Local Versus Global Binarization Techniques After Frangi Filtering for Optical Coherence Tomography Angiography Based Retinal Vessel Density Assessment in Diabetic Retinopathy
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Eligibility Criteria
2.3. Ophthalmologic Examination and Imaging Protocol
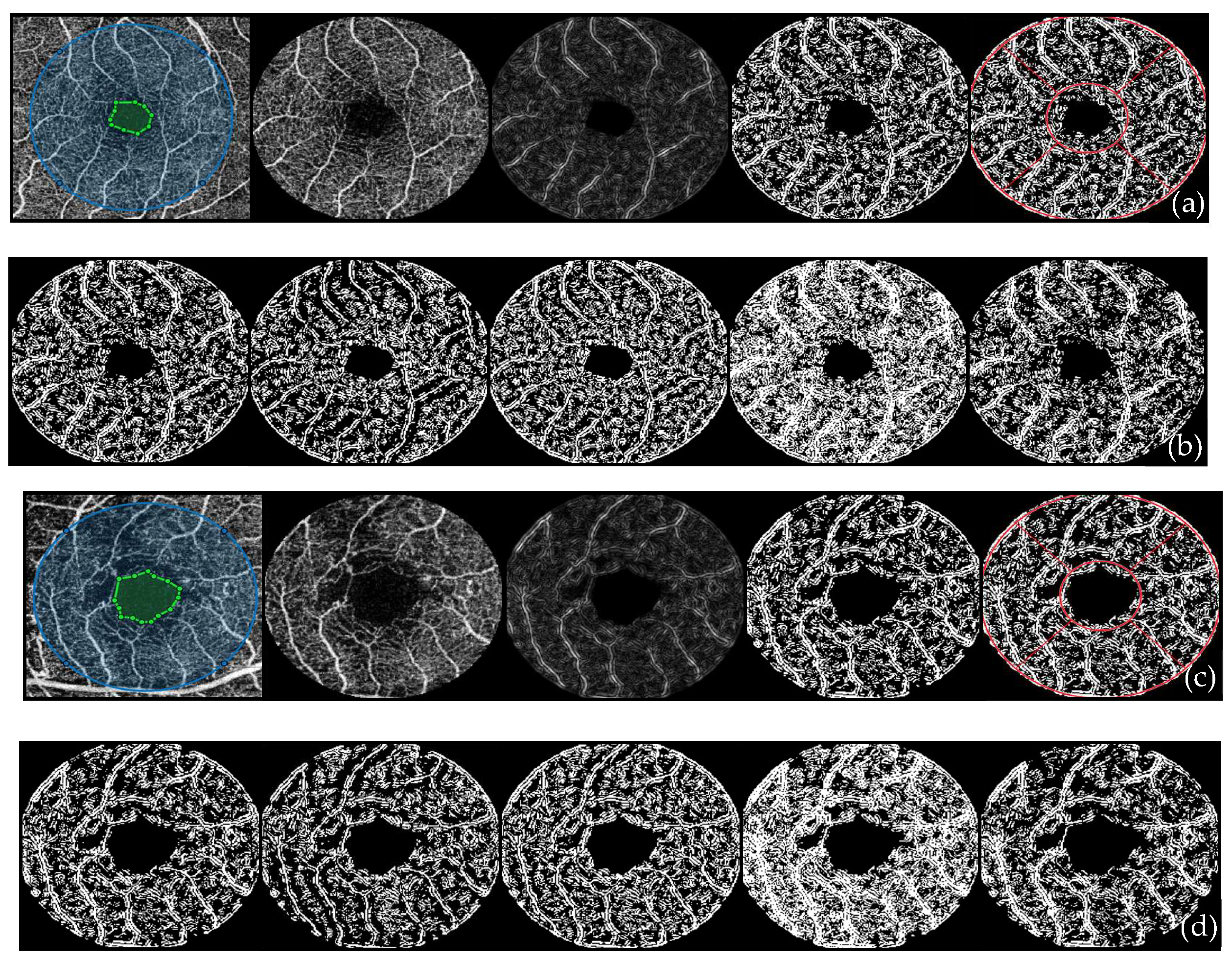
2.4. OCTA Image Acquisition and Preprocessing
2.5. Statistical Analysis
3. Results
