2019, 286, 4378–4401. [Google Scholar] [CrossRef]
- Di Filippo, E.S.; Checcaglini, F.; Fanò-Illic, G.; Fulle, S. H2O2/Ca2+/Zn2+ Complex Can Be Considered a “Collaborative Sensor” of the Mitochondrial Capacity? Antioxidants 2022, 11, 342. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Maes, M. Oxidative and Nitrosative Stress and Immune-Inflammatory Pathways in Patients with Myalgic Encephalomyelitis (ME)/Chronic Fatigue Syndrome (CFS). Curr. Neuropharmacol. 2014, 12, 168–185. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Ozigbo, A.A.; Varon, J.; Halma, M.; Laezzo, M.; Ang, S.P.; Iglesias, J. Mitochondrial Reactive Oxygen Species: A Unifying Mechanism in Long COVID and Spike Protein-Associated Injury: A Narrative Review. Biomolecules 2025, 15, 1339. [Google Scholar] [CrossRef] [PubMed]
- Severinsen, M.C.K.; Pedersen, B.K. Muscle-Organ Crosstalk: The Emerging Roles of Myokines. Endocr. Rev. 2020, 41, 594–609, Erratum in Endocr. Rev. 2021, 42, 97–99. https://doi.org/10.1210/endrev/bnaa024. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chmielewski, G.; Kuna, J.; Jaśkiewicz, Ł.; Knapik, M.; Mikiewicz, M.; Majewski, M.S.; Krajewska-Włodarczyk, M. The Dual Role of Myokines in Fatigue Associated with Inflammatory Joint Diseases. J. Inflamm. Res. 2025, 18, 11999–12020. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, W.; Wang, L.; You, W.; Shan, T. Myokines Mediate the Cross Talk between Skeletal Muscle and Other Organs. J. Cell Physiol. 2021, 236, 2393–2412. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, Y.; Yan, W. The Role of Extracellular Vesicles in Skeletal Muscle Wasting. J. Cachexia Sarcopenia Muscle 2023, 14, 2462–2472. [Google Scholar] [CrossRef]
- Watanabe, S.; Sudo, Y.; Makino, T.; Kimura, S.; Tomita, K.; Noguchi, M.; Sakurai, H.; Shimizu, M.; Takahashi, Y.; Sato, R.; et al. Skeletal Muscle Releases Extracellular Vesicles with Distinct Protein and microRNA Signatures That Function in the Muscle Microenvironment. PNAS Nexus 2022, 1, pgac173. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscles, Exercise and Obesity: Skeletal Muscle as a Secretory Organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [Google Scholar] [CrossRef]
- Castaño, C.; Mirasierra, M.; Vallejo, M.; Novials, A.; Párrizas, M. Delivery of Muscle-Derived Exosomal miRNAs Induced by HIIT Improves Insulin Sensitivity through down-Regulation of Hepatic FoxO1 in Mice. Proc. Natl. Acad. Sci. USA 2020, 117, 30335–30343. [Google Scholar] [CrossRef] [PubMed]
- Pal, M.; Febbraio, M.A.; Whitham, M. From Cytokine to Myokine: The Emerging Role of Interleukin-6 in Metabolic Regulation. Immunol. Cell Biol. 2014, 92, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, A.; Maeshige, N.; Yan, J.; Ma, X.; Uemura, M.; Matsuda, M.; Nishimura, Y.; Hasunuma, T.; Kondo, H.; Fujino, H.; et al. Skeletal Myotube-Derived Extracellular Vesicles Enhance Itaconate Production and Attenuate Inflammatory Responses of Macrophages. Front. Immunol. 2023, 14, 1099799. [Google Scholar] [CrossRef] [PubMed]
- Crescioli, C. Targeting Age-Dependent Functional and Metabolic Decline of Human Skeletal Muscle: The Geroprotective Role of Exercise, Myokine IL-6, and Vitamin, D. Int. J. Mol. Sci. 2020, 21, 1010. [Google Scholar] [CrossRef]
- Kim, J.-S.; Galvão, D.A.; Newton, R.U.; Gray, E.; Taaffe, D.R. Exercise-Induced Myokines and Their Effect on Prostate Cancer. Nat. Rev. Urol. 2021, 18, 519–542. [Google Scholar] [CrossRef]
- Whitham, M.; Febbraio, M.A. The Ever-Expanding Myokinome: Discovery Challenges and Therapeutic Implications. Nat. Rev. Drug Discov. 2016, 15, 719–729. [Google Scholar] [CrossRef]
- Giloteaux, L.; Glass, K.A.; Germain, A.; Franconi, C.J.; Zhang, S.; Hanson, M.R. Dysregulation of Extracellular Vesicle Protein Cargo in Female Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Cases and Sedentary Controls in Response to Maximal Exercise. J. Extracell. Vesicles 2024, 13, e12403. [Google Scholar] [CrossRef]
