68Ga-FAPI-04 PET/CT in the Diagnosis of Hepatocellular Carcinoma Associated with Cirrhosis: Diagnostic Value, Correlation Between PET Parameters of the Tumor and Its Size, and PIVKA-II Levels
Abstract
1. Introduction
2. Materials and Methods
2.1. Pathologic Evaluation
2.2. The Procedure of PET/CT Imaging
2.3. Evaluation of Images and Lesion-Based Analysis
2.4. Reference Standard
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics
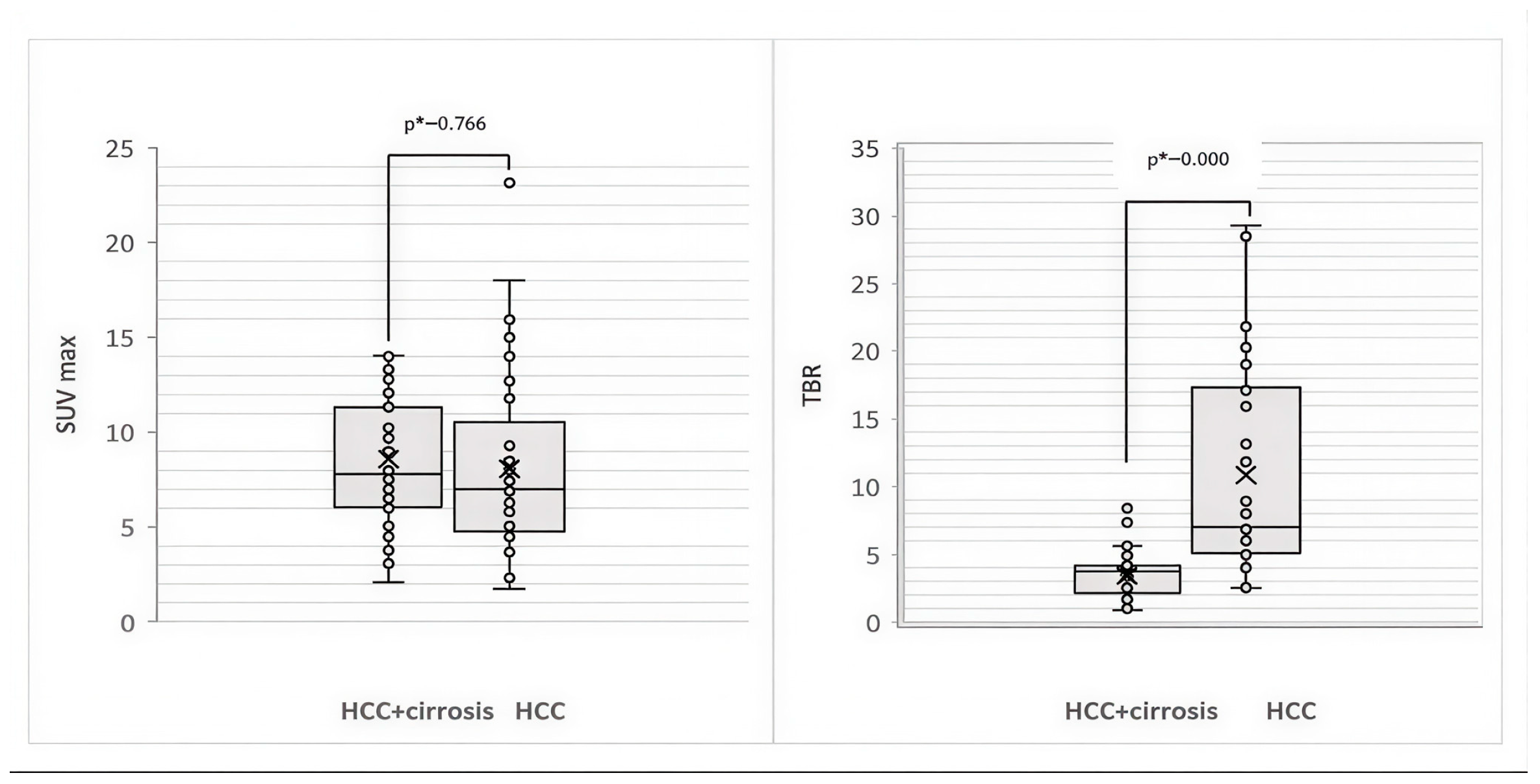
3.2. Comparative Analysis of 68Ga-FAPI-04 PET/CT Parameters for Visualization of HCC According to the Presence of Liver Cirrhosis
3.3. Analysis of the Relationship Between 68Ga-FAPI-04 PET/CT Parameters and the Size of HCC Tumor Lesions Associated with Cirrhosis
3.4. Analysis of the Relationship Between PET Parameters of HCC Tumor Lesions on 68Ga-FAPI-04 PET/CT and the Serum Marker PIVKA-II Level
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AASLD | American Association for the Study of the Liver Diseases |
| AFP | alpha-fetoprotein |
| ALT | alanine aminotransferase |
| AST | aspartate aminotransferase |
| CAF | cancer-associated fibroblasts |
| CPT | Child–Pugh–Turcotte |
| CT | computed tomography |
| DICOM | Digital Imaging and Communications in Medicine |
| EASL | European Association for the Study of the Liver |
| ELISA | enzyme-linked immunosorbent assay |
| FAP | fibroblast activation protein |
| FAPI | fibroblast activation protein inhibitor |