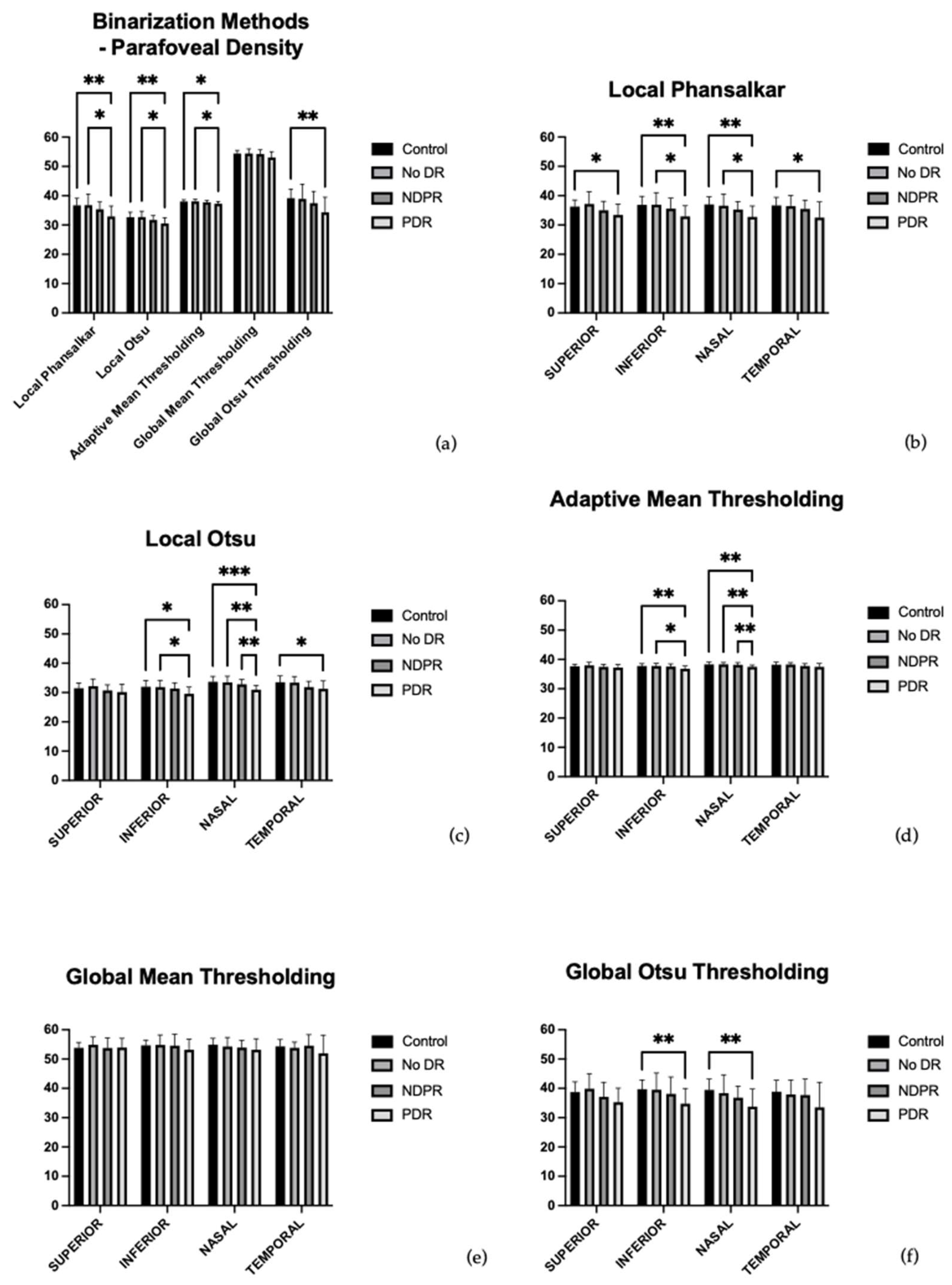
3.1. Comparison Between Binarization Methods
3.2. Results of Each Binarization Methods
3.2.1. Local Phansalkar
- Superior quadrant: p adj (control, RDP) = 0.0370,
- Inferior quadrant: p adj (control, RDP) = 0.0048, p adj (no DR and RDP) = 0.0325,
- Nasal quadrant: p adj (control and RDP) = 0.0013, p adj (no DR and RDP) = 0.0423,
- Temporal quadrant: p adj (control and RDP) = 0.0250.