- Glass, K.A.; Giloteaux, L.; Zhang, S.; Hanson, M.R. Extracellular Vesicle Proteomics Uncovers Energy Metabolism, Complement System, and Endoplasmic Reticulum Stress Response Dysregulation Postexercise in Males with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Clin. Transl. Med. 2025, 15, e70346. [Google Scholar] [CrossRef]
- Germain, A.; Glass, K.A.; Eckert, M.A.; Giloteaux, L.; Hanson, M.R. Temporal Dynamics of the Plasma Proteomic Landscape Reveals Maladaptation in ME/CFS Following Exertion. Mol. Cell Proteom. 2025, 24, 101467. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mancinelli, R.; Checcaglini, F.; Coscia, F.; Gigliotti, P.; Fulle, S.; Fanò-Illic, G. Biological Aspects of Selected Myokines in Skeletal Muscle: Focus on Aging. Int. J. Mol. Sci. 2021, 22, 8520. [Google Scholar] [CrossRef]
- Visconti, N.R.G.; Rocha, N.N.; Nascimento, G.S.; Menário, C.V.B.; Silva, J.D.; Martins, C.M.; Caruso-Neves, C.; Cruz, F.F.; Rocco, P.R.M.; Mello, F.C.Q.; et al. Elevated adipokines and myokines are associated with fatigue in long COVID patients. Front. Med. 2025, 12, 1547886. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jones, D.E.; Hollingsworth, K.G.; Jakovljevic, D.G.; Fattakhova, G.; Pairman, J.; Blamire, A.M.; Trenell, M.I.; Newton, J.L. Loss of capacity to recover from acidosis on repeat exercise in chronic fatigue syndrome: A case–control study. Eur. J. Clin. Investig. 2012, 42, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Brenu, E.W.; van Driel, M.L.; Staines, D.R.; Ashton, K.J.; Ramos, S.B.; Keane, J.; Klimas, N.G.; Marshall-Gradisnik, S.M. Immunological Abnormalities as Potential Biomarkers in Chronic Fatigue Syndrome/Myalgic Encephalomyelitis. J. Transl. Med. 2011, 9, 81. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Yang, J.; Zhao, J.; Chen, Z.; Yang, H.; Cai, D. Evaluating the Causal Role of Genetically Inferred Immune Cells and Inflammatory Cytokines on Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Biomedicines 2025, 13, 1200. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Su, J. Toll-like Receptor Signaling in Teleosts. Sci. China Life Sci. 2025, 68, 1889–1911. [Google Scholar] [CrossRef] [PubMed]
- Almutairi, B.; Langley, C.; Crawley, E.; Thai, N.J. Using structural and functional MRI as a neuroimaging technique to investigate chronic fatigue syndrome/myalgic enceph-alopathy: A systematic review. BMJ Open 2020, 10, e031672. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Victorio, M.; Dieffenderfer, J.; Songkakul, T.; Willeke, J.; Bozkurt, A.; Pozdin, V.A. Wearable Wireless Functional Near-Infrared Spectroscopy System for Cognitive Activity Monitoring. Biosensors 2025, 15, 92. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- VanElzakker, M.B.; Brumfield, S.A.; Lara Mejia, P.S. Neuroinflammation and Cytokines in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): A Critical Review of Research Methods. Front. Neurol. 2019, 9, 1033. [Google Scholar] [CrossRef]
- Nakatomi, Y.; Mizuno, K.; Ishii, A.; Wada, Y.; Tanaka, M.; Tazawa, S.; Onoe, K.; Fukuda, S.; Kawabe, J.; Takahashi, K.; et al. Neuroinflammation in Patients with Chronic Fatigue Syndrome/Myalgic Encephalomyelitis: An 11C-(R)-PK11195 PET Study. J. Nucl. Med. 2014, 55, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Rutherford, G.; Manning, P.; Newton, J.L. Understanding Muscle Dysfunction in Chronic Fatigue Syndrome. J. Aging Res. 2016, 2016, 2497348. [Google Scholar] [CrossRef]
- Tsamou, M.; Kremers, F.A.C.; Samaritakis, K.A.; Roggen, E.L. Identifying microRNAs Possibly Implicated in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome and Fibromyalgia: A Review. Int. J. Mol. Sci. 2024, 25, 9551. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Lidbury, B.A.; Thomas, N.; Gooley, P.R.; Armstrong, C.W. Machine Learning and Multi-Omics in Precision Medicine for ME/CFS. J. Transl. Med. 2025, 23, 68. [Google Scholar] [CrossRef] [PubMed]