| FDG | fluorodeoxyglucose |
| FN | false-negative |
| FP | false-positive |
| GMP | Good Manufacturing Practice |
| HCC | hepatocellular carcinoma |
| HBs Ag | hepatitis B surface antigen |
| MELD | Model for End-Stage Liver Disease |
| MIP | maximum intensity projection |
| MRI | magnetic resonance imaging |
| PET/CT | positron emission tomography/computed tomography |
| PIVKA-II | protein induced by vitamin K absence or antagonist II |
| PT-INR | prothrombin time—international normalized ratio |
| PTA | prothrombin time activity |
| ROI | region of interest |
| rs | Spearman rank correlation coefficient |
| SUV | standardized uptake value |
| SUVmax | maximum standardized uptake value |
| SUVmean | mean standardized uptake value |
| TBR | tumor-to-background ratio |
| TN | true-negative |
| TP | true-positive |
| UL | upper limit |
| VOI | volume of interest |
| WGO | World Gastroenterology Organization |
References
- Vogel, A.; Meyer, T.; Sapisochin, G.; Salem, R.; Saborowski, A. Hepatocellular carcinoma. Lancet 2022, 400, 1345–1362. [Google Scholar] [CrossRef]
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef]
- Nyakale, N.; Filippi, L.; Aldous, C.; Sathekge, M. Update on PET Radiopharmaceuticals for Imaging Hepatocellular Carcinoma. Cancers 2023, 15, 1975. [Google Scholar] [CrossRef]
- Cancer Today. Available online: https://gco.iarc.who.int/today/ (accessed on 20 February 2024).
