Early Prediction of Biliary Atresia Using Combi-Elastography in Infants ≤ 60 Days of Age
Abstract
1. Introduction
2. Method and Materials
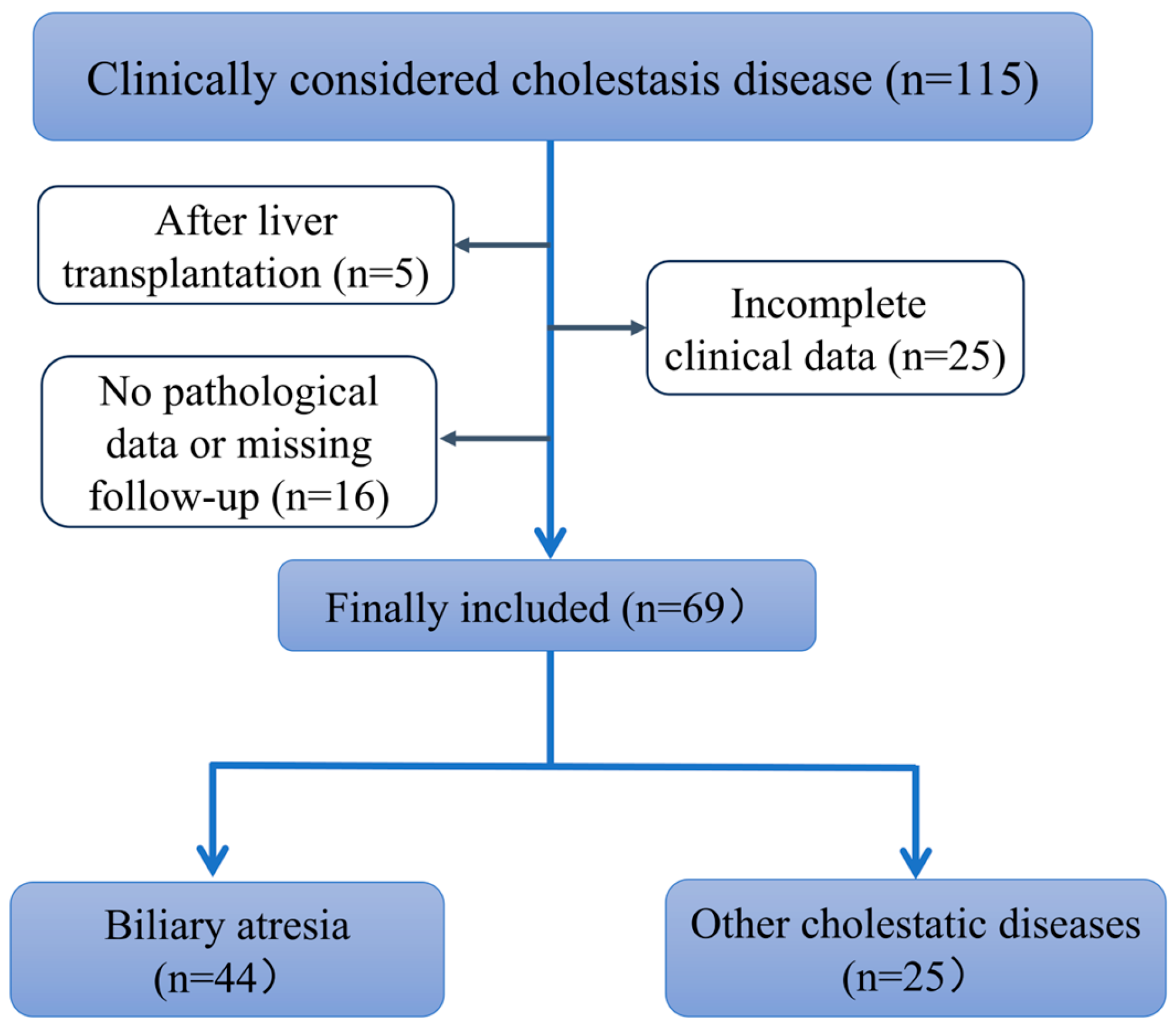
2.1. Study Population
2.2. Laboratory Indexes
2.3. Conventional Ultrasound
2.4. Combi-Elastography Evaluation
2.5. Statistical Analyses
3. Results
3.1. General Characteristics
3.2. Nomogram Construction
3.3. Diagnostic Performance of Laboratory and Ultrasound Indexes
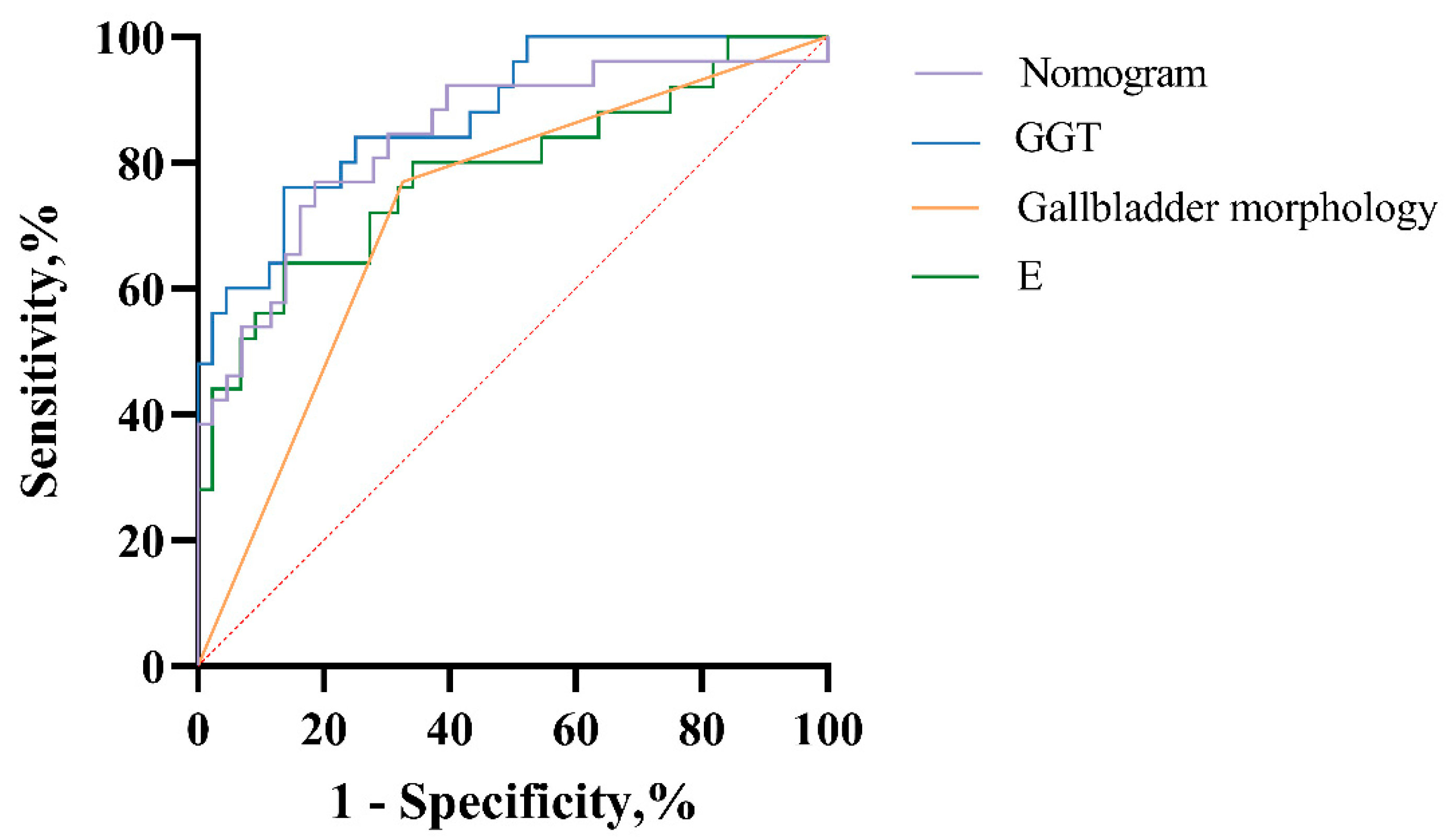
3.4. Evaluation of Nomogram Performance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Harpavat, S.; Garcia-Prats, J.A.; Anaya, C.; Brandt, M.L.; Lupo, P.J.; Finegold, M.J.; Obuobi, A.; ElHennawy, A.A.; Jarriel, W.S.; Shneider, B.L. Diagnostic Yield of Newborn Screening for Biliary Atresia Using Direct or Conjugated Bilirubin Measurements. JAMA 2020, 323, 1141–1150. [Google Scholar] [CrossRef]
- Lendahl, U.; Lui, V.C.H.; Chung, P.H.Y.; Tam, P.K.H. Biliary Atresia—Emerging diagnostic and therapy opportunities. eBioMedicine 2021, 74, 103689. [Google Scholar] [CrossRef] [PubMed]
- Evans, H.M.; Siew, S.M. Neonatal liver disease. J. Paediatr. Child Health 2020, 56, 1760–1768. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.Y.; Zhan, X.; Li, S.Y. Prompt distinction of Alagille syndrome and biliary Atresia in infants: A comparative study. BMC Pediatr. 2025, 25, 543. [Google Scholar] [CrossRef] [PubMed]
- Anouti, A.; Sudhakara, P.; Pratt, C.; Mourya, R.; VanWagner, L.; Shivakumar, P.; Pandurangi, S. Advances in prognostic biomarkers for biliary atresia: Current insights and future directions. J. Pediatr. Gastroenterol. Nutr. 2025, 81, 497–506. [Google Scholar] [CrossRef]
- He, J.P.; Hao, Y.; Wang, X.L.; Yang, X.J.; Shao, J.F.; Feng, J.X. Comparison of different noninvasive diagnostic methods for biliary atresia: A meta-analysis. World J. Pediatr. 2016, 12, 35–43. [Google Scholar] [CrossRef]
- Chan, W.K.; Chung, P.H.Y.; Wong, K.K.Y. The Value of Hepatic Scintigraphy in the Diagnosis of Biliary Atresia. Front. Pediatr. 2022, 10, 874809. [Google Scholar] [CrossRef]