- Komaroff, A.L.; Lipkin, W.I. ME/CFS and Long COVID Share Similar Symptoms and Biological Abnormalities: Road Map to the Literature. Front. Med. 2023, 10, 1187163. [Google Scholar] [CrossRef] [PubMed]
- Shekhar Patil, M.; Richter, E.; Fanning, L.; Hendrix, J.; Wyns, A.; Barrero Santiago, L.; Nijs, J.; Godderis, L.; Polli, A. Epigenetic Changes in Patients with Post-Acute COVID-19 Symptoms (PACS) and Long-COVID: A Systematic Review. Expert. Rev. Mol. Med. 2024, 26, e29. [Google Scholar] [CrossRef]
- Deumer, U.S.; Varesi, A.; Floris, V.; Savioli, G.; Mantovani, E.; López-Carrasco, P.; Rosati, G.M.; Prasad, S.; Ricevuti, G. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): An Overview. J. Clin. Med. 2021, 10, 4786. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Li, Q.; Xiao, N.; Zhang, H.; Liang, G.; Lin, Y.; Qian, Z.; Yang, X.; Yang, J.; Fu, Y.; Zhang, C.; et al. Systemic aging and aging-related diseases. FASEB J. 2025, 39, e70430. [Google Scholar] [CrossRef] [PubMed]
- Syed, A.M.; Karius, A.K.; Ma, J.; Wang, P.Y.; Hwang, P.M. Mitochondrial Dysfunction in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Physiology 2025, 40, 319–328. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kedor, C.; Freitag, H.; Meyer-Arndt, L.; Wittke, K.; Hanitsch, L.G.; Zoller, T.; Steinbeis, F.; Haffke, M.; Rudolf, G.; Heidecker, B.; et al. A Prospective Observational Study of Post-COVID-19 Chronic Fatigue Syndrome Following the First Pandemic Wave in Germany and Biomarkers Associated with Symptom Severity. Nat. Commun. 2022, 13, 5104. [Google Scholar] [CrossRef]
- Dehlia, A.; Guthridge, M.A. The Persistence of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) after SARS-CoV-2 Infection: A Systematic Review and Meta-Analysis. J. Infect. 2024, 89, 106297. [Google Scholar] [CrossRef]
- Paul, B.D.; Lemle, M.D.; Komaroff, A.L.; Snyder, S.H. Redox Imbalance Links COVID-19 and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Proc. Natl. Acad. Sci. USA 2021, 118, e2024358118. [Google Scholar] [CrossRef]
- Coscia, F.; Mancinelli, R.; Gigliotti, P.V.; Checcaglini, F.; Fanò-Illic, G. Physical Activity Effects on Muscle Fatigue in Sport in Active Adults with Long COVID-19: An Observational Study. Diagnostics 2023, 13, 1336. [Google Scholar] [CrossRef]
- Carraro, U.; Albertin, G.; Martini, A.; Giuriati, W.; Guidolin, D.; Masiero, S.; Kern, H.; Hofer, C.; Marcante, A.; Ravara, B. To Contrast and Reverse Skeletal Muscle Weakness by Full-Body In-Bed Gym in Chronic COVID-19 Pandemic Syndrome. Eur. J. Transl. Myol. 2021, 31, 9641. [Google Scholar] [CrossRef] [PubMed]
- Larun, L.; Brurberg, K.G.; Odgaard-Jensen, J.; Price, J.R. Exercise therapy for chronic fatigue syndrome. Cochrane Database Syst Rev. 2019, 10, CD003200. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Malouff, J.M.; Thorsteinsson, E.B.; Rooke, S.E.; Bhullar, N.; Schutte, N.S. Efficacy of cognitive behavioral therapy for chronic fatigue syndrome: A meta-analysis. Clin. Psychol. Rev. 2008, 28, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Vink, M.; Vink-Niese, F. Graded exercise therapy does not restore the ability to work in ME/CFS—Rethinking of a Cochrane review. Work 2020, 66, 283–308. [Google Scholar] [CrossRef] [PubMed]
- Coscia, F.; Di Filippo, E.S.; Gigliotti, P.V.; Fano Illic, G. Effect of Physical Activity on Long COVID Fatigue: An Unsolved Enigma. Eur. J. Transl. Myol. 2023, 33, 11639. [Google Scholar] [CrossRef]