- Siripongsatian, D.; Promteangtrong, C.; Kunawudhi, A.; Kiatkittikul, P.; Boonkawin, N.; Chinnanthachai, C.; Jantarato, A.; Chotipanich, C. FAPI-PET/CT and [18F]F-FDG PET/CT in liver malignancies. Mol. Imaging Biol. 2022, 24, 818–829. [Google Scholar] [CrossRef]
- Liang, J.; Peng, H.; Li, W.; Liu, Z. Head-to-head study of [18F]FAPI-04 PET/CT and [18F]FDG PET/CT for non-invasive assessment of liver cancer and its immunohistochemical markers. BMC Cancer 2024, 24, 1378. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, S.; Li, M.; Xue, H.; Zhong, X.; Li, S.; Peng, H.; Liang, J.; Liu, Z.; Rao, S.; et al. Head-to-head comparison of 18F-FAPI and 18F-FDG PET/CT in staging and therapeutic management of hepatocellular carcinoma. Cancer Imaging 2023, 23, 106. [Google Scholar] [CrossRef]
- Izuishi, K.; Yamamoto, Y.; Mori, H.; Kameyama, R.; Fujihara, S.; Masaki, T.; Suzuki, Y. Molecular mechanisms of [18F]fluorodeoxyglucose accumulation in liver cancer. Oncol. Rep. 2014, 31, 701–706. [Google Scholar] [CrossRef]
- Xu, H.; Wang, H.; Yu, S.; Li, X.-M.; Jiang, D.-L.; Wu, Y.-F.; Ren, S.-H.; Qin, L.-X.; Guan, Y.-H.; Lu, L.; et al. Prognostic and diagnostic value of [18F]FDG, 11C-acetate, and [68Ga]Ga-FAPI-04 PET/CT for hepatocellular carcinoma. Eur. Radiol. 2025, 35, 4121–4131. [Google Scholar] [CrossRef]
- Ho, C.L.; Yu, S.C.; Yeung, D.W. 11C-acetate PET imaging in hepatocellular carcinoma and other liver masses. J. Nucl. Med. 2003, 44, 213–221. [Google Scholar]
- Peng, D.; Cao, J.; Guo, C.; He, J.; Yang, L.; Zhang, J.; Yang, J.; Feng, Y.; Xu, T.; Chen, Y. Influence of Cirrhosis on 68Ga-FAPI PET/CT in Intrahepatic Tumors. Radiology 2023, 307, e222448. [Google Scholar] [CrossRef]
- Affo, S.; Yu, L.X.; Schwabe, R.F. The Role of Cancer-Associated Fibroblasts and Fibrosis in Liver Cancer. Annu. Rev. Pathol. 2017, 12, 153–186. [Google Scholar] [CrossRef]
- Yang, T.; Yang, Y.; Hu, S.; Xie, Y.; Shen, Z.; Zhang, Y. 18F-FAPI-42 PET/CT for preoperatively identifying intrahepatic cholangiocarcinoma and hepatocellular carcinoma. Quant. Imaging Med. Surg. 2025, 15, 741–751. [Google Scholar] [CrossRef]
- Mule, S.; Chalaye, J.; Legou, F.; Tenenhaus, A.; Calderaro, J.; Galletto Pregliasco, A.; Laurent, A.; Kharrat, R.; Amaddeo, G.; Regnault, H.; et al. Hepatobiliary MR contrast agent uptake as a predictive biomarker of aggressive features on pathology and reduced recurrence-free survival in resectable hepatocellular carcinoma: Comparison with dual-tracer 18F-FDG and 18F-FCH PET/CT. Eur. Radiol. 2020, 30, 5348–5357. [Google Scholar] [CrossRef]
- Lindner, T.; Loktev, A.; Altmann, A.; Giesel, F.; Kratochwil, C.; Debus, J.; Jäger, D.; Mier, W.; Haberkorn, U. Development of quinoline-based theranostic ligands for the targeting of fibroblast activation protein. J. Nucl. Med. 2018, 59, 1415–1422. [Google Scholar] [CrossRef]
