Comprehensive Sonographic Paradigm and Trend Pattern of Median Nerve Indices in Carpal Tunnel Syndrome from Wrist to Forearm: What We Need to Know
Abstract
1. Introduction
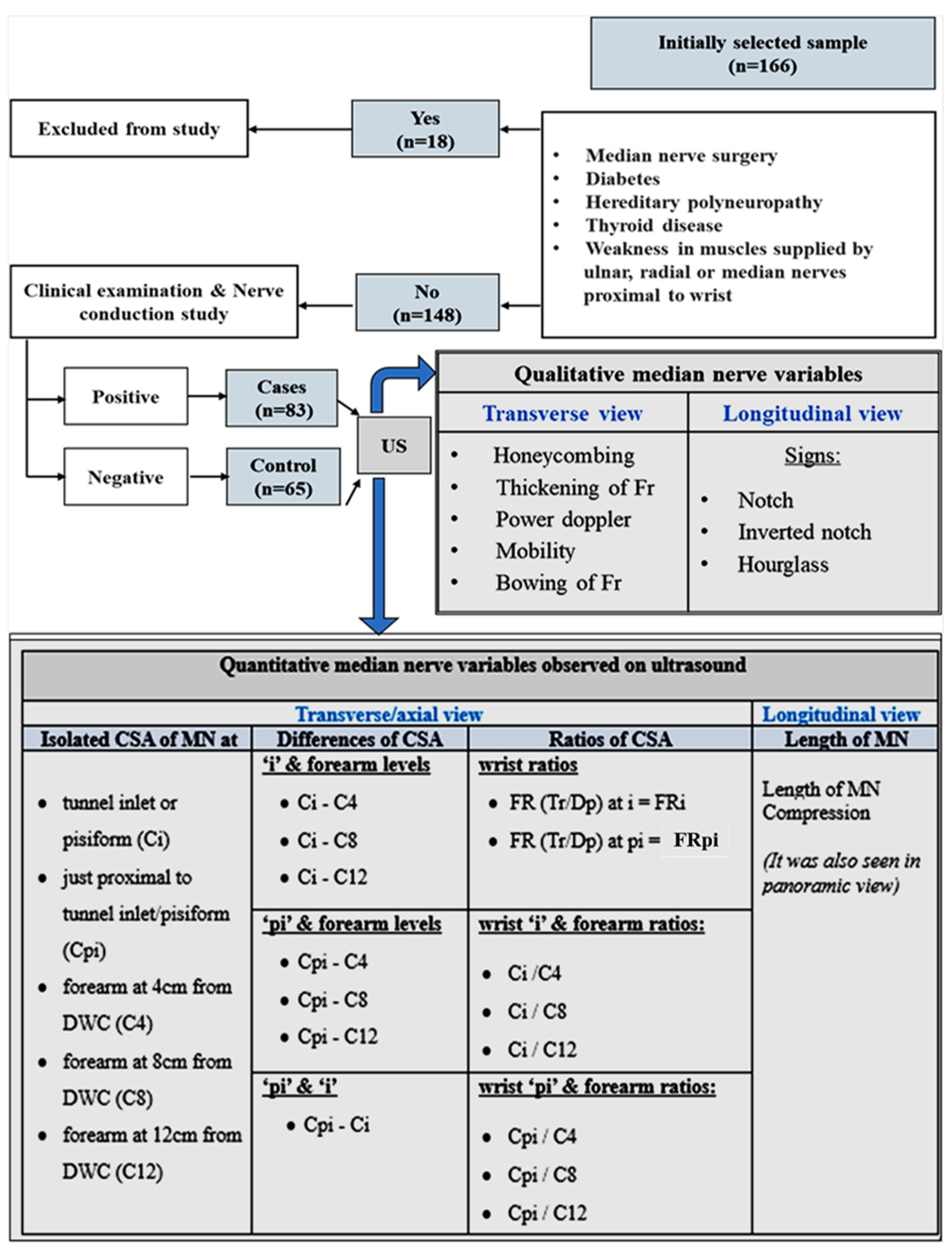
2. Materials and Methods
2.1. Patient Selection
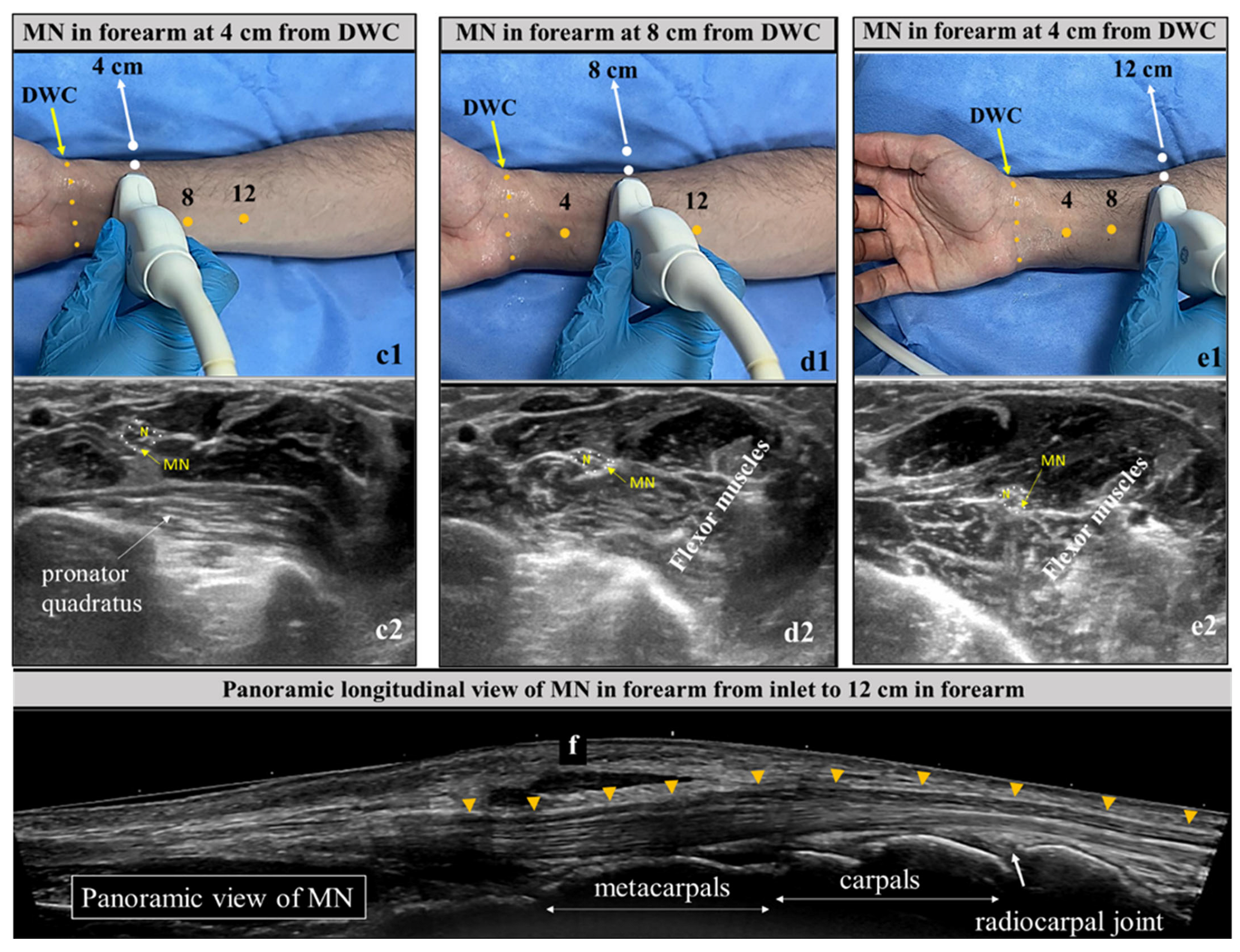
2.2. Ultrasound Technique
2.3. Images Recording and Interpretation
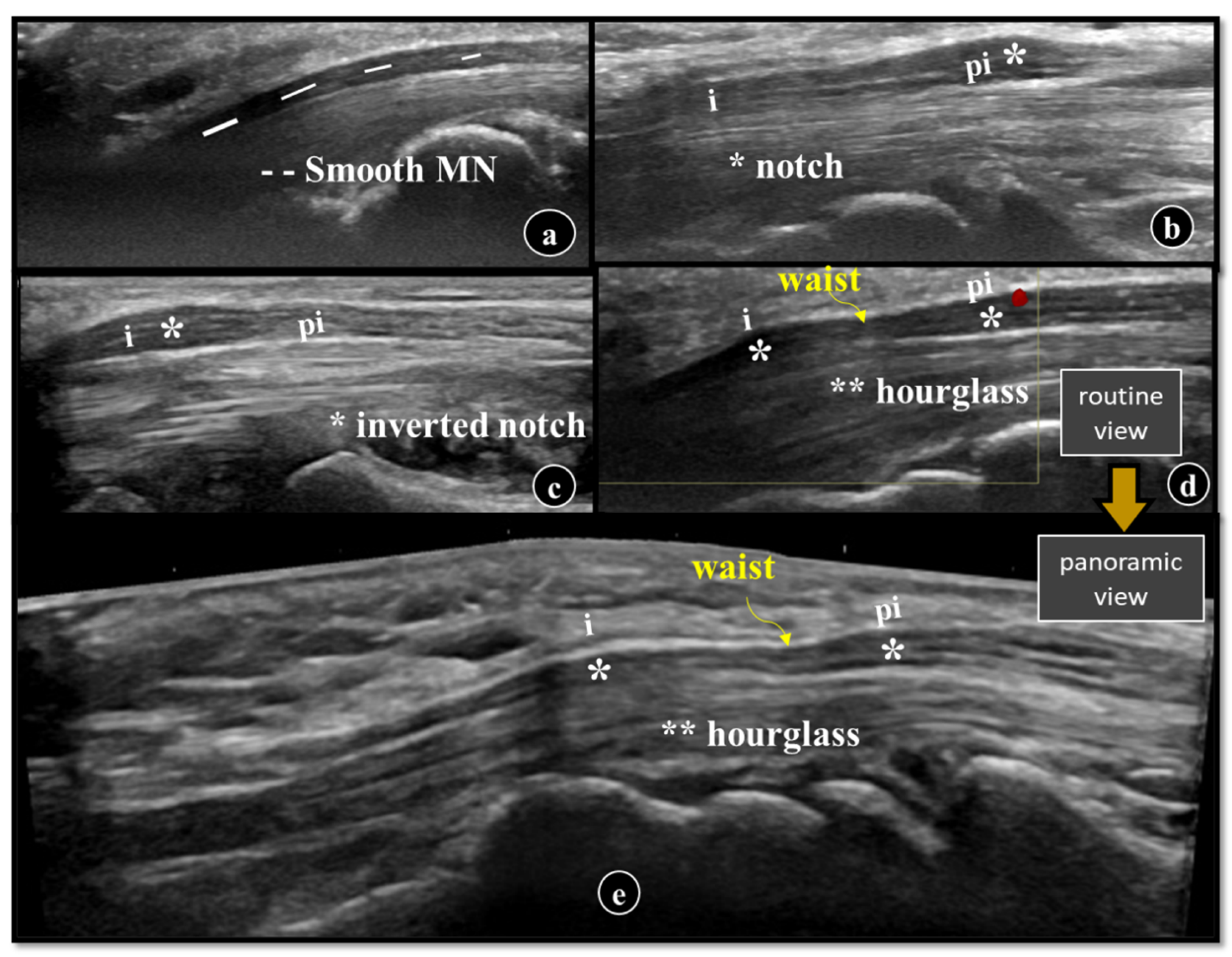
2.3.1. Definition of Inlet (i)
2.3.2. Definition of Proximal to Inlet (pi)
2.4. Statistical Analysis
3. Results
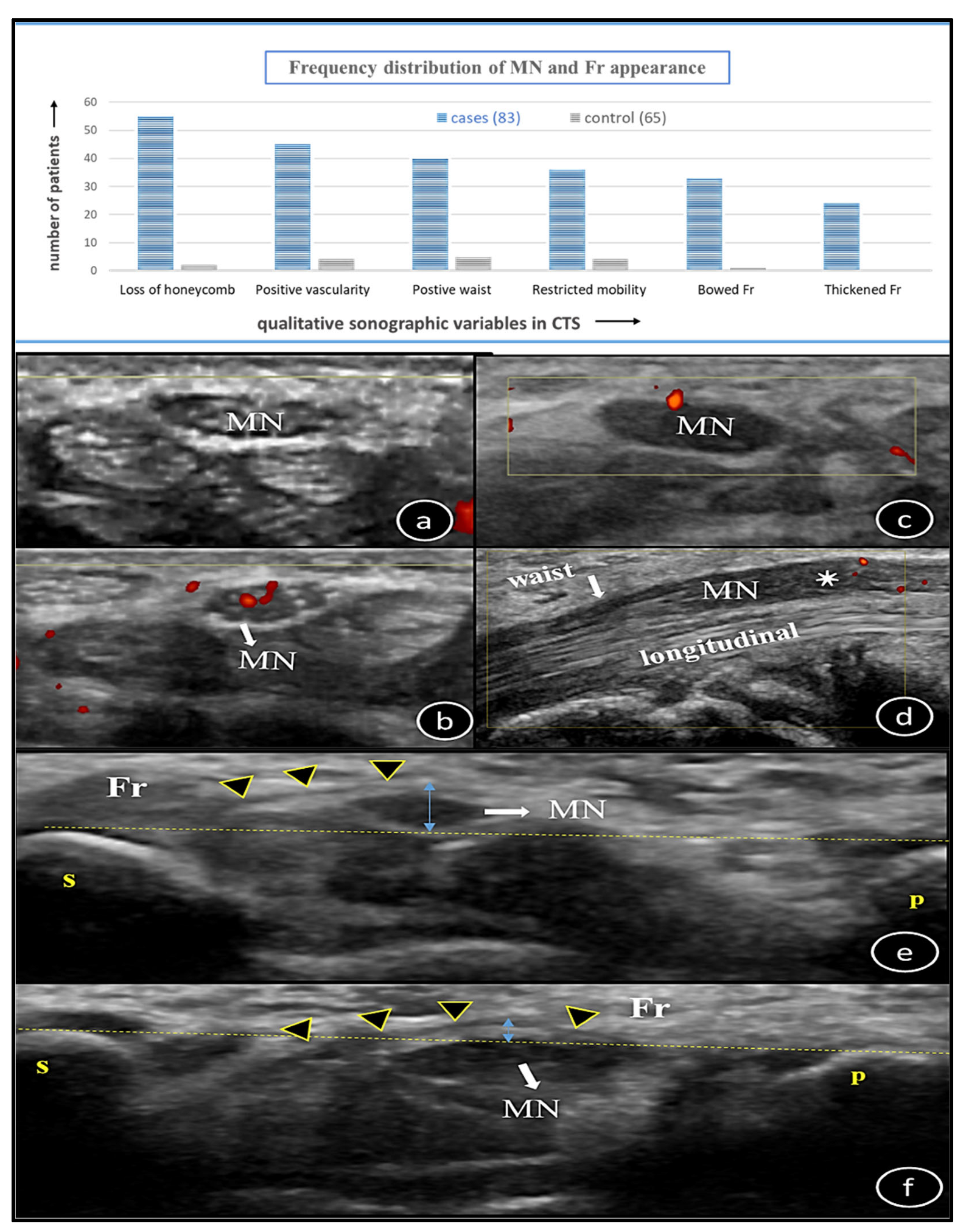
3.1. Subject Characteristics and Frequency Distribution of Qualitative Data Variables
3.2. Analysis of Quantitative Data Variables
3.3. Median Nerve Dimensions
3.3.1. Cross-Sectional Area
Wrist
Forearm
3.3.2. Transverse Length and Depth of Wrist MN
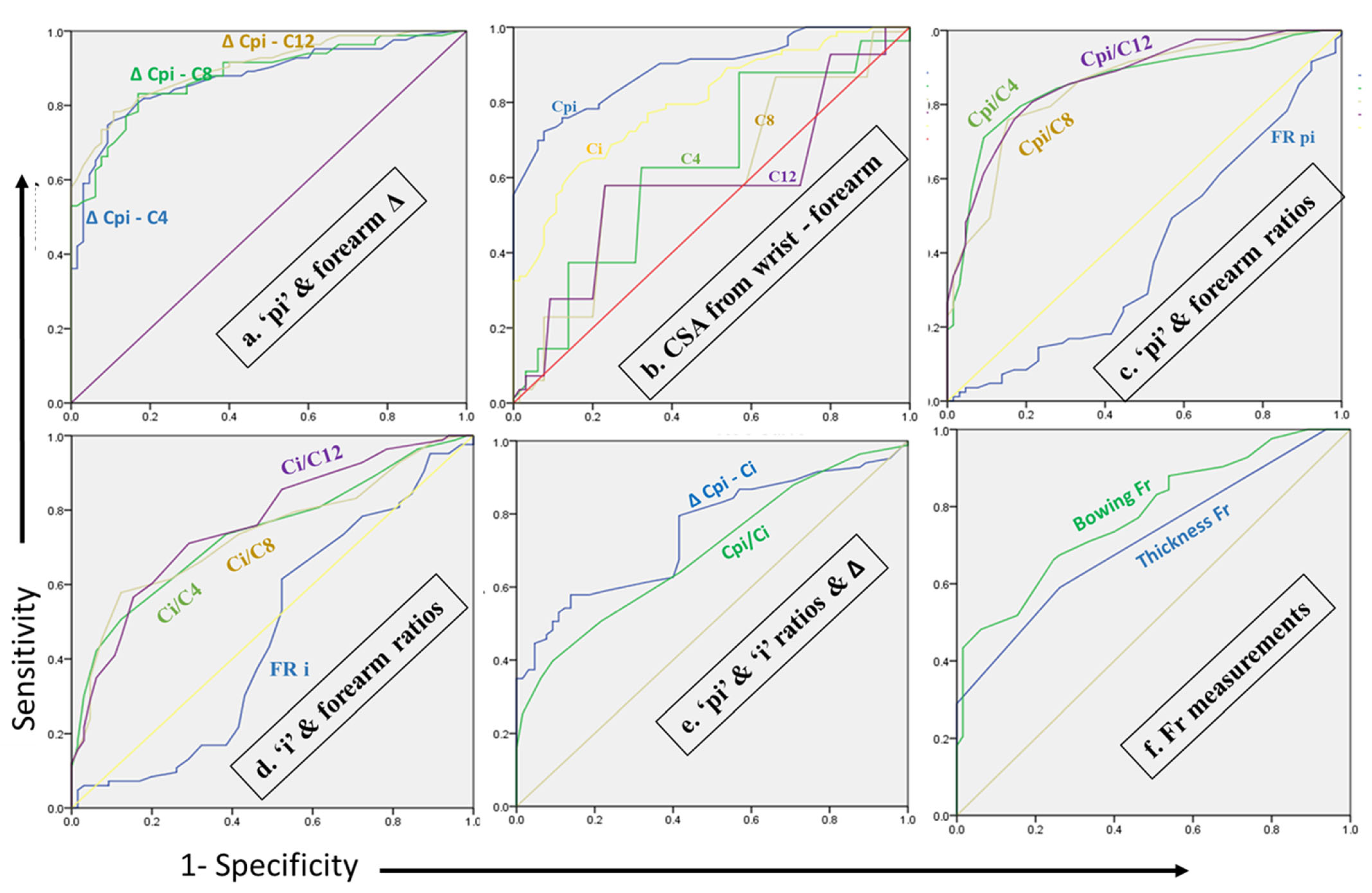
3.3.3. Differences (Δ) of Median Nerve CSA
Δ Proximal to Inlet—Forearm
Δ Proximal to Inlet—Inlet (Cpi-Ci)
3.3.4. Ratios of Median Nerve
Wrist Proximal to Inlet/Forearm
Wrist Inlet/Forearm
Proximal to Inlet/Inlet (pi/i)
Flattening Ratio in Inlet (FR i) and Proximal to Inlet (FR pi)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MN | Median Nerve |
| CTS | Carpal Tunnel Syndrome |
| CSA | Cross-Sectional Area |
| AUC | Area Under Curve |
| US | Ultrasound |
| NCS | nerve conduction study |
| DWC | distal wrist crease |
| PML | probe marker line |
| PUB | Probe upper border |
| Fr | flexor retinaculum |
| i | inlet |
| pi | proximal to inlet |
| Ci | Cross-Sectional Area of median nerve at inlet |
| Cpi | Cross-Sectional Area of median nerve at proximal to inlet |
| C4 | Cross-Sectional Area of median nerve in forearm at 4 cm from distal wrist crease |
| C8 | Cross-Sectional Area of median nerve in forearm at 8 cm from distal wrist crease |
| C12 | Cross-Sectional Area of median nerve in forearm at 12 cm from distal wrist crease |
| Tr | transverse of median nerve |
| Dp | depth of median nerve |
| FR | flattening ratio of median nerve |
| Ci/C4 | ratios of Cross-Sectional Area of median nerve at inlet and forearm at 4 cm from distal wrist crease |
| Ci/C8 | ratios of Cross-Sectional Area of median nerve at inlet and forearm at 8 cm from distal wrist crease |
| Ci/C12 | ratios of Cross-Sectional Area of median nerve at inlet and forearm at 12 cm from distal wrist crease |
