Soluble CD14 and Lipopolysaccharide-Binding Protein Are Not Superior to Soluble CD25 as Biomarkers for Sarcoidosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Laboratory Measurements
2.3. Statistical Analysis
2.4. Ethics Approval
3. Results
3.1. Demographics
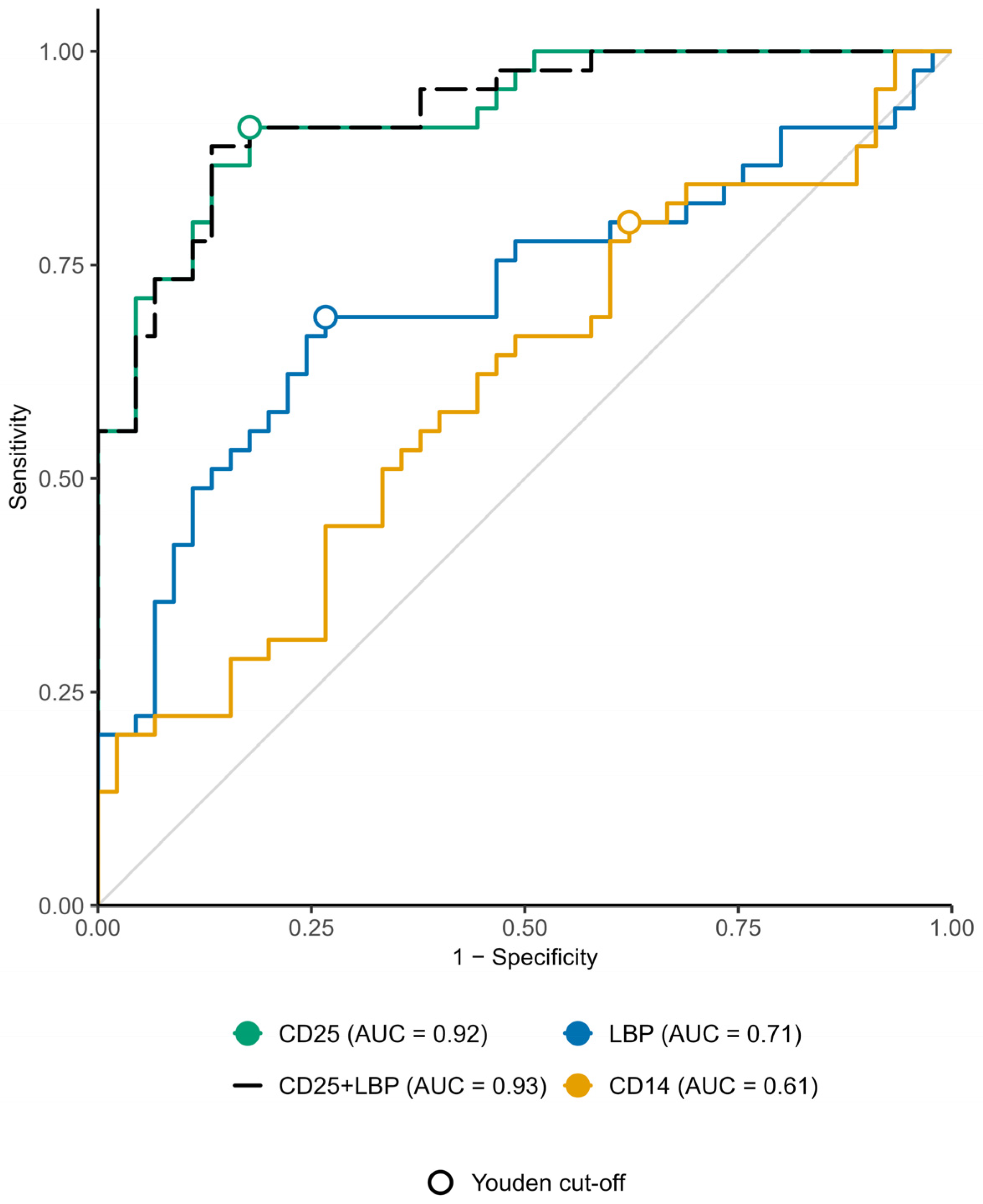
3.2. Biomarker Analysis
4. Discussion
4.1. Diagnostic Performance of Circulating Biomarkers
4.2. Longitudinal Changes Under Immunosuppressive Therapy
4.3. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACE | Angiotensin-Converting Enzyme |
| LBP | Lipopolysaccharide-binding protein |
| sCD25 | soluble interleukin-2 receptor (sCD25) |
| CD14 | Cluster of Differentiation 14 |
| BMI | Body Mass Index |
| FDG-PET | 18F-Fluorodeoxyglucose Positron Emission Tomography |
| PAMPs | Pathogen-Associated Molecular Patterns |
| WASOG | World Association of Sarcoidosis and Other Granulomatous Disorders |
| JCS | Japanese Circulation Society |
References
- Schupp, J.C.; Freitag-Wolf, S.; Bargagli, E.; Mihailović-Vučinić, V.; Rottoli, P.; Grubanovic, A.; Müller, A.; Jochens, A.; Tittmann, L.; Schnerch, J.; et al. Phenotypes of Organ Involvement in Sarcoidosis. Eur. Respir. J. 2018, 51, 1700991. [Google Scholar] [CrossRef]
- Baughman, R.P.; Teirstein, A.S.; Judson, M.A.; Rossman, M.D.; Yeager, H.; Bresnitz, E.A.; DePalo, L.; Hunninghake, G.; Iannuzzi, M.C.; Johns, C.J.; et al. Clinical Characteristics of Patients in a Case Control Study of Sarcoidosis. Am. J. Respir. Crit. Care Med. 2001, 164, 1885–1889. [Google Scholar] [CrossRef]
- Hu, X.; Zou, L.; Wang, S.; Zeng, T.; Li, P.; Shen, Y.; Chen, L. Performance of Serum Angiotensin-Converting Enzyme in Diagnosing Sarcoidosis and Predicting the Active Status of Sarcoidosis: A Meta-Analysis. Biomolecules 2022, 12, 1400. [Google Scholar] [CrossRef] [PubMed]
- Fløe, A.; Hoffmann, H.J.; Nissen, P.H.; Møller, H.J.; Hilberg, O. Genotyping Increases the Yield of Angiotensin-Converting Enzyme in Sarcoidosis—A Systematic Review. Dan. Med. J. 2014, 61, A4815. [Google Scholar] [PubMed]
- Rigat, B.; Hubert, C.; Alhenc-Gelas, F.; Cambien, F.; Corvol, P.; Soubrier, F. An Insertion/Deletion Polymorphism in the Angiotensin I-Converting Enzyme Gene Accounting for Half the Variance of Serum Enzyme Levels. J. Clin. Investig. 1990, 86, 1343–1346. [Google Scholar] [CrossRef]
- Gupta, S.; Miloni Parmar, M.P.; Padappayil, R.P.; Bansal, A.; Daouk, S. Role of Serum Soluble Interleukin-2 Receptor Level in the Diagnosis of Sarcoidosis: A Systematic Review and Meta-Analysis. Sarcoidosis Vasc. Diffus. Lung Dis. 2023, 40, e2023005. [Google Scholar] [CrossRef]