- Loktev, A.; Lindner, T.; Mier, W.; Debus, J.; Altmann, A.; Jäger, D.; Giesel, F.; Kratochwil, C.; Barthe, P.; Roumestand, C.; et al. A tumor-imaging method targeting cancer-associated fibroblasts. J. Nucl. Med. 2018, 59, 1423–1429. [Google Scholar] [CrossRef]
- Mori, Y.; Dendl, K.; Cardinale, J.; Kratochwil, C.; Giesel, F.L.; Haberkorn, U. FAPI PET: Fibroblast Activation Protein Inhibitor Use in Oncologic and Nononcologic Disease. Radiology 2023, 306, e220749. [Google Scholar] [CrossRef]
- Rajaraman, V.; Meenakshi, L.A.; Selvaraj, A.J.; Pottakkat, B.M.; Halanaik, D. Role of 68Ga-FAPI PET/CT in Assessing Hepatobiliary Malignancies: A Prospective Pilot Study. Clin. Nucl. Med. 2023, 48, e281–e288. [Google Scholar] [CrossRef]
- Manuppella, F.; Pisano, G.; Taralli, S.; Caldarella, C.; Calcagni, M.L. Diagnostic Performances of PET/CT Using Fibroblast Activation Protein Inhibitors in Patients with Primary and Metastatic Liver Tumors: A Comprehensive Literature Review. Int. J. Mol. Sci. 2024, 25, 7197. [Google Scholar] [CrossRef]
- Shi, X.; Xing, H.; Yang, X.; Li, F.; Yao, S.; Zhang, H.; Zhao, H.; Hacker, M.; Huo, L.; Li, X. Fibroblast imaging of hepatic carcinoma with 68Ga-Ga-FAPI-04 PET/CT: A pilot study in patients with suspected hepatic nodules. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 196–203. [Google Scholar] [CrossRef]
- Chen, H.; Pang, Y.; Wu, J.; Zhao, L.; Hao, B.; Wu, J.; Wei, J.; Wu, S.; Zhao, L.; Luo, Z.; et al. Comparison of [68Ga]Ga-DOTA-FAPI-04 and [18F]FDG PET/CT for the diagnosis of primary and metastatic lesions in patients with various types of cancer. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1820–1832. [Google Scholar] [CrossRef]
- Giesel, F.L.; Kratochwil, C.; Lindner, T.; Marschalek, M.M.; Loktev, A.; Lehnert, W.; Debus, J.; Jäger, D.; Flechsig, P.; Altmann, A.; et al. 68Ga-FAPI PET/CT: Biodistribution and preliminary dosimetry estimate of 2 DOTA-containing FAP-targeting agents in patients with various cancers. J. Nucl. Med. 2019, 60, 386–392. [Google Scholar] [CrossRef]
- Wang, H.; Zhu, W.; Ren, S.; Kong, Y.; Huang, Q.; Zhao, J.; Guan, Y.; Jia, H.; Chen, J.; Lu, L.; et al. (68)Ga-FAPI-04 Versus (18)F-FDG PET/CT in the Detection of Hepatocellular Carcinoma. Front. Oncol. 2021, 11, 693640. [Google Scholar] [CrossRef]
- Guo, W.; Pang, Y.; Yao, L.; Zhao, L.; Fan, C.; Ke, J.; Guo, P.; Hao, B.; Fu, H.; Xie, C.; et al. Imaging fibroblast activation protein in liver cancer: A single-center post hoc retrospective analysis to compare [68Ga]Ga-FAPI-04 PET/CT versus MRI and [18F]-FDG PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1604–1617. [Google Scholar] [CrossRef]
- Shi, X.; Xing, H.; Yang, X.; Li, F.; Yao, S.; Congwei, J.; Zhao, H.; Hacker, M.; Huo, L.; Li, X. Comparison of PET imaging of activated fibroblasts and 18F-FDG for diagnosis of primary hepatic tumours: A prospective pilot study. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 1593–1603. [Google Scholar] [CrossRef]