- Napolitano, M.; Franchi-Abella, S.; Damasio, B.M.; Augdal, T.A.; Avni, F.E.; Bruno, C.; Darge, K.; Kljucevsek, D.; Littooij, A.S.; Lobo, L.; et al. Practical approach for the diagnosis of biliary atresia on imaging, part 2: Magnetic resonance cholecystopancreatography, hepatobiliary scintigraphy, percutaneous cholecysto-cholangiography, endoscopic retrograde cholangiopancreatography, percutaneous liver biopsy, risk scores and decisional flowchart. Pediatr. Radiol. 2021, 51, 1545–1554. [Google Scholar] [CrossRef]
- Parra, D.A.; Peters, S.E.; Kohli, R.; Chamlati, R.; Connolly, B.L.; Wolinska, J.M.; Ng, V.L.; Temple, M.J.; John, P.R.; Kamath, B.M.; et al. Findings in percutaneous trans-hepatic cholecysto-cholangiography in neonates and infants presenting with conjugated hyperbilirubinemia: Emphasis on differential diagnosis and cholangiographic patterns. BMC Pediatr. 2023, 23, 22. [Google Scholar] [CrossRef]
- Wang, J.; Guo, L.; Shi, X.; Pan, W.; Bai, Y.; Ai, H. Real-time elastography with a novel quantitative technology for assessment of liver fibrosis in chronic hepatitis B. Eur. J. Radiol. 2012, 81, e31–e36. [Google Scholar] [CrossRef]
- Fujimoto, K.; Kato, M.; Kudo, M.; Yada, N.; Shiina, T.; Ueshima, K.; Yamada, Y.; Ishida, T.; Azuma, M.; Yamasaki, M.; et al. Novel image analysis method using ultrasound elastography for noninvasive evaluation of hepatic fibrosis in patients with chronic hepatitis C. Oncology 2013, 84, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, C.F.; Bamber, J.; Berzigotti, A.; Bota, S.; Cantisani, V.; Castera, L.; Cosgrove, D.; Ferraioli, G.; Friedrich-Rust, M.; Gilja, O.H.; et al. EFSUMB Guidelines and Recommendations on the Clinical Use of Liver Ultrasound Elastography, Update 2017 (Long Version). Ultraschall Med. 2017, 38, e16–e47. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Liang, J.; Shan, Q.; Chen, H.; Gao, P.; Cao, Q.; Wang, G.; Xie, X.; Zhou, L. Comparison of Two Kinds of Two-Dimensional Shear Wave Elastography Techniques in the Evaluation of Jaundiced Infants Suspected of Biliary Atresia. Diagnostics 2022, 12, 1092. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wu, L.; Qin, H.; Li, Q.; Shen, C.; He, Y.; Yang, H. Preoperative combi-elastography for the prediction of early recurrence after curative resection of hepatocellular carcinoma. Clin. Imaging 2021, 79, 173–178. [Google Scholar] [CrossRef]