- Groen-Hakan, F.; Eurelings, L.; Ten Berge, J.C.; Van Laar, J.; Ramakers, C.R.B.; Dik, W.A.; Rothova, A. Diagnostic Value of Serum-Soluble Interleukin 2 Receptor Levels vs Angiotensin-Converting Enzyme in Patients with Sarcoidosis-Associated Uveitis. JAMA Ophthalmol. 2017, 135, 1352. [Google Scholar] [CrossRef]
- Damoiseaux, J. The IL-2—IL-2 Receptor Pathway in Health and Disease: The Role of the Soluble IL-2 Receptor. Clin. Immunol. 2020, 218, 108515. [Google Scholar] [CrossRef]
- Karim, A.F.; Eurelings, L.E.M.; Bansie, R.D.; Van Hagen, P.M.; Van Laar, J.A.M.; Dik, W.A. Soluble Interleukin-2 Receptor: A Potential Marker for Monitoring Disease Activity in IgG4-Related Disease. Mediat. Inflamm. 2018, 2018, 6103064. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, Q.; Li, M.; Huang, G.; Guo, L.; Chen, Z.; Ding, Q.; Gu, Y. Clinical Diagnostic Value of Serum Soluble IL-2 Receptor for Stage I Sarcoidosis in Benign Isolated Mediastinal and Hilar Lymphadenopathy. Clin. Chim. Acta 2023, 545, 117372. [Google Scholar] [CrossRef]
- Futami, Y.; Takeda, Y.; Koba, T.; Narumi, R.; Nojima, Y.; Ito, M.; Nakayama, M.; Ishida, M.; Yoshimura, H.; Naito, Y.; et al. Identification of CD14 and Lipopolysaccharide-Binding Protein as Novel Biomarkers for Sarcoidosis Using Proteomics of Serum Extracellular Vesicles. Int. Immunol. 2022, 34, 327–340. [Google Scholar] [CrossRef] [PubMed]
- Anas, A.; van der Poll, T.; de Vos, A.F. Role of CD14 in Lung Inflammation and Infection. Crit. Care Lond. Engl. 2010, 14, 209. [Google Scholar] [CrossRef]
- Pforte, A.; Schiessler, A.; Gais, P.; Beer, B.; Ehlers, M.; Schütt, C.; Ziegler-Heitbrock, H.W. Expression of CD14 Correlates with Lung Function Impairment in Pulmonary Sarcoidosis. Chest 1994, 105, 349–354. [Google Scholar] [CrossRef]
- Schröder, N.W.J.; Heine, H.; Alexander, C.; Manukyan, M.; Eckert, J.; Hamann, L.; Göbel, U.B.; Schumann, R.R. Lipopolysaccharide Binding Protein Binds to Triacylated and Diacylated Lipopeptides and Mediates Innate Immune Responses. J. Immunol 2004, 173, 2683–2691. [Google Scholar] [CrossRef]
- Judson, M.A.; Costabel, U.; Drent, M.; Wells, A.; Maier, L.; Koth, L.; Shigemitsu, H.; Culver, D.A.; Gelfand, J.; Valeyre, D.; et al. The WASOG Sarcoidosis Organ Assessment Instrument: An Update of a Previous Clinical Tool. Sarcoidosis Vasc. Diffuse Lung Dis. 2014, 31, 19–27. [Google Scholar]
- Terasaki, F.; Azuma, A.; Anzai, T.; Ishizaka, N.; Ishida, Y.; Isobe, M.; Inomata, T.; Ishibashi-Ueda, H.; Eishi, Y.; Kitakaze, M.; et al. JCS 2016 guideline on diagnosis and treatment of cardiac sarcoidosis―digest version―. Circ. J. 2019, 83, 2329–2388. [Google Scholar] [CrossRef] [PubMed]
- Birnie, D.H.; Sauer, W.H.; Bogun, F.; Cooper, J.M.; Culver, D.A.; Duvernoy, C.S.; Judson, M.A.; Kron, J.; Mehta, D.; Cosedis Nielsen, J.; et al. HRS Expert Consensus Statement on the Diagnosis and Management of Arrhythmias Associated with Cardiac Sarcoidosis. Heart Rhythm 2014, 11, 1305–1323. [Google Scholar] [CrossRef] [PubMed]
