Prognostic Value of Blood Urea Nitrogen for Acute Kidney Injury and Mortality in Vasculitis: A Large Cohort Study Using Multivariate Joint Model and Machine Learning
Abstract
1. Introduction
2. Methods
2.1. Data Source
2.2. Study Population
2.3. Data Extraction and Definitions
2.4. Statistical Analysis
2.5. Machine Learning
3. Results
3.1. Baseline Characteristics
3.2. Relationship Between Vasculitis and BUN Levels and Their Association with AKI Incidence
3.3. Association of BUN Levels and Risk Factors with Short- and Long-Term Mortality in Vasculitis Patients with AKI
3.4. Nonlinear Analyses
3.5. KM Survival Analyses
3.6. Time-Dependent AUC Curve
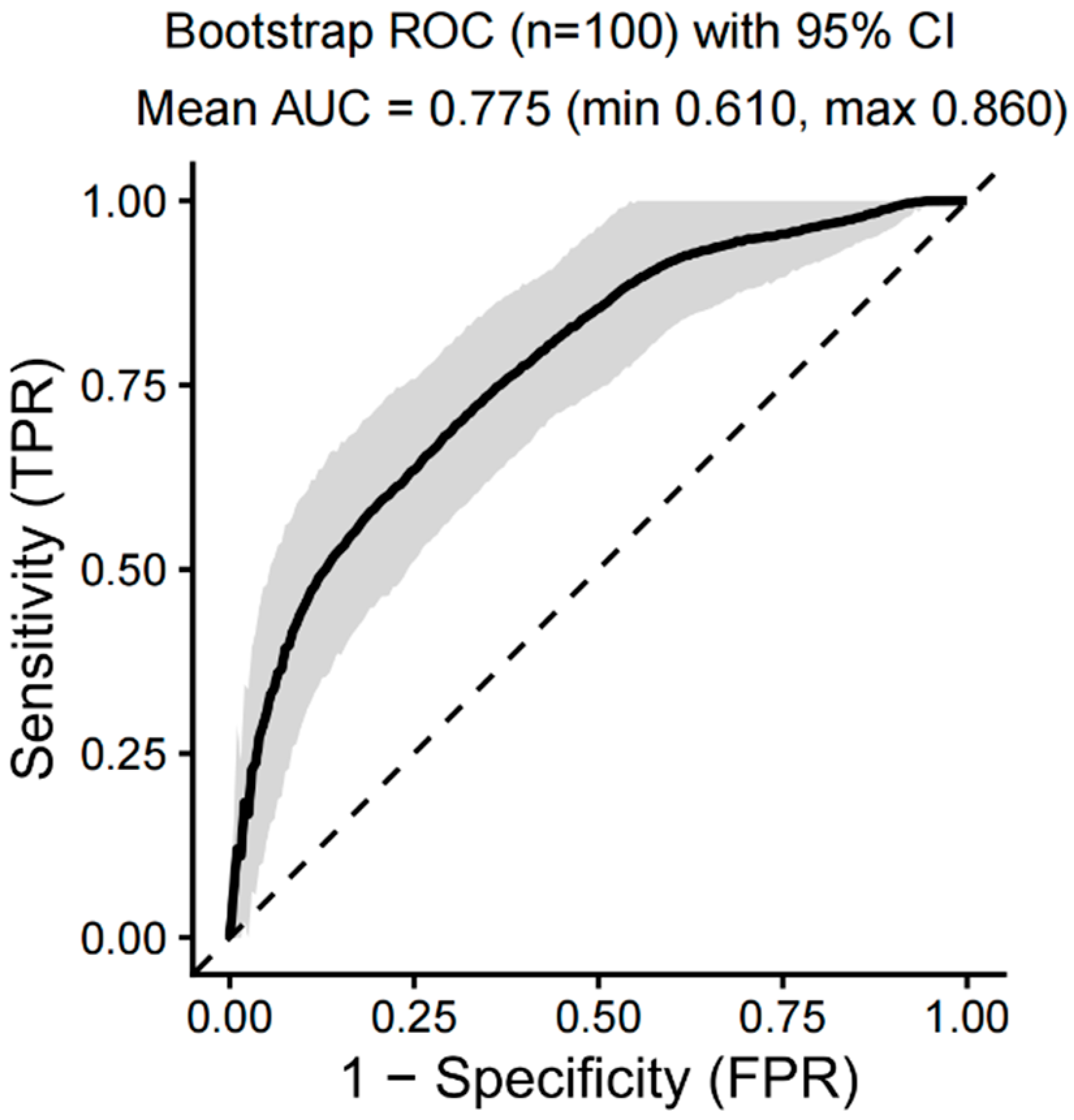
3.7. The Results of Machine Learning
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jennette, J.C.; Falk, R.J.; Bacon, P.A.; Basu, N.; Cid, M.C.; Ferrario, F.; Flores-Suarez, L.F.; Gross, W.L.; Guillevin, L.; Hagen, E.C.; et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013, 65, 1–11. [Google Scholar] [CrossRef]
- John, R.; Herzenberg, A.M. Vasculitis affecting the kidney. Semin. Diagn. Pathol. 2009, 26, 89–102. [Google Scholar] [CrossRef]
- Klein, R.G.; Hunder, G.G.; Stanson, A.W.; Sheps, S.G. Large artery involvement in giant cell (temporal) arteritis. Ann. Intern. Med. 1975, 83, 806–812. [Google Scholar] [CrossRef]
- Jennette, J.C.; Falk, R.J. The pathology of vasculitis involving the kidney. Am. J. Kidney Dis. 1994, 24, 130–141. [Google Scholar] [CrossRef]
- Huang, X.; Chen, L.; Lan, L.; Ren, P.; Ni, A.; Ma, Y.; Wang, Y.; Zhu, Y.; Xu, Y.; Chen, J.; et al. Antineutrophil Cytoplasmic Antibody-Associated Vasculitis with Acute Kidney Injury: Short-Term Recovery Predicts Long-Term Outcome. Front. Immunol. 2021, 12, 641655. [Google Scholar] [CrossRef]
- Booth, A.D.; Almond, M.K.; Burns, A.; Ellis, P.; Gaskin, G.; Neild, G.H.; Plaisance, M.; Pusey, C.D.; Jayne, D.R.; Pan-Thames Renal Research, G. Outcome of ANCA-associated renal vasculitis: A 5-year retrospective study. Am. J. Kidney Dis. 2003, 41, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, K.; Miyayama, S.; Ushiogi, Y.; Matsui, O. Renal involvement of polyarteritis nodosa: CT and MR findings. Abdom. Imaging 2009, 34, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Medvedev, G.; Al-Shamari, A.E.; Copland, M.A.; Magil, A.B. Isolated renal giant cell arteritis. Am. J. Kidney Dis. 2002, 40, 658–661. [Google Scholar] [CrossRef] [PubMed]