- Ferraioli, G.; Wong, V.W.; Castera, L.; Berzigotti, A.; Sporea, I.; Dietrich, C.F.; Choi, B.I.; Wilson, S.R.; Kudo, M.; Barr, R.G. Liver Ultrasound Elastography: An Update to the World Federation for Ultrasound in Medicine and Biology Guidelines and Recommendations. Ultrasound Med. Biol. 2018, 44, 2419–2440. [Google Scholar] [CrossRef]
- Yada, N.; Tamaki, N.; Koizumi, Y.; Hirooka, M.; Nakashima, O.; Hiasa, Y.; Izumi, N.; Kudo, M. Diagnosis of Fibrosis and Activity by a Combined Use of Strain and Shear Wave Imaging in Patients with Liver Disease. Dig. Dis. 2017, 35, 515–520. [Google Scholar] [CrossRef]
- Li, R.; Hu, C.; Xu, F.; Zhang, Q.; Zhou, F.; Zheng, C.; Gao, Y.; Tang, Y.; Chen, J. Combi-Elasto Evaluation of the Degree of Liver Fibrosis in Children with Cholestatic Liver Disease. Diagnostics 2023, 13, 3229. [Google Scholar] [CrossRef]
- Gao, Y.; Xu, F.; Zhu, L.; Tang, Y.; Yang, C.; Xu, J.; Wang, Z.G.; Chen, J. Application of combi-elastography for follow-up after pediatric liver transplantation: A prospective study. Eur. J. Radiol. 2025, 188, 112127. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, T.; Wang, T.; Ji, Q.; Zhan, J. Comparison for the diagnostic performance of early diagnostic methods for biliary atresia: A systematic review and network meta-analysis. Pediatr. Surg. Int. 2024, 40, 146. [Google Scholar] [CrossRef]
- Huang, C.; Lu, Y.; Wang, Z.; Jiang, Q.; Dong, Y.; Cao, L.; Yan, J.; Xu, Z.; Wang, F.; Gao, Y.; et al. Correlation Between Clinical Indicators and Liver Pathology in Children with Chronic Hepatitis B. Biomedicines 2024, 12, 2903. [Google Scholar] [CrossRef]
- Yang, P.; Tang, Y.; Wang, H.; Zhang, X.; Yang, B. Latest diagnostic performance of different ultrasonic features for biliary atresia. Acta Radiol. 2022, 63, 1593–1602. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Shan, Q.; Tian, W.; Wang, Z.; Liang, J.; Xie, X. Ultrasound for the Diagnosis of Biliary Atresia: A Meta-Analysis. Am. J. Roentgenol. 2016, 206, W73–W82. [Google Scholar] [CrossRef] [PubMed]
- Farrant, P.; Meire, H.B.; Mieli-Vergani, G. Improved diagnosis of extraheptic biliary atresia by high frequency ultrasound of the gall bladder. Br. J. Radiol. 2001, 74, 952–954. [Google Scholar] [CrossRef]