| Cpi/C4 | ratios of Cross-Sectional Area of median nerve at proximal to inlet and forearm at 4 cm from distal wrist crease |
| Cpi/C8 | ratios of Cross-Sectional Area of median nerve at proximal to inlet and forearm at 8 cm from distal wrist crease |
| Cpi/C12 | ratios of Cross-Sectional Area of median nerve at proximal to inlet and forearm at 12 cm from distal wrist crease |
| Δ | difference |
| Cpi-C4 | difference between Cross-Sectional Area of median nerve at proximal to inlet and forearm at 4 cm from distal wrist crease |
| Cpi-C8 | difference between Cross-Sectional Area of median nerve at proximal to inlet and forearm at 8 cm from distal wrist crease |
| Cpi-C12 | difference between Cross-Sectional Area of median nerve at proximal to inlet and forearm at 12 cm from distal wrist crease |
| Sn/sp | sensitivity and specificity |
References
- Erickson, M.; Lawrence, M.; Jansen, C.W.; Coker, D.; Amadio, P.; Cleary, C. Hand pain and sensory deficits: Carpal Tunnel syndrome. J. Orthop. Sports Phys. Ther. 2019, 49, CPG1–CPG85. [Google Scholar] [CrossRef]
- Yoshii, Y.; Zhao, C.; Amadio, P.C. Recent advances in ultrasound diagnosis of carpal tunnel syndrome. Diagnostics 2020, 10, 596. [Google Scholar] [CrossRef]
- Salamh, F.; Habib, S.S.; AlRouq, F.; Albarrak, A.; Al-Khlaiwi, T.; Khan, A. Relationship of obesity indices with clinical severity and nerve conduction studies in females presenting with median nerve compression neuropathy at the wrist. J. Fam. Med. Prim. Care 2024, 13, 1291–1295. [Google Scholar] [CrossRef]
- Fowler, J.R.; Byrne, K.; Pan, T.; Goitz, R.J. False-positive rates for nerve conduction studies and ultrasound in patients without clinical signs and symptoms of carpal tunnel syndrome. J. Hand Surg. Am. 2019, 44, 181–185. [Google Scholar] [CrossRef]
- Gervasio, A.; Stelitano, C.; Bollani, P.; Giardini, A.; Vanzetti, E.; Ferrari, M. Ultrasound-based grading of carpal tunnel syndrome: A comparative study of cross-sectional area and shear wave elastography at different wrist joint angles. Br. J. Radiol. 2025, 98, 58–67. [Google Scholar] [CrossRef]
- Gervasio, A.; Stelitano, C.; Bollani, P.; Giardini, A.; Vanzetti, E.; Ferrari, M. Carpal tunnel sonography. J. Ultrasound 2020, 23, 337–347. [Google Scholar] [CrossRef]