3.2.2. Local Otsu
- Inferior quadrant: p adj (control and RDP) = 0.0119, p adj (no DR and RDP) = 0.0388,
- Nasal quadrant: p adj (control and RDP) = 0.0003), p adj (no DR and RDP) = 0.0048, p adj (RDNP and RDP) = 0.0054,
- Temporal quadrant: p adj (control and RDP) = 0.0417.
3.2.3. Local Adaptive Mean Thresholding
- Inferior quadrant: p adj (control and RDP) = 0.0093, p adj (no DR and RDP) = 0.0272,
- Nasal quadrant: p adj (control and RDP) = 0.0032, p adj (no DR and RDP) = 0.0045, p adj (RDNP and RDP) = 0.0100.
3.2.4. Global Mean Thresholding
3.2.5. Global Otsu Thresholding
- Inferior quadrant: p adj (control and RDP) = 0.0070,
- Nasal quadrant: p adj (control and RDP) = 0.0068.
4. Discussion
4.1. Two-Dimensional Frangi Filter and Local Versus Global Binarization Methods
4.2. Parafoveal Vessel Density of the SCP
4.3. Vessel Density in the Four Quadrants of the Inner ETDRS Grid
4.4. Limitations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| DR | diabetic retinopathy |
| NPDR | non-proliferative diabetic retinopathy |
| PDR | proliferative diabetic retinopathy |
| OCTA | optical coherence tomography angiography |
| SCP | superficial capillary plexus |
| DCP | deep capillary plexus |
| FAZ | foveal avascular zone |
| ETDRS | The Early Treatment Diabetic Retinopathy Study |
| No DR | diabetic patients without diabetic retinopathy |
| Anti-VEGF | anti-vascular endothelial growth factor |
| BCVA | best-corrected visual acuity |
| OCT | optical coherence tomography |
| PRP | panretinal photocoagulation |
References
- AbdelAl, O.; Ashraf, M.; Sampani, K.; Sun, J.K. “For Mass Eye and Ear Special Issue” Adaptive Optics in the Evaluation of Diabetic Retinopathy. Semin. Ophthalmol. 2019, 34, 189–197. [Google Scholar] [CrossRef] [PubMed]
- International Diabetes Federation. Diabetes Atlas; International Diabetes Federation: Brussels, Belgium, 2025. [Google Scholar]
- Wong, T.Y.; Cheung, C.M.; Larsen, M.; Sharma, S.; Simó, R. Diabetic retinopathy. Nat. Rev. Dis. Primers 2016, 2, 16012. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, M.; Denmead, P.; Tay, N.; Rajendram, R.; Michaelides, M.; Patterson, E. How early can we detect diabetic retinopathy? A narrative review of imaging tools for structural assessment of the retina. Graefe’s Arch. Clin. Exp. Ophthalmol. 2025, 263, 2413–2425. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Yang, D.; Tang, Z.; Ng, D.S.; Cheung, C.Y. Optical coherence tomography angiography in diabetic retinopathy: An updated review. Eye 2021, 35, 149–161. [Google Scholar] [CrossRef]
- Yu, L.; Chen, Z. Doppler variance imaging for three-dimensional retina and choroid angiography. J. Biomed. Opt. 2010, 15, 016029. [Google Scholar] [CrossRef]
- Jia, Y.; Tan, O.; Tokayer, J.; Potsaid, B.; Wang, Y.; Liu, J.J.; Kraus, M.F.; Subhash, H.; Fujimoto, J.G.; Hornegger, J.; et al. Split-spectrum amplitude-decorrelation angiography with optical coherence tomography. Opt. Express 2012, 20, 4710–4725. [Google Scholar] [CrossRef]
- Jia, Y.; Bailey, S.T.; Hwang, T.S.; McClintic, S.M.; Gao, S.S.; Pennesi, M.E.; Flaxel, C.J.; Lauer, A.K.; Wilson, D.J.; Hornegger, J.; et al. Quantitative optical coherence tomography angiography of vascular abnormalities in the living human eye. Proc. Natl. Acad. Sci. USA 2015, 112, E2395–E2402. [Google Scholar] [CrossRef]
- Nemiroff, J.; Kuehlewein, L.; Rahimy, E.; Tsui, I.; Doshi, R.; Gaudric, A.; Gorin, M.B.; Sadda, S.; Sarraf, D. Assessing Deep Retinal Capillary Ischemia in Paracentral Acute Middle Maculopathy by Optical Coherence Tomography Angiography. Am. J. Ophthalmol. 2016, 162, 121–132.e1. [Google Scholar] [CrossRef]