- Cavallo, L.; Kovar, E.M.; Aqul, A.; McLoughlin, L.; Mittal, N.K.; Rodriguez-Baez, N.; Shneider, B.L.; Zwiener, R.J.; Chambers, T.M.; Langlois, P.H.; et al. The Epidemiology of Biliary Atresia: Exploring the Role of Developmental Factors on Birth Prevalence. J. Pediatr. 2022, 246, 89–94.e2. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhao, D.; Gu, S.; Li, Y.; Pan, W.; Zhang, Y. Three-color risk stratification for improving the diagnostic accuracy for biliary atresia. Eur. Radiol. 2020, 30, 3852–3861. [Google Scholar] [CrossRef]
- Dai, S.Y.; Sun, Y.Q.; Wu, Y.; Chen, G.; Sun, S.; Dong, R.; Zheng, S. Development and Assessment of Screening Nomogram for Biliary Atresia Based on Hepatobiliary Ultrasonographic Features. Front. Pediatr. 2021, 9, 625451. [Google Scholar] [CrossRef]
- Wang, Y.; Jia, L.Q.; Hu, Y.X.; Xin, Y.; Yang, X.; Wang, X.M. Development and Validation of a Nomogram Incorporating Ultrasonic and Elastic Findings for the Preoperative Diagnosis of Biliary Atresia. Acad. Radiol. 2021, 28, S55–S63. [Google Scholar] [CrossRef]
- Yan, H.; Liu, J.; Jin, S.; Du, L.; Wang, Q.; Luo, Y. A novel prediction tool based on shear wave elastography, gallbladder ultrasound, and serum biomarkers for the early diagnosis of biliary atresia in infants younger than 60 days old. Quant. Imaging Med. Surg. 2023, 13, 259–270. [Google Scholar] [CrossRef]






| All Children | BA | Non-BA | p | |
|---|---|---|---|---|
| Age, days | 51.1 (29.2, 62.1) | 47.9 (20.2) | 52.9 (20.6) | 0.323 |
| Sex, n (%) | 0.065 | |||
| Male | 34 (49.30%) | 18 (40.90%) | 16 (64.00%) | |
| Female | 35 (50.70%) | 26 (59.10%) | 19 (36.00%) | |
| AST, U/L | 200.0 (128.00, 277.00) | 225.50 (145.75, 298.00) | 172.00 (102.00, 252.00) | 0.060 |
| ALT, U/L | 131.0 (84.00, 234.00) | 128.00 (87.25, 248.25) | 134.00 (71.50, 222.50) | 0.644 |
| TBIL, μmol/L | 157.52 (64.10) | 173.01 (57.16) | 130.26 (67.64) | 0.007 |
| ALP, U/L | 534.00 (430.00, 670.00) | 543.61 (152.92) | 567.32 (218.91) | 0.599 |
| GGT, U/L | 278.00 (131.50, 489.50) | 386.00 (246.25, 614.25) | 128.00 (62.50, 214.00) | <0.001 |
| TP, g/L | 57.60 (7.01) | 58.09 (5.98) | 56.73 (8.59) | 0.442 |
| ALB, g/L | 40.49 (5.37) | 40.76 (5.02) | 40.02 (6.00) | 0.583 |
| PLT, 109/L | 418.05 (144.48) | 422.98 (142.25) | 421.00 (352.00, 501.00) | 0.100 |