- Ng, A.W.H.; Griffith, J.F.; Lee, R.K.L.; Tse, W.L.; Wong, C.W.Y.; Ho, P.C. Ultrasound Carpal Tunnel Syndrome: Additional criteria for diagnosis. Clin Radiol. 2018, 73, 214.e11–214.e18. [Google Scholar] [CrossRef]
- Braham, S.; Moussa, A.; Bouhoula, M.; Ben Meriem, N.; Annen, I.; Sakly, G.; Chouchane, A.; Ben Abdelkader, M.; Aloui, A.; Kacem, I.; et al. Exploring ultrasound and electromyography for carpal tunnel syndrome diagnosis: A comprehensive comparative study and implications for occupational medicine. Front. Neurol. 2024, 15, 1490873. [Google Scholar] [CrossRef]
- Lee, D.; van Holsbeeck, M.T.; Janevski, P.K.; Ganos, D.L.; Ditmars, D.M.; Darian, V.B. Diagnosis of carpal tunnel syndrome. Radiol. Clin. N. Am. 1999, 37, 859–872. [Google Scholar] [CrossRef]
- Schwabl, C.; Schmidle, G.; Kaiser, P.; Drakonaki, E.; Taljanovic, M.S.; Klauser, A.S. Nerve entrapment syndromes: Detection by ultrasound. Ultrasonography 2023, 42, 376–387. [Google Scholar] [CrossRef]
- Falsetti, P.; Conticini, E.; Baldi, C.; D’ignazio, E.; Al Khayyat, S.G.; Bardelli, M.; Gentileschi, S.; D’alessandro, R.; D’alessandro, M.; Acciai, C.; et al. A novel ultrasonographic anthropometric-independent measurement of median nerve swelling in carpal tunnel syndrome: The “nerve/tendon ratio” (NTR). Diagnostics 2022, 12, 2621. [Google Scholar] [CrossRef]
- Yao, B.; Yang, L.; Evans, K.D.; Roll, S.C. Doppler Sonography Evaluation of Median Nerve Intraneural Blood Flow: A Systematic Review. J. Diagn. Med. Sonogr. 2024, 40, 480–495. [Google Scholar] [CrossRef]
- Jacobson, J.A.; Wilson, T.J.; Yang, L.J.-S. Sonography of common peripheral nerve disorders with clinical correlation. J. Ultrasound Med. 2016, 35, 683–693. [Google Scholar] [CrossRef]
- Presazzi, A.; Bortolotto, C.; Zacchino, M.; Madonia, L.; Draghi, F. Carpal tunnel: Normal anatomy, anatomical variants and ultrasound technique. J. Ultrasound 2011, 14, 40–46. [Google Scholar] [CrossRef]
- Angelopoulou, C.; Chrysafis, I.; Keskinis, A.; Tilkeridis, K.; Trypsianis, G.; Paraskevopoulos, K.; Drosos, G.; Ververidis, A. Does ultrasonography, in conjunction with nerve conduction study, plays any role in the diagnosis and the evaluation of severity in patients with clinically diagnosed carpal tunnel syndrome? A prospective study. Arch. Med. Clin. Res. 2021, 1, 1–13. [Google Scholar] [CrossRef]