- Akpolat, T.; Dilek, M.; Aksu, K.; Keser, G.; Toprak, O.; Cirit, M.; Oguz, Y.; Taskapan, H.; Adibelli, Z.; Akar, H.; et al. Renal Behcet’s disease: An update. Semin. Arthritis Rheum. 2008, 38, 241–248. [Google Scholar] [CrossRef]
- Zheng, W.; Li, G.; Zhou, M.; Chen, L.; Tian, X.; Zhang, F. Renal involvement in Chinese patients with Behcet’s disease: A report of 16 cases. Int. J. Rheum. Dis. 2015, 18, 892–897. [Google Scholar] [CrossRef]
- Ardalan, M.R.; Sadreddini, S.; Noshad, H.; Ebrahimi, A.; Molaeefard, M.; Somi, M.H.; Shoja, M.M. Renal involvement in Behcet’s disease. Saudi J. Kidney Dis. Transpl. 2009, 20, 618–622. [Google Scholar]
- Stevens, L.A.; Coresh, J.; Greene, T.; Levey, A.S. Assessing kidney function--measured and estimated glomerular filtration rate. N. Engl. J. Med. 2006, 354, 2473–2483. [Google Scholar] [CrossRef]
- Waikar, S.S.; Bonventre, J.V. Creatinine kinetics and the definition of acute kidney injury. J. Am. Soc. Nephrol. 2009, 20, 672–679. [Google Scholar] [CrossRef]
- Shemesh, O.; Golbetz, H.; Kriss, J.P.; Myers, B.D. Limitations of creatinine as a filtration marker in glomerulopathic patients. Kidney Int. 1985, 28, 830–838. [Google Scholar] [CrossRef]
- Doi, K.; Yuen, P.S.; Eisner, C.; Hu, X.; Leelahavanichkul, A.; Schnermann, J.; Star, R.A. Reduced production of creatinine limits its use as marker of kidney injury in sepsis. J. Am. Soc. Nephrol. 2009, 20, 1217–1221. [Google Scholar] [CrossRef] [PubMed]
- Aragane, M.; Matsumoto, L.; Yasuda, H.; Nosaka, M.; Ishida, Y.; Yamamoto, H.; Kondo, T.; Ishigami, A. An autopsy case of fatal Japanese spotted fever in Wakayama. Leg. Med. 2024, 66, 102355. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Han, S.; Liang, F.; Hu, C.; Zhang, B.; Hou, Q.; Zhao, S. Machine Learning for Predicting Risk and Prognosis of Acute Kidney Disease in Critically Ill Elderly Patients During Hospitalization: Internet-Based and Interpretable Model Study. J. Med. Internet Res. 2024, 26, e51354. [Google Scholar] [CrossRef]
- Chiang, C.H.; Lan, T.Y.; Hsieh, J.H.; Lin, S.C.; Chen, J.W.; Chang, T.T. Diosgenin Reduces Acute Kidney Injury and Ameliorates the Progression to Chronic Kidney Disease by Modifying the NOX4/p65 Signaling Pathways. J. Agric. Food Chem. 2024, 72, 17444–17454. [Google Scholar] [CrossRef]
- Kodger, J.; Durant, T.J.S.; Yurtsever, N.; El-Khoury, J.M. Validating CALIPER pediatric reference intervals in a U.S. population using retrospective outpatient data and RefineR. Clin. Chim. Acta 2026, 584, 120846. [Google Scholar] [CrossRef]