| CRP, mg/L | 20 (29.00%) | 13 (29.5%) | 7 (28.00%) | 0.892 |
| Liver enlargement, n (%) | 34 (49.30%) | 22 (50.00%) | 12 (48.00%) | 0.873 |
| E, kPa | 12.43 (9.04, 161.17) | 13.83 (11.00, 17.63) | 8.89 (6.24, 12.39) | <0.001 |
| ATT, dB/cm/MHz | 0.61 (0.53, 0.66) | 0.609 (0.09) | 0.6 (0.10) | 0.062 |
| FI | 2.35 (0.77) | 2.59 (0.72) | 1.92 (0.70) | <0.001 |
| AI | 1.47 (0.32) | 1.569 (0.29) | 1.30 (0.30) | <0.001 |
| Gallbladder morphology | 35 (50.70%) | 29 (65.90%) | 6 (24.00%) | 0.001 |
| Variable | β | SE | OR (95% CI) | p |
|---|---|---|---|---|
| FI | 1.265 | 0.529 | 3.542 (1.257, 9.984) | 0.017 |
| GGT | 0.003 | 0.001 | 1.003 (1.001, 1.026) | 0.015 |
| TBIL | 0.013 | 0.006 | 1.013 (1.000, 1.026) | 0.050 |
| Gallbladder morphology | 1.771 | 0.741 | 5.878 (1.376, 25.114) | 0.017 |
| ATT | 5.584 | 3.297 | 266.017 (0.415, 170, 404.681) | 0.090 |
| Variables | Cutoff-Value | AUC (95% CI) | Sensitivity, % | Specificity, % | p |
|---|---|---|---|---|---|
| E | 10.055 | 0.785 (0.664, 0.905) | 0.864 | 0.640 | <0.001 |
| GGT | 232 | 0.819 (0.735, 0.902) | 0.818 | 0.80 | <0.001 |
| Gallbladder morphology | — | 0.851 (0.770, 0.933) | 0.659 | 0.76 | <0.001 |
| Nomogram | — | 0.887 (0.823, 0.952) | 0.864 | 0.76 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Xu, F.; Zheng, C.; Hu, C.; Yu, M.; Li, X.; Gao, Y.; Tang, Y.; Chen, J. Early Prediction of Biliary Atresia Using Combi-Elastography in Infants ≤ 60 Days of Age. Diagnostics 2026, 16, 571. https://doi.org/10.3390/diagnostics16040571
Xu F, Zheng C, Hu C, Yu M, Li X, Gao Y, Tang Y, Chen J. Early Prediction of Biliary Atresia Using Combi-Elastography in Infants ≤ 60 Days of Age. Diagnostics. 2026; 16(4):571. https://doi.org/10.3390/diagnostics16040571
Chicago/Turabian StyleXu, Fenglin, Chenpeng Zheng, Caihui Hu, Mingzhu Yu, Xiang Li, Yang Gao, Yi Tang, and Jingyu Chen. 2026. "Early Prediction of Biliary Atresia Using Combi-Elastography in Infants ≤ 60 Days of Age" Diagnostics 16, no. 4: 571. https://doi.org/10.3390/diagnostics16040571
APA StyleXu, F., Zheng, C., Hu, C., Yu, M., Li, X., Gao, Y., Tang, Y., & Chen, J. (2026). Early Prediction of Biliary Atresia Using Combi-Elastography in Infants ≤ 60 Days of Age. Diagnostics, 16(4), 571. https://doi.org/10.3390/diagnostics16040571