- Min, Y.S.; Han, J.S.; Kim, S.H. Normal Sonographic Anatomy of the Wrist and Hand. Clin. Pain 2022, 21, 15–20. [Google Scholar] [CrossRef]
- Hobson-Webb, L.D.; Massey, J.M.; Juel, V.C.; Sanders, D.B. The ultrasonographic wrist-to-forearm median nerve area ratio in carpal tunnel syndrome. Clin. Neurophysiol. 2008, 119, 1353–1357. [Google Scholar] [CrossRef]
- Peng, F.; Zhang, L.; Rehemutula, A.; Yu, C.; Wang, T.; Chen, L. Does the ratio of the carpal tunnel inlet and outlet cross-sectional areas in the median nerve reflect carpal tunnel syndrome severity? Neural Regen. Res. 2015, 10, 1172. [Google Scholar] [CrossRef]
- Dejaco, C.; Stradner, M.; Zauner, D.; Seel, W.; Simmet, N.E.; Klammer, A.; Heitzer, P.; Brickmann, K.; Gretler, J.; Fürst-Moazedi, F.C.; et al. Ultrasound for diagnosis of carpal tunnel syndrome: Comparison of different methods to determine median nerve volume and value of power Doppler sonography. Ann. Rheum. Dis. 2012, 72, 1934–1939. [Google Scholar] [CrossRef]
- Chen, S.-F.; Lu, C.-H.; Huang, C.-R.; Chuang, Y.-C.; Tsai, N.-W.; Chang, C.-C.; Chang, W.-N. Ultrasonographic median nerve cross-section areas measured by 8-point “Inching test” for idiopathic carpal tunnel syndrome: A correlation of nerve conduction study severity and duration of clinical symptoms. BMC Med. Imaging 2011, 11, 22. [Google Scholar] [CrossRef]
- Pace, V.; Marzano, F.; Placella, G. Update on surgical procedures for carpal tunnel syndrome: What is the current evidence and practice? what are the future research directions? World J. Orthop. 2023, 14, 6–12. [Google Scholar] [CrossRef]
- Luchetti, R.; Amadio, P. (Eds.) Carpal Tunnel Syndrome; Springer-Verlag: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- McDonagh, C.; Alexander, M.; Kane, D. The role of ultrasound in the diagnosis and management of Carpal Tunnel Syndrome: A new paradigm. Rheumatology 2014, 54, 9–19. [Google Scholar] [CrossRef]
- El-Shewi, I.E.H.; Tawfeek, A.A.; Mohamed, A.A.; Mostafa, M.A. Role of conventional ultrasound and shear wave elastography of median nerve in diagnosis and differentiation of carpal tunnel syndrome severity in correlation with electrodiagnostic studies. Egypt. J. Radiol. Nucl. Med. 2024, 55, 53. [Google Scholar] [CrossRef]
- Battaglia, F.; Troisi, L.; Cigna, E.; D’alcontres, F.S.; Rizzo, V.; Delia, G. Standardized high-resolution ultrasound protocol for the diagnosis and monitoring of carpal tunnel syndrome: A mixed-design observational study. Diagnostics 2025, 15, 1593. [Google Scholar] [CrossRef]