- Du, R.; Wang, L.; Wang, Y.; Zhao, Z.; Zhang, D.; Zuo, S. AKI prediction model in acute aortic dissection surgery: Nomogram development and validation. Front. Med. 2025, 12, 1562956. [Google Scholar] [CrossRef] [PubMed]
- Lincoln, M.; McGimsey, T.; O’Driscoll, D. Rituximab in the Intensive Care Unit: A Review of Indications and Clinical Considerations. J. Intensive Care Med. 2025; in press. [Google Scholar] [CrossRef]
- Yokota, K.; Inoue, T.; Akiyama, Y.; Kajiyama, H.; Funakubo Asanuma, Y.; Arai, E.; Suzuki, H.; Mimura, T. Acute kidney injury in a patient with polyarteritis nodosa and multiple myeloma. Intern. Med. 2014, 53, 263–267. [Google Scholar] [CrossRef]
- Akpolat, T.; Diri, B.; Oguz, Y.; Yilmaz, E.; Yavuz, M.; Dilek, M. Behcet’s disease and renal failure. Nephrol. Dial. Transplant. 2003, 18, 888–891. [Google Scholar] [CrossRef]
- Bacani, R.A.; Velasquez, F.; Kanter, A.; Pirani, C.L.; Pollak, V.E. Rapidly progressive (nonstreptococcal) glomerulonephritis. Ann. Intern. Med. 1968, 69, 463–485. [Google Scholar] [CrossRef]
- Mohamadlou, H.; Lynn-Palevsky, A.; Barton, C.; Chettipally, U.; Shieh, L.; Calvert, J.; Saber, N.R.; Das, R. Prediction of Acute Kidney Injury with a Machine Learning Algorithm Using Electronic Health Record Data. Can. J. Kidney Health Dis. 2018, 5, 2054358118776326. [Google Scholar] [CrossRef]
- Tomasev, N.; Glorot, X.; Rae, J.W.; Zielinski, M.; Askham, H.; Saraiva, A.; Mottram, A.; Meyer, C.; Ravuri, S.; Protsyuk, I.; et al. A clinically applicable approach to continuous prediction of future acute kidney injury. Nature 2019, 572, 116–119. [Google Scholar] [CrossRef]
- Johnson, A.E.; Pollard, T.J.; Shen, L.; Lehman, L.W.; Feng, M.; Ghassemi, M.; Moody, B.; Szolovits, P.; Celi, L.A.; Mark, R.G. MIMIC-III, a freely accessible critical care database. Sci. Data 2016, 3, 160035. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.E.W.; Bulgarelli, L.; Shen, L.; Gayles, A.; Shammout, A.; Horng, S.; Pollard, T.J.; Hao, S.; Moody, B.; Gow, B.; et al. MIMIC-IV, a freely accessible electronic health record dataset. Sci. Data 2023, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.; Pollard, T.; Mark, R. MIMIC-III Clinical Database (version 1.4). PhysioNet 2016, RRID:SCR_007345. [Google Scholar] [CrossRef]
- Johnson, A.; Bulgarelli, L.; Pollard, T.; Gow, B.; Moody, B.; Horng, S.; Celi, L.A.; Mark, R. MIMIC-IV (version 3.1). PhysioNet 2024, RRID:SCR_007345. [Google Scholar] [CrossRef]
- Team DC. DecisionLinnc. 1.0. 2023. Available online: https://www.statsape.com/ (accessed on 18 February 2026).