- Simonett, J.M.; Scarinci, F.; Picconi, F.; Giorno, P.; De Geronimo, D.; Di Renzo, A.; Varano, M.; Frontoni, S.; Parravano, M. Early microvascular retinal changes in optical coherence tomography angiography in patients with type 1 diabetes mellitus. Acta Ophthalmol. 2017, 95, e751–e755. [Google Scholar] [CrossRef]
- Ishibazawa, A.; Nagaoka, T.; Takahashi, A.; Omae, T.; Tani, T.; Sogawa, K.; Yokota, H.; Yoshida, A. Optical Coherence Tomography Angiography in Diabetic Retinopathy: A Prospective Pilot Study. Am. J. Ophthalmol. 2015, 160, 35–44.e31. [Google Scholar] [CrossRef]
- Yu, S.; Pang, C.E.; Gong, Y.; Freund, K.B.; Yannuzzi, L.A.; Rahimy, E.; Lujan, B.J.; Tabandeh, H.; Cooney, M.J.; Sarraf, D. The spectrum of superficial and deep capillary ischemia in retinal artery occlusion. Am. J. Ophthalmol. 2015, 159, 53–63.e2. [Google Scholar] [CrossRef]
- Freiberg, F.J.; Pfau, M.; Wons, J.; Wirth, M.A.; Becker, M.D.; Michels, S. Optical coherence tomography angiography of the foveal avascular zone in diabetic retinopathy. Graefe’s Arch. Clin. Exp. Ophthalmol. 2016, 254, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Hwang, T.S.; Jia, Y.; Gao, S.S.; Bailey, S.T.; Lauer, A.K.; Flaxel, C.J.; Wilson, D.J.; Huang, D. Optical coherence tomography angiography features of diabetic retinopathy. Retina 2015, 35, 2371–2376. [Google Scholar] [CrossRef] [PubMed]
- Hwang, T.S.; Gao, S.S.; Liu, L.; Lauer, A.K.; Bailey, S.T.; Flaxel, C.J.; Wilson, D.J.; Huang, D.; Jia, Y. Automated Quantification of Capillary Nonperfusion Using Optical Coherence Tomography Angiography in Diabetic Retinopathy. JAMA Ophthalmol. 2016, 134, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Maruko, I.; Arakawa, H.; Koizumi, H.; Izumi, R.; Sunagawa, H.; Iida, T. Age-Dependent Morphologic Alterations in the Outer Retinal and Choroidal Thicknesses Using Swept Source Optical Coherence Tomography. PLoS ONE 2016, 11, e0159439. [Google Scholar] [CrossRef]
- Untracht, G.R.; Matos, R.S.; Dikaios, N.; Bapir, M.; Durrani, A.K.; Butsabong, T.; Campagnolo, P.; Sampson, D.D.; Heiss, C.; Sampson, D.M. OCTAVA: An open-source toolbox for quantitative analysis of optical coherence tomography angiography images. PLoS ONE 2021, 16, e0261052. [Google Scholar] [CrossRef]
- Freedman, I.G.; Li, E.; Hui, L.; Adelman, R.A.; Nwanyanwu, K.; Wang, J.C. The Impact of Image Processing Algorithms on Optical Coherence Tomography Angiography Metrics and Study Conclusions in Diabetic Retinopathy. Transl. Vis. Sci. Technol. 2022, 11, 7. [Google Scholar] [CrossRef]
- Untracht, G.R.; Durkee, M.S.; Zhao, M.; Kwok-Cheung Lam, A.; Sikorski, B.L.; Sarunic, M.V.; Andersen, P.E.; Sampson, D.D.; Chen, F.K.; Sampson, D.M. Towards standardising retinal OCT angiography image analysis with open-source toolbox OCTAVA. Sci. Rep. 2024, 14, 5979. [Google Scholar] [CrossRef]
- Frangi, A.F.; Niessen, W.J.; Vincken, K.L.; Viergever, M.A. Multiscale vessel enhancement filtering. In Proceedings of the Medical Image Computing and Computer-Assisted Intervention—MICCAI’98, Cambridge, MA, USA, 11–13 October 1998; pp. 130–137. [Google Scholar]
- Meiburger, K.M.; Salvi, M.; Rotunno, G.; Drexler, W.; Liu, M. Automatic Segmentation and Classification Methods Using Optical Coherence Tomography Angiography (OCTA): A Review and Handbook. Appl. Sci. 2021, 11, 9734. [Google Scholar] [CrossRef]