- Won, S.J.; Kim, B.; Park, K.S.; Yoon, J.S.; Choi, H. Reference values for nerve ultrasonography in the upper extremity. Muscle Nerve 2013, 47, 864–871. [Google Scholar] [CrossRef]
- Kapuścińska, K.; Urbanik, A. High-frequency ultrasound in carpal tunnel syndrome: Assessment of patient eligibility for surgical treatment. J. Ultrason. 2015, 15, 283–291. [Google Scholar] [CrossRef]
- Xu, C.; Zhou, Y.; He, Z.; Liu, W.; Zou, M.; Sun, Y.; Qiu, J.; Ren, Y.; Mao, G.; Wang, Y.; et al. Difference and ratio of the cross-sectional area of median nerve at the carpal tunnel and the pronator quadratus muscle in diagnosing carpal tunnel syndrome: A cross-sectional study. Ann. Transl. Med. 2022, 10, 340. [Google Scholar] [CrossRef]
- Chang, Y.-W.; Hsieh, T.-C.; Tzeng, I.-S.; Chiu, V.; Huang, P.-J.; Horng, Y.-S. Ratio and difference of the cross-sectional area of median nerve to ulnar nerve in diagnosing carpal tunnel syndrome: A case control study. BMC Med. Imaging 2019, 19, 52. [Google Scholar] [CrossRef]
- Kanagasabai, K. Ultrasound of median nerve in the diagnosis of carpal tunnel syndrome—Correlation with Electrophysiological Studies. Indian. J. Radiol. Imaging 2022, 32, 016–029. [Google Scholar] [CrossRef]
- Mianehsaz, E.; Talari, H.; Naghavi Ravandi, M.; Tabatabaei, M.H.; Azadchehr, M.J.; Eshraqi, S.; Heidari, M.M. Evaluating various ultrasound criteria for determining carpal tunnel syndrome severity. Radiol. Res. Pract. 2025, 2025, 4936187. [Google Scholar] [CrossRef]
- Panicker, P.; Iype, T. Nerve ultrasound findings in carpal tunnel syndrome and its correlation with clinical and electrophysiological data. J. Med. Sci. Health 2022, 8, 59–64. [Google Scholar] [CrossRef]
- Osiak, K.; Elnazir, P.; Walocha, J.A.; Pasternak, A. Carpal tunnel syndrome: State-of-the-art review. Folia Morphol. 2022, 81, 851–862. [Google Scholar] [CrossRef]
- Nalbant, M.; Ümit Yemişci, O.; Özen, S.; Tezcan, Ş. Ultrasonographic and electrophysiological outcomes of carpal tunnel syndrome treated with low-level laser therapy: A double-blind, prospective, randomized, sham-controlled study. Arch. Rheumatol. 2022, 37, 19–30. [Google Scholar] [CrossRef]