- Kim, D.Y.; Toan, B.N.; Tan, C.-K.; Hasan, I.; Setiawan, L.; Yu, M.-L.; Izumi, N.; Huyen, N.N.; Chow, P.K.-H.; Mohamed, R.; et al. Utility of combining PIVKA-II and AFP in the surveillance and monitoring of hepatocellular carcinoma in the Asia-Pacific region. Clin. Mol. Hepatol. 2023, 29, 277–292. [Google Scholar] [CrossRef]
- Feng, H.; Li, B.; Li, Z.; Wei, Q.; Ren, L. PIVKA-II serves as a potential biomarker that complements AFP for the diagnosis of hepatocellular carcinoma. BMC Cancer 2021, 21, 401. [Google Scholar] [CrossRef]
- Loy, L.M.; Low, H.M.; Choi, J.Y.; Rhee, H.; Wong, C.F.; Tan, C.H. Variant Hepatocellular Carcinoma Subtypes According to the 2019 WHO Classification: An Imaging-Focused Review. AJR Am. J. Roentgenol. 2022, 219, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Perdigoto, D.N.; Figueiredo, P.; Tomé, L. The Role of the CLIF-C OF and the 2016 MELD in Prognosis of Cirrhosis with and without Acute-on-Chronic Liver Failure. Ann. Hepatol. 2019, 18, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.-C.; She, B.; Gao, W.-T.; Ji, Y.-H.; Xu, D.-D.; Wang, Q.-S.; Wang, S.-B. Positron-emission tomography for hepatocellular carcinoma: Current status and future prospects. World J. Gastroenterol. 2019, 25, 4682–4695. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatocellular carcinoma. J. Hepatol. 2025, 82, 315–374. [Google Scholar] [CrossRef]
- Pabst, K.M.; Weber, M.M.; Lashinsky, C.; Sandach, P.; Bartel, T.; Küper, A.T.; Kessler, L.; Trajkovic-Arsic, M.; Eckstein, M.; Gilman, E.; et al. [68Ga]Ga-FAPI-46 PET accuracy for cancer imaging with histopathology validation: A single-centre, single-arm, interventional, phase 2 trial. Lancet Oncol. 2025, 26, 1204–1214. [Google Scholar] [CrossRef]
- Kratochwil, C.; Flechsig, P.; Lindner, T.; Abderrahim, L.; Altmann, A.; Mier, W.; Adeberg, S.; Rathke, H.; Röhrich, M.; Winter, H.; et al. (68)Ga-FAPI PET/CT: Tracer Uptake in 28 Different Kinds of Cancer. J. Nucl. Med. 2019, 60, 801–805. [Google Scholar] [CrossRef]
- Geist, B.K.; Xing, H.; Wang, J.; Shi, X.; Zhao, H.; Hacker, M.; Sang, X.; Huo, L.; Li, X. A methodological investigation of healthy tissue, hepatocellular carcinoma, and other lesions with dynamic 68Ga-FAPI PET/CT imaging. EJNMMI Phys. 2021, 8, 8. [Google Scholar] [CrossRef]
- Bughda, R.; Dimou, P.; D’Souza, R.R.; Klampatsa, A. Fibroblast Activation Protein (FAP)-targeted CAR-T cells: Launching an attack on tumor stroma. Immunotargets Ther. 2021, 10, 313–323. [Google Scholar] [CrossRef]
- Zhang, J.; He, Q.; Jiang, S.; Li, M.; Xue, H.; Zhang, D.; Li, S.; Peng, H.; Liang, J.; Liu, Z.; et al. [18F]FAPI PET/CT in the evaluation of focal liver lesions with [18F]FDG non-avidity. Eur. J. Nucl. Med. Mol. Imaging 2023, 50, 937–950. [Google Scholar] [CrossRef]
- Liu, X.; Liu, H.; Gao, C.; Zeng, W. Comparison of 68Ga-FAPI and 18F-FDG PET/CT for the diagnosis of primary and metastatic lesions in abdominal and pelvic malignancies: A systematic review and meta-analysis. Front. Oncol. 2023, 13, 1093861. [Google Scholar] [CrossRef]