- Tao, J.; Liu, S.; Montez-Rath, M.; Charu, V.; Chertow, G.M. Antineutrophil Cytoplasmic Antibody-Associated Vasculitis with Active Kidney Involvement in the United States: 2016–2020. Glomerular Dis. 2024, 4, 33–42. [Google Scholar]
- Chen, Y.; Lu, Y.; Zhou, Y.; Yu, C. Clinical Features and Prognosis in ANCA Associated Vasculitis Patients with Acute Kidney Injury. Arch. Iran. Med. 2022, 25, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Ahn, S.H. Granulomatosis with Polyangiitis Presenting as Cholangitis and Acute Kidney Injury. Chonnam Med. J. 2019, 55, 124–125. [Google Scholar] [CrossRef]
- El Madhoun, I.; Warnock, N.G.; Roy, A.; Jones, C.H. Bilateral renal hemorrhage due to polyarteritis nodosa wrongly attributed to blunt trauma. Nat. Rev. Urol. 2009, 6, 563–567. [Google Scholar] [CrossRef]
- Elling, H.; Kristensen, I.B. Fatal renal failure in polymyalgia rheumatica caused by disseminated giant cell arteritis. Scand. J. Rheumatol. 1980, 9, 206–208. [Google Scholar] [CrossRef] [PubMed]
- Candan, C.; Civilibal, M.; Oktay, G.; Canpolat, N.; Caliskan, S.; Kilicaslan, I.; Sever, L.; Kasapcopur, O.; Arisoy, N. Classic polyarteritis nodosa presenting with acute anuric renal failure. Pediatr. Int. 2010, 52, e76–e78. [Google Scholar] [CrossRef] [PubMed]
- Lima, C.; Macedo, E. Biomarkers in acute kidney injury and cirrhosis. J. Transl. Crit. Care Med. 2024, 6, e23-0014. [Google Scholar] [CrossRef]
- Endre, Z.H.; Westhuyzen, J. Early detection of acute kidney injury: Emerging new biomarkers. Nephrology 2008, 13, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Murty, M.S.; Sharma, U.K.; Pandey, V.B.; Kankare, S.B. Serum cystatin C as a marker of renal function in detection of early acute kidney injury. Indian J. Nephrol. 2013, 23, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Neha, F.; Bansal, A.K. Convnext-pca: A parameter-efficient model for accurate kidney abnormality classification. In Proceedings of the 2024 IEEE 34th International Workshop on Machine Learning for Signal Processing (MLSP), London, UK, 22–25 September 2024; pp. 1–6. [Google Scholar]





| Variable | Overall (n = 701) | Without AKI (n = 525) | AKI (n = 176) | p-Value |
|---|---|---|---|---|
| Age, years | 75.76 (65.00–83.00) | 76.00 (66.00–83.00) | 74.00 (64.00–83.00) | 0.4 |
| Gender, n (%) | 0.004 | |||
| Female | 445.00 (63.48%) | 349.00 (66.48%) | 96.00 (54.55%) | |
| Male | 256.00 (36.52%) | 176.00 (33.52%) | 80.00 (45.45%) | |
| LOS of hospital, day | 4.66 (2.50–7.89) | 3.88 (2.15–6.95) | 7.03 (3.87–13.00) | <0.001 |
| Hospital mortality, n (%) | <0.001 | |||
| No | 668.00 (95.29%) | 512.00 (97.52%) | 156.00 (88.64%) | |
| Yes | 33.00 (4.71%) | 13.00 (2.48%) | 20.00 (11.36%) | |
| 30-day mortality, n (%) | <0.001 | |||
| No | 660.00 (94.15%) | 504.00 (96.00%) | 156.00 (88.64%) | |
| Yes | 41.00 (5.85%) | 21.00 (4.00%) | 20.00 (11.36%) | |
| 60-day mortality, n (%) | 0.001 | |||