- Kuo, T.-T.; Lee, M.-R.; Liao, Y.-Y.; Chen, J.-P.; Hsu, Y.-W.; Yeh, C.-K. Assessment of median nerve mobility by ultrasound dynamic imaging for diagnosing carpal tunnel syndrome. PLoS ONE 2016, 11, e0147051. [Google Scholar] [CrossRef]
- Yau, Y.-C.; Yang, C.-P.; Lin, C.-P.; Tsai, I.-J.; Chang, C.-M.; Yang, C.-C.; Shih, P.-H.; Liao, Y.-Y. The morphological and dynamic changes of ultrasound in the evaluation of effects of oral steroids treatment for patients with carpal tunnel syndrome. Diagnostics 2021, 11, 1336. [Google Scholar] [CrossRef]
- Houben, M.P.W.A.; Maars, M.; Beekman, R. High prevalence of increased nerve vascularization in healthy individuals. Muscle Nerve 2015, 51, 938–939. [Google Scholar] [CrossRef]
- Habib, S.S.; Alanazy, M.H. Predictive Value of Markers of Adiposity in Carpal Tunnel Syndrome: A Clinical and Electrophysiological Evaluation. J. Coll. Physicians Surg. Pak. 2020, 30, 828–832. [Google Scholar] [CrossRef]

| (a) Demographic Data | ||||||
| n = 83~65 | Males (n) | Females (n) | Age (years) | Height (cm) | Weight (kg) | BMI (kg/m2) |
| case~control | 16~19 | 67~46 | 50.6~40.16 | 158.3~164.7 | 81.2~76.21 | 32.3~27.9 |
| ±SD | - | 8.92~9.98 | 7.10~8.49 | 14.43~16.13 | 5.08~4.89 | |
| pvalue | - | 0.00 | 0.00 | 0.03 | 0.00 | |
| (b) Frequency (%) Distribution of MN and Fr Appearance (In Decreasing Order from Left to Right) | ||||||
| Features | Loss of Honeycomb | Positive Vascularity | Positive Waist Sign | Restricted Mobility | Bowed Fr | Thickened Fr |
| case~control (%) | 66.3~3.1 | 54.2~6.2 | 48.2~7.7 | 43.4~6.2 | 39.7~1.5 | 28.9~0 |
| (n, cases)~(n, control) | (55)~(2) | (45)~(4) | (40)~(5) | (36)~(4) | (33)~(1) | (24)~(0) |
| p-value | 1 × 10−5 | |||||
| (c) Flexor retinaculum measurements and MN compression length | ||||||
| Features Mean ± SD (range) | Thickness of Fr (mm) case~control | Bowing height of Fr (mm) case~control | Length of waist (mm) case~control | |||
| Mean | 0.3~0.2 | 2.0~1.1 | 4.9~5.8 | |||
| ±SD | 0.13~0.05 | 0.94~0.59 | 2.11~1.3 | |||
| AUC | 0.71 | 0.77 | - | |||
| Cut-off value | 0.3 | 1.4 | - | |||
| Min. | 0.2~0.1 | 0.4~−0.3 | 2.0~4.6 | |||
| Max. | 0.8~0.3 | 4.7~2.7 | 10.0~7.6 | |||
| p-value | 0.00 | 0.22 | ||||
| AUC | Variables | Mean Case~Control | ±SD Case~Control | Min. Case~Control | Max. Case~Control | Cut-Off | Sn/Sp % | p-Value | |
|---|---|---|---|---|---|---|---|---|---|
| (a) | Flexor retinaculum (mm) | ||||||||