- Fridlender, Z.G.; Schwartz, A.; Kohan, M.; Amir, G.; Glazer, M.; Berkman, N. Association between CD14 Gene Polymorphisms and Disease Phenotype in Sarcoidosis. Respir. Med. 2010, 104, 1336–1343. [Google Scholar] [CrossRef]
- Schimmelpennink, M.; Quanjel, M.; Vorselaars, A.; Wiertz, I.; Veltkamp, M.; Van Moorsel, C.; Grutters, J. Value of Serum Soluble Interleukin-2 Receptor as a Diagnostic and Predictive Biomarker in Sarcoidosis. Expert Rev. Respir. Med. 2020, 14, 749–756. [Google Scholar] [CrossRef]
- Eurelings, L.E.M.; Miedema, J.R.; Dalm, V.A.S.H.; Van Daele, P.L.A.; Van Hagen, P.M.; Van Laar, J.A.M.; Dik, W.A. Sensitivity and Specificity of Serum Soluble Interleukin-2 Receptor for Diagnosing Sarcoidosis in a Population of Patients Suspected of Sarcoidosis. PLoS ONE 2019, 14, e0223897. [Google Scholar] [CrossRef]
- Gundlach, E.; Hoffmann, M.M.; Prasse, A.; Heinzelmann, S.; Ness, T. Interleukin-2 Receptor and Angiotensin-Converting Enzyme as Markers for Ocular Sarcoidosis. PLoS ONE 2016, 11, e0147258. [Google Scholar] [CrossRef] [PubMed]
- Mortaz, E.; Adcock, I.M.; Abedini, A.; Kiani, A.; Kazempour-Dizaji, M.; Movassaghi, M.; Garssen, J. The Role of Pattern Recognition Receptors in Lung Sarcoidosis. Eur. J. Pharmacol. 2017, 808, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Striz, I.; Zheng, L.; Wang, Y.M.; Pokorná, H.; Bauer, P.C.; Costabel, U. Soluble CD14 Is Increased in Bronchoalveolar Lavage of Active Sarcoidosis and Correlates with Alveolar Macrophage Membrane-Bound CD14. Am. J. Respir. Crit. Care Med. 1995, 151, 544–547. [Google Scholar] [CrossRef]
- Guerrero, C.R.; Maier, L.A.; Griffin, T.J.; Higgins, L.; Najt, C.P.; Perlman, D.M.; Bhargava, M. Application of Proteomics in Sarcoidosis. Am. J. Respir. Cell Mol. Biol. 2020, 63, 727–738. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Li, X. Role of Leptin and Adiponectin in Immune Response and Inflammation. Int. Immunopharmacol. 2025, 161, 115082. [Google Scholar] [CrossRef]
- Taylor, E.B. The Complex Role of Adipokines in Obesity, Inflammation, and Autoimmunity. Clin. Sci. 2021, 135, 731–752. [Google Scholar] [CrossRef]
- Dumas, O.; Boggs, K.M.; Cozier, Y.C.; Stampfer, M.J.; Camargo, C.A. Prospective Study of Body Mass Index and Risk of Sarcoidosis in US Women. Eur. Respir. J. 2017, 50, 1701397. [Google Scholar] [CrossRef]
- Cozier, Y.C.; Coogan, P.F.; Govender, P.; Berman, J.S.; Palmer, J.R.; Rosenberg, L. Obesity and Weight Gain in Relation to Incidence of Sarcoidosis in US Black Women: Data from the Black Women’s Health Study. Chest 2015, 147, 1086–1093. [Google Scholar] [CrossRef]