| No | 639.00 (91.16%) | 489.00 (93.14%) | 150.00 (85.23%) | |
| Yes | 62.00 (8.84%) | 36.00 (6.86%) | 26.00 (14.77%) | |
| 90-day mortality, n (%) | <0.001 | |||
| No | 626.00 (89.30%) | 481.00 (91.62%) | 145.00 (82.39%) | |
| Yes | 75.00 (10.70%) | 44.00 (8.38%) | 31.00 (17.61%) | |
| 180-day mortality, n (%) | <0.001 | |||
| No | 602.00 (85.88%) | 466.00 (88.76%) | 136.00 (77.27%) | |
| Yes | 99.00 (14.12%) | 59.00 (11.24%) | 40.00 (22.73%) | |
| 365-day mortality, n (%) | 0.002 | |||
| No | 565.00 (80.60%) | 437.00 (83.24%) | 128.00 (72.73%) | |
| Yes | 136.00 (19.40%) | 88.00 (16.76%) | 48.00 (27.27%) | |
| Hypertension, n (%) | 0.015 | |||
| No | 407.00 (58.06%) | 291.00 (55.43%) | 116.00 (65.91%) | |
| Yes | 294.00 (41.94%) | 234.00 (44.57%) | 60.00 (34.09%) | |
| Heart failure, n (%) | 0.047 | |||
| No | 540.00 (77.03%) | 414.00 (78.86%) | 126.00 (71.59%) | |
| Yes | 161.00 (22.97%) | 111.00 (21.14%) | 50.00 (28.41%) | |
| Myocardial infarction, n (%) | 0.46 | |||
| No | 686.00 (97.86%) | 515.00 (98.10%) | 171.00 (97.16%) | |
| Yes | 15.00 (2.14%) | 10.00 (1.90%) | 5.00 (2.84%) | |
| Stroke, n (%) | 0.22 | |||
| No | 637.00 (90.87%) | 473.00 (90.10%) | 164.00 (93.18%) | |
| Yes | 64.00 (9.13%) | 52.00 (9.90%) | 12.00 (6.82%) | |
| Immunosuppressants, n (%) | 0.021 | |||
| No | 626.00 (89.30%) | 477.00 (90.86%) | 149.00 (84.66%) | |
| Yes | 75.00 (10.70%) | 48.00 (9.14%) | 27.00 (15.34%) | |
| GCs, n (%) | 0.72 | |||
| No | 303.00 (43.22%) | 229.00 (43.62%) | 74.00 (42.05%) | |
| Yes | 398.00 (56.78%) | 296.00 (56.38%) | 102.00 (57.95%) | |
| Monoclonal antibody agent, n (%) | <0.001 | |||
| No | 664.00 (94.72%) | 509.00 (96.95%) | 155.00 (88.07%) | |
| Yes | 37.00 (5.28%) | 16.00 (3.05%) | 21.00 (11.93%) | |
| Diuretics, n (%) | 0.007 | |||
| No | 430.00 (61.34%) | 337.00 (64.19%) | 93.00 (52.84%) | |
| Yes | 271.00 (38.66%) | 188.00 (35.81%) | 83.00 (47.16%) | |
| White blood cell, K/µL | 9.00 (6.60–12.10) | 8.60 (6.50–11.60) | 9.50 (6.75–12.30) | 0.032 |
| Hemoglobin, g/dL | 10.70 (9.30–12.10) | 10.90 (9.70–12.20) | 10.05 (8.65–11.50) | <0.001 |
| Platelet, K/µL | 239.64 (182.00–311.00) | 244.00 (185.00–321.00) | 235.50 (168.50–287.00) | 0.023 |
| Anion gap, mmol/L | 14.00 (12.00–16.00) | 14.00 (12.00–16.00) | 15.00 (13.00–18.00) | <0.001 |
| Total calcium, mmol/L | 8.70 (8.30–9.20) | 8.80 (8.30–9.20) | 8.60 (8.05–9.10) | <0.001 |
| Chloride, mmol/L | 103.00 (100.00–106.00) | 103.00 (99.00–106.00) | 103.00 (100.00–107.00) | 0.092 |
| Glucose, mg/dL | 109.00 (90.00–148.00) | 109.00 (90.00–147.00) | 115.00 (91.00–153.50) | 0.44 |
| Potassium, mmol/L | 4.10 (3.80–4.50) | 4.00 (3.70–4.40) | 4.30 (3.90–4.90) | <0.001 |
| Sodium, mmol/L | 139.00 (136.00–141.00) | 139.00 (137.00–141.00) | 138.00 (136.00–141.00) | 0.018 |
| Cr, mg/dL | 1.00 (0.70–1.50) | 0.90 (0.70–1.10) | 1.65 (1.20–2.85) | <0.001 |