- Levy, M.T.; McCaughan, G.W.; Abbott, C.A.; Park, J.E.; Cunningham, A.M.; Müller, E.; Rettig, W.J.; Gorrell, M.D. Fibroblast Activation Protein: A Cell Surface, Dipeptidyl Peptidase and Gelatinase Expressed by Stellate Cells at the Tissue Remodelling Interface in Human Cirrhosis. Hepatology 1999, 29, 1768–1778. [Google Scholar] [CrossRef]
- Chen, H.; Zhao, L.; Ruan, D.; Pang, Y.; Hao, B.; Dai, Y.; Wu, X.; Guo, W.; Fan, C.; Wu, J.; et al. Usefulness of [68Ga]Ga-DOTA-FAPI-04 PET/CT in patients presenting with inconclusive [18F]FDG PET/CT findings. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 73–86. [Google Scholar] [CrossRef]
- Peng, D.; He, J.; Liu, H.; Cao, J.; Wang, Y.; Chen, Y. FAPI PET/CT research progress in digestive system tumours. Dig. Liver Dis. 2022, 54, 164–169. [Google Scholar] [CrossRef]
- Serumula, W.; Pillay, V.; Hadebe, B.; Vorster, M. Fibroblast Activation Protein Inhibitor (FAPI)-Based Theranostics. Pharmaceuticals 2025, 18, 522. [Google Scholar] [CrossRef]



| Total Number of Patients | 59 |
|---|---|
| Median, age (y, range) | 61.0 (range 38–85) |
| Gender | |
| Male | 45 (76.27%) |
| Female | 14 (23.73%) |
| Groups | |
| Group one: HCC and cirrhosis | 37 (62.71%) |
| Group two: HCC | 22 (37.29%) |
| Liver tumor lesions | |
| Number of patients/solitary tumor | 36/36 (61.02%) |
| Number of patients/multiple tumor lesions | 23/62 (38.98%) |
| Child–Pugh–Turcotte score for group one | |
| Class A | 15 (40.54%) |
| Class B | 18 (48.65%) |
| Class С | 4 (10.81%) |
| MELD score [1] (Me = 12) | |
| ≤12 | 14 |
| >12 | 19 |
| Child–Pugh–Turcotte/MELD [29] | |
| CPT Class A: MELD score, median, (score, range) | 8 (range 6–16) |
| CPT Class B: MELD score, median, (score, range) | 15 (range 12–21) |
| Histological types of HCC [28] | |
| NOS-HCC | 49 (83.05%) |
| HCC of specific variants | |
| SH-HCC | 9 (15.25%) |
| Fibrolamellar carcinoma | 1 (1.70%) |
| Differentiation | |
| G1 | 10 (16.95%) |
| G2 | 42 (71.19%) |
| G3 | 7 (11.86%) |
| Differentiation for group | |
| G1: Group one/group two | 6 (16.22%)/4 (18.18%) |
| G2: Group one/group two | 26 (70.27%)/16 (72.73%) |
| G3: Group one/group two | 5 (13.51%)/2 (9.09%) |
| HBs Ag (+) | 44 (74.58%) |
| Serum tumor markers | |
| AFP > 10.0 ng/mL | 54 (91.53%) |
| PIVKA-II > 50.8 mAU/mL | 52 (88.14%) |
| Blood transaminases | |
| For men ALT > 45 U/L, for women ALT > 35 U/L | 42 (71.19%) |
| For men AST > 45 U/L, for women AST > 35 U/L | 46 (77.97%) |
| Metastatic HCC | 6 (10.17%) |
| Characteristics | HCC | HCC and Cirrhosis | HCC Without Cirrhosis | |||
|---|---|---|---|---|---|---|
| D “+” | D “-” | D “+” | D “-” | D “+” | D “-” | |
| Positive | TP = 88 | FP = 4 | TP = 55 | FP = 3 | TP = 33 | FP = 1 |
| Negative | FN = 10 | TN = 6 | FN = 8 | TN = 4 | FN = 2 | TN = 2 |