- Judson, M.A.; Tiwari, A.; Gemoets, D.E. The Relationship of Obesity and OSA to the Development of Sarcoidosis: A Large Retrospective Case-Control US Veterans Administration Analysis. Chest 2022, 162, 1086–1092. [Google Scholar] [CrossRef]
- Colamatteo, A.; Fusco, C.; Matarese, A.; Matarese, G. Obesity and Autoimmunity Epidemic: The Role of Immunometabolism. Annu. Rev. Nutr. 2025, 45, 115–140. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, X.; Zhao, M.; Lower, E.E.; Baughman, R.P. SACE and IL-2R as Serum Biomarkers for Evaluation of Multi-Organ Involvement and Prognosis of Sarcoidosis. Respir. Res. 2023, 24, 219. [Google Scholar] [CrossRef]
- López-Martínez, J.; Fernández-Valmaña, A.; Mayer-Fuentes, Á.; Mercadé-Torras, J.M.; Díaz-Martín, E.; García-González, M.; Mas-Maresma, L.; Feijoo-Massó, C. Diagnostic Value of Angiotensin-Converting Enzyme Levels for Assessing Organ Involvement in Sarcoidosis: A Retrospective Single-Centre Study. Med. Clin. 2026, 166, 107278. [Google Scholar] [CrossRef]
- Alía, P.; Mañá, J.; Capdevila, O.; Alvarez, A.; Navarro, M.A. Association between ACE Gene I/D Polymorphism and Clinical Presentation and Prognosis of Sarcoidosis. Scand. J. Clin. Lab. Investig. 2005, 65, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.-Y.; Du, X.; Dong, J.-Z. Re-Evaluating Serum Angiotensin-Converting Enzyme in Sarcoidosis. Front. Immunol. 2023, 14, 950095. [Google Scholar] [CrossRef] [PubMed]
- d’Alessandro, M.; Bergantini, L.; Perrone, A.; Cameli, P.; Cameli, M.; Prasse, A.; Plataroti, D.; Sestini, P.; Bargagli, E. Serial Investigation of Angiotensin-Converting Enzyme in Sarcoidosis Patients Treated with Angiotensin-Converting Enzyme Inhibitor. Eur. J. Intern. Med. 2020, 78, 58–62. [Google Scholar] [CrossRef]
- Thi Hong Nguyen, C.; Kambe, N.; Kishimoto, I.; Ueda-Hayakawa, I.; Okamoto, H. Serum Soluble Interleukin-2 Receptor Level Is More Sensitive than Angiotensin-Converting Enzyme or Lysozyme for Diagnosis of Sarcoidosis and May Be a Marker of Multiple Organ Involvement. J. Dermatol. 2017, 44, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Vanmaris, R.M.M.; Rijkers, G.T. Biological Role of the Soluble Interleukin-2 Receptor in Sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 2017, 34, 122–129. [Google Scholar] [CrossRef]
- Benamore, R.; Kendrick, Y.R.; Repapi, E.; Helm, E.; Cole, S.L.; Taylor, S.; Ho, L.-P. CTAS: A CT Score to Quantify Disease Activity in Pulmonary Sarcoidosis. Thorax 2016, 71, 1161–1163. [Google Scholar] [CrossRef]

| Variables | Control (n = 46) | Sarcoidosis (n = 46) | p-Value |
|---|---|---|---|
| Male sex, n (%) | 28 (60.9) | 28 (60.9) | |
| Caucasian, n (%) | 42 (91.3) | ||
| Age, mean (SD) | 50 (13.0) | 50 (13.1) | |