| BUN, mg/dL | 21.00 (14.00–32.00) | 18.00 (13.00–26.00) | 32.50 (22.50–58.00) | <0.001 |
| Variable | Overall (n = 176) | 30-Day Mortality | 365-Day Mortality | ||||
|---|---|---|---|---|---|---|---|
| Survivors (n = 156) | Non-Survivors (n = 20) | p-Value | Survivors (n = 128) | Non-Survivors (n = 48) | p-Value | ||
| Age, years | 74.00 (64.00–83.00) | 73.00 (63.00–81.01) | 81.00 (72.99–86.31) | 0.011 | 71.00 (60.56–79.32) | 81.50 (71.67–87.58) | <0.001 |
| Gender, n (%) | 0.97 | 0.16 | |||||
| Female | 96.00 (54.55%) | 85.00 (54.49%) | 11.00 (55.00%) | 74.00 (57.81%) | 22.00 (45.83%) | ||
| Male | 80.00 (45.45%) | 71.00 (45.51%) | 9.00 (45.00%) | 54.00 (42.19%) | 26.00 (54.17%) | ||
| LOS of hospital, day | 7.03 (3.87–13.00) | 6.87 (3.90–13.28) | 7.79 (3.54–12.54) | 0.91 | 6.54 (3.42–12.24) | 9.68 (4.47–15.68) | 0.038 |
| Hypertension, n (%) | 0.93 | 0.4 | |||||
| No | 116.00 (65.91%) | 103.00 (66.03%) | 13.00 (65.00%) | 82.00 (64.06%) | 34.00 (70.83%) | ||
| Yes | 60.00 (34.09%) | 53.00 (33.97%) | 7.00 (35.00%) | 46.00 (35.94%) | 14.00 (29.17%) | ||
| Heart failure, n (%) | 0.22 | 0.017 | |||||
| No | 126.00 (71.59%) | 114.00 (73.08%) | 12.00 (60.00%) | 98.00 (76.56%) | 28.00 (58.33%) | ||
| Yes | 50.00 (28.41%) | 42.00 (26.92%) | 8.00 (40.00%) | 30.00 (23.44%) | 20.00 (41.67%) | ||
| Myocardial infarction, n (%) | 0.54 | 0.52 | |||||
| No | 171.00 (97.16%) | 152.00 (97.44%) | 19.00 (95.00%) | 125.00 (97.66%) | 46.00 (95.83%) | ||
| Yes | 5.00 (2.84%) | 4.00 (2.56%) | 1.00 (5.00%) | 3.00 (2.34%) | 2.00 (4.17%) | ||
| Stroke, n (%) | 0.55 | 0.012 | |||||
| No | 164.00 (93.18%) | 146.00 (93.59%) | 18.00 (90.00%) | 123.00 (96.09%) | 41.00 (85.42%) | ||
| Yes | 12.00 (6.82%) | 10.00 (6.41%) | 2.00 (10.00%) | 5.00 (3.91%) | 7.00 (14.58%) | ||
| Immunosuppressants, n (%) | 0.2 | 0.44 | |||||
| No | 149.00 (84.66%) | 134.00 (85.90%) | 15.00 (75.00%) | 110.00 (85.94%) | 39.00 (81.25%) | ||
| Yes | 27.00 (15.34%) | 22.00 (14.10%) | 5.00 (25.00%) | 18.00 (14.06%) | 9.00 (18.75%) | ||
| GCs, n (%) | 0.78 | 0.15 | |||||
| No | 74.00 (42.05%) | 65.00 (41.67%) | 9.00 (45.00%) | 58.00 (45.31%) | 16.00 (33.33%) | ||
| Yes | 102.00 (57.95%) | 91.00 (58.33%) | 11.00 (55.00%) | 70.00 (54.69%) | 32.00 (66.67%) | ||
| Monoclonal antibody agent, n (%) | 0.31 | 0.89 | |||||
| No | 155.00 (88.07%) | 136.00 (87.18%) | 19.00 (95.00%) | 113.00 (88.28%) | 42.00 (87.50%) | ||
| Yes | 21.00 (11.93%) | 20.00 (12.82%) | 1.00 (5.00%) | 15.00 (11.72%) | 6.00 (12.50%) | ||
| Diuretics, n (%) | 0.002 | 0.005 | |||||
| No | 93.00 (52.84%) | 89.00 (57.05%) | 4.00 (20.00%) | 76.00 (59.38%) | 17.00 (35.42%) | ||
| Yes | 83.00 (47.16%) | 67.00 (42.95%) | 16.00 (80.00%) | 52.00 (40.63%) | 31.00 (64.58%) | ||
| White blood cell, K/µL | 9.50 (6.75–12.30) | 9.40 (6.70–12.05) | 13.45 (9.95–26.90) | 0.003 | 9.35 (6.55–11.75) | 11.35 (7.95–16.10) | 0.006 |