| 0.71 | Thickness | 0.3~0.2 | 0.13~0.05 | 0.2~0.1 | 0.8~0.3 | 0.3 | 59/73.8 | 0.00 | |
| 0.77 | Bowing height | 2.0~1.1 | 0.94~0.59 | 0.4~0.2 | 4.7~2.7 | 1.4 | 71.1/67 | 0.00 | |
| - | Waist length | 4.9~5.8 | 2.11~1.32 | 2~4.6 | 10~7.6 | - | - | 0.22 | |
| (b) | Wrist median nerve CSA (mm2) | ||||||||
| 0.80 | Inlet (Ci) | 9.2~6.4 | 3.32~1.43 | 4.5~3.4 | 22~9.5 | 7.2 | 72.3/71 | 0.00 | |
| 0.89 * | proximal to inlet(Cpi) * | 12.3~7.2 | 5.57~1.57 | 6~3.5 | 33~10 | 9.1 * | 73.5/89.2 * | ||
| (c) | Forearm median nerve CSA at (mm2) | ||||||||
| 0.63 | 4 cm (C4) | 7.1~6.5 | 1.61~1.50 | 4.5~4.1 | 12~11 | 6.9 | 62.7/67.7 | 0.01 | |
| 0.61 | 8 cm (C8) | 5.8~5.4 | 1.19~1.27 | 3.2~3 | 9.1~10 | 5.9 | 57.8/76.9 | 0.05 | |
| 0.58 | 12 cm (C12) | 4.9~4.5 | 1.18~1.05 | 2.7~2.1 | 9~8 | 4.9 | 82/81.5 | 0.04 | |
| (d) | 2D dimensions of median nerve in wrist inlet (mm) | ||||||||
| - | Tr | 5.4~4.7 | 1.03~0.97 | 4.1~3 | 10~7 | - | - | 0.00 | |
| - | Dp | 1.6~1.2 | 0.62~0.36 | 1~1 | 4~2 | - | - | ||
| (e) | 2D dimensions of median nerve in wrist justproximal to inlet(mm) | ||||||||
| - | Tr | 6.6~5.2 | 1.41~0.85 | 4.9~4 | 11~7 | - | - | 0.00 | |
| - | Dp | 2.2~1.6 | 0.51~0.32 | 1.2~1 | 3.7~2 | - | - | ||
| AUC | Variables (mm2) | Mean Case~Control | SD± Case~Control | Min. Case~Control | Max. Case~Control | Cut-Off | Sn/Sp % | p Value | |
|---|---|---|---|---|---|---|---|---|---|
| (a) | Δ proximal to inlet—forearm CSA at 4, 8, 12 cm distances from DWC | ||||||||
| 0.87 | Cpi-C4 | 5.2~1.3 | 4.68~1.08 | 1.4~2.5 | 23.9~4.9 | 2.2 | 83/83.1 | 0.00 | |
| 0.88 | Cpi-C8 | 6.5~1.9 | 5.11~1.36 | 0.1~1.9 | 26.2~5.2 | 3.0 | 82/83.1 | ||
| 0.90 * | Cpi-C12 * | 7.4~2.6 * | 5.07~1.35 | 1.3~0.3 | 28~5.7 | 3.7 * | 78.3/89.2 * | ||
| (b) | Δ proximal to inlet—inlet CSA | ||||||||
| 0.74 | Cpi-Ci | 3.2~1.0 | 3.44~0.80 | 0~0 | 19~3.4 | 1.2 | 61.4/66.2 | 0.00 | |
| AUC | Variables (mm2) | Mean Case~Control | SD± Case~Control | Min. Case~Control | Max. Case~Control | Cut Off | Sn/Sp % | p Value | |
|---|---|---|---|---|---|---|---|---|---|
| (a) | Wrist to forearm ratio: CSA at ‘pi’ ÷ CSA in forearm at 4, 8, 12 cm | ||||||||
| 0.86 | Cpi/C4 | 1.7~1.1 | 0.55~0.26 | 0.8~0.6 | 4.0~2.0 | 1.3 | 80/82 | 0.00 | |
| 0.85 | Cpi/C8 | 2.1~1.4 | 0.78~0.36 | 1~0.6 | 5.5~2.4 | 1.5 | 80/74 | ||
| 0.86 * | Cpi/C12* | 2.5~1.6 | 0.88~0.37 | 1.3~0.9 | 6.6~2.7 | 1.8 * | 81/79 * | ||