| Accuracy/Acc, % (95%CI) | 87.04 (79.21–92.73) | 84.29 (73.62–91.89) | 92.11 (78.62–98.34) | |||
| Sensitivity/Sn, % (95%CI) | 89.80 (82.03–95.00) | 87.30 (76.50–94.35) | 94.29 (80.84–99.30) | |||
| Specificity/Sp, % (95%CI) | 60.0 (26.24–87.84) | 57.14 (18.41–90.10) | 66.67 (9.43–99.16) | |||
| PET Parameters | The Calculated Spearman Correlation Coefficient, rs | p-Value |
|---|---|---|
| All lesions, n = 88 | ||
| SUVmax of lesion | 0.504 | 0.000 * |
| TBR | 0.122 | 0.258 |
| Group one: HCC and cirrhosis, n = 55 | ||
| SUVmax of lesion | 0.207 | 0.129 |
| TBR | −0.121 | 0.379 |
| Group two: HCC without cirrhosis, n = 33 | ||
| SUVmax of lesion | 0.815 | 0.000 * |
| TBR | 0.673 | 0.000 * |
| PET Parameters | Calculated Spearman Correlation Coefficient, rs | p-Value |
|---|---|---|
| All lesions, n = 88 | ||
| SUVmax of lesion | 0.177 | 0.098 |
| TBR | 0.128 | 0.234 |
| Size of lesion | 0.072 | 0.506 |
| Group one: HCC and cirrhosis, n = 55 | ||
| SUVmax of lesion | 0.438 | 0.001 ** |
| TBR | 0.105 | 0.444 |
| Size of lesion | 0.296 | 0.028 * |
| Group two: HCC without cirrhosis, n = 33 | ||
| SUVmax of lesion | −0.087 | 0.632 |
| TBR | −0.214 | 0.231 |
| Size of lesion | −0.320 | 0.070 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zholdybay, Z.; Zhakenova, Z.; Issamatov, B.; Gabdullina, M.; Filippenko, Y.; Yessentayeva, S.; Alisherov, G.; Amankulov, J.; Fakhradiyev, I. 68Ga-FAPI-04 PET/CT in the Diagnosis of Hepatocellular Carcinoma Associated with Cirrhosis: Diagnostic Value, Correlation Between PET Parameters of the Tumor and Its Size, and PIVKA-II Levels. Diagnostics 2026, 16, 249. https://doi.org/10.3390/diagnostics16020249
Zholdybay Z, Zhakenova Z, Issamatov B, Gabdullina M, Filippenko Y, Yessentayeva S, Alisherov G, Amankulov J, Fakhradiyev I. 68Ga-FAPI-04 PET/CT in the Diagnosis of Hepatocellular Carcinoma Associated with Cirrhosis: Diagnostic Value, Correlation Between PET Parameters of the Tumor and Its Size, and PIVKA-II Levels. Diagnostics. 2026; 16(2):249. https://doi.org/10.3390/diagnostics16020249
Chicago/Turabian StyleZholdybay, Zhamilya, Zhanar Zhakenova, Bekzhan Issamatov, Madina Gabdullina, Yevgeniya Filippenko, Suriya Yessentayeva, Galymzhan Alisherov, Jandos Amankulov, and Ildar Fakhradiyev. 2026. "68Ga-FAPI-04 PET/CT in the Diagnosis of Hepatocellular Carcinoma Associated with Cirrhosis: Diagnostic Value, Correlation Between PET Parameters of the Tumor and Its Size, and PIVKA-II Levels" Diagnostics 16, no. 2: 249. https://doi.org/10.3390/diagnostics16020249
APA StyleZholdybay, Z., Zhakenova, Z., Issamatov, B., Gabdullina, M., Filippenko, Y., Yessentayeva, S., Alisherov, G., Amankulov, J., & Fakhradiyev, I. (2026). 68Ga-FAPI-04 PET/CT in the Diagnosis of Hepatocellular Carcinoma Associated with Cirrhosis: Diagnostic Value, Correlation Between PET Parameters of the Tumor and Its Size, and PIVKA-II Levels. Diagnostics, 16(2), 249. https://doi.org/10.3390/diagnostics16020249