| Weight, mean (SD) | 72.6 (13.7) | 80.7 (19.4) | 0.029 |
| Height, mean (SD) | 1.73 (0.09) | 1.71 (0.09) | 0.243 |
| Body mass index, kg/m2, mean (SD) | 24.2 (3.5) | 27.7 (6.8) | 0.004 |
| Former or active smoker, n (%) | 12 (26) | ||
| ACE inhibitor use, n (%) | 4 (8.7) |
| Variables | Sarcoidosis (n = 46) |
|---|---|
| Isolated pulmonary disease | 3 (6.5) |
| Multisystemic sarcoidosis | 43 (93.5) |
| Type of extra-thoracic involvement | |
| Extra-thoracic lymph nodes | 26 (56.5) |
| Heart | 24 (52.2) |
| Spleen | 14 (30.4) |
| Liver | 11 (23.9) |
| Bone | 11 (23.9) |
| Skin | 9 (19.6) |
| Nervous system | 5 (10.9) |
| Eye | 3 (6.5) |
| Kidney | 2 (4.3) |
| Number of organs involved, median [IQR] | 3 [2–5] |
| Variables | Control | Sarcoidosis | p-Value |
|---|---|---|---|
| (n = 46) | (n = 46) | ||
| LBP (µg/mL) | 9.7 [8.3–10.8] | 12.5 [10.0–13.9] | <0.001 |
| sCD14 (ng/mL) | 1180 [1010–1452.5] | 1320 [1120.0–1570.0] | 0.028 |
| sCD25 (pg/mL) | 640.2 [557.9–763.6] | 1666.3 [1071.3–2454.9] | <0.001 |
| Variables | p-Value | Odds Ratio | 95% Confidence Interval |
|---|---|---|---|
| sCD25 (per 100 pg/mL) | <0.001 | 1.53 | 1.258–1.862 |
| BMI (per 1 kg/m2) | 0.044 | 1.179 | 1.005–1.383 |
| Biomarker (Units) | n Pairs | T1 Median [IQR] | T2 Median [IQR] | Wilcoxon V | p-Value |
|---|---|---|---|---|---|
| sCD25 (pg/mL) | 32 | 1620.5 [945.1–2349.5] | 978.7 [689.5–1299.2] | 497.0 | <0.0001 |
| sCD14 (ng/mL) | 31 | 1380.0 [1135.0–1570.0] | 1160.0 [1040.0–1245.0] | 458.5 | <0.0001 |
| LBP (µg/mL) | 32 | 12.8 [10.8–14.0] | 10.7 [8.6–12.3] | 432.0 | 0.0017 |
| ACE (U/L) | 32 | 48.4 [34.2–84.1] | 33.0 [23.9–42.4] | 438.5 | 0.0011 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ammann, S.; Marques-Vidal, P.; Perreau, M.; Ribi, C. Soluble CD14 and Lipopolysaccharide-Binding Protein Are Not Superior to Soluble CD25 as Biomarkers for Sarcoidosis. Diagnostics 2026, 16, 1018. https://doi.org/10.3390/diagnostics16071018
Ammann S, Marques-Vidal P, Perreau M, Ribi C. Soluble CD14 and Lipopolysaccharide-Binding Protein Are Not Superior to Soluble CD25 as Biomarkers for Sarcoidosis. Diagnostics. 2026; 16(7):1018. https://doi.org/10.3390/diagnostics16071018
Chicago/Turabian StyleAmmann, Sabine, Pedro Marques-Vidal, Matthieu Perreau, and Camillo Ribi. 2026. "Soluble CD14 and Lipopolysaccharide-Binding Protein Are Not Superior to Soluble CD25 as Biomarkers for Sarcoidosis" Diagnostics 16, no. 7: 1018. https://doi.org/10.3390/diagnostics16071018
APA StyleAmmann, S., Marques-Vidal, P., Perreau, M., & Ribi, C. (2026). Soluble CD14 and Lipopolysaccharide-Binding Protein Are Not Superior to Soluble CD25 as Biomarkers for Sarcoidosis. Diagnostics, 16(7), 1018. https://doi.org/10.3390/diagnostics16071018