| Hemoglobin, g/dL | 10.05 (8.65–11.50) | 10.15 (8.80–11.75) | 8.75 (7.95–9.30) | 0.002 | 10.25 (9.00–11.80) | 9.00 (7.95–10.55) | 0.001 |
| Platelet, K/µL | 235.50 (168.50–287.00) | 236.00 (179.00–291.50) | 171.50 (127.50–242.00) | 0.011 | 236.00 (180.00–284.00) | 221.00 (144.00–297.00) | 0.19 |
| Anion gap, mmol/L | 15.00 (13.00–18.00) | 15.00 (12.00–17.00) | 17.00 (14.00–20.00) | 0.014 | 14.00 (12.00–17.00) | 16.00 (13.00–18.50) | 0.025 |
| Total calcium, mmol/L | 8.60 (8.05–9.10) | 8.60 (8.00–9.10) | 8.35 (8.10–8.75) | 0.4 | 8.60 (8.10–9.10) | 8.40 (8.00–8.95) | 0.38 |
| Chloride, mmol/L | 103.00 (100.00–107.00) | 103.00 (100.00–107.00) | 104.50 (99.00–107.50) | 0.83 | 103.50 (101.00–107.00) | 103.00 (98.50–106.00) | 0.33 |
| Glucose, mg/dL | 115.00 (91.00–153.50) | 115.50 (91.50–151.00) | 108.50 (84.50–160.00) | 0.88 | 116.00 (90.00–155.00) | 106.50 (96.50–144.50) | 0.68 |
| Potassium, mmol/L | 4.30 (3.90–4.90) | 4.30 (3.90–4.90) | 4.60 (4.10–5.00) | 0.14 | 4.30 (3.80–4.90) | 4.30 (4.00–4.85) | 0.56 |
| Sodium, mmol/L | 138.00 (136.00–141.00) | 138.00 (136.00–140.50) | 140.50 (135.50–143.00) | 0.13 | 138.00 (136.00–140.50) | 138.50 (136.00–141.00) | 0.39 |
| Cr, mg/dL | 1.65 (1.20–2.85) | 1.60 (1.15–2.55) | 2.85 (1.40–3.95) | 0.033 | 1.55 (1.15–2.30) | 2.45 (1.20–3.95) | 0.018 |
| BUN, mg/dL | 32.50 (22.50–58.00) | 31.50 (22.00–51.00) | 65.00 (45.50–109.00) | <0.001 | 29.50 (21.00–48.00) | 52.00 (28.00–87.00) | <0.001 |
| Variables | Multivariate Logistic Analysis | LASSO-Logistic Analysis | ||
|---|---|---|---|---|
| ORs (95% CIs) | p-Value | ORs (95% CIs) | p-Value | |
| Gender | 1.54 (1.00–2.36) | 0.047 | ||
| Monoclonal antibody agent | 3.68 (1.62–8.44) | 0.002 | 3.66 (1.62–8.30) | 0.002 |
| Chloride | 1.10 (1.04–1.17) | 0.003 | 1.10 (1.04–1.16) | 0.002 |
| Potassium | 1.60 (1.16–2.24) | 0.005 | 1.61 (1.16–2.24) | 0.004 |
| Sodium | 0.91 (0.84–0.98) | 0.01 | 0.92 (0.86–0.99) | 0.02 |
| BUN | 1.03 (1.02–1.05) | <0.0001 | 1.03 (1.02–1.04) | <0.0001 |
| Model 1 | Model 2 | Model 3 | |||||
|---|---|---|---|---|---|---|---|
| ORs (95% CIs) | p-Value | ORs (95% CIs) | p-Value | ORs (95% CIs) | p-Value | ||
| BUN | 1.04 (1.03–1.05) | <0.0001 | 1.04 (1.03–1.05) | <0.0001 | 1.03 (1.02–1.05) | <0.0001 | |
| BUN (tertile) | |||||||
| Q1 | 12 (10–14) | reference | reference | reference | |||
| Q2 | 21 (19–23) | 2.57 (1.49–4.59) | 0.0009 | 2.72 (1.55–4.92) | 0.0006 | 2.08 (1.15–3.87) | 0.02 |
| Q3 | 44.5 (32–67.5) | 10.24 (6.17–17.78) | <0.0001 | 10.37 (6.12–18.36) | <0.0001 | 5.67 (3.00–11.04) | <0.0001 |
| p for trend | <0.0001 | <0.0001 | <0.0001 | ||||
| Variables | Multivariate Cox Analysis | LASSO-Cox Analysis | ||
|---|---|---|---|---|
| HRs (95% CIs) | p-Value | HRs (95% CIs) | p-Value | |
| 30-day mortality | ||||
| Age | 1.12 (1.05–1.21) | 0.001 | 1.10 (1.04–1.17) | 0.002 |
| Diuretics | 5.17 (1.24–21.50) | 0.024 | ||
| White blood cell | 1.08 (1.02–1.14) | 0.009 | 1.08 (1.04–1.13) | <0.0001 |