- Thapaliya, K.; Marshall-Gradisnik, S.; Barth, M.; Eaton-Fitch, N.; Barnden, L. Brainstem volume changes in myalgic encephalomyelitis/chronic fatigue syndrome and long COVID patients. Front. Neurosci. 2023, 17, 1125208. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Castro-Marrero, J.; Segundo, M.J.; Lacasa, M.; Martinez-Martinez, A.; Sentañes, R.S.; Alegre-Martin, J. Effect of Dietary Coenzyme Q10 Plus NADH Supplementation on Fatigue Perception and Health-Related Quality of Life in Individuals with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A Prospective, Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2021, 13, 2658. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Eckey, M.; Li, P.; Morrison, B.; Bergquist, J.; Davis, R.W.; Xiao, W. Patient-reported treatment outcomes in ME/CFS and long COVID. Proc. Natl. Acad. Sci. USA 2025, 122, e2426874122. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Barnden, L.R.; Shan, Z.Y.; Staines, D.R.; Marshall-Gradisnik, S.; Finegan, K.; Ireland, T.; Bhuta, S. Intra brainstem connectivity is impaired in chronic fatigue syndrome. Neuroimage Clin. 2019, 24, 102045. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]



| Feature | CDC (1994) | CCC (2003) | IOM/SEID (2015) |
|---|---|---|---|
| Primary Focus | Unexplained chronic fatigue. | Neurological and Immunological features. | Post-Exertional Malaise (PEM) and core symptoms. |
| Pathognomonic Sign (PEM) | Optional (one of 8 minor criteria). | Mandatory. | Mandatory. |
| Sensitivity | High. Identifies a broad and heterogeneous patient population. | Moderate. More restrictive; excludes patients without systemic involvement. | High. Designed for clinical utility and inclusivity of severe cases. |
| Specificity | Low. High risk of misclassifying primary psychiatric or other fatiguing conditions. | High. Highly effective at distinguishing ME from other pathologies. | Moderate/High. More specific than Fukuda, though less granular than CCC. |
| Application | Former gold standard. | Clinical research and rigorous diagnosis. | Rapid clinical diagnosis (Primary Care). |
| Secretome Component | Pathological Deviation | Pathophysiological Mechanism | Clinical Aspects |
|---|---|---|---|
| Pro-inflammatory Myokines | Post-exertional elevation/Prolonged expression | Systemic Inflammation: Activation of the HPA axis and systemic immune response. | Flu-like symptoms and systemic PEM. |
| Extracellular Vesicles (EVs) | Altered cargo (miRNAs, mtDNA) | Intercellular Communication: Transport of “danger signals” to the Central Nervous System. | Neuroinflammation and cognitive impairment (“brain fog”). |
| Lactate & Metabolic Acids | Accelerated accumulation/Impaired clearance | Peripheral Sensitization: Irritation of muscle nociceptors (acid-sensing ion channels). | Muscle pain and deep somatic hyperalgesia. |
| Reactive Oxygen Species (ROS) | Excessive production vs. low antioxidant capacity | Oxidative Stress: Damage to mitochondrial membranes and proteins. | Non-restorative fatigue and cellular energy depletion. |
| Growth Factors (e.g., IGF-1, BDNF) | Reduced bioavailability or signaling | Impaired Recovery: Decreased regenerative capacity and neuroplasticity. | Prolonged recovery time (lasting days or weeks). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Fanò-Illic, G.; Coscia, F.; Gigliotti, P.V.; Checcaglini, F.; Carraro, U.; Fulle, S.; Mancinelli, R. Pathophysiological, Translational, and Diagnostic Aspects of ME/CFS: A Focus on Skeletal Muscle Involvement. Diagnostics 2026, 16, 1019. https://doi.org/10.3390/diagnostics16071019
Fanò-Illic G, Coscia F, Gigliotti PV, Checcaglini F, Carraro U, Fulle S, Mancinelli R. Pathophysiological, Translational, and Diagnostic Aspects of ME/CFS: A Focus on Skeletal Muscle Involvement. Diagnostics. 2026; 16(7):1019. https://doi.org/10.3390/diagnostics16071019
Chicago/Turabian StyleFanò-Illic, Giorgio, Francesco Coscia, Paola V. Gigliotti, Franco Checcaglini, Ugo Carraro, Stefania Fulle, and Rosa Mancinelli. 2026. "Pathophysiological, Translational, and Diagnostic Aspects of ME/CFS: A Focus on Skeletal Muscle Involvement" Diagnostics 16, no. 7: 1019. https://doi.org/10.3390/diagnostics16071019
APA StyleFanò-Illic, G., Coscia, F., Gigliotti, P. V., Checcaglini, F., Carraro, U., Fulle, S., & Mancinelli, R. (2026). Pathophysiological, Translational, and Diagnostic Aspects of ME/CFS: A Focus on Skeletal Muscle Involvement. Diagnostics, 16(7), 1019. https://doi.org/10.3390/diagnostics16071019