- Chapman, B.E.; Parker, D.L. 3D multi-scale vessel enhancement filtering based on curvature measurements: Application to time-of-flight MRA. Med. Image Anal. 2005, 9, 191–208. [Google Scholar] [CrossRef]
- Hennersperger, C.; Baust, M.; Waelkens, P.; Karamalis, A.; Ahmadi, S.A.; Navab, N. Multi-scale tubular structure detection in ultrasound imaging. IEEE Trans. Med. Imaging 2015, 34, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Wang, G.; Lin, R.; Gong, X.; Song, L.; Li, T.; Wang, W.; Zhang, K.; Qian, X.; Zhang, H.; et al. Three-dimensional Hessian matrix-based quantitative vascular imaging of rat iris with optical-resolution photoacoustic microscopy in vivo. J. Biomed. Opt. 2018, 23, 046006. [Google Scholar] [CrossRef] [PubMed]
- Stefan, S.; Lee, J. Deep learning toolbox for automated enhancement, segmentation, and graphing of cortical optical coherence tomography microangiograms. Biomed. Opt. Express 2020, 11, 7325–7342. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.; Sim, R.; Chua, J.; Wong, D.W.K.; Yao, X.; Garhöfer, G.; Schmidl, D.; Werkmeister, R.M.; Schmetterer, L. Approaches to quantify optical coherence tomography angiography metrics. Ann. Transl. Med. 2020, 8, 1205. [Google Scholar] [CrossRef]
- Phansalkar, N.; More, S.; Sabale, A.; Joshi, M. Adaptive local thresholding for detection of nuclei in diversity stained cytology images. In Proceedings of the 2011 International Conference on Communications and Signal Processing, Kerala, India, 10–12 February 2011; pp. 218–220. [Google Scholar]
- Laiginhas, R.; Cabral, D.; Falcão, M. Evaluation of the different thresholding strategies for quantifying choriocapillaris using optical coherence tomography angiography. Quant. Imaging Med. Surg. 2020, 10, 1994. [Google Scholar] [CrossRef]
- Borrelli, E.; Sacconi, R.; Querques, L.; Battista, M.; Bandello, F.; Querques, G. Quantification of diabetic macular ischemia using novel three-dimensional optical coherence tomography angiography metrics. J. Biophotonics 2020, 13, e202000152. [Google Scholar] [CrossRef]
- Mehta, N.; Liu, K.; Alibhai, A.Y.; Gendelman, I.; Braun, P.X.; Ishibazawa, A.; Sorour, O.; Duker, J.S.; Waheed, N.K. Impact of binarization thresholding and brightness/contrast adjustment methodology on optical coherence tomography angiography image quantification. Am. J. Ophthalmol. 2019, 205, 54–65. [Google Scholar] [CrossRef]
- Su, L.; Ji, Y.-S.; Tong, N.; Sarraf, D.; He, X.; Sun, X.; Xu, X.; Sadda, S.R. Quantitative assessment of the retinal microvasculature and choriocapillaris in myopic patients using swept-source optical coherence tomography angiography. Graefe’s Arch. Clin. Exp. Ophthalmol. 2020, 258, 1173–1180. [Google Scholar] [CrossRef]
- Chu, Z.; Cheng, Y.; Zhang, Q.; Zhou, H.; Dai, Y.; Shi, Y.; Gregori, G.; Rosenfeld, P.J.; Wang, R.K. Quantification of choriocapillaris with phansalkar local thresholding: Pitfalls to avoid. Am. J. Ophthalmol. 2020, 213, 161–176. [Google Scholar] [CrossRef]
- Chu, Z.; Lin, J.; Gao, C.; Xin, C.; Zhang, Q.; Chen, C.-L.; Roisman, L.; Gregori, G.; Rosenfeld, P.J.; Wang, R.K. Quantitative assessment of the retinal microvasculature using optical coherence tomography angiography. J. Biomed. Opt. 2016, 21, 066008. [Google Scholar] [CrossRef]