| Hemoglobin | 0.73 (0.56–0.97) | 0.03 | ||
| BUN | 1.03 (1.01–1.05) | 0.005 | 1.02 (1.01–1.04) | <0.0001 |
| 365-day mortality | ||||
| Age | 1.08 (1.04–1.12) | <0.0001 | 1.08 (1.05–1.12) | <0.0001 |
| Diuretics | 2.01 (1.09–3.71) | 0.025 | ||
| White blood cell | 1.06 (1.02–1.10) | 0.002 | 1.05 (1.02–1.08) | <0.0001 |
| Hemoglobin | 0.78 (0.64–0.95) | 0.013 | 0.79 (0.67–0.93) | 0.005 |
| BUN | 1.02 (1.00–1.03) | 0.008 | 1.02 (1.00–1.02) | <0.0001 |
| Model 1 | Model 2 | Model 3 | |||||
|---|---|---|---|---|---|---|---|
| ORs (95% CIs) | p-Value | ORs (95% CIs) | p-Value | ORs (95% CIs) | p-Value | ||
| 30-day mortality | |||||||
| BUN | 1.02 (1.01–1.03) | 0.001 | 1.03 (1.01–1.04) | 0.0002 | 1.03 (1.01–1.04) | 0.0002 | |
| BUN (tertile) | |||||||
| Q1 | 20 (16–23) | reference | reference | reference | |||
| Q2 | 35 (29.5–41) | 0.75 (0.10–4.72) | 0.76 | 0.67 (0.08–4.27) | 0.67 | 0.62 (0.08–4.04) | 0.62 |
| Q3 | 77 (60.25–102) | 6.98 (2.14–31.47) | 0.0034 | 8.14 (2.36–38.50) | 0.0024 | 8.30 (2.33–40.56) | 0.0028 |
| p for trend | 0.001 | 0.0008 | 0.0009 | ||||
| 365-day mortality | |||||||
| BUN | 1.02 (1.01–1.03) | 0.0011 | 1.02 (1.01–1.03) | 0.0001 | 1.02 (1.01–1.03) | 0.0002 | |
| BUN (tertile) | |||||||
| Q1 | 20 (16–23) | reference | reference | reference | |||
| Q2 | 35 (29.5–41) | 1.33 (0.51–3.46) | 0.56 | 1.24 (0.45–3.47) | 0.67 | 1.24 (0.44–3.55) | 0.69 |
| Q3 | 77 (60.25–102) | 4.62 (2.02–11.22) | 0.0001 | 6.34 (2.50–17.65) | 0.0002 | 6.12 (2.37–17.34) | 0.0003 |
| p for trend | 0.0003 | 0.0001 | 0.0002 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Chen, S.; Liu, R.; Zhang, Y.; Wang, Y.; Luan, H.; Zeng, X.; Yuan, H. Prognostic Value of Blood Urea Nitrogen for Acute Kidney Injury and Mortality in Vasculitis: A Large Cohort Study Using Multivariate Joint Model and Machine Learning. Diagnostics 2026, 16, 665. https://doi.org/10.3390/diagnostics16050665
Chen S, Liu R, Zhang Y, Wang Y, Luan H, Zeng X, Yuan H. Prognostic Value of Blood Urea Nitrogen for Acute Kidney Injury and Mortality in Vasculitis: A Large Cohort Study Using Multivariate Joint Model and Machine Learning. Diagnostics. 2026; 16(5):665. https://doi.org/10.3390/diagnostics16050665
Chicago/Turabian StyleChen, Si, Rongfeng Liu, Yongzhi Zhang, Yan Wang, Haixia Luan, Xiaoli Zeng, and Hui Yuan. 2026. "Prognostic Value of Blood Urea Nitrogen for Acute Kidney Injury and Mortality in Vasculitis: A Large Cohort Study Using Multivariate Joint Model and Machine Learning" Diagnostics 16, no. 5: 665. https://doi.org/10.3390/diagnostics16050665
APA StyleChen, S., Liu, R., Zhang, Y., Wang, Y., Luan, H., Zeng, X., & Yuan, H. (2026). Prognostic Value of Blood Urea Nitrogen for Acute Kidney Injury and Mortality in Vasculitis: A Large Cohort Study Using Multivariate Joint Model and Machine Learning. Diagnostics, 16(5), 665. https://doi.org/10.3390/diagnostics16050665