| (b) | Wrist to forearm ratio: CSA at ‘i’ ÷ CSA in forearm at 4, 8, 12 cm | ||||||||
| 0.74 | Ci/C4 | 1.3~1.0 | 0.38~2.29 | 0.6~0.5 | 2.7~1.7 | 1.1 | 74/62 | 0.00 | |
| 0.74 | Ci/C8 | 1.6~1.2 | 0.51~0.31 | 0.8~0.6 | 3.8~2.1 | 1.3 | 74/59 | ||
| 0.76 | Ci/C12 | 1.9~1.4 | 0.54~0.35 | 1.0~0.9 | 3.6~2.5 | 1.5 | 71/71 | ||
| (c) | Wrist ratios: CSA at ‘pi’ ÷ CSA at ‘i’ | ||||||||
| 0.69 | Cpi/Ci | 1.3~1.1 | 0.34~0.16 | 0.7~0.9 | 2.7~1.6 | 1.1 | 63.9/58.5 | 0.00 | |
| (d) | Flattening ratio of MN at ‘i’ (Tr ÷ Dp) | ||||||||
| 0.46 | FR i | 3.1~3.3 | 0.77~0.89 | 1.5~1.7 | 5.5~5.9 | 3.0 | 53/48 | 0.49 | |
| (e) | Flattening ratio of MN at ‘pi’ (Tr ÷ Dp) | ||||||||
| 0.40 | FR pi | 3.3~3.6 | 0.76~1.13 | 2~2.1 | 6.2~9.8 | 3.2 | 50/43.1 | 0.04 | |
| Overall Trend Pattern of MN Parameters from Wrist to Forearm | |
|---|---|
| (a) dimensions |
|
| (b) differences |
|
| (c) ratios |
|
| (d) overall |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Khan, A.; Salamah, F.T.; Habib, S.S.; Fawzy, W.; AlRouq, F.; Alkhliwi, H.T.; Sultan, M.; Alsabih, A.O. Comprehensive Sonographic Paradigm and Trend Pattern of Median Nerve Indices in Carpal Tunnel Syndrome from Wrist to Forearm: What We Need to Know. Diagnostics 2026, 16, 641. https://doi.org/10.3390/diagnostics16040641
Khan A, Salamah FT, Habib SS, Fawzy W, AlRouq F, Alkhliwi HT, Sultan M, Alsabih AO. Comprehensive Sonographic Paradigm and Trend Pattern of Median Nerve Indices in Carpal Tunnel Syndrome from Wrist to Forearm: What We Need to Know. Diagnostics. 2026; 16(4):641. https://doi.org/10.3390/diagnostics16040641
Chicago/Turabian StyleKhan, Adeena, Fawaz T. Salamah, Syed S. Habib, Waleed Fawzy, Fawzia AlRouq, Huthayfah T. Alkhliwi, Mamoona Sultan, and Ahmed O. Alsabih. 2026. "Comprehensive Sonographic Paradigm and Trend Pattern of Median Nerve Indices in Carpal Tunnel Syndrome from Wrist to Forearm: What We Need to Know" Diagnostics 16, no. 4: 641. https://doi.org/10.3390/diagnostics16040641
APA StyleKhan, A., Salamah, F. T., Habib, S. S., Fawzy, W., AlRouq, F., Alkhliwi, H. T., Sultan, M., & Alsabih, A. O. (2026). Comprehensive Sonographic Paradigm and Trend Pattern of Median Nerve Indices in Carpal Tunnel Syndrome from Wrist to Forearm: What We Need to Know. Diagnostics, 16(4), 641. https://doi.org/10.3390/diagnostics16040641