- Kim, A.Y.; Chu, Z.; Shahidzadeh, A.; Wang, R.K.; Puliafito, C.A.; Kashani, A.H. Quantifying microvascular density and morphology in diabetic retinopathy using spectral-domain optical coherence tomography angiography. Investig. Ophthalmol. Vis. Sci. 2016, 57, OCT362–OCT370. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, H.; Cao, T.; Chen, R.; Qiu, H.; Gu, Y.; Li, P. Automatic 3D adaptive vessel segmentation based on linear relationship between intensity and complex-decorrelation in optical coherence tomography angiography. Quant. Imaging Med. Surg. 2021, 11, 895. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.; Toslak, D.; Lim, J.I.; Yao, X. OCT feature analysis guided artery-vein differentiation in OCTA. Biomed. Opt. Express 2019, 10, 2055–2066. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.; Le, D.; Lim, J.I.; Chan, R.V.P.; Yao, X. Supervised Machine Learning Based Multi-Task Artificial Intelligence Classification of Retinopathies. J. Clin. Med. 2019, 8, 872. [Google Scholar] [CrossRef] [PubMed]
- BahadarKhan, K.; Amir, A.K.; Shahid, M. A Morphological Hessian Based Approach for Retinal Blood Vessels Segmentation and Denoising Using Region Based Otsu Thresholding. PLoS ONE 2016, 11, e0158996. [Google Scholar] [CrossRef]
- Arrigo, A.; Aragona, E.; Saladino, A.; Amato, A.; Bandello, F.; Battaglia Parodi, M. The impact of different thresholds on optical coherence tomography angiography images binarization and quantitative metrics. Sci. Rep. 2021, 11, 14758. [Google Scholar] [CrossRef]
- Glasbey, C.A. An analysis of histogram-based thresholding algorithms. CVGIP Graph. Models Image Process. 1993, 55, 532–537. [Google Scholar] [CrossRef]
- Ma, Z.; Yu, F.; Li, H.; Liu, Y.; Deng, B.; Zeng, Y. Divergent nasal-temporal changes in superficial vascular density as early biomarkers of diabetic retinopathy. BMC Ophthalmol. 2026, 26, 11. [Google Scholar] [CrossRef]
- Saif, P.S.; Salman, A.E.G.; Omran, N.A.H.; Farweez, Y.A.T. Assessment of Diabetic Retinopathy Vascular Density Maps. Clin. Ophthalmol. 2020, 14, 3941–3953. [Google Scholar] [CrossRef]
- Agemy, S.A.; Scripsema, N.K.; Shah, C.M.; Chui, T.; Garcia, P.M.; Lee, J.G.; Gentile, R.C.; Hsiao, Y.-S.; Zhou, Q.; Ko, T. Retinal vascular perfusion density mapping using optical coherence tomography angiography in normals and diabetic retinopathy patients. Retina 2015, 35, 2353–2363. [Google Scholar] [CrossRef]
- Mirescu, A.E.; Deleanu, D.G.; Jurja, S.; Popa-Cherecheanu, A.; Balta, F.; Garhofer, G.; Balta, G.; Cristescu, I.E.; Tofolean, I.T. Multimodal Imaging of Diabetic Retinopathy: Insights from Optical Coherence Tomography Angiography and Adaptive Optics. Diagnostics 2025, 15, 1732. [Google Scholar] [CrossRef] [PubMed]
- Chaher, A.; Fajnkuchen, F.; Tabary, S.; Giocanti-Aurégan, A. Reduced Vessel Density in the Mid-Periphery and Peripapillary Area of the Superficial Capillary Plexus in Non-Proliferative Diabetic Retinopathy. J. Clin. Med. 2022, 11, 532. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Yeo, J.H.; Son, G.; Kang, H.; Sung, Y.S.; Lee, J.Y.; Kim, J.-G.; Yoon, Y.H. Efficacy of intravitreal AFlibercept injection For Improvement of retinal Nonperfusion In diabeTic retinopathY (AFFINITY study). BMJ Open Diabetes Res. Care 2020, 8, e001616. [Google Scholar] [CrossRef] [PubMed]
- Massengill, M.T.; Cubillos, S.; Sheth, N.; Sethi, A.; Lim, J.I. Response of diabetic macular edema to anti-VEGF medications correlates with improvement in macular vessel architecture measured with OCT angiography. Ophthalmol. Sci. 2024, 4, 100478. [Google Scholar] [CrossRef]
- Abdelhalim, A.S.; Abdelkader, M.F.S.O.; Mahmoud, M.S.E.-D.; Mohamed Mohamed, A.A. Macular vessel density before and after panretinal photocoagulation in patients with proliferative diabetic retinopathy. Int. J. Retin. Vitr. 2022, 8, 21. [Google Scholar] [CrossRef]
- Chatziralli, I.; Dimitriou, E.; Agapitou, C.; Kazantzis, D.; Kapsis, P.; Morogiannis, N.; Kandarakis, S.; Theodossiadis, G.; Theodossiadis, P. Optical Coherence Tomography Angiography Changes in Macular Area in Patients with Proliferative Diabetic Retinopathy Treated with Panretinal Photocoagulation. Biomedicines 2023, 11, 3146. [Google Scholar] [CrossRef]
- Fawzi, A.A.; Fayed, A.E.; Linsenmeier, R.A.; Gao, J.; Yu, F. Improved macular capillary flow on optical coherence tomography angiography after panretinal photocoagulation for proliferative diabetic retinopathy. Am. J. Ophthalmol. 2019, 206, 217–227. [Google Scholar] [CrossRef]
- Takahashi, A.; Nagaoka, T.; Sato, E.; Yoshida, A. Effect of panretinal photocoagulation on choroidal circulation in the foveal region in patients with severe diabetic retinopathy. Br. J. Ophthalmol. 2008, 92, 1369–1373. [Google Scholar] [CrossRef]
- Zhao, T.; Chen, Y.; Liu, D.; Stewart, J.M. Optical coherence tomography angiography assessment of macular choriocapillaris and choroid following panretinal photocoagulation in a diverse population with advanced diabetic retinopathy. Asia-Pac. J. Ophthalmol. 2021, 10, 203–207. [Google Scholar] [CrossRef]
- Petrou Sr, P.; Angelidis, C.D.; Andreanos, K.; Kanakis, M.; Kandarakis, S.; Karamaounas, A.; Papakonstantinou, E.; Mamas, N.; Droutsas, K.; Georgalas, I. Reduction of foveal avascular zone after vitrectomy demonstrated by optical coherence tomography angiography. Cureus 2021, 13, e13757. [Google Scholar] [CrossRef]
- Russell, J.F.; Scott, N.L.; Townsend, J.H.; Shi, Y.; Gregori, G.; Crane, A.M.; Flynn, H.W., Jr.; Sridhar, J.; Rosenfeld, P.J. Wide-field swept-source optical coherence tomography angiography of diabetic tractional retinal detachments before and after surgical repair. Retina 2021, 41, 1587–1596. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mirescu, A.-E.; Tofolean, I.T.; Jurja, S.; Balta, F.; Popa-Cherecheanu, A.; Pirvulescu, R.A.; Garhofer, G.; Balta, G.; Cristescu, I.-E.; Deleanu, D.G. Local Versus Global Binarization Techniques After Frangi Filtering for Optical Coherence Tomography Angiography Based Retinal Vessel Density Assessment in Diabetic Retinopathy. Diagnostics 2026, 16, 934. https://doi.org/10.3390/diagnostics16060934
Mirescu A-E, Tofolean IT, Jurja S, Balta F, Popa-Cherecheanu A, Pirvulescu RA, Garhofer G, Balta G, Cristescu I-E, Deleanu DG. Local Versus Global Binarization Techniques After Frangi Filtering for Optical Coherence Tomography Angiography Based Retinal Vessel Density Assessment in Diabetic Retinopathy. Diagnostics. 2026; 16(6):934. https://doi.org/10.3390/diagnostics16060934
Chicago/Turabian StyleMirescu, Andrada-Elena, Ioana Teodora Tofolean, Sanda Jurja, Florian Balta, Alina Popa-Cherecheanu, Ruxandra Angela Pirvulescu, Gerhard Garhofer, George Balta, Irina-Elena Cristescu, and Dan George Deleanu. 2026. "Local Versus Global Binarization Techniques After Frangi Filtering for Optical Coherence Tomography Angiography Based Retinal Vessel Density Assessment in Diabetic Retinopathy" Diagnostics 16, no. 6: 934. https://doi.org/10.3390/diagnostics16060934
APA StyleMirescu, A.-E., Tofolean, I. T., Jurja, S., Balta, F., Popa-Cherecheanu, A., Pirvulescu, R. A., Garhofer, G., Balta, G., Cristescu, I.-E., & Deleanu, D. G. (2026). Local Versus Global Binarization Techniques After Frangi Filtering for Optical Coherence Tomography Angiography Based Retinal Vessel Density Assessment in Diabetic Retinopathy. Diagnostics, 16(6), 934. https://doi.org/10.3390/diagnostics16060934





